
49
CHAPTER 3
Research Ethics and
Philosophies
The primary focus of this chapter is on research ethics. While each methods chapter in
this book provides a discussion of ethical issues devoted specifically to a particular method
(e.g., experimental design, survey), this chapter will highlight the general ethical
considerations everyone should consider before beginning his or her research. Every
researcher needs to consider how to practice his or her discipline ethically. Whenever we
interact with other people as social scientists, we must place great importance on the
concerns and emotional needs that shape their responses to our actions. It is here that
ethical research practice begins, with the recognition that our research procedures involve
people who deserve respect. At the end of the chapter, we conclude with a brief discussion
of different social research philosophies that will set the stage for the remainder of the
book.
WHAT DO WE HAVE IN MIND?
Consider the following scenario: One day as you are drinking coffee and reading the
newspaper during your summer in California, you notice a small ad recruiting college
students for a study at Stanford University. You go to the campus and complete an
application. The ad read as follows:
Male college students needed for psychological study of prison life. $80 per day
for 1–2 weeks beginning Aug. 14. For further information & applications, come to
Room 248, Jordan Hall, Stanford U. (Zimbardo 1973: 38)
After you arrive at the university, you are given an information form with more details
about the research (Zimbardo 1973).
FUNDAMENTALS OF RESEARCH IN CRIMINOLOGY AND CRIMINAL JUSTICE
50
Purpose: A simulated prison will be established somewhere in the vicinity of Palo Alto, Stanford [sic], to
study a number of problems of psychological and sociological relevance. Paid volunteers will be randomly
assigned to play the roles of either prisoners and guards [sic] for the duration of the study. This time
period will vary somewhat from about five days to two weeks for any one volunteer—depending upon
several factors, such as the “sentence” for the prisoner or the work effectiveness of the guards. Payment
will be $80 a day for performing various activities and work associated with the operation of our prison.
Each volunteer must enter a contractual arrangement with the principal investigator (Dr. P. G. Zimbardo)
agreeing to participate for the full duration of the study. It is obviously essential that no prisoner can
leave once jailed, except through established procedures. In addition, guards must report for their 8-hour
work shifts promptly and regularly since surveillance by the guards will be around-the-clock—three work
shifts will be rotated or guards will be assigned a regular shift—day, evening, or early morning. Failure
to fulfill this contract will result in a partial loss of salary accumulated—according to a prearranged
schedule to be agreed upon. Food and accommodations for the prisoners will be provided which will meet
minimal standard nutrition, health, and sanitation requirements. A warden and several prison staff will
be housed in adjacent cell blocks, meals and bedding also provided for them. Medical and psychiatric
facilities will be accessible should any of the participants desire or require such services. All participants
will agree to having their behavior observed and to be interviewed and perhaps also taking psychological
tests. Films of parts of the study will be taken, participants agreeing to allow them to be shown, assuming
their content has information of scientific value.
[The information form then summarizes two of the “problems to be studied” and provides a few more details.]
Thanks for your interest in this study. We hope it will be possible for you to participate and to share
your experiences with us.
Philip G. Zimbardo, PhD
Professor of Social Psychology
Stanford University
Source: Zimbardo (1973).
Prison Life Study: General Information
First, you are asked to complete a long questionnaire about your family background,
physical and mental health history, and prior criminal involvement. Next, you are
interviewed by someone, and then you finally sign a consent form. A few days later, you
are informed that you and 20 other young men have been selected to participate in the
experiment. You return to the university to complete a battery of “psychological tests” and
are told you will be picked up for the study the next day (Haney, Banks, & Zimbardo
1973: 73).
The next morning, you hear a siren just before a squad car stops in front of your house.
A police officer charges you with assault and battery, warns you of your constitutional
rights, searches and handcuffs you, and drives you off to the police station. After
fingerprinting and a short stay in a detention cell, you are blindfolded and driven to the
“Stanford County Prison.” Upon arrival, your blindfold is removed and you are stripped
naked, skin-searched, deloused, and issued a uniform (a loosely fitting smock with an ID
number printed on it), bedding, soap, and a towel. You don’t recognize anyone, but you

CHAPTER 3 Research Ethics and Philosophies
51
notice that the other “prisoners” and the “guards” are college-age, apparently almost all
middle-class white men (except for one Asian) like you (Haney et al. 1973; Zimbardo et al.
1973).
The prison warden welcomes you:
As you probably know, I’m your warden. All of you have shown that you are
unable to function outside in the real world for one reason or another—that
somehow you lack the responsibility of good citizens of this great country. We of
this prison, your correctional staff, are going to help you learn what your
responsibilities as citizens of this country are. . . . If you follow all of these rules
and keep your hands clean, repent for your misdeeds and show a proper attitude
of penitence, you and I will get along just fine. (Zimbardo et al. 1973: 38)
Among other behavioral restrictions, the rules stipulate that prisoners must remain silent
during rest periods, during meals, and after lights out. They must address each other only by
their assigned ID numbers, they are to address guards as “Mr. Correctional Officer,” and
everyone is warned that punishment will follow any rule violation (Zimbardo et al. 1973).
You look around and can tell that you are in the basement of a building. You are led
down a corridor to a small cell (6’ x 9’) with three cots, where you are locked behind a steel-
barred black door with two other prisoners (Exhibit 3.1). Located across the hall, there is a
small solitary confinement room (2’ x 2’ x 7’)
for those who misbehave. There is little
privacy, since you realize that the uniformed
guards, behind the mirrored lenses of their
sunglasses, can always observe the prisoners.
After you go to sleep, you are awakened by a
whistle summoning you and the others for a
roll call periodically through the night.
The next morning, you and the other eight
prisoners must stand in line outside your cells
and recite the rules until you remember all 17
of them. Prisoners must chant, “It’s a wonderful
day, Mr. Correctional Officer.” Two prisoners
who get out of line are put in the solitary
confinement unit. After a bit, the prisoners in
Cell 1 decide to resist: They barricade their cell
door and call on the prisoners in other cells to
join in their resistance. The guards respond by
pulling the beds out from the other cells and
spraying several of the inmates with a fire
extinguisher. The guards succeed in enforcing
control and become more authoritarian, while
the prisoners become increasingly docile.
Punishments are regularly meted out for
infractions of rules and sometimes for
Exhibit 3.1
Prisoner in His Cell
Source: From The Lucifer Effect, by Philip Zimbardo
2008, p. 155. Reprinted with permission.

