
User manual
Dream Sta tion
CPAP
CPAP Pro
Auto CPAP
© Koninklijke Philips N.V., 2021. All rights reserved.
Table of Contents
Intended Use ........................................................................................................................................................... 1
Important ................................................................................................................................................................. 1
Warnings .................................................................................................................................................................. 1
Cautions ................................................................................................................................................................... 3
Contraindications .................................................................................................................................................. 3
Symbols Glossary ...................................................................................................................................................4
System Contents .................................................................................................................................................... 5
How to Contact Philips Respironics .................................................................................................................5
System Overview ...................................................................................................................................................6
Installing/Replacing the Air Filters ...................................................................................................................... 7
Where to Place the Device .................................................................................................................................8
Supplying AC Power to the Device ....................................................................................................................8
Connecting the Breathing Circuit ...................................................................................................................... 9
Navigating the Device Screens ..........................................................................................................................10
Starting the Device .............................................................................................................................................. 10
Menu Navigation (Therapy ON) and Optional Humidication Settings .................................................11
Ramp Feature ........................................................................................................................................................11
Menu Navigation (Therapy OFF) ......................................................................................................................12
Bluetooth
®
Wireless Technology .........................................................................................................................16
Check Mask Fit ..................................................................................................................................................... 17
Sleep Progress ......................................................................................................................................................17
Altitude Compensation ......................................................................................................................................17
Device Alerts ........................................................................................................................................................18
Troubleshooting ...................................................................................................................................................22
Accessories ...........................................................................................................................................................24
Traveling with the System ..................................................................................................................................26
Home and Hospital/Institution Cleaning: Device and Humidier Exterior ............................................27
Home and Hospital: Rinsing and Replacing Filters .......................................................................................27
Home and Hospital Cleaning: Non-heated Flexible Tubing ........................................................................27
Service .................................................................................................................................................................... 28
Additional Notices ...............................................................................................................................................28
Specications ........................................................................................................................................................29
Disposal ..................................................................................................................................................................30
EMC Information .................................................................................................................................................31
Limited Warranty ................................................................................................................................... Back Page

1User Manual
Caution: U. S. federal law restricts this device to sale by or on the order of a physician.
Intended Use
The Philips Respironics DreamStation systems deliver positive airway pressure therapy for the treatment of Obstructive Sleep Apnea in
spontaneously breathing patients weighing over 30 kg (66 lbs). It is for use in the home or hospital/institutional environment.
Important
The device is to be used only on the instruction of a licensed physician. Your home care provider will make the correct pressure
settings and device congurations including accessories, according to your health care professional’s prescription.
Several accessories are available to make your OSA treatment with the DreamStation system as convenient and comfortable as
possible.
Warning: Use only the cleaning methods outlined in your user manual. Cleaning and disinfection methods using Ozone-based
products are not approved or recommended, and may adversely affect your device safety and performance.
Warnings
A warning indicates the possibility of injury to the user or the operator.
• This manual serves as a reference. The instructions in this manual are not intended to supersede the health care professional’s
instructions regarding the use of the device.
• The prescription and other device settings should only be changed on the order of the supervising physician.
• The operator should read and understand this entire manual before using the device.
• This device is not intended for life support.
• The device should only be used with compatible patient interfaces (e.g., masks, circuits and exhalation ports). Proper operation of the
device with other circuits has not been veried by Philips Respironics and is the responsibility of the health care professional. A mask
should not be used unless the device is turned on and operating properly. The exhalation port(s) associated with the mask should
never be blocked. Explanation of the Warning: The device is intended to be used with special masks or connectors that have
exhalation ports to allow continuous ow of air out of the mask. When the device is turned on and functioning properly, new air
from the device ushes the exhaled air out through the mask exhalation port. However, when the device is not operating, enough
fresh air will not be provided through the mask, and exhaled air may be rebreathed. Rebreathing of exhaled air for longer than
several minutes can in some circumstances lead to suffocation.
• An exhalation port is required. Do not block the exhalation port. This can reduce airow and result in rebreathing of exhaled air.
• At low expiratory pressures, the ow through the exhalation port may be inadequate to clear all exhaled gas from the tubing –
some rebreathing may occur.
• If you are using a full face mask (a mask covering both your mouth and your nose), the mask must be equipped with a safety
(entrainment) valve.
• To ensure that you receive the safe, effective therapy prescribed for you, use only Philips Respironics accessories. The use
of accessories, transducers, and cables other than those specied by Philips Respironics may result in increased emissions or
decreased immunity of the device.
• When using oxygen with this system, the oxygen supply must comply with local regulations for medical oxygen.
• Oxygen supports combustion. Oxygen should not be used while smoking or in the presence of an open ame.
• When using oxygen with this system, turn the device on before turning on the oxygen. Turn the oxygen off before turning the
device off. This will prevent oxygen accumulation in the device. Explanation of the Warning: When the device is not in
operation and the oxygen ow is left on, oxygen delivered into the tubing may accumulate within the device’s enclosure. Oxygen
accumulated in the device enclosure will create a risk of re.
• When using oxygen with this system, a Philips Respironics Pressure Valve must be placed in-line with the patient circuit between
the device and the oxygen source. The pressure valve helps prevent the backow of oxygen from the patient circuit into the
device when the unit is off. Failure to use the pressure valve could result in a re hazard.
• Do not connect the device to an unregulated or high pressure oxygen source.
• Do not use the device in the presence of a ammable anaesthetic mixture in combination with oxygen or air, or in the presence
of nitrous oxide.
• Do not use the device near a source of toxic or harmful vapors.
• Do not use this device if the room temperature is warmer than 35° C (95° F). If the device is used at room temperatures warmer
than 35° C (95° F), the temperature of the airow may exceed 43° C (109° F). This could cause irritation or injury to your airway.
• Do not operate the device in direct sunlight or near a heating appliance because these conditions can increase the temperature of
the air coming out of the device.
• Contact your health care professional if symptoms of sleep apnea recur.
• If you notice any unexplained changes in the performance of this device, if it is making unusual or harsh sounds, if it has been
dropped or mishandled, if water is spilled into the enclosure, or if the enclosure is broken, disconnect the power cord and
discontinue use. Contact your home care provider.
2 User Manual
• Repairs and adjustments must be performed by Philips Respironics-authorized service personnel only. Unauthorized service could
cause injury, invalidate the warranty, or result in costly damage. Contact your home care service provider for maintenance.
• Do not use any accessories, detachable parts, and materials not recommended by Philips Respironics. Incompatible parts or
accessories can result in degraded performance.
• Use only approved cables and accessories. Misuse may affect EMC performance and should be avoided.
• The Health Industry Manufacturers Association recommends that a minimum separation of six inches be maintained between
a wireless phone and a pacemaker to avoid potential interference with the pacemaker. The DreamStation on-board Bluetooth
communication should be considered a wireless phone in this regard.
• Use only power cords supplied by Philips Respironics for this device. Use of power cords not supplied by Philips Respironics may cause
overheating or damage to the device and may result in increased emissions or decreased immunity of the equipment or system.
• The device should not be used while stacked or in close approximation to other non-approved devices.
• Portable and Mobile RF Communications Equipment can affect Medical Electrical Equipment. See the EMC section of this manual
for distances to observe between RF Generators and the device to avoid interference.
• Do not use this device near active high frequency surgical equipment and the Radio Frequency shielded room of a Medical
Electrical system for magnetic resonance imaging, where the intensity of electromagnetic disturbances is high.
• MR Unsafe - Projectile hazard. Keep outside of MRI scan room (Zone IV).
• Use of this equipment adjacent to or stacked with other equipment should be avoided because it could result in improper
operation. If such use is necessary, this equipment and the other equipment should be observed to verify that they are operating
normally.
• Do not pull or stretch the tubing. This could result in circuit leaks.
• Do not cover the tubing with a blanket or heat it in an incubator or with an overhead heater. This can affect the quality of the
therapy or injure the patient.
• Inspect the tubing for damage or wear. Discard and replace the tubing as necessary.
• Periodically inspect electrical cords and cables for damage or signs of wear. Discontinue use and replace if damaged.
• Never operate the device if any parts are damaged or if it is not working properly. Replace damaged parts before continuing use.
• To avoid electrical shock, always unplug the power cord from the wall outlet before cleaning the device.
• Do not immerse the device in any uids or spray the device with water or cleaners. Clean the device with a cloth dampened with
an approved cleaner.
• If the device is used by multiple persons (such as rental devices), a low-resistance, main ow bacteria lter should be installed in-
line between the device and the circuit tubing to prevent contamination.
• If using a humidier, do not use the humidier at an altitude above 2286 m (7500 ft) or outside a temperature of 5° C to 40° C
(41° F to 104° F). Using the humidier outside of this temperature range or above this altitude can affect the quality of the therapy
or injure the patient.
• To prevent disconnection of the tubing or tubing system during use, only tubes in compliance with ISO 5367 or ISO 80601-2-74
should be used.
• Humidication can increase the resistance of the bacteria lter and the operator must monitor the bacteria lter frequently for
increased resistance and blockage to ensure the delivery of the therapeutic pressure.
• Be sure to route the power cord to the outlet in a way that will prevent the cord from being tripped over or interfered with by
chairs or other furniture.
• To avoid strangulation hazards, ensure that all cords connected to the device are properly routed.
• This device is activated when the power cord is connected.
• For safe operation when using a humidier, the humidier must always be positioned below the breathing circuit connection at
the mask. The humidier must be level for proper operation.
• Use only Philips Respironics recommended pulse oximeter and sensors. Use of incompatible sensors can result in inaccurate pulse
oximeter performance.
• Do not use a damaged pulse oximeter or sensor.
• Before use, carefully read these instructions and the instructions for use provided with the pulse oximeter and sensor.
Note: Please see the “Limited Warranty” section of this manual for information on warranty coverage.
3User Manual
Cautions
A Caution indicates the possibility of damage to the device.
• Medical electrical equipment needs special precautions regarding EMC and needs to be installed according to EMC information.
Contact your home care provider regarding EMC installation information.
• Do not use antistatic or conductive hoses or conductive patient tubing with the device.
• Pins of connectors marked with the ESD warning symbol shall not be touched and connections shall not be made without
special precautions. Precautionary procedures include methods to prevent build-up of electrostatic charge (e.g., air conditioning,
humidication, conductive oor coverings, non-synthetic clothing), discharging one’s body to the frame of the equipment or
system or to earth. It is recommended that all individuals that will handle this device understand these precautionary procedures
at a minimum as part of their training.
• Before operating the device, ensure that the SD card/lter access door and the modem access door are both closed whenever any
of the accessories such as the Link Module or Modem are not installed. Refer to the instructions that came with your accessory.
• Condensation may damage the device. If this device has been exposed to either very hot or very cold temperatures, allow it to
adjust to room temperature (operating temperature) for 24 hours before starting therapy. Do not operate the device outside of
the environmental operating ranges listed in the “Specications” section later in this manual.
• Make sure the air inlet lter area on the side of the device is not blocked by bedding, curtains, or other items. Air must ow
freely around the device for the system to work properly.
• Do not place the device directly onto carpet, fabric, or other ammable materials.
• Do not place the device in or on any container that can collect or hold water.
• Do not plug the device into an outlet controlled by a wall switch.
• A properly installed, undamaged Philips Respironics blue pollen lter is required for proper operation.
• Tobacco smoke may cause tar build-up within the device, which may result in the device malfunctioning.
• Dirty inlet lters may cause high operating temperatures that may affect device performance. Regularly examine the inlet lters as
needed for integrity and to check for accumulated debris.
• Never install a wet lter into the device. You must ensure sufcient drying time for the rinsed lter.
• Always ensure that the DC power cord securely ts into your therapy device prior to use. Contact your home care provider or
Philips Respironics to determine if you have the appropriate DC cord for your specic therapy device.
• When DC power is obtained from a vehicle battery, the device should not be used while the vehicle’s engine is running. Damage
to the device may occur.
