
Bioanalytical Method Validation
05/24/18
Bioanalytical Method
Validation
Guidance for Industry
U.S. Department of Health and Human Services
Food and Drug Administration
Center for Drug Evaluation and Research (CDER)
Center for Veterinary Medicine (CVM)
May 2018
Biopharmaceutics

Bioanalytical Method Validation
05/24/18
Bioanalytical Method
Validation
Guidance for Industry
Additional copies are available from:
Office of Communications,
Division of Drug Information
Center for Drug Evaluation and Research
Food and Drug Administration
10001 New Hampshire Ave., Hillandale Bldg., 4
th
Floor
Silver Spring, MD 20993-0002
Phone: 855-543-3784 or 301-796-3400; Fax: 301-431-6353
Email: druginfo@fda.hhs.gov
http://www.fda.gov/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/default.htm
and/or
Policy and Regulations Staff, HFV-6
Center for Veterinary Medicine
Food and Drug Administration
7500 Standish Place, Rockville, MD 20855
http://www.fda.gov/AnimalVeterinary/GuidanceComplianceEnforcement/GuidanceforIndustry/default.htm
U.S. Department of Health and Human Services
Food and Drug Administration
Center for Drug Evaluation and Research (CDER)
Center for Veterinary Medicine (CVM)
May 2018
Biopharmaceutics
Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
TABLE OF CONTENTS
I.
INTRODUCTION ...................................................................................................... 1
II. BACKGROUND ........................................................................................................ 2
III. BIOANALYTICAL METHOD DEVELOPMENT AND VALIDATION ................. 4
A. Guiding Principles ........................................................................................................ 4
B. Bioanalytical Parameters of CCs and LBAs .................................................................... 5
1. Reference Standards and Critical Reagents ........................................................................ 5
2. Calibration Curve........................................................................................................... 6
3. Quality Control Samples.................................................................................................. 7
4. Selectivity and Specificity................................................................................................. 7
5. Sensitivity...................................................................................................................... 8
6. Accuracy, Precision, and Recovery ................................................................................... 8
7. Stability ........................................................................................................................ 9
8. Dilution Effects .............................................................................................................10
9. Partial and Cross Validations..........................................................................................11
C. Validated Methods: Expectations of In-Study Analysis and Reporting ............................12
IV. INCURRED SAMPLE REANALYSIS .................................................................... 14
V. ADDITIONAL ISSUES............................................................................................ 15
A. Endogenous Compounds ..............................................................................................15
B. Biomarkers .................................................................................................................15
C. Diagnostic Kits ............................................................................................................16
D. Bridging Data From Multiple Bioanalytical Technologies ...............................................17
E. Dried Blood Spots ........................................................................................................18
VI. DOCUMENTATION ............................................................................................... 18
A. Summary Information..................................................................................................
18
B. Documentation for Method Validation and Bioanalytical Reports ...................................19
VII. APPENDIX............................................................................................................... 20
Table 1. Recommendations and Acceptance Criteria for Bioanalytical Method Validation and
In-Study Conduct. ...............................................................................................................20
Table 2. Documentation and Reporting ................................................................................28
Table 3.
Example of an Overall Summary Table for a Method Validation Report* or a Clinical
Study Report.......................................................................................................................33
Table 4. Example of Summary Analytical Runs for a Bioanalytical Study Report....................36
VIII. GLOSSARY ............................................................................................................. 37

Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
1
Bioanalytical Method Validation
Guidance for Industry
1
This guidance represents the current thinking of the Food and Drug Administration (FDA or Agency) on
this topic. It does not establish any rights for any person and is not binding on FDA or the public. You
can use an alternative approach if it satisfies the requirements of the applicable statutes and regulations.
To discuss an alternative approach, contact the FDA office responsible for this guidance as listed on the
title page.
I. INTRODUCTION
This guidance helps sponsors of investigational new drug applications (INDs) or applicants of
new drug applications (NDAs), abbreviated new drug applications (ANDAs), biologic license
applications (BLAs), and supplements validate bioanalytical methods used in human clinical
pharmacology, bioavailability (BA), and bioequivalence (BE) studies that require
pharmacokinetic, toxicokinetic, or biomarker concentration evaluation.
2
This guidance can also
inform the development of bioanalytical methods used for nonclinical studies that require
toxicokinetic or biomarker concentration data. For studies related to the veterinary drug
approval process such as investigational new animal drug applications (INADs), new animal
drug applications (NADAs), and abbreviated new animal drug applications (ANADAs), this
guidance may apply to blood and urine BA, BE, and pharmacokinetic studies.
The information in this guidance applies to bioanalytical procedures such as chromatographic
assays (CCs) and ligand binding assays (LBAs) that quantitatively determine the levels of drugs,
their metabolites, therapeutic proteins, and biomarkers in biologica l matrices such as blood,
serum, plasma, urine, and tissue such as skin.
This final guidance incorporates public comments to the revised draft published in 2013 and
provides recommendations for the development, validation, and in-study use of bioanalytical
methods. The recommendations can be modified with justification, depending on the specific
type of bioanalytical method. This guidance reflects advances in science and technology related
to validating bioanalytical methods.
In general, FDA’s guidance documents do not establish legally enforceable responsibilities.
Instead, guidances describe the Agency’s current thinking on a topic and should be viewed only
1
This guidance has been prepared by the Office of Clinical Pharmacology in the Center for Drug Evaluation and
Research and the Center for Veterinary Medicine at the Food and Drug Administration.
2
This guidance applies to both sponsors and applicants. The use of the word sponsor applies to both sponsors and
applicants and hence, INDs, NDAs, BLAs, and ANDAs.

Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
2
as recommendations, unless specific regulatory or statutory requirements are cited. The use of
the word should in Agency guidances means that something is suggested or recommended, but
not required.
II. BACKGROUND
The 2001 guidance for industry on Bioanalytical Method Validation was originally based on the
deliberations of two workshops described in publications entitled:
• Analytical Methods Validation: Bioavailability, Bioequivalence, and Pharmacokinetic
Studies
3
• Bioanalytical Methods Validation: A Revisit With a Decade of Progress
4
Additional workshops, summarized in the following publications, have informed subsequent
revisions (e.g., the 2013 draft guidance for industry entitled Bioanalytical Method Validation
5
):
• Quantitative Bioanalytical Methods Validation and Implementation: Best Practices for
Chromatographic and Ligand Binding Assays
6
• The AAPS/FDA Workshop on Incurred Sample Reanalysis
7
• The AAPS Workshop on Crystal City V — Quantitative Bioanalytical Method Validation
and Implementation: 2013 Revised FDA Guidance
8
3
Shah, VP, KK Midha, S Dighe, IJ McGilveray, JP Skelly, A Yacobi, T Layloff, CT Viswanathan, CE Cook, RD
McDowell, KA Pittman, S Spector, 1992, Analytical Methods Validation: Bioavailability, Bioequivalence, and
Pharmacokinetic Studies, Pharm Res, 9:588-592.
4
Shah, VP, KK Midha, JW Findlay, HM Hill, JD Hulse, IJ McGilveray, G McKay, KJ Miller, RN Patnaik, ML
Powell, A Tonelli, CT Vis wanathan, A Yacobi, 2000, Bioanalytical Methods Validation: A Revisit With a Decade
of Progress, Pharm Res, 17:1551-1557.
5
When final, this guidance will represent the FDA’s current thinking on this topic. For the most recent version of a
guidance, check the FDA Drugs guidance Web page at
http://www.fda.gov/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/default.htm
6
Viswanathan, CT, B Surendra, B Booth, AJ DeStefano, MJ Rose, J Sailstad, VP Shah, JP Skelly, PG Swann, R
Weiner, 2007, Quantitative Bioanalytical Methods Validation and Implementation: Best Practices for
Chromatographic and Ligand Binding Assays, Pharm Res, 24:1962-1973.
7
Fast, DM, M Kelley, CT Viswanathan, J O’Shaughnessy, SP King, A Chaudhary, R Weiner, AJ DeStefano, D
Tang, 2009, Workshop Report and Follow-Up — AAPS Workshop on Current Topics in GLP Bioanalysis: Assay
Reproducibility for Incurred Samples — Implications of Crystal City Recommendations, AAPS J, 11:238-241.
8
Booth, B, ME Arnold, B DeSilva, L Amaravadi, S Dudal, E Fluhler, B Gorovits, SH Haidar, J Kadavil, S Lowes,
R Nicholson, M Rock, M Skelly, L Stevenson, S Subramaniam, R Weiner, E Woolf, 2015, Workshop Report:

Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
3
Validated analytical methods for the quantitative evaluation of analytes (i.e., drugs, including
biologic products, and their metabolites) and biomarkers in a given biological matrix (e.g. blood,
plasma, serum, or urine) are critical for the successful conduct of nonclinical, biopharmaceutics,
and clinical pharmacology studies. These validated methods provide critical data to support the
safety and effectiveness of drugs and biologic products. Validating the analytical method
ensures that the data are reliable by addressing certain key questions, including:
• Does the method measure the intended analyte? For example, does anything interfere
with the measurement, and is the method specific or selective for the analyte?
• What is the variability associated with these measurements? For example, what are the
accuracy and precision
of the method?
• What is the range in measurements that provide reliable data? For example, what is the
sensitivity of the method (e.g., what is the lower limit of quantitation (LLOQ) of the
method, and what is the upper limit of quantitation the method (ULOQ)?)
• How do sample collection, handling, and storage affect the reliability of the data from the
bioanalytical method? For example, what steps need to be followed while collecting
samples? Do the samples need to be frozen during shipping? What temperatures are
required to store the samples, and how long can the samples be stored?
When changes are made to a validated method, the sponsor should conduct additional validation
(i.e., partial or cross validation).
The fit-for-purpose (FFP) concept states that the level of validation should be appropriate for the
intended purpose of the study. The key questions listed above should be evaluated relative to the
stage of drug development. Pivotal studies submitted in an NDA, BLA, or ANDA that require
regulatory decision making for approval, safety or labeling, such as BE or pharmacokinetic
studies, should include bioanalytical methods that are fully validated. Exploratory methods that
would not be used to support regulatory decision making (e.g., candidate selection) may not
require such stringent validation. This FFP concept applies to drugs, their metabolites, and
biomarkers.
The analytical laboratory conducting toxicology studies for regulatory submissions should
adhere to 21 CFR 58, Good Laboratory Practices (GLPs).
9
The bioanalytical method for human
BA, BE, and pharmacokinetic studies must meet the criteria specified in 21 CFR 320
Bioequivalence and Bioavailability Requirements (i.e., 21 CFR 320.29).
Crys tal City V — Quantitative Bioanalytical Method Validation and Implementation: The 2013 Revised FDA
Guidance, AAPS J, 17:277-288.
9
For the Center for Veterinary Medicine, all BE studies are subject to Good Laboratory Practices.
Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
4
The following sections discuss the development, validation, and in-study use of bioanalytica l
methods and how best to document validation methods and results. Refer to the Glossary for the
definitions of assay parameters and analytical terms used in this guidance.
III. BIOANALYTICAL METHOD DEVELOPMENT AND VALIDATION
A. Guiding Principles
The purpose of bioanalytical method development is to define the design, operating conditions,
limitations, and suitability of the method for its intended purpose and to ensure that the method is
optimized for validation.
Before the development of a bioanalytical method, the sponsor should understand the analyte of
interest (e.g., determine the physicochemical properties of the drug, in vitro and in vivo
metabolism, and protein binding) and consider aspects of any prior analytical methods that may
be applicable.
The elements and acceptance criteria of method development and validation are summarized in
Table 1. Table 2 describes how the sponsor should document the development and validation of
the bioanalytical assay and where it should be stored or submitted.
Method development involves optimizing the procedures and conditions involved with extracting
and detecting the analyte. Method development includes the optimization of the following
bioanalytical parameters (which are discussed in greater detail in section III.B) to ensure that the
method is suitable for validation:
• Reference standards
• Critical reagents
• Calibration curve
• Quality control samples (QCs)
• Selectivity and specificity
• Sensitivity
• Accuracy
• Precision
• Recovery
• Stability of the analyte in the matrix
Bioanalytical method development does not require extensive record keeping or notation.
However, the sponsor should record the changes to procedures as well as any issues and their
resolutions during development of the bioanalytical method to provide a rationale for any
changes during the development of the method.
Bioanalytical method validation proves that the optimized method is suited to the analysis of the
study samples. The sponsor should:
Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
5
• Conduct a full validation of any new bioanalytical method for the analysis of a new drug
entity, its metabolite(s), or biomarkers.
• Conduct a full validation for any revisions to an existing validated method that adds a
metabolite or an additional analyte.
• Establish a detailed, written description (e.g., protocol, study plan, and/or standard
operating procedure (SOP)) for the bioanalytical method before initiating validation. The
description should identify procedures that control critical parameters in the method (e.g.,
environmental, matrix, procedural variables) from the time of collection of the samples to
the time of analysis to minimize their effects on the measurement of the analyte in the
matrix.
• Document and report (in the method validation report) all experiments used to make
claims or draw conclusions about the validity of the method.
• Validate the measurement of each analyte in the biological matrix. The specific
recommendations and acceptance criteria for each bioanalytical parameter are listed in
Table 1.
B. Bioanalytical Parameters of CCs and LBAs
The bioanalytical parameters applicable to CCs and LBAs are discussed below. Issues unique to
either CCs or LBAs are specifically identified.
1. Reference Standards and Critical Reagents
The sponsor should appropriately characterize and document (e.g. determine the identity, purity,
and stability) all reference standards and critical reagents, such as antibodies, labeled analytes,
and matrices and store them under defined conditions.
a. Reference standards
The purity of reference standards used to prepare calibrators and QCs can affect the study data.
Therefore, the sponsor should use authenticated analytical reference standards with known
identities and purities to prepare solutions of known concentrations. The reference standard
should be identical to the analyte; however, when this scenario is not possible, the sponsor can
use an established chemical form (e.g., free base, free acid, or salt) of known purity.
The sponsor should provide the certificates of analyses (CoA), including the source, lot number,
and expiration date (with the exception of United States Pharmacopeia (USP) standards) for
commercially available reference standards. For internally or externally generated reference
standards that do not have a CoA, the sponsor should provide evidence of the standard’s identity
and purity in addition to the source and the lot number. When using expired reference standards,
the sponsor should provide an updated CoA or re-establish the identity and purity of the
standard. If the reference standard expires, the sponsor should not make stock solutions with this
Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
6
lot of standard unless the standard’s purity is re-established. For internal standards (ISs), the
sponsor does not have to provide a CoA or evidence of purity if it demonstrates that the IS is
suitable for the specific use (e.g., lack of interference with an analyte).
b. Critical reagents
The sponsor should appropriately characterize and document (i.e., determine the identity, purity
and stability) the critical reagents, including – but not limited to – any reference standards,
antibodies, labeled analytes, and matrices.
Assay validation is important when there are changes to the critical reagents, such as lot-to-lot
changes or switches to another reagent. For example, if there are changes to the labeled analytes,
detector reagents, or antibodies, the sponsor should:
• Evaluate binding and re-optimize assays
• Verify performance with a standard curve and QCs
• Evaluate cross-reactivities
2. Calibration Curve
During method development, the sponsor should choose the quantitation range of the assay and
the concentrations of the calibration standards on the basis of the concentration range expected in
a particular study. For LBAs, in addition to the calibration standards, anchor points outside the
range of quantification can facilitate the fitting of the curve.
Anchor points should not be used as
part of the acceptance criteria for the run. For most LBAs, calibration (standard) curves are
inherently nonlinear, and in general, more calibration standards are needed to define the fit over
the calibration curve range for LBAs than for CCs. In addition, the response-error relationship
for LBA standard curves is a variable function of the mean response (i.e., heteroscadisticity).
The sponsor should use the simplest model that adequately describes the concentration-response
relationship, as well as an appropriate weighting scheme and regression equation. For LBAs, the
concentration-response relationship is most often fitted to a four- or five-parameter logistic
model, although other models can be assessed.
When the method is validated, the calibration curve should be continuous and reproducible. The
sponsor should prepare the calibration standards in the same biological matrix as the samples in
the intended study. Study samples may contain more than one analyte. The sponsor should
generate a calibration curve for each analyte in the sample. When surrogate matrices are
necessary, the sponsor should justify and validate the calibration curves.
The requirements for the calibration curve, including the LLOQ, ULOQ, as well as the
acceptance criteria are listed in Table 1.
Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
7
3. Quality Control Samples
Quality controls are used to assess the precision and accuracy of an assay and the stability of the
samples. Sponsors should prepare QCs in the same matrix as the study samples to be assayed
with the validated method. Freshly prepared QCs are recommended for precision and accuracy
analyses during method development, as stability data are generally not available at this time.
During method validation, QCs evaluate the performance of a method and the stability of an
analyte. Performance QCs are included in validation runs to determine the precision and
accuracy of the method (see section III.B). Stability QCs evaluate the stability of an analyte
under various stress conditions (Refer to section III.B for the selection of QC concentrations).
The sponsor should prepare any calibration standards and QCs from separate stock solutions.
However, if the sponsor can demonstrate the precision and accuracy in one validation run using
calibrators and QCs prepared from separate stock solutions, then the sponsor can use calibrators
and QCs prepared from the same stock solution in subsequent runs. The sponsor should make up
calibrators and QCs in lots of blank matrix that is free of interference or matrix effects.
4. Selectivity and Specificity
During method development, the sponsor should verify that the substance being measured is the
intended analyte to minimize or avoid interference. Selectivity of the method is routinely
demonstrated by analyzing blank samples of the appropriate biological matrix (e.g., plasma)
from multiple sources. Depending on the intended use of the assay, the impact of hemolyzed
samples, lipemic samples, or samples from special populations can be included in the selectivity
assessment. When using liquid chromatography/mass spectrometry (LC/MS) methods, the
sponsor or applicant should determine the effects of the matrix on ion suppression, ion
enhancement, or extraction efficiency.
Internal standards should be assessed to avoid
interference with the analyte. Potential interfering substances in a biological matrix include
endogenous matrix components such as metabolites, decomposition products – and from the
actual study – concomitant medications and other xenobiotics.
If a stabilizer or enzyme inhibitor
is used during sample collection, the sponsor should evaluate the potential for interference on the
quantitation of the analyte. Sponsors should make a scientific judgment about the need to assess
these (and any other) potential interferences during method development.
During validation, the sponsor should confirm that the assay is free of potential interfering
substances including endogenous matrix components, metabolites, anticipated concomitant
medications, etc. If the study sample contains more than one analyte and the analytes are
intended to be quantified by different methods, the sponsor should test each method for
interference from the other analyte.
The sponsor should analyze blank samples of the appropriate biological matrix (e.g. plasma)
from at least six (for CCs) or ten (for LBAs) individual sources. The sponsor should ensure that
there are no matrix effects throughout the application of the method. Refer to Table 1 for
details of selectivity and specificity requirements and acceptance criteria.
Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
8
For LBAs, it is important to investigate any interference originating from structurally or
physiologically similar analytes (i.e., exogenous interference) or matrix effects (i.e., endogenous
interference). Investigating exogenous interference involves determining the cross-reactivity of
molecules that could potentially interfere with the binding interaction, including molecules
structurally related to the drug, any metabolites, concomitant medications (and their significant
metabolites), or endogenous matrix components. The sponsor should evaluate each factor
individually and in combination with the analyte of interest to determine its ability to cause
interference. Matrix effects evaluation involves comparing calibration curves in multiple sources
of the biological matrix against a calibration curve in the matrix for parallelism (serial dilution of
incurred samples) and nonspecific binding. The sponsor should eliminate or minimize any
significant interference. If such attempts are unsuccessful, the sponsor could consider the
development of an orthogonal method to eliminate or minimize the interference.
Carryover between samples can occur in analytical methods. The sponsor should eliminate any
carryover during method development. If carryover cannot be eliminated, the sponsor should
assess the impact of any carryover during method validation on the accuracy of the study sample
concentrations.
5. Sensitivity
The LLOQ defines the method sensitivity and should be determined during method
development. The method should be developed and validated such that it will be able to meet
the requirements necessary for the intended study samples. The LLOQ evaluation can be done
separately or as part of the precision and accuracy assessment for the calibration range. The
specific recommendations to validate sensitivity are listed in Table 1.
6. Accuracy, Precision, and Recovery
Evaluating the accuracy and precision across the quantitation range during method development
is essential to determine whether the method is ready for validation and involves analyzing
replicate QCs at multiple concentrations across the assay range. Specifically, the sponsor should
evaluate the performance at the LLOQ, low, mid and high QCs (and the ULOQ for LBAs) to
determine if the method is suitable to analyze study samples.
Method validation experiments for estimating accuracy and precision should include a minimum
of three (for CCs) and six (for LBAs) independent runs (i.e., accuracy and precision (A & P)
runs; see Table 1) conducted over several days. Each A & P run should include a calibration
curve and multiple QC concentrations that are analyzed in replicates. The sponsor should
determine the accuracy and precision of the method based on the performance of the QC in the A
& P runs. The specific validation requirements for accuracy and precision and A & P runs are
listed in Table 1. The sponsor should use freshly prepared calibrators and QCs in all A & P runs.
Use of freshly prepared QCs in all A & P runs is preferred; however, if this is not possible, the
sponsor should use freshly prepared QCs in one or more A & P runs.
The sponsor should optimize the recovery of the analyte to ensure that the extraction is efficient
and reproducible. Recovery need not be 100 percent, but the extent of the recovery of an analyte
Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
9
and of the ISs should be consistent and reproducible. The sponsor should perform recovery
experiments by comparing the analytical results of extracted samples with corresponding extracts
of blanks spiked with the analyte post-extraction (i.e., to represent 100 percent recovery).
