
Recommended methods for the
Identification and Analysis of
Cocaine in Seized Materials
Manual for use by national drug analysis laboratories

Photo credits:
UNODC Photo Library; UNODC/Ioulia Kondratovitch; Alessandro Scotti.

Laboratory and Scientific Section
UNITED NATIONS OFFICE ON DRUGS AND CRIME
Vienna
Recommended Methods for the
Identification and Analysis of
Cocaine in Seized Materials
(Revised and updated)
MANUAL FOR USE BY
NATIONAL DRUG ANALYSIS LABORATORIES
UNITED NATIONS
New York, 2012

ii
Note
Operating and experimental conditions are reproduced from the original reference
materials, including unpublished methods, validated and used in selected national
laboratories as per the list of references. A number of alternative conditions and
substitution of named commercial products may provide comparable results in many
cases, but any modification has to be validated before it is integrated into laboratory
routines.
Mention of names of firms and commercial products does not imply the endorse-
ment of the United Nations.
Original language: English
© United Nations, March 2012. All rights reserved.
The designations employed and the presentation of material in this publication do
not imply the expression of any opinion whatsoever on the part of the Secretariat
of the United Nations concerning the legal status of any country, territory, city or
area, or of its authorities, or concerning the delimitation of its frontiers or
boundaries.
This publication has not been formally edited.
Publishing production: English, Publishing and Library Section, United Nations
Office at Vienna.
ST/NAR/7/REV.1
iii
Contents
Page
1. Introduction ................................................. 1
1.1 Background .............................................. 1
1.2 Purpose and use of the manual .............................. 1
2. Physical appearance and chemical characteristics of coca leaf and
illicit materials containing cocaine ................................. 3
3. Description of the pure compounds ................................ 5
4. Illicit production of cocaine ...................................... 13
4.1 Production from coca leaves ................................ 13
4.2 Chemical synthesis of cocaine ............................... 15
5. Qualitative and quantitative analysis of materials containing cocaine ..... 17
5.1 Sampling ................................................ 17
5.2 Analysis of coca leaf ...................................... 18
5.2.1 Physical identification ............................... 18
5.2.2 Chemical analysis of coca leaf (whole or powdered) ..... 18
5.3 Analysis of coca paste and cocaine ........................... 20
5.3.1 Presumptive tests for cocaine ......................... 20
5.3.1.1 Colour test ................................ 20
5.3.1.2 Odour test ................................. 21
5.3.1.3 Microcrystal tests ........................... 22
5.3.1.4 Solubility tests ............................. 24
5.3.1.5 Anion tests ................................ 24
5.3.2 Thin Layer Chromatography (TLC) ................... 26
5.3.3 Gas Chromatography-Mass Spectrometry (GC-MS) ...... 29
5.3.4 Gas Chromatography (GC) with flame ionization detection
(GC-FID) ......................................... 31
5.3.5 High Performance Liquid Chromatography (HPLC) ...... 33
5.3.6 Fourier Transform Infrared (FTIR) Spectroscopy ......... 35
5.3.7 Ultraviolet (UV) Spectrophotometry ................... 35
5.4 The analysis of cocaine enantiomers .......................... 36
6. References ................................................. 39
iv
Acknowledgements
UNODC’s Laboratory and Scientific Section (LSS, headed by Dr. Justice Tettey)
wishes to express its appreciation and thanks to Dr. Michael Collins, Dr. Helen
Salouros and Mr. Hilton Swan for the preparation of the first draft of the present
revised and updated manual.
LSS would also like to thank Prof. Niamh Nic Daeid, Dr. Kathleen Savage, Dr. Udo
Zerell and Dr. Pierre Esseiva for their expert reviews and valuable contributions.
The preparation of the present manual was coordinated by Ms. Yen Ling Wong,
staff of LSS. The contribution of other UNODC staff is gratefully acknowledged.
1
1. Introduction
1.1 Background
Cocaine is a highly addictive stimulant that occurs naturally as an alkaloid of the
coca plant (Erythroxylon coca or Erythroxylon novogranatense). Traditionally, the
coca leaves are chewed or brewed as tea. In the early 1900s, purified cocaine was
used in most tonics and elixirs to treat a wide variety of illnesses.
Due to its high potential for abuse and dependence, coca leaf and cocaine were put
under international control in the Schedule I of the Single Convention on Narcotics
Drugs, 1961. However, in some countries, cocaine still has legitimate medical uses,
such as local anaesthesia for eye, ear and throat surgeries.
The two main chemical forms of cocaine are the water-soluble salt form and the
water-insoluble cocaine base form. Generally, the salt form can be injected or snorted
while the base form (“crack”) is usually smoked.
With excessive or prolonged abuse, cocaine can cause development of tolerance,
strong psychological dependence, malnutrition, disorientation, hallucination and
paranoid psychosis.
1.2 Purpose and use of the manual
The present manual is one in a series of similar publications dealing with the
identification and analysis of various types of drugs under international control.
These manuals are the outcome of a programme pursued by UNODC since the early
1980s, aimed at the harmonization and establishment of recommended methods of
analysis for national drug analysis laboratories.
The present manual is a revision of the manual on Recommended methods for testing
cocaine (ST/NAR/7), which was published in 1986. It has been prepared taking into
account developments in analytical technology with a view to providing the basis
for reliable forensic evidence on cocaine-containing seized materials.
In line with the overall objective of the series, the present manual suggests approaches
that may assist drug analysts in the selection of methods appropriate to the sample

2 Recommended Methods for the Identification and Analysis of Cocaine in Seized Materials
under examination and provide data suitable for the purpose at hand, leaving room
also for adaptation to the level of sophistication of different laboratories and the
various legal needs. The majority of methods included in the present manual are
validated methods, which have been used for a number of years in reputable labo-
ratories and as part of inter-laboratory studies, collaborative exercises and proficiency
tests. The reader should be aware, however, that there are a number of other methods,
including those published in the forensic science literature, which may also produce
acceptable results. Any new method that is about to be used in the reader’s labora-
tory must be validated and/or verified prior to routine use.
In addition, there are a number of more sophisticated approaches, but they may not
be necessary for routine operational applications. Therefore, the methods described
here should be understood as guidance, that is, minor modifications to suit local
circumstances should not normally change the validity of the results. The choice of
the methodology and approach to analysis as well as the decision whether or not
additional methods are required remain with the analyst and may also be dependent
on the availability of appropriate instrumentation and the level of legally acceptable
proof in the jurisdiction within which the analyst works.
Attention is also drawn to the vital importance of the availability to drug analysts
of reference materials and books on drugs of abuse and analytical techniques. Moreo-
ver, the analyst must of necessity keep abreast of current trends in drug analysis,
consistently following current analytical and forensic science literature.
UNODC’s Laboratory and Scientific Section would welcome observations on the
contents and usefulness of the present manual. Comments and suggestions may be
addressed to:
Laboratory and Scientific Section
United Nations Office on Drugs and Crime
Vienna International Centre
P.O. Box 500
1400 Vienna
Austria
Fax: (+43-1) 26060-5967
E-mail: [email protected]
All manuals, as well as guidelines and other scientific-technical publications may
be requested by contacting the address above.
3
2. Physical appearance and chemical
characteristics of coca leaf and illicit
materials containing cocaine
Coca leaf
Coca leaves are somewhat similar in appearance to Laurus nobilis leaves. Different
Erythroxylon species produce leaves varying in size and appearance. In all species
the upper side of the leaf is darker than the underside which may be grey-green in
colour. On the underside of the leaves are found two lines parallel to the midrib
which are considered to be characteristic of coca leaf.
The leaves of Erythroxylon coca Lam. are characteristically large and thick, broadly
elliptic in shape, more or less pointed at the apex and dark green in colour. The
leaves of Erythroxylon novogranatense (Morris) Hieron are smaller, narrower, thin-
ner and rounded at the apex. They are bright yellow-green coloured. The leaves of
Erythroxylon novograntense var. truxillense (Rusby) Plowman are even smaller and
narrower. However, they are thicker then the other types and have a rich green
colour.
Coca paste
This is an off-white, creamy or beige-coloured powder; it is rarely fine, often con-
tains aggregates and is generally damp. Unless the aggregates are crystalline (which
is rare) they usually break down under slight pressure. It has a characteristic odour.
Cocaine
Although produced from a somewhat variable natural product, by a batch process
capable of wide variation, cocaine varies comparatively little when compared for
example to heroin products. Nevertheless no two illicit samples of cocaine are
exactly identical. For the most part it is a white or off-white crystalline powder
which is often fine, and rarely damp.