• Only use a Philips Respironics DC Power Cord and Battery Adapter Cable. Use of any other system may cause damage to the device.
• Do not immerse the device or allow any liquid to enter the enclosure or the inlet lter.
• Do not steam autoclave the device. Doing so will destroy the device.
• Do not use harsh detergents, abrasive cleaners, or brushes to clean the system.
• Only the cleaning procedures listed in this manual are recommended by Philips Respironics. Use of other cleaning processes, not
specied by Philips Respironics, may affect the performance of the product.
Contraindications
When assessing the relative risks and benets of using this equipment, the clinician should understand that this device can deliver
pressures up to 20 cm H
2
O. In the event of certain fault conditions, a maximum pressure of 40 cm H
2
O is possible. Studies have
shown that the following pre-existing conditions may contraindicate the use of CPAP therapy for some patients:
• Bullous Lung Disease
• Pathologically Low Blood Pressure
• Bypassed Upper Airway
• Pneumothorax
• Pneumocephalus has been reported in a patient using nasal Continuous Positive Airway Pressure. Caution should be used when
prescribing CPAP for susceptible patients such as those with: cerebral spinal uid (CSF) leaks, abnormalities of the cribriform
plate, prior history of head trauma, and/or pneumocephalus. (Chest 1989; 96:1425-1426)
The use of positive airway pressure therapy may be temporarily contraindicated if you exhibit signs of a sinus or middle ear
infection. Not for use with patients whose upper airways are bypassed. Contact your health care professional if you have any
questions concerning your therapy.

4 User Manual
Symbols Glossary
The following symbols may appear on the device, power supply and accessories. For a full list and description of the symbols used on
the device and its packaging, see www.symbols.philips.com.
Symbol Denition
Operator’s manual; operating instructions
Consult instructions for use.
MR unsafe
Do not use device in a Magnetic Resonance (MR) environment.
AC power (Alternating current)
Indicates on the rating plate that the equipment is suitable for alternating current only; to identify
relevant terminals.
DC power (Direct current)
Indicates on the rating plate that the equipment is suitable for direct current only; to identify relevant
terminals.
IP22
Drip Proof Equipment
Caution, consult accompanying documents.
Electrostatic sensitive devices (ESD warning symbol)
Attention – Observe precautions for handling electrostatic sensitive devices.
Class II equipment (Double Insulated)
To identify equipment meeting the safety requirements specied for Class II equipment.
Type BF applied part
To identify a type BF applied part complying with IEC 60601-1.
For indoor use only
Equipment is designed primarily for indoor use.
Approved for airline use.
Separate collection for electrical and electronic equipment per EC Directive 2012/19/EU.
Bluetooth
®
symbol
Indicates the device has Bluetooth capabilities.
Non-ionizing electromagnetic radiation
Indicates that the equipment includes RF transmitters.
Oximeter Connection
Serial Connection
Identies a connector for a serial data connection.
Keep away from sunlight
Indicates the medical device needs protection from light sources.
Prescription device
Caution: U. S. federal law restricts this device to sale by or on the order of a physician.
Do not disassemble.
Therapy on/off (Stand-by)
Identies the button to turn therapy on or off (puts the device in a stand-by condition).

5User Manual
Symbol Denition
Ramp (Variability)
To identify the control device by means of which a quantity is controlled. The controlled quantity
increases with the gure width.
Keep dry
Indicates the medical device that needs to be protected from moisture.
Fragile, handle with care
Indicates the medical device can be broken or damaged if not handled carefully.
Temperature limit
Indicates the storage temperature limits to which the medical device can be safely exposed.
Humidity limitation
Indicates the range of humidity to which the medical device can be safely exposed.
Manufacturer
Indicates the medical device manufacturer.
Date of manufacture
Indicates the date when the medical device was manufactured.
CC
Country of Manufacture - Indicates the country of manufacture of the product. Note: When applied to
the label, “CC” is replaced by the country code.
Reorder number
Indicates the manufacturer’s catalogue number so the medical device can be identied.
Serial number
Identies the manufacturer’s serial number for the medical device.
MD
Medical Device - Indicates that the item is a medical device.
System Contents
Your DreamStation system may include the following items:
• Device • SD card
• User manual • Flexible tubing
• Carrying case • Reusable blue pollen lter
• Power cord • Disposable light-blue ultra-ne lter (optional)
• Power supply ( 1118499) • Humidier (optional)
Note: If any of these items are missing, contact your home care provider.
How to Contact Philips Respironics
Should you experience trouble with this equipment or require assistance setting up, using, or maintaining the device
or accessories, please contact your home care provider. If you need to contact Philips Respironics directly, call
the Philips Respironics Customer Service department at 1-800-345-6443 or 1-724-387-4000. You can also use the
following address:
Respironics, Inc.
1001 Murry Ridge Lane
Murrysville, PA 15668
6 User Manual
System Overview
The DreamStation CPAP is a Continuous Positive Airway Pressure therapy device designed for the treatment
of Obstructive Sleep Apnea (OSA). The DreamStation CPAP Pro can also deliver CPAP-check therapy, and the
DreamStation Auto CPAP can also deliver CPAP-Check and Auto-CPAP therapy. Your home care provider will choose
the appropriate pressure settings for you.
When prescribed for you, the device provides several special features to help make your therapy more comfortable.
The ramp function allows you to lower the pressure when you are trying to fall asleep. The air pressure will gradually
increase until your prescription pressure is reached. Also, the Flex comfort feature provides you with pressure relief
when you exhale during therapy.
Several accessories are also available for use with your device. Contact your home care provider to purchase any
accessories not included with your system.
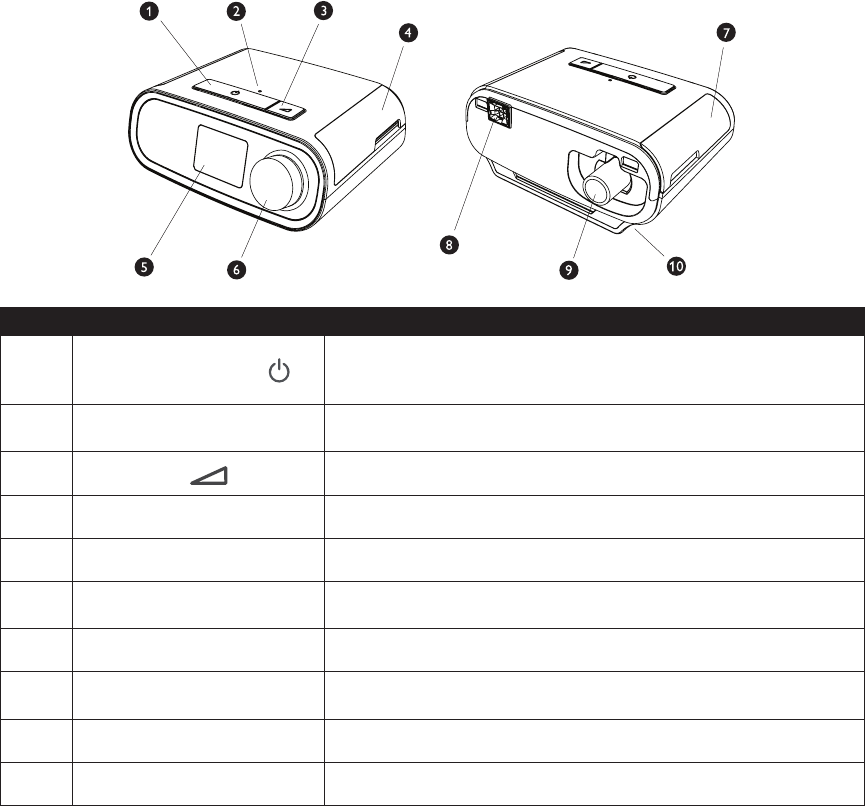
This gure illustrates some of the device features, described in the following table.
# Device Feature Description
1
Therapy On/Off Button
Starts and stops the airow for therapy.
If the Therapy On/Off button LED is ashing, you may have a pending
message. Press or turn the knob to display the message.
2 Ambient Light Sensor
Detects room light levels and adjusts brightness of LCD Display
Screen.
3
Ramp Button
Activates the ramp feature during therapy.
4 Door, SD card & Filter Access This door lifts open for access to the SD card and lter area.
5 LCD Display Screen This is the User Interface for the therapy device.
6 Control Dial
Turn the dial to scroll between options on the screen. Press the dial to
choose an option.
7 Door, Accessory Access This door lifts open for access to the (optional) accessories.
8 Humidier Connector
Humidier connects to the back of the therapy device. The humidier
pin connector will attach here.
9 Air Outlet Port Connect the tubing here.
10 Power Inlet Connect the power cord here.
7User Manual
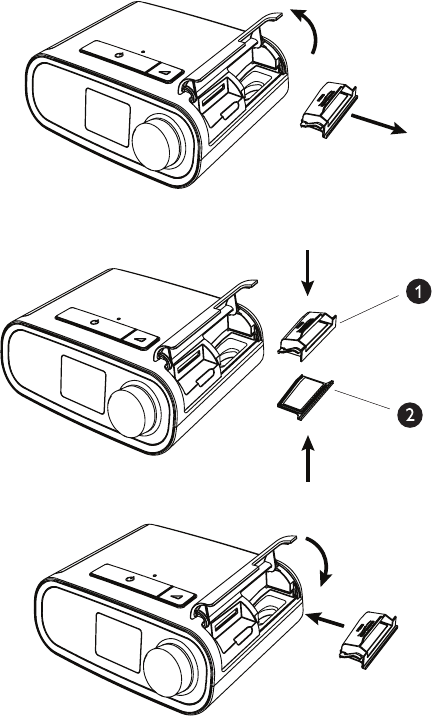
Installing/Replacing the Air Filters
Caution: A properly installed, undamaged Philips Respironics blue pollen lter is required for proper operation.
The device uses a reusable blue pollen lter that can be rinsed and and a disposable light-blue ultra-ne lter. The
reusable blue lter screens out normal household dust and pollens, while the light-blue ultra-ne lter provides more
complete ltration of very ne particles. The reusable blue lter must be in place at all times when the device is
operating. The ultra-ne lter is recommended for people who are sensitive to tobacco smoke or other small particles.
The reusable blue lter is supplied with the device. A disposable light-blue ultra-ne lter may also be included. If your
lter is not already installed when you receive your device, you must at least install the reusable lter before using the
device.
This device has an automatic air lter reminder. Every 30 days, the device will display a message reminding you to
check your lters and replace them as directed.
Note: This message is a reminder only. The device does not detect the performance of the lters nor does it
recognize when a lter has been rinsed or replaced.
1. Lift up on the lter access door and swing open. If replacing, pull out the old lter assembly.
2. If applicable, place a dry, reusable blue pollen lter (1) on top of a new, optional disposable light-blue ultra-ne lter
(2) and rmly snap them together.
3. Place the new lter assembly back in the side of the therapy device. Swing the door closed.
8 User Manual
Where to Place the Device
Place the device on a rm, at surface somewhere within easy reach of where you will use it at a level lower than
your sleeping position. Make sure the device is away from any heating or cooling equipment (e.g., forced air vents,
radiators, air conditioners).
Note: When positioning the device, make sure that the power cable is accessible because removing power is the
only way to turn off the device.
Caution: Make sure the lter area on the side of the device is not blocked by bedding, curtains, or other items.
Air must ow freely around the device for the system to work properly.
Caution: Do not place the device directly onto carpet, fabric, or other ammable materials.
Caution: Do not place the device in or on any container that can collect or hold water.
