EDEXCEL INTERNATIONAL GCSE CHEMISTRY
EDEXCEL CERTIFICATE IN CHEMISTRY
ANSWERS
SECTION B
Chapter 7
1. a)
b) 2H
2
O
2
(aq) ----> 2H
2
O(l) + O
2
(g)
c) Relights a glowing splint.
2. a) non-metal (sulfur)
b) metal (sodium)
c) metal (copper)
d) non-metal (carbon as graphite)
3. a) React with a named heated metal (most obvious would be to pass over heated copper). Valid
equation for reaction used, e.g.:
2Cu(s) + O
2
(g) ----> 2CuO(s)
b) e.g. pass through a solution of an alkali. Lime water will probably be suggested, but this isn't
very effective because of the very dilute solution used. Sodium hydroxide solution is better.
(Useful discussion point)
c) 3Mg(s) + N
2
(g) ----> Mg
3
N
2
(s)
d) Denser. Mixing it with nitrogen increases the density of the nitrogen.
e) Any two noble gases apart from argon.
4. a) Carbon dioxide is heavier than air and doesn’t burn or support combustion (except for burning
metals like magnesium). It smothers the flame preventing oxygen getting at the fuel.
b) Gases are more soluble in water if you increase the pressure, and so carbon dioxide is
dissolved in water under pressure. When the top is released, the pressure drops to atmospheric.
The gas becomes less soluble, and bubbles out of solution.
c) The sulfur (or sulfur compound) burns to make sulfur dioxide. The sulfur dioxide reacts with
water and oxygen in the atmosphere to produce very dilute sulfuric acid which falls as acid rain.
d) The spark in the engine which is intended to ignite the petrol/air mixture also causes nitrogen to
react with oxygen to give various oxides of nitrogen.
e) The catalyst has to be hot to work properly. During a short journey it may not reach a high
enough temperature.
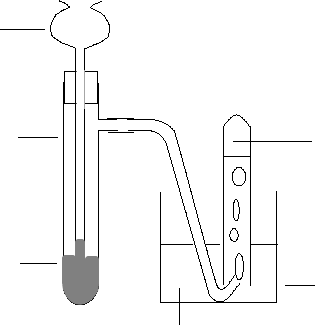
thistle funnel
side arm
boiling tube
hydrogen peroxide
solution and solid
manganese(IV)
oxide
water
beaker
oxygen collects
Chapter 8
1. a) Na, Al, Fe, Cu
b) (i) magnesium oxide, copper
(ii) Mg(s) + CuO(s) ----> MgO(s) + Cu(s) (Include state symbols in all equations for
preference.)
(iii) CuO
(iv) CuO
c) (i) zinc
(ii) A reducing agent is a substance which reduces something else. Zinc removes oxygen
from the cobalt(II) oxide. Removal of oxygen is reduction.
(iii) zinc
d) Al, Mn, Cr (Statement 1: Al is above Cr. Statement 2: Mn is below Al. Statement 3: Mn is
above Cr. Putting this together gives the final list.)
2. a) oxidised; gain of oxygen
b) reduced; loss of oxygen
c) oxidised; loss of electrons
d) reduced; gain of electrons
3. a) magnesium is above lead because it removes the oxygen from the lead(II) oxide.
b) Mg(s) + Pb
2+
(s) ----> Mg
2+
(s) + Pb(s)
4. a) Either: grey iron filings become coated with brown solid. Or: solution fades from blue to
colourless (very pale green).
b) iron
c) Fe(s) + CuSO
4
(aq) ----> FeSO
4
(aq) + Cu(s)
5. a) Ni, Cu, Ag
b) (i) Either: colour of solution changes from blue to green. Or: nickel becomes coated with
brown solid.
(ii) Ni(s) + CuSO
4
(aq) ----> NiSO
4
(aq) + Cu(s)
(iii) Ni(s) + Cu
2+
(aq) ---->Ni
2+
(aq) + Cu(s)
Nickel has been oxidised by loss of electrons.