FUNDAMENTALS OF RESEARCH IN CRIMINOLOGY AND CRIMINAL JUSTICE
52
seemingly no reason at all; punishments include doing push-ups, being stripped naked,
having legs chained, and being repeatedly wakened during the night. If this were you, would
you join in the resistance? How would you react to this deprivation of your liberty by these
authoritarian guards? How would you respond given that you signed a consent form allowing
you to be subjected to this kind of treatment?
By the fifth day of the actual Stanford Prison Experiment, five student prisoners had to
be released due to evident extreme stress (Zimbardo 2008). On the sixth day, Philip
Zimbardo terminated the experiment. A prisoner subsequently reported,
The way we were made to degrade ourselves really brought us down and that’s
why we all sat docile towards the end of the experiment. (Haney et al. 1973: 88)
One guard later recounted his experience:
I was surprised at myself. . . . I made them call each other names and clean the
toilets out with their bare hands. I practically considered the prisoners cattle, and I
kept thinking: “I have to watch out for them in case they try something.”
(Zimbardo et al. 1973: 174)
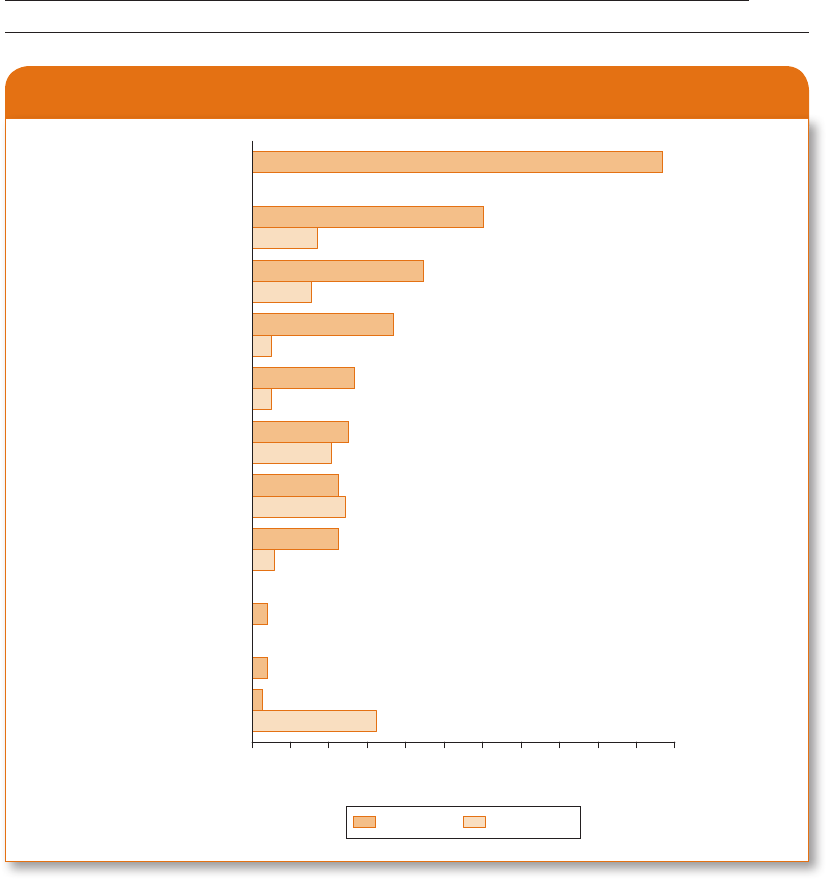
Exhibit 3.2 gives some idea of the difference in how the prisoners and guards behaved.
What is most striking about this result is that all the guards and prisoners had been screened
before the study began to ensure that they were physically and mentally healthy. The roles
of guard and prisoner had been assigned randomly, by the toss of a coin, so the two groups
were very similar when the study began. Something about the “situation” appears to have
led to the deterioration of the prisoners’ mental states and the different behavior of the
guards. Being a guard or a prisoner, with rules and physical arrangements reinforcing
distinctive roles, changed their behavior.
Are you surprised by the outcome of the experiment? By the guard’s report of his
unexpected, abusive behavior? By the prisoners’ ultimate submissiveness and the
considerable psychic distress some felt? (We leave it to you to assess how you would have
responded if you had been an actual research participant.)
Of course, our purpose in introducing this small “experiment” is not to focus attention
on the prediction of behavior in prisons but to introduce the topic of research ethics. We
will refer to Philip Zimbardo’s Stanford Prison Experiment throughout this chapter, since
it is fair to say that this research ultimately had a profound influence on the way that social
scientists think about research ethics as well as on the way that criminologists understand
behavior in prisons. We will also refer to Stanley Milgram’s (1963) experiments on
obedience to authority, since that research also pertains to criminal justice issues and has
stimulated much debate about research ethics.
HISTORICAL BACKGROUND
Formal procedures regarding the protection of research participants emerged only after the
revelation of several very questionable and damaging research practices. A defining event
CHAPTER 3 Research Ethics and Philosophies
53
occurred in 1946, when the Nuremberg War Crime Trials exposed horrific medical
experiments conducted by Nazi doctors and others in the name of “science.” In the 1970s,
Americans were shocked to learn that researchers funded by the U.S. Public Health Service
had followed 399 low-income African American men with syphilis in the 1930s, collecting
data to study the “natural” course of the illness (Exhibit 3.3). Many participants were not
informed of their illness and were denied treatment until 1972, even though a cure
(penicillin) was developed in the 1950s.
Commands
Insults
Deindividuating
reference
Aggression
Threats
Questions
Information
Use of
instruments
Individuating
reference
Helping
Resistance
0102030405060
Frequency
70 80 90 100 110
Guards Prisoners
Exhibit 3.2
Chart of Guard and Prisoner Behavior
Source: From The Lucifer Effect by Philip G. Zimbardo, copyright © 2007 by Philip G. Zimbardo, Inc. Used by permission of
Random House Inc.

FUNDAMENTALS OF RESEARCH IN CRIMINOLOGY AND CRIMINAL JUSTICE
54
Horrible violations of human rights similar to these resulted, in the United States, in the
creation of a National Commission for the Protection of Human Subjects of Biomedical and
Behavioral Research. The commission’s 1979 Belmont Report (from the Department of
Health, Education, and Welfare) established three basic ethical principles for the protection
of human subjects (Exhibit 3.4):
Exhibit 3.3
Tuskegee Syphilis Experiment
Source: Tuskegee Syphilis Study Administrative Records. Records of the Centers for Disease Control and Prevention. National
Archives—Southeast Region (Atlanta).
Respect for Persons
Beneficence Justice
Exhibit 3.4
Belmont Report Principles

CHAPTER 3 Research Ethics and Philosophies
55
• Respect for persons: Treating persons as autonomous agents and protecting those
with diminished autonomy
• Beneficence: Minimizing possible harms and maximizing benefits
• Justice: Distributing benefits and risks of research fairly
The Department of Health and Human Services and the Food and Drug Administration
then translated these principles into specific regulations that were adopted in 1991 as the
Federal Policy for the Protection of Human Subjects. This policy has shaped the course of
social science research ever since. This section introduces these regulations.
Federal regulations require that every institution, including universities that seek federal
funding for biomedical or behavioral research on human subjects, have an institutional
review board (IRB) to review research proposals. IRBs at universities and other agencies
adopt a review process that is principally guided by federally regulated ethical standards
but can be expanded by the IRB itself (Sieber 1992). To promote adequate review of ethical
issues, the regulations require that IRBs include members with diverse backgrounds. The
Office for Protection From Research Risks in the National Institutes of Health monitors
IRBs, with the exception of research involving drugs (which is the responsibility of the
federal Food and Drug Administration).
The Academy of Criminal Justice Sciences (ACJS) and the American Society of
Criminology (ASC), like most professional social science organizations, have adopted
ethical guidelines for practicing criminologists that are more specific than the federal
regulations. The ACJS Code of Ethics also establishes procedures for investigating and
resolving complaints concerning the ethical conduct of the organization’s members. The
Code of Ethics of the ACJS (2000) is available on the ACJS Web site (www.acjs.org). The ASC
follows the American Sociological Association’s code (ASA 1999).
ETHICAL PRINCIPLES
Achieving Valid Results
A commitment to achieving valid results is the necessary starting point for ethical research
practice. Simply put, we have no business asking people to answer questions, submit to
observations, or participate in experimental procedures if we are simply seeking to verify
our preexisting prejudices or convince others to take action on behalf of our personal
interests. It is the pursuit of objective knowledge about human behavior—the goal of
validity—that motivates and justifies our investigations and gives us some claim to the right
to influence others to participate in our research. If we approach our research projects
objectively, setting aside our personal predilections in the service of learning a bit more
about human behavior, we can honestly represent our actions as potentially contributing
to the advancement of knowledge.
The details in Zimbardo’s articles and his recent book (2008) on the prison
experiment make a compelling case for his commitment to achieving valid results—to
learning how and why a prison-like situation influences behavior. In Zimbardo’s (2009)
own words,

FUNDAMENTALS OF RESEARCH IN CRIMINOLOGY AND CRIMINAL JUSTICE
56
Social-psychological studies were showing that human nature was more pliable
than previously imagined and more responsive to situational pressures than we
cared to acknowledge. . . . Missing from the body of social-science research at the
time was the direct confrontation . . . of good people pitted against the forces
inherent in bad situations. . . . I decided that what was needed was to create a
situation in a controlled experimental setting in which we could array on one side
a host of variables, such as . . . coercive rules, power differentials, anonymity. . . .
On the other side, we lined up a collection of the “best and brightest” of young
college men. . . . I wanted to know who wins—good people or an evil situation—
when they were brought into direct confrontation.
Zimbardo (Haney et al. 1973) devised his experiment so the situation would seem
realistic to the participants and still allow careful measurement of important variables and
observation of behavior at all times. Questionnaires and rating scales, interviews with
participants as the research proceeded and after it was over, ongoing video and audio
recording, and documented logs maintained by the guards all ensured that very little would
escape the researcher’s gaze.
Zimbardo’s (Haney et al. 1973) attention to validity is also apparent in his design of the
physical conditions and organizational procedures for the experiment. The “prison” was
constructed in a basement without any windows so that participants were denied a sense
of time and place. Their isolation was reinforced by the practice of placing paper bags over
their heads when they moved around “the facility,” meals were bland, and conditions were
generally demeaning. This was a very different “situation” from what the participants were
used to—suffice it to say that it was no college dorm experience.
However, not all social scientists agree that Zimbardo’s approach achieved valid
results. British psychologists Stephen Reicher and S. Alexander Haslam (2006) argue
that guard behavior was not so consistent and that it was determined by the instructions
Zimbardo gave the guards at the start of the experiment, rather than by becoming a
guard in itself. For example, in another experiment, when guards were trained to
respect prisoners, their behavior was less malicious (Lovibond, Mithiran, & Adams
1979).
In response to such criticism, Zimbardo (2007) has pointed to several replications of his
basic experiment that support his conclusions—as well as to the evidence of patterns of
abuse in the real world of prisons, including the behavior of guards who tormented
prisoners at Abu Ghraib during the war in Iraq.
Do you agree with Zimbardo’s assumption that the effects of being a prisoner or guard
could fruitfully be studied in a mock prison, with “pretend” prisoners? Do you find merit
in the criticisms? Will your evaluation of the ethics of Zimbardo’s experiment be influenced
by your answers to these questions? Should our ethical judgments differ when we are
confident a study’s results provide valid information about important social processes?
As you attempt to answer such questions, bear in mind that both Zimbardo and his
critics support their conflicting ethical arguments with assertions about the validity (or
invalidity) of the experimental results. It is hard to justify any risk for human subjects,
or any expenditure of time and resources, if our findings tell us nothing about the reality
of crime and punishment.