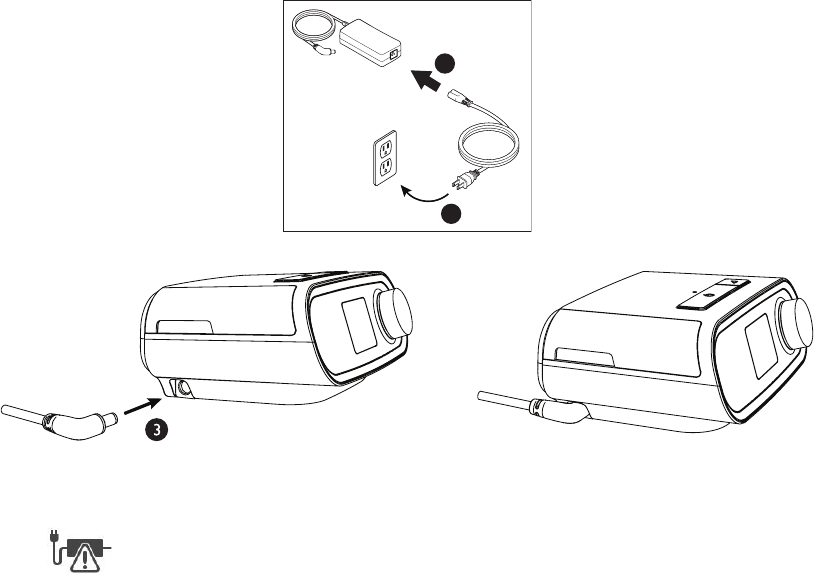
Supplying AC Power to the Device
Complete the following steps to operate the device using AC power:
1. Plug the socket end of the AC power cord (included) into the power supply (also included).
2. Plug the pronged end of the AC power cord into an electrical outlet that is not controlled by a wall switch.
1
1
2
2
3. Plug the power supply cord’s connector into the power inlet on the side of the device.
4. Verify that the plug at the side of the device, at the power supply, and at the electrical outlet are fully inserted. This
will help to ensure that a secure, reliable electrical connection has been made.
Note: If the following Check Power icon appears on the screen, please repeat step 4.
Important: To remove AC power, disconnect the power supply cord from the electrical outlet.
Warning: Periodically inspect electrical cords and cables for damage or signs of wear. Discontinue use and replace
if damaged.
9User Manual
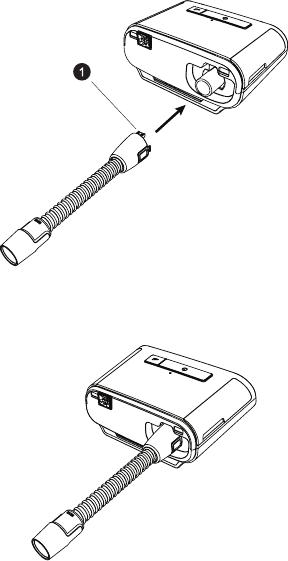
Connecting the Breathing Circuit
To use the system, you will need the following accessories in order to assemble the recommended breathing circuit:
• Philips Respironics interface (nasal mask or full face mask) with integrated exhalation port, or Philips Respironics
interface with a separate exhalation device (such as the Whisper Swivel II)
• Philips Respironics exible tubing, 1.83 m (6 ft.)
• Philips Respironics headgear (for the mask)
To connect your breathing circuit to the device, complete the following steps:
1. Connect the exible tubing to the air outlet on the therapy device.
To connect heated tubing (shown) to the air outlet on the back of the therapy device, line up the connector (1) at
the top of the heated tube to the top of the air outlet port on the back of the device.
2. Press the heated tubing into place over the air outlet port until the tabs on the side of the tube click into place in
the slots on the sides of the outlet port.
If you are using standard tubing (not shown), simply slide the tubing over the air outlet port on the device.
Note: If you are using the optional 12 mm tubing, an adaptor is required to connect to the therapy device.
Note: If required, connect a bacteria lter to the device air outlet, and then connect the exible tubing to the
outlet of the bacteria lter. When using the bacteria lter, the device performance may be affected. However, the
device will remain functional and deliver therapy.
3. If you are using the optional 12 mm performance tubing, connect the provided mask adapter to the mask
connection end of the tubing.
4. Connect the tubing to the mask. For proper placement and positioning, refer to the instructions that came with
your mask.
Warning: Do not pull or stretch the tubing. This could result in circuit leaks.
Warning: Inspect the tubing for damage or wear. Discard and replace the tubing as necessary.
5. Attach the headgear to the mask if necessary. Refer to the instructions that came with your headgear.
10 User Manual
Navigating the Device Screens
The User Interface (UI) on this device allows you to adjust the device settings and view information about your
therapy. The UI is comprised of the display screen and the control dial. Rotate the control dial in either direction to
scroll through the menu options on the display screen.
Note: The display is not a touch screen. You must use the control dial to navigate the device menu.
To adjust a setting:
1. Rotate the control dial to your desired menu option.
2. Press the control dial to select that setting.
3. Rotate the control dial to change the setting.
4. Press the control dial again to save the change.
Note: The rotate dial icon on any screen indicates to rotate the dial to perform an action. The click dial icon
on any screen indicates to press the dial to perform an action.
Note: Pressing the dial when the down arrow appears on any screen will take you to a sub-menu with more
menu options. Pressing the dial when the up arrow appears on any sub-menu will return you back to the main
menu.
Note: The screens shown throughout this manual are examples for reference only. Actual screens may vary based
upon device model and provider settings.
Starting the Device
1. Ensure power is supplied to the device. The rst screen to display will be the Philips Respironics logo, followed by
the device model screen, and then the Home screen.
Home Screen
The rst time the device is powered on, a pop-up may prompt you to set the time on the device. The default setting
is Greenwich Mean Time, but if prompted you may adjust the time in 30 minute increments to match your local
time zone. The time can always be adjusted under the “My Setup” menu.
Note: This time setting is not displayed as a clock function on the device. It is only used to align your therapy data
for your Provider’s data reports.
2. Put on your mask assembly. Refer to the instructions supplied with the mask.
3. Press the Therapy button ( ) on top of the device to turn on airow and begin therapy. The current delivered
pressure will display on the screen.
4. Make sure that no air is leaking from your mask. If necessary, adjust the mask and headgear until the air leak stops.
See the instructions provided with your mask for more information.
Note: A small amount of mask leak is normal and acceptable. Correct large mask leaks or eye irritation from an air
leak as soon as possible.
5. If you are using the device in a bed with a headboard, try placing the tubing over the headboard. This may reduce
tension on the mask.
6. Press the Therapy button again to turn off therapy.
Note: During therapy, if there is a mains interruption (i.e. power loss) the device will return to the Home screen
once power is restored. You may resume therapy as needed.
11User Manual
Menu Navigation (Therapy ON) and Optional Humidication Settings
While the device is delivering therapy, you can adjust Tube Temperature or Humidier Settings. Rotate the control dial
to choose either setting. Press and rotate the dial to change the setting.
Note: If you are using the Humidier without the Heated Tube, simply just rotate the control dial to change the
Humidier setting.
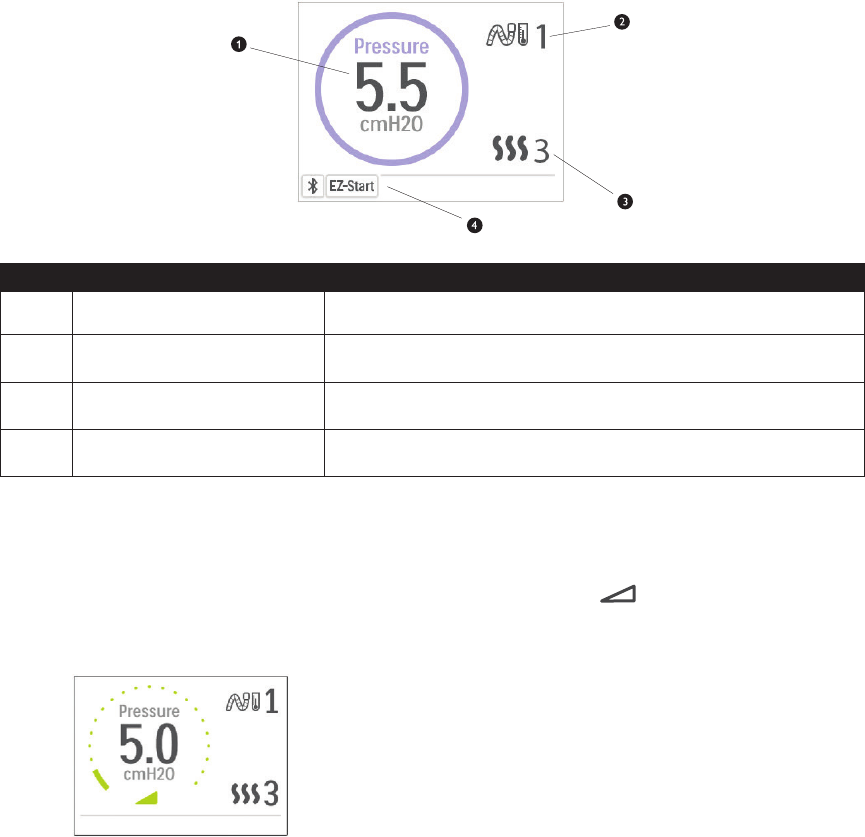
Therapy Pressure Screen
# Feature Description
1 Therapy Pressure Displays the current delivered pressure.
2
Adjustable Tube Temperature
Setting
You can change this setting from 0 to 5. Only displays when optional
heated tube is connected.
3 Adjustable Humidier Setting
You can change this setting from 0 to 5. Only displays when
humidier is attached.
4 Enabled Features
Depending on setup, certain enabled therapy features will display
here.
Ramp Feature
The device is equipped with an optional ramp feature that your home care provider can enable or disable. This
feature reduces the air pressure when you are trying to fall asleep and then gradually increases (ramps) the pressure
until your prescription setting is reached, allowing you to fall asleep more comfortably.
If ramp is enabled on your device, after you turn on the airow, press the Ramp ( ) button on the top of the
device. You can use the Ramp button as often as you wish during the night.
When you click the ramp button, the Therapy screen will change to reect the Ramp pressure, and the green circle
will reect the gradual increase in pressure.
Ramp Pressure Screen
Your device has two ramp modes. Your Provider will select the one that is most appropriate for you. The standard
ramp mode increases pressure at a steady rate. Alternately, the SmartRamp mode maintains a constant lower
pressure until the device detects that you require more pressure.
