
Page 1
Optimizing Unit Operations In Biopharmaceutical Manufacturing
Introduction
Biopharmaceutical manufacturing may come in a wide
variety of forms, but every iteration of unit operation must
adhere to an unbending set of operational parameters and
structures if the desired outcome – a viable, contaminant-
free drug suitable for human or animal administration – is
to be realized.
Three of the more common unit operations within
the biopharmaceutical-manufacturing universe are
chromatography, virus filtration and tangential flow
filtration (TFF). In order for these unique operations to
be implemented successfully, though, the operator must
be aware of their specific operating characteristics. For
example, chromatography requires constant fluid-flow rates
during their operations, but may have varying pumping
pressures. Virus filtration, on the other hand, will feature
constant pumping pressures, but flow rates will change as
Chromatography, virus filtration and tangential flow filtration (TFF) are three of the more common unit operations that can be used in the manufacture of biopharmaceuticals. In
order to provide efficient, reliable and cost-effective processes in all three unit operations, as well as guaranteeing the production of drugs that are suitable for human or animal
consumption, manufacturers must utilize a pump technology that features low-pulsation and low-shear operation even when encountering variable flow rates and pumping
pressures. A technology that meets all of those operational requirements is the Quaternary Diaphragm Pump from Quattroflow™, which also possesses the versatility to operate
effectively in a fixed stainless-steel production setup, as well as in the increasingly popular single-use manufacturing applications.
the filters become clogged or fouled. And in TFF, the main
challenge is attempting to keep the flow rate and pressure
unchanging throughout the process.
While fluid transfer is taking place in any of these specific
unit operations, it is important to know that the materials
that are being transferred can be highly sensitive and
delicate (and, in many cases, expensive), meaning that the
pumping action must be low-pulsating and low-shear, lest
the material be damaged.
This white paper will examine the material-handling
challenges pertaining to flow rate and pressure in
chromatography, virus filtration and TFF processes and
illustrate why the design and operation of the quaternary
diaphragm pump – rather than other technologies such as
the lobe or peristaltic (hose) pump – makes it ideal for use
in critical biopharmaceutical-manufacturing applications.
Additionally, the paper will show how the quaternary
diaphragm pump’s ability to operate consistently whether
Optimizing Unit Operations In Biopharmaceutical Manufacturing
CHROMATOGRAPHY, VIRUS FILTRATION AND TANGENTIAL FLOW FILTRATION ALL HAVE UNIQUE DEMANDS
WITH QUATERNARY DIAPHRAGM PUMPS OFFERING THE LOW PULSATION AND SHEAR THAT IS CRITICAL TO THESE TECHNIQUES
By Glenn Hiroyasu
April 2017

Page 2
Optimizing Unit Operations In Biopharmaceutical Manufacturing
in a fixed stainless-steel production regime or in the
increasingly popular single-use applications gives it the
versatility to optimize biopharmaceutical-manufacturing
maintenance, downtime, changeover and operational costs.
The Unit Operations
Let’s start by taking a closer look at three of the
more popular unit operations in biopharmaceutical
manufacturing:
Chromatography Columns
A typical chromatography column, whether it is glass,
steel or plastic, is filled with resins that are compressed in
a certain format through which the feed stream product
flows and purifies the product by selective adsorption
to a stationary phase (resin). Chromatography columns
contain complex target-product adsorbing media that
need careful handling. Protein A resin, for example, can
cost as much as $10,000 a liter, making proper feeding of
the resin extremely important.
Some chromatography systems require buffer gradients
in order to achieve purification of the proteins. Buffers
are compounds that are immune to changes in their pH
level when limited amounts of acids or bases are added to
them. For example, buffering salts have a wide pH range
and can effectively stabilize the pH level of the material.
Quite often more than one buffer is required, which
creates the need to use two or more pumps. In this
application, high- and low-salt buffers are mixed
continuously and with changing ratios in order to
affect the adsorption of the target molecule to the
chromatography resin. Because of this, precise pumping
is required to achieve the right pH/conductivity
conditions for specific adsorption and high-resolution
purification. For example, a Buffer A and a Buffer B
can be used to create a gradient that ranges from a
low-salt buffer to a high-salt buffer in a linear fashion.