CHAPTER 3 Research Ethics and Philosophies
57
Honesty and Openness
The scientific concern with validity requires that scientists openly disclose their methods
and honestly present their findings. In contrast, research distorted by political or personal
pressures to find particular outcomes or to achieve the most marketable results is unlikely
to be carried out in an honest and open fashion. To assess the validity of a researcher’s
conclusions and the ethics of his or her procedures, you need to know exactly how the
research was conducted. This means that articles or other reports must include a detailed
methodology section, perhaps supplemented by appendices containing the research
instruments or websites or an address where more information can be obtained.
Philip Zimbardo’s research reports seemed to present an honest and forthright account
of the methods used in the Stanford experiment. His initial article (Haney et al. 1973)
contained a detailed description of study procedures, including the physical aspects of the
prison, the instructions to participants, the uniforms used, the induction procedure, and
the specific data collection methods and measures. Many more details, including forms and
pictures, are available on Zimbardo’s website (www.prisonexperiment.org) and in his
recent book (Zimbardo 2008).
The act of publication itself is a vital element in maintaining openness and honesty. It
allows others to review and question study procedures and generate an open dialogue with
the researcher. Although Zimbardo disagreed sharply with his critics about many aspects
of his experiment, their mutual commitment to public discourse in publications resulted
in a more comprehensive presentation of study procedures and a more thoughtful
discourse about research ethics (Savin 1973; Zimbardo 1973). Almost 40 years later, this
commentary continues to inform debates about research ethics (Reicher & Haslam 2006;
Zimbardo 2007).
Openness about research procedures and results goes hand in hand with honesty in
research design. Openness is also essential if researchers are to learn from the work of
others. In spite of this need for openness, some researchers may hesitate to disclose their
procedures or results to prevent others from building on their ideas and taking some of the
credit. Scientists are like other people in their desire to be first. Enforcing standards of
honesty and encouraging openness about research are the best solutions to this problem.
Protecting Research Participants
The ACJS code’s standards concerning the treatment of human subjects include
federal regulations and ethical guidelines emphasized by most professional social science
organizations:
• Research should expose participants to no more than minimal risk of personal
harm. (#16)
• Researchers should fully disclose the purposes of their research. (#13)
• Participation in research should be voluntary, and therefore subjects must give
their informed consent to participate in the research. (#16)
• Confidentiality must be maintained for individual research participants unless it is
voluntarily and explicitly waived. (#14, #18, #19)

FUNDAMENTALS OF RESEARCH IN CRIMINOLOGY AND CRIMINAL JUSTICE
58
Philip Zimbardo (2008) himself decided that his Stanford Prison Experiment was
unethical because it violated the first two of these principles: First, participants “did suffer
considerable anguish . . . and [the experiment] resulted in such extreme stress and
emotional turmoil that five of the sample of initially healthy young prisoners had to be
released early” (pp. 233–234). Second, Zimbardo’s research team did not disclose in
advance the nature of the arrest or booking procedures at police headquarters nor did they
disclose to the participants’ parents how bad the situation had become when they came to
a visiting night. Nonetheless, Zimbardo (Zimbardo et al. 1973; Zimbardo 2008) argued that
there was no long-lasting harm to participants and that there were some long-term social
benefits from this research. In particular, debriefing participants—discussing their
experiences and revealing the logic behind the experiment—and follow-up interviews
enabled the participants to recover from the experience without lasting harm (Zimbardo
2007). Also, the experience led several participants in the experiment, including Zimbardo,
to dedicate their careers to investigating and improving prison conditions. As a result,
publicity about the experiment has also helped focus attention on problems in prison
management.
Do you agree with Zimbardo’s conclusion that his experiment was not ethical? Do you
think it should have been prevented from happening in the first place? Are you relieved to
learn that current standards in the United States for the protection of human subjects in
research would not allow his experiment to be conducted?
In contrast to Zimbardo, Stanley Milgram (1963) believed that his controversial
experiments on obedience to authority were entirely ethical, so debate about this study
persists today. His experiments raise most of the relevant issues we want to highlight here.
Milgram had recruited community members to participate in his experiment at Yale
University. His research was prompted by the ability of Germany’s Nazi regime of the 1930s
and 1940s to enlist the participation of ordinary citizens in unconscionable acts of terror
and genocide. Milgram set out to identify through laboratory experiments the conditions
under which ordinary citizens will be obedient to authority figures’ instructions to inflict
pain on others. He operationalized this obedience by asking subjects to deliver electric
shocks (fake, of course) to students supposedly learning a memory task. Subjects
(“teachers”) were told to administer a shock to the learner each time he gave a wrong
response and to incrementally raise the voltage with each incorrect response. They were
told to increase the shocks over time and many did so, even after the “students,” behind a
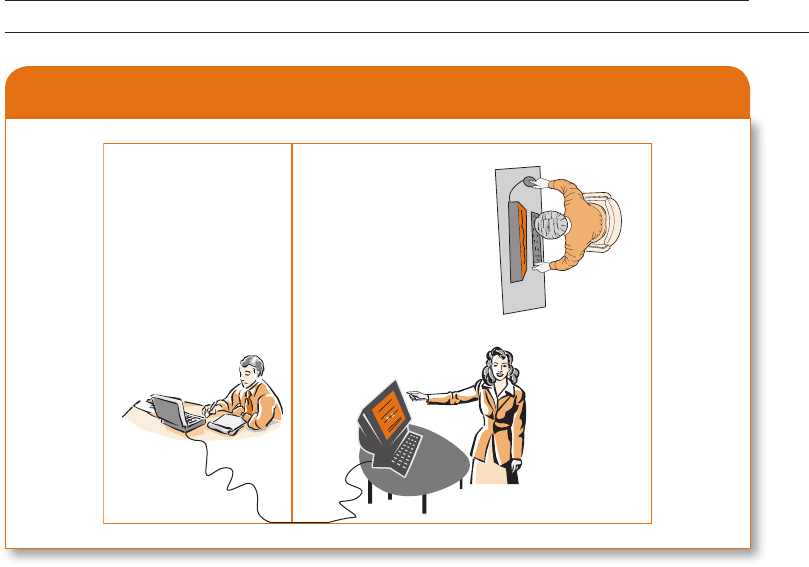
partition, began to cry out in (simulated) pain (Exhibit 3.5). The participants became very
tense, and some resisted as the shocks increased to the (supposedly) lethal range, but many
still complied with the authority in that situation and increased the shocks. Like Zimbardo,
Milgram debriefed participants afterward and followed up later to check on their well-
being. It seemed that none had suffered long-term harm (Milgram 1974).
As we discuss how the ACJS Code of Ethics standards apply to Milgram’s experiments,
you will begin to realize that there is no simple answer to the question, “What is (or isn’t)
ethical research practice?” The issues are just too complicated and the relevant principles
too subject to different interpretations. But we do promise that by the end of this chapter,
you will be aware of the major issues in research ethics and be able to make informed,
defensible decisions about the ethical conduct of social science research.
CHAPTER 3 Research Ethics and Philosophies
59
Avoid Harming Research Participants
Although this standard may seem straightforward, it can be difficult to interpret in
specific cases and harder yet to define in a way that is agreeable to all social scientists.
Does it mean that subjects should not be harmed at all, psychologically or physically?
That they should feel no anxiety or distress whatsoever during the study or even after
their involvement ends? Should the possibility of any harm, no matter how remote,
deter research?
Before we address these questions with respect to Milgram’s experiments, a verbatim
transcript of one session will give you an idea of what participants experienced (Milgram
1965):
150 volts delivered. You want me to keep going?
165 volts delivered
. That guy is hollering in there. There’s a lot of them here.
He’s liable to have a heart condition. You want me to go on?
180 volts delivered. He can’t stand it! I’m not going to kill that man in there!
You hear him hollering? He’s hollering. He can’t stand
it. . . . I mean who is going to take responsibility if
anything happens to that gentleman?
[The experimenter accepts responsibility.] All right.
Experimenter
Teacher
Student
Exhibit 3.5
Diagram of Milgram’s Experiment