4 Recommended Methods for the Identification and Analysis of Cocaine in Seized Materials
Adulteration is comparatively rare (but not unknown) for material being internation-
ally trafficked with a purity often of 80-90% (as cocaine hydrochloride). Subsequent
adulteration and transformation for trafficking purposes usually involves the addition
of uncontrolled substances such as levamisole (and/or tetramisole), phenacetin, lido-
caine, caffeine, diltiazam, hydroxyzine, procaine, benzocaine or sugars (e.g. man-
nitol, lactose or glucose). In either case the physical appearance is changed only
slightly, since all the known adulterants are themselves fine dry white powders.
For trafficking within countries, the cocaine purity is typically about 30%; the inter-
nationally trafficked material is subjected to adulteration with about three times its
own weight of the diluent.
“Crack” cocaine
A flaky, hard material obtained by adding ammonia or sodium bicarbonate (baking
soda) and water to cocaine hydrochloride and heating the resulting precipitated
powder. The term “crack” which is the street name given to freebase cocaine, refers
to the crackling sound produced when the mixture is heated.
Deviations/variations of materials submitted for forensic examination to the physical
characteristics described here, should not be interpreted to mean the absence of
cocaine or a cocaine containing product.
5
3. Description of the pure compounds
The compounds listed below include cocaine, major components (> 1% by weight)
and minor components (usually < 1% by weight). Trace components (usually < 0.1%
by weight and typically requiring an extraction step) are not described here.
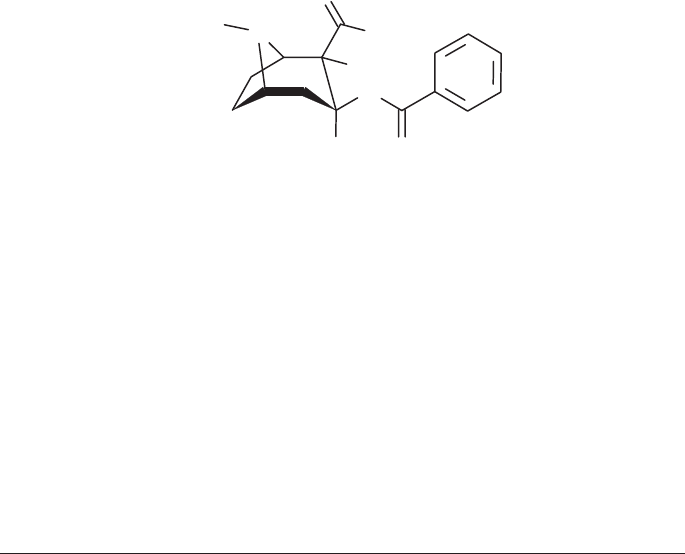
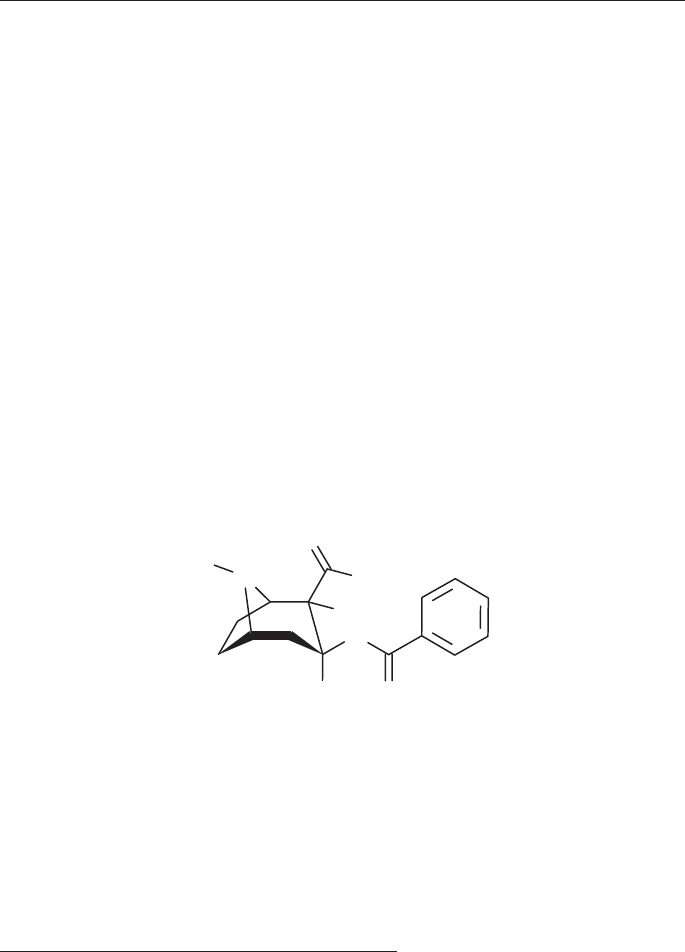
Cocaine
Synonyms: [1R-(exo,exo)]-3-(Benzoyloxy)-8-methyl-8-azabicyclo[3.2.1]octane-
2-carboxylic acid methyl ester
3β-Hydroxy-1αH,5αH-tropane-2β-carboxylic acid methyl ester benzoate
Ecgonine methyl ester benzoate
l-Cocaine
β-Cocaine
Benzoylmethylecgonine
C
17
H
21
NO
4
Molecular Weight = 303.4 (base), 339.8 (hydrochloride)
Melting point: 98° C (base), 195° C (hydrochloride)
Solubilities (1g/ml): Base Hydrochloride
Water slightly soluble (1 in 600) soluble (1 in 0.4)
Ethanol soluble (1 in 6.5) soluble (1 in 3.2)
Diethyl ether soluble (1 in 3.5) practically insoluble
Chloroform soluble (1 in 0.7) soluble (1 in 12.5)
O
O
N
O
OCH
3
H
3
C
H
H
6 Recommended Methods for the Identification and Analysis of Cocaine in Seized Materials
GC-MS data (percentage abundance):
303 (M
+
, 17), 182 (71), 105 (29), 96 (24), 94 (36), 82 (100), 77 (35) m/z
NMR data (hydrochloride):
1
H NMR 600 MHz; (D
2
O): δ 2.90 (3H, s), 3.63 (3H, s), 3.65 (1H, dd,), 4.10 (1H,
bm), 4.24 (1H, bm), 5.59 (1H, ddd), 7.54, (1H, t), 7.70 (1H, t), 7.96 (1H, d) ppm
13
C NMR (151 MHz; D
2
O): δ 22.8, 23.9, 32.8, 39.1, 46.3, 53.6, 63.4, 64.1, 64.7,
128.7, 129.2, 129.8, 134.7, 167.5, 173.6 ppm
Infrared data:
Principal peaks at wavenumbers 1710, 1738, 1275, 1110, 712, 1037 cm
-1
(KBr disk).
UV Data:
Aqueous acid—233 nm (A¦ = 430), 275 nm
Major and minor components
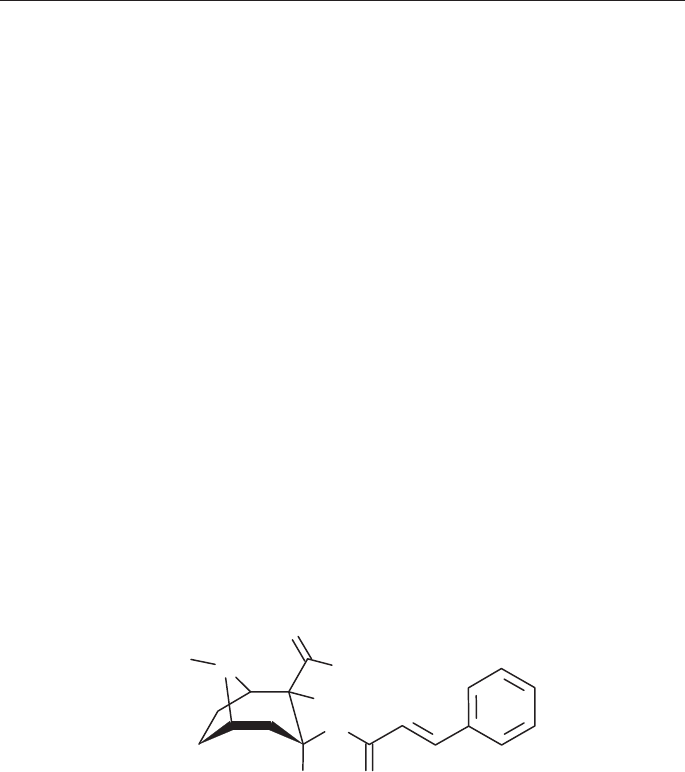
Cinnamoylcocaine
Synonyms: [1R-(exo,exo)]-8-Methyl-3-[(1-oxo-3-phenyl-2-propenyl)oxy]-8-azabi-
cyclo[3.2.1]octane-2-carboxylic acid methyl ester
Ecgonine cinnamate methyl ester
Cinnamoylecgoninmethyl ester
Cinnamoylmethylecgonine
Cinnamylcocaine
C
19
H
23
NO
4
Molecular Weight: 329.4
Melting point: 121° C (base)
O
O
N
O
OCH
3
H
3
C
H
H