12 User Manual
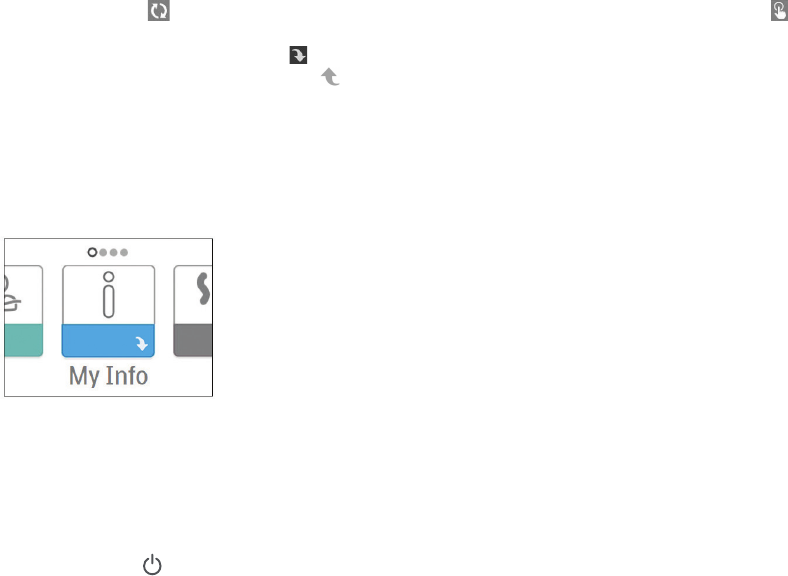
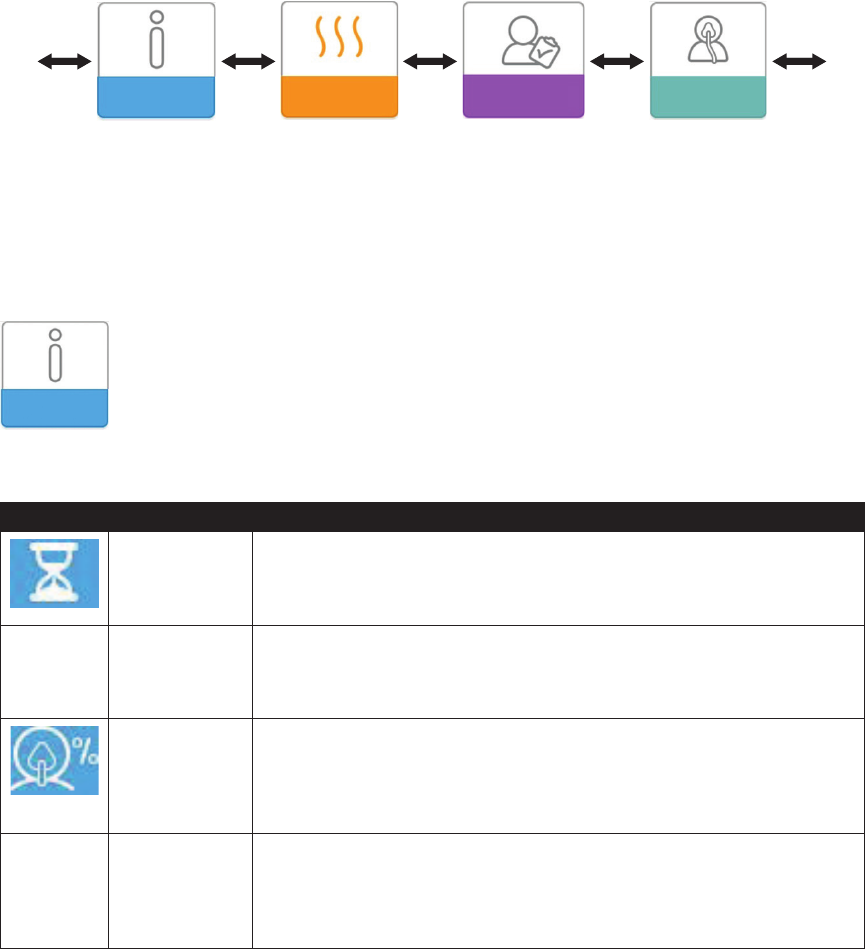
Menu Navigation (Therapy OFF)
From the Home screen, you can scroll between the following menus. Only the menus available and enabled on your
device will display.
My Info Preheat My Provider My Setup
My Info: This menu provides summary statistics of your therapy use.
Preheat (if available): This function lets you warm up your humidier for 30 minutes before starting a therapy
session.
My Provider: This menu contains information that your provider may direct you to read to them so they can
better assist you over the phone.
My Setup: This menu contains comfort settings that you can adjust as needed.
My Info:
When you select “My Info”, you will be able to view the following screens. These screens will only display if they are
available and enabled on your device. You cannot change settings in the My Info menu. These screens are only for
reference. Your home care provider may periodically ask you for this information.
Icon Text Description
Therapy Hours This screen displays the amount of time the user is actually receiving therapy
on the device for the most recent 1 day time frame. It also displays the
average amount of time the patient is actually receiving therapy over the last 7
days and 30 days.
AHI AHI This screen displays the nightly Apnea/Hypopnea indices (AHI) value for the most
recent 1 day time frame. It also displays the average of these individual nightly
AHI values over a 7 day and a 30 day time frame. Only available on CPAP Pro and
Auto CPAP devices.
Mask Fit Displays the value “100% minus Large Leak”. Large Leak is the percentage of
time that the mask leak was so high that it is no longer possible for the device
to identify respiratory events with statistical accuracy. Displays the value for the
most recent 1 day, as well as the values over the last 7 days and 30 days. Only
available on CPAP Pro and Auto CPAP devices.
Periodic
Breathing
Periodic
Breathing
Displays the percentage of time that the user experienced periodic breathing.
Displays the value for the most recent 1 day time frame, as well as values for the
last 7 days and 30 days. If you observe a large increase in the percent of time in
periodic breathing indicated here, contact your home care provider for assistance.
Only available on CPAP Pro and Auto CPAP devices.
13User Manual
Icon Text Description
90%
Pressure
90% Pressure This screen displays the nightly value of 90% Pressure for the most recent 1 day
time frame. It also displays the average of these individual nightly values of 90%
Pressure over a 7 day and a 30 day time frame. Available on the Auto model.
Preheat (if available):
Preheat On Screen Preheat Off Screen
Note: The Preheat menu will only display if it is available on your device.
When using a humidier, the device can preheat the water tank for up to 30 minutes prior to starting therapy.
In order to activate the preheat mode, the blower must be “off” and a humidier must be attached. When “Preheat”
is selected, you will be able to turn the control dial to choose between “on” or “off”. Press the control dial again to
make your selection. During the 30 minute preheat, you will still be able to use the control dial to select other menu
options from the Home screen.
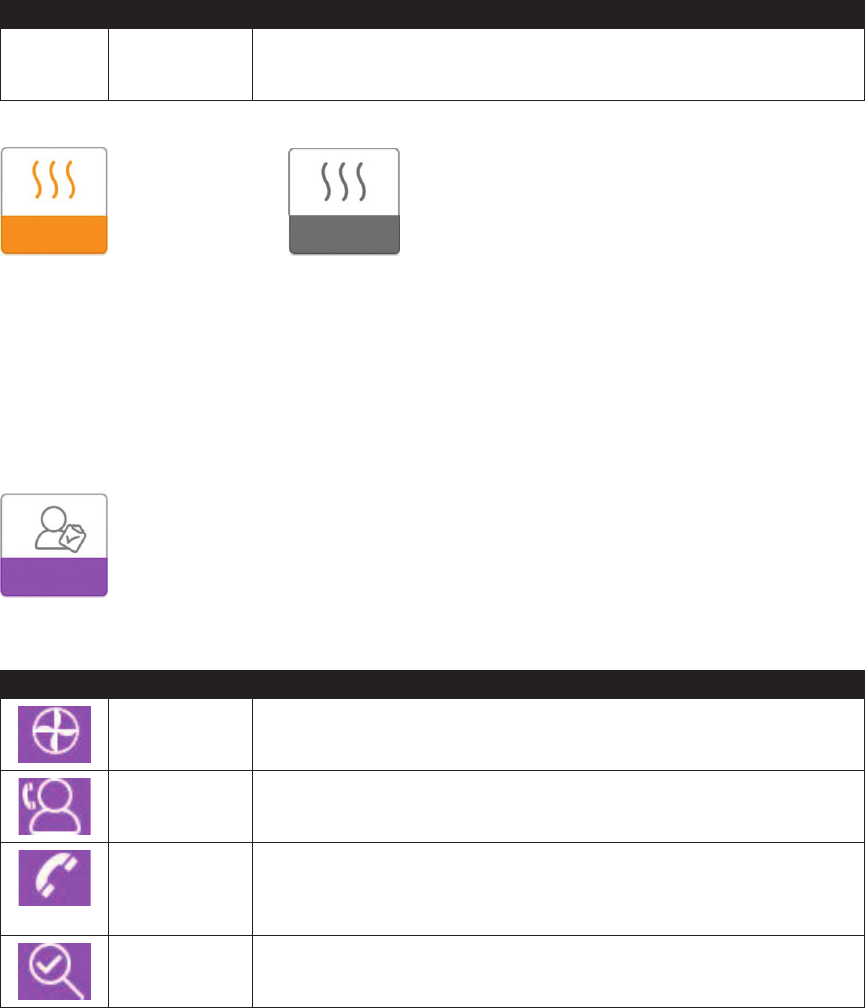
My Provider:
When you select “My Provider”, you will be able to view the following screens. These screens will only display if they
are available and enabled on your device. You cannot change settings in the Provider menu. These screens are only for
reference. Your home care provider may periodically ask you for this information.
Icon Text Description
Device Info This screen displays your therapy device information: serial number, model and
software version.
Provider Contact
Info
This screen will display the contact information for your provider if it has been
uploaded to your device.
Phone-In This screen displays the total therapy hours for the device, the total blower
hours, the total number of days used when the sessions were greater than 4
hours, and a compliance check number used by your home care provider to
validate that the data provided by you is the data taken from this screen.
Compliance This screen displays your start date, the total number of days used when the
sessions were greater than 4 hours, and a check code number used by your
home care provider.
14 User Manual
Icon Text Description
VIC90 VIC90 This Visual Inspection Check screen will display a check code number created
from information gathered over the most recent 90 day period. This 15 digit
number will display as: xxx.xxxx.xxxx.xxxx. Your home care provider may
periodically ask you for this information. This screen will not display if your device
has a modem installed.
A-TRIAL A-Trial If Auto-Trial mode is available, this screen displays Days: xx/xx (where xx/xx is
the number of accumulated trial days / number of selected trial days).
Upload Allows user to initiate a modem call when an optional Cellular Modem or Wi-Fi
Accessory is installed. Signal strength is indicated at the top right of this screen.
After the modem upload has nished, the screen will either display a green
checkmark with the text “Completed” to indicate a successful upload, or a red X
with the text “Failed” to indicate an unsuccessful upload. If the upload fails, initiate
an upload a second time, or contact your home care provider if the issue persists.
This screen is only available if your device has a modem installed.
Performance
Check
Your device is equipped with a self-diagnostic tool called “Performance Check.”
This tool can evaluate your device for certain errors. It also allows you to share
key device settings with your home care provider. Use Performance Check when
directed to by your home care provider.
At conclusion of the scan, the screen displays a green checkmark if no issue is
detected. If device displays a red “X,” please contact your home care provider for
assistance.
My Setup:
When you select “My Setup”, you will be able to view the following screens. These screens will only display if they are
available and enabled on your device. You can change the settings in the Setup menu.
Icon Text Description
Ramp This displays the ramp starting pressure. You can increase or decrease the ramp
starting pressure in 0.5 cm H
2
O increments.
Ramp Time When you set the Ramp time, the device increases the pressure from the value
set on the Ramp screen to the therapy pressure setting over the length of time
specified here.
Flex This allows you to adjust the level of air pressure relief that you feel when you
exhale during therapy. Your home care provider can enable or disable this
feature. When your provider enables Flex, a level will already be set for you on
the device. You can increase or decrease the setting from 1 to 3. The setting of
“1” provides a small amount of pressure relief, with higher numbers providing
additional relief.
Note: If a lock icon is displayed on this screen, it indicates that your
provider has locked this setting and you cannot change it.

15User Manual
Icon Text Description
Humidication This displays the Humidication Mode being used. You can choose between
Fixed or Adaptive Humidication. If a heated tube is being used, the device will
automatically switch to Heated Tube Humidication Mode. A “lock” symbol will
appear next to the mode setting indicating that so long as the heated tube is
attached to the device, this mode cannot be changed. However, the heater plate
and tube temperature settings can still be adjusted on the device Therapy screen
as normal.
Mask Type This setting allows you to adjust the level of air pressure relief based on the
specic Philips Respironics mask. Each Philips Respironics mask may have a
“System One” resistance control setting. Contact your home care provider if
you cannot nd this resistance setting for your mask.
Note: If a lock icon is displayed on this screen, it indicates that your
provider has locked this setting and you cannot change it.
Tube Type This setting allows you to select the correct size diameter tubing that you are
using with the device. You can choose either (22) for the Philips Respironics 22
mm tubing, (15) for the Philips Respironics 15 mm tubing, or (12) for the optional
Philips Respironics 12 mm tubing. When using Heated Tubing, the device will
automatically change this setting to the appropriate tubing type (HT15) and you
will not be able to change it.
Note: Tubing is identied on the cuff with the tubing identier symbol: “12”,
“15”, or “15H”. 22 mm tubing contains no symbol.
Note: If a lock icon is displayed on this screen, it indicates that your
provider has locked this setting and you cannot change it.
Language This feature allows you to choose which language to display on the interface. You
can choose English or Spanish.