FUNDAMENTALS OF RESEARCH IN CRIMINOLOGY AND CRIMINAL JUSTICE
60
195 volts delivered. You see he’s hollering. Hear that? Gee, I don’t know.
[The experimenter says: “The experiment requires that
you go on.”] I know it does, sir, but I mean—Hugh—he
don’t know what he’s in for. He’s up to 195 volts.
210 volts delivered.
225 volts delivered.
240 volts delivered. (p. 67)
This experimental manipulation generated “extraordinary tension” (Milgram 1963):
Subjects were observed to sweat, tremble, stutter, bite their lips, groan and dig
their fingernails into their flesh. . . . Full-blown, uncontrollable seizures were
observed for 3 subjects. [O]ne . . . seizure [was] so violently convulsive that it was
necessary to call a halt to the experiment [for that individual]. (p. 375)
An observer (behind a one-way mirror) reported, “I observed a mature and initially poised
businessman enter the laboratory smiling and confident. Within 20 minutes he was
reduced to a twitching, stuttering wreck, who was rapidly approaching a point of nervous
collapse” (p. 377).
Psychologist Diana Baumrind (1964) disagreed sharply with Milgram’s approach,
concluding that the emotional disturbance subjects experienced was “potentially harmful
because it could easily affect an alteration in the subject’s self-image or ability to trust adult
authorities in the future” (p. 422). Stanley Milgram (1964) quickly countered, “As the
experiment progressed there was no indication of injurious effects in the subjects; and as
the subjects themselves strongly endorsed the experiment, the judgment I made was to
continue the experiment” (p. 849).
When Milgram (1964) surveyed the subjects in a follow-up, 83.7% endorsed the
statement that they were “very glad” or “glad” “to have been in the experiment,” 15.1%
were “neither sorry nor glad,” and just 1.3% were “sorry” or “very sorry” to have
participated (p. 849). Interviews by a psychiatrist a year later found no evidence “of any
traumatic reactions” (p. 197). Subsequently, Milgram (1974) argued that “the central moral
justification for allowing my experiment is that it was judged acceptable by those who took
part in it” (p. 21).
Milgram (1964) also attempted to minimize harm to subjects with post-experimental
procedures “to assure that the subject would leave the laboratory in a state of well being”
(p. 374). A friendly reconciliation was arranged between the subject and the victim, and an
effort was made to reduce any tensions that arose as a result of the experiment. In some
cases, the “dehoaxing” (or “debriefing”) discussion was extensive, and all subjects were
promised (and later received) a comprehensive report (p. 849).
In a later article, Baumrind (1985) dismissed the value of the self-reported “lack of harm”
of subjects who had been willing to participate in the experiment—and noted that 16% did
not endorse the statement that they were “glad” they had participated in the experiment
(p. 168). Baumrind also argued that research indicates most students who have participated

CHAPTER 3 Research Ethics and Philosophies
61
in a deception experiment report a decreased trust in authorities as a result—a tangible
harm in itself.
Many social scientists, ethicists, and others concluded that Milgram’s procedures had
not harmed the subjects and so were justified for the knowledge they produced, but others
sided with Baumrind’s criticisms (Miller 1986). What is your opinion at this point? Does
Milgram’s debriefing process relieve your concerns? Are you as persuaded by the subjects’
own endorsement of the procedures as was Milgram?
Would you ban such experiments because of the potential for harm to subjects? Does
the fact that Zimbardo’s and Milgram’s experiments seemed to yield significant insights
into the effect of a social situation on human behavior—insights that could be used to
improve prisons or perhaps lessen the likelihood of another holocaust—make any
difference (Reynolds 1979)? Do you believe that this benefit outweighs the foreseeable
risks?
Obtain Informed Consent
The requirement of informed consent is also more difficult to define than it first appears.
To be informed consent, it must be given by the persons who are competent to consent,
can consent voluntarily, are fully informed about the research, and comprehend what they
have been told (Reynolds 1979). Still, even well-intentioned researchers may not foresee
all the potential problems and may not point them out in advance to potential participants
(Baumrind 1985). In Zimbardo’s prison-simulation study, all the participants signed con-
sent forms, but they were not “fully informed” in advance about potential risks. The
researchers themselves did not realize that the study participants would experience so
much stress so quickly, that some prisoners would have to be released for severe negative
reactions within the first few days, or that even those who were not severely stressed would
soon be begging to be released from the mock prison. But on the other hand, are you con-
cerned that real harm “could result from not doing research on destructive obedience” and
other troubling human behavior (Miller 1986:138, italics original)?
Obtaining informed consent creates additional challenges for researchers. The language
of the consent form must be clear and understandable to the research participants yet
sufficiently long and detailed to explain what will actually happen in the research.
Examples A (Exhibit 3.6) and B (Exhibit 3.7) illustrate two different approaches to these
trade-offs.
Consent form A was approved by the University of Delaware IRB for in-depth interviews
with former inmates about their experiences after release from prison. Consent form B is
the one used by Philip Zimbardo. It is brief and to the point, leaving out many of the details
that current standards for the protection of human subjects require. Zimbardo’s consent
form also released the researchers from any liability for problems arising out of the
research (Such a statement is no longer allowed.).
As in Milgram’s (1963) study, experimental researchers whose research design requires
some type of subject deception try to minimize disclosure of experimental details by
withholding some information before the experiment begins but then debrief subjects at
the end. In the debriefing, the researcher explains to the subjects what happened in the
experiment and why, and then addresses participants’ concerns or questions. A carefully

FUNDAMENTALS OF RESEARCH IN CRIMINOLOGY AND CRIMINAL JUSTICE
62
INFORMED CONSENT
ROADS DIVERGE: LONG-TERM PATTERNS OF RELAPSE, RECIDIVISM, AND DESIS TANCE
FOR A RE-ENTRY COHORT (National Institute of Justice, 2008-IJ-CX-0017)
PURPOSE: You are one of approximately 300 people being asked to participate in a research
project conducted by the Center for Drug and Alcohol Studies at the University of Delaware.
You were part of the original study of offenders in Delaware leaving prison in the 1990s, and we
want to find out how things in your life have changed since that time. The overall purpose of this
research is to help us understand what factors lead to changes in criminal activity and drug use
over time.
PROCEDURES: If you agree to take part in this study, you will be asked to complete a survey,
which will last approximately 60 to 90 minutes. We will ask you to provide us with some contact
information so that we can locate you again if we are able to do another follow up study in the
future. You will be asked about your employment, family history, criminal involvement, health his-
tory, drug use, and how these have changed over time. We will use this information, as well as
information that you have previously provided or which is publicly available. We will not ask you
for the names of anyone, or the specific dates or specific places of any of your activities. The
interviews will be tape-recorded, but you will not be identified by name on the tape. The tapes will
be stored in a locked cabinet until they can be transcribed to an electronic word processor. After
the tapes have been transcribed and checked for accuracy they will be destroyed. Anonymous
transcribed data will be kept indefinitely – no audio data will be kept.
RISKS: There are some risks to participating in this study. You may experience distress or dis-
comfort when asked questions about your drug use, criminal history, and other experiences.
Should this occur, you may choose not to answer such questions. If emotional distress occurs,
our staff will make referrals to services you may need, including counseling, and drug abuse
treatment and support services.
The risk that confidentiality could be broken is a concern, but it is very unlikely to occur. You will
not be identified on the audiotape of the interview. We request that you not mention names of
other people or places, but if this happens, those names will be deleted from the audiotape prior
to transcription. All study materials are kept in locked file cabinets. Only three members of [the]
research team will have access to study materials.
BENEFITS: You will have the opportunity to participate in an important research project, which
may lead to the better understanding of what factors both help and prevent an individual’s recov-
ery from drug use and criminal activity.
COMPENSATION: You will receive $100 to compensate you for your time and travel costs for
this interview.
CONFIDENTIALITY: Your records will be kept confidential. They will be kept under lock and key
and will not be shared with anyone without your written permission. Your name will not appear
on any data file or research report.
Exhibit 3.6
Consent Form A