Recommended Methods for the Identification and Analysis of Cocaine in Seized Materials 7
Solubilities (1g/ml): Base Hydrochloride
Water almost insoluble soluble
Ethanol soluble soluble
Diethyl ether soluble soluble
Chloroform soluble slightly
GC-MS data (percentage abundance):
329 (M
+
, 15), 238 (14), 182 (72), 131 (33), 103 (24), 96 (59), 94 (35), 82 (100),
42 (27) m/z
NMR data (hydrochloride):
1
H NMR (300 MHz; CDCl
3
): (Key spectral data): δ 2.21 (3H, s), 2.40 (1H, ddd),
3.71 (3H, s), 5.11 (1H, ddd), 6.44 (1H, d), 7.36 (3H, m), 7.51 (2H, m), 7.65
(1H, d) ppm
13
C NMR (75.5 MHz; CDCl
3
): δ 25.2, 25.4, 35.5, 41.2, 50.1, 51.4, 61.6, 64.8, 66.6,
118.3, 128.1 (x 2), 128.8 (x 2), 130.2, 134.4, 144.9, 166.7, 170.8 ppm
Infrared data:
Principal peaks at wavenumbers 2959, 2856, 2804, 1749, 1699, 1630, 1319, 1179,
1037, 1008, 767, 683 cm
-1
(KBr disk)
Methylecgonine
Synonyms: [1R-(exo,exo)]-3-Hydroxy-8-methyl-8-azabicyclo[3.2.1]octane-2-
carboxylic acid methyl ester
Ecgonine methyl ester
3β-Hydroxy-1αH,5αH-tropane-2β-carboxylic acid methyl ester
C
10
H
17
NO
3
Molecular Weight: 199.3 (base), 235.7 (hydrochloride)
Melting point: oil (base), 215° C (hydrochloride)
OH
N
O
OCH
3
H
3
C
H
H
8 Recommended Methods for the Identification and Analysis of Cocaine in Seized Materials
GC-MS data (percentage abundance):
199 (M
+
, 30), 168 (18), 112 (12), 96 (78), 94 (38), 82 (100), 68 (8), 42 (32) m/z
NMR data (hydrochloride):
1
H NMR (500 MHz; D
2
O): δ 2.03-2.14 (3H, m), 2.20-2.24 (1H, m), 2.30-2.48
(2H, m), 2.83 (3H, s), 3.31 (1H, dd, J = 2.2, 7.2 Hz), 3.80 (3H, s) 3.99 (1H, m),
4.15 (1H, bd, J = 7.0 Hz), 4.43-4.48 (1H, m) ppm
13
C NMR (125 MHz; D
2
O): δ 22.5, 23.5, 34.8, 38.4, 48.8, 52.8, 60.3, 63.1, 63.8,
174.2 ppm
Infrared data:
Principal peaks at wavenumbers 3269, 2963, 2132, 1704, 1481, 1428, 1350, 1215,
1140, 1049, 1013, 968, 777, 616 cm
-1
(KBr disk).
Benzoylecgonine
Synonyms: 3-(Benzoyloxy)-8-methyl-8-azabicyclo[3.2.1]octane-2-carboxylic acid
3β-Hydroxy-1αH,5αH-tropane-2β-carboxylic acid benzoate
Ecgonine benzoate
C
16
H
19
NO
4
Molecular Weight: 289.3
Melting point: 195° C (anhydrous) (decomposes), 86-92° C (tetrahydrate), 200° C
hydrochloride
Solubilities (1g/ml): Base Hydrochloride
Water, boiling soluble soluble
Ethanol soluble soluble
GC-MS data (percentage abundance):
289 (M
+
, 5), 168 (26), 124 (100), 105 (31), 96 (19), 94 (26), 82 (61), 77 (40), 67
(11) m/z
O
O
N
O
OH
H
3
C
H
H

Recommended Methods for the Identification and Analysis of Cocaine in Seized Materials 9
NMR data (hydrochloride):
1
H NMR (300 MHz; D
2
O): (Key spectral data): δ 2.61-2.17 (6H, m) 2.88 (3H, s),
3.22 (1H, dd), 4.07 (2H, bd), 5.54 (1H, m), 7.59 (2H, dd), 7.76 (1H, dd), 8.06 (2H,
d) ppm
13
C NMR (75.5 MHz; D
2
O): δ 23.2, 32.6, 37.6, 48.8, 62.3, 64.8, 128.7, 128.9, 129.5,
133.9, 167.2, 176.9 ppm
Infrared data:
Principal peaks at wavenumbers 1275, 1720, 1618, 717, 1116, 1316 cm
-1
UV Data:
Aqueous acid—234 nm (A¦ = 376), 274 nm
Ecgonine
Synonyms: [1R-(exo,exo)]-3-Hydroxy-8-methyl-8-azabicyclo[3.2.1]octane-2-
carboxylic acid
3β-Hydroxy-1αH,5αH-tropane-2β-carboxylic acid
C
9
H
15
NO
3
Molecular Weight: 185.2 (base), 221.7 (hydrochloride)
Melting point: 198° C (base), 246° C (hydrochloride)
Solubilities (1g/ml): Base Hydrochloride
Water soluble soluble
Ethanol slightly soluble slightly soluble
Diethyl ether sparingly soluble
Chloroform sparingly soluble
GC-MS data (percentage abundance):
185 (M
+
, 9), 124 (33), 96 (82), 82 (100), 57 (54), 42 (89) m/z
OH
N
O
OH
H
3
C
H
H
10 Recommended Methods for the Identification and Analysis of Cocaine in Seized Materials
NMR data (hydrochloride):
1
H NMR (600 MHz; D
2
O): δ 1.98-2.19 (4H, m), 2.25-2.41 (2H, m), 2.78 (3H, s),
3.18 (1H, dd, J = 2.3, 7.1 Hz), 3.92 (1H, m), 4.10 (1H, d, J = 7.3 Hz), 4.41 (1H,
m) ppm
13
C NMR (150 MHz; D
2
O): δ 23.2, 24.0, 35.5, 38.9, 49.5, 60.8, 63.6, 64.6,
176.4 ppm
Infrared data:
Principle peaks at wavenumbers 1688, 1210, 1200, 1223, 1134, 1179 cm
-1
(ecgonine
hydrochloride, KBr disk)
UV Data:
Ethanol—275 nm
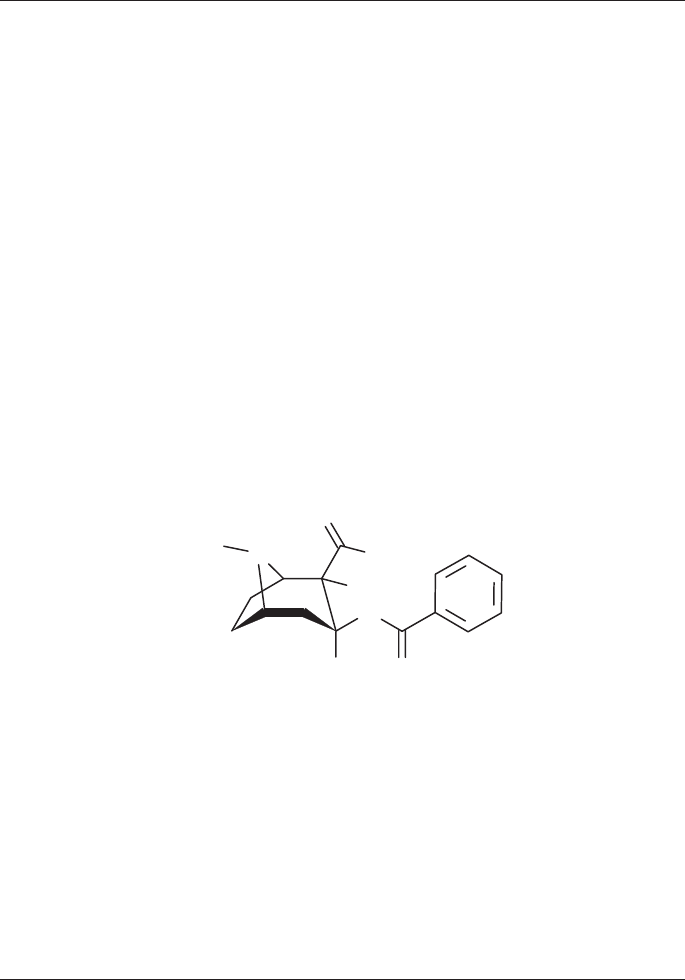
Norcocaine
Synonyms: 1R-(exo,exo)-3-(Benzoyloxy)-8-azabicyclo[3.2.1]octane-2-carboxylic
acid methyl ester
C
16
H
19
NO
4
Molecular Weight: 289.3 (base), 325.8 (hydrochloride)
Melting point: 115-116° C (hydrochloride)
Solubilities (1g/ml): Hydrochloride
Water soluble
Ethanol slightly soluble
GC-MS data (percentage abundance):
289 (M
+
, 11), 168 (100), 136 (37), 108 (25), 105 (23), 82 (13), 80 (23), 77 (33),
68 (41) m/z
O
O
N
O
OCH
3
H
H
H
Recommended Methods for the Identification and Analysis of Cocaine in Seized Materials 11
NMR data (hydrochloride):
1
H NMR (500 MHz; D
2
O): (Key spectral data): δ 3.59 (1H, dd), 3.64 (3H, s), 4.38
(1H, bd), 5.56 (1H, ddd), 7.54 (2H, t), 7.71 (1H, t), 7.95 (1H, d) ppm
13
C NMR (75.5 MHz; CDCl
3
): δ 24.4, 25.1, 31.1, 44.9, 53.0, 54.3, 55.4, 65.0, 128.4,
128.8, 129.4, 134.3, 167.0, 173.0 ppm
Infrared data:
Principle peaks at wavenumbers 3597, 3408, 3152, 2951, 2772, 2744, 2527, 1721,
1440, 1350, 1275, 717 cm
-1
Cinnamic acid (trans-)
Synonyms: 3-phenyl-2-propenoic acid
β-phenylacrylic acid
C
9
H
8
O
2
Molecular Weight: 148.2
Melting point: 133° C (base)
Solubilities (1g/ml): Base
Water slightly soluble
Ethanol soluble
Diethyl ether soluble
Chloroform soluble
GC-MS data (percentage abundance):
148 (M
+
, 74), 148 (100), 147 (100), 131 (22), 103 (61), 77 (47), 51 (40) m/z
UV Data:
Ethanol—273 nm
OH
O
12 Recommended Methods for the Identification and Analysis of Cocaine in Seized Materials
N-Formylnorcocaine
Synonyms: [1R-(exo,exo)]-3-(Benzoyloxy)-8-formyl-8-azabicyclo[3.2.1]octane-2-
carboxylic acid methyl ester
N-Formylcocaine
C
17
H
19
NO
5
Molecular Weight: 317.3
GC-MS data (percentage abundance):
289 (38), 195 (39), 168 (100), 136 (42), 105 (94), 77 (58), 68 (48) m/z.
NMR data:
1
H NMR (600 MHz; CDCl
3
): Double resonances observed due to restricted rotation
about the amide bond. Rotamers present in ca. 1:1 ratio at room temperature. Key
spectral data:
Rotamer A δ 2.37 (1H, ddd), 3.25 (1H, bdd,), 3.65 (3H, s), 4.30 (1H, bd), 4.81
(1H, m), 5.53 (1H, ddd,), 8.02 (0.5H, s) ppm
Rotamer B δ 2.53 (1H, ddd), 3.17 (1H, bdd), 3.68 (3H, s), 4.27 (1H, m), 4.95
(1H, bd), 5.49 (1H, ddd), 8.16 (1H, s) ppm
13
C NMR (CDCl
3
): δ 26.9, 27.4, 27.9, 28.4, 33.4, 35.4, 48.5, 48.7, 49.3, 51.1, 51.9,
52.0, 53.6, 55.5, 66.2, 66.3, 128.4, 129.6, 129.7, 133.3, 157.8, 158.0, 165.6, 165.7,
169.6, 170.0 ppm
For additional details related to the substances, the reader is referred to the
Multilingual Dictionary of Narcotic Drugs and Psychotropic Substances Under
International Control (http://www.unodc.org/unodc/en/scientists/multilingual- dictionary-
of-narcotic-drugs-and-psychotropic-substances-under-international-control.html.
html), the widely used Merck Index [1] and Clarke’s Analysis of Drugs and
Poisons [2].
O
O
N
O
OCH
3
H
H
H
O
13
4. Illicit production of cocaine
4.1 Production from coca leaves
The production of illicit cocaine can be achieved in a number of ways. The details
outlined here represent one of the approaches to the illicit production of cocaine [3].
In illicit production, variation of technique, reagents and quantities are to be expected.
The first step involves the extraction of the crude coca paste from the coca leaf.
The coca leaves are stripped from the plant. The fresh or dried leaves are mixed
with water and lime. The alkaline mixture is crushed and kerosene, or some other
hydrocarbon, is added so as to extract the cocaine from the leaves. The kerosene
contains the coca alkaloids and may additionally contain waxy material from the
leaves. This waxy material can be removed by heating then cooling the kerosene
mixture which results in solidification of the unwanted wax. The kerosene is
separated from the coca leaves and wax.
The kerosene is then back-extracted with dilute acidified water (such as sulphuric acid).
The acid converts the cocaine free base to cocaine sulphate and this process extracts the
alkaloids into the aqueous layer. At this point the kerosene is discarded and the aqueous
layer is made alkaline with lime or ammonia. This converts the cocaine sulphate back
to the free-base, which results in precipitation of crude cocaine, the more basic alkaloids
as well as inorganic salts. The product is then filtered and dried to give coca paste.
Another technique employed in the extraction of the crude coca paste from the coca
leaf is the acid extraction method. The coca leaves are directly extracted with dilute
sulphuric acid which converts the cocaine free base to cocaine sulphate. The mixture
is filtered and to the aqueous layer is then added excess lime or carbonate resulting
in precipitation of crude coca paste. The coca paste is then back extracted with
kerosene and the kerosene layer is treated as mentioned above in the earlier method.
The second step in the production of illicit cocaine is the purification of the coca
paste to cocaine base. The coca paste is dissolved in dilute sulphuric acid. The solu-
tion has a yellow-brown colour and is treated with potassium permanganate. The
potassium permanganate is slowly added until the solution turns from a yellow-brown
colour to a colourless liquid. The purpose of the addition of potassium permanganate
is to oxidise the cinnamoylcocaine isomers present in the cocaine. The oxidation
process also makes the cocaine whiter in appearance. The solution is filtered, and