6. a) X is between iron and hydrogen in the reactivity series. It displaces hydrogen from dilute
hydrochloric acid, and copper from copper(II) sulphate, and so must be above hydrogen and
copper. It won't displace iron from iron(II) sulphate, and so must be below iron.
b) (i) Yes: X nitrate and silver
(ii) No
(iii) No (at least, not in the short term)
(iv) Yes: X chloride and copper
(v) Yes: X sulphate and hydrogen
7. a) hydrogen
b) aluminium chloride
c) 2Al(s) + 6HCl(aq) ----> 2AlCl
3
(aq) + 3H
2
(g)
d) Aluminium is covered by a very thin, but very strong, layer of aluminium oxide which prevents
the acid getting at the aluminium underneath. On heating, the acid reacts with the oxide and
removes it. The aluminium then shows its true reactivity, and produces a vigorous reaction.
8. Drop a very small piece into cold water. If it reacts, judge its reactivity relative to K, Na, Ca or Mg.
If it doesn't react, add a small piece to dilute hydrochloric acid and warm if necessary. Rapid
reaction in the cold would place it as “similar to magnesium”. A few bubbles of hydrogen in the
cold, but more on heating would place it as "similar to iron or zinc".
If no reaction, then it is "below hydrogen". (A good student might mention that "no reaction" could
also be caused by an effect similar to the oxide coating on aluminium. That would be worth some
praise.)
Note: Students might also suggest reacting the metal with steam. Point out that this is much more
tedious than dropping a piece of metal into dilute hydrochloric acid, and that they should go for the
easiest option available.
Chapter 9
1. a) copper(II) oxide, copper(II) hydroxide, copper(II) carbonate.
b) CuO(s) + H
2
SO
4
(aq) ----> CuSO
4
(aq) + H
2
O(l)
Cu(OH)
2
(s) + H
2
SO
4
(aq) ----> CuSO
4
(aq) + 2H
2
O(l)
CuCO
3
(s) + H
2
SO
4
(aq) ----> CuSO
4
(aq) + CO
2
(g) + H
2
O(l)
2. a)
(A thistle funnel isn’t essential. It would work perfectly well with a simple bung.)
Mg(s) + 2HCl(aq) ----> MgCl
2
(aq) + H
2
(g)
b) Pops with a lighted splint.
c) Anything underneath hydrogen in the Reactivity Series - e.g. copper, silver, gold, platinum.
d) Anything above calcium in the Reactivity Series - e.g. potassium, sodium, lithium. Calcium is
safe as long as the acid is really dilute, but it still gets very hot!
e) (i) water: 2H
2
(g) + O
2
(g) ----> 2H
2
O(l)
(ii) The hydrogen is much less dense than air and so rises quickly. The flame rises with
the hydrogen.
3. a) A = copper; B = copper(II) oxide; C = copper(II) sulfate; D = copper(II) carbonate; E = carbon
dioxide; F = copper(II) nitrate
b) (i) CuO(s) + H
2
SO
4
(aq) ----> CuSO4(aq) + H
2
O(l)
(ii) CuCO
3
(s) + 2HNO
3
(aq) ----> Cu(NO
3
)
2
(aq) + CO
2
(g) + H
2
O(l)
4. a) Description could include: Silvery metal. Colourless acid. Slow bubbles of gas on warming.
Formation of green solution. Possible disappearance of nickel if very small quantities were used.
Testing for gas: Pops with lighted splint held to mouth of tube.
b) Ni(s) + H
2
SO
4
(aq) ----> NiSO
4
(aq) + H
2
(g)
(c) Description could include: Green solid. Colourless acid. Bubbles of gas produced in the cold.
Formation of green solution. Green powder disappears if excess acid is used.
Testing for gas: Bubble through lime water to give a white precipitate (milky, cloudy).
