
NEORAL
®
Soft Gelatin Capsules (cyclosporine capsules, USP) MODIFIED
NEORAL
®
Oral Solution (cyclosporine oral solution, USP) MODIFIED
Rx only
Prescribing Information
WARNING
Only physicians experienced in management of systemic immunosuppressive therapy for the indicated
disease should prescribe Neoral
®
. At doses used in solid organ transplantation, only physicians
experienced in immunosuppressive therapy and management of organ transplant recipients should
prescribe Neoral
®
. Patients receiving the drug should be managed in facilities equipped and staffed with
adequate laboratory and supportive medical resources. The physician responsible for maintenance therapy
should have complete information requisite for the follow-up of the patient.
Neoral
®
, a systemic immunosuppressant, may increase the susceptibility to infection and the development
of neoplasia. In kidney, liver, and heart transplant patients Neoral
®
may be administered with other
immunosuppressive agents. Increased susceptibility to infection and the possible development of
lymphoma and other neoplasms may result from the increase in the degree of immunosuppression in
transplant patients.
Neoral
®
Soft Gelatin Capsules (cyclosporine capsules, USP) MODIFIED and Neoral
®
Oral Solution
(cyclosporine oral solution, USP) MODIFIED have increased bioavailability in comparison to
Sandimmune
®
Soft Gelatin Capsules (cyclosporine capsules, USP) and Sandimmune
®
Oral Solution
(cyclosporine oral solution, USP). Neoral
®
and Sandimmune
®
are not bioequivalent and cannot be used
interchangeably without physician supervision. For a given trough concentration, cyclosporine exposure
will be greater with Neoral
®
than with Sandimmune
®
. If a patient who is receiving exceptionally high
doses of Sandimmune
®
is converted to Neoral
®
, particular caution should be exercised. Cyclosporine
blood concentrations should be monitored in transplant and rheumatoid arthritis patients taking Neoral
®
to
avoid toxicity due to high concentrations. Dose adjustments should be made in transplant patients to
minimize possible organ rejection due to low concentrations. Comparison of blood concentrations in the
published literature with blood concentrations obtained using current assays must be done with detailed
knowledge of the assay methods employed.
For Psoriasis Patients (See also Boxed WARNINGS above)
Psoriasis patients previously treated with PUVA and to a lesser extent, methotrexate or other
immunosuppressive agents, UVB, coal tar, or radiation therapy, are at an increased risk of developing
skin malignancies when taking Neoral
®
.
Cyclosporine, the active ingredient in Neoral
®
, in recommended dosages, can cause systemic
hypertension and nephrotoxicity. The risk increases with increasing dose and duration of cyclosporine
therapy. Renal dysfunction, including structural kidney damage, is a potential consequence of
cyclosporine, and therefore, renal function must be monitored during therapy.
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
DESCRIPTION
Neoral
®
is an oral formulation of cyclosporine that immediately forms a microemulsion in an aqueous
environment.
Cyclosporine, the active principle in Neoral
®
, is a cyclic polypeptide immunosuppressant agent consisting
of 11 amino acids. It is produced as a metabolite by the fungus species Beauveria nivea.
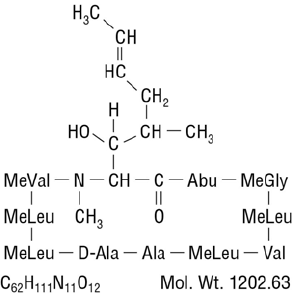
Chemically, cyclosporine is designated as [R-[R*,R*-(E)]]-cyclic-(L-alanyl-D-alanyl-N-methyl-L-leucyl-
N-methyl-L-leucyl-N-methyl-L-valyl-3-hydroxy-N,4-dimethyl-L-2-amino-6-octenoyl-L-α -amino-
butyryl-N-methylglycyl-N-methyl-L-leucyl-L-valyl-N-methyl-L-leucyl).
Neoral
®
Soft Gelatin Capsules
(cyclosporine capsules, USP) MODIFIED are available in 25 mg and 100 mg strengths.
Each 25 mg capsule contains:
cyclosporine ...................................................................................................................... 25 mg
alcohol, USP dehydrated.....................................................................11.9% v/v (9.5% wt/vol.)
Each 100 mg capsule contains:
cyclosporine .................................................................................................................... 100 mg
alcohol, USP dehydrated.....................................................................11.9% v/v (9.5% wt/vol.)
Inactive Ingredients: Corn oil-mono-di-triglycerides, polyoxyl 40 hydrogenated castor oil NF, DL-α-
tocopherol USP, gelatin NF, glycerol, iron oxide black, propylene glycol USP, titanium dioxide USP,
carmine, and other ingredients.
Neoral
®
Oral Solution
(cyclosporine oral solution, USP) MODIFIED is available in 50 mL bottles.
Each mL contains:
cyclosporine ..............................................................................................................100 mg/mL
alcohol, USP dehydrated.....................................................................11.9% v/v (9.5% wt/vol.)
Inactive Ingredients: Corn oil-mono-di-triglycerides, polyoxyl 40 hydrogenated castor oil NF, DL-α -
tocopherol USP, propylene glycol USP.
The chemical structure of cyclosporine (also known as cyclosporin A) is:
CLINICAL PHARMACOLOGY
Cyclosporine is a potent immunosuppressive agent that in animals prolongs survival of allogeneic
transplants involving skin, kidney, liver, heart, pancreas, bone marrow, small intestine, and lung.
Cyclosporine has been demonstrated to suppress some humoral immunity and to a greater extent, cell-
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
mediated immune reactions such as allograft rejection, delayed hypersensitivity, experimental allergic
encephalomyelitis, Freund’s adjuvant arthritis, and graft vs. host disease in many animal species for a
variety of organs.
The effectiveness of cyclosporine results from specific and reversible inhibition of immunocompetent
lymphocytes in the G
0
- and G
1
-phase of the cell cycle. T-lymphocytes are preferentially inhibited. The T-
helper cell is the main target, although the T-suppressor cell may also be suppressed. Cyclosporine also
inhibits lymphokine production and release including interleukin-2.
No effects on phagocytic function (changes in enzyme secretions, chemotactic migration of granulocytes,
macrophage migration, carbon clearance in vivo) have been detected in animals. Cyclosporine does not
cause bone marrow suppression in animal models or man.
Pharmacokinetics
The immunosuppressive activity of cyclosporine is primarily due to parent drug. Following oral
administration, absorption of cyclosporine is incomplete. The extent of absorption of cyclosporine is
dependent on the individual patient, the patient population, and the formulation. Elimination of
cyclosporine is primarily biliary with only 6% of the dose (parent drug and metabolites) excreted in urine.
The disposition of cyclosporine from blood is generally biphasic, with a terminal half-life of
approximately 8.4 hours (range 5-18 hours). Following intravenous administration, the blood clearance of
cyclosporine (assay: HPLC) is approximately 5-7 mL/min/kg in adult recipients of renal or liver
allografts. Blood cyclosporine clearance appears to be slightly slower in cardiac transplant patients.
The Neoral
®
Soft Gelatin Capsules (cyclosporine capsules, USP) MODIFIED and Neoral
®
Oral Solution
(cyclosporine oral solution, USP) MODIFIED are bioequivalent. Neoral
®
Oral Solution diluted with
orange juice or apple juice is bioequivalent to Neoral Oral Solution diluted with water. The effect of milk
on the bioavailability of cyclosporine when administered as Neoral Oral Solution has not been evaluated.
The relationship between administered dose and exposure (area under the concentration versus time
curve, AUC) is linear within the therapeutic dose range. The intersubject variability (total, %CV) of
cyclosporine exposure (AUC) when Neoral
®
or Sandimmune
®
is administered ranges from approximately
20% to 50% in renal transplant patients. This intersubject variability contributes to the need for
individualization of the dosing regimen for optimal therapy (see DOSAGE AND ADMINISTRATION).
Intrasubject variability of AUC in renal transplant recipients (%CV) was 9%-21% for Neoral
®
and
19%-26% for Sandimmune
®
. In the same studies, intrasubject variability of trough concentrations (%CV)
was 17%-30% for Neoral
®
and 16%-38% for Sandimmune
®
.
Absorption
Neoral
®
has increased bioavailability compared to Sandimmune
®
. The absolute bioavailability of
cyclosporine administered as Sandimmune
®
is dependent on the patient population, estimated to be less
than 10% in liver transplant patients and as great as 89% in some renal transplant patients. The absolute
bioavailability of cyclosporine administered as Neoral
®
has not been determined in adults. In studies of
renal transplant, rheumatoid arthritis and psoriasis patients, the mean cyclosporine AUC was
approximately 20% to 50% greater and the peak blood cyclosporine concentration (C
max
) was
approximately 40% to 106% greater following administration of Neoral
®
compared to following
administration of Sandimmune
®
. The dose normalized AUC in de novo liver transplant patients
administered Neoral
®
28 days after transplantation was 50% greater and C
max
was 90% greater than in
those patients administered Sandimmune
®
. AUC and C
max
are also increased (Neoral
®
relative to
Sandimmune
®
) in heart transplant patients, but data are very limited. Although the AUC and C
max
values
are higher on Neoral
®
relative to Sandimmune
®
, the pre-dose trough concentrations (dose-normalized) are
similar for the two formulations.
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
Following oral administration of Neoral
®
, the time to peak blood cyclosporine concentrations (T
max
)
ranged from 1.5-2.0 hours. The administration of food with Neoral
®
decreases the cyclosporine AUC and
C
max
. A high fat meal (669 kcal, 45 grams fat) consumed within one-half hour before Neoral
®
administration decreased the AUC by 13% and C
max
by 33%. The effects of a low fat meal (667 kcal, 15
grams fat) were similar.
The effect of T-tube diversion of bile on the absorption of cyclosporine from Neoral
®
was investigated in
eleven de novo liver transplant patients. When the patients were administered Neoral
®
with and without
T-tube diversion of bile, very little difference in absorption was observed, as measured by the change in
maximal cyclosporine blood concentrations from pre-dose values with the T-tube closed relative to when
it was open: 6.9±41% (range -55% to 68%).