14 Recommended Methods for the Identification and Analysis of Cocaine in Seized Materials
the filtrate is made basic with ammonia, resulting in precipitation of cocaine base
and other alkaloids. The cocaine base is filtered, washed with water and dried.
The final stage of the production involves the conversion of the crude cocaine base
to cocaine hydrochloride. The cocaine base is dissolved in diethyl ether. The solu-
tion is filtered and concentrated hydrochloric acid and acetone are added, resulting
is precipitation of cocaine hydrochloride. The cocaine hydrochloride is then filtered
and dried.
Figure 1. Flow chart showing the illicit production of cocaine from coca leaves
Filtered
Cocaine hydrochloride
Filtrate
(discarded)
Coca leaves
(+ H
2
O + lime + kerosene)
Crushed and stirred
Extracted coca leaf
(discarded)
Kerosene
(+ acidified H
2
O)
Layers separated
Kerosene layer
(discarded)
Solid waste
(discarded)
Acidified H
2
O layer
(+ lime or NH
3
)
Aqueous filtrate
(+ NH
3
)
Filtered
Filtered
Filtered
Coca paste
(+ dilute sulphuric acid
+ potassium permanganate)
Cocaine base
(+ solvent [e.g. ether,
acetone, etc]
+ hydrochloric acid)
Aqueous filtrate
(discarded)
Aqueous filtrate
(discarded)
Recommended Methods for the Identification and Analysis of Cocaine in Seized Materials 15
4.2 Chemical synthesis of cocaine
One of the routes for synthesizing cocaine from the starting material, methyl ecgo-
nine hydrochloride is described as follows [4]:
Step 1. Conversion of methyl ecgonine hydrochloride to methyl ecgonine base
Methyl ecgonine hydrochloride (5.0 g) is dissolved in aqueous NaHCO
3
solution
(10%, 100 ml) and extracted with chloroform (5 x 50 ml). The chloroform layer is
washed with brine (2 x 50 ml), dried over anhydrous Na
2
SO
4
, and concentrated
under reduced pressure to yield an oil. The oil is then dried under high vacuum for
2 hours to yield methyl ecgonine as an oil (3.62 g, 85%).
Step 2. Benzoylation of methyl ecgonine to cocaine base
Methyl ecgonine base (3.62 g) is dissolved in dry benzene (200 ml) and cooled in
an ice bath. Benzoyl chloride (3.05 g) in dry benzene (50 ml) is added slowly to
the methyl ecgonine solution and the mixture is refluxed for 16 hours. The reaction
mixture becomes more viscous and white crystals of cocaine HCl are observed to
precipitate. The mixture is cooled and extracted with 1M aqueous HCl solution
(3 x 50 ml). The combined aqueous HCl extract is then basified with aqueous
ammonia solution and the resulting white precipitate of cocaine base is filtered, and
washed with water. The cocaine base is dissolved in diethyl ether, washed with
water (1 x 50 ml), brine (1 x 50 ml), dried (Na
2
SO
4
), filtered and concentrated under
reduced pressure to a white solid. The solid is purified by flash column chromatog-
raphy (eluting with CHCl
3
/EtOAc 80/20) to yield cocaine base (2.23 g, 41% yield).
The cocaine base is recrystallized from n-hexane to give white needle like crystals.
The crystals are then ground and dried under high vacuum at 40° C to give cocaine
base (1.57 g, 29% yield).
N
H
3
C
OMe
O
C
10
H
17
NO
3
Exac tMass: 199. 12
N
H
3
C
OMe
O
C
10
H
18
ClNO
3
+
ExactMass: 23 5.10
NaHC O
3
.HC l
OH
OH
N
H
3
C
N
H
3
C
Benzene
O
Cl
O
O
+
OMe
O
OMe
O
C
10
H
17
NO
3
ExactMass: 199. 12
C
7
H
5
ClO
ExactMass: 14 0.00
C
17H21 NO 4
ExactMass: 303.15
OH
17
5. Qualitative and quantitative
analysis of materials
containing cocaine
Generally, in attempting to establish the identity of a controlled drug in suspect
material, the analytical approach must entail the determination of at least two
uncorrelated parameters, one of which should provide information on the
chemical structure of the analyte (for example, IR, MS; or tandem methods such
as GC-MS).
It is recognized that the selection of these parameters in any particular case would
take into account the drug involved and the laboratory resources available to the
analyst. It is also accepted that unique requirements in different jurisdictions may
dictate the actual practices followed by a particular laboratory.
5.1 Sampling
The principal reason for a sampling procedure is to permit an accurate and meaningful
chemical analysis. Because most methods—qualitative and quantitative—used in
forensic drug analysis laboratories require very small aliquots of material, it is vital
that these small aliquots be representative of the bulk from which they have been
drawn. Sampling should conform to the principles of analytical chemistry, as laid
down, for example, in national pharmacopoeias or by regional or international
organizations. For general aspects of qualitative sampling of multi-unit samples,
refer to the Guidelines on Representative Drug Sampling (http://www.unodc.org/
unodc/en/ scientists/publications_manuals.html). For seized material with obvious
external characteristics, a sampling method based on the Bayes’ model may be
preferred over the hypergeometric approach.
The use of an approved sampling system also helps to preserve valuable resources
and time by reducing the number of determinations needed. It is recognized that
there may be situations where, for legal reasons, the normal rules of sampling and
homogenization cannot be followed.

18 Recommended Methods for the Identification and Analysis of Cocaine in Seized Materials
5.2 Analysis of coca leaf
Coca leaf, being a vegetable product, requires a different analytical approach to that
to be applied to the extracted material, whether it be impure coca paste or the purer
cocaine. The sampling methods may be used on seizures of coca leaf, provided the
analyst varies the sampling procedure to allow for the different physical makeup of
leaf material as opposed to powder.
Trafficking in coca leaf is rare (but not unknown) outside those countries where
coca is grown. This section has therefore been included in the manual to assist the
analyst on those rare occasions when he or she may be required to deal with this
material.
The identification of both coca leaf and powdered coca leaf material should be by
a two part process—botanical and chemical. Ideally the analyst should be trained
in both botany and chemistry and should have appropriate reference materials for
both techniques.
5.2.1 Physical identification
(i) Whole coca leaf
This is described in section 2. Confirmation should include microscopy.
(ii) Powdered coca leaf
This may be identified by microscopy. The standard textbooks dealing with powdered
vegetable drugs usually contain a section devoted to coca leaf (e.g. Trease and
Evans Pharmacognosy, 15th edition, W.C. Evans, W.B. Saunders, 2002, 349 and
529).
5.2.2 Chemical analysis of coca leaf (whole or powdered)
Short immersion in boiling ethanol produces effective extraction of ecgonine-type
alkaloids and minimizes the breakdown of cocaine. Extraction with hot methanol
has also been found to be effective.
If quantitative extraction of the alkaloids is not required, a short extraction at room
temperature may be sufficient. The leaves (preferably chopped or powdered) may
be pounded with ethanol or methanol in a mortar.
The alcoholic extract is subjected to TLC (with visualization by spray reagent) or
GC-MS for qualitative analysis of the coca leaf (see sections 5.3.2 and 5.3.3 for
detailed procedures).