Check Mask Fit This feature allows you to check the t of your mask prior to starting therapy.
This is done by measuring the amount of leak.
Modem Allows you to turn modem off temporarily or turn it back on. When modem
is turned off, it will automatically turn on again after 3 days. Only displays when
modem is installed.
Bluetooth Allows you to turn Bluetooth off and on. Also, it allows you to clear the pairing
with a compatible Bluetooth device.
Time Allows you to adjust the time. The default setting is Greenwich Mean Time, but
you may adjust the time in 30 minute increments to match your local time zone.
Note: This time setting is not displayed as a clock function on the device. It is
only used to align your therapy data for your Provider’s data reports.

16 User Manual
Bluetooth
Wireless Technology
Your device has Bluetooth wireless technology. which is one method by which you can transfer your therapy device’s
data to DreamMapper. DreamMapper is a mobile and web-based system designed to help Obstructive Sleep Apnea
(OSA) patients enhance their sleep therapy experience.
Pairing your therapy device to your Bluetooth enabled Mobile Device
Note: You can only pair your therapy device to one mobile device at any given time.
Note: Pairing works best when your therapy device and mobile device are in the same room.
Note: The current version of DreamMapper will guide you through these instructions.
Note: After initiating pairing, you will have 30 seconds to complete the setup. After this time, it will be
cancelled automatically.
Follow the steps below to manually pair to your mobile phone or tablet.
1. With your therapy device powered up and the blower off, initiate Bluetooth Setup from the DreamMapper mobile
app.
2. If you need to select from a list of available Bluetooth devices, the therapy device will appear as “PR BT XXXX”
(XXXX will be the last four digits of the serial number listed on your therapy device).
3. You will be required to conrm pairing via one of these two methods:
• Your mobile device may ask you to enter a PIN code
The following icon will appear on your therapy device screen with “Pair?”:
Rotate the therapy device’s Control Dial to select “yes,” and press the Control Dial. Your therapy device will display
a 6 digit PIN. Enter this PIN on your mobile device to complete pairing.
• Your mobile device may ask you to conrm a PIN code
The following icon will appear on your therapy device screen with a 6 digit PIN and “Pair?”:
Verify that the PIN is the same on both the therapy device and the mobile device. If so, rotate the therapy device’s
Control Dial to select “yes” and press the Control Dial. Then, accept on the mobile device to complete pairing.

17User Manual
Check Mask Fit
The optional check mask t feature can be enabled or disabled by your home care provider. This feature allows you to
check the t of your mask prior to starting therapy. This is done by measuring the amount of leak. Put on your mask
assembly. Refer to your mask instructions if needed. Navigate to the Check Mask Fit screen under “My Setup” and
press the control dial to initiate the check.
The device will deliver a test pressure while the screen counts down 40 seconds. A green bar indicates good t, while
a red bar indicates improvement is needed. After the test, normal therapy will start and the screen will either display
a green checkmark or a red “X”. The green checkmark indicates that the leak found allows for optimal performance
of the device. The red “X” indicates that the leak may affect device performance, however, the device will remain
functional and deliver therapy.
Check Mask Fit Screen
Note: If you choose to try to improve your mask t, you can stop therapy, adjust the t of your mask, and rerun
the check mask t. Please refer to the instructions that came with your mask and headgear for the proper tting
procedure.
Sleep Progress
Your device provides summary information about your therapy use each time the therapy is turned off. The rst
screen displays your “Three Night Summary.” It shows your nightly usage for the last 3 sleep sessions (measured in
24 hour periods, ending at noon each day). The most recent session is displayed in the right hand bar, labeled with the
number of hours slept. A green bar indicates that you slept more than 4 hours, and a yellow bar indicates less than 4
hours of use.
The second screen shows the total number of 4+ hour nights that you have slept in the last 30 days. It provides a goal
of sleeping at least 4 hours per night for 70% of the last 30 nights. Therefore the goal is 21 “good nights” of use. This
screen provides a simple way to track your progress. The screen will stop displaying when you reach the goal, or after
the rst 90 days of use has passed, whichever comes rst.
Three Night Summary Screen Goal Progress Screen
Altitude Compensation
This device automatically compensates for altitude up to 7,500 feet. No manual adjustment is necessary.
18 User Manual
Device Alerts
Device alerts are pop-ups that show up on the UI screen. There are 5 types of alerts described here:
• Status: These alerts are just the pop-up screen.
• Notication: These alerts consist of the pop-up screen in addition to a blinking Power LED on top of the device.
• Alert 1: These alerts consist of the pop-up screen, a blinking Power LED and an audible beep when displayed. This
alert will not occur during therapy.
• Alert 2: These alerts consist of the pop-up screen, a blinking Power LED and an audible beep when displayed. This
alert can occur during therapy.
• Safe State: These alerts consist of the pop-up screen, a blinking Power LED and a repeating audible beep.
Note: Status alerts automatically time out after 30 seconds and their pop up screens disappear. All other alerts
must be acknowledged to clear.
Alert Summary Table: The following table summarizes the alerts.
Alert Icon Type Description Possible Cause Action
Data Activity: Do
not remove SD
card.
Status SD card read/write
underway.
n/a No action needed.
Change Accepted Status Conrms acceptance of
prescription change or
device upgrade.
n/a No action needed.
EZ-Start Pressure
Incremented to
xx.x
Status Displays when EZ-Start
mode is enabled and
device is increasing
therapy pressure
setting for the next
session.
n/a No action needed.
Oximetry: Good
Connection
(icon only)
Status Displays on the
therapy screen when
the blower is on and
3 seconds of good
connection is detected.
Appears at the
beginning of therapy.
This screen will not
display again if the
nger probe is removed
and reapplied unless
therapy is stopped and
restarted.
n/a No action needed.
Pair?: 123456
Yes/No
Status Prompts to accept or
decline pairing to a
Bluetooth compatible
device. This device can
be identied by the
digits displayed.
n/a Rotate control dial to
accept pairing (Yes),
or decline (No), then
press control dial to
conrm selection.

19User Manual
Alert Icon Type Description Possible Cause Action
SD Card
Removed.
Notication
or Alert 2
Indicates SD card has
been removed from
therapy device and not
reinserted before the
start of the current
therapy session.
SD card was not
reinserted into device.
Reinsert SD card, or
click to clear alert.
Oximetry: Good
Study
(icon only)
Notication Noties that user has
achieved at least 4
hours of therapy and
oximetry use. Appears
at the end of therapy.
n/a Press Control Dial to
acknowledge and clear
the message.
SD Card Error:
Remove and
Reinsert
Notication SD card error detected Device cannot read
the SD card. A
problem may exist
with the SD card or
it was ejected during
a writing activity,
or it was inserted
incorrectly.
Remove SD card
and reinsert. If alert
continues to occur,
replace with another
card or contact your
provider.
SD Card Full. Notication SD card is full. SD card is full. Remove SD card and
replace with a new
card, or contact your
provider for a new SD
card.
Patient Message
(Refer to section)
Notication Message from your
Provider.
n/a Press Control Dial to
acknowledge and clear
the message.
Change Rejected Alert 1 A prescription or
settings change was
rejected.
Change missing or
incorrect.
Contact your provider.
Humidication
Error. Contact
support if the
problem persists.
Status Humidier error (only
when humidier is
present)
Humidier heater
plate error or
humidier not
properly connected to
therapy device
Turn off device and
disconnect from
power. Detach the
humidier, visually
check that electrical
contacts are clear,
then reconnect
humidier and
power cord. If alert
continues, contact
your provider.
20 User Manual
Alert Icon Type Description Possible Cause Action
Heated Tube
Error. Contact
support if the
problem persists.
Status Heated tube error
(only when heated tube
is present)
Heated tube may
be overheated or
damaged.
Turn off device.
Detach heated tube
from humidier,
make sure that tube
is not covered or
obstructed, and then
reattach to humidier.
If alert continues,
contact your provider.
The attached
power supply
does not support
humidication.
Alert 2 Indicates that the
attached power
supply is not capable
of supporting
humidication or
heated tube.
Incorrect power
supply.
Switch to a Philips
Respironics
DreamStation
power supply that is
capable of supporting
humidication. Or
operate therapy
device without
humidier.
Service Required Safe State Indicates an error
which enters device
into “Safe State.” This
allows power to remain
on but airow is
disabled.
Device error. Press Control Dial
to silence alert.
Disconnect device
from power. Reattach
power cord to restore
power. If the alert
continues to occur,
contact your home
care provider.
Check Power Notication Indicates an
incompatible power
supply is attached.
Incompatible power
supply, or power cord
is not fully inserted
into device’s power
inlet.
Conrm power cord
is fully inserted into
device’s power inlet.
Conrm a compatible
Philips Respironics
power supply is
attached. Switch to
compatible power
supply if needed.
21User Manual
Alert Icon Type Description Possible Cause Action
Low Voltage Notication Low voltage. Incompatible power
supply is attached.
Conrm a compatible
Philips Respironics
power supply is
attached. Switch to
compatible power
supply if needed. If
battery is being used,
ensure battery is
adequately charged.
Automatic Off Status Displayed when therapy
ends due to automatic
off function.
The mask has been
removed.
Put your mask back
on, conrm good t,
and turn airow on to
resume therapy.
Inlet blocked.
Check lter.
Notication Blocked airway Blockage at device
inlet.
Check device air inlet
is not obstructed.
Check air lter(s)
are installed properly;
replace if needed.
Low Leak: Check
Mask and Tube
Notication Blocked airway Blockage at tube or
mask.
Check tube is not
crushed or folded
such that air ow
is restricted. Check
mask is attached
properly and without
any obstruction.
Check Mask Fit n/a Status Displayed when Check
Mask Fit function is
enabled from Patient
Menu.
n/a This alert can be
cleared by pressing
the Control Dial.
Otherwise it will time
out after 60 seconds.
Loading Language
and Rebooting
Status Displayed when a new
language is selected
from the menu.
n/a No action needed.
Times out when
complete.
Busy Status Displayed when the
device is temporarily
inaccessible due to data
communication.
n/a No action needed.
“Sleep Progress” n/a Status Displays last 3 nights
hourly use on rst
screen, and nights of
use on second screen.
n/a Press Control Dial
to acknowledge and
clear each screen.
Otherwise message
times out after 30
seconds.
22 User Manual
Troubleshooting
Your device is equipped with a self-diagnostic tool call “Performance Check”. This tool can evaluate your device for certain errors. It
also allows you to share key device settings with your Provider. Use Performance Check when directed by your provider.
The table below lists some of the problems you may experience with your device and possible solutions to those problems.
Problem Why It Happened What To Do
Nothing happens
when you apply
power to the device.
The backlights on the
buttons do not light.
There’s no power
at the outlet or the
device is unplugged.
If you are using AC power, check the outlet and verify that the device is properly
plugged in. Make sure there is power available at the outlet. Make sure the AC
power cord is connected correctly to the power supply and the power supply cord
is securely connected to the device’s power inlet. If the problem continues to occur,
contact your home care provider. Return both the device and power supply to your
provider, so they can determine if the problem is with the device or power supply.
If you are using DC power, make sure your DC power cord and battery adaptor
cable connections are secure. Check your battery. It may need recharged or
replaced. If the problem persists, check the DC cord’s fuse following the instructions
supplied with your DC cord. The fuse may need to be replaced. If the problem still
occurs, contact your home care provider.
The airow does not
turn on.
There may be a
problem with the
blower.
Make sure the device is powered correctly. Make sure the Home screen appears on
the user interface. Press the Therapy button on top of the device to start airow.
If the airow does not turn on, there may be a problem with your device. Contact
your home care provider for assistance.
The device’s display
is erratic.
The device has
been dropped or
mishandled, or
the device is in
an area with high
Electromagnetic
Interference (EMI)
emissions.
Unplug the device. Reapply power to the device. If the problem continues, relocate
the device to an area with lower EMI emissions (away from electronic equipment
such as cellular phones, cordless phones, computers, TVs, electronic games, hair
dryers, etc.). If the problem still occurs, contact your home care provider for
assistance.