Pharmacokinetic Parameters (mean±SD)
Dose/day
1
Dose/weight AUC
2
C
max
Trough
3
CL/F CL/F
Patient Population (mg/d) (mg/kg/d) (ng·hr/mL) (ng/mL) (ng/mL) (mL/min) (mL/min/kg)
De novo renal
transplant
4
597±174 7.95±2.81 8772±2089 1802±428 361±129 593±204 7.8±2.9
Week 4 (N=37)
Stable renal
transplant
4
344±122 4.10±1.58 6035±2194 1333±469 251±116 492±140 5.9±2.1
(N=55)
De novo liver
transplant
5
458±190 6.89±3.68 7187±2816 1555±740 268±101 577±309 8.6±5.7
Week 4 (N=18)
De novo rheumatoid
arthritis
6
182±55.6 2.37±0.36 2641±877 728±263 96.4±37.7 613±196 8.3±2.8
(N=23)
De novo psoriasis
6
189±69.8 2.48±0.65 2324±1048 655±186 74.9±46.7 723±186 10.2±3.9
Week 4 (N=18)
1
Total daily dose was divided into two doses administered every 12 hours
2
AUC was measured over one dosing interval
3
Trough concentration was measured just prior to the morning Neoral
®
dose, approximately 12 hours after the previous dose
4
Assay: TDx specific monoclonal fluorescence polarization immunoassay
5
Assay: Cyclo-trac specific monoclonal radioimmunoassay
6
Assay: INCSTAR specific monoclonal radioimmunoassay
Distribution
Cyclosporine is distributed largely outside the blood volume. The steady state volume of distribution
during intravenous dosing has been reported as 3-5 L/kg in solid organ transplant recipients. In blood, the
distribution is concentration dependent. Approximately 33%-47% is in plasma, 4%-9% in lymphocytes,
5%-12% in granulocytes, and 41%-58% in erythrocytes. At high concentrations, the binding capacity of
leukocytes and erythrocytes becomes saturated. In plasma, approximately 90% is bound to proteins,
primarily lipoproteins. Cyclosporine is excreted in human milk. (See PRECAUTIONS, Nursing Mothers)
Metabolism
Cyclosporine is extensively metabolized by the cytochrome P-450 3A enzyme system in the liver, and to
a lesser degree in the gastrointestinal tract, and the kidney. The metabolism of cyclosporine can be altered
by the co-administration of a variety of agents. (See PRECAUTIONS, Drug Interactions) At least 25
metabolites have been identified from human bile, feces, blood, and urine. The biological activity of the
metabolites and their contributions to toxicity are considerably less than those of the parent compound.
The major metabolites (M1, M9, and M4N) result from oxidation at the 1-beta, 9-gamma, and
4-N-demethylated positions, respectively. At steady state following the oral administration of
Sandimmune
®
, the mean AUCs for blood concentrations of M1, M9, and M4N are about 70%, 21%, and
7.5% of the AUC for blood cyclosporine concentrations, respectively. Based on blood concentration data
from stable renal transplant patients (13 patients administered Neoral
®
and Sandimmune
®
in a crossover
study), and bile concentration data from de novo liver transplant patients (4 administered Neoral
®
, 3
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda

administered Sandimmune
®
), the percentage of dose present as M1, M9, and M4N metabolites is similar
when either Neoral
®
or Sandimmune
®
is administered.
Excretion
Only 0.1% of a cyclosporine dose is excreted unchanged in the urine. Elimination is primarily biliary with
only 6% of the dose (parent drug and metabolites) excreted in the urine. Neither dialysis nor renal failure
alter cyclosporine clearance significantly.
Drug Interactions
(See PRECAUTIONS, Drug Interactions) When diclofenac or methotrexate was co-administered with
cyclosporine in rheumatoid arthritis patients, the AUC of diclofenac and methotrexate, each was
significantly increased. (See PRECAUTIONS, Drug Interactions) No clinically significant
pharmacokinetic interactions occurred between cyclosporine and aspirin, ketoprofen, piroxicam, or
indomethacin.
Special Populations
Pediatric Population
Pharmacokinetic data from pediatric patients administered Neoral
®
or Sandimmune
®
are very limited. In
15 renal transplant patients aged 3-16 years, cyclosporine whole blood clearance after IV administration
of Sandimmune
®
was 10.6±3.7 mL/min/kg (assay: Cyclo-trac specific RIA). In a study of 7 renal
transplant patients aged 2-16, the cyclosporine clearance ranged from 9.8-15.5 mL/min/kg. In 9 liver
transplant patients aged 0.6-5.6 years, clearance was 9.3±5.4 mL/min/kg (assay: HPLC).
In the pediatric population, Neoral
®
also demonstrates an increased bioavailability as compared to
Sandimmune
®
. In 7 liver de novo transplant patients aged 1.4-10 years, the absolute bioavailability of
Neoral
®
was 43% (range 30%-68%) and for Sandimmune
®
in the same individuals absolute
bioavailability was 28% (range 17%-42%).
Pediatric Pharmacokinetic Parameters (mean±SD)
Dose/day Dose/weight AUC
1
C
max
CL/F CL/F
Patient Population (mg/d) (mg/kg/d) (ng·hr/mL) (ng/mL) (mL/min) (mL/min/kg)
Stable liver transplant
2
Age 2-8, Dosed TID (N=9) 101±25 5.95±1.32 2163±801 629±219 285±94 16.6±4.3
Age 8-15, Dosed BID (N=8) 188±55 4.96±2.09 4272±1462 975±281 378±80 10.2±4.0
Stable liver transplant
3
Age 3, Dosed BID (N=1) 120 8.33 5832 1050 171 11.9
Age 8-15, Dosed BID (N=5) 158±55 5.51±1.91 4452±2475 1013±635 328±121 11.0±1.9
Stable renal transplant
3
Age 7-15, Dosed BID (N=5) 328±83 7.37±4.11 6922±1988 1827±487 418±143 8.7±2.9
1
AUC was measured over one dosing interval
2
Assay: Cyclo-trac specific monoclonal radioimmunoassay
3
Assay: TDx specific monoclonal fluorescence polarization immunoassay
Geriatric Population
Comparison of single dose data from both normal elderly volunteers (N=18, mean age 69 years) and
elderly rheumatoid arthritis patients (N=16, mean age 68 years) to single dose data in young adult
volunteers (N=16, mean age 26 years) showed no significant difference in the pharmacokinetic
parameters.
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
CLINICAL TRIALS
Rheumatoid Arthritis
The effectiveness of Sandimmune
®
and Neoral
®
in the treatment of severe rheumatoid arthritis was
evaluated in 5 clinical studies involving a total of 728 cyclosporine treated patients and 273 placebo
treated patients.
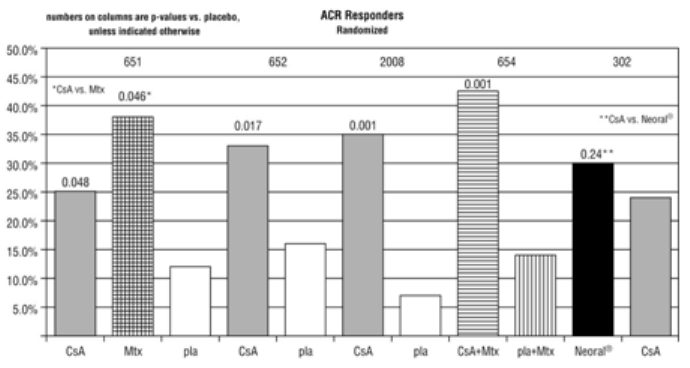
A summary of the results is presented for the “responder” rates per treatment group, with a responder
being defined as a patient having completed the trial with a 20% improvement in the tender and the
swollen joint count and a 20% improvement in 2 of 4 of investigator global, patient global, disability, and
erythrocyte sedimentation rates (ESR) for the Studies 651 and 652 and 3 of 5 of investigator global,
patient global, disability, visual analog pain, and ESR for Studies 2008, 654 and 302.
Study 651 enrolled 264 patients with active rheumatoid arthritis with at least 20 involved joints, who had
failed at least one major RA drug, using a 3:3:2 randomization to one of the following three groups: (1)
cyclosporine dosed at 2.5-5 mg/kg/day, (2) methotrexate at 7.5-15 mg/week, or (3) placebo. Treatment
duration was 24 weeks. The mean cyclosporine dose at the last visit was 3.1 mg/kg/day. See Graph below.
Study 652 enrolled 250 patients with active RA with >6 active painful or tender joints who had failed at
least one major RA drug. Patients were randomized using a 3:3:2 randomization to 1 of 3 treatment arms:
(1) 1.5-5 mg/kg/day of cyclosporine, (2) 2.5-5 mg/kg/day of cyclosporine, and (3) placebo. Treatment
duration was 16 weeks. The mean cyclosporine dose for group 2 at the last visit was 2.92 mg/kg/day. See
Graph below.
Study 2008 enrolled 144 patients with active RA and >6 active joints who had unsuccessful treatment
courses of aspirin and gold or Penicillamine. Patients were randomized to 1 of 2 treatment groups (1)
cyclosporine 2.5-5 mg/kg/day with adjustments after the first month to achieve a target trough level and
(2) placebo. Treatment duration was 24 weeks. The mean cyclosporine dose at the last visit was 3.63
mg/kg/day. See Graph below.
Study 654 enrolled 148 patients who remained with active joint counts of 6 or more despite treatment
with maximally tolerated methotrexate doses for at least three months. Patients continued to take their
current dose of methotrexate and were randomized to receive, in addition, one of the following
medications: (1) cyclosporine 2.5 mg/kg/day with dose increases of 0.5 mg/kg/day at weeks 2 and 4 if
there was no evidence of toxicity and further increases of 0.5 mg/kg/day at weeks 8 and 16 if a <30%
decrease in active joint count occurred without any significant toxicity; dose decreases could be made at
any time for toxicity or (2) placebo. Treatment duration was 24 weeks. The mean cyclosporine dose at the
last visit was 2.8 mg/kg/day (range: 1.3-4.1). See Graph below.
Study 302 enrolled 299 patients with severe active RA, 99% of whom were unresponsive or intolerant to
at least one prior major RA drug. Patients were randomized to 1 of 2 treatment groups (1) Neoral
®
and (2)
cyclosporine, both of which were started at 2.5 mg/kg/day and increased after 4 weeks for inefficacy in
increments of 0.5 mg/kg/day to a maximum of 5 mg/kg/day and decreased at any time for toxicity.
Treatment duration was 24 weeks. The mean cyclosporine dose at the last visit was 2.91 mg/kg/day
(range: 0.72-5.17) for Neoral
®
and 3.27 mg/kg/day (range: 0.73-5.68) for cyclosporine. See Graph below.
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
INDICATIONS AND USAGE
Kidney, Liver, and Heart Transplantation
Neoral
®
is indicated for the prophylaxis of organ rejection in kidney, liver, and heart allogeneic
transplants. Neoral
®
has been used in combination with azathioprine and corticosteroids.
Rheumatoid Arthritis
Neoral
®
is indicated for the treatment of patients with severe active, rheumatoid arthritis where the
disease has not adequately responded to methotrexate. Neoral
®
can be used in combination with
methotrexate in rheumatoid arthritis patients who do not respond adequately to methotrexate alone.
Psoriasis
Neoral
®
is indicated for the treatment of adult, nonimmunocompromised patients with severe (i.e.,
extensive and/or disabling), recalcitrant, plaque psoriasis who have failed to respond to at least one
systemic therapy (eg., PUVA, retinoids, or methotrexate) or in patients for whom other systemic therapies
are contraindicated, or cannot be tolerated.
While rebound rarely occurs, most patients will experience relapse with Neoral
®
as with other therapies
upon cessation of treatment.
CONTRAINDICATIONS
General
Neoral
®
is contraindicated in patients with a hypersensitivity to cyclosporine or to any of the ingredients
of the formulation.
Rheumatoid Arthritis
Rheumatoid arthritis patients with abnormal renal function, uncontrolled hypertension, or malignancies
should not receive Neoral
®
.
Psoriasis
Psoriasis patients who are treated with Neoral
®
should not receive concomitant PUVA or UVB therapy,
methotrexate or other immunosuppressive agents, coal tar or radiation therapy. Psoriasis patients with
abnormal renal function, uncontrolled hypertension, or malignancies should not receive Neoral
®
.