Recommended Methods for the Identification and Analysis of Cocaine in Seized Materials 19
For quantitative analysis, a systematic extraction procedure follows the scheme illus-
trated below (figure 2) [5]. The resulting extract may be subjected to GC or HPLC
analysis to estimate the cocaine/alkaloidal content of the coca leaf (see sections
5.3.4 and 5.3.5 for detailed procedures). Typical amounts of cocaine present in dried
coca leaf could be found in table 1.
Table 1. Typical amounts of cocaine present in three Erythroxylon varieties
Erythroxylon variety Typical cocaine content in dry leaf
coca var. ipadu 0.11-0.41%
novo var. novogranatense 0.17-0.76%
novo var. truxillense 0.42-1.02%
Figure 2. Flow chart showing the extraction procedures for the chemical
analysis of coca leaves
Manicured dried coca-leaves
(1 gram)
Discard chloroform layer
Discard extracted leaves Filtrate
Aqueous layer
Residue
Chloroform layer
GC or HPLC analysis
Discard aqueous layer
Add 40 ml 95% ethanol
Reflux 15 minutes
Cool
Filter
Evaporate
Add 20 ml chloroform
Extract with 2x10 ml 1.5% (w/v) citric acid
Adjust to pH 8.2 with NaCO
3
Extract with 2x20 ml chloroform
Dry over Na
2
SO
4
Evaporate
Add internal standard

20 Recommended Methods for the Identification and Analysis of Cocaine in Seized Materials
5.3 Analysis of coca paste and cocaine
Cocaine exhibits generally come in powder form and commonly contain adulterants
or cutting agents such as levamisole (and/or tetramisole) phenacetin, lidocaine, caf-
feine, diltiazem, hydroxyzine, procaine, benzocaine or sugars (e.g. mannitol, lactose
or glucose).
For cocaine containing exhibits, combinations of testing methods such as Scott’s
colour test, TLC, FTIR, GC coupled with FID and/or MS and HPLC are considered
to be appropriate tests for positive identification of cocaine. Recommended minimum
guidelines for method selection have been formulated by the Scientific Working
Group on Drugs (SWGDRUG) [8].
In cases where the cocaine is present in other matrices such as clothing, synthetic
resins or liquids it must be first extracted before these tests are applied.
5.3.1 Presumptive tests for cocaine
Presumptive tests are fast screening procedures that are designed to provide an
indication of the presence or absence of drug classes in the test sample and quickly
eliminate negative samples. Good presumptive test techniques, as all analytical tech-
niques, maximize the probability of a “true” result, and minimize the probability of
a false positive. However, presumptive tests are not considered sufficient for drug
identification and results must be confirmed by additional laboratory tests.
5.3.1.1 Colour test [9,10]
Colour reactions are produced by compounds with a particular chemical structure.
The colour obtained in any particular test may vary depending on the conditions of
the test, amount of substance present and extraneous material in the test sample.
Colour test reagents must be checked with known substances when prepared. A
blank should be run to preclude false positive results.
It must be stressed that positive results to colour tests are only a presumptive indi-
cation of the possible presence of cocaine. The colour tests for cocaine are especially
prone to produce false positives. Many other materials, often harmless and uncon-
trolled by national legislation or international treaties, may give similar colours with
the test reagents. A number of these are either other controlled drugs, often encoun-
tered as white powders (e.g. methaqualone), or the synthetic local anaesthetics which
are often substituted for cocaine in the illicit traffic. It is mandatory for analysts to
confirm such results by the use of alternative techniques.
The colour test described below is known as the Scott’s Test (modified Cobalt
Thiocyanate Test).

Recommended Methods for the Identification and Analysis of Cocaine in Seized Materials 21
Reagent 1: Dissolve 1.0 g cobalt thiocyanate in 50 ml of 10% (vol/vol) acetic
acid, then add 50 ml of glycerine
Reagent 2: Hydrochloric acid (concentrated)
Reagent 3: Chloroform
Method:
Step 1: Place a small amount (no more than 1 mg) of the suspected material in a
test tube. Add 5 drops of Reagent 1 and shake the test tube for 10 seconds.
Cocaine and related substances produce a blue precipitate and a blue
solution.
Step 2: Add a drop of Reagent 2 and shake the mixture for a few seconds. The
blue solution should turn pink. If the blue colour still does not change,
add one further drop. If still no change, repeat the test with a smaller
portion of suspected material.
Step 3: Add 5 drops of Reagent 3 and shake. If cocaine is present, the lower
chloroform layer will develop an intense blue colour, while the upper layer
will be pink.
Results:
A positive result at each stage is required in order to be considered as a positive
test for cocaine. Only a few non-controlled or controlled drugs will give a similar
colour sequence.
5.3.1.2 Odour test [11]
As is typical of many drugs, cocaine’s biological activity is not matched by a com-
parable degree of chemical reactivity. However, it is unique among commonly abused
drugs in being a benzoate ester. While colour tests for this functional group are not
available, the lower alkyl esters of benzoic acid have quite distinctive odours detect-
able at very low concentrations relative to the average colour test. The transfer of
the benzoate function of cocaine from methyl ecgonine to methanol is readily
accomplished in the presence of dry methanolic potassium or sodium hydroxide.
Analytical notes
• It is important to use less than 1 mg of sample.
• The amounts used of Reagents 1 and 3 are not critical. However, the ratio
of Reagent 1 to 2 is critical. If excess hydrochloric acid is added to Reagent
1 after the blue colour has developed with cocaine, a blue rather than pink
solution will result; this blue will not be extracted into the chloroform layer.
If excess cocaine is used in Step 1, then it is sometimes necessary to add
1-2 drops of hydrochloric acid; no more should be used.

22 Recommended Methods for the Identification and Analysis of Cocaine in Seized Materials
Evaporation of the excess methanol leaves a residue containing methyl benzoate
readily apparent by its odour.
Reagent:
Dissolve 1 g of potassium or sodium hydroxide in 20 ml of methanol to give
methanolic sodium or potassium hydroxide.
Method:
Thoroughly moisten dried test material with the reagent. After allowing excess alco-
hol to evaporate, compare the odour characteristic of the sample with that of standard
cocaine material.
5.3.1.3 Microcrystal tests [12]
Microcrystal tests are quick, simple and extremely sensitive test for the identification
of substances. They involve the formation of crystals from the reaction of the target
compound with a chemical reagent, followed by the analysis of the resulting crystals
by means of a polarizing microscope and comparison with reference material.
Platinic chloride test
Reagent: Dissolve 1 g of platinic chloride in 20 ml of distilled water.
Method:
Place 2 drops of sample solution (approximately 2-3 mg of the sample/5 drops of
10% hydrochloric acid) on a clean microscope slide. Then place 2 drops of the
reagent near the drops of the sample and use a glass rod to create a tiny channel
connecting the solutions. Observe the reaction and resulting crystals without a cover
slip at 100-200 magnification using a polarized microscope.
Analytical notes
• Over one hundred drugs were tested for positive interference and only
piperocaine (also a benzoate ester) gave a positive result. Certain amines
such as amphetamines will produce a “weak, fishy odour”.
• The sensitivity is greater than that of existing field tests, e.g. the Scott’s Test.
• The sample and reagent must be kept free of water which interferes with
the reaction.
• Ideally the test should be performed concomitantly with a test on standard
cocaine material and the odours compared.