Recovery evaluation is not necessary for LBAs unless sample extraction is involved. Recovery
experiments should be performed as described in Table 1.
7. Stability
During method development, the sponsor should determine the chemical stability of the analyte
in a given matrix, including the effects of sample collection, handling, and storage of the analyte.
The sponsor should assess autosampler, benchtop, processed or extracted samples, freeze-thaw,
stock solution, and long-term stability of the analyte. The sponsor should assess the stability in
the same matrix as that intended for in-study samples; however, when the matrix is rare, the
sponsor can explore the use of suitable surrogate matrices.
For drugs administered as fixed combinations, or part of a specific drug regimen, the stability of
the analyte should be assessed in the presence of the other drug. The sponsor should also
consider the stability of the analyte in the presence of other co-medications that are known to be
regularly administered to patients for the indication of the drug under development.
Depending on the analyte as well as the sample collection and assay conditions, evaluating the
stability of the analyte in whole blood during method development can be useful. For example, a
drug can be unstable in whole blood or adsorb to cellular components during collection.
During validation, stability evaluations should cover the expected sample conditions before
receipt at the analytical site (e.g., at the clinical site, during shipment,
and at all other secondary
sites) as well as during receipt and analysis at the analytical site. Validation of drug stability in a
biological fluid is a function of the storage conditions, the physicochemical properties of the
drug, the matrix, and the container system. The stability of an analyte in a particular matrix and
container system is relevant only to that matrix and container system and should not be
extrapolated to other matrices and container systems.
If the storage conditions changed or the sample analysis occurred outside of the validated storage
condition , the stability should be re-established under these new conditions. Stability testing of
the analyte in whole blood should be revalidated if necessary (e.g., if the analytes are unstable
during blood collection). The specific recommendations and acceptance criteria for stability are
listed in Table 1.
Matrix-related stability experiments should compare stability QCs against freshly prepared
calibration curves and freshly prepared QCs. Although the use of freshly prepared calibrators
and QCs is the preferred approach, in some cases, (e.g., for macromolecules), it may be
necessary to freeze them overnight. In such cases, the sponsor should provide valid justification
and demonstrate the freeze-thaw stability.
Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
10
All stability determinations (see list below) should use a set of samples prepared from a freshly
made stock solution of the analyte in the appropriate analyte-free, interference-free biological
matrix.
• Autosampler stability: The sponsor should demonstrate the stability of extracts in the
autosampler only if the autosampler storage conditions are different or not covered by
extract (processed sample) stability.
• Bench-top stability: The sponsor should determine the stability of samples under the
laboratory handling conditions that are expected for the study samples (e.g., the stability
of samples maintained at room temperature or stored in an ice bucket).
• Extract (or processed sample) stability: The sponsor should assess the stability of
processed samples, including the residence time in the autosampler against freshly
prepared calibrators.
• Freeze-thaw stability: The sponsor should assess the stability of the sample after a
minimum of three freeze-thaw cycles. QC samples should be thawed and analyzed
according to the same procedures as the study samples. QC samples should be frozen for
at least 12 hours between cycles. Freeze-thaw stability QCs should be compared to
freshly prepared calibration curves and QCs.
• Long-term stability: The sponsor should determine the long-term stability of the sample
over a period of time equal to or exceeding the time between the date of first sample
collection and the date of last sample analysis. The storage temperatures studied should
be the same as those used to store study samples. Long-term stability QCs should be
compared to freshly prepared calibration curves and QCs. Determination of stability at
minus 20ºC would cover stability at colder temperatures.
• Stock solution stability: Stock solutions should not be made from reference materials
that are about to expire unless the purity of the analyte in the stock solutions is re-
established. When the stock solution exists in a different state (e.g., solution versus solid)
or in a different buffer composition (which is generally the case for macromolecules)
from the certified reference standard, the sponsor should generate stability data on stock
solutions to justify the duration of stock solution storage stability.
8. Dilution Effects
If the method measures diluted samples, the integrity of the dilution should be monitored during
validation by diluting QC samples above the ULOQ with like matrix to bring to within
quantitation range, and the accuracy and precision of these diluted QCs should be demonstrated.
Dilutions used during the validation should mimic the expected dilutions in the study. The
prozone effect should be demonstrated in LBAs. Refer to the specific recommendations and
acceptance criteria in Table 1.
Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
11
9. Partial and Cross Validations
The following section defines other types of methods validation.
a. Partial validation
Partial validations evaluate modifications of already validated bioanalytical methods. Partial
validation can range from as little as one intra-assay accuracy and precision determination to a
nearly full validation. Raw data on partial validations should be retained at the analytical site for
inspection when requested. Typical bioanalytical method modifications or changes that fall into
this category include, but are not limited to, the following:
• Bioanalytical method transfers between laboratories
• Changes in analytical methodology (e.g., a change in detection systems)
• Changes in sample processing procedures
• Changes in sample volume (e.g., the smaller volume of pediatric samples)
• Changes in instruments and/or software platforms
• Extensions of the assay range
• Changes in the anticoagulant (but not changes in the counter-ion) in harvesting biological
fluids (e.g., heparin to EDTA)
• Changes in the matrix within species (e.g., switching from human plasma to human
blood) or changes to the species within the matrix (e.g., switching from rat plasma to
mouse plasma)
• Changes to the matrices (e.g., cerebrospinal fluid)
• Demonstrating the selectivity of an analyte in the presence of concomitant medications
• Changes in LBA critical reagents (e.g., lot-to-lot changes, changes in reagents)
b. Cross validation
Cross validation is a comparison of validation parameters of two or more bioanalytical methods
or techniques that are used to generate data within the same study or across different studies.
Also, cross validation is necessary when sample analyses within a single study are conducted at
more than one site or more than one laboratory. In such cases, cross validation with shared
matrix QCs and nonpooled subject samples should be conducted at each site or laboratory to
establish interlaboratory reliability. Pooled incurred samples can be used when insufficient
volume exists. An SOP or validation plan should define the criteria a priori.
Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
12
C. Validated Methods: Expectations of In-Study Analysis and Reporting
This section describes the expectations for the use of a validated bioanalytical method for routine
drug analysis. The specific recommendations and acceptance criteria are listed in Table 1.
• If system suitability is assessed, a specific SOP should be used. System suitability,
including apparatus conditioning and instrument performance, should be determined
using samples that are independent of the current study calibrators, QCs, and study
samples. Records of system suitability should be maintained and available for audits.
• Calibration curves and QCs should be included in all analytical runs (see Table 1 for
details). The QCs should cover the expected study sample concentration range.
• Typically, the same curve fitting, weighting, and goodness-of-fit determined during
validation should be used for the calibration curve within the study. Changes in the
response-function relationship between the validation and study sample analyses indicate
potential problems. A SOP should be developed a priori to address such issues.
• Total QCs should number at least five percent of the total samples analyzed, or be at least
six in number (low-, mid-, and high-QCs, in duplicate), whichever is greater (see Table 1
for details). Duplicate low-, mid-, and high-QCs should be used on all distinct processing
batches within a run.
• If the study sample concentrations are clustered in a narrow range of the standard curve,
additional QCs should be added to cover the sample range. If the additional QC
concentrations are not bracketed by QCs validated before the study, the accuracy and
precision of the additional QCs should be demonstrated before continuing with the
analysis. If the partial validation is acceptable, samples that have already been analyzed
do not require re-analysis.
• The QCs should be interspersed with study samples during processing and analysis.
• In each analytical run, the lack of analyte interference at the LLOQ should be confirmed
(see Table 1 for Selectivity and Sensitivity).
• The analytical run fails if the calibration and/or QC acceptance criteria are not met (see
Table 1).
• QC results (including outliers) from analytical runs that meet the acceptance criteria
should be included in the estimation of accuracy and precision during the study’s sample
analysis. The QC results from all analytical runs (passed and failed) should be reported,
but QCs results from failed runs need not be included as part of the estimation of
accuracy and precision.
Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
13
• If the bioanalytical method necessitates separation of the overall analytical run into
distinct processing batches (e.g., groups of samples processed at distinctly different times
or by different analysts), each distinct batch should process duplicate QCs at all levels
(e.g., low, middle, high) along with the study samples. Examples might include when the
number of samples exceeds the capacity of a 96-well plate or when a solid phase
extraction manifold cannot accommodate all samples. See Table 1 for what constitutes
an acceptable run based on QC acceptance criteria. A distinct batch or batches in an
analytical run may be rejected when it fails to meet QC acceptance criteria, but the
remaining batches may pass provided that the analytical run meets the overall QC
acceptance criteria.
• Study samples with concentrations listed below the LLOQ should be reported as below
the LLOQ (BQL). Study samples with concentrations above the ULOQ should be diluted
and re-analyzed, or the standard curve should be extended and revalidated.
• Study sample dilutions should use the same matrix (e.g., human plasma to human
plasma).
• Assays of all study samples of an analyte in a biological matrix should be completed
within the time period for which stability data are available. If sample handling
conditions are changed or exceed validated stability data, then the stability of the sample
should be established at the new conditions.
• For CCs, the IS response should be monitored for variability. An SOP should be
developed a priori to address issues with IS variability.
• Drift should be monitored and its impact on the accuracy of the estimated unknown
sample concentrations, if any, should be addressed (e.g., the impact of drift on the
accuracy of interspersed QCs).
• All study samples from a subject should be analyzed in a single run, especially for studies
designed with repeated measures from individual subjects (e.g., crossover or sequential
design required for BE studies). If other approaches are taken, the sponsor or applicant
should justify the approach and take steps to minimize the variability between periods.
• Carryover, if any, should be monitored, and its impact on the quantitation of study
samples should be addressed.
• Incurred sample reanalysis (ISR) should be performed (See section IV, Table 1 and Table
2).
• An SOP or guideline describing the reasons for a repeat analysis should be established a
priori. Repeat analysis is acceptable only for assignable causes (e.g., the samples are
above the ULOQ, there are sample processing errors, there is an equipment failure, the
chromatography is poor). The SOP should include the acceptance criteria for re-analysis,
and the sponsor or applicant should report final values. The specific recommendations

Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
14
are described in Table 1 and Table 2. The rationale, approach, and all data for the repeat
analysis and reporting should be clearly documented.
• For study samples involving multiple analytes, a valid result for one analyte should not
be rejected because of another analyte failing the acceptance criteria.
• If a unique or disproportionately high concentration of a metabolite is discovered in
human studies, a fully validated assay may need to be developed for the metabolite,
depending upon its activity (refer to the FDA guidance for industry entitled Safety
Testing of Drug Metabolites
10
).
• An SOP or guideline for sample data reintegration for CCs should be established a priori.
This SOP or guideline should define the criteria for re-integration and how the re-
integration will be performed. The rationale for the re-integration should be clearly
described and documented. Audit trails should be maintained. Original and re-integrated
data should be documented and reported.