CHAPTER 3 Research Ethics and Philosophies
63
A Privacy Certificate has been approved by the U.S. Department of Justice. The data will be
protected from being revealed to non-research interests by court subpoena in any federal, state,
or local civil, criminal, administrative, legislative or other proceedings.
You should understand that a Privacy Certificate does not prevent you or a member of your
family from voluntarily releasing information about yourself or your involvement in this research.
If you give anyone written consent to receive research information, then we may not use the
Certificate to withhold that information.
The Privacy Certificate does not prevent research staff from voluntary disclosures to authorities
if we learn that you intend to harm yourself or someone else. These incidents would be reported
as required by state and federal law. However, we will not ask you questions about these areas.
Because this research is paid for by the National Institute of Justice, staff of this research office
may review copies of your records, but they also are required to keep that information confidential.
RIGHT TO QUIT THE STUDY: Participation in this research project is voluntary and you have
the right to leave the study at any time. The researchers and their assistants have the right to
remove you from this study if needed.
You may ask and will receive answers to any questions concerning this study. If you have any
questions about this study, you may contact Ronet Bachman or Daniel O’Connell at (302) 831-
6107. If you have any questions about your rights as a research participant you may contact the
Chairperson of the University of Delaware’s Human Subjects Review Board at (302) 831-2136.
CONSENT TO BE INTERVIEWED
I have read and understand this form (or it has been read to me), and I agree to participate in the
in-depth interview portion of this research project.
____________________________________________
Participant Signature Date
____________________________________________
Signature of Witness/Interviewer Date
CONSENT TO BE CONTACTED IN FUTURE
I have read and understand this form (or it has been read to me), and I agree to be recontacted
in the future as part of this research project.
____________________________________________
Participant Signature Date
____________________________________________
Signature of Witness/Interviewer Date
Ronet Bachman, PhD
Principal Investigator
University of Delaware
Telephone: (302) 831-6107

FUNDAMENTALS OF RESEARCH IN CRIMINOLOGY AND CRIMINAL JUSTICE
64
CONSENT
Prison Life Study
Dr. Zimbardo
August 1971
__________________________________________________________
(date) (name of volunteer)
I, _________________________________________, the undersigned, hereby consent to par-
ticipate as a volunteer in a prison life study research project to be conducted by the Stanford
University Psychology Department.
The nature of the research project has been fully explained to me, including, without limi-
tation, the fact that paid volunteers will be randomly assigned to the roles of either “prison-
ers” or “guards” for the duration of the study. I understand that participation in the research
project will involve a loss of privacy, that I will be expected to participate for the full duration
of the study, that I will only be released from participation for reasons of health deemed
adequate by the medical advisers to the research project or for other reasons deemed
appropriate by Dr. Philip Zimbardo, Principal Investigator of the project, and that I will be
expected to follow directions from staff members of the project or from other participants in
the research project.
I am submitting myself for participation in this research project with full knowledge and
understanding of the nature of the research project and of what will be expected of me. I specifi-
cally release the Principal Investigator and the staff members of the research project, Stanford
University, its agents and employees, and the Federal Government, its agents and employees,
from any liability to me arising in any way out of my participation in the project.
________________________________________________________
(signature of volunteer)
Witness: ________________________________________________
If volunteer is a minor:
________________________________________________________
(signature of person authorized to consent for volunteer)
Witness: ________________________________________________
________________________________________________________
(relationship to volunteer)
Exhibit 3.7 Consent Form B

CHAPTER 3 Research Ethics and Philosophies
65
designed debriefing procedure can help the research participants learn from the
experimental research and grapple constructively with feelings elicited by the realization
that they were deceived (Sieber 1992). However, even though debriefing can be viewed as a
substitute, in some cases, for securing fully informed consent prior to the experiment,
debriefed subjects who disclose the nature of the experiment to other participants can
contaminate subsequent results (Adair, Dushenko, & Lindsay 1985). Unfortunately, if the
debriefing process is delayed, the ability to lessen any harm resulting from the deception
may also be compromised.
If you were to serve on your university’s IRB, would you allow this type of research to
be conducted? Can students who are asked to participate in research by their professor
be considered able to give informed consent? Do you consider “informed consent” to be
meaningful if the true purpose or nature of an experimental manipulation is not
revealed?
The process and even possibility of obtaining informed consent must take into
account the capacity of prospective participants to give informed consent. For example,
children cannot legally give consent to participate in research. Instead, minors must in
most circumstances be given the opportunity to give or withhold their assent or
compliance to participate in research, usually by a verbal response to an explanation
of the research. In addition, a child’s legal guardian typically must grant additional
written informed consent to have the child participate in research (Sieber 1992). There
are also special protections for other populations that are likely to be vulnerable to
coercion—prisoners, pregnant women, mentally disabled persons, and educationally
or economically disadvantaged persons. Would you allow research on prisoners, whose
ability to give “informed consent” can be questioned? If so, what special protections do
you think would be appropriate?
Avoid Deception in Research, Except in Limited Circumstances
Deception occurs when subjects are misled about research procedures in an effort to deter-
mine how they would react to the treatment if they were not research subjects. In other
words, researchers deceive their subjects when they believe that knowledge of the experi-
mental premise may actually change the subjects’ behavior. Deception is a critical compo-
nent of many experiments, in part because of the difficulty of simulating real-world
stresses and dilemmas in a laboratory setting. The goal is to get subjects “to accept as true
what is false or to give a false impression” (Korn 1997: 4). In Milgram’s (1963) experiment,
for example, deception seemed necessary because the subjects could not be permitted to
administer real electric shocks to the “student,” yet it would not have made sense to order
the subjects to do something that they didn’t find to be so troubling. Milgram (1992)
insisted that the deception was absolutely essential. The results of many other experiments
would be worthless if subjects understood what was really happening to them while the
experiment was in progress. The real question is this: Is that sufficient justification to allow
the use of deception?
There are many examples of research efforts that employ placebos, ruses, or guises
to ensure that participants’ behavior is genuine. For example, Piliavin and Piliavin
(1972) staged fake seizures on subway trains to study helpfulness. Would you vote to

FUNDAMENTALS OF RESEARCH IN CRIMINOLOGY AND CRIMINAL JUSTICE
66
allow such deceptive practices in research if you were a member of your university’s
IRB? What about less dramatic instances of deception in laboratory experiments with
students like yourself? Do you react differently to the debriefing by Milgram compared
to that by Zimbardo?
What scientific or educational or applied “value” would make deception justifiable, even
if there is some potential for harm? Who determines whether a nondeceptive intervention
is “equally effective” (Miller 1986: 103)? Diana Baumrind (1985) suggested that personal
“introspection” would have been sufficient to test Milgram’s hypothesis and has argued
subsequently that intentional deception in research violates the ethical principles of self-
determination, protection of others, and maintenance of trust between people and so can
never be justified. How much risk, discomfort, or unpleasantness might be seen as affecting
willingness to participate? When should a post-experimental “attempt to correct any
misconception” due to deception be deemed sufficient?
Can you see why an IRB, representing a range of perspectives, is an important tool for
making reasonable, ethical research decisions when confronted with such ambiguity?
Maintain Privacy and Confidentiality
Maintaining privacy and confidentiality is another key ethical standard for protecting research
participants, and the researcher’s commitment to that standard should be included in the
informed consent agreement (Sieber 1992). Procedures to protect each subject’s privacy, such
as locking records and creating special identifying codes, must be created to minimize the
risk of access by unauthorized persons. However, statements about confidentiality should be
realistic: In some cases, laws allow research records to be subpoenaed and may require report-
ing child abuse; a researcher may feel compelled to release information if a health- or life-
threatening situation arises and participants need to be alerted. Also, the standard of
confidentiality does not apply to observation in public places and information available in
public records.
There are two exceptions to some of these constraints: The National Institute of Justice
can issue a “Privacy Certificate,” and the National Institutes of Health can issue a
“Certificate of Confidentiality.” Both of these documents protect researchers from being
legally required to disclose confidential information. Researchers who are focusing on
high-risk populations or behaviors, such as crime, substance abuse, sexual activity, or
genetic information, can request such a certificate. Suspicions of child abuse or neglect
must still be reported, as well as instances where respondents may immediately harm
themselves or others. In some states, researchers also may be required to report crimes
such as elder abuse (Arwood & Panicker 2007).
The Health Insurance Portability and Accountability Act (HIPAA) passed by Congress in
1996 created much more stringent regulations for the protection of health care data. As
implemented by the U.S. Department of Health and Human Services in 2000 (and revised in
2002), the HIPAA Final Privacy Rule applies to oral, written, and electronic information that
“relates to the past, present, or future physical or mental health or condition of an individual.”
The HIPAA rule requires that researchers have valid authorization for any use or disclosure
of “protected health information” (PHI) from a health care provider. Waivers of authorization
can be granted in special circumstances (Cava, Cushman, & Goodman 2007).