Recommended Methods for the Identification and Analysis of Cocaine in Seized Materials 23
Results:
Cocaine forms V-shaped long, thin needles with branching.
Figure 3. Cocaine and platinic chloride, 100x
Source: Reprinted, with permission, from Journal of Forensic Sciences, vol. 48, Issue 3, copyright ASTM
International, 100 Barr Harbor Drive, West Conshohocken, PA 19428.
Gold chloride test
Reagent: Dissolve 1 g of gold chloride in 20 ml of distilled water.
Method:
Place 2 drops of sample solution (approximately 2-3 mg of the sample/5 drops of
10% hydrochloric acid) on a clean microscope slide. Then place 2 drops of the
reagent near the drops of the sample and use a glass rod to create a tiny channel
connecting the solutions. Observe the reaction and resulting crystals without a cover
slip at 100-200 magnification using a polarized microscope.
Results:
Cocaine forms radiating clusters of fine needles with perpendicular branches.
Figure 4. Cocaine and gold chloride, 200x
Source: Reprinted, with permission, from Journal of Forensic Sciences, vol. 48, Issue 3, copyright ASTM
International, 100 Barr Harbor Drive, West Conshohocken, PA 19428.
20
Method:
Thoroughly moisten dried test material with the reagent. After allowing excess
alcohol to evaporate, compare the odour characteristic of the sample with that of
standard cocaine material.
5.3.1.3 Microcrystal tests [12]
Microcrystal tests are quick, simple and extremely sensitive test for the identification
of substances. They involve the formation of crystals from the reaction of the target
compound with a chemical reagent, followed by the analysis of the resulting crystals
by means of a polarizing microscope and comparison with reference material.
Platinic Chloride Test
Reagent: Dissolve 1 g of platinic chloride in 20 mL of distilled water.
Method:
Place two drops of sample solution (approximately 2-3 mg of the sample/5 drops of
10% hydrochloric acid) on a clean microscope slide. Then place 2 drops of the
reagent near the drops of the sample and use a glass rod to create a tiny channel
connecting the solutions. Observe the reaction and resulting crystals without a cover
slip at 100-200 magnification using a polarized microscope.
Results:
Cocaine forms V-shaped long, thin needles with branching.
Figure 3: Cocaine and platinic chloride, 100 (Reprinted, with permission, from
Journal of Forensic Sciences, Vol. 48, Issue 3, copyright ASTM International, 100
Barr Harbor Drive, West Conshohocken, PA 19428.)
Analytical notes
Over one hundred drugs were tested for positive interference and only piperocaine (also a
benzoate ester) gave a positive result. Certain amines such as amphetamines will produce
a “weak, fishy odour”.
The sensitivity is greater than that of existing field tests, e.g. the Scott’s Test.
The sample and reagent must be kept free of water which interferes with the reaction.
Ideally the test should be performed concomitantly with a test on standard cocaine
material and the odours compared.
21
Gold Chloride Test
Reagent: Dissolve 1 g of gold chloride in 20 mL of distilled water.
Method:
Place two drops of sample solution (approximately 2-3 mg of the sample/5 drops of
10% hydrochloric acid) on a clean microscope slide. Then place 2 drops of the
reagent near the drops of the sample and use a glass rod to create a tiny channel
connecting the solutions. Observe the reaction and resulting crystals without a cover
slip at 100-200 magnification using a polarized microscope.
Results:
Cocaine forms radiating clusters of fine needles with perpendicular branches.
Figure 4: Cocaine and gold chloride, 200 (Reprinted, with permission, from Journal
of Forensic Sciences, Vol. 48, Issue 3, copyright ASTM International, 100 Barr
Harbor Drive, West Conshohocken, PA 19428.)
5.3.1.4 Solubility Tests
If it is suspected that the material has cutting agents added to it, then a solubility test
is typically carried out. The solubility of a small amount of the material in water and
in ethanol may give an indication of the form in which the drug is present. Cocaine
hydrochloride is soluble in water and ethanol. Whereas, cocaine base is soluble in
ethanol and almost insoluble in water as are many adulterants. The presence and
amount of insoluble material may give some indication of the likely purity, as sugar
diluents are largely insoluble in ethanol. The insoluble material may be filtered, dried
and subjected to further testing by, for example IR spectroscopy.
Analytical notes
Standard cocaine should be analysed concomitantly.
The dilution of the test material or the hydrochloric acid may be varied to give the
optimum results.

24 Recommended Methods for the Identification and Analysis of Cocaine in Seized Materials
5.3.1.4 Solubility tests
If it is suspected that the material has cutting agents added to it, then a solubility
test is typically carried out. The solubility of a small amount of the material in
water and in ethanol may give an indication of the form in which the drug is present.
Cocaine hydrochloride is soluble in water and ethanol. Whereas, cocaine base is
soluble in ethanol and almost insoluble in water as are many adulterants. The pres-
ence and amount of insoluble material may give some indication of the likely purity,
as sugar diluents are largely insoluble in ethanol. The insoluble material may be
filtered, dried and subjected to further testing by, for example IR spectroscopy.
Method:
Step 1: Dissolve a portion (approximately 1 g) of the powder or material in
approximately 5 ml of distilled or deionized water. For small seizures of
0.1 g, 0.5 ml of water should be used.
Step 2: Dissolve a portion (approximately 1 g) of the powder or material in
approximately 5 ml of ethanol. For small seizures 0.1 g, 0.5 ml of ethanol
should be used. This will show the presence of any ethanol insolubles such
as carbohydrates. The carbohydrates have low solubility in ethanol.
5.3.1.5 Anion tests
Anion testing for forensic purposes typically makes use of solubilities combined
with selected reactions where results are determined by the presence or absence,
and solubility, of a precipitate.
The hydrochloride salt of cocaine is by far the most commonly encountered while
sulphates salts are rarely encountered in cocaine products with the exception of coca
Analytical notes
• Standard cocaine should be analysed concomitantly.
• The dilution of the test material or the hydrochloric acid may be varied to
give the optimum results.
Analytical notes
• This test is most useful when the sample size is large and a considerable
quantity of the powder can be used without seriously reducing the total
amount of exhibit that can be produced in court.
• It may be used on small seizures by reducing both the amount of test material
and solvent.

Recommended Methods for the Identification and Analysis of Cocaine in Seized Materials 25
paste. This test should be confirmed, if possible, by IR spectroscopy and/or X-ray
diffraction methods.
Chlorides—Silver nitrate test
Reagent: Dissolve 1.0 g of silver nitrate in 20 ml of distilled water (to result in
a 5% aqueous silver nitrate solution).
Method:
Dissolve a small amount of solid material in distilled water. Test pH with indicator
paper, and acidify if necessary with a few drops of nitric acid. Add 1-2 drops of
reagent and observe for precipitate. If a white or yellow precipitate develops, add
ammonia until alkaline.
Results:
Solutions of chlorides, when treated with silver nitrate solution, yield a white curdy
precipitate which is insoluble in nitric acid. After washing with water, the precipitate
is soluble in ammonia solution, from which it can be re-precipitated by the addition
of nitric acid.
Sulphates—Barium chloride test
Reagent: Dissolve 1.0 g of barium chloride in 10 ml of distilled water. (to result
in a 10% aqueous barium chloride solution).
Method:
Dissolve a small amount of material in distilled water, acidify with a few drops of
dilute hydrochloric acid and then add 1-2 drops of reagent.
Results:
Solutions of sulphates, when treated with barium chloride solution, yield a white
precipitate which is insoluble in hydrochloric acid.
Analytical notes
• Washing the precipitate with water before performing the test for dissolution
of the precipitate is critical to remove any soluble (non-precipitated) anion.
• A blank should be carried out concurrently to preclude false positive results.

26 Recommended Methods for the Identification and Analysis of Cocaine in Seized Materials
5.3.2 Thin Layer Chromatography (TLC)
TLC is a commonly used technique for the separation and identification of illicitly
manufactured drugs. It is inexpensive, rapid, sensitive (sub-milligram quantities of
analyte required), flexible in the selection of both the stationary and mobile phase
and amenable to a wide variety of substances, in base and salt form, ranging from
the most polar to non-polar materials.
TLC plates (stationary phases)
Coating: Silica gel G with layer thickness of 0.25 mm and containing an inert
indicator, which fluoresces under UV light wavelength 254 nm (silica gel GF254).
Typical plate sizes: 20 x 20 cm; 20 x 10 cm; 10 x 5 cm (the latter should be used
with the 10 cm side vertical with the TLC tank).
Plates prepared by the analyst must be activated before use by placing them into
an oven at 120° C for at least 10 to 30 minutes. Plates are then stored in a grease-
free desiccator over blue silica gel. Heat activation is not required for commercially
available coated plates.
Methods:
Solvent systems
Prepare developing solvent system (System A, B or C as shown in table 2) as
accurately as possible by use of pipettes, dispensers and measuring cylinders. Leave
the solvent system in the TLC tank for a time sufficient to allow vapour phase
saturation to be achieved prior to the analysis (with adsorbent paper-lined tanks,
this takes approximately 5 minutes).
Table 2. Developing solvent systems suitable for the analysis of cocaine
System Solvents
Solvent proportions
(by volume)
System A Chloroform
Dioxane
Ethyl acetate
Ammonia (29%)
25
60
10
5
System B Methanol
Ammonia (29%)
100
1.5
System C Cyclohexane
Toluene
Diethylamine
75
15
10

Recommended Methods for the Identification and Analysis of Cocaine in Seized Materials 27
Preparation of standard and sample solutions
Standard and sample solutions are prepared at a concentration of 1 mg per ml in
methanol. The form of cocaine standard used, i.e., salt or base, is unimportant as
on the TLC plates, compounds move as the free base.
Spotting and developing
Apply as separate spots 1 µl and 5 µl aliquots of sample solution, 2 µl of the standard
solutions and 2 µl of solvent (as a negative control) on the TLC plate. Spotting must
be done carefully to avoid damaging the surface of the plate.
Visualization/detection
The plates must be dried prior to visualization. The solvent can be allowed to
evaporate at room temperature or with a hot air blower. In the later case, care must
be exercised that no component of interest is thermally labile. It is important for
proper colour development that all traces of ammonia or other bases are removed
from the plate.
Visualization methods
A. UV light at 254 nm
Dark spots against a green background are observed.
B. Acidified potassium iodoplatinate reagent
Dissolve 0.25 g of platonic chloride and 5 g of potassium iodide in distilled
water and make up to 100 ml; add 2 ml of concentrated hydrochloric acid to
Analytical notes
• The starting point of the run i.e. the “spotting line” should be 2 cm from
the bottom of the plate.
• The spacing between applications of sample (spotting points) should be at
least 1 cm and spots should not be placed closer than 1.5 cm to the side
edge of the plate.
• To avoid diffuse spots during development, the size of the sample spot should
be as small as possible (2 mm) by applying solutions in aliquots rather than
a single discharge.
• Allow spots to dry and place plate into solvent-saturated tank (saturation of
the vapour phase is achieved by using solvent-saturated pads or filter paper
as lining of the tank).
• Remove plate from the development tank as soon as possible as the solvent
reaches the development line (10 cm from starting line) marked beforehand;
otherwise, diffused spots will occur.