Specifically, the process will begin with Buffer A
producing 95% of the flow and Buffer B the remaining
5%. Over the course of the operation, the flow rates of
Buffer A and Buffer B will decrease and increase in a
linear fashion (90% for A and 10% for B, 75% for A and
25% for B, all the way to 5% for Buffer A and 95% for
Buffer B)
1
.
This requires a pumping technology that can produce
a highly accurate flow with a high turndown ratio that
can deliver low and high flow rates as the elution stage
continues. Pump pulsation should also be minimized to
prevent disturbance of the packed column
2
. If the pump
is not able to meet these requirements, the correct buffer
concentration may not be attained. Also, if the pumping
action produces excessive pulsation, the buffers can be
susceptible to experiencing spikes in their conductivity.
This can affect the purification level of the product as the
salt level in the buffer could be compromised.
Also, during the loading of the sample, it is not
uncommon for the system’s back pressure to increase.
Pumps that do not slip offer benefits in these situations
since their flow rates will remain consistent and the
linear velocity will remain stable. Simply put, a pump
with minimal slip will have a more easily controlled flow
rate that will need only incremental adjustments to the
pump’s speed (measured in RPMs).
Virus Filtration
In biopharmaceutical manufacturing, virus-filtration
systems are used to ensure the viability and safety of
the drugs that are produced through the removal of
potential contaminants from products that are created
using cell cultures. Whereas chromatography features
constant flow rates and variable pressures, the operation
of virus-filtration systems is the opposite – most virus-
filter applications use constant pressures with variable
flows. In other words, you may have to raise and lower
the operation’s flow rate or speed in order to maintain a
constant pressure.
As mentioned, the flows change as the virus filter
becomes clogged. Most typical virus-filtration systems
run at a constant pressure, for example, 2 bar (29 psi),
due to the nature of the tight pores in the filtering
medium, but the flow rates will decrease as the filter’s
pores become fouled. When this happens, the flow rate
will not decrease in a linear fashion, which will adversely
affect the performance of the filter, product yield and
overall quality.
After its invention in 2000, quaternary diaphragm pump technology from Quattroflow™
quickly gained popularity as a low-pulse, low-shear option for the various unit
operations that can be used in the manufacture of biopharmaceuticals.
Page 3
Optimizing Unit Operations In Biopharmaceutical Manufacturing
Some virus filters have been designed with a flux-
decay capacity of up to 90% of the starting flux rate,
which requires a pump that has both a high turndown
ratio and produces minimal pulsation in the pumped
fluid. Evaluation of viral clearance strategies requires
demonstration of the equivalence of scalability from
bench to manufacturing scale and vice versa
3
. Spiking
studies for virus-filtration use a pressure vessel with a
small surface area, which can be as little as 5 cm
2
and
demand a pump that has low-shear and low-pulsation
operation if commercial-scale production levels are to
replicate the small-scale studies. The use of low-pulsing
pumps in these circumstances can ensure that pressure
conditions during validation of the particular filter are
not outside of the validated range.