(d) (i) NiCO
3
(s) + 2HCl(aq) ----> NiCl
2
(aq) + CO
2
(g) + H
2
O(l)
(ii) CO
3
2-
(s) + 2H
+
(aq) ----> CO
2
(g) + H
2
O(l)
Some teachers may prefer to write this as
NiCO
3
(s) + 2H
+
(aq) ----> Ni
2+
(aq) + CO
2
(g) + H
2
O(l)
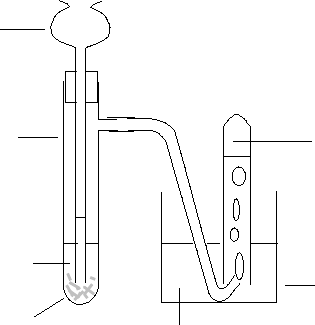
thistle funnel
side arm
boiling tube
dilute hydrochloric
acid
water
beaker
hydrogen collects
magnesium
5. a) acid: H
2
SO
4
base: MgO
b) acid: H
+
base: CO
3
2-
c) not acid-base (actually a redox reaction)
d) acid: HCl base: H
2
O
e) not acid-base (another redox reaction)
f) acid: HCl base: NH
3
g) acid: HCl base: NaOH
6. a) (i) Water: gives a hydrogen ion to a hydride ion.
(ii) Hydride ion: accepts a hydrogen ion.
b) sodium hydroxide, hydrogen
c) Fizzing. Colourless gas. White solid dissolves in colourless liquid to give colourless solution.
d) Accept any pH from reasonably to very alkaline.
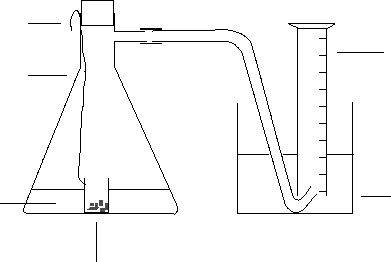
7. The most obvious way of doing this would be to use an initial rate experiment, and measure the
time taken for a small fixed volume (say 5 cm
3
) of hydrogen to be produced. The apparatus
should allow mixing of the zinc and acid without any possible loss of hydrogen. For example:
The reaction would be started by shaking the flask vigorously to upset the weighing bottle and mix
all the reactants. Record how long it takes for a small fixed volume of hydrogen to be produced.
The reason for choosing very small zinc granules is because it would otherwise be very difficult to
get exactly the same mass and surface area of zinc from one reaction to the next. In each
experiment, the mass of zinc should be the same, and the same volume of the same sulfuric acid
should be used. It has to be assumed that the lab temperature doesn’t change significantly during
the practical session.
To vary the amount of copper(II) sulfate present, it would be easiest to start with, say, 10 cm
3
of
copper(II) sulfate solution for the first experiment. For the second experiment, use 9 cm
3
topped
up with 1 cm
3
of water, and then continue diluting it in this way in subsequent experiments. In
each case, the total volume of copper(II) sulfate solution added to the sulfuric acid must be
constant, otherwise the acid is being diluted by varying amounts throughout the experiments.
Process the results by plotting rate (in terms of cm
3
of hydrogen per second) against the volume of
copper(II) sulfate solution used.
beaker of water
small measuring cylinder
cotton
side arm flask
dilute sulfuric acid
+ copper(II) sulfate
solution
weighing bottle containing
very small zinc granules
Chapter 10
1. soluble insoluble
sodium chloride lead(II) sulfate
zinc nitrate calcium carbonate
iron(III) sulfate lead(II) chloride
potassium sulfate copper(II) carbonate
aluminium nitrate silver chloride
ammonium chloride barium sulfate
magnesium nitrate calcium sulfate (almost insoluble)
sodium phosphate nickel(II) carbonate
potassium dichromate(VI) chromium(III) hydroxide
2. a) Dilute sulphuric acid in a beaker / flask. Heat gently. Add copper(II) oxide a little at a time until
no more reacts. Filter into an evaporating basin. Evaporate gently until a sample will crystallise on
cooling. Leave to crystallise. Separate and dry crystals.