28 Recommended Methods for the Identification and Analysis of Cocaine in Seized Materials
the resulting solution. Cocaine appears as a blue spot when the plate is sprayed
with the reagent.
C. Dragendorff’s reagent (Munier)
Solution 1: Dissolve 2 g of bismuth subnitrate in 25 ml of concentrated (glacial)
acetic acid and add 100 ml of distilled water.
Solution 2: Dissolve 40 g of potassium iodide in 100 ml of distilled water.
Mix 10 ml of solution 1, 10 ml of solution 2, 20 ml of concentrated (glacial)
acetic acid and 100 ml of distilled water to produce Dragendorff’s reagent.
Cocaine appears as an orange spot when the plate is sprayed with the
reagent.
Interpretation
After visualization, mark spots (e.g. by pencil) and calculate retardation factor (R
f
)
values.
R
f
=
Migration distance: from origin to centre of spot
Development distance: from origin to solvent front
Results
Compound
Developing System (R
f
x 100)
A B C
Cocaine 81 59 56
Ecgonine 0 84 0
Methylecgonine 61 65 44
Benzoylecgonine 0 25 0
Cinnamoylcocaine 83 59 51
Tetracaine 63 56 25
Benzocaine 77 80 11
Lidocaine 77 69 40-55 (s)*
Pethidine 61 49 69
Methaqualone 81 78 38
Methadone 75 31-45 (s)* 74
Procaine 61 55 8-16 (s)*
*(s): streak, not spot produced on the TLC plate.

Recommended Methods for the Identification and Analysis of Cocaine in Seized Materials 29
5.3.3 Gas Chromatography-Mass Spectrometry (GC-MS)
GC-MS is one of the most commonly used techniques for the identification of
forensic drug samples. As a hyphenated technique, it unifies the separation power
and sensitivity of a GC with the analyte specificity of a spectroscopic technique. It
can provide high specific spectral data on individual compounds in a complex
mixture without prior isolation.
Sample preparation and extraction procedure
Solid samples are pulverized and homogenized with a mortar and pestle. A suitable
quantity of sample material is extracted with an appropriate solvent (e.g., methanol,
chloroform or 1:1 methanol:chloroform) to approximately 1 mg/ml sample solution.
Some sample presentations and analytes may necessitate the use of other solvents
or solvent mixtures.
Preparation of standard solutions
Prepare a standard solution of cocaine at a concentration of 1 mg/ml with an appro-
priate solvent (e.g., methanol, chloroform or 1:1 methanol:chloroform).
Preparation of Internal Standard (IS) e.g. benzopinacolone (for Retention Locking
if required)
Dissolve 50 mg of 2,2,2-triphenylacetophenone (benzopinacolone) in 1l of an appro-
priate solvent (e.g. chloroform or 1:1 chloroform:methanol). Add an aliquot of the
internal standard to the sample/standard solution if retention time locking of the
analysis is required.
Analytical notes
• R
f
values are not always reproducible due to small changes in plate
composition and activation, in solvent systems, tank saturation or development
distance. Therefore, the R
f
values provided are indications of the chromato-
graphic behaviour of the substances listed.
• It is essential that reference standards be run simultaneously on the same
plate.
• For identification purposes, both the R
f
value and the colour of the spots
after spraying with the appropriate visualization reagents should always be
considered.

30 Recommended Methods for the Identification and Analysis of Cocaine in Seized Materials
Results:
Identification is accomplished by comparing the retention time and mass spectrum
of the analyte with that of a reference standard. All compounds identified by GC-MS
and reported by the analyst must be compared to a current mass spectrum of the
appropriate reference standard, preferably obtained from the same instrument, oper-
ated under the same conditions. Commercial mass spectral libraries or user generated
spectra should be used for reference purposes only.
GC retention times for cocaine and known adulterants using the above operating
conditions are as follows:
Compound GC RT (mins) Base, P1, P2, M
+
ions (m/z)
Benzocaine 7.59 120, 165M
+
, 92, 65
Phenacetin 7.97 108, 109, 179M
+
Caffeine 8.40 194M
+
, 109, 55, 67
Lignocaine 8.53 86, 58, 30, 234M
+
Levamisole 8.83 204M
+
, 148, 73, 101
Procaine 8.91 86, 99, 120, 236M
+
Cocaine 9.36 82, 182, 94, 303M
+
Benzopinacolone (IS) 11.11 243, 165, 105, 348M
+
Hydroxyzine 11.83 201, 165, 299, 374M
+
Diltiazem 12.36 58, 71, 73, 414M
+
GC-MS operating conditions
GC oven conditions: Column temperature initially set at 60° C and held
isothermal for 3 minutes immediately after injection
and ramped to 300° C at a rate of 40° C/min with a
final isotherm of 6 minutes.
Column: DB-5MS, HP-5MS, 30 m x 250 µm; d
f
0.25 µm.
Inlet: Mode: splitless (purge flow 60 ml/min at 0.5 min);
Temp: 240° C;
Carrier gas: Helium, 1 ml/min, constant flow.
Detector: Ionization mode: EI mode, 70 eV;
Transfer line temp: 300° C;
Ion source temp: 230° C.
MS parameters: Solvent delay 3.00 minutes;
Scan parameters: TIC,
Scan range: 40–450 amu at 1 scan/sec.
Note: The above conditions may be altered as long as appropriate validation is carried out.

Recommended Methods for the Identification and Analysis of Cocaine in Seized Materials 31
5.3.4 Gas Chromatography (GC) with flame ionization
detection (GC-FID)
The GC instrument of choice for routine analytical work is the narrow bore capillary
gas chromatograph, using columns with internal diameter between 0.2 and 0.32 mm.
The packed column technique is no longer included in this manual as GC systems
are now typically equipped with capillary columns. Information on packed column
techniques for the analysis of cocaine can be found in an earlier edition of this
manual.
The method provided below for the quantitative GC-FID analysis of cocaine does
not require derivatization and is described here for general use.
Single standard method without derivatization [13]
This method involves the preparation of only one cocaine standard solution in a
concentration similar to the anticipated concentration of the analyte.
Preparation of internal standard solution (IS)
Dissolve isopropylcocaine hydrochloride (IPC) in chloroform to give a concentration
of 1.0 mg/ml. The solution is stored at 4°
C when not in use. Solutions can be
stored for one year at 4° C without detectable degradation. The solution should be
warmed to room temperature before use. Each analysis requires 5 ml.
Preparation of standard solution
Accurately weigh 20 mg of cocaine (hydrochloride or base) standard and transfer
quantitatively into a 50 ml Erlenmeyer flask. Accurately transfer 5 ml of internal
standard solution to the flask followed by 20 ml of chloroform containing 50 µl of
diethylamine. Allow the solution to sit for 5 minutes and then inject 1-2 µl of the
solution into the gas chromatograph.
Preparation of sample solution
Obtain a representative sample from the seized material. Grind and homogenize to
a fine powder. Accurately weigh a quantity of seized material containing approxi-
mately 20 mg of cocaine hydrochloride and transfer quantitatively to a 50 ml Erlen-
meyer flask. Accurately transfer 5 ml of internal standard solution to the flask fol-
lowed by 20 ml of chloroform containing 50 µl of diethylamine. Allow the solution
to sit for 5 minutes and then inject 1-2 µl of the solution into the gas
chromatograph.

32 Recommended Methods for the Identification and Analysis of Cocaine in Seized Materials
Calculations
The percentage of cocaine (as base) in the sample can be calculated using the general
formula below:
Content (%) =
Wsampcalc
x 100
Wsampnom
where
Wsampcalc
=
PAR
sam
x W
std
= calculated weight of analyte in the sample
PAR
std
Wsampnom
= nominal amount of sample used in preparation of the sample solution
PARsam
=
Area of cocaine peak in sample solution
Area of internal standard peak in sample solution
PARstd
=
Area of cocaine peak in sample solution
Area of internal standard peak in sample solution
W
std
= amount of standard (as base) used in preparation of the standard solution
GC operating conditions
Detector: FID
Column: HP-1 or equivalent, 30 m length, 0.25 mm ID, 0.25 µm
film thickness
Carrier gas: Hydrogen 1.1 ml/min
Injector temp: 280° C
Detector temp: 280° C
Oven temp: 250° C
Injection volume: 2 µl
Split ratio: 25:1
Run time: 7 min

Recommended Methods for the Identification and Analysis of Cocaine in Seized Materials 33
Results
Elution order and the corresponding relative retention time with respect to cocaine
are as follows:
Compound Relative retention time (min)
Ecgonine methyl ester 0.48
Benzocaine 0.49
Acetaminophen 0.52
Caffeine 0.61
Lidocaine 0.65
Procaine 0.76
Cocaine 1.00
Isopropylcocaine (IS) 1.14
cis-Cinnamoylcocaine 1.36
trans-Cinnamoylcocaine 1.78
Benzoylecgonine 1.89
5.3.5 High Performance Liquid Chromatography (HPLC)
In addition to GC, HPLC is another major separation technique commonly used in
forensic drug analysis. For ease of sample preparation, best reproducibility and
detectability, reversed phase chromatography is recommended for the analysis of
cocaine. The most universal and versatile column is a bonded octadecyl silica column
(C18). Column length, diameter, particle size, pore size and carbon load should be
considered before final selection of the column.
Since there is a large variety of stationary and mobile phases available to the analyst,
one method for quantitative HPLC method is described below where it can be modi-
fied for best performance. All methods must be properly validated and/or verified
prior to routine use.
Analytical notes
• This method can be modified from a single to a multiple standard method
by sequentially diluting aliquots of a concentrated cocaine standard stock
solution with the internal standard solution using volumetric flasks.
• The use of a structurally related internal standard such as IPC maximizes
precision and accuracy. If they are not available, alternative internal standards
such as n-eicosane, n-tetracosane or tetraphenylethylene may be used. Selec-
tion of a suitable solvent will be required to ensure that both the internal
standard and the cocaine sample are fully dissolved.