Tangential Flow Filtration (TFF)
Also known as cross-flow filtration, in TFF the biologic
feed stream flows tangentially across the filter membrane
at positive pressure. As it passes across the membrane,
the portion of the feed stream that is smaller than the
membrane’s pore size passes through the membrane. This
is different from what is known as normal-flow (NFF), or
“dead-end,” filtration, in which the feed flows entirely
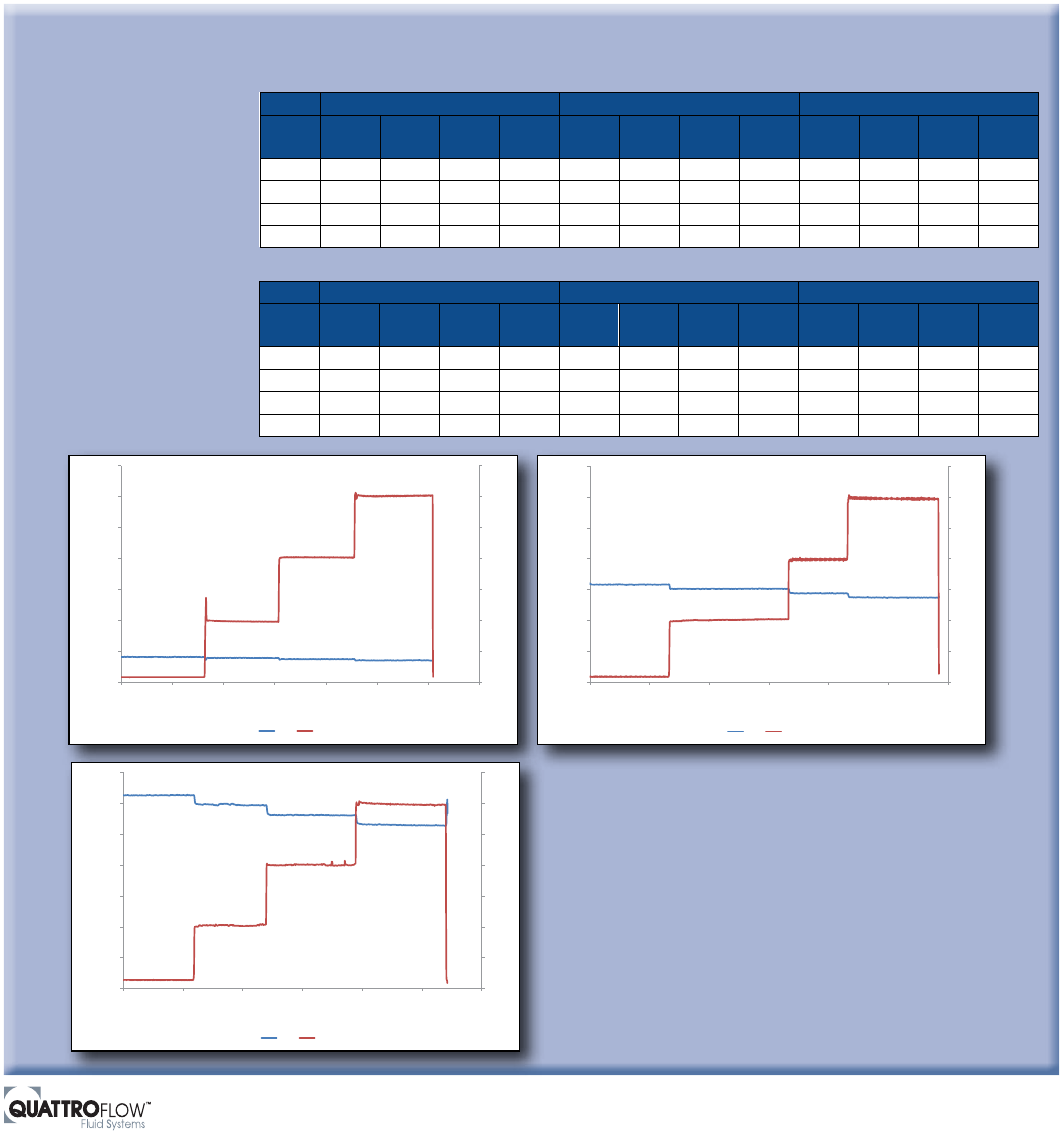
Determining Flow and Pressure Pulsation of Quaternary Diaphragm Pumps
Flow Pulsation
250 rpm 1000 rpm 2000 rpm
Pressure
min
[LPH]
max
[LPH]
average
[LPH]
max.
pulsati
on
[LPH]
min
[LPH]
max
[LPH]
average
[LPH]
max.
pulsati
on
[LPH]
min
[LPH]
max
[LPH]
average
[LPH]
max.
pulsation
[LPH]
free flow
158
163
160
5
630
637
632
7
1249
1255
1252
6
2 bar
152
156
154
4
602
607
605
5
1184
1197
1190
13
4 bar 143 148 146 5 573 578 576 5 1120 1126 1123 6
6 bar 136 141 138 5 545 552 548 7 1054 1067 1059 13
250 rpm 1000 rpm 2000 rpm
Pressure min
[bar]
max
[bar]
average
[bar]
max.
pulsati
on
[bar]
min
[bar]
max
[bar]
average
[bar]
max.