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda

WARNINGS
(See also Boxed WARNING)
All Patients
Cyclosporine, the active ingredient of Neoral
®
, can cause nephrotoxicity and hepatotoxicity. The risk
increases with increasing doses of cyclosporine. Renal dysfunction including structural kidney damage is
a potential consequence of Neoral
®
and therefore renal function must be monitored during therapy. Care
should be taken in using cyclosporine with nephrotoxic drugs. (See PRECAUTIONS)
Patients receiving Neoral
®
require frequent monitoring of serum creatinine. (See Special Monitoring
under DOSAGE AND ADMINISTRATION) Elderly patients should be monitored with particular care,
since decreases in renal function also occur with age. If patients are not properly monitored and doses are
not properly adjusted, cyclosporine therapy can be associated with the occurrence of structural kidney
damage and persistent renal dysfunction.
An increase in serum creatinine and BUN may occur during Neoral
®
therapy and reflect a reduction in the
glomerular filtration rate. Impaired renal function at any time requires close monitoring, and frequent
dosage adjustment may be indicated. The frequency and severity of serum creatinine elevations increase
with dose and duration of cyclosporine therapy. These elevations are likely to become more pronounced
without dose reduction or discontinuation.
Because Neoral
®
is not bioequivalent to Sandimmune
®
, conversion from Neoral
®
to Sandimmune
®
using a 1:1 ratio (mg/kg/day) may result in lower cyclosporine blood concentrations. Conversion
from Neoral
®
to Sandimmune
®
should be made with increased monitoring to avoid the potential of
underdosing.
Kidney, Liver, and Heart Transplant
Cyclosporine, the active ingredient of Neoral
®
, can cause nephrotoxicity and hepatotoxicity when used in
high doses. It is not unusual for serum creatinine and BUN levels to be elevated during cyclosporine
therapy. These elevations in renal transplant patients do not necessarily indicate rejection, and each
patient must be fully evaluated before dosage adjustment is initiated.
Based on the historical Sandimmune
®
experience with oral solution, nephrotoxicity associated with
cyclosporine had been noted in 25% of cases of renal transplantation, 38% of cases of cardiac
transplantation, and 37% of cases of liver transplantation. Mild nephrotoxicity was generally noted 2-3
months after renal transplant and consisted of an arrest in the fall of the pre-operative elevations of BUN
and creatinine at a range of 35-45 mg/dl and 2.0-2.5 mg/dl respectively. These elevations were often
responsive to cyclosporine dosage reduction.
More overt nephrotoxicity was seen early after transplantation and was characterized by a rapidly rising
BUN and creatinine. Since these events are similar to renal rejection episodes, care must be taken to
differentiate between them. This form of nephrotoxicity is usually responsive to cyclosporine dosage
reduction.
Although specific diagnostic criteria which reliably differentiate renal graft rejection from drug toxicity
have not been found, a number of parameters have been significantly associated with one or the other. It
should be noted however, that up to 20% of patients may have simultaneous nephrotoxicity and rejection.
Nephrotoxicity vs. Rejection
Parameter Nephrotoxicity Rejection
History
Donor >50 years old or hypotensive
Prolonged kidney preservation
Anti-donor immune response
Retransplant patient
Prolonged anastomosis time
Concomitant nephrotoxic drugs
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda

Clinical
Often >6 weeks postop
b
Prolonged initial nonfunction (acute tubular
necrosis)
Often < 4 weeks postop
b
Fever > 37.5°C
Weight gain > 0.5 kg
Graft swelling and tenderness
Decrease in daily urine volume > 500 mL (or 50%)
Laboratory
CyA serum trough level > 200 ng/mL
Gradual rise in Cr (< 0.15 mg/dl/day)
a
Cr plateau < 25% above baseline
BUN/Cr ≥ 20
CyA serum trough level < 150 ng/mL
Rapid rise in Cr (> 0.3 mg/dl/day)
a
Cr > 25% above baseline
BUN/Cr < 20
Biopsy
Arteriolopathy (medial hypertrophy
a
, hyalinosis,
nodular deposits, intimal thickening, endothelial
vacuolization, progressive scarring)
Tubular atrophy, isometric vacuolization, isolated
calcifications
Minimal edema
Mild focal infiltrates
c
Endovasculitis
c
(proliferation
a
, intimal arteritis
b
,
necrosis, sclerosis)
Tubulitis with RBC
b
and WBC
b
casts, some
irregular vacuolization
Interstitial edema
c
and hemorrhage
b
Diffuse moderate to severe mononuclear
infiltrates
d
Aspiration Cytology
Diffuse interstitial fibrosis, often striped form
CyA deposits in tubular and endothelial cells
Fine isometric vacuolization of tubular cells
Glomerulitis (mononuclear cells)
c
Inflammatory infiltrate with mononuclear
phagocytes, macrophages, lymphoblastoid cells,
and activated T-cells
These strongly express HLA-DR antigens
Urine Cytology
Tubular cells with vacuolization and
granularization
Degenerative tubular cells, plasma cells, and
lymphocyturia > 20% of sediment
Manometry
Ultrasonography
Intracapsular pressure < 40 mm Hg
b
Unchanged graft cross sectional area
Intracapsular pressure > 40 mm Hg
b
Increase in graft cross sectional area
AP diameter ≥ Transverse diameter
Magnetic Resonance
Imagery
Normal appearance
Loss of distinct corticomedullary junction, swelling
image intensity of parachyma approaching that of
psoas, loss of hilar fat
Radionuclide Scan Normal or generally decreased perfusion
Decrease in tubular function
(
131
I-hippuran) > decrease in perfusion (
99m
Tc
DTPA)
Patchy arterial flow
Decrease in perfusion > decrease in tubular
function
Increased uptake of Indium 111 labeled platelets
or Tc-99m in colloid
Therapy Responds to decreased cyclosporine
Responds to increased steroids or antilymphocyte
globulin
a b d
p < 0.05, p < 0.01,
c
p < 0.001, p < 0.0001
A form of a cyclosporine-associated nephropathy is characterized by serial deterioration in renal function
and morphologic changes in the kidneys. From 5%-15% of transplant recipients who have received
cyclosporine will fail to show a reduction in rising serum creatinine despite a decrease or discontinuation
of cyclosporine therapy. Renal biopsies from these patients will demonstrate one or several of the
following alterations: tubular vacuolization, tubular microcalcifications, peritubular capillary congestion,
arteriolopathy, and a striped form of interstitial fibrosis with tubular atrophy. Though none of these
morphologic changes is entirely specific, a diagnosis of cyclosporine-associated structural nephrotoxicity
requires evidence of these findings.
When considering the development of cyclosporine-associated nephropathy, it is noteworthy that several
authors have reported an association between the appearance of interstitial fibrosis and higher cumulative
doses or persistently high circulating trough levels of cyclosporine. This is particularly true during the
first 6 post-transplant months when the dosage tends to be highest and when, in kidney recipients, the
organ appears to be most vulnerable to the toxic effects of cyclosporine. Among other contributing factors
to the development of interstitial fibrosis in these patients are prolonged perfusion time, warm ischemia
time, as well as episodes of acute toxicity, and acute and chronic rejection. The reversibility of interstitial
fibrosis and its correlation to renal function have not yet been determined. Reversibility of arteriolopathy
has been reported after stopping cyclosporine or lowering the dosage.
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
Impaired renal function at any time requires close monitoring, and frequent dosage adjustment may be
indicated.
In the event of severe and unremitting rejection, when rescue therapy with pulse steroids and monoclonal
antibodies fail to reverse the rejection episode, it may be preferable to switch to alternative
immunosuppressive therapy rather than increase the Neoral
®
dose to excessive levels.
Occasionally patients have developed a syndrome of thrombocytopenia and microangiopathic hemolytic
anemia which may result in graft failure. The vasculopathy can occur in the absence of rejection and is
accompanied by avid platelet consumption within the graft as demonstrated by Indium 111 labeled
platelet studies. Neither the pathogenesis nor the management of this syndrome is clear. Though
resolution has occurred after reduction or discontinuation of cyclosporine and 1) administration of
streptokinase and heparin or 2) plasmapheresis, this appears to depend upon early detection with Indium
111 labeled platelet scans. (See ADVERSE REACTIONS)
Significant hyperkalemia (sometimes associated with hyperchloremic metabolic acidosis) and
hyperuricemia have been seen occasionally in individual patients.
Hepatotoxicity associated with cyclosporine use had been noted in 4% of cases of renal transplantation,
7% of cases of cardiac transplantation, and 4% of cases of liver transplantation. This was usually noted
during the first month of therapy when high doses of cyclosporine were used and consisted of elevations
of hepatic enzymes and bilirubin. The chemistry elevations usually decreased with a reduction in dosage.
As in patients receiving other immunosuppressants, those patients receiving cyclosporine are at increased
risk for development of lymphomas and other malignancies, particularly those of the skin. Patients taking
cyclosporine should be warned to avoid excess ultraviolet light exposure. The increased risk appears
related to the intensity and duration of immunosuppression rather than to the use of specific agents.
Because of the danger of oversuppression of the immune system resulting in increased risk of infection or
malignancy, a treatment regimen containing multiple immunosuppressants should be used with caution.
Some malignancies may be fatal. Transplant patients receiving cyclosporine are at increased risk for
serious infection with fatal outcome.
Latent Viral Infections
Immunosuppressed patients are at increased risk for opportunistic infections, including activation of latent
viral infections. These include BK virus-associated nephropathy which has been observed in patients
receiving immunosuppressants, including Neoral. This infection is associated with serious outcomes,
including deteriorating renal function and renal graft loss. Patient monitoring may help detect patients at
risk for BK virus-associated nephropathy. Reduction in immunosuppression should be considered for
patients who develop evidence of BK virus-associated nephropathy.
There have been reports of convulsions in adult and pediatric patients receiving cyclosporine, particularly
in combination with high dose methylprednisolone.
Encephalopathy has been described both in post-marketing reports and in the literature. Manifestations
include impaired consciousness, convulsions, visual disturbances (including blindness), loss of motor
function, movement disorders and psychiatric disturbances. In many cases, changes in the white matter
have been detected using imaging techniques and pathologic specimens. Predisposing factors such as
hypertension, hypomagnesemia, hypocholesterolemia, high-dose corticosteroids, high cyclosporine blood
concentrations, and graft-versus-host disease have been noted in many but not all of the reported cases.
The changes in most cases have been reversible upon discontinuation of cyclosporine, and in some cases
improvement was noted after reduction of dose. It appears that patients receiving liver transplant are more
susceptible to encephalopathy than those receiving kidney transplant. Another rare manifestation of
cyclosporine-induced neurotoxicity, occurring in transplant patients more frequently than in other
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
indications, is optic disc edema including papilloedema, with possible visual impairment, secondary to
benign intracranial hypertension.
Care should be taken in using cyclosporine with nephrotoxic drugs. (See PRECAUTIONS)
Rheumatoid Arthritis
Cyclosporine nephropathy was detected in renal biopsies of 6 out of 60 (10%) rheumatoid arthritis
patients after the average treatment duration of 19 months. Only one patient, out of these 6 patients, was
treated with a dose ≤4 mg/kg/day. Serum creatinine improved in all but one patient after discontinuation
of cyclosporine. The “maximal creatinine increase” appears to be a factor in predicting cyclosporine
nephropathy.