(b) (i) CuO(s) + H
2
SO
4
(aq) ----> CuSO
4
(aq) + H
2
O(l)
(ii) CuSO
4
(aq) + 5H
2
O(l) ----> CuSO
4
.5H
2
O(s)
3. a) using a pipette: Need to be able to measure the volume of sodium carbonate solution
accurately so that exactly the same volume can be used later without the indicator.
few drops of methyl orange . . .: An indicator to show when the solution becomes “neutral”. (In
fact the colour change happens around pH 4. The pH changes very quickly around the end point
for the indicator, and the difference in volume of acid added to take the pH from the desired pH 7 to
the actual pH 4 for the indicator will be a fraction of a drop.)
. . . becomes orange: Exactly the right volume of acid has been added (the "neutral" colour for
methyl orange).
without the methyl orange: Otherwise the crystals would be contaminated by methyl orange.
evaporated until . . .: Shows when the solution is sufficiently concentrated to crystallise on cooling.
If a sample will crystallise, so will the bulk of the solution.
. . . left to cool: Crystals form on cooling because the solubility of the sodium sulphate is lower in
the cold.
(b) (i) Na
2
CO
3
(aq) + H
2
SO
4
(aq) ----> Na
2
SO
4
(aq) + CO
2
(g) + H
2
O
(l)
(ii) Na
2
SO
4
(aq) + 10H
2
O(l)
----> Na
2
SO
4
.10H
2
O(s)
4. a) A solution of any soluble silver salt (silver nitrate is always used) plus a solution of any soluble
chloride including dilute hydrochloric acid.
Ag
+
(aq) + Cl
-
(aq) ----> AgCl(s)
b) A solution of any soluble calcium salt (calcium chloride or nitrate) plus a solution of a soluble
carbonate (sodium, potassium or ammonium carbonate).
Ca
2+
(aq) + CO
3
2-
(aq) ----> CaCO
3
(s)
c) A solution of a soluble lead(II) salt (most commonly, lead(II) nitrate) plus a solution of any
soluble sulfate including dilute sulfuric acid.
Pb
2+
(aq) + SO
4
2-
(aq) ----> PbSO
4
(s)
d) A solution of a soluble lead(II) salt (most commonly, lead(II) nitrate) plus a solution of any
soluble chloride including dilute hydrochloric acid.
Pb
2+
(aq) + 2Cl
-
(aq) ----> PbCl
2
(s)
5. Mix solutions of barium chloride or barium nitrate and sodium carbonate, potassium carbonate or
ammonium carbonate. Filter, wash and dry the precipitate.
Ba
2+
(aq) + CO
3
2-
(aq) ----> BaCO
3
(s)
6. a) A: dilute sulfuric acid + solid zinc (or zinc oxide, hydroxide or carbonate)
H
2
SO
4
(aq) + Zn(s) ----> ZnSO
4
(aq) + H
2
(g)
or H
2
SO
4
(aq) + ZnO(s) ----> ZnSO
4
(aq) + H
2
O(l)
or H
2
SO
4
(aq) + Zn(OH)
2
(s) ----> ZnSO
4
(aq) + 2H
2
O(l)
or H
2
SO
4
(aq) + ZnCO
3
(s) ----> ZnSO
4
(aq) + H
2
O(l) + CO
2
(g)
b) C: solutions of a soluble barium salt + soluble sulfate (including H
2
SO
4
)
Ba
2+
(aq) + SO
4
2-
(aq) ----> BaSO
4
(s)
There is no point in writing full equations for precipitation reactions like this. The ionic equation is
always easier.
c) B: dilute nitric acid + potassium hydroxide or carbonate solution
HNO
3
(aq) + KOH(aq) ----> KNO
3
(aq) + H
2
O(l)
or 2HNO
3
(aq) + K
2
CO
3
(aq) ----> 2KNO
3
(aq) + H
2
O(l) + CO
2
(g)
d) A: dilute nitric acid + solid copper(II) oxide or hydroxide or carbonate.