CHAPTER 3 Research Ethics and Philosophies
67
The Uses of Research
Although many scientists believe that personal values should be left outside the laboratory,
some feel that it is proper—even necessary—for scientists to concern themselves with the
way their research is used. Philip Zimbardo made it clear that he was concerned about the
phenomenon of situational influence on behavior precisely because of its implications for
people’s welfare. As you have already learned, his first article (Haney et al. 1973) high-
lighted abuses in the treatment of prisoners. In his more comprehensive book, Zimbardo
(2007) used his findings to explain the atrocities committed at Abu Ghraib. He also urged
reforms in prison policy.
It is also impossible to ignore the very practical implications of Milgram’s investigations,
which Milgram (1974) took pains to emphasize. His research highlighted the extent of
obedience to authority and identified multiple factors that could be manipulated to lessen
blind obedience (such as encouraging dissent by just one group member, removing the
subject from direct contact with the authority figure, and increasing the contact between
the subject and the victim).
The evaluation research by Lawrence Sherman and Richard Berk (1984) on the police
response to domestic violence provides an interesting cautionary tale about the uses of
science. As you will recall from Chapter 2, the results of this field experiment indicated that
those who were arrested were less likely to subsequently commit violent acts against their
partners. Sherman (1992) explicitly cautioned police departments not to adopt mandatory
arrest policies based solely on the results of the Minneapolis experiment, but the results were
publicized in the mass media and encouraged many jurisdictions to change their policies
(Binder & Meeker 1993; Lempert 1989). Although we now know that the original finding of
a deterrent effect of arrest did not hold up in other cities where the experiment was repeated,
Sherman (1992) later suggested that implementing mandatory arrest policies might have
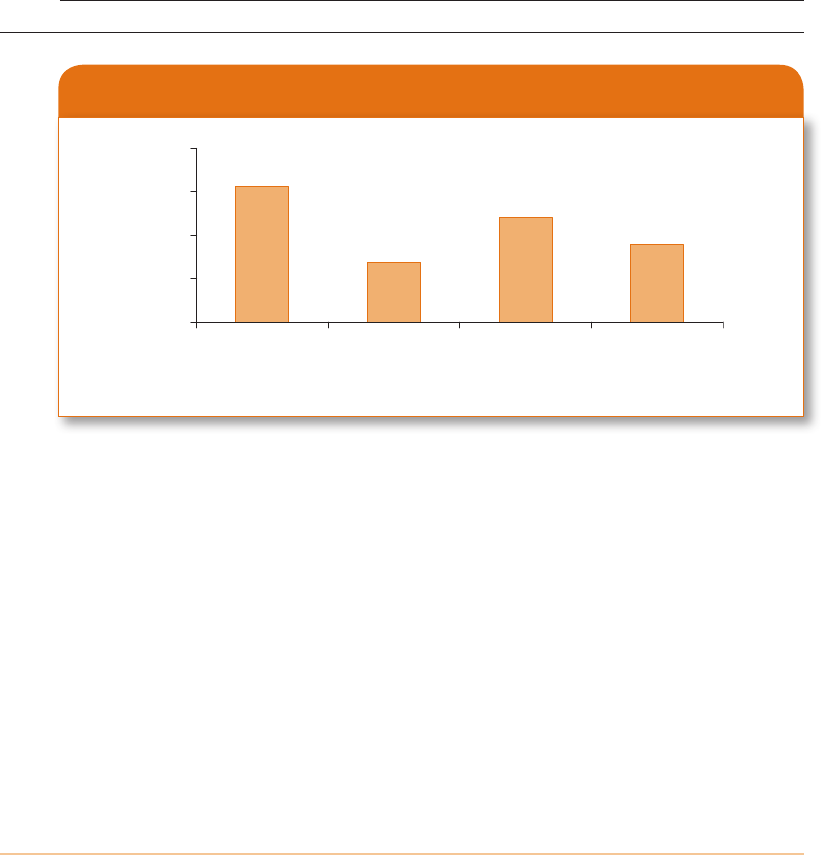
prevented some subsequent cases of spouse abuse. JoAnn Miller’s (2003) analysis of victims’
experiences and perceptions concerning their safety after the mandatory arrest experiment
in Dade County, Florida, found that victims reported less violence if their abuser had been
arrested (or assigned to a police-based counseling program called “Safe Streets”) (Exhibit 3.8).
Should this Dade County finding be publicized in the popular press so it could be used to
improve police policies? What about the results of the other replication studies where arrest
led to increased domestic assault? The answers to such questions are never easy.
Social scientists who conduct research on behalf of specific organizations may face
additional difficulties when the organization, instead of the researcher, controls the final
report and the publicity it receives. If organizational leaders decide that particular research
results are unwelcome, the researcher’s desire to have findings used appropriately and
reported fully can conflict with contractual obligations. Researchers can often anticipate
such dilemmas in advance and resolve them when the contract for research is negotiated—
or simply decline a particular research opportunity altogether. But other times, such
problems come up only after a report has been drafted, or the problems are ignored by a
researcher who needs a job or needs to maintain particular professional relationships.
These possibilities cannot be avoided entirely, but because of them, it is always important
to acknowledge the source of research funding in reports and to consider carefully the
sources of funding for research reports written by others.
FUNDAMENTALS OF RESEARCH IN CRIMINOLOGY AND CRIMINAL JUSTICE
68
0
10
20
30
40
Control Arrest Safe Streets Arrest and
Safe Streets
Treatment Condition
Percent
Exhibit 3.8
Victim Reports of Violence Following Police Intervention
Source: Adapted from Miller (2003: 704).
The withholding of a beneficial treatment from some subjects also is a cause for ethical
concern. Recall that the Sherman and Berk (1984) experiment required the random
assignment of subjects to treatment conditions and thus had the potential of causing harm
to the victims of domestic violence whose batterers were not arrested. The justification for
the study design, however, is quite persuasive: The researchers didn’t know prior to the
experiment which response to a domestic violence complaint would be most likely to deter
future incidents (Sherman 1992). The experiment provided clear evidence about the value
of arrest, so it can be argued that the benefits outweighed the risks.
In later chapters, we will continue to highlight the ethical dilemmas faced by research
that utilizes particular types of methods. Before we begin our examination of various
research methods, however, we first want to introduce you to the primary philosophies.
SOCIAL RESEARCH PHILOSOPHIES
What influences the decision to choose one research strategy over another? The motive for
conducting research is critical: An explanatory or evaluative motive generally leads a researcher
to use quantitative methods, whereas an exploratory motive often results in the use of qualitative
methods. Of course, a descriptive motive means choosing a descriptive research strategy.
Positivism and Postpositivism
A researcher’s philosophical perspective on reality and on the appropriate role of the
researcher also will shape his or her choice of methodological preferences. Researchers with
a philosophy of positivism believe that there is an objective reality that exists apart from the
perceptions of those who observe it; the goal of science is to better understand this reality.

CHAPTER 3 Research Ethics and Philosophies
69
Whatever nature “really” is, we assume that it presents itself in precisely the same
way to the same human observer standing at different points in time and space. . . .
We assume that it also presents itself in precisely the same way across different
human observers standing at the same point in time and space. (Wallace 1983: 461)
This philosophy is traditionally associated with science (Weber 1949), with the expectation
that there are universal laws of human behavior, and with the belief that scientists must be
objective and unbiased to see reality clearly.
Postpositivism is a philosophy of reality that is closely related to positivism.
Postpositivists believe that there is an external, objective reality, but are very sensitive to
the complexity of this reality and the limitations of the scientists who study it. Social
scientists, in particular, recognize the biases they bring to their research as they are social
beings themselves (Guba & Lincoln 1994: 109–111). As a result, they do not think scientists
can ever be sure that their methods allow them to perceive objective reality. Rather, the goal
of science can only be to achieve intersubjective agreement among scientists about the
nature of reality (Wallace 1983: 461). For example, postpositivists may worry that
researchers’ predispositions may bias them in favor of deterrence theory. Therefore, they
will remain somewhat skeptical of results that support predictions based on deterrence
until a number of researchers feel that they have found supportive evidence. The
postpositivist retains much more confidence in the ability of the community of social
researchers to develop an unbiased account of reality than in the ability of any individual
social scientist to do so (Campbell & Russo 1999: 144).
Positivist Research Guidelines
To achieve an accurate understanding of the social world, a researcher operating within the
positivist or postpositivist tradition must adhere to some basic guidelines about how to
conduct research:
1. Test ideas against empirical reality without becoming too personally invested in a
particular outcome. This guideline requires a commitment to “testing,” as opposed
to just reacting to events as they happen or looking for what we want to or expect
to see (Kincaid 1996: 51–54).
2. Plan and carry out investigations systematically. Social researchers have little hope
of conducting a careful test of their ideas if they do not fully think through in
advance how they should go about the test and then proceed accordingly.
3. Document all procedures and disclose them publicly. Social researchers should
disclose the methods on which their conclusions are based so that others can
evaluate for themselves the likely soundness of these conclusions (Kincaid 1996).
4. Clarify assumptions. No investigation is complete in itself. Whatever the
researcher’s method(s), the effort rests on some background assumptions. For
example, research to determine whether arrest has a deterrent effect assumes that
potential law violators think rationally and that they calculate potential costs and
benefits prior to committing crimes.