There is a potential, as with other immunosuppressive agents, for an increase in the occurrence of
malignant lymphomas with cyclosporine. It is not clear whether the risk with cyclosporine is greater than
that in rheumatoid arthritis patients or in rheumatoid arthritis patients on cytotoxic treatment for this
indication. Five cases of lymphoma were detected: four in a survey of approximately 2,300 patients
treated with cyclosporine for rheumatoid arthritis, and another case of lymphoma was reported in a
clinical trial. Although other tumors (12 skin cancers, 24 solid tumors of diverse types, and 1 multiple
myeloma) were also reported in this survey, epidemiologic analyses did not support a relationship to
cyclosporine other than for malignant lymphomas.
Patients should be thoroughly evaluated before and during Neoral
®
treatment for the development of
malignancies. Moreover, use of Neoral
®
therapy with other immunosuppressive agents may induce an
excessive immunosuppression which is known to increase the risk of malignancy.
Psoriasis
(See also Boxed WARNINGS for Psoriasis)
Since cyclosporine is a potent immunosuppressive agent with a number of potentially serious side effects,
the risks and benefits of using Neoral
®
should be considered before treatment of patients with psoriasis.
Cyclosporine, the active ingredient in Neoral
®
, can cause nephrotoxicity and hypertension (see
PRECAUTIONS) and the risk increases with increasing dose and duration of therapy. Patients who may
be at increased risk such as those with abnormal renal function, uncontrolled hypertension or
malignancies, should not receive Neoral
®
.
Renal dysfunction is a potential consequence of Neoral
®
therefore renal function must be monitored
during therapy.
Patients receiving Neoral
®
require frequent monitoring of serum creatinine. (See Special Monitoring
under DOSAGE AND ADMINISTRATION) Elderly patients should be monitored with particular care,
since decreases in renal function also occur with age. If patients are not properly monitored and doses are
not properly adjusted, cyclosporine therapy can cause structural kidney damage and persistent renal
dysfunction.
An increase in serum creatinine and BUN may occur during Neoral
®
therapy and reflects a reduction in
the glomerular filtration rate.
Kidney biopsies from 86 psoriasis patients treated for a mean duration of 23 months with 1.2-7.6
mg/kg/day of cyclosporine showed evidence of cyclosporine nephropathy in 18/86 (21%) of the patients.
The pathology consisted of renal tubular atrophy and interstitial fibrosis. On repeat biopsy of 13 of these
patients maintained on various dosages of cyclosporine for a mean of 2 additional years, the number with
cyclosporine induced nephropathy rose to 26/86 (30%). The majority of patients (19/26) were on a dose
of ≥5.0 mg/kg/day (the highest recommended dose is 4 mg/kg/day). The patients were also on
cyclosporine for greater than 15 months (18/26) and/or had a clinically significant increase in serum
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
creatinine for greater than 1 month (21/26). Creatinine levels returned to normal range in 7 of 11 patients
in whom cyclosporine therapy was discontinued.
There is an increased risk for the development of skin and lymphoproliferative malignancies in
cyclosporine-treated psoriasis patients. The relative risk of malignancies is comparable to that observed in
psoriasis patients treated with other immunosuppressive agents.
Tumors were reported in 32 (2.2%) of 1439 psoriasis patients treated with cyclosporine worldwide from
clinical trials. Additional tumors have been reported in 7 patients in cyclosporine postmarketing
experience. Skin malignancies were reported in 16 (1.1%) of these patients; all but 2 of them had
previously received PUVA therapy. Methotrexate was received by 7 patients. UVB and coal tar had been
used by 2 and 3 patients, respectively. Seven patients had either a history of previous skin cancer or a
potentially predisposing lesion was present prior to cyclosporine exposure. Of the 16 patients with skin
cancer, 11 patients had 18 squamous cell carcinomas and 7 patients had 10 basal cell carcinomas.
There were two lymphoproliferative malignancies; one case of non-Hodgkin’s lymphoma which required
chemotherapy, and one case of mycosis fungoides which regressed spontaneously upon discontinuation of
cyclosporine. There were four cases of benign lymphocytic infiltration: 3 regressed spontaneously upon
discontinuation of cyclosporine, while the fourth regressed despite continuation of the drug. The
remainder of the malignancies, 13 cases (0.9%), involved various organs.
Patients should not be treated concurrently with cyclosporine and PUVA or UVB, other radiation
therapy, or other immunosuppressive agents, because of the possibility of excessive
immunosuppression and the subsequent risk of malignancies. (See CONTRAINDICATIONS) Patients
should also be warned to protect themselves appropriately when in the sun, and to avoid excessive sun
exposure. Patients should be thoroughly evaluated before and during treatment for the presence of
malignancies remembering that malignant lesions may be hidden by psoriatic plaques. Skin lesions not
typical of psoriasis should be biopsied before starting treatment. Patients should be treated with Neoral
®
only after complete resolution of suspicious lesions, and only if there are no other treatment options. (See
Special Monitoring for Psoriasis Patients)
PRECAUTIONS
General
Hypertension
Cyclosporine is the active ingredient of Neoral
®
. Hypertension is a common side effect of cyclosporine
therapy which may persist. (See ADVERSE REACTIONS and DOSAGE AND ADMINISTRATION for
monitoring recommendations) Mild or moderate hypertension is encountered more frequently than severe
hypertension and the incidence decreases over time. In recipients of kidney, liver, and heart allografts
treated with cyclosporine, antihypertensive therapy may be required. (See Special Monitoring of
Rheumatoid Arthritis and Psoriasis Patients) However, since cyclosporine may cause hyperkalemia,
potassium-sparing diuretics should not be used. While calcium antagonists can be effective agents in
treating cyclosporine-associated hypertension, they can interfere with cyclosporine metabolism. (See
Drug Interactions)
Vaccination
During treatment with cyclosporine, vaccination may be less effective; and the use of live attenuated
vaccines should be avoided.
Special Monitoring of Rheumatoid Arthritis Patients
Before initiating treatment, a careful physical examination, including blood pressure measurements (on at
least two occasions) and two creatinine levels to estimate baseline should be performed. Blood pressure
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
and serum creatinine should be evaluated every 2 weeks during the initial 3 months and then monthly if
the patient is stable. It is advisable to monitor serum creatinine and blood pressure always after an
increase of the dose of nonsteroidal anti-inflammatory drugs and after initiation of new nonsteroidal anti-
inflammatory drug therapy during Neoral
®
treatment. If co-administered with methotrexate, CBC and
liver function tests are recommended to be monitored monthly. (See also PRECAUTIONS, General,
Hypertension)
In patients who are receiving cyclosporine, the dose of Neoral
®
should be decreased by 25%-50% if
hypertension occurs. If hypertension persists, the dose of Neoral
®
should be further reduced or blood
pressure should be controlled with antihypertensive agents. In most cases, blood pressure has returned to
baseline when cyclosporine was discontinued.
In placebo-controlled trials of rheumatoid arthritis patients, systolic hypertension (defined as an
occurrence of two systolic blood pressure readings >140 mmHg) and diastolic hypertension (defined as
two diastolic blood pressure readings >90 mmHg) occurred in 33% and 19% of patients treated with
cyclosporine, respectively. The corresponding placebo rates were 22% and 8%.
Special Monitoring for Psoriasis Patients
Before initiating treatment, a careful dermatological and physical examination, including blood pressure
measurements (on at least two occasions) should be performed. Since Neoral
®
is an immunosuppressive
agent, patients should be evaluated for the presence of occult infection on their first physical examination
and for the presence of tumors initially, and throughout treatment with Neoral
®
. Skin lesions not typical
for psoriasis should be biopsied before starting Neoral
®
. Patients with malignant or premalignant changes
of the skin should be treated with Neoral
®
only after appropriate treatment of such lesions and if no other
treatment option exists.
Baseline laboratories should include serum creatinine (on two occasions), BUN, CBC, serum magnesium,
potassium, uric acid, and lipids.
The risk of cyclosporine nephropathy is reduced when the starting dose is low (2.5 mg/kg/day), the
maximum dose does not exceed 4.0 mg/kg/day, serum creatinine is monitored regularly while
cyclosporine is administered, and the dose of Neoral
®
is decreased when the rise in creatinine is greater
than or equal to 25% above the patient’s pretreatment level. The increase in creatinine is generally
reversible upon timely decrease of the dose of Neoral
®
or its discontinuation.
Serum creatinine and BUN should be evaluated every 2 weeks during the initial 3 months of therapy and
then monthly if the patient is stable. If the serum creatinine is greater than or equal to 25% above the
patient’s pretreatment level, serum creatinine should be repeated within two weeks. If the change in serum
creatinine remains greater than or equal to 25% above baseline, Neoral
®
should be reduced by 25%-50%.
If at any time the serum creatinine increases by greater than or equal to 50% above pretreatment level,
Neoral
®
should be reduced by 25%-50%. Neoral
®
should be discontinued if reversibility (within 25% of
baseline) of serum creatinine is not achievable after two dosage modifications. It is advisable to monitor
serum creatinine after an increase of the dose of nonsteroidal anti-inflammatory drug and after initiation
of new nonsteroidal anti-inflammatory therapy during Neoral
®
treatment.
Blood pressure should be evaluated every 2 weeks during the initial 3 months of therapy and then
monthly if the patient is stable, or more frequently when dosage adjustments are made. Patients without a
history of previous hypertension before initiation of treatment with Neoral
®
, should have the drug reduced
by 25%-50% if found to have sustained hypertension. If the patient continues to be hypertensive despite
multiple reductions of Neoral
®
, then Neoral
®
should be discontinued. For patients with treated
hypertension, before the initiation of Neoral
®
therapy, their medication should be adjusted to control
hypertension while on Neoral
®
. Neoral
®
should be discontinued if a change in hypertension management
is not effective or tolerable.
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda

CBC, uric acid, potassium, lipids, and magnesium should also be monitored every 2 weeks for the first 3
months of therapy, and then monthly if the patient is stable or more frequently when dosage adjustments
are made. Neoral
®
dosage should be reduced by 25%-50% for any abnormality of clinical concern.
In controlled trials of cyclosporine in psoriasis patients, cyclosporine blood concentrations did not
correlate well with either improvement or with side effects such as renal dysfunction.
Information for Patients: Patients should be advised that any change of cyclosporine formulation
should be made cautiously and only under physician supervision because it may result in the need
for a change in dosage.
Patients should be informed of the necessity of repeated laboratory tests while they are receiving
cyclosporine. Patients should be advised of the potential risks during pregnancy and informed of the
increased risk of neoplasia. Patients should also be informed of the risk of hypertension and renal
dysfunction.
Patients should be advised that during treatment with cyclosporine, vaccination may be less effective and
the use of live attenuated vaccines should be avoided.
Patients should be given careful dosage instructions. Neoral
®
Oral Solution (cyclosporine oral solution,
USP) MODIFIED should be diluted, preferably with orange or apple juice that is at room temperature.
The combination of Neoral
®
Oral Solution (cyclosporine oral solution, USP) MODIFIED with milk can
be unpalatable.
Patients should be advised to take Neoral
®
on a consistent schedule with regard to time of day and
relation to meals. Grapefruit and grapefruit juice affect metabolism, increasing blood concentration of
cyclosporine, thus should be avoided.