The Ramp feature
does not work when
you press the Ramp
button.
Your home care
provider did not
prescribe Ramp
for you, or your
therapy pressure is
already set to the
minimum setting.
If Ramp has not been prescribed for you, discuss this feature with your home care
provider to see if they will change your prescription.
If your provider has enabled Ramp, but the feature still does not work, check the
current pressure setting on the Therapy screen. If the therapy pressure is set to the
minimum setting (4.0 cm H
2
O), or the Ramp starting pressure is the same as the
therapy pressure, the Ramp feature will not work. Make sure that the ramp time
setting is >0.
The airow is
much warmer than
usual.
The air lters may
be dirty. The device
may be operating
in direct sunlight or
near a heater.
Rinse or replace the reusable air lter or replace the disposable ultra-ne lter.
The temperature of the air may vary somewhat based on your room temperature.
Make sure that the device is properly ventilated. Keep the device away from bedding
or curtains that could block the ow of air around the device. Make sure the device
is away from direct sunlight and heating equipment.
If using the humidier with the device, check the humidier settings. Refer to the
humidier instructions to make sure the humidier is working properly.
If the problem continues, contact your home care provider.
The airow pressure
feels too high or too
low.
The Tubing type
setting may be
incorrect.
Make sure the Tubing type setting (22, 15, or 12) matches the tubing that you are
using (Philips Respironics 22, 15, or 12 mm tubing).
If you are using the Heated Tubing, this setting will be 15H and you cannot change it.
Tube Temperature is
turned on in “Setup”
screen but Heated
Tubing is not warm.
Incorrect power
supply is being used.
Make sure the 80W power supply is being used or a compatible battery or DC cable
is being used.
23User Manual
Problem Why It Happened What To Do
I’m having difculty
adjusting the heated
humidier setting
or the heated tube
temperature setting.
The blower is not
turned on, or the
humidier or heated
tube is not fully
connected.
The humidier setting and tube temperature settings can only be adjusted from the
Therapy ON display screen. Conrm that the blower is turned on, and that the
settings are visible on the right side of the screen, then adjust to desired comfort. If
the blower is on but the humidier settings are not displayed on the Therapy ON
screen, then unplug the device. Conrm that the humidier and/or heated tube
electrical contacts are not obstructed or damaged. Then reconnect the humidier
and/or heated tube, and reconnect the device’s power supply. Turn the blower on; if
the settings are still not visible, contact your provider for assistance.
The water in the
water chamber runs
out before morning.
Water chamber
was not full at start
of session. Mask
leak is excessively
high. The ambient
conditions are very
dry/cool.
Under most conditions, a full water chamber should last for a typical sleep
session. However, many factors impact water consumption, including: the ambient
temperature and humidity in your bedroom, your humidier or heated tube settings,
the level of mask leak, and the duration of your sleep session.
First, make sure that the water chamber is lled to the maximum ll line at the start
of your sleep session. Check that your mask is tted properly, and adjust as needed
to reduce mask leak to normal levels. You may use the Check Mask Fit function to
evaluate your mask t. Also, conrm that the device, humidier, humidier seals
and tube are connected properly and not leaking. You may also choose to lower
your humidier and/or heated tube settings or change the humidication mode from
Fixed to Adaptive humidication mode to increase the time that your humidier
water will last.
I hear a leak or
whistling sound
coming from my
therapy device or
humidier (not
related to mask leak).
The therapy device
air inlet may be
obstructed. The
humidier or tube is
not fully connected.
The humidier seals
are not fully seated
or are missing.
Check therapy device air inlet is not obstructed, and lters are free of debris and
properly inserted. Conrm that the device, humidier, and tube are connected
properly and not leaking. Conrm that the humidier lid seal and dry box seal are
present and properly seated; if needed, gently press around the perimeter of the
seals to reseat them.
I accidentally spilled
water into my
humidier basin.
The water
chamber has been
lled beyond the
maximum ll line.
A small amount of water spilled in the basin of the humidier will not harm your
device. A small spill in the humidier will evaporate under normal humidier use.
However, too much water in the humidier basin could spill over the humidier lid
hinge and might damage your furniture.
Disconnect power from the device. Remove the water chamber, pour out any
excess water until the water level is at or below the maximum ll line and set the
chamber aside. Separate the humidier from the therapy device, and pour out the
spilled water. Once the heater plate has cooled, wipe the inside of the humidier
with a paper towel or soft cloth. If needed, dry the underside of the humidier and
conrm that your table top is dry. Reconnect the humidier and power supply, and
reinstall the water chamber.
24 User Manual
Accessories
There are several accessories available for your DreamStation system such as a Humidier, Cellular Modem,
Wi-Fi Accessory or a Link Module. Contact your home care provider for additional information on the available
accessories. When using optional accessories, always follow the instructions enclosed with the accessories.
List of Compatible Accessories
DSXH DreamStation Humidier
1127627 Humidier Air Outlet Port Disinfection Aid
HT15 15 mm Heated Tube
302418 Pressure Valve Single
312710 O
2
Enrichment Attachment
342077 Bacteria Filter, 10 pack
1124009 PAP Lithium Ion Battery Kit
1126947 Li-ion Battery Case
1121736 Lithium Ion Battery, PAP Device Cable
1121694 DreamStation, Nonin SpO
2
Cable
936 Reusable Finger Sensor (clip)
953 Nonin Adult Flex SpO
2
Sensor (ex)
1062050 Reusable Finger Sensor (soft), Medium
1062051 Reusable Finger Sensor (soft), Small
1120135 DreamStation travel case, 65 W power supply w/USB port
1124301 DreamStation travel case, 65W power supply w/USB port, 5/case
1032907 22 mm Performance Tubing
PR15 15 mm Performance Tubing
PR12 12 mm Performance Tubing
1147466 DreamStation 12mm Tube Device Adapter with OIS
100600C DreamStation Cellular Modem with Oximetry, US
100603C DreamStation Cellular Modem with Oximetry, US
100604C DreamStation Cellular Modem, US
100605C DreamStation Cellular Modem, US
100606C DreamStation Cellular Modem, US with Oximetry, AT&T
100607C DreamStation Cellular Modem, US, AT&T
100700W DreamStation Wi-Fi Accessory with Oximetry, US
100705W DreamStation Wi-Fi Accessory, US
1120293 Link Module
1131859 Dreamstation Roll Stand
1120746 Shielded DC Cord
1120747 Shielded 12V DC Power Cord System
532209 DC Batter Adapter Cable
1144267 DreamStation 80W Power Supply
1005894 Power Cord
1063859 Philips Respironics SD Cards (10 pack)
1114784 PAP Laptop/Travel Briefcase
25User Manual
1122446 Pollen Filter, Reusable (1 pack)
1122447 Ultra-ne Filter, Disposable (1 pack)
1122518 Ultra-ne Filter, Disposable (2 pack)
1122519 Ultra-ne Filter, Disposable (6 pack)
Caution: Pins of connectors should not be touched. Connections should not be made to these connectors
unless ESD precautionary procedures are used. Precautionary procedures include methods to prevent build-up
of electrostatic charge (e.g., air conditioning, humidication, conductive oor coverings, non-synthetic clothing),
discharging one’s body to the frame of the equipment or system or to earth or a large metal object, and bonding
oneself by means of a wrist strap to the equipment or system or to earth.
Adding a Humidier with or without Heated Tubing
You can use the heated humidier and the heated tube with your device. They are available from your home care
provider. A humidier may reduce nasal dryness and irritation by adding moisture to the airow.
Warning: For safe operation, the humidier must always be positioned below the breathing circuit connection
at the mask. The humidier must be level for proper operation.
Note: Refer to the humidier’s instructions for complete setup information.
Using the SD Card
The DreamStation system comes with an SD card inserted in the SD card slot on the side of the device to record
information for the home care provider. Your home care provider may ask you to periodically remove the SD card
and send it to them for evaluation.
Updating Software Using the SD card
To check which version of software is currently on your device, navigate to My Provider and select Device Info.
You can update the device software using the SD card. The software update must be done when the therapy is off.
1. Insert an SD card with the new software version into the device. A pop-up screen appears asking “Would you
like to upgrade software?”
2. Turn the control dial to select Yes and then press the control dial to start the upgrade. The busy icon appears
while the upgrade is in progress. Do not remove power from the device.
3. If the software update is successful, the Change Accepted icon appears on the screen. Remove the SD card
from the device to restart the device and use the new software.
4. If an SD card error is detected, the Change Rejected icon appears. Remove the SD card and reinsert. If the
alert continues to occur, contact Philips Respironics at 1-800-345-6443 or 1-724-387-4000 for a new SD card.
Using the DreamStation Link Module
The Link Module is able to receive oximetry data and transfer it to the therapy device for home use or in a laboratory
setting. For use in a laboratory setting, the Link Module also includes an RS-232 (or “DB9”) port to allow remote
control of the DreamStation Sleep Therapy Device by a personal computer.
Note: Please consult the instructions that accompany the Link Module for installation and removal.
Note: There are no SpO
2
alarms available.
Note: Oximetry data is not displayed.
Dispose of the module following the same disposal instructions for your therapy device.
Warnings:
• If you notice any unexplained changes in the performance of this device, if it has been dropped or mishandled,
if water is spilled into the enclosure, or if the enclosure is broken, discontinue use. Contact your home care
provider.
• Repairs and adjustments must be performed by Philips Respironics-authorized service personnel only.
Unauthorized service could cause injury, invalidate the warranty, or result in costly damage.
• Do not use any accessories, detachable parts, and materials not recommended by Philips Respironics.
Incompatible parts or accessories can result in degraded performance.
26 User Manual
Adding Supplemental Oxygen
Oxygen can be added to the patient circuit. Please note the warnings listed below when using oxygen with the device.
Warnings:
• When using oxygen with this system, the oxygen supply must comply with local regulations for medical oxygen.
• Oxygen supports combustion. Oxygen should not be used while smoking or in the presence of an open ame.
• If supplemental oxygen is added at the exit of the ow generator or humidier, a Philips Respironics Pressure Valve
must be placed in-line with the patient circuit between the device and the oxygen source. The pressure valve helps
prevent the backow of oxygen from the patient circuit into the device when the unit is off. Failure to use the
pressure valve could result in a re hazard.
• When adding oxygen at the mask end of the hose, a Philips Respironics Pressure Valve is not required for oxygen
ow rates of ≤4 liters per minute. However, the reusable and disposable lters must be in place on the ow
generator. Failure to install both the reusable and disposable lters could result in a re hazard.
Note: Refer to the pressure valve’s instructions for complete setup information.
• When using oxygen with this system, turn the device on before turning on the oxygen. Turn the oxygen off
before turning the device off. This will prevent oxygen accumulation in the device.
• Do not connect the device to an unregulated or high pressure oxygen source.
Supplying DC Power to the Device
A Philips Respironics DC power cord can be used to operate this device in a stationary recreational vehicle, boat,
or motor home. In addition, a Philips Respironics DC battery adapter cable, when used with a DC power cord,
allows the device to be operated from a 12 VDC free-standing battery.
Caution: Always ensure that the DC power cord securely ts into your therapy device prior to use. Contact
your home care provider or Philips Respironics to determine if you have the appropriate DC cord for your
specic therapy device.
Caution: When DC power is obtained from a vehicle battery, the device should not be used while the vehicle’s
engine is running. Damage to the device may occur.
Caution: Only use a Philips Respironics DC Power Cord and Battery Adapter Cable. Use of any other system
may cause damage to the device.
Refer to the instructions supplied with the DC power cord and adapter cable for information on how to operate
the device using DC power.
Traveling with the System
When traveling, the carrying case is for carry-on luggage only. The carrying case will not protect the system if it is put
through checked baggage. If traveling with the optional humidier, do not travel with water in the water tank.
For your convenience at security stations, there is a symbol on the bottom of the device indicating that it is medical
equipment and is suitable for airline use. It may be helpful to bring this manual along with you to help security
personnel understand the DreamStation device.