IV. INCURRED SAMPLE REANALYSIS
ISR is a necessary component of bioanalytical method validation and verifies the reliability of
the reported study sample analyte concentrations. ISR is conducted by repeating the analysis of
a subset of subject or patient samples from a given study in separate runs, preferably during the
study, to critically support the precision and accuracy measurements established with the QCs.
The original and repeat analyses should be conducted using the same bioanalytical method
procedures. If a bulk frozen calibration curve was used for the original analysis, then it is
acceptable to use a frozen curve for the ISR evaluation. The calibration curve, QCs, and study
samples for the ISR evaluation should be extracted or processed separately from those used in
the original runs. Incurred samples should not be pooled. ISR should be conducted in all studies
submitted in an NDA, BLA, or ANDA that provide pivotal data for the approval or labeling of
the product, regardless of the matrix. For instance, ISR is expected for all in vivo human BE
studies in ANDAs, or all pivotal pharmacokinetic, pharmacodynamic, and biomarker studies in
NDAs or BLAs. For nonclinical safety studies, the performing laboratory should conduct ISR at
least once for each method and species. Table 1 lists the sample requirements and acceptance
criteria for ISR. Written SOPs should be established for the conduct of ISR and to guide an
investigation in the event of ISR failure to resolve the lack of reproducibility. All aspects of ISR
evaluations should be documented to allow reconstruction of the study, as well as guide any
investigations (see Table 2).
The percentage difference of the results between the original study and the repeat study is
determined with the following equation:
10
This guidance is available on the Internet at http://www.fda.gov/Drugs/default.htm under Guidances (Drugs).

Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
15
(Repeat – Original) * 100
Mean
V. ADDITIONAL ISSUES
A. Endogenous Compounds
For analytes that are also endogenous compounds, the accuracy of the measurement of the
analytes poses a challenge when the assay cannot distinguish between the therapeutic agent and
the endogenous counterpart. In such situations, the following approaches are recommended to
validate and monitor assay performance. Other approaches, if justified by scientific principles,
can also be considered.
• The biological matrix used to prepare calibration standards should be the same as the
study samples and free of the endogenous analyte. To address the suitability of using an
analyte-free biological matrix, the matrix should be demonstrated to have: (1) no
measurable endogenous analyte; and (2) no matrix effect or interference when compared
to the biological matrix. The use of alternate matrices (e.g., buffers, dialyzed serum) for
the preparation of calibration standards should be justified. The QCs should be prepared
by spiking known quantities of the analyte in the same biological matrix as the study
samples. The endogenous concentrations of the analyte in the biological matrix should
be evaluated before QC preparation (e.g., by replicate analysis). The concentrations for
the QCs should account for the endogenous concentrations in the biological matrix (i.e.,
additive) and be representative of the expected study concentrations.
• Parallelism should be evaluated for assays for endogenous compounds.
B. Biomarkers
The recommendations in this guidance only pertain to the validation of assays to measure in vivo
biomarker concentrations in biological matrices such as blood or urine. Considerable effort also
goes into defining the biological function of biomarkers, and confusion may arise regarding
terminology (e.g. biomarker method validation vs biomarker qualification).
Biomarkers are increasingly used to assess the effects of new drugs and therapeutic biological
products in patient populations. Because of the important roles biomarkers can play in
evaluating the safety, activity, or effectiveness of a new medical product, it is critical to ensure
the integrity of the data generated by assays used to measure them. Biomarkers can be used for a
wide variety of purposes during drug development; therefore, a FFP approach should be used
when determining the appropriate extent of method validation. When biomarker data will be
used to support a regulatory decision making, such as the pivotal determination of safety and/or
effectiveness or to support dosing instructions in product labeling, the assay should be fully
validated.

Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
16
For assays intended to support early drug development (e.g., candidate selection, go-no-go
decisions, proof-of-concept), the sponsor should incorporate the extent of method validation they
deem appropriate.
Method validation for biomarker assays should address the same questions as method validation
for drug assays. The accuracy, precision, sensitivity, selectivity, parallelism, range,
reproducibility, and stability of a biomarker assay are important characteristics that define the
method. The approach used for drug assays should be the starting point for validation of
biomarker assays, although the FDA realizes that some characteristics may not apply or that
different considerations may need to be addressed.
C. Diagnostic Kits
Diagnostic kits are sometimes co-developed with new drug or therapeutic biological products as
analytical methods that are used during the development of new drugs and therapeutic biologics.
The recommendations in this section of the guidance do not apply to commercial diagnostic kits
intended for point-of-care patient diagnosis (e.g., companion diagnostic kits), which are
addressed in the following CDRH guidance documents:
• Principles for Codevelopment of an In Vitro Companion Diagnostic Device with a
Therapeutic Product
11
• In Vitro Companion Diagnostic Devices
However, when commercial diagnostic kits are repurposed as analytical methods to measure the
concentrations of drugs, therapeutic biologics, or biomarkers in development, the FDA has the
following recommendations:
• LBA kits with various detection platforms are sometimes used to determine analyte
concentrations in pharmacokinetic or pharmacodynamic studies when the reported results
must exhibit sufficient precision and accuracy. Because such kits are generally
developed for use as clinical diagnostic tools, their suitability for use in such studies
should be demonstrated.
• Diagnostic kit validation data provided by the manufacturer may not ensure that the kit
method is reliable for drug development purposes. In such situations, the performance of
diagnostic kits should be assessed in the facility conducting the sample analysis.
Validation considerations for kit assays include, but are not limited to, the following
examples:
11
When final, this guidance will represent the FDA’s current thinking on this topic. For the most recent version of a
guidance, check the FDA Drugs guidance Web page at
http://www.fda.gov/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/default.htm
Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
17
- Site-specific validation should be performed. The specificity, accuracy,
precision, and stability of the assay should be demonstrated under actual
conditions of use. Modifications from kit processing instructions should be
completely validated.
- Kits that use sparse calibration standards (e.g., one- or two-point calibration
curves) should include in-house validation experiments to establish the
calibration curve with a sufficient number of standards across the calibration
range as specified in Table 1.
- Actual QC concentrations should be known. Concentrations of QCs
expressed as ranges are not sufficient for quantitative applications. In such
cases, QCs with known concentrations should be prepared and used,
independent of the kit-supplied QCs.
- Standards and QCs should be prepared in the same matrix as the subject
samples. Kits with standards and QCs prepared in a matrix different from the
subject samples should be justified, and appropriate cross-validation
experiments should be performed. Refer to section V.A of this guidance for
additional discussion.
- If the analyte source (i.e., reference standard) in the kit differs from that of the
subject samples (e.g., the sample is a protein isoform of the reference
standard), testing should evaluate differences in assay performance of the kit
reagents.
- If multiple kit lots are used within a study, lot-to-lot variability and
comparability should be addressed for any critical reagents.
- Individual batches using multiple assay plates (e.g., 96-well ELISA plates)
should include sufficient replicate QCs on each plate to monitor the accuracy
of the assay. Acceptance criteria should be established for the individual
plates and the overall analytical run (refer to Table 1 and section III.B).
D. Bridging Data From Multiple Bioanalytical Technologies
The FDA encourages the development and use of new bioanalytical technologies. However, the
use of two different bioanalytical technologies for the development of a drug may generate data
for the same product that could be difficult to interpret. This outcome can occur when one
platform generates drug concentrations that differ from another platform. Therefore, when a new
platform is used in the development of a drug, the data it produces should be bridged to that of
the other method. This is best accomplished by assessing the output of both methods with a set
of incurred samples (a minimum of 20 samples). In cases where one method produces data with
a constant bias relative to the other, concentrations can be mathematically transformed by that
factor to allow for appropriate study interpretation. Sponsors are encouraged to seek feedback
Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
18
from the appropriate FDA review division early in drug development. The use of two methods
for BE studies in ANDAs is discouraged.
E. Dried Blood Spots
Dried blood spot (DBS) technology has been under development for several years. The benefits
of DBS include reduced blood sample volumes collected for drug analysis as well as ease of
collection, storage, and transportation. Additional validation of this sampling approach is
essential before using DBS in regulatory studies. This validation should address, at a minimum,
the effects of the following issues: storage and handling temperatures, homogeneity of sample
spotting, hematocrit, stability, carryover, and reproducibility, including ISR. Correlative studies
with traditional sampling should be conducted during drug development. Sponsors are
encouraged to seek feedback from the appropriate FDA review division early in drug
development.
VI. DOCUMENTATION
General and specific SOPs and good record keeping are essential to a properly validated
analytical method. The data generated for bioanalytical method development and/or validation
should be documented and available for data audit and inspection. Documentation at the
analytical site and for submission to the FDA is described in Table 2.
All relevant documentation necessary for reconstructing the study as it was conducted and
reported should be maintained in a secure environment. Relevant documentation includes, but is
not limited to, source data, protocols and reports, records supporting procedural, operational, and
environmental concerns, and correspondence records between all involved parties.
Regardless of the documentation format (i.e., paper or electronic), records should be
contemporaneous with the event, and subsequent alterations should not obscure the original data.
The basis for changing or reprocessing data should be documented with sufficient detail, and the
original record should be maintained.
A. Summary Information
Summary information should include the following items:
• Α summary of assay methods used for each study protocol should be included. Each
summary should provide the protocol number, the protocol title, the assay type, the assay
method identification code, the bioanalytical report code, and the effective date of the
method.
• For each analyte, a summary table of all the relevant method validation reports should be
provided, including partial validation and cross validation reports. The table should
include the assay method identification code, the type of assay, the reason for the new
method or additional validation (e.g., to lower the limit of quantification), and the dates
of final reports. Changes made to the method should be clearly identified.
Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
19
• A summary table cross-referencing multiple identification codes should be provided
when an assay has different codes for the assay method, the validation reports, and the
bioanalytical reports.
B. Documentation for Method Validation and Bioanalytical Reports
Refer to Table 2 for the FDA’s recommended documentation for method validation and
bioanalytical reports.
Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
20
VII. APPENDIX
Table 1. Recommendations and Acceptance Criteria for Bioanalytical Method Validation and In-Study Conduct (refer to
sections III.A and III.B for additional information).