pulsati
on
[bar]
min
[bar]
max
[bar]
average
[bar]
max.
pulsati
on
[bar]
free flow 0.14 0.15 0.15 0.01 0.17 0.19 0.18 0.02 0.27 0.29 0.28 0.02
2 bar 1.93 1.99 1.96 0.06 1.97 2.05 2.02 0.08 2.00 2.09 2.04 0.09
4 bar
4.01
4.04
4.03
0.03
3.90
4.04
3.97
0.14
3.97
4.14
4.00
0.17
6 bar 5.99 6.05 6.02 0.06 5.90 6.00 5.95 0.10 5.91 6.04 5.97 0.13
Pressure Pulsation
The maximum flow pulsation measured by the quaternary diaphragm
pump was 13 L/hr (3.4 gph), which was approximately 1.2% of the
average flow rate. Regarding pressure pulsation, the maximum value
was 0.17 bar (2.5 psi), which is 4.2% of the average pressure. These
results indicate that operation of quaternary diaphragm pumps is quite
capable of minimizing harmful pulsation in critical biopharmaceutical-
manufacturing and handling applications.
Flow Pulsation
250 rpm 1000 rpm 2000 rpm
Pressure
min
[LPH]
max
[LPH]
average
[LPH]
max.
pulsati
on
[LPH]
min
[LPH]
max
[LPH]
average
[LPH]
max.
pulsati
on
[LPH]
min
[LPH]
max
[LPH]
average
[LPH]
max.
pulsation
[LPH]
free flow
158
163
160
5
630
637
632
7
1249
1255
1252
6
2 bar
152
156
154
4
602
607
605
5
1184
1197
1190
13
4 bar 143 148 146 5 573 578 576 5 1120 1126 1123 6
6 bar
136
141
138
5
545
552
548
7
1054
1067
1059
13
250 rpm 1000 rpm 2000 rpm
Pressure min
[bar]
max
[bar]
average
[bar]
max.
pulsati
on
[bar]
min
[bar]
max
[bar]
average
[bar]
max.
pulsati
on
[bar]
min
[bar]
max
[bar]
average
[bar]
max.
pulsati
on
[bar]
free flow 0.14 0.15 0.15 0.01 0.17 0.19 0.18 0.02 0.27 0.29 0.28 0.02
2 bar 1.93 1.99 1.96 0.06 1.97 2.05 2.02 0.08 2.00 2.09 2.04 0.09
4 bar
4.01
4.04
4.03
0.03
3.90
4.04
3.97
0.14
3.97
4.14
4.00
0.17
6 bar 5.99 6.05 6.02 0.06 5.90 6.00 5.95 0.10 5.91 6.04 5.97 0.13
Pressure Pulsation
0
1
2
3
4
5
6
7
0
200
400
600
800
1000
1200
1400
0 200 400 600 800 1000 1200 1400
P [barg]
Q [L/H]
Time [sec]
QF1200S; 250RPM
LPH BAR
0
1
2
3
4
5
6
7
0
200
400
600
800
1000
1200
1400
0 200 400 600 800 1000 1200
P [barg]
Q [L/H]
Time [sec]
QF1200S; 1000RPM
LPH BAR
0
1
2
3
4
5
6
7
0
200
400
600
800
1000
1200
1400
0 200 400 600 800 1000 1200
P [barg]
Q [L/H]
Time [sec]
QF1200S; 2000RPM
LPH BAR
0
1
2
3
4
5
6
7
0
200
400
600
800
1000
1200
1400
0 200 400 600 800 1000 1200 1400
P [barg]
Q [L/H]
Time [sec]
QF1200S; 250RPM
LPH BAR
0
1
2
3
4
5
6
7
0
200
400
600
800
1000
1200
1400
0 200 400 600 800 1000 1200
P [barg]
Q [L/H]
Time [sec]
QF1200S; 1000RPM
LPH BAR
0
1
2
3
4
5
6
7
0
200
400
600
800
1000
1200
1400
0 200 400 600 800 1000 1200
P [barg]
Q [L/H]
Time [sec]
QF1200S; 2000RPM
LPH BAR
0
1
2
3
4
5
6
7
0
200
400
600
800
1000
1200
1400
0 200 400 600 800 1000 1200 1400
P [barg]
Q [L/H]
Time [sec]
QF1200S; 250RPM
LPH BAR
0
1
2
3
4
5
6
7
0
200
400
600
800
1000
1200
1400
0 200 400 600 800 1000 1200
P [barg]
Q [L/H]
Time [sec]
QF1200S; 1000RPM
LPH BAR
0
1
2
3
4
5
6
7
0
200
400
600
800
1000
1200
1400
0 200 400 600 800 1000 1200
P [barg]
Q [L/H]
Time [sec]
QF1200S; 2000RPM
LPH BAR
Using water at ambient temperature
as the medium, pressure and flow
rates were recorded for a quaternary
diaphragm pump at free flow,
2 bar, 4 bar and 6 bar (29, 58 and
87 psi) pressure and at motor
speeds of 250, 1,000 and 2,000 rpm.