If you are traveling to a country with a line voltage different than the one you are currently using, a different power
cord or an international plug adaptor may be required to make your power cord compatible with the power outlets
of the country to which you are traveling. Contact your home care provider for additional information.
Airline Travel
The device is suitable for use on airlines when the device is operating from an AC or DC power source.
Note: It is not suitable for airline use with any of the modems or humidiers installed in the unit.
27User Manual
Home and Hospital/Institution Cleaning: Device and Humidier Exterior
Warning: To avoid electrical shock, always unplug the power cord from the wall outlet before cleaning the device.
DO NOT immerse the device in any uids.
The device’s exterior surface should be cleaned weekly or more often if necessary. In the hospital or institutional
environment, clean the device’s exterior surface weekly and between patients.
If using the device and humidier on multiple users, the device and humidier exterior should be cleaned between
patient use and as needed by performing the following steps:
1. Turn the device off and disconnect from the power source. Detach all accessories and connectors.
2. Remove the reusable blue pollen lter and disposable light-blue ultra-ne lter (if using). Refer to “Caring for the
Filters” for more information.
Warning: If you are using the device on multiple users, discard and replace the bacteria lter each time the device
is used on a different person.
3. Use a lint-free cloth dampened with water and a mild liquid dish washing detergent solution to clean the exterior of
the enclosure. Use 1 teaspoon (5 milliliters) of liquid dish washing detergent per gallon (3.8 liters) of water.
4. Pay close attention to all corners and crevices of the device exterior surfaces. Be sure all visible soil is removed.
5. Wipe with a lint-free cloth dampened (not dripping) with potable water for at least one minute, turning the cloth
frequently, to remove all detergent residue.
6. Let the device air dry completely before plugging in the power cord.
7. Inspect the device and all circuit parts for damage after cleaning. If any parts are damaged, contact Philips
Respironics Customer Service. Replace any damaged parts.
Caution: Allow the device and humidier to dry completely before reconnecting to the power source.
Home and Hospital: Rinsing and Replacing Filters
Reusable Blue Pollen Filter
Under normal home usage, rinse the reusable blue pollen lter monthly. Replace it with a new one every six
months. In the hospital or institutional environment, rinse the reusable blue pollen lter weekly and replace
it with a new one every six months and between patients.
Caution: Dirty inlet lters may cause high operating temperatures that may affect device performance. Regularly
examine the inlet lters as needed for integrity and cleanliness.
Follow these steps to rinse the reusable blue pollen lter:
1. Turn the device off and disconnect the device from the power source.
2. Remove the lter(s) from the device (refer to the Installing and replacing the air lters section of this manual).
Examine the lters for cleanliness and integrity.
3. To rinse off the reusable blue pollen lter, rst detach the light-blue ultra-ne lter (if applicable), and set aside or
dispose of as needed.
4. Take the reusable lter to the sink, turn it upside down, and run warm tap water through the white lter media to
rinse away any debris. Next, lightly shake the lter to remove as much water as possible.
5. Allow the lter to air dry completely before reinstalling it.
6. If the reusable blue pollen lter is torn or otherwise damaged, replace it.
Note: Only Philips Respironics-supplied lters should be used as replacement lters.
7. If the light-blue ultra-ne lter is dirty or torn, replace it.
8. Reinstall the lters. Refer to the “Installing/Replacing the Filters” section of this manual.
Caution: Never install a wet lter into the device. Allow sufcient drying time for the lter.
Light-blue Ultra-ne Filter
In the home, the light-blue ultra-ne lter is disposable. Replace it with a new one every 30 days or sooner if it
appears dirty. DO NOT rinse the ultra-ne lter. In the hospital or institutional environment, the ultra-ne lter
should be replaced with a new one every 30 days or sooner and should be replaced between patients.
Home and Hospital Cleaning: Non-heated Flexible Tubing
Clean the non-heated exible tubing bef ore rst use and weekly. Discard and replace the tubing every six (6)
months and between patients.
1. Disconnect the exible tubing from the device.
2. Gently wash the 12, 15, or 22 mm exible tubing by completely immersing in a solution of warm water and a mild
liquid dish washing detergent. Use 1 teaspoon (5 ml) of liquid dish washing detergent per gallon (3.8 liters) of warm
water for 3 minutes.
28 User Manual
3. During immersion, gently move the tubing back and forth to loosen and adequately remove adhering substances
from the tubing and connectors.
Note: Be sure to clean the entire inner surface of the tube by ensuring it is fully immersed in the detergent
solution during gentle agitation by hand.
4 Rinse thoroughly with potable water for at least 1 minute to remove all soap residue from the tubing and
connectors.
5. Allow to air dry completely out of direct sunlight.
6. Inspect the tubing for damage or wear (cracking, crazing, tears, punctures, etc.). Discard and replace if necessary.
Note: Refer to the humidier manual for the instructions on how to clean the heated tube.
Service
The device does not require routine servicing.
Warning: If you notice any unexplained changes in the performance of this device, if it is making unusual or harsh
sounds, if it has been dropped or mishandled, if water is spilled into the enclosure, or if the enclosure is broken,
disconnect the power cord and discontinue use. Contact your home care provider.
Additional Notices
Notice: The Bluetooth
®
word mark and logos are registered trademarks owned by Bluetooth SIG, Inc. and
any use of such marks by Philips Respironics is under license. Other trademarks and trade names are
those of their respective owners.
Notice: The DreamStation Therapy Device is capable of transmitting data between the therapy device and a
mobile device. This connection between the therapy device and a mobile device is encrypted.
Notice: A small portion of the rmware that performs data encryption on the DreamStation device is being
utilized under the Apache 2.0 and Mozilla 2.0 licenses. These licenses are available at:
www.apache.org/licenses/LICENSE-2.0 and https://www.mozilla.org/en-US/MPL/2.0/
Notice: This device contains a FCC certied Bluetooth radio module (located on the main board).
Only the co-location of this Bluetooth radio with the radio transceivers of the DreamStation Wi-Fi
Accessory and Cellular Modem has been FCC approved and is permitted.
For compliance with FCC RF exposure guidelines a minimum distance of 20 cm between the Wi-Fi
Accessory or the Cellular Modem and the user’s body should be maintained during operation of one of
those accessories together with the DreamStation.
Notice: FCC ID: THO1116426
Notice: THO1116426 is the FCC ID of the FCC certied Bluetooth module contained in this device.
Notice: Use of non-original manufacturer-approved accessories may violate your local RF exposure
guidelines and should be avoided.
Notice: This device complies with part 15 of the FCC Rules. Operation is subject to the following two
conditions: (1) This device may not cause harmful interference, and (2) this device must accept any
interference received, including interference that may cause undesired operation.
This equipment has been tested and found to comply with the limits for a Class B digital device,
pursuant to Part 15 of the FCC Rules. These limits are designed to provide reasonable protection
against harmful interference in a residential installation. This equipment generates, uses, and can
radiate radio frequency energy and, if not installed and used in accordance with the instructions,
may cause harmful interference to radio communications. However, there is no guarantee that
interference will not occur in a particular installation. If this equipment does cause harmful
interference to radio, TV reception, or other devices which can be determined by turning the
equipment on and off, the user is encouraged to try to correct the interference by one or more of
the following measures:
• Reorient or relocate the receiving antenna (on the radio, TV, or other device).
• Increase the separation between the equipment and receiver.
• Connect the equipment into an outlet on a circuit different from that to which the receiver is
connected.
• Consult the dealer of the device for help.
Notice: Any changes or modications made to the device that are not expressly approved by Respironics
may void the user’s authority to operate the equipment.
29User Manual
Specications
Environmental
Operating Temperature: 5° to 35° C (41° to 95° F)
Storage Temperature: -20° to 60° C (-4° to 140° F)
Relative Humidity (operating & storage): 15 to 95% (non-condensing)
Atmospheric Pressure: 101 to 77 kPa (0 - 2286 m / 0 - 7500 ft)
Physical
Dimensions: 15.7 x 19.3 x 8.4 cm (6.2” L x 7.6” W x 3.3” H)
Weight (Device with power supply): Approximately 1.33 kg (2.94 lbs)
Service Life
The expected service life of the DreamStation Therapy Device and Link Module is 5 years.
Standards Compliance This device is designed to conform to the following standards:
IEC 60601-1 General Requirements for Basic Safety and Essential Performance of Medical Electrical Equipment
ISO 80601-2-70 Sleep Apnea Breathing Therapy Equipment
ISO 80601-2-74 Medical Electrical Equipment — Part 2-74: Particular Requirements for Basic Safety and Essential
Performance of respiratory humidifying equipment
EN 60601-1-2 Electromagnetic Compatibility
RTCA/DO-160G section 21, category M; Emission of Radio Frequency Energy
IEC 60601-1 Classication
Type of Protection Against Electric Shock: Class II Equipment
Degree of Protection Against Electric Shock: Type BF Applied Part
Degree of Protection against Ingress of Water:
Device: Drip Proof, IP22
Link Module: Drip Proof, IP22
80W power supply: Drip Proof, IP22
Mode of Operation: Continuous
Electrical
AC Power Consumption (with 80W power supply): 100 – 240 VAC, 50/60 Hz, 2.0-1.0 A
Note: Power supply is part of the medical electrical equipment.
DC Power Consumption: 12 VDC, 6.67 A
Fuses: There are no user-replaceable fuses.
Radio Specications
Operating Frequency Range: 2402 - 2480 MHz
Maximum Output Power: <10 dBm
Modulation: GFSK, P/4 DQPSK, 8DQPSK
Intake Port Filters
Pollen Filter: 100% Polyester
88% Efcient @ 7-10 micron size
Ultra-ne Filter: Blended Synthetic Fiber
95% Efcient @ 0.5-0.7 micron size
Declared Dual-Number Noise Emissions Values In accordance with ISO 4871
The A-weighted sound pressure level is:
Device: 27 dB(A) with and uncertainty of 2 dB.
Device with Humidier: 29 dB(A) with and uncertainty of 2 dB.
The A-weighted sound power level is:
Device: 35 dB(A) with an uncertainty of 2 dB.
Device with Humidier: 37 dB(A) with an uncertainty of 2 dB.
Note: Values determined according to noise test code given in ISO 80601-2-70:2015, using the basic
standards ISO 3744 and ISO 4871.

30 User Manual
Pressure Accuracy
Pressure Increments: 4.0 to 20.0 cm H
2
O (in 0.5 cm H
2
O increments)
Maximum static pressure accuracy, according to ISO 80601-2-70:2015:
Pressure Static Accuracy
10 cm H
2
O ± 0.5 cm H
2
O
Static pressure accuracy has a measurement uncertainty of 3.7%
Note: All tests were performed with and without humidier and with 22 mm standard, 15 mm standard, 15 mm heated tubes.
Pressure Static Accuracy
10 cm H
2
O ± 1.0 cm H
2
O
Static pressure accuracy has a measurement uncertainty of 3.7%
Note: All tests were performed with and without humidier and with 12 mm standard tubes.
Maximum dynamic pressure variation, according to ISO 80601-2-70:2015:
Pressure 10 BPM 15 BPM 20 BPM
< 10 cm H
2
O ± 0.4 cm H
2
O ± 0.5 cm H
2
O ± 0.8 cm H
2
O
≥ 10.0 to 20 cm H
2
O ± 0.5 cm H
2
O ± 0.8 cm H
2
O ± 1.0 cm H
2
O
Dynamic pressure accuracy has a measurement uncertainty of 4.3%
Note: All tests were performed with and without humidier and with 22 mm standard, 15 mm standard, 15 mm heated, and 12mm
standard tubes.