34 Recommended Methods for the Identification and Analysis of Cocaine in Seized Materials
Method
Preparation of cocaine standard and sample solutions
Dissolve an appropriate amount of standard or sample in the mobile phase, targeting
a concentration of the cocaine between 0.05-0.50 mg/ml.
Stock and standard solutions must be prepared from reference standards. Working
standards should be within the linear range of the detector and approximately
80-120% of the target concentration. Multiple point calibration is desirable but a
single standard method is also acceptable.
Quantitation
External or internal standard calibration may be used. The use of peak area for
HPLC quantitation is recommended, because peak broadening (decreases in peak
height) may occur as a result of deterioration of the stationary phase, rendering peak
height unsuitable for quantitation.
Results
Elution order and the corresponding capacity factor (k) are as follows:
Compound Capacity factor (k)
Procaine 0
Lignocaine 0.79
Cocaine 2.68
Benzoylecgonine 5.68
cis-Cinnamolycocaine 6.30
Amylocaine 7.19
Butacaine 8.97
trans-Cinnamoylcocaine 10.65
Benzocaine 20.06
Chromatographic conditions
Column: C18 Hypersil (or equivalent), 160 mm x 5.0 mm ID
Mobile phase: methanol:water:1% phosphoric acid:n-hexylamine
(300:700:1000:14 by volume; pH = 2.5)
Flow rate: 2.0 ml/min (slower flow values should be considered with
shorter columns and smaller particle sizes)
Detection: UV at 230 nm
Injection volume: 5-20 µl

Recommended Methods for the Identification and Analysis of Cocaine in Seized Materials 35
5.3.6 Fourier Transform Infrared (FTIR) Spectroscopy
The confirmation of the identity of a substance can be achieved by FTIR. Unequivo-
cal identification of cocaine is thus possible from each unique spectrum. For powders,
considered from prior chromatographic analysis to be reasonably pure, the infrared
spectrum of the powder may be run directly in a KBr disc for comparison with
those of cocaine free bases or HCl salts.
Results
Major peaks occur at the following wavenumbers (cm
-1
) which are listed in order
of magnitude of absorbance. The sequence may, however, vary from sample to
sample.
Compound Principal peaks at wavenumbers
Cocaine base 1275, 1700, 1106, 1728, 710, 1040, 1280 cm
-1
Cocaine HCl 1712, 1730, 1276, 1230 (side peak), 732, 1106, 1075, 1025 cm
-1
Cis- and trans-cinnamoylcocaine could be differentiated by:
" The large absorbance at 1320 cm
-1
in the spectrum of trans-cinnamoyl-
cocaine is absent in the spectrum of cis-cinnamoylcocaine.
" In the spectrum of trans-cinnamoylcocaine the absorbance at 1625 cm
-1
is
of about the same magnitude as the absorbance at 1745 and 1695 cm
-1
,
whereas in the spectrum of cis-cinnamoylcocaine the absorbance at
1625 cm
-1
is smaller than the absorbances at 1745 and 1715 cm
-1
.
5.3.7 Ultraviolet (UV) Spectrophotometry
Cocaine show the following absorption peaks in aqueous acid media: 233 nm
{A (1% 1 cm) = 430}, 275 nm.
Analytical notes
• The KBr disc method consists of grinding a dry sample to a very fine powder,
then mixing about 2 mg of homogenized sample powder with 200 mg of
carefully dried and ground KBr. After grinding, the mixture is pressed into a
thin transparent disk.
• KBr should be “IR Grade” and dried at 105° C for a minimum of one hour.
It can be stored in a desiccator containing a strong desiccant (silica gel) or
left in the oven and removed when required.

36 Recommended Methods for the Identification and Analysis of Cocaine in Seized Materials
5.4 The analysis of cocaine enantiomers
Four pairs of enantiomers can be predicted from the structural formula of cocaine.
Each member of a given pair of enantiomers has a diastereoisomeric relationship
to members of all the other pairs. All diastereoisomers have been synthesized and
their configurations and conformations determined by various methods. The only
natural occurring cocaine enantiomer is the l-cocaine.
Many methods have been developed to differentiate these cocaine enantiomers as
certain countries control only the l-cocaine enantiomer. The microcrystal test method
is described in detail below:
Microcrystal test
Reagents
1. Dissolve 10 mg of di-p-toluoyl-d-tartaric acid (TDTA) in 1 ml of ethanol
in a 10 ml volumetric flask. Add 1 ml of glycerine and top up to mark
with distilled water.
2. Dissolve 10 mg of di-l-toluoyl-d-tartaric acid (TLTA) in 1 ml of ethanol in
a 10 ml volumetric flask. Add 1 ml of glycerine and top up to mark with
distilled water.
Method
Place 1 drop of reagent onto the microscope slide, then add a small quantity of
sample to the reagent and stir. View the resulting crystals with a microscopic
magnification of about 100.
Results
With TDTA, l-cocaine HCl, after about 1 minute gives almost perfectly symmetrical
rosettes. The crystals, when first formed, will be greyish-white to white under
polarized light. After growing for a few minutes, some rosettes will exhibit different
colours, (red, blue, green, yellow) on the arms of the rosettes, depending on
orientation.
Analytical notes
• Crystals form in reagents after about three months. Fresh solutions should
be made if old ones fail to give results with authentic cocaine.
• If cocaine is not present as the hydrochloride salt, it must be converted to
this form.

Recommended Methods for the Identification and Analysis of Cocaine in Seized Materials 37
With TLTA, l-cocaine HCl immediately forms greyish-white crystals. The formation
of these crystals varies from a multitude of single needles, to tufts, to fan-shaped,
to sheaves.
D-cocaine HCl gives the completely opposite crystal formation as l-cocaine HCl,
i.e. after about one minute, it gives almost perfectly symmetrical rosettes with TLTA
and crystals varying from single needles, to tufts, to fan-shaped, to sheaves with
TDTA.
Alternative methods to differentiate cocaine enantiomers
There are a variety of other methods and instrumentation (e.g. HPLC, GC, GC-MS,
TLC, IR and NMR) that could be used to differentiate cocaine enantiomers which
would not be mentioned here. Please refer to references [15], [16] and [17] for more
information.
39
6. References
1. O’Neil, M. J., (Ed.). (2006) The Merck Index: An Encyclopedia of Chemicals,
Drugs, and Biologicals, 13th Edition, NJ: Merck.
2. Moffat A. C., Osselton M. D. and Widdop B, (2004) Clarke’s Analysis of
Drugs and Poisons, 3rd Edition, Pharmaceutical Press.
3. Casale J. F. and Klein R. F. X., Illicit production of cocaine, Forensic Science
Review; 5 (1993) 95-107.
4. Patent US 7,855,296 B1 Dec 21, 2010 “Method for synthesizing 2-carbo-
methoxytropinone” Vladimir F Kuznetsov; Cody, WY (US)
5. Turner C. E., Ma C. Y. and Elsohly M. A, Constituents of erythroxylon coca II.
Gas chromatographic analysis of cocaine and other alkaloids in coca leaves.
Journal of Ethnopharmacology, 3 (1981) 293-298.
6. Bieri S., Brachet A., Veuthey J. and Christen P., Cocaine distribution in wild
Erythroxylum species. Journal of Ethnopharmacology, 103 (2006) 439-447.
7. Jenkins A. J., Llosa T., Montoya I. and Cone E. J., Identification and quantitation
of alkaloids in coca tea. Forensic Science International, 77 (1996) 179-189.
8. SWGDRUG, Scientific Working Group for the Analysis of Seized Drugs
(SWGDRUG) Recommendations, www.swgdrug.org
9. Rapid Testing Methods of Drugs of Abuse (ST/NAR/13/REV.1), United
Nations, New York (1995).
10. Tsumura Y., Mitome T. and Kimoto S., False positives and false negatives with
a cocaine-specific field test and modification of test protocol to reduce false
decision. Forensic Science International, 155 (2005) 158-164.
11. Grant F. W., Martin C. and Quackenbush R. W., A simple sensitive specific
field test for cocaine based on the recognition of the odour of methyl benzoate
as a test product. Bulletin on Narcotics, 27 (1975) 33-35.
12. Swiatko J., De Forest P. R. and Zedeck M. S., Further studies on spot tests
and microcrystal tests for identification of cocaine. Journal of Forensic
Sciences, 48 (3) (2003) 581-585.

40 Recommended Methods for the Identification and Analysis of Cocaine in Seized Materials
13. Piñero E. L. and Casale J. F., Quantitation of cocaine by gas chromatography-
flame ionization detection utilizing isopropylcocaine as a structurally related
internal standard. Microgram Journal, 4(1-4) (2006), 47-53.
14. Gill R., Abbott R. W.and Moffat A. C, High-performance liquid chromato-
graphy systems for the separation of local anaesthetic drugs with applicability
to the analysis of illicit cocaine samples. Journal of Chromatography, 301
(1984) 155-163.
15. Olieman C., Maat L. and Beyerman H. C., Analysis of cocaine, pseudococaine,
allococaine and allopseudococaine by ion-pair reverse-phase high-performance
liquid chromatography. Recueil des travaux chimiques des Pays-Bas, 98(10)
(1979) 501-502.
16. Lewin A. H., Parker S. R. and Carroll F. I., Positive identification and
quantitation of isomeric cocaines by high-performance liquid chromatography.
Journal of Chromatography, 193(3) (1980) 371-380.
17. Allen A. C., Cooper D. A., Cottreli R. C. and Kiser W. O., The cocaine
diastereoisomers. Journal of Forensic Sciences, 26(1) (1981) 12-26.

Vienna International Centre, PO Box 500, 1400 Vienna, Austria
Tel.: (+43-1) 26060-0, Fax: (+43-1) 26060-5866, www.unodc.org
United Nations publication
Printed in Austria
ST/NAR/7/REV.1
*1187637*
V.11-87637—March 2012—300