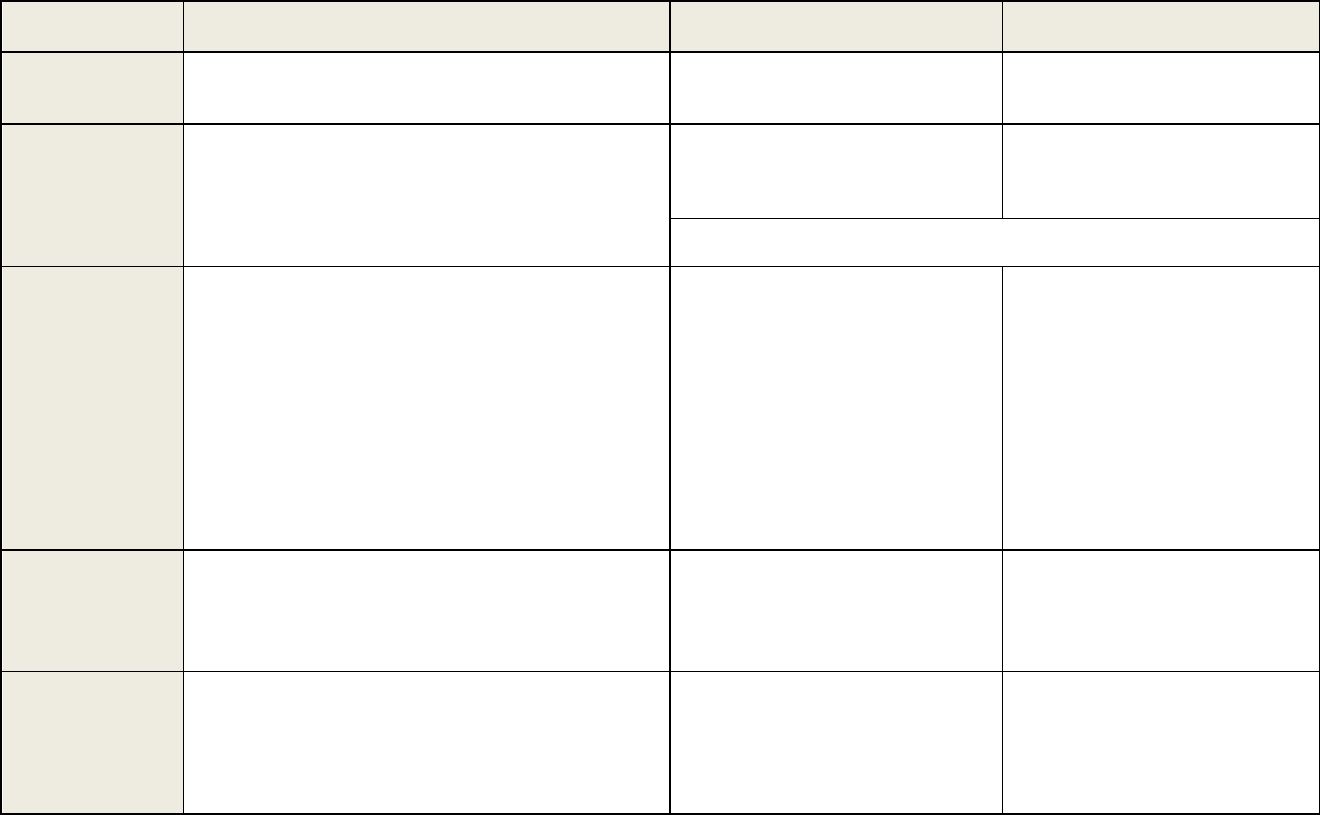
Parameters
Validation Recommendations
In-Study Analysis Recommendations
Chromatographic Assays (CCs)
Ligand Binding Assays (LBAs)
Calibration
Curve
Elements:
• A blank (no analyte, no IS), a
zero calibrator
(blank plus IS), and at least six, non-zero
calibrator levels covering the quantitation
range, including LLOQ in every run.
• All blanks and calibrators should be in the
same matrix as the study samples.
• The concentration-response relationship
should be fit with the simples t regression
model.
Acceptance Criteria:
• Non-zero calibrators should be ± 15% of
nominal (theoretical) concentrations, except
at LLOQ where the calibrator should be ±
20% of the nominal concentrations in each
validation run.
• 75% and a minimum of six non-zero
calibrator levels should meet the above
criteria in each validation run.
Elements:
• A blank and at leas t six, non-zero
calibrator levels covering the quantitation
range, including LLOQ per validation
run.
• Calibration curves are usually run in
duplicate.
• Additional calibrators may be used as
anchor points.
• All blanks and calibrators should be in the
same matrix as the study samples.
• The concentration-response relationship is
usually fit with a four- or five-parameter
logis tic model. Other models may be
acceptable with justification.
Acceptance Criteria:
• Non-zero calibrators should be ± 20% of
nominal (theoretical) concentrations,
except at LLOQ and ULOQ where the
calibrator should be
± 25% of the nominal
concentrations in each validation run.
• 75% and a minimum of six non-zero
calibrator levels should meet the above
criteria in each validation run.
•
Anchor points should not be included in
the curve fit.
Elements:
• A blank, a zero, and at least six, (in duplicate
for LBAs) non-zero calibrator levels covering
the expected range, including LLOQ per
analytical run.
• All blanks and calibrators should be in the same
matrix as the study samples.
• The in-study analysis should use the same
regression model as used in validation.
Acceptance Criteria:
• CC: Non-zero calibrators should be ± 15%,
e
xcept at LLOQ where the calibrator should be
± 20%
of nominal concentrations in each run.
• LBA: Non-zero calibrators should be ± 20%,
except at LLOQ and ULOQ where the
calibrator should be ± 25% of nominal
concentrations in each run.
• CC and LBA: 75% and a minimu m of s ix
non-zero calibrator levels should meet the
above criteria in each run.
Only data points that fail to meet acceptance criteria may be excluded. Exclusion should not change the model used.
Continued

Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
21
Table 1 continued. Recommendations and Acceptance Criteria for Bioanalytical Method Validation and In-Study Conduct
Parameters
Validation Recommendations
In-Study Analysis Recommendations
Chromatographic Assays (CCs)
Ligand Binding Assays (LBAs)
Quality
Controls
(QC)
Elements:
• For A & P Runs: Four QCs, including
LLOQ, low (L: defined as three times the
LLOQ), mid (M: defined as mid-range), and
high (H: defined as high-range) from at least
five replicates in at least three runs
• For Other Validation Runs: L, M, and H
QCs in duplicates
Acceptance Criteria:
• Refer to A & P Runs, Other Validation
Runs, and Stability Evaluations.
Elements:
• For A& P Runs: Five QCs, including
LLOQ, L, M, H, and ULOQ from at least
three replicates in at least six runs
• For Other Validation Runs: L, M, and
H QCs in duplicates
Acceptance Criteria:
• Refer to A & P Runs, Other Validation
Runs, and Stability Evaluations.
Elements:
• ≥ three QC levels (L, M & H) and ≥ two
replicates per QC level in each analytical run.
•
Total QCs should be 5% of unknown samples
or ≥ six, whichever number is greater.
• If the analytical runs consist of dis tinct
processing batches, the QC acceptance criteria
should be applied for the whole run and for
each distinct batch within the runs.
Acceptance Criteria:
• CC: ≥ 67% of QCs should be ± 15% of the
n
ominal, and ≥ 50% of QCs per level should be
± 15% of their nominal.
• LBA: ≥ 67% of QCs should be ± 20% of the
nominal, and ≥ 50% of QCs per level should be
± 20% of their nominal.
Continued
Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
22
Table 1 continued. Recommendations and Acceptance Criteria for Bioanalytical Method Validation and In-Study Conduct
Parameters
Validation Recommendations
In-Study Analysis Recommendations
Chromatographic Assays (CCs)
Ligand Binding Assays (LBAs)
Selectivity
Elements:
• Analyze blank samples of the appropriate
biological matrix from at least six
individual
sources.
Acceptance Criteria:
• Blank and zero calibrators should be free of
interference at the retention times of the
analyte(s) and the IS.
• Spiked samples should be ± 20% LLOQ.
• The IS response in the blank should not
exceed 5% of the average IS responses of
the calibrators and QCs.
Elements:
• Investigate parallelism (for endogenous
products).
• Conduct an analysis of blank samples in
the
matrix from ≥ 10 individual sources
.
Acceptance Criteria:
• Fo r ≥ 80% of sources, unspiked mat rix
should be BQL, and spiked samples
should be ± 25% at LLOQ, and ± 20% at
H QC.
CC Acceptance Criteria:
• In each analytical run, the blank and zero
calibrators should be free of interference at the
retention times of the analyte and the internal
standard
• In each analytical run, the internal standard
response in the blank should not exceed 5% of
average internal standard response of the
calibrators and QCs.
LBA Acceptance Criteria:
•
The blank should be free of interference for the
analyte.
• Parallelism should be conducted if not done
during validation.
Specificity
Elements:
•
The method specificity should be assessed
for interference by cross-
reacting molecules,
concomitant medications, bio-transformed
species, etc.
Acceptance Criteria:
• See Selectivity above.
Elements:
• The method specificity should be assessed
for interference by cross-reacting
molecules, concomitant medications, bio-
transformed species, etc.
• Potential interfering materials should be
added to calibration curves in buffer.
Acceptance Criteria:
• QCs should meet ± 20%, or 25% at the
LLOQ and ULOQ.
Elements:
• Check as needed.
Continued
Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
23
Table 1 continued. Recommendations and Acceptance Criteria for Bioanalytical Method Validation and In-Study Conduct
Parameters
Validation Recommendations
In-Study Analysis Recommendations
Chromatographic Assays (CCs)
Ligand Binding Assays (LBAs)
Carryover
Elements:
• The impact of carryover on the accuracy of
the study sample concentrations should be
assessed.
Acceptance Criteria:
• Carryover should not exceed 20% of
LLOQ.
• Not applicable
Elements:
•
Carryover, if any, should be monitored, and its
impact on the quantitation of study samples
should be addressed.
Acceptance Criteria:
• Carryover should not exceed 20% of LLOQ.
Sensitivity
Elements:
• The lowest nonzero standard on the
calibration curve defines the sensitivity
(LLOQ).
Acceptance Criteria:
• The analyte response at the LLOQ should
be ≥ five times the analyte response of the
zero calibrator.
• The accuracy should be ± 20% of nominal
concentration (from ≥ five replicates in at
least three runs).
•
The precision should be ± 20% CV (from ≥
five replicates in at least three runs).
Elements:
• The lowest nonzero standard on the
calibration curve defines the sensitivity
(LLOQ).
Acceptance Criteria:
• The accuracy should be ± 25% of the
nominal concentration (from ≥ three
replicates in at least six runs).
•
The precision should be ± 25% CV (from
≥ three replicates in at least six runs).
• The total error should be ≤ 40%.
Acceptance Criteria:
In each analytical run:
•
The analyte response at the LLOQ should be ≥
five times the analyte response of the zero
calibrator (CC).
• The A & P for CC should be ±
20% of nominal
concentration.
• The A & P for LBA should be ± 25% of
nominal concentration.
• If the
above criteria are not met, the next higher
calibrator can be selected as the new LLOQ or
the next lower point if the ULOQ fails
(provided the resulting calibration curve meets
acceptance criteria) and does not change the
calibration model.