Maximum Flow Rate (typical)
Test pressures (cm H
2
O)
4.0 8.0 12.0 16.0 20.0
22 mm
tubing
Measured pressure at the patient
connection port (cm H
2
O)
3.7 7.7 11.2 14.9 18.9
Average ow at the patient
connection port (l/min)
85 124 131 132 128
15 mm
tubing
(heated
or non-
heated)
Measured pressure at the patient
connection port (cm H
2
O)
3.7 7.4 10.9 14.9 18.8
Average ow at the patient
connection port (l/min)
86 127 134 133 117
12 mm
tubing
Measured pressure at the patient
connection port (cm H
2
O)
4.0 7.0 11.0 15.0 19.0
Average ow at the patient
connection port (l/min)
85 95 94 100 102
Tubing Specications*
Tubing Tubing resistance (RI/RE) Tubing compliance (@ 60 hPa)
12 mm performance tubing (PR12)
@30 l/min: 0,03 hPa/l/min (cmH
2
O/l/min) 0.44 ml/hPa (ml/cmH
2
O)
15 mm performance tubing (PR15)
@15 l/min: 0.006 hPa/l/min (cmH
2
O/l/min)
@30 l/min: 0.01 hPa/l/min (cmH
2
O/l/min)
0.52 ml/hPa (ml/cmH
2
O)
22 mm performance tubing
@15 l/min: 0.00 hPa/l/min (cmH
2
O/l/min)
@30 l/min: 0.003 hPa/l/ min (cmH
2
O/l/min)
0.73 ml/hPa (ml/cmH
2
O)
*Note: See the DreamStation heated humidier user manual for heated tubing specications.
Disposal
Dispose of this device in accordance with local regulations.
31User Manual
EMC Information
Your unit has been designed to meet EMC standards throughout its Service Life without additional maintenance. There is always
an opportunity to relocate your DreamStation Therapy Device within an environment that contains other devices with their own
unknown EMC behavior. If you believe your unit is affected by locating it closer to another device, simply separate the devices to
remove the condition.
Pressure and Flow Accuracy
The DreamStation Therapy Device is designed to perform within the pressure and owrate accuracies specied in the user manual.
If you suspect that the pressure and/or ow rate accuracy is affected by EMC interference remove power and relocate the device to
another area. If performance continues to be affected discontinue use and contact your home care provider.
SpO2 and Pulse Rate Accuracy
The DreamStation Therapy Device is designed to capture the SpO
2
and Pulse Rate oximetry data within the accuracy specication
described in the sensor manufacture’s instructions for use. When 4 hours of successful oximetry data have been achieved the device
indicates this to the user by displaying “Oximetry: Good Study”. If you suspect that your unit is affected by EMC interference remove
power and relocate the device to another area. If performance continues to be affected discontinue use and contact your home care
provider.
Guidance and Manufacturer’s Declaration - Electromagnetic Emissions – This device is intended for use in the electromagnetic
environment specied below. The user of this device should make sure it is used in such an environment.
Emissions Test Compliance Electromagnetic Environment - Guidance
RF radiated emissions
CISPR 11
Group 1
Class B
The device uses RF energy only for its internal function. Therefore, its RF emissions are
very low and are not likely to cause any interference in nearby electronic equipment.
RF conducted emissions
CISPR 11
Group 1
Class B
The device is suitable for use in all establishments, including domestic establishments
and those directly connected to the public low-voltage power supply network.
Harmonic emissions
IEC 61000-3-2
Class A
Voltage uctuations/Flicker emissions
IEC 61000-3-3
Complies
Emission of Radio Frequency Energy
RTCA/DO-160G Section 21
Category M This device is suitable for use onboard commercial airplanes inside passenger cabin.
Guidance and Manufacturer’s Declaration - Electromagnetic Immunity – This device is intended for use in the electromagnetic
environment specied below. The user of this device should make sure it is used in such an environment.
Immunity Test IEC 60601 Test Level Compliance Level Electromagnetic Environment - Guidance
Electrostatic Discharge (ESD)
IEC 61000-4-2
±8 kV contact discharges
±2 kV, ±4 kV, ±8 kV, &
±15 kV air discharges
±8 kV contact discharges
±2 kV, ±4 kV, ±8 kV, &
±15 kV air discharges
Floors should be wood, concrete or ceramic tile.
If oors are covered with synthetic material, the
relative humidity should be at least 35%.
Electrical fast Transient/burst
IEC 61000-4-4
±2 kV for power supply lines;
100 kHz repetition rate
±2 kV for power supply lines;
100 kHz repetition rate
Mains power quality should be that of a typical
home or hospital environment.
±1 kV for input-output lines;
100 kHz repetition rate
±1 kV for input-output lines;
100 kHz repetition rate
Surge
IEC 61000-4-5
±1 kV differential mode ±1 kV differential mode Mains power quality should be that of a typical
home or hospital environment.
±2 kV common mode ±2 kV for common mode
Voltage dips, short
interruptions and voltage
variations on power supply
input lines
IEC 61000-4-11
<5% U
T
(>95% dip in U
T
)
for 0.5 cycle at 45 degree
increments
<5% U
T
(>95% dip in U
T
) for
1 cycle
70% U
T
(30% dip in U
T
) for 0.5
seconds
<5% U
T
(>95% dip in U
T
)
for 5 seconds
<5% U
T
(>95% dip in U
T
)
for 0.5 cycle at 45 degree
increments
<5% U
T
(>95% dip in U
T
) for
1 cycle
70% U
T
(30% dip in U
T
) for 0.5
seconds
<5% U
T
(>95% dip in U
T
)
for 5 seconds
Mains power quality should be that of a typical
home or hospital environment.
If the user of the device requires continued
operation during power mains interruptions, it is
recommended that the device be powered from
an uninterruptible power supply or a battery.
Power frequency (50/60 Hz)
magnetic eld
IEC 61000-4-8
30 A/m 30 A/m Power frequency magnetic elds should be at
levels characteristic of a typical location in a typical
hospital or home environment.

32 User Manual
Immunity Test IEC 60601 Test Level Compliance Level Electromagnetic Environment - Guidance
Immunity to RFID Readers
AIM 7351731
RFID Reader frequencies as
specied in AIM 7351731:
134.2 kHz at 65 A/m
13.56 MHz at 12 A/m
RFID Reader frequencies as
specied in AIM 7351731:
134.2 kHz at 65 A/m
13.56 MHz at 12 A/m
Magnetic elds should be at levels characteristic
of a typical location in a typical hospital or home
environment.
NOTE: U
T
is the a.c. mains voltage prior to application of the test level.
Guidance and Manufacturer’s Declaration - Electromagnetic Immunity – This device is intended for use in the electromagnetic
environment specied below. The user of this device should make sure it is used in such an environment.
Immunity Test IEC 60601 Test Level Compliance Level Electromagnetic Environment - Guidance
Conducted RF
IEC 61000-4-6
3 Vrms
150 kHz to 80 MHz
6 Vrms
Amateur Radio & ISM
Bands between 150 kHz
and 80 MHz
3 Vrms
150 kHz to 80 MHz
6 Vrms
Amateur Radio & ISM
Bands between 150 kHz
and 80 MHz
Portable and mobile RF communications equipment should be
used no closer to any part of the device, including cables, than the
recommended 30 cm separation distance.
Interference may occur in the vicinity of equipment marked with the
following symbol:
Radiated RF
IEC 61000-4-3
10 V/m
80 MHz to 2.7 GHz
Telecommunication
frequencies as specied in
clause 8.10 of IEC 60601-
1-2:2014:
450, 810, 870, 930, 1720,
1845, 1970, and 2450 MHz
at 28 V/m
385 MHz at 27 V/m
710, 745, 780, 5240, 5500,
and 5785 MHz at 9 V/m
10 V/m
80 MHz to 2.7 GHz
Telecommunication
frequencies as specied in
clause 8.10 of IEC 60601-
1-2:2014:
450, 810, 870, 930, 1720,
1845, 1970, and 2450 MHz
at 28 V/m
385 MHz at 27 V/m
710, 745, 780, 5240, 5500,
and 5785 MHz at 9 V/m
Immunity to RFID
Readers
AIM 7351731
RFID Reader frequencies
as specied in AIM
7351731:
433 MHz at 3 V/m
860 MHz to 960 MHz at
54 V/m
2450 MHz at 54 V/m
RFID Reader frequencies
as specied in AIM
7351731:
433 MHz at 3 V/m
860 MHz to 960 MHz at
54 V/m
2450 MHz at 54 V/m
Limited Warranty
Respironics, Inc., a Philips company (“Philips Respironics”) provides this non-transferable, limited warranty for DreamStation CPAP,
DreamStation CPAP Pro, and DreamStation Auto CPAP (“Product”) to the customer who originally purchased the Product directly
from Philips Respironics.
What this Warranty Covers: Philips Respironics warrants each new Product will be free from defects in materials and workmanship
and will perform in accordance with the Product specications under normal and proper use and maintenance in accordance with
applicable instructions, subject to the exclusions below.
How Long does this Warranty Last: Two (2) years from the longer of the date of shipment to the purchaser or date of setup by
purchaser for the end user, except:
The warranty period for accessories, replacement parts, and disposables including, but not limited to, lters, tubing,
carrying case, is 90 days from the date of shipment to the original purchaser.
What this Warranty does not cover: This warranty does not apply to any software included with the Product as the software
warranty is included in the software license. This warranty does not cover damage or injury whether to the Products, personal
property, or persons caused by accident, misuse, abuse, Acts of God, water ingress, repair or alteration by anyone other than Philips
Respironics or its authorized service center, failure to operate in accordance with the terms of the operating manual and instructions,
lack of reasonable care, the discontinuance of a network (e.g. 2G, 3G, etc.) by a carrier (e.g. ATT, Verizon, etc., or other defects not
related to material or workmanship. This warranty is not transferable. If Philips Respironics nds that a Product returned for service
or the issue raised is not covered under this limited warranty, Philips Respironics may charge an evaluation fee and return shipping.
What Philips Respironics will do: If a Product does not meet the warranty above in the rst 90 days after the original shipment date,
Philips Respironics will replace the device with a new Product. Thereafter, if a Product fails to conform to the warranties set forth
above during the applicable warranty period, Philips Respironics will repair or replace the Product or refund the original purchase
price, in Philips Respironics sole discretion. Philips Respironics may use new or remanufactured assemblies, components, and parts
in repair and new or recertied refurbished devices for replacement. The balance of the original warranty period will apply to any
Product or component of a Product repaired or replaced under this warranty.
Warranty Disclaimer; Limitation of Liability: EXCEPT AS SET FORTH IN THIS LIMITED WARRANTY, PHILIPS RESPIRONICS MAKES
NO WARRANTIES, EXPRESSED OR IMPLIED, STATUTORY OR OTHERWISE, REGARDING THE PRODUCT OR ITS QUALITY
OR PERFORMANCE. PHILIPS RESPIRONICS SPECIFICALLY DISCLAIMS THE IMPLIED WARRANTY OF MERCHANTABILITY
AND THE IMPLIED WARRANTY OF FITNESS FOR A PARTICULAR PURPOSE. IN NO EVENT WILL PHILIPS RESPIRONICS
MAXIMUM LIABILITY UNDER THESE WARRANTIES EXCEED THE ORIGINAL PURCHASE PRICE OR WILL PHILIPS
RESPIRONICS BE LIABLE FOR ANY ECONOMIC LOSS, LOSS OF PROFITS, OVERHEAD, OR SPECIAL, INCIDENTAL, OR
CONSEQUENTIAL DAMAGES. Repair, replacement, or return of purchase price by Philips Respironics is the original purchaser’s sole
and exclusive remedy under this warranty.
This warranty gives you specic legal rights, and you may also have other rights that vary from state to state. Some states do not
allow the exclusion or limitation of incidental or consequential damages, so the above exclusion and limitations may not apply to you.
How to get warranty support: Patients contact your local authorized Philips Respironics dealer and dealers contact Respironics, Inc.
at:
1001 Murry Ridge Lane
Murrysville, Pennsylvania 15668-8550
+1-724-387-4000

1152070
1152070 R00
JDW 08/27/2021
EN-DOM
Respironics Inc.
1001 Murry Ridge Lane
Murrysville, PA 15668 USA