2HNO
3
(aq) + CuO(s) ----> Cu(NO
3
)
2
(aq) + H
2
O(l)
or 2HNO
3
(aq) + Cu(OH)
2
(s) ----> Cu(NO
3
)
2
(aq) + 2H
2
O(l)
or 2HNO
3
(aq) + CuCO
3
(s) ----> Cu(NO
3
)
2
(aq) + H
2
O(l) + CO
2
(g)
(Unlike the other simple acids, nitric acid also reacts with copper metal, but it doesn’t produce
hydrogen, and is not on the Edexcel IGCSE syllabus. Any equation using copper to produce
hydrogen should be disallowed.)
e) C: solutions of a soluble lead(II) salt (normally the nitrate) + a soluble chromate(VI). (Since you
don’t know anything specifically about chromates, choose one of sodium, potassium or
ammonium chromate(VI) - these are bound to be soluble because all sodium, potassium and
ammonium salts are soluble.)
Pb
2+
(aq) + CrO
4
2-
(aq) ----> PbCrO
4
(s)
Chapter 11
1. a) A
b) D
c) C
d) B
2. For example: Stir with a large enough volume of cold water to dissolve all the sugar. Filter to
leave the diamonds on the filter paper. Wash on the filter paper with more water to remove any
last traces of sugar solution. Allow to dry.
3. a) chlorine
b) ammonia
c) carbon dioxide
d) hydrogen
e) oxygen
4. a) M
b) R
c) G and T
d) P
5. a) Clean a nichrome or platinum wire by dipping it into concentrated hydrochloric acid and then into
a flame until no colour shows. Moisten the wire with concentrated hydrochloric acid, dip it into the
solid, and then back into flame. Lithium ions give a red flame colour.
b) Warm the solid very gently with sodium hydroxide solution. Test any gases given off with a
piece of damp red litmus paper. If it turns blue, ammonia is being given off from an ammonium
compound.
c) Make a solution in pure water. Add dilute hydrochloric acid + barium chloride solution. A white
precipitate shows the sulfate ions.
d) Drop the liquid onto blue cobalt chloride paper (which turns pink), or onto white anhydrous
copper(II) sulfate (which turns blue).
e) Add dilute hydrochloric acid or dilute nitric acid. (Not sulfuric acid which forms an insoluble layer
of calcium sulfate around the calcium carbonate, which stops the reaction.) Look for a colourless
odourless gas turning lime water milky.
(f) Make a solution in pure water. Add dilute nitric acid + silver nitrate solution. A yellow precipitate
shows the iodide ions.
6. a) A = iron(III) chloride; B = iron(III) hydroxide; C = silver chloride
b) B: Fe
3+
(aq) + 3OH
-
(aq) ----> Fe(OH)
3
(s)
or: FeCl
3
(aq) + 3NaOH(aq) ----> Fe(OH)
3
(s) + 3NaCl(aq)
C: Ag
+
(aq) + Cl
-
(aq) ----> AgCl(s)
or 3AgNO
3
(aq) + FeCl
3
(aq) ----> 3AgCl(s) + Fe(NO
3
)
3
(aq)
7. a) D = iron(II) sulphate; E = iron(II) hydroxide; F = barium sulphate
b) E: Fe
2+
(aq) + 2OH
-
(aq) ----> Fe(OH)
2
(s)
or: FeSO
4
(aq) + 2NaOH(aq) ----> Fe(OH)
2
(s) + Na
2
SO
4
(aq)
F: Ba
2+
(aq) + SO
4
2-
(aq) ----> BaSO
4
(s)
or BaCl
2
(aq) + FeSO
4
(aq) ----> BaSO
4
(s) + FeCl
2
(aq)
8. a) G = potassium carbonate; H = potassium nitrate; I = carbon dioxide (Note: G could also be
potassium hydrogencarbonate, but, because hydrogencarbonates haven’t been covered in the
course, a student is unlikely to come up with it.)