Continued

Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
24
Table 1 continued. Recommendations and Acceptance Criteria for Bioanalytical Method Validation and In-Study Conduct
Parameters
Validation Recommendations
In-Study Analysis Recommendations
Chromatographic Assays (CCs) Ligand Binding Assays (LBAs)
Accuracy
and
Precision
(A & P)
Elements:
• A & P should be es tablished with at least
three independent A & P runs, four QC
levels per run (LLOQ, L, M, H QC), and ≥
five replicates per QC level.
A & P Run Acceptance Criteria:
• The run should meet the calibration curve
acceptance criteria and include the LLOQ
calibrator.
• This run has no QC acceptance criteria.
Accuracy: Within-run and between runs:
• ± 15% of nominal concentrations; except
± 20% at LLOQ.
Precision: Within-run and between runs:
• ± 15% CV, except
± 20% CV at LLOQ
Total Error:
• Not applicable
Elements:
• A & P should be es tablished with at least
six independent A & P runs, five QC
levels per run (LLOQ, L, M, H, ULOQ
QC), and ≥ three replicates per QC level.
A & P Run Acceptance Criteria:
• The run should meet the calibration
acceptance criteria and include the LLOQ
calibrator.
• This run has no QC acceptance criteria.
Accuracy: Within-run and between runs:
• ± 20% of nominal concentrations; except
±25% at LLOQ, ULOQ
Precision: Within-run and between runs:
• ± 20% CV, except
± 25% at LLOQ, ULOQ
Total Error:
• QCs should be ±30%, except at LLOQ,
ULOQ ±40%
Elements:
• Not applicable
Accuracy: Between runs:
• CC: ± 15% of nominal concentrations
• LBA: ± 20% of nominal concentrations
Precision: Between runs:
• CC: ± 15% CV
• LBA: ±20% CV
Total Error:
• Not applicable
Continued
Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
25
Table 1 continued. Recommendations and Acceptance Criteria for Bioanalytical Method Validation and In-Study Conduct
Parameters
Validation Recommendations
In-Study Analysis Recommendations
Chromatographic Assays (CCs) Ligand Binding Assays (LBAs)
Other
Validation
Runs
Elements:
• ≥ three QC levels (L, M, H) in at least
duplicates in each run.
Run Acceptance Criteria:
• Meet the calibration acceptance criteria
• ≥ 67% of QCs should be ± 15% of the
nominal (theoretical) values, ≥ 50% of QCs
per level should be ± 15% of their nominal
concentrations
Elements:
• ≥ three QC levels (L, M, H) in at least
duplicates in each run
Run Acceptance Criteria:
• Meet the calibration acceptance criteria
• ≥ 67% of QCs should be ± 20% of the
nominal (theoretical) values, and ≥ 50%
of QCs per level should be ± 20% of their
nominal concentrations
• Not applicable
Recovery
Elements:
• Extracted samples at L, M, and H QC
concentrations versus extracts of blanks
spiked with the analyte post extraction (at
L, M, and H)
Elements:
• Need to be demonstrated only if
extraction is involved
Continued

Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
26
Table 1 continued. Recommendations and Acceptance Criteria for Bioanalytical Method Validation and In-Study Conduct
Parameters
Validation Recommendations
In-Study Analysis Recommendations
Chromatographic Assays (CCs) Ligand Binding Assays (LBAs)
Stability
Elements:
• For auto-sampler, bench-top, extract,
freeze-thaw, stock solution and long-term
stability, perform at least three replicates at
L and H QC concentrations.
Acceptance Criteria:
• The accuracy (% nominal) at each level
should be ± 15%.
Elements:
• For auto-sampler, bench-top, extract,
freeze-thaw, s tock solution/reagent and
long-term s tability, perform at least three
replicates at L and H QC concentrations.
Acceptance Criteria:
• The accuracy (% nominal) at each level
should be ± 20%.
Elements:
• Update stability parameters (e.g., long-term)
as needed.
Dilution
Elements:
• QCs for planned dilutions, 5 replicates per
dilution factor:
o Accuracy: ± 15% of nominal
concentrations
o Precision: ± 15% CV
Elements:
• QCs for planned dilutions
• Demonstrate dilution linearity
• Demonstrate lack of prozone effect, i.e.,
increasing analyte concentration results in
no change or decreased signals compared
to the preceding concentration
• 5 replicates per dilution factor:
o Accuracy: ± 20% of nominal
concentrations
o Precision: ± 20% CV
Elements:
• Dilution QC (if not a validated pre-study)
Acceptance Criteria:
• Same as described under ‘QCs’ above
Continued

Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
27
Table 1 continued. Recommendations and Acceptance Criteria for Bioanalytical Method Validation and In-Study Conduct
Parameters
Validation Recommendations
In-Study Analysis Recommendations
Chromatographic Assays (CCs) Ligand Binding Assays (LBAs)
Incurred
Sample
Reanalysis
(ISR)
• Not applicable
• Not applicable
Elements:
• Sample size:
o 10% reanalysis of the first 1000 samples,
and
o
5% reanalysis of the remaining samples
• Sample selection:
o Around C
max
and in the elimination
phase
Acceptance Criteria:
• CC: 67% should be ± 20% of the mean
• LBA: 67% should be ± 30% of the mean
Repeat
Analysis
• No re-analys is of individual calibrators and
QCs is permitted.
• No re-analysis of individual calibrators
and QCs is permitted.
• Re-analysis should be based on reasons
described in a pre-exis ting SOP
• No re-analysis of calibrators and QCs
• At least the same number of replicates for
repeats as originally tested
• No confirmatory repeats for BE studies
Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
28
Table 2. Documentation and Reporting (refer to sections III.B and VI for additional information)
Items
Documentation at the Analytical Site Validation Report* Analytical Study Report*
System
Suitability
• Dates, times, QCs or samples used for suitability testing
• Not applicable
• Not applicable
Synopsis
• Not applicable
• Synopsis of method development
(e.g., evolution of methods with
multiple revisions, unique aspects)
• Not applicable
• Overall summary information
Reference
Standards
and
Critical
Reagents
• Certificate of analysis (CoA) or purity,
stability/expiration data, batch number, and
manufacturer
• Log records of receipt, use, and storage.
• If expired, recertified CoA, or retest of purity &
identity with retest dates
• Internal standard CoA, purity or demonstration of
suitability
• Batch/lot number, purity, and
expiration (see appendix VII, Table
4)
• If expired, purity and stability at the
time of use and retest dates
• Batch/Lot number, purity, and
expiration (see appendix VII,
Table 4)
• If expired, purity and stability at
the time of use and retest dates
Stock Solutions
• Log records of preparation, and use
• Storage location and condition
• Brief description of preparation
• Preparation dates
• Stock s olution stability
• Storage conditions
• Brief description of preparation
• Preparation dates
• Stock s olution stability
• Storage conditions
Blank Matrix
• Records of matrix descriptions, receipt dates, and
storage
• Records of interference checks
•
Matrix effect results
• Des cription, lot number, receipt
dates
• Description of interference check
•
Matrix effect results
• Des cription, lot number, receipt
dates
• Description of interference check
Continued
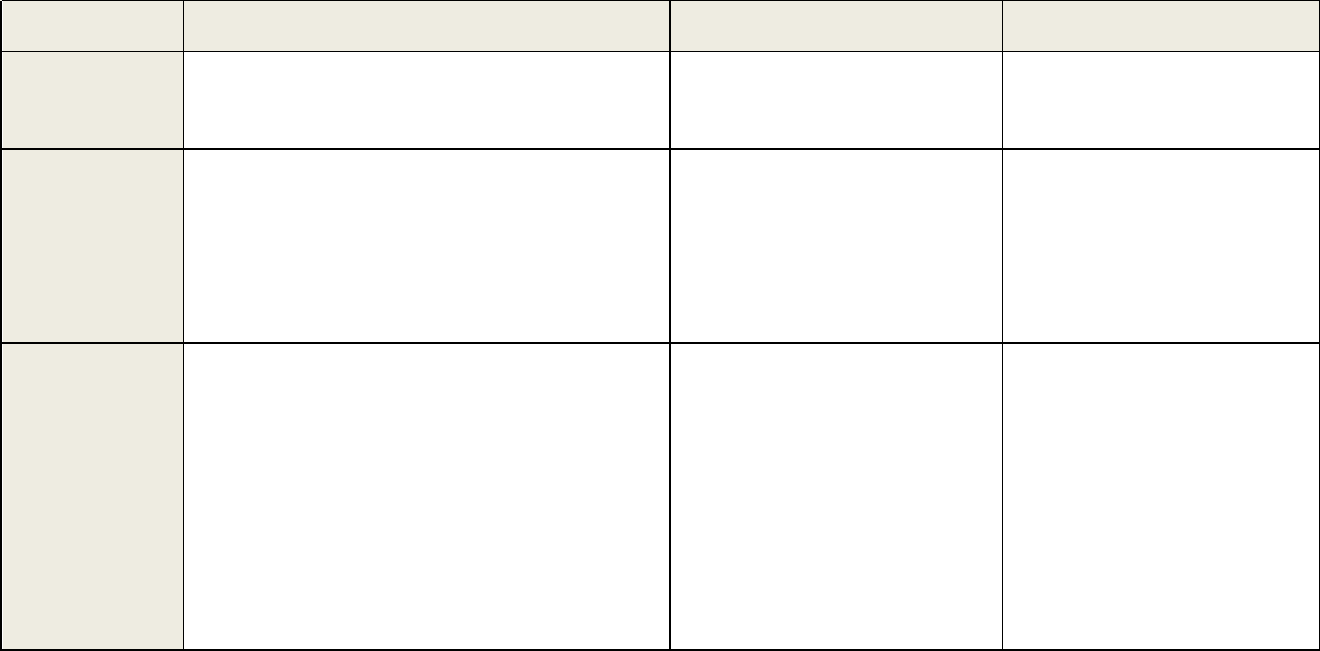
Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
29
Table 2 continued: Documentation and Reporting
Items
Documentation at the Analytical Site Validation Report* Analytical Study Report*
Calibrators
and QCs
• Records of preparation
• Record of storage (e.g., in/out dates, temperatures)
• Brief description of preparation
• Preparation dates
• Storage conditions
• Brief description of preparation
• Preparation dates
• Storage conditions
SOPs
SOPs for all aspects of analysis, such as:
• Method/procedure (validation/analytical)
• Acceptance criteria (e.g., run, calibration curve, QCs)
• Instrumentation
• Re-analysis
• ISR
• Record of changes to SOP (change, date, reason, etc.)