The measuring frequency was one
measuring point per second (1 Hz),
and the duration measuring point
was approximately five minutes.
At those operational parameters,
the following results were observed:
Page 4
Optimizing Unit Operations In Biopharmaceutical Manufacturing
through the filter membrane with the size of the pores
determining which portion of the feed is allowed to
pass through and which will remain trapped in the filter
membrane.
TFF is different from NFF in biologic applications because
the tangential motion of the fluid across the membrane
prevents molecules from building up a compact gel layer
on the surface of the membrane. This mode of operation
means that a TFF process can operate continuously with
relatively high protein concentrations with less fouling or
binding of the filter.
To scale up a TFF process there are two variables that
need to be successfully controlled. Recirculation (cross-
flow) is required to minimize formation of the gel layer
and pressure as the driving force to push the permeate
through the membrane. The recirculation rate needs to
work in conjunction with the pressure (known as the
trans-membrane pressure, or TMP, which is the average
amount of pressure that is applied to the membrane).
Maintaining a constant TMP is critical because if it is
too high it can cause gel-layer formation that cannot
be removed by recirculation, and if it is too low it
results in low flux that will reduce process efficiency.
In this instance, pumps that deliver low-pulsation flow
characteristics will perform most reliably by decreasing
the fluctuation in the variables.
So, in considering the functional design of
chromatography columns, virus-filtration systems
and TFF systems, the common thread in guaranteeing
efficient, reliable, cost-effective operation is found in
identifying and using a pump technology that is capable
of producing both low-pulsation and low-shear operation
despite varying flow rates and pumping pressures.
The Challenge
With these operational requirements in mind, over the years
various pump technologies have been used or tested for
chromatography, virus filtration and TFF processes. Two that
are among the more popular choices of biopharmaceutical
manufacturers are lobe and peristaltic pumps. Both, however,
have been found to feature operational inefficiencies that may
make them insufficient for use in the processes described above.
Lobe Pumps
Since many biopharmaceutical materials are contained in a
low-viscosity aqueous solution, lobe pumps may not be a good
choice because slippage can occur during their operation, which
can vary between 10% to 100%, depending on the system’s
back pressure. Slip will also result in increased shear damage
and energy consumption, and if used in a long-duration
recirculation loop, such as a TFF filtration system, there can be
noticeable heat addition to the product that requires significant
cooling efforts to protect the product from overheating.
Lobe pumps also have mechanical seals, which is a controlled
product leak and does not provide full containment unless
special (and oftentimes expensive) seals and seal barriers are
used. The sterility required in biopharmaceutical handling also
means that no outside contaminants can be introduced into
the purification process, which is something that pumps with
mechanical seals cannot reliably ensure.
Additionally, the necessary contact between a lobe pump’s
internal parts can lead to wear and the generation of particles
that can result in product contamination. Solid particulates,
such as undissolved salt crystals, can cause severe damage to the
lobes, resulting in damage to the entire manufacturing batch.
Lobe pumps will ultimately cost more to operate because of the
increase in power required to overcome the pump’s slippage.
Fixed Speed Curves
Performance of Quattroflow
™
Pumps and Lobe Pumps Compared
Quattroow pump at maximum speed. Pump is only slightly
inuenced by pressure and wear over time.