b) CO
3
2-
(s) + 2H
+
(aq) ----> CO
2
(g) + H
2
O(l)
or K
2
CO
3
(s) + 2HNO
3
(aq) ----> 2KNO
3
(aq) + CO
2
(g) + H
2
O(l)
9. a) Dissolve the mixture in the minimum possible amount of hot water, and then allow it to cool
again. The small amount of the more soluble potassium carbonate will stay in solution, but crystals
of potassium nitrate will be formed on cooling. Filter these off, and wash them on the filter paper
with a small amount of very cold water. Allow them to dry.
b) Take a sample and add any named dilute acid. If the crystals are free of potassium carbonate,
there won’t be any fizzing.
10.a) A = copper(II) carbonate
B = sulfuric acid
C = copper(II) sulfate
D = carbon dioxide
E = barium sulfate
F = copper(II) oxide
G = carbon dioxide
H = copper(II) chloride
I = copper(II) hydroxide
b) (i) CuCO
3
(s) + H
2
SO
4
(aq) ----> CuSO
4
(aq) + CO
2
(g) + H
2
O(l)
or: CO
3
2-
(s) + 2H
+
(aq) ----> CO
2
(g) + H
2
O(l)
(ii) Ba
2+
(aq) + SO
4
2-
(aq) ----> BaSO
4
(s)
or BaCl
2
(aq) + CuSO
4
(aq) ----> BaSO
4
(s) + CuCl
2
(aq)
(iii) Cu
2+
(aq) + 2OH
-
(aq) ----> Cu(OH)
2
(s)
or CuSO
4
(aq) + 2NaOH(aq) ----> Cu(OH)
2
(s) + Na
2
SO
4
(aq)
(iv) CuO(s) + 2HCl(aq) ----> CuCl
2
(aq) + H
2
O(l)
(v) CuCO
3
(s) ----> CuO(s) + CO
2
(g)
(Hardly anyone would write the ionic equations for the last two.)
Chapter 12
1. a) (i) strontium
(ii) chlorine
(iii) nitrogen
b) (i) 2; (ii) 7; (iii) 5
c) metals: caesium, molybdenum, nickel, strontium, tin
non-metals: chlorine, neon, nitrogen
d) molybdenum and nickel
e) (i) caesium
(ii) neon
2. a) A = lithium
b) B = potassium; C = hydrogen; D = potassium hydroxide
c) E = sodium; F = chlorine; G = sodium chloride
d) (i) 2K(s) + 2H
2
O(l) ----> 2KOH(aq) + H
2
(g)
(ii) 2Na(s) + Cl
2
(g) ----> 2NaCl(s)
(You could easily justify (l) as the state symbol for both K and Na.)
e) red turns blue; no change to the blue.
b) Lots of heat evolved. Melting point of potassium is low.
3. a)
b) solid
c) less
d) steamy gas; solution with a pH about 1
e) A white solid, soluble in water. It will be an ionic solid, because that’s typical of compounds
formed when Group 1 and Group VII elements combine. The other Group 1 halides are soluble in
water. There isn’t a transition element present, so it is unlikely to be coloured.
f) 2At
-
(aq) + Cl
2
(aq) ----> At
2
(s) + 2Cl
-
(aq)
A redox reaction is one in which reduction and oxidation occur. The astatide ions have lost
electrons, and so have been oxidised. The chlorine molecules have gained electrons, and so have
been reduced.
4. 5 from: high melting point, high boiling point, high density, hard, good conductor of electricity, good
conductor of heat, malleable and ductile, useful catalyst, compounds are coloured (+ anything else
which seems sensible)
At At At
At
atom ion molecule
5. a) Ne atoms have 8 electrons in their outer level. No room to share electrons to form covalent
bonds and therefore monatomic. Gases because the only forces possible between the individual
atoms are weak intermolecular attractions. Unreactive because of the lack of possibility of forming
covalent bonds or stable ions.
b) Both metals react by their atoms losing their single outer electron to form 1+ ions. In potassium,
this electron is further from the attraction of the nucleus and so lost more easily.