• A detailed description of the assay
procedure
Not applicable
Sample
Tracking
• Study sample receipt, and condition on receipt
• Temperature during shipment
• Sample inventory and reasons for missing samples
• Location of storage
• Tracking logs of QC, calibrators, and study samples
• Freezer logs for QC, calibrators, and study samples
entry and exit
• Storage condition and location of
QCs and calibrators
• Dates of receipt of shipments and
contents
• Sample condition on receipt
• Analytical s ite s torage condition
and location
• Total duration of sample storage
• Any deviations from planned
storage conditions
Continued

Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
30
Table 2 continued: Documentation and Reporting
Items Documentation at the Analytical Site Validation Report* Analytical Study Report*
Analysis
• Documentation and data for system suitability checks
• Instrument use log, including d
ates of analysis for each
run
• Sample extraction logs, including documentation of
processing of calibrators, QCs, and study samples for
each run, including dates of extraction
•
Identity of QC & calibrator lots, and study samples in
each run
•
Documentation of instrument settings and maintenance
• 100% of run summary sheets of passed and failed runs,
including calibration curve, regression, weighting
function, analyte and IS response, response ratio,
integration type
• 100% e-chromatograms of original and re-integrations
from passed and fail runs
• Laboratory information management system (LIMS)
• Validation information, including documentation and
data for:
o Selectivity, sensitivity, precision and accuracy,
carryover, dilution, recovery, matrix effect
o Bench-top, freeze-thaw, long-term, and extract
stability
o Cross/partial validations, if applicable
• Table of all runs (including failed
runs), instrument ID, and analysis
dates
• Tables of calibrator concentration
and response functions results of all
runs with accuracy and precision.
• Tables of within- and between- run
QC results (from accuracy and
precision runs)
• Interference/matrix effect,
sensitivity, carryover, dilution,
recovery
• Bench-top, freeze-thaw, long-term,
extract, and stock solution stability
• Stability QC storage and handling
conditions (dates, duration,
temperature, etc.)
• Partial/cross-validation, if applicable
• Append separate report for
additional validation, if any
• Include total error for LBA methods
• Table of all runs, status (pass and
fail), reason for failure, instrument
ID, and analysis dates (see
appendix VII, Table 4).
• Table of calibrator concentration
& response function results of all
runs (pass and fail) with accuracy
and precision
• Table of QC results of all runs
(pass and fail) with accuracy and
precision results of the QC
samples and bet
ween run accuracy
and precision results from
successful runs
• Table of re-injected runs with
results from original and re-
injected runs and reason(s) for
reinjection
• QC graphs trend analysis
encouraged
•
Study concentration results table
Continued
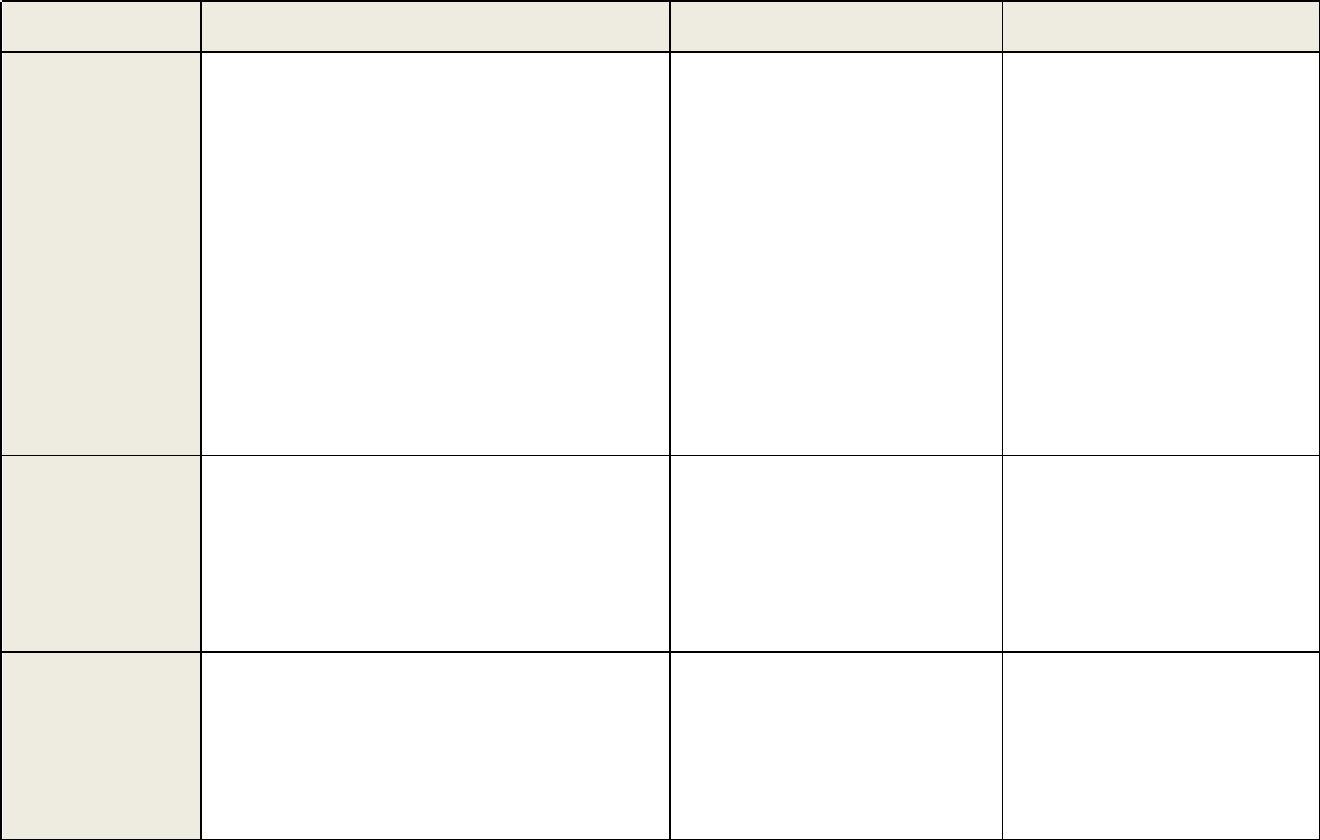
Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
31
Table 2 continued: Documentation and Reporting
Items
Documentation at the Analytical Site Validation Report* Analytical Study Report*
Chromatograms
and
Reintegration
• Electronic audit trail: original and re-integration
• Reason for re-integration
• Mode of re-integration
• Representative chromatograms
(original and re-integration)
• Reason for re-integration
• Chromatograms from 20% of
serially selected subjects for BE
studies in ANDAs
• Randomly selected
chromatograms from 5% of
studies submitted in NDAs and
BLAs
• Original and re-integrated
chromatograms and initial and
repeat integration results for BE
studies
• Reason for re-integration
•
SOP for re-integration
Deviations from
Procedures
• Contemporaneous documentation of deviations/
unexpected events
• Investigation of unexpected events
• Impact assessment
• ISR failure investigations
• Description of deviations
• Impact on study results
•
Description and supporting data of
significant investigations
• Description of deviations
• Impact on study results
• Description and supporting data of
significant investigations
Repeat Analysis
• SOP for re-analysis (Refer to Analysis)
• 100% of repeat data
• Contemporaneous records of reason for repeats
• Not applicable
• Table of sample IDs, reason for
re-assay, original and re-assay
values, reason for reported values,
and run IDs and percent difference
between original and re-assay
values
• Re-analysis SOP
Continued
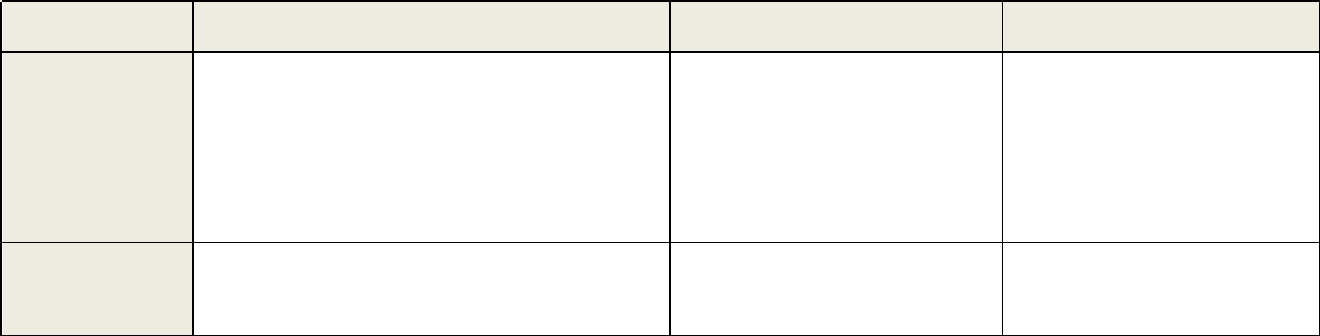
Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
32
Table 2 continued: Documentation and Reporting
Items
Documentation at the Analytical Site Validation Report* Analytical Study Report*
ISR
• SOP for ISR
• ISR data: Run IDs, run summary sheets,
chromatograms or other electronic instrument data
files
• Document ISR failure investigations, if any
• Not applicable
• SOP for ISR
• ISR data table (original,
reanalysis, percent difference,
percent passed)
• ISR failure investigations, if any
Communication
• Between involved parties (Sponsor, contract research
organizations (CROs), and consultants) related to
study/assay
• Not applicable
• Not applicable
*The FDA expects the sponsor to maintain data at the analytical site to support summary data submitted in Validation and Analytical Study Reports.
Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
33
Table 3.
&
Example of an Overall Summary Table for a Method Validation Report* or a Clinical Study Report (this table
contains fictitious information, which serves illustrative purposes only)
Items
Results
Hyperlink
†
Comments
Methodology
LC/MS/MS
01-SOP-001
Method Validation
Report (MVR)
Number
MVR-001
MVR-001
Biological matrix
Human plasma
MVR-001
Anticoagulant (if
applicable)
EDTA
MVR-001
Calibration curve
range
XXX-YYY ng/mL
Summary tables
001MVR-01/CCTables
Report text
001MVR-01/CCText
Analyte of interest
Compound A
NA
Internal standard
Compound A internal
standard
NA
Inter-run accuracy
(for each QC
concentration)
Low QC (AA ng/mL): X%
Medium QC (e.g., BB
ng/mL): Y%
High QC (e.g. CC ng/mL):
Z%
Summary tables
001MVR-01/APTables
Report text
001MVR-01/APText
Inter-run precision
(for each QC
concentration)
Low QC (AA ng/mL): X%
Medium QC (BB
ng/mL): Y%
High QC (CC
ng/mL): Z%
Continued
Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
34
Table 3 continued. Example of an Overall Summary Table for a Method Validation
Report* or a Clinical Study Report
Items
Results
Hyperlink
†
Comments
Dilution integrity
(specify dilution
factors, QC
concentrations, and
matrices that were
evaluated)
Dilution QC: CC ng/mL
(dilution factor: X)
Accuracy: Y%
Precision: Z%
Summary tables
001MVR-01/DILTables
Report text
001MVR-01/DILText
Selectivity
< 20% of the lower limit of
quantification (LLOQ)
-list drugs tested
Summary tables
001MVR-01/SELTables
Report text
001MVR-01/SELText
Short-term or bench-
top temperature
stability
Demonstrated for X hours at
Y°C
Summary tables
001MVR-01/STSTables
Report text
001MVR-01/STSText
Long-term stability
Demonstrated for X days at
Y°C
Summary tables
001MVR-01/LTSTables
Report text
001MVR-01/LTSText
Freeze-thaw stability
Demonstrated for Y cycles at
Z°C
Summary tables
001MVR-01/FTSTables
Report text
001MVR-01/FTSText
Stock solution stability
Demonstrated for X weeks at
YºC
Summary tables
001MVR-01/SSSTables
Report text
001MVR-01/SSSText
Continued
Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
35
Table 3 continued. Example of an Overall Summary Table for a Method Validation
Report* or a Clinical Study Report
Items
Results
Hyperlink
†
Comments
Processed sample
stability
Demonstrated for Y hours at
ZºC
Summary tables
001MVR-01/PSSTables
Report text
001MVR-01/PSSText
ISR
> 67% of samples acceptable
Summary tables
001MVR-01/ISRTables
Report text
001MVR-01/ISRText
Recovery: extraction
efficiency
Summary tables
001MVR-01/EXTTables
Report text
001MVR-01/EXTText
Matrix effects
Summary tables
001MVR-01/MATTables
Report text
001MVR-01/MATText
&
Report Format examples are pertinent for applications to either CDER or CVM. Summary tables should be included in Module 2 of the eCTD.