Laboratory Tests
In all patients treated with cyclosporine, renal and liver functions should be assessed repeatedly by
measurement of serum creatinine, BUN, serum bilirubin, and liver enzymes. Serum lipids, magnesium,
and potassium should also be monitored. Cyclosporine blood concentrations should be routinely
monitored in transplant patients (see DOSAGE AND ADMINISTRATION, Blood Concentration
Monitoring in Transplant Patients), and periodically monitored in rheumatoid arthritis patients.
Drug Interactions
All of the individual drugs cited below are well substantiated to interact with cyclosporine. In addition,
concomitant non-steroidal anti-inflammatory drugs, particularly in the setting of dehydration, may
potentiate renal dysfunction.
Drugs That May Potentiate Renal Dysfunction
Anti-
Antibiotics
Antineoplastics
Antifungals
inflammatory
Drugs
Gastrointestinal
Agents
Immunosuppressives Other Drugs
fibric acid
ciprofloxacin melphalan amphotericin B azapropazon cimetidine tacrolimus derivatives
gentamicin ketoconazole colchicine ranitidine
(e.g., bezafibrate,
fenofibrate)
methotrexate
tobramycin
diclofenac
vancomycin naproxen
trimethoprim with
sulfamethoxazole
sulindac
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda

Drugs That Alter Cyclosporine Concentrations
Cyclosporine is extensively metabolized by CYP 3A isoenzymes, in particular CYP3A4, and is a
substrate of the multidrug efflux transporter P-glycoprotein. Various agents are known to either increase
or decrease plasma or whole blood of cyclosporine levels usually by inhibition or induction of CYP3A4
or P-glycoprotein transporter or both. Compounds that decrease cyclosporine absorption such as orlistat
should be avoided. Monitoring of circulating cyclosporine concentrations and appropriate Neoral
®
dosage
adjustment are essential when these drugs are used concomitantly. (See Blood Concentration Monitoring)
Drugs That Increase Cyclosporine Concentrations
Calcium Channel Blockers Antifungals Antibiotics Glucocorticoids Other Drugs
diltiazem fluconazole azithromycin methylprednisolone Allopurinol
nicardipine itraconazole clarithromycin Amiodarone
verapamil ketoconazole erythromycin Bromocriptine
voriconazole quinupristin/ dalfopristin colchicine
danazol
imatinib
metoclopramide
nefazodone
oral contraceptives
The HIV protease inhibitors (e.g., indinavir, nelfinavir, ritonavir, and saquinavir) are known to inhibit
cytochrome P-450 3A and thus could potentially increase the concentrations of cyclosporine, however no
formal studies of the interaction are available. Care should be exercised when these drugs are
administered concomitantly.
Grapefruit and grapefruit juice affect metabolism, increasing blood concentrations of cyclosporine, thus
should be avoided.
Drugs/Dietary Supplements That Decrease Cyclosporine Concentrations
Antibiotics Anticonvulsants Other Drugs/Dietary Supplements
nafcillin carbamazepine bosentan St. John’s Wort
rifampin oxcarbazepine octreotide
phenobarbital orlistat
phenytoin sulfinpyrazone
terbinafine
ticlopidine
There have been reports of a serious drug interaction between cyclosporine and the herbal dietary
supplement, St. John’s Wort. This interaction has been reported to produce a marked reduction in
the blood concentrations of cyclosporine, resulting in subtherapeutic levels, rejection of
transplanted organs, and graft loss.
Rifabutin is known to increase the metabolism of other drugs metabolized by the cytochrome P-450
system. The interaction between rifabutin and cyclosporine has not been studied. Care should be exercised
when these two drugs are administered concomitantly.
Nonsteroidal Anti-inflammatory Drug (NSAID) Interactions
Clinical status and serum creatinine should be closely monitored when cyclosporine is used with
nonsteroidal anti-inflammatory agents in rheumatoid arthritis patients. (See WARNINGS)
Pharmacodynamic interactions have been reported to occur between cyclosporine and both naproxen and
sulindac, in that concomitant use is associated with additive decreases in renal function, as determined by
99m
Tc-diethylenetriaminepentaacetic acid (DTPA) and (p-aminohippuric acid) PAH clearances. Although
concomitant administration of diclofenac does not affect blood levels of cyclosporine, it has been
associated with approximate doubling of diclofenac blood levels and occasional reports of reversible
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
decreases in renal function. Consequently, the dose of diclofenac should be in the lower end of the
therapeutic range.
Methotrexate Interaction
Preliminary data indicate that when methotrexate and cyclosporine were co-administered to rheumatoid
arthritis patients (N=20), methotrexate concentrations (AUCs) were increased approximately 30% and the
concentrations (AUCs) of its metabolite, 7-hydroxy methotrexate, were decreased by approximately 80%.
The clinical significance of this interaction is not known. Cyclosporine concentrations do not appear to
have been altered (N=6).
Other Drug Interactions
Cyclosporine is an inhibitor of CYP3A4 and of the multidrug efflux transporter P-glycoprotein and
may increase plasma concentrations of comedications that are substrates of CYP3A4 or P-
glycoprotein or both.
Cyclosporine may reduce the clearance of digoxin, colchicine, prednisolone, HMG-CoA reductase
inhibitors (statins) and etoposide. Severe digitalis toxicity has been seen within days of starting
cyclosporine in several patients taking digoxin. There are also reports on the potential of
cyclosporine to enhance the toxic effects of colchicine such as myopathy and neuropathy, especially
in patients with renal dysfunction. If digoxin or colchicine are used concurrently with cyclosporine,
close clinical observation is required in order to enable early detection of toxic manifestations of
digoxin or colchicine, followed by reduction of dosage or its withdrawal.
Literature and postmarketing cases of myotoxicity, including muscle pain and weakness, myositis,
and rhabdomyolysis, have been reported with concomitant administration of cyclosporine with
lovastatin, simvastatin, atorvastatin, pravastatin, and, rarely fluvastatin. When concurrently
administered with cyclosporine, the dosage of these statins should be reduced according to label
recommendations. Statin therapy needs to be temporarily withheld or discontinued in patients with
signs and symptoms of myopathy or those with risk factors predisposing to severe renal injury,
including renal failure, secondary to rhabdomyolysis.
Cyclosporine may increase the plasma concentrations of repaglinide and thereby increase the risk of
hypoglycemia. In 12 healthy male subjects who received two doses of 100mg cyclosporine capsule
orally 12 hours apart with a single dose of 0.25mg repaglinide tablet (one half of a 0.5mg tablet)
orally 13 hours after the cyclosporine initial dose, the repaglinide mean Cmax and AUC were
increased 1.8 fold (range: 0.6 - 3.7 fold) and 2.4 fold (range 1.2 - 5.3 fold), respectively. Close
monitoring of blood glucose level is advisable for a patient taking cyclosporine and repaglinide
concomitantly.
Cyclosporine should not be used with potassium-sparing diuretics because hyperkalemia can occur.
Caution is also required when cyclosporine is co-administered with potassium sparing drugs (e.g.
angiotensin converting enzyme inhibitors, angiotensin II receptor antagonists), potassium containing
drugs as well as in patients on a potassium rich diet. Control of potassium levels in these situations is
advisable.
Elevations in serum creatinine were observed in studies using sirolimus in combination with full-dose
cyclosporine. This effect is often reversible with cyclosporine dose reduction. Simultaneous co-
administration of cyclosporine significantly increases blood levels of sirolimus. To minimize increases in
sirolimus concentrations, it is recommended that sirolimus be given 4 hours after cyclosporine
administration.
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
During treatment with cyclosporine, vaccination may be less effective. The use of live vaccines should be
avoided. Frequent gingival hyperplasia with nifedipine, and convulsions with high dose
methylprednisolone have been reported.
Psoriasis patients receiving other immunosuppressive agents or radiation therapy (including PUVA and
UVB) should not receive concurrent cyclosporine because of the possibility of excessive
immunosuppression.
For additional information on Cyclosporine Drug Interactions please contact Novartis Medical Affairs
Department at 888-NOW-NOVA [888-669-6682].
Carcinogenesis, Mutagenesis, and Impairment of Fertility
Carcinogenicity studies were carried out in male and female rats and mice. In the 78-week mouse study,
evidence of a statistically significant trend was found for lymphocytic lymphomas in females, and the
incidence of hepatocellular carcinomas in mid-dose males significantly exceeded the control value. In the
24-month rat study, pancreatic islet cell adenomas significantly exceeded the control rate in the low dose
level. Doses used in the mouse and rat studies were 0.01 to 0.16 times the clinical maintenance dose (6
mg/kg). The hepatocellular carcinomas and pancreatic islet cell adenomas were not dose related.
Published reports indicate that co-treatment of hairless mice with UV irradiation and cyclosporine or other
immunosuppressive agents shorten the time to skin tumor formation compared to UV irradiation alone.
Cyclosporine was not mutagenic in appropriate test systems. Cyclosporine has not been found to be
mutagenic/genotoxic in the Ames Test, the V79-HGPRT Test, the micronucleus test in mice and Chinese
hamsters, the chromosome-aberration tests in Chinese hamster bone-marrow, the mouse dominant lethal
assay, and the DNA-repair test in sperm from treated mice. A recent study analyzing sister chromatid
exchange (SCE) induction by cyclosporine using human lymphocytes in vitro gave indication of a
positive effect (i.e., induction of SCE), at high concentrations in this system. In two published research
studies, rabbits exposed to cyclosporine in utero (10 mg/kg/day subcutaneously) demonstrated reduced
numbers of nephrons, renal hypertrophy, systemic hypertension and progressive renal insufficiency up to
35 weeks of age. Pregnant rats which received 12 mg/kg/day of cyclosporine intravenously (twice the
recommended human intravenous dose) had fetuses with an increased incidence of ventricular septal
defect. These findings have not been demonstrated in other species and their relevance for humans is
unknown.
No impairment in fertility was demonstrated in studies in male and female rats.
Widely distributed papillomatosis of the skin was observed after chronic treatment of dogs with
cyclosporine at 9 times the human initial psoriasis treatment dose of 2.5 mg/kg, where doses are
expressed on a body surface area basis. This papillomatosis showed a spontaneous regression upon
discontinuation of cyclosporine.
An increased incidence of malignancy is a recognized complication of immunosuppression in recipients
of organ transplants and patients with rheumatoid arthritis and psoriasis. The most common forms of
neoplasms are non-Hodgkin’s lymphoma and carcinomas of the skin. The risk of malignancies in
cyclosporine recipients is higher than in the normal, healthy population but similar to that in patients
receiving other immunosuppressive therapies. Reduction or discontinuance of immunosuppression may
cause the lesions to regress.
In psoriasis patients on cyclosporine, development of malignancies, especially those of the skin has been
reported. (See WARNINGS) Skin lesions not typical for psoriasis should be biopsied before starting
cyclosporine treatment. Patients with malignant or premalignant changes of the skin should be treated
with cyclosporine only after appropriate treatment of such lesions and if no other treatment option exists.