FUNDAMENTALS OF RESEARCH IN CRIMINOLOGY AND CRIMINAL JUSTICE
70
5. Specify the meaning of all terms. Words often have multiple or unclear meanings.
“Recidivism,” “self-control,” “poverty,” “overcrowded,” and so on can mean
different things to different people. In scientific research, all terms must be defined
explicitly and used consistently.
6. Maintain a skeptical stance toward current knowledge. The results of any particular
investigation must be examined critically, although confidence about
interpretations of the social or natural world increases after repeated investigations
yield similar results.
7. Replicate research and build social theory. No one study is definitive by itself. We
cannot fully understand a single study’s results apart from the larger body of
knowledge to which it is related, and we cannot place much confidence in these
results until the study has been replicated.
8. Search for regularities or patterns. Positivist and postpositivist scientists assume
that the natural world has some underlying order of relationships, so that unique
events and individuals can be understood at least in part in terms of general
principles (Grinnell 1992: 27–29).
Real investigations by social scientists do not always include much attention to theory,
specific definitions of all terms, and so forth. However, all social researchers should be
compelled to study these guidelines and to consider the consequences of not following any
with which they do not agree.
A Positivist Research Goal: Advancing Knowledge
The goal of the traditional positivist scientific approach is to advance scientific knowledge.
This goal is achieved when research results are published in academic journals or pre-
sented at academic conferences.
The positivist approach regards value considerations to be beyond the scope of science.
In Max Weber’s (1949) words, “An empirical science cannot tell anyone what he should
do—but rather what he can do—and under certain circumstances—what he wishes to do”
(p. 54). The idea is that developing valid knowledge about how society is organized, or how
we live our lives, does not tell us how society should be organized or how we should live
our lives. The determination of empirical facts should be a separate process from the
evaluation of these facts as satisfactory or unsatisfactory (p. 11).
Intersubjective agreement An agreement by different observers on what is happening in
the natural or social world
Positivism The belief, shared by most scientists, that there is a reality that exists quite
apart from our own perception of it, although our knowledge of this reality may never be
complete
Postpositivism The belief that there is an empirical reality but that our understanding of
it is limited by its complexity and by the biases and other limitations of researchers

CHAPTER 3 Research Ethics and Philosophies
71
Interpretivism and Constructivism
Qualitative research is often guided by a philosophy of interpretivism. Interpretive
social scientists believe that reality is socially constructed and that the goal of social
scientists is to understand what meanings people give to reality, not to determine how
reality works apart from these interpretations. This philosophy rejects the positivist
belief that there is a concrete, objective reality that scientific methods help us to under-
stand (Lynch & Bogen 1997); instead, interpretivists believe that scientists construct an
image of reality based on their own preferences and prejudices and their interactions
with others.
Here is the basic argument: The empirical data we collect all come to us through our
own senses and must be interpreted with our own minds. This suggests that we can never
be sure that we have understood reality properly, or that we ever can, or that our own
understandings can really be judged more valid than someone else’s.
Searching for universally applicable social laws can distract from learning what
people know and how they understand their lives. The interpretive social
researcher examines meanings that have been socially constructed. . . . There is
not one reality out there to be measured; objects and events are understood by
different people differently, and those perceptions are the reality—or realities—
that social science should focus on. (Rubin & Rubin 1995: 35)
The paradigm of constructivism extends interpretivist philosophy by emphasizing the
importance of exploring how different stakeholders in a social setting construct their
beliefs (Guba & Lincoln 1989: 44–45). It gives particular attention to the different goals of
researchers and other participants in a research setting and seeks to develop a consensus
among participants about how to understand the focus of inquiry. The constructivist
research report will highlight different views of the social program or other issue and
explain how a consensus can be reached among participants.
Interpretivism The belief that reality is socially constructed and that the goal of social
scientists is to understand what meanings people give to that reality. Max Weber termed
the goal of interpretivist research verstehen, or “understanding.”
Constructivism A perspective that emphasizes how different stakeholders in social
settings construct their beliefs
Constructivist inquiry uses an interactive research process, in which a researcher
begins an evaluation in some social setting by identifying the different interest groups
in that setting. The researcher goes on to learn what each group thinks, and then
gradually tries to develop a shared perspective on the problem being evaluated (Guba
& Lincoln 1989: 42).

FUNDAMENTALS OF RESEARCH IN CRIMINOLOGY AND CRIMINAL JUSTICE
72
Interpretivist/Constructivist Research Guidelines
Researchers guided by an interpretivist philosophy reject some of the guidelines to which
positivist researchers seek to adhere. In fact, there is a wide variety of specific approaches
that can be termed “interpretivist,” and each has some guidelines that it highlights. For
those working within the constructivist perspective, Guba and Lincoln (1989) suggest four
key steps for researchers, each of which may be repeated many times in a given study:
1. Identify stakeholders and solicit their “claims, concerns, and issues.”
2. Introduce the claims, concerns, and issues of each stakeholder group to the other
stakeholder groups and ask for their reactions.
3. Focus further information collection on claims, concerns, and issues about which
there is disagreement among stakeholder groups.
4. Negotiate with stakeholder groups about the information collected, and attempt
to reach consensus on the issues about which there is disagreement (p. 42).
An Interpretivist or Constructivist Research Goal: Creating Change
Some social researchers with an interpretivist or constructivist orientation often reject
explicitly the traditional positivist distinction between facts and values (Sjoberg & Nett
1968). Bellah et al. (1985) have instead proposed a model of “social science as public phi-
losophy.” In this model, social scientists focus explicit attention on achieving a more just
society.
Whyte (1991) proposed a more activist approach to research called participatory action
research. As the name implies, this approach encourages social researchers to get “out of
the academic rut” and bring values into the research process (p. 285).
In participatory action research, the researcher involves as active participants some
members of the setting studied. Both the organizational members and the researcher are
assumed to want to develop valid conclusions, to bring unique insights, and to desire
change, but Whyte (1991) believes these objectives are more likely to be obtained if the
researcher collaborates actively with the persons studied.
An Integrated Philosophy
It is tempting to think of positivism and postpositivism as representing an opposing
research philosophy to interpretivism and constructivism. Then it seems that we should
choose the one philosophy that seems closest to our own preferences and condemn the
other as “unscientific,” “uncaring,” or perhaps just “unrealistic.” But there are good reasons
to prefer a research philosophy that integrates some of the differences between these phi-
losophies (Smith 1991).
And what about the important positivist distinction between facts and values in social
research? Here, too, there is evidence that neither the “value-free” presumption of
positivists nor the constructivist critique of this position is entirely correct. For example,
Savelsberg, King, and Cleveland (2002) examined influences on the focus and findings of