(A full answer should really include a mention of increased nuclear charge being offset by
increased screening.)
c) Cl
2
(aq) + 2Br
-
(aq) ----> 2Cl
-
(aq) + Br
2
(aq)
Chlorine removes an electron from a Br
-
ion and uses it to form Cl
-
. This happens because
chlorine is a smaller atom than bromine and so an extra electron is closer to, and more attracted
to, the chlorine nucleus than it was to the bromine nucleus.
(A full answer should really include a mention of increased nuclear charge being offset by
increased screening.)
6. a) Noble gas: Physical data shows a gas. Lack of reactivity suggests a noble gas rather than
fluorine or chlorine from Group 7.
b) Transition element: High melting point metal (conducts electricity) with coloured compounds.
Not Group 1 or 2 because of lowish reactivity and coloured compounds.
c) Group 1 or 2 element: Physical data and reactions suggest a metal. Reactivity suggests
Groups 1 or 2. Lack of coloured compounds suggests not a transition metal.
7. There’s no “right” answer to this. A sample response might be:
Test Observation
Appearance Shiny grey solid
Hit with a hammer Very little effect. Edges bent slightly.
Put into electrical circuit with battery
and bulb
Bulb lights up. Conducts electricity.
Warm with dilute hydrochloric acid No reaction
From its appearance, its lack of brittleness and its electrical conductivity, the element is a metal.
Its lack of reaction with dilute hydrochloric acid must be because it is either below hydrogen in the
Reactivity Series, or has a very strong oxide coating.
Most students would give a positive result with the acid. The negative result is included here to
make the point that any inconsistent result should be explained.
Chapter 13
1. cathode anode
a) lead bromine
b) zinc chlorine
c) hydrogen iodine
d) sodium iodine
e) copper chlorine
f) hydrogen chlorine
g) hydrogen oxygen
h) hydrogen oxygen
2. a) Ions weren't free to move.
b) anode
c) iodine: 2I
-
(l) ----> I
2
(g) + 2e
-
d) K
+
(l) + e
-
----> K(l)
e) Orange flashes around the cathode (sodium burning) and brown fumes (bromine) around the
anode.
f) cathode: Na
+
(l) + e
-
----> Na(l)
anode: 2Br
-
(l) ----> Br
2
(g) + 2e
-
3. a) (i) Pb
2+
(l) + 2e
-
----> Pb(l)
(ii) 2Br
-
(l) ----> Br
2
(g) + 2e
-
(iii) Oxidised: bromide ions. Reduced: lead(II) ions.
b) (i) 2H
+
(aq) + 2e
-
----> H
2
(g)
(ii) 2Cl
-
(aq) ----> Cl
2
(g) + 2e
-
(iii) Oxidised: chloride ions. Reduced: hydrogen ions.
c) (i) 2H
+
(aq) + 2e
-
----> H
2
(g)
(ii) 2Br
-
(aq) ----> Br
2
(aq or l) + 2e
-
(iii) Oxidised: bromide ions. Reduced: hydrogen ions.
d) (i) Cu
2+
(aq) + 2e
-
----> Cu(s)
(ii) 4OH
-
(aq) ----> 2H
2
O(l) + O
2
(g) + 4e
-
(iii) Oxidised: hydroxide ions. Reduced: copper(II) ions.
e) (i) 2H
+
(aq) + 2e
-
----> H
2
(g)
(ii) 4OH
-
(aq) ----> 2H
2
O(l) + O
2
(g) + 4e
-
(iii) Oxidised: hydroxide ions. Reduced: hydrogen ions.
f) (i) Mg
2+
(l) + 2e
-
----> Mg(s or l)
(ii) 2I
-
(l) ----> I
2
(g) + 2e
-
(iii) Oxidised: iodide ions. Reduced: magnesium ions.
g) (i) 2H
+
(aq) + 2e
-
----> H
2
(g)
(ii) 2Cl
-
(aq) ----> Cl
2(g)
+ 2e
-
(iii) Oxidised: chloride ions. Reduced: hydrogen ions.