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
Pregnancy:
Pregnancy Category C
Animal studies have shown reproductive toxicity in rats and rabbits. Cyclosporine gave no evidence of
mutagenic or teratogenic effects in the standard test systems with oral application (rats up to 17 mg/kg
and rabbits up to 30 mg/kg per day orally.) Only at dose levels toxic to dams, were adverse effects seen in
reproduction studies in rats. Cyclosporine has been shown to be embryo- and fetotoxic in rats and rabbits
following oral administration at maternally toxic doses. Fetal toxicity was noted in rats at 0.8 and rabbits
at 5.4 times the transplant doses in humans of 6.0 mg/kg, where dose corrections are based on body
surface area. Cyclosporine was embryo- and fetotoxic as indicated by increased pre- and postnatal
mortality and reduced fetal weight together with related skeletal retardation.
There are no adequate and well-controlled studies in pregnant women therefore, Neoral
®
should not be
used during pregnancy unless the potential benefit to the mother justifies the potential risk to the fetus.
In pregnant transplant recipients who are being treated with immunosuppressants the risk of premature
birth is increased. The following data represent the reported outcomes of 116 pregnancies in women
receiving cyclosporine during pregnancy, 90% of whom were transplant patients, and most of whom
received cyclosporine throughout the entire gestational period. The only consistent patterns of
abnormality were premature birth (gestational period of 28 to 36 weeks) and low birth weight for
gestational age. Sixteen fetal losses occurred. Most of the pregnancies (85 of 100) were complicated by
disorders; including, pre-eclampsia, eclampsia, premature labor, abruptio placentae, oligohydramnios, Rh
incompatibility, and fetoplacental dysfunction. Pre-term delivery occurred in 47%. Seven malformations
were reported in 5 viable infants and in 2 cases of fetal loss. Twenty-eight percent of the infants were
small for gestational age. Neonatal complications occurred in 27%. Therefore, the risks and benefits of
using Neoral
®
during pregnancy should be carefully weighed.
A limited number of observations in children exposed to cyclosporine in utero are available, up to an age
of approximately 7 years. Renal function and blood pressure in these children were normal.
Because of the possible disruption of maternal-fetal interaction, the risk/benefit ratio of using Neoral
®
in
psoriasis patients during pregnancy should carefully be weighed with serious consideration for
discontinuation of Neoral
®
.
Nursing Mothers
Cyclosporine passes into breast milk. Mothers receiving treatment with Neoral
®
should not breast-feed.
Pediatric Use
Although no adequate and well-controlled studies have been completed in children, transplant recipients
as young as one year of age have received Neoral
®
with no unusual adverse effects. The safety and
efficacy of Neoral
®
treatment in children with juvenile rheumatoid arthritis or psoriasis below the age of
18 have not been established.
Geriatric Use
In rheumatoid arthritis clinical trials with cyclosporine, 17.5% of patients were age 65 or older. These
patients were more likely to develop systolic hypertension on therapy, and more likely to show serum
creatinine rises ≥50% above the baseline after 3-4 months of therapy.
Clinical studies of Neoral
®
in transplant and psoriasis patients did not include a sufficient number of
subjects aged 65 and over to determine whether they respond differently from younger subjects. Other
reported clinical experiences have not identified differences in response between the elderly and younger
patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda

of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and
of concomitant disease or other drug therapy.
ADVERSE REACTIONS
Kidney, Liver, and Heart Transplantation
The principal adverse reactions of cyclosporine therapy are renal dysfunction, tremor, hirsutism,
hypertension, and gum hyperplasia.
Hypertension, which is usually mild to moderate, may occur in approximately 50% of patients following
renal transplantation and in most cardiac transplant patients.
Glomerular capillary thrombosis has been found in patients treated with cyclosporine and may progress to
graft failure. The pathologic changes resembled those seen in the hemolytic-uremic syndrome and
included thrombosis of the renal microvasculature, with platelet-fibrin thrombi occluding glomerular
capillaries and afferent arterioles, microangiopathic hemolytic anemia, thrombocytopenia, and decreased
renal function. Similar findings have been observed when other immunosuppressives have been employed
post-transplantation.
Hypomagnesemia has been reported in some, but not all, patients exhibiting convulsions while on
cyclosporine therapy. Although magnesium-depletion studies in normal subjects suggest that
hypomagnesemia is associated with neurologic disorders, multiple factors, including hypertension, high
dose methylprednisolone, hypocholesterolemia, and nephrotoxicity associated with high plasma
concentrations of cyclosporine appear to be related to the neurological manifestations of cyclosporine
toxicity.
In controlled studies, the nature, severity, and incidence of the adverse events that were observed in 493
transplanted patients treated with Neoral
®
were comparable with those observed in 208 transplanted
patients who received Sandimmune
®
in these same studies when the dosage of the two drugs was adjusted
to achieve the same cyclosporine blood trough concentrations.
Based on the historical experience with Sandimmune
®
, the following reactions occurred in 3% or greater
of 892 patients involved in clinical trials of kidney, heart, and liver transplants.
Randomized Kidney Patients Cyclosporine Patients (Sandimmune
®
)
Sandimmune
®
Azathioprine Kidney Heart Liver
Body System Adverse Reactions (N=227)% (N=228)% (N=705)% (N=112)% (N=75)%
Genitourinary Renal Dysfunction 32 6 25 38 37
Cardiovascular Hypertension 26 18 13 53 27
Cramps 4 <1 2 <1 0
Skin Hirsutism 21 <1 21 28 45
Acne 6 8 2 2 1
Central Nervous System Tremor 12 0 21 31 55
Convulsions 3 1 1 4 5
Headache 2 <1 2 15 4
Gastrointestinal Gum Hyperplasia 4 0 9 5 16
Diarrhea 3 <1 3 4 8
Nausea/Vomiting 2 <1 4 10 4
Hepatotoxicity <1 <1 4 7 4
Abdominal Discomfort <1 0 <1 7 0
Autonomic Nervous
System Paresthesia 3 0 1 2 1
Flushing <1 0 4 0 4
Hematopoietic Leukopenia 2 19 <1 6 0
Lymphoma <1 0 1 6 1
Respiratory Sinusitis <1 0 4 3 7
Miscellaneous Gynecomastia <1 0 <1 4 3
Among 705 kidney transplant patients treated with cyclosporine oral solution (Sandimmune
®
)
in clinical
trials, the reason for treatment discontinuation was renal toxicity in 5.4%, infection in 0.9%, lack of
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda

efficacy in 1.4%, acute tubular necrosis in 1.0%, lymphoproliferative disorders in 0.3%, hypertension in
0.3%, and other reasons in 0.7% of the patients.
The following reactions occurred in 2% or less of Sandimmune
®
-treated patients: allergic reactions,
anemia, anorexia, confusion, conjunctivitis, edema, fever, brittle fingernails, gastritis, hearing loss,
hiccups, hyperglycemia, muscle pain, peptic ulcer, thrombocytopenia, tinnitus.
The following reactions occurred rarely: anxiety, chest pain, constipation, depression, hair breaking,
hematuria, joint pain, lethargy, mouth sores, myocardial infarction, night sweats, pancreatitis, pruritus,
swallowing difficulty, tingling, upper GI bleeding, visual disturbance, weakness, weight loss.
Patients receiving immunosuppressive therapies, including cyclosporine and cyclosporine -containing
regimens, are at increased risk of infections (viral, bacterial, fungal, parasitic). Both generalized and
localized infections can occur. Pre-existing infections may also be aggravated. Fatal outcomes have been
reported. (see Warnings)
Infectious Complications in Historical Randomized Studies
in Renal Transplant Patients Using Sandimmune®
Cyclosporine Treatment Azathioprine with Steroids*
(N=227) (N=228)
Complication % of Complications % of Complications
Septicemia 5.3 4.8
Abscesses 4.4 5.3
Systemic Fungal Infection 2.2 3.9
Local Fungal Infection 7.5 9.6
Cytomegalovirus 4.8 12.3
Other Viral Infections 15.9 18.4
Urinary Tract Infections 21.1 20.2
Wound and Skin Infections 7.0 10.1
Pneumonia 6.2 9.2
*Some patients also received ALG.
Postmarketing Experience, Kidney, Liver and Heart Transplantation
BK virus associated nephropathy has been observed in patients receiving immunosuppressants, including
Neoral. This infection is associated with serious outcomes, including deteriorating renal function and
renal graft loss. (see WARNINGS, Kidney, Liver and Heart Transplant).
Rheumatoid Arthritis
The principal adverse reactions associated with the use of cyclosporine in rheumatoid arthritis are renal
dysfunction (see WARNINGS), hypertension (see PRECAUTIONS), headache, gastrointestinal
disturbances, and hirsutism/hypertrichosis.
In rheumatoid arthritis patients treated in clinical trials within the recommended dose range, cyclosporine
therapy was discontinued in 5.3% of the patients because of hypertension and in 7% of the patients
because of increased creatinine. These changes are usually reversible with timely dose decrease or drug
discontinuation. The frequency and severity of serum creatinine elevations increase with dose and
duration of cyclosporine therapy. These elevations are likely to become more pronounced without dose
reduction or discontinuation.