*Failed method validation experiments should be listed, and data may be requested.
†For eCTD submissions, a hyperlink should be provided for the summary tables and report text.
Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
36
Table 4. Example of Summary Analytical Runs for a Bioanalytical Study Report* (this
table contains fictitious information, which serves illustrative purposes only)
Sponsors and applicants should provide a table summarizing both the failed and accepted runs
for each study.
Clinical Study XXYY-0032456
Analytical
run *
Batch
number
within
analytical
run
Dates of
analysis
Results
(Accepted
/Rejected)
Hyperlink
†
Comments
(e.g. information on runs
that failed)
001-100-01
Not
applicable
MM/DD/YY
Rejected
Summary tables for
calibration curve
standards and QCs
001BR-
01/01CALTables
001BR-
01/01QCTables
Report text
001BR-01/01CALText
001BR-01/01QCText
Raw Data
001BR-
01/01CALData
001BR-01/01QCData
001BR-01/01Failure
67% of the QCs passed;
however both QCs that
exceeded ±15%
were at the low QC
concentration. The follow-
up investigation concluded
that the LC/MS/MS
instrument required a
recalibration.
001-100-02
Not
applicable
MM/DD/YY
Accepted
Summary tables for
calibration curve
standards and QCs
001BR-
01/02CALTables
001BR-
01/02QCTables
Report text
001BR-01/02CALText
001BR-01/02QCText
Raw Data
001BR-
01/02CALData
001BR-01/02QCData
This is the reanalysis of the
samples from run 001-100-
01
*If multiple batches are analyzed within an analytical run, each batch should be separately evaluated to determine if
the batch meets acceptance criteria.
†For eCTD submissions, a hyperlink should be provided for the summary tables, report text, and raw data.
Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
37
VIII. GLOSSARY
Accuracy: Accuracy is the degree of closeness of the determined value to the nominal or known
true value under prescribed conditions. Accuracy is also sometimes termed trueness.
Analyte: An analyte is the specific chemical moiety being measured; it can be an intact drug, a
biomolecule or its derivative, a metabolite, or a degradation product in a biologic matrix.
Analytical run: An analytical run is a complete set of analytical and study samples with an
appropriate number of standards and QCs for their validation. Several runs can be completed in
one day, or one run may take several days to complete.
Autosampler stability: Autosampler stability is the stability of the analyte in the processed
sample under the conditions in the autosampler.
Biological matrix: A biological matrix is discrete material of biological origin that can be
sampled and processed in a reproducible manner. Examples are blood, serum, plasma, urine,
feces, cerebrospinal fluid, saliva, sputum, and various discrete tissues.
Batch: For purposes of this guidance, a batch is a number of unknown samples from one or
more patients in a study and QCs that are processed at one time.
Between run: Between run refers to the distinct period between or among several analytical or
validation runs.
Bench-top stability: Bench-top stability is the stability of an analyte in a matrix under
conditions of sample handling during sample processing.
Blank: A blank is a sample of a biological matrix to which no analytes have been added that is
used to assess the selectivity of the bioanalytical method.
Calibration curve: The calibration curve — also known as the standard curve — is the
relationship between the instrument response and the calibration standards within the intended
quantitation range.
Calibrators/Calibration standards: Calibrators, or calibration standards, refer to a biological
matrix to which a known amount of analyte has been added. Calibration standards are used to
construct calibration curves from which the concentrations of analytes in QC samples and in-
study samples are determined.
Carryover: Carryover is the appearance of an analyte in a sample from a preceding sample.
Critical reagents: Critical reagents are requisite components of an assay, which include
antibodies, labeled analytes, matrices, etc.
Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
38
Dilutional linearity: Dilutional linearity demonstrates the accurate measurement of
concentrations of spiked samples (i.e., QCs) exceeding the quantitation range when serially
diluted to within the quantitative assay range.
Extract: An extract is a sample treated to remove impurities or interfering substances (also
known as a processed sample).
Extract stability: Extract stability assesses the degradation of the processed sample relative to
the starting material.
Freeze-thaw stability: Freeze-thaw stability refers to the stability of the analyte in the matrix
upon freezing and thawing.
Freshly prepared: Freshly prepared refers to QC sample preparation (i.e., spiked) on the day of
the experiment; not frozen before use.
Full validation: Full validation refers to the establishment of all validation parameters that
apply to sample analysis for the bioanalytical method for each analyte.
Heteroscadisticity:
Heteroscadisticity occurs when the variance of a response is not constant
but changes with the response.
Hook effect: The hook effect occurs when increasing analyte concentrations result in no change
or decreased signals when compared to the preceding concentration.
Incurred samples: Incurred samples are study samples or samples from subjects or patients
who were dosed.
Incurred Sample Reanalysis (ISR): ISR is the repeated measurement of an analyte’s
concentration from study samples to demonstrate reproducibility.
Interference: Interference refers to the action of sample components, including structurally
similar analytes, metabolites, impurities, degradants, or matrix components that may impact
quantitation of the analyte of interest. Refer to Selectivity and Matrix effect for further
information.
Internal standard (IS): ISs are test compounds (e.g., structurally similar analogs, stable isotope
labeled compounds) added to both calibration standards and samples at known and constant
concentrations to facilitate quantification of the target analyte(s).
Long-term stability: Long-term stability assesses the degradation of an analyte in the matrix
relative to the starting material after periods of frozen storage.
Lower limit of quantification (LLOQ): The LLOQ is the lowest amount of an analyte that can
be quantitatively determined with acceptable precision and accuracy.
Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
39
Matrix effect: The matrix effect is a direct or indirect alteration or interference in response
because of the presence of unintended analytes (for analysis) or other interfering substances in
the sample.
Method: A method is a comprehensive description of all procedures used in the collection,
storage, and analysis of samples.
Non-zero calibrator: A non-zero calibrator is a calibrator to which the internal standard is
added.
Nominal concentration: The nominal concentration is the actual or intended concentration of
the calibrator or quality control samples.
Parallelism: Parallelism demonstrates that the serially diluted incurred sample response curve is
parallel to the calibration curve. Parallelism is a performance characteristic that can detect
potential matrix effects and interactions between critical reagents in an assay.
Precision: Precision is the closeness of agreement (i.e., degree of scatter) among a series of
measurements obtained from multiple sampling of the same homogenous sample under the
prescribed conditions.
Processed sample: A processed sample is the final extract (before instrumental analysis) of a
sample that has been subjected to various manipulations (e.g., extraction, dilution,
concentration).
Processing batch: A processing batch is a group of unknown samples from one or more study
subjects, calibrators, and a set of QCs that are subjected to the analytical methodology together.
Prozone: The prozone is an effect observed when increasing analyte concentrations result in
either no change or decreased detector response when compared to the preceding concentration.
(Also see the Hook effe ct)
Quality control sample (QC): A QC is a biologica l matrix with a known quantity of analyte
that is used to monitor the performance of a bioanalytical method and to assess the integrity and
validity of the results of study samples analyzed in an individual run.
Quantification range: The quantification range is the range of concentrations, including the
ULOQ and the LLOQ that can be reliably and reproducibly quantified with accuracy and
precision with a concentration-response relationship.
Recovery: Recovery refers to the extraction efficiency of an analytical process, reported as a
percentage of the known amount of an analyte carried through the sample extraction and
processing steps of the method.
Reintegration: Reintegration is a reanalysis of the chromatographic peak.
Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
40
Reference standard: A reference standard is a chemical substance of known purity and identity
which is used to prepare calibration standards and quality controls. Three types of reference
standards are usually used: (1) certified (e.g., USP compendial standards), (2) commercially-
supplied, and (3) custom-synthesized.
Reproducibility: Reproducibility is the precision between two laboratories. It also represents
the precision of the method under the same operating conditions over a short period of time.
Response function: Response function is the mathematical expression that describes the
relationship between known sample concentrations and the response of the instrument (Also
refer to Calibration curve).
Sample: A sample is a generic term encompassing controls, blanks, unknowns, and processed
samples.
Selectivity: Selectivity is the extent to which the method can determine a particular compound
in the analyzed matrices without interference from matrix components.
Sensitivity: Sensitivity is defined as the lowest analyte concentration in the matrix that can be
measured with acceptable accuracy and precision (i.e., LLOQ).
Specificity: Specificity is the ability of the method to assess, unequivocally, the analyte in the
presence of other components that are expected to be present (e.g., impurities, degradation
products, matrix components, etc.).
Spiked samples: A spiked sample is a general term that refers to calibrators (calibration
standards) and quality controls.
Stability: Stability is a measure of the intactness an analyte (lack of degradation) in a given
matrix under specific storage and use conditions relative to the starting material for given time
intervals.
Standard curve: Refer to Calibration curve.
Stock Solution: A stock solution refers to an analyte in a solvent or mixture of solvents at a
known concentration, which is used to prepare calibrators or QCs.
Study samples: Study samples refer to samples from subjects or patients enrolled in a study.
System suitability: System suitability is a determination of instrument performance (e.g.,
sensitivity and chromatographic retention) by analyzing a set of reference standards before the
analytical run.
Total error: Total error is the sum of the absolute value of the errors in accuracy (%) and
precision (%). Total error is reported as percent (%) error.
Contains Nonbinding Recommendations
Bioanalytical Method Validation
05/24/18
41
Unknown: An unknown is a biological sample that is the subject of the analysis.
Upper limit of quantification (ULOQ): The ULOQ is the highest amount of an analyte in a
sample that can be quantitatively determined with precision and accuracy.
Within-run: Within-run refers to the time period during a single analytical or validation run.
Zero calibrator: A zero calibrator is a blank sample to which the internal standard is added.