The same Quattroow pump at half speed. Pump is only
slightly inuenced by pressure and wear over time. Pump is
able to match a lobe pump that slips at maximum speed.
Larger traditional lobe pump slips and needs to be oversized.
Smaller traditional lobe pump does not have needed ow
range (turn-down) to meet ow.
* For applications that experience loss of performance
from pump wear.
*

Page 5
Optimizing Unit Operations In Biopharmaceutical Manufacturing
Peristaltic (Hose) Pumps
The main shortcoming of peristaltic pumps is also the
most obvious: their method of operation will undoubtedly
produce pulsation, and, as noted, pulsation is always bad
in biopharmaceutical manufacturing. Peristaltic pumps
also have limited flow and pressure-handling abilities. For
example, they cannot reliably produce the higher discharge
pressures (such as 4 bar, or 58 psi) that are required in some
fluid-handling applications.
They are also known to release some small quantity of
hose material – in a process known as “spalling” – into
the pumped product, which can compromise its purity. If
the spalled hose material makes its way to the filter, it can
foul the filter, making its operation not as efficient as it
needs to be, which will also lead to contamination. Also,
inconsistency of flow rate will result due to mechanical
deformation of the hose during the pumping process.
In the end, the shortcomings of lobe and peristaltic pumps
come down to two main things:
• If there is shear, which is common in lobe pumps,
you will damage the pumped material
• If there is pulsation, an operational certainty with
peristaltic pumps, you won’t have even flow, and
without even flow, you won’t have accurate flow
The Solution
An effective solution to the operational shortcomings of
lobe and peristaltic pumps can be the quaternary diaphragm
pump.
The motivation behind the invention of the quaternary
diaphragm pump goes back 30 years to the mid-1980s, at
the time of what is now referred to as the “birth of the
modern biotech industry.” In 1986, Frank Glabiszewski was
an engineer for a German filter manufacturer and he was
growing increasingly frustrated with the overall operation
of the pumping technologies that were commonly used in
chromatography and TFF applications.
“We were using peristaltic pumps but we found out that
these pumps were not made for TFF applications. We were
checking the marketplace looking for better pumps and
spent 80% of our time looking for pumps,” Glabiszewski
recalled. “One night I went home and I was sitting in
my car frustrated after a pump failure and asked myself a
question, ‘Which type of pump did Mother Nature invent
to process sensitive biologic fluids like blood?’”
4
The answer, of course, was the human heart, and with that
in mind, Glabiszewski began working with his friend and
engineering partner, Josef Zitron, to perfect the design and
operation of the quaternary diaphragm pump technology.
When the new technology was finalized in 2000, the
pair created a company that would begin producing
quaternary diaphragm pumps for use in biopharmaceutical-
manufacturing processes. As the duo’s invention grew in
popularity over the past 15 years, the technology was also
modified so that it could employ disposable plastic heads
and wetted parts to make it applicable in the burgeoning
single-use biopharmaceutical-production marketplace.
The operating principle of the quaternary diaphragm pump
most closely resembles the operation of the human heart
because the four-piston diaphragm technology enables
a gentle pumping action through soft “heartbeats.” This
action produces four overlapping pumping strokes of the
pistons that efficiently reduce pulsation since each stroke of
the four diaphragms is generated by an eccentric shaft that
is connected to an electric motor.
The quaternary diaphragm pump’s method of operation
allows it to gently, safely and securely convey low-viscosity
aqueous solutions and biopharmaceutical materials that are
highly sensitive to shear forces and pulsation while being
pumped. Since the four-piston design of the pump does
not require any mechanical seals or wetted rotating parts,
total product containment is ensured without any abrasion
or particulate generation. The pump’s method of operation
also produces risk-free dry-running and self-priming
capabilities with high turndown ratios. A pump technology
with high turndown ratios allows for the creation of a
broad flow range, which makes the pump applicable for
utilization in a wide range of process applications.
With regards to specific unit operations, quaternary
diaphragm pumps can be used to pack chromatography
columns and then pump the biopharmaceutical material
through the column, both of which are critical concerns
that require low pulsation with accurate and constant
flow rates and pressures. In TFF applications, quaternary
diaphragm pumps deliver the consistent flow control that
is essential in producing optimal filtrate yields.