The following adverse events occurred in controlled clinical trials:
Neoral
®
/Sandimmune
®
Rheumatoid Arthritis
Percentage of Patients with Adverse Events ≥3% in any Cyclosporine Treated Group
Studies Study Study Study Study Studies
651+652+2008 302 654 654 302 651+652+2008
Methotrexate & Methotrexate
Body
System
Preferred
Term
Sandimmune
®
†
(N=269)
Sandimmune
®
(N=155)
Sandimmune
®
(N=74)
& Placebo
(N=73)
Neoral
®
(N=143)
Placebo
(N=201)
Autonomic Nervous System Disorders
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
Flushing 2% 2% 3% 0% 5% 2%
Body As A Whole–General Disorders
Accidental
Trauma 0% 1% 10% 4% 4% 0%
Edema NOS* 5% 14% 12% 4% 10% <1%
Fatigue 6% 3% 8% 12% 3% 7%
Fever 2% 3% 0% 0% 2% 4%
Influenza-like
symptoms <1% 6% 1% 0% 3% 2%
Pain 6% 9% 10% 15% 13% 4%
Rigors 1% 1% 4% 0% 3% 1%
Cardiovascular Disorders
Arrhythmia 2% 5% 5% 6% 2% 1%
Chest Pain 4% 5% 1% 1% 6% 1%
Hypertension 8% 26% 16% 12% 25% 2%
Central and Peripheral Nervous System Disorders
Dizziness 8% 6% 7% 3% 8% 3%
Headache 17% 23% 22% 11% 25% 9%
Migraine 2% 3% 0% 0% 3% 1%
Paresthesia 8% 7% 8% 4% 11% 1%
Tremor 8% 7% 7% 3% 13% 4%
Gastrointestinal System Disorders
Abdominal Pain 15% 15% 15% 7% 15% 10%
Anorexia 3% 3% 1% 0% 3% 3%
Diarrhea 12%
12% 18% 15% 13% 8%
Dyspepsia 12% 12% 10% 8% 8% 4%
Flatulence 5% 5% 5% 4% 4% 1%
Gastrointestinal
Disorder NOS* 0% 2% 1% 4% 4% 0%
Gingivitis 4% 3% 0% 0% 0% 1%
Gum Hyperplasia 2% 4% 1% 3% 4% 1%
Nausea 23% 14% 24% 15% 18% 14%
Rectal
Hemorrhage 0% 3% 0% 0% 1% 1%
Stomatitis 7% 5% 16% 12% 6% 8%
Vomiting 9% 8% 14% 7% 6% 5%
Hearing and Vestibular Disorders
Ear Disorder
NOS* 0% 5% 0% 0% 1% 0%
Metabolic and Nutritional Disorders
Hypomagnesemia
Musculoskeletal System Disorders
0% 4% 0% 0% 6% 0%
Arthropathy 0% 5% 0% 1% 4% 0%
Leg Cramps /
Involuntary
Muscle
Contractions 2% 11% 11% 3% 12% 1%
Psychiatric Disorders
Depression 3% 6% 3% 1% 1% 2%
Insomnia 4% 1% 1% 0% 3% 2%
Renal
Creatinine
elevations ≥30% 43% 39% 55% 19% 48% 13%
Creatinine
elevations ≥50%
24% 18% 26% 8% 18%
3%
Reproductive Disorders, Female
Leukorrhea 1% 0% 4% 0% 1% 0%
Menstrual
Disorder 3% 2% 1% 0% 1% 1%
Respiratory System Disorders
Bronchitis 1% 3% 1% 0% 1% 3%
Coughing 5% 3% 5% 7% 4% 4%
Dyspnea 5% 1% 3% 3% 1% 2%
Infection NOS* 9% 5% 0% 7% 3% 10%
Pharyngitis 3% 5% 5% 6% 4% 4%
Pneumonia 1% 0% 4% 0% 1% 1%
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda

Rhinitis 0% 3% 11% 10% 1% 0%
Sinusitis 4% 4% 8% 4% 3% 3%
Upper Respiratory
Tract
0% 14% 23% 15% 13% 0%
Skin and Appendages Disorders
Alopecia 3% 0% 1% 1% 4% 4%
Bullous Eruption 1% 0% 4% 1% 1% 1%
Hypertrichosis 19% 17% 12% 0% 15% 3%
Rash 7% 12% 10% 7% 8% 10%
Skin Ulceration 1% 1% 3% 4% 0% 2%
Urinary System Disorders
Dysuria
Micturition
0% 0% 11% 3% 1% 2%
Frequency
2% 4% 3% 1% 2% 2%
NPN, Increased 0% 19% 12% 0% 18% 0%
Urinary Tract
Infection 0% 3% 5% 4% 3% 0%
Vascular (Extracardiac) Disorders
Purpura 3% 4% 1% 1% 2% 0%
† Includes patients in 2.5 mg/kg/day dose group only. *NOS = Not Otherwise Specified.
In addition, the following adverse events have been reported in 1% to <3% of the rheumatoid arthritis
patients in the cyclosporine treatment group in controlled clinical trials.
Autonomic Nervous System: dry mouth, increased sweating;
Body as a Whole: allergy, asthenia, hot flushes, malaise, overdose, procedure NOS*, tumor NOS*,
weight decrease, weight increase;
Cardiovascular: abnormal heart sounds, cardiac failure, myocardial infarction, peripheral ischemia;
Central and Peripheral Nervous System: hypoesthesia, neuropathy, vertigo;
Endocrine: goiter;
Gastrointestinal: constipation, dysphagia, enanthema, eructation, esophagitis, gastric ulcer, gastritis,
gastroenteritis, gingival bleeding, glossitis, peptic ulcer, salivary gland enlargement, tongue disorder,
tooth disorder;
Infection: abscess, bacterial infection, cellulitis, folliculitis, fungal infection, herpes simplex, herpes
zoster, renal abscess, moniliasis, tonsillitis, viral infection;
Hematologic: anemia, epistaxis, leukopenia, lymphadenopathy;
Liver and Biliary System: bilirubinemia;
Metabolic and Nutritional: diabetes mellitus, hyperkalemia, hyperuricemia, hypoglycemia;
Musculoskeletal System: arthralgia, bone fracture, bursitis, joint dislocation, myalgia, stiffness,
synovial cyst, tendon disorder;
Neoplasms: breast fibroadenosis, carcinoma;
Psychiatric: anxiety, confusion, decreased libido, emotional lability, impaired concentration, increased
libido, nervousness, paroniria, somnolence;
Reproductive (Female): breast pain, uterine hemorrhage;
Respiratory System: abnormal chest sounds, bronchospasm;
Skin and Appendages: abnormal pigmentation, angioedema, dermatitis, dry skin, eczema, nail
disorder, pruritus, skin disorder, urticaria;
Special Senses: abnormal vision, cataract, conjunctivitis, deafness, eye pain, taste perversion, tinnitus,
vestibular disorder;
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda

Urinary System: abnormal urine, hematuria, increased BUN, micturition urgency, nocturia, polyuria,
pyelonephritis, urinary incontinence.
*NOS = Not Otherwise Specified.
Psoriasis
The principal adverse reactions associated with the use of cyclosporine in patients with psoriasis are renal
dysfunction, headache, hypertension, hypertriglyceridemia, hirsutism/hypertrichosis, paresthesia or
hyperesthesia, influenza-like symptoms, nausea/vomiting, diarrhea, abdominal discomfort, lethargy, and
musculoskeletal or joint pain.
In psoriasis patients treated in US controlled clinical studies within the recommended dose range,
cyclosporine therapy was discontinued in 1.0% of the patients because of hypertension and in 5.4% of the
patients because of increased creatinine. In the majority of cases, these changes were reversible after dose
reduction or discontinuation of cyclosporine.
There has been one reported death associated with the use of cyclosporine in psoriasis. A 27-year-old
male developed renal deterioration and was continued on cyclosporine. He had progressive renal failure
leading to death.
Frequency and severity of serum creatinine increases with dose and duration of cyclosporine therapy.
These elevations are likely to become more pronounced and may result in irreversible renal damage
without dose reduction or discontinuation.
Adverse Events Occurring in 3% or More of Psoriasis Patients in Controlled Clinical Trials
Body System* Preferred Term Neoral
®
(N=182) Sandimmune
®
(N=185)
Infection or Potential Infection 24.7% 24.3%
Influenza-Like Symptoms 9.9% 8.1%
Upper Respiratory Tract Infections 7.7% 11.3%
Cardiovascular System 28.0% 25.4%
Hypertension** 27.5% 25.4%
Urinary System 24.2% 16.2%
Increased Creatinine 19.8% 15.7%
Central and Peripheral Nervous
System 26.4% 20.5%
Headache 15.9% 14.0%
Paresthesia 7.1% 4.8%
Musculoskeletal System 13.2% 8.7%
Arthralgia 6.0% 1.1%
Body As a Whole–General 29.1% 22.2%
Pain 4.4% 3.2%
Metabolic and Nutritional 9.3% 9.7%
Reproductive, Female 8.5% (4 of 47 females) 11.5% (6 of 52 females)
Resistance Mechanism 18.7% 21.1%
Skin and Appendages 17.6% 15.1%
Hypertrichosis 6.6% 5.4%
Respiratory System
Bronchospasm, Coughing, Dyspnea,
5.0% 6.5%
Rhinitis
5.0% 4.9%
Psychiatric
5.0% 3.8%
Gastrointestinal System 19.8% 28.7%
Abdominal Pain 2.7% 6.0%
Diarrhea 5.0% 5.9%
Dyspepsia 2.2% 3.2%
Gum Hyperplasia 3.8% 6.0%
Nausea 5.5% 5.9%
White cell and RES 4.4% 2.7%
*Total percentage of events within the system
**Newly occurring hypertension = SBP≥160 mm Hg and/or DBP≥90 mm Hg
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
The following events occurred in 1% to less than 3% of psoriasis patients treated with cyclosporine:
Body as a Whole: fever, flushes, hot flushes;
Cardiovascular: chest pain;
Central and Peripheral Nervous System: appetite increased, insomnia, dizziness, nervousness,
vertigo;
Gastrointestinal: abdominal distention, constipation, gingival bleeding;
Liver and Biliary System: hyperbilirubinemia;
Neoplasms: skin malignancies [squamous cell (0.9%) and basal cell (0.4%) carcinomas];
Reticuloendothelial: platelet, bleeding, and clotting disorders, red blood cell disorder;
Respiratory: infection, viral and other infection;
Skin and Appendages: acne, folliculitis, keratosis, pruritus, rash, dry skin;
Urinary System: micturition frequency;
Vision: abnormal vision.
Mild hypomagnesemia and hyperkalemia may occur but are asymptomatic. Increases in uric acid may
occur and attacks of gout have been rarely reported. A minor and dose related hyperbilirubinemia has
been observed in the absence of hepatocellular damage. Cyclosporine therapy may be associated with a
modest increase of serum triglycerides or cholesterol. Elevations of triglycerides (>750 mg/dL) occur in
about 15% of psoriasis patients; elevations of cholesterol (>300 mg/dL) are observed in less than 3% of
psoriasis patients. Generally these laboratory abnormalities are reversible upon dose reduction or
discontinuation of cyclosporine.
OVERDOSAGE
There is a minimal experience with cyclosporine overdosage. Forced emesis and gastric lavage can be of
value up to 2 hours after administration of Neoral
®
. Transient hepatotoxicity and nephrotoxicity may
occur which should resolve following drug withdrawal. Oral doses of cyclosporine up to 10 g (about 150
mg/kg) have been tolerated with relatively minor clinical consequences, such as vomiting, drowsiness,
headache, tachycardia and, in a few patients, moderately severe, reversible impairment of renal function.
However, serious symptoms of intoxication have been reported following accidental parenteral
overdosage with cyclosporine in premature neonates. General supportive measures and symptomatic
treatment should be followed in all cases of overdosage. Cyclosporine is not dialyzable to any great
extent, nor is it cleared well by charcoal hemoperfusion. The oral dosage at which half of experimental
animals are estimated to die is 31 times, 39 times, and >54 times the human maintenance dose for
transplant patients (6mg/kg; corrections based on body surface area) in mice, rats, and rabbits.
DOSAGE AND ADMINISTRATION
Neoral
®
Soft Gelatin Capsules (cyclosporine capsules, USP) MODIFIED and Neoral
®
Oral Solution
(cyclosporine oral solution, USP) MODIFIED
Neoral
®
has increased bioavailability in comparison to Sandimmune
®
. Neoral
®
and Sandimmune
®
are not bioequivalent and cannot be used interchangeably without physician supervision.
The daily dose of Neoral
®
should always be given in two divided doses (BID). It is recommended that
Neoral
®
be administered on a consistent schedule with regard to time of day and relation to meals.
Grapefruit and grapefruit juice affect metabolism, increasing blood concentration of cyclosporine, thus
should be avoided.
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
Newly Transplanted Patients
The initial oral dose of Neoral
®
can be given 4-12 hours prior to transplantation or be given
postoperatively. The initial dose of Neoral
®
varies depending on the transplanted organ and the other
immunosuppressive agents included in the immunosuppressive protocol. In newly transplanted patients,
the initial oral dose of Neoral
®
is the same as the initial oral dose of Sandimmune
®
. Suggested initial
doses are available from the results of a 1994 survey of the use of Sandimmune
®
in US transplant centers.