CHAPTER 3 Research Ethics and Philosophies
73
published criminal justice scholarship. They found that criminal justice research was more
likely to be oriented to topics and theories suggested by the state when it was funded by
government agencies. This reflects a political influence on scholarship. However,
government funding did not have any bearing on the researchers’ conclusions about the
criminal justice processes they examined. This suggests that scientific procedures can
insulate the research.
Which philosophy makes the most sense to you? Do you agree with positivists and
postpositivists that scientific methods can help us understand the social world as it is, not
just as we would like to think it is? Does the interpretivist focus on meanings sound like a
good idea? Whatever your answers to these questions, you would probably agree that
developing a valid understanding of the social world is not an easy task for social
scientists.
CONCLUSION
The extent to which ethical issues present methodological challenges for researchers
varies dramatically with the type of research design. Survey research, in particular,
creates few ethical problems. In fact, researchers from Michigan’s Institute for Social
Research Survey Center interviewed a representative national sample of adults and found
that 68% of those who had participated in a survey were somewhat or very interested in
participating in another; the more times respondents had been interviewed, the more
willing they were to participate again. Presumably, they would have felt differently if they
had been treated unethically (Reynolds 1979). On the other hand, some experimental
studies in the social sciences that have put people in uncomfortable or embarrassing
situations have generated vociferous complaints and years of debate about ethics
(Reynolds 1979; Sjoberg 1967).
The evaluation of ethical issues in a research project should be based on a realistic
assessment of the overall potential for harm and benefit to research subjects rather than
an apparent inconsistency between any particular aspect of a research plan and a specific
ethical guideline. For example, full disclosure of “what is really going on” in an experimental
study is unnecessary if subjects are unlikely to be harmed. Nevertheless, researchers
should make every effort to foresee all possible risks and to weigh the possible benefits of
the research against these risks. They should consult with individuals with different
perspectives to develop a realistic risk–benefit assessment, and they should try to
maximize the benefits to, as well as minimize the risks for, subjects of the research (Sieber
1992).
Ultimately, these decisions about ethical procedures are not just up to you, as a
researcher, to make. Your university’s IRB sets the human subjects’ protection standards for
your institution and will require researchers—even, in most cases, students—to submit
their research proposal to the IRB for review. So we leave you with the instruction to review
the human subjects guidelines of the ACJS or other professional association in your field,
consult your university’s procedures for the conduct of research with human subjects, and
then proceed accordingly.
FUNDAMENTALS OF RESEARCH IN CRIMINOLOGY AND CRIMINAL JUSTICE
74
KEY TERMS
Belmont Report
Beneficence
Certificate of Confidentiality
Code of Ethics
Constructivism
Debriefing
Federal Policy for the
Protection of Human
Subjects
Institutional review board (IRB)
Interpretivism
Intersubjective agreement
Nuremberg War Crime Trials
Office for Protection From
Research Risks in the
National Institutes of
Health
Participatory action research
Philip Zimbardo’s Stanford
Prison Experiment
Positivism
Postpositivism
Privacy Certificate
Respect for persons
Stanley Milgram’s experiments
on obedience to authority
Verstehen
HIGHLIGHTS
• Philip Zimbardo’s prison-simulation study and Stanley Milgram’s obedience experiments led
to intensive debate about the extent to which deception could be tolerated in social science
research and how harm to subjects should be evaluated.
• Egregious violations of human rights by researchers, including scientists in Nazi Germany and
researchers in the Tuskegee syphilis study, led to the adoption of federal ethical standards for
research on human subjects.
• The 1979 Belmont Report, developed by a national commission, established three basic
ethical standards for the protection of human subjects: respect for persons, beneficence, and
justice.
• The Department of Health and Human Services adopted in 1991 a Federal Policy for the
Protection of Human Subjects. This policy requires that every institution seeking federal
funding for biomedical or behavioral research on human subjects have an institutional review
board (IRB) to exercise oversight.
• The ACJS standards for the protection of human subjects require avoiding harm, obtaining
informed consent, avoiding deception except in limited circumstances, and maintaining
privacy and confidentiality.
• Scientific research should maintain high standards for validity and be conducted and reported
in an honest and open fashion.
• Effective debriefing of subjects after an experiment can help reduce the risk of harm resulting
from the use of deception in the experiment.
• Positivism is the belief that there is a reality quite apart from one’s own perception of it that
is amenable to observation.
• Intersubjective agreement is an agreement by different observers on what is happening in the
natural or social world.

CHAPTER 3 Research Ethics and Philosophies
75
• Postpositivism is the belief that there is an empirical reality but that our understanding of it is
limited by its complexity and by the biases and other limitations of researchers.
• Interpretivism is the belief that reality is socially constructed, and the goal of social science
should be to understand what meanings people give to that reality.
• The constructivist paradigm emphasizes the importance of exploring and representing the
ways in which different stakeholders in a social setting construct their beliefs. Constructivists
interact with research subjects to gradually develop a shared perspective on the issue being
studied.
EXERCISES
Discussing research
1. What policy would you recommend that researchers such as Sherman and Berk follow in
reporting the results of their research? Should social scientists try to correct misinformation
in the popular press about their research, or should they just focus on what is published in
academic journals? Should researchers speak to audiences like at police conventions in order
to influence policies related to their research results?
2. Now go to this book’s study site at www.sagepub.com/bachmanfrccj2e and choose the
Learning From Journal Articles option. Read one article based on research involving human
subjects. What ethical issues did the research pose, and how were they resolved? Does it seem
that subjects were appropriately protected?
3. Outline your own research philosophy. You can base your outline primarily on your reactions
to the points you have read in this chapter, but try also to think seriously about which
perspective seems the most reasonable to you.
4. Researchers should consider their research philosophy as well as their theoretical stance
prior to designing a research project. The “Theories and Philosophies” lesson on the text’s
study site will help you think about the options. To use these lessons, choose one of the four
“Theories and Philosophies” exercises from the opening menu for the Interactive Exercises.
Follow the instructions for entering your answers and responding to the program’s comments.
Finding Research on the Web
1. The Collaborative Institutional Training Initiative (CITI) offers an extensive online training
course in the basics of human subjects protections issues. Go to the public access CITI site at
www.citiprogram.org/rcrpage.asp?affiliation=100 and complete the course in social and
behavioral research. Write a short summary of what you have learned.
2. Philip Zimbardo provides extensive documentation about the Stanford Prison Experiment at
www.prisonexperiment.org. Read several documents that you find on this website, and write
a short report about them.
3. Read the entire ACJS Code of Ethics at www.acjs.org. Discuss the meaning of each research
standard.

FUNDAMENTALS OF RESEARCH IN CRIMINOLOGY AND CRIMINAL JUSTICE
76
4. There are many interesting websites that discuss philosophy of science issues. Read the
summaries of positivism and interpretivism at www.misq.org/archivist/vol/no28/issue1/
EdCommentsV28N1.pdf. What do these summaries add to your understanding of these
philosophical alternatives?
Critiquing Research
1. Investigate the standards and operations of your university’s IRB. Interview one IRB member
and one researcher whose research has been reviewed by the IRB (after receiving the
appropriate permissions!). How well do typical IRB meetings work to identify the ethical
issues in proposed research? Do researchers feel that their proposals are treated fairly? Why
or why not?
2. Continue the debate between positivism and interpretivism with an in-class discussion. Be
sure to review the guidelines for these research philosophies and the associated goals. You
might also consider whether an integrated philosophy is preferable.
3. How do you evaluate the current ACJS ethical code? Is it too strict, too lenient, or just about
right? Are the enforcement provisions adequate? What provisions could be strengthened?
Making Research Ethical
1. Should criminologists be permitted to conduct replications of Zimbardo’s prison simulation?
Of Milgram’s obedience experiments? Can you justify such research as permissible within the
current ACJS ethical standards? If not, do you believe that these standards should be altered
so as to permit this type of research?
2. Why does unethical research occur? Is it inherent in science? Does it reflect “human nature”?
What makes ethical research more or less likely?
3. Does debriefing solve the problem of subject deception? How much must researchers reveal
after the experiment is over as well as before it begins?
Developing a Research Proposal
Now it’s time to consider the potential ethical issues in your proposed study and the research
philosophy that will guide your research. The following exercises involve very critical “Decisions
in Research.”
1. List the elements in your research plans that an IRB might consider to be relevant to the
protection of human subjects. Rate each element from 1 to 5, where 1 indicates no more than
a minor ethical issue and 5 indicates a major ethical problem that probably cannot be
resolved.
2. Write one page for the application to the IRB that explains how you will ensure that your
research adheres to each relevant ASA standard.
3. Draft a consent form to be administered to your subjects when they enroll in your research.
Use underlining and margin notes to indicate where each standard for informed consent
statements is met.

CHAPTER 3 Research Ethics and Philosophies
77
Performing Data Analysis in SPSS or Excel
1. Access HOMICIDE.por. Obtain a pie chart for MURCON, which is a variable that gives the
disposition of a homicide case according to three values: (1) no conviction, (2) a murder
conviction, or (3) some other felony conviction. What do you conclude about this sample of
homicide cases after analyzing the pie chart for this variable?
2. Using these same data, ask for a bar chart to be made for the variable giving the number of
prior arrests the homicide defendant had (PRIARR). What do you conclude about the criminal
history for this sample of homicide defendants?
3. Access YOUTH.por. Ask for a frequency distribution for the variable V63, which provides
information on how often respondents’ parents know where they are when they are away
from home, using the following response options: 1 = never, 2 = sometimes, 3 = usually,
4 = always. You can also ask for a bar chart to be included in the output. What do you
conclude about parents’ knowledge of the whereabouts of their children from this sample of
adolescents?
Student Study Site
The companion Student Study Site for Fundamentals of Research in Criminology and Criminal
Justice can be found at www.sagepub.com/bachmanfrccj2e.
Visit the Student Study Site to enhance your understanding of the chapter content and to
discover additional resources that will take your learning one step further. You can enhance
your understanding of the chapters by using the comprehensive study material, which includes
interactive exercises, e-flashcards, web exercises, practice self-tests, and more. You will also
find special features, such as Learning From Journal Articles, which incorporates Sage’s online
journal collection.