In today’s evolving biopharmaceutical-manufacturing
processes, quaternary diaphragm pumps are also rapidly
becoming a first-choice technology in increasingly popular
single-use production setups. Basically, a single-use pump
enables biopharmaceutical manufacturers to eliminate
the cost of cleaning and validating their pumps by using
a pump with a replaceable pump head. The result is not
only a quicker production process, but one that delivers
preferred levels of product purity and sterility with no
chance for cross-batch or cross-product contamination.

Page 6
Here are some of the other advantages that can be realized
when quaternary diaphragm single-use pumps are used:
• Can be used for one product or in one production
campaign
• At the conclusion of the production campaign, the
pump chamber that has come in contact with the
fluids is disposed of
• Can be used for a set amount of time before the
wetted parts are replaced, which eliminates elevated
maintenance costs
• If the operator needs to use a stainless-steel pump,
the plastic pumping chamber can be replaced with a
stainless-steel one
• If there’s a pump failure, the old chamber can be taken
out and replaced with a new one in five minutes
• Used when cleaning in place (CIP) or steam
sterilization is not practical or possible. This represents
a significant simplification and cost reduction to the
overall process as there are no contaminated cleaning
chemical and water solutions that need to be treated
and disposed of. The costs to properly treat and
dispose the cleaning fluids can alone be the driver to
require use of single-use alternatives.
Of course, not every pump technology is completely
perfect for every characteristic of a specific fluid-handling
application. In this instance, the design and operation of the
quaternary diaphragm pump limits it to handling fluids that
have a maximum viscosity of 1,000 centipoise (cPs) and that
contains particulates up to 0.1 millimeter in diameter.
Conclusion
The importance of biopharmaceuticals means that any
and all products must meet strict demands regarding their
manufacture. This includes ensuring that no damage is done
to component materials during critical chromatography,
virus-filtration or TFF processes. While lobe and peristaltic
pumps have been preferred pumping technologies for these
unit operations, a better choice can be the quaternary
diaphragm pump, the operation of which greatly reduces the
chance that pulsation and shear will compromise the safety
and effectiveness of the end product.
References:
1. http://www.pall.com/main/biopharmaceuticals/product.page?id=33058
2. L. Hagel, G. Jagschies and G. Sofer, Handbook of Process
Chromatography: Development, Manufacturing, Validation and
Economics, 1997
3. H. Aranha and S. Forbes, “Viral Clearance Strategies for
Biopharmaceutical Safety” Pharmaceutical Technology, June 2001
4. Video: “Quattroflow
™
: The New Standard in Biopharmaceutical Pump
Excellence”
About the Author:
Glenn Hiroyasu is the Americas Development Manager for
Quattroflow
™
Fluid Systems GmbH, Kamp-Lintfort, Germany.
He can be reached at [email protected]. Quattroflow
offers innovative quaternary diaphragm pump technology for
use in pharmaceutical and biopharmaceutical applications that
require gentle displacement, reliability, product safety, purity and
cleanability. Quattroflow
™
is a brand of Almatec
®
Maschinenbau
GmbH, Kamp-Lintfort, Germany, which is a product brand
of PSG
®
, Oakbrook Terrace, IL, USA, a Dover company. PSG
is comprised of several of the world’s leading pump brands,
including Abaque
®
, Almatec
®
, Blackmer
®
, Ebsray
®
, EnviroGear
®
,
Finder, Griswold
™
, Mouvex
®
, Neptune
™
, Quattroflow
™
, RedScrew
™
and Wilden
®
. You can find more information on Quattroflow at
quattroflow.com and on PSG at psgdover.com.
Quattrolfow
™
offers a complete family of stainless-steel quaternary diaphragm pumps
for use in biopharmaceutical manufacturing, many of which can also be outfitted with
disposable plastic wetted parts and pump heads.
quattroflow.com
PSG
ALMATEC Maschinenbau GmbH
Carl-Friedrich-Gauß-Straße 5
47475 Kamp-Lintfort, Germany
O: +49/2842/961-0 / F: +49/2842/961-40