The mean ± SD initial doses were 9±3 mg/kg/day for renal transplant patients (75 centers), 8±4
mg/kg/day for liver transplant patients (30 centers), and 7±3 mg/kg/day for heart transplant patients
(24 centers). Total daily doses were divided into two equal daily doses. The Neoral
®
dose is subsequently
adjusted to achieve a pre-defined cyclosporine blood concentration. (See Blood Concentration Monitoring
in Transplant Patients, below) If cyclosporine trough blood concentrations are used, the target range is the
same for Neoral
®
as for Sandimmune
®
. Using the same trough concentration target range for Neoral
®
as
for Sandimmune
®
results in greater cyclosporine exposure when Neoral
®
is administered. (See
Pharmacokinetics, Absorption) Dosing should be titrated based on clinical assessments of rejection and
tolerability. Lower Neoral
®
doses may be sufficient as maintenance therapy.
Adjunct therapy with adrenal corticosteroids is recommended initially. Different tapering dosage
schedules of prednisone appear to achieve similar results. A representative dosage schedule based on the
patient’s weight started with 2.0 mg/kg/day for the first 4 days tapered to 1.0 mg/kg/day by 1 week, 0.6
mg/kg/day by 2 weeks, 0.3 mg/kg/day by 1 month, and 0.15 mg/kg/day by 2 months and thereafter as a
maintenance dose. Steroid doses may be further tapered on an individualized basis depending on status of
patient and function of graft. Adjustments in dosage of prednisone must be made according to the clinical
situation.
Conversion from Sandimmune
®
to Neoral
®
in Transplant Patients
In transplanted patients who are considered for conversion to Neoral
®
from Sandimmune
®
, Neoral
®
should be started with the same daily dose as was previously used with Sandimmune
®
(1:1 dose
conversion). The Neoral
®
dose should subsequently be adjusted to attain the pre-conversion cyclosporine
blood trough concentration. Using the same trough concentration target range for Neoral
®
as for
Sandimmune
®
results in greater cyclosporine exposure when Neoral
®
is administered. (See
Pharmacokinetics, Absorption) Patients with suspected poor absorption of Sandimmune
®
require different
dosing strategies. (See Transplant Patients with Poor Absorption of Sandimmune
®
, below) In some
patients, the increase in blood trough concentration is more pronounced and may be of clinical
significance.
Until the blood trough concentration attains the pre-conversion value, it is strongly recommended
that the cyclosporine blood trough concentration be monitored every 4 to 7 days after conversion to
Neoral
®
. In addition, clinical safety parameters such as serum creatinine and blood pressure should be
monitored every two weeks during the first two months after conversion. If the blood trough
concentrations are outside the desired range and/or if the clinical safety parameters worsen, the dosage of
Neoral
®
must be adjusted accordingly.
Transplant Patients with Poor Absorption of Sandimmune
®
Patients with lower than expected cyclosporine blood trough concentrations in relation to the oral dose of
Sandimmune
®
may have poor or inconsistent absorption of cyclosporine from Sandimmune
®
. After
conversion to Neoral
®
, patients tend to have higher cyclosporine concentrations. Due to the increase in
bioavailability of cyclosporine following conversion to Neoral
®
, the cyclosporine blood trough
concentration may exceed the target range. Particular caution should be exercised when converting
patients to Neoral
®
at doses greater than 10 mg/kg/day. The dose of Neoral
®
should be titrated
individually based on cyclosporine trough concentrations, tolerability, and clinical response. In this
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
population the cyclosporine blood trough concentration should be measured more frequently, at least
twice a week (daily, if initial dose exceeds 10 mg/kg/day) until the concentration stabilizes within the
desired range.
Rheumatoid Arthritis
The initial dose of Neoral
®
is 2.5 mg/kg/day, taken twice daily as a divided (BID) oral dose. Salicylates,
nonsteroidal anti-inflammatory agents, and oral corticosteroids may be continued. (See WARNINGS and
PRECAUTIONS, Drug Interactions) Onset of action generally occurs between 4 and 8 weeks. If
insufficient clinical benefit is seen and tolerability is good (including serum creatinine less than 30%
above baseline), the dose may be increased by 0.5-0.75 mg/kg/day after 8 weeks and again after 12 weeks
to a maximum of 4 mg/kg/day. If no benefit is seen by 16 weeks of therapy, Neoral
®
therapy should be
discontinued.
Dose decreases by 25%-50% should be made at any time to control adverse events, e.g., hypertension
elevations in serum creatinine (30% above patient’s pretreatment level) or clinically significant laboratory
abnormalities. (See WARNINGS and PRECAUTIONS)
If dose reduction is not effective in controlling abnormalities or if the adverse event or abnormality is
severe, Neoral
®
should be discontinued. The same initial dose and dosage range should be used if Neoral
®
is combined with the recommended dose of methotrexate. Most patients can be treated with Neoral
®
doses of 3 mg/kg/day or below when combined with methotrexate doses of up to 15 mg/week. (See
CLINICAL PHARMACOLOGY, Clinical Trials)
There is limited long-term treatment data. Recurrence of rheumatoid arthritis disease activity is generally
apparent within 4 weeks after stopping cyclosporine.
Psoriasis
The initial dose of Neoral
®
should be 2.5 mg/kg/day. Neoral
®
should be taken twice daily, as a divided
(1.25 mg/kg BID) oral dose. Patients should be kept at that dose for at least 4 weeks, barring adverse
events. If significant clinical improvement has not occurred in patients by that time, the patient’s dosage
should be increased at 2-week intervals. Based on patient response, dose increases of approximately 0.5
mg/kg/day should be made to a maximum of 4.0 mg/kg/day.
Dose decreases by 25%-50% should be made at any time to control adverse events, e.g., hypertension,
elevations in serum creatinine (≥25% above the patient’s pretreatment level), or clinically significant
laboratory abnormalities. If dose reduction is not effective in controlling abnormalities, or if the adverse
event or abnormality is severe, Neoral
®
should be discontinued. (See Special Monitoring of Psoriasis
Patients)
Patients generally show some improvement in the clinical manifestations of psoriasis in 2 weeks.
Satisfactory control and stabilization of the disease may take 12-16 weeks to achieve. Results of a dose-
titration clinical trial with Neoral
®
indicate that an improvement of psoriasis by 75% or more (based on
PASI) was achieved in 51% of the patients after 8 weeks and in 79% of the patients after 16 weeks.
Treatment should be discontinued if satisfactory response cannot be achieved after 6 weeks at 4
mg/kg/day or the patient’s maximum tolerated dose. Once a patient is adequately controlled and appears
stable the dose of Neoral
®
should be lowered, and the patient treated with the lowest dose that maintains
an adequate response (this should not necessarily be total clearing of the patient). In clinical trials,
cyclosporine doses at the lower end of the recommended dosage range were effective in maintaining a
satisfactory response in 60% of the patients. Doses below 2.5 mg/kg/day may also be equally effective.
Upon stopping treatment with cyclosporine, relapse will occur in approximately 6 weeks (50% of the
patients) to 16 weeks (75% of the patients). In the majority of patients rebound does not occur after
cessation of treatment with cyclosporine. Thirteen cases of transformation of chronic plaque psoriasis to
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
more severe forms of psoriasis have been reported. There were 9 cases of pustular and 4 cases of
erythrodermic psoriasis. Long term experience with Neoral
®
in psoriasis patients is limited and
continuous treatment for extended periods greater than one year is not recommended. Alternation with
other forms of treatment should be considered in the long term management of patients with this life long
disease.
Neoral
®
Oral Solution (cyclosporine oral solution, USP) MODIFIED–
Recommendations for Administration
To make Neoral
®
Oral Solution (cyclosporine oral solution, USP) MODIFIED more palatable, it should
be diluted with orange or apple juice that is at room temperature. Patients should avoid switching diluents
frequently. Grapefruit juice affects metabolism of cyclosporine and should be avoided. The combination
of Neoral
®
solution with milk can be unpalatable. The effect of milk on the bioavailability of cyclosporine
when adminstered as Neoral Oral Solution has not been evaluated.
Take the prescribed amount of Neoral
®
Oral Solution (cyclosporine oral solution, USP) MODIFIED from
the container using the dosing syringe supplied, after removal of the protective cover, and transfer the
solution to a glass of orange or apple juice. Stir well and drink at once. Do not allow diluted oral solution
to stand before drinking. Use a glass container (not plastic). Rinse the glass with more diluent to ensure
that the total dose is consumed. After use, dry the outside of the dosing syringe with a clean towel and
replace the protective cover. Do not rinse the dosing syringe with water or other cleaning agents. If the
syringe requires cleaning, it must be completely dry before resuming use.
Blood Concentration Monitoring in Transplant Patients
Transplant centers have found blood concentration monitoring of cyclosporine to be an essential
component of patient management. Of importance to blood concentration analysis are the type of assay
used, the transplanted organ, and other immunosuppressant agents being administered. While no fixed
relationship has been established, blood concentration monitoring may assist in the clinical evaluation of
rejection and toxicity, dose adjustments, and the assessment of compliance.
Various assays have been used to measure blood concentrations of cyclosporine. Older studies using a
nonspecific assay often cited concentrations that were roughly twice those of the specific assays.
Therefore, comparison between concentrations in the published literature and an individual patient
concentration using current assays must be made with detailed knowledge of the assay methods
employed. Current assay results are also not interchangeable and their use should be guided by their
approved labeling. A discussion of the different assay methods is contained in Annals of Clinical
Biochemistry 1994;31:420-446. While several assays and assay matrices are available, there is a
consensus that parent-compound-specific assays correlate best with clinical events. Of these, HPLC is the
standard reference, but the monoclonal antibody RIAs and the monoclonal antibody FPIA offer
sensitivity, reproducibility, and convenience. Most clinicians base their monitoring on trough
cyclosporine concentrations. Applied Pharmacokinetics, Principles of Therapeutic Drug Monitoring
(1992) contains a broad discussion of cyclosporine pharmacokinetics and drug monitoring techniques.
Blood concentration monitoring is not a replacement for renal function monitoring or tissue biopsies.
HOW SUPPLIED
Neoral
®
Soft Gelatin Capsules (cyclosporine capsules, USP) MODIFIED
25 mg
Oval, blue-gray imprinted in red, “Neoral” over “25 mg.”
Packages of 30 unit-dose blisters (NDC 0078-0246-15).
100 mg
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
Oblong, blue-gray imprinted in red, “NEORAL” over “100 mg.”
Packages of 30 unit-dose blisters (NDC 0078-0248-15).
Store and Dispense
In the original unit-dose container at controlled room temperature 68°-77°F (20°-25°C).
Neoral
®
Oral Solution (cyclosporine oral solution, USP) MODIFIED
A clear, yellow liquid supplied in 50 mL bottles containing 100 mg/mL (NDC 0078-0274-22).
Store and Dispense
In the original container at controlled room temperature 68°-77°F (20°-25°C). Do not store in the
refrigerator. Once opened, the contents must be used within two months. At temperatures below 68°F
(20°C) the solution may gel; light flocculation or the formation of a light sediment may also occur. There
is no impact on product performance or dosing using the syringe provided. Allow to warm to room
temperature 77°F (25°C) to reverse these changes.
Neoral
®
Soft Gelatin Capsules (cyclosporine capsules, USP) MODIFIED
Neoral
®
Oral Solution (cyclosporine oral solution, USP) MODIFIED
Distributed by:
Novartis Pharmaceuticals Corporation, East Hanover, New Jersey 07936
REV: October 2009 T2009-16
© Novartis
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