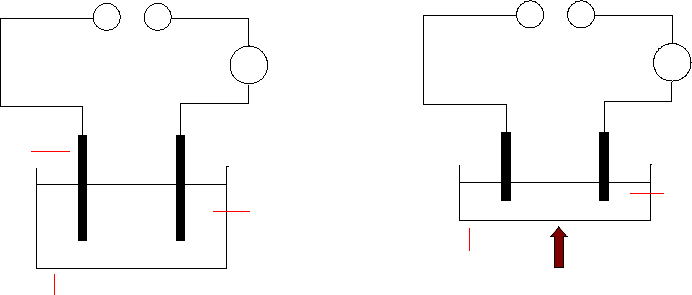
4. The melting point of S is too high to reach using a bunsen, and so you would have to test a solution
in water.
On the other hand, T would melt easily, and won’t dissolve. Heat it until it melts.
(You could also do the electrolysis of the solution using the more complicated apparatus in the
chapter, but since there is no need to collect anything, there isn’t much point.)
If the substances are electrolytes, the bulbs will light up, and there will be signs of activity around
the electrodes (gases given off, solids deposited, etc).
6v DC power supply
bulb
carbon electrodes
small beaker
S solution
heat
bulb
pyrex dish
or crucible
6v DC power supply
molten T
Chapter 14
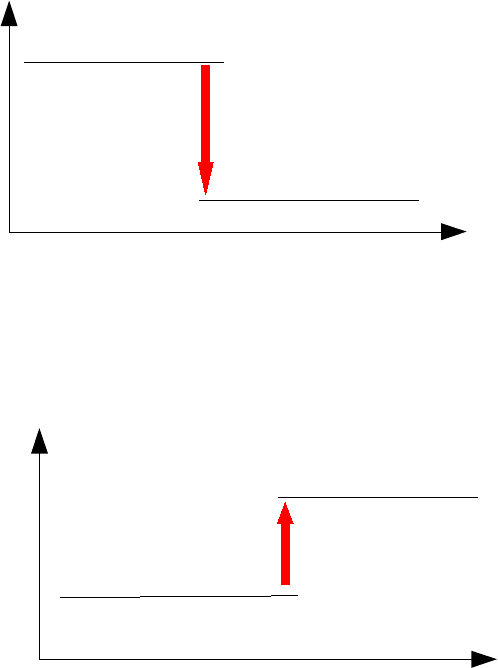
1. a) A reaction in which heat energy is produced. Correctly balanced equations (including state
symbols) for any two exothermic reactions - e.g. the obvious ones are any combustion reactions
(metals, hydrogen, hydrocarbons, etc, in oxygen), neutralisation reactions involving oxides or
hydroxides and acids, magnesium and acids. See chapter for specific examples.
b)
c) Energy is needed to break the bonds in heptane and in oxygen. Energy is released when new
bonds are made to produce the carbon dioxide and water. More energy is released when the new
bonds are made than was used to break the old ones.
2. a) A reaction in which energy is absorbed. The chapter uses the effect of heat on carbonates as
examples of endothermic reactions. It is easiest to choose any two of these.
b)
3. a) exothermic
b) exothermic
c) endothermic
d) exothermic
e) endothermic
f) exothermic
4. The cans are double skinned - the gap between the skins containing calcium oxide and (kept
separate) water. Pushing a button on the bottom of the can breaks a seal and allows the reactants
into contact, and you get the reaction described in the chapter.
CaO(s) + H
2
O(l) ----> Ca(OH)
2
(s)
It is easy to find lots of information on this by an Internet search.
energy
progress of reaction
heptane and oxygen
carbon dioxide + water
-4187 kJ
energy
progress of reaction
glucose and oxygen
carbon dioxide + water
+2820 kJ
Chapter 15
1. a) If a dynamic equilibrium is disturbed by changing the conditions, the reaction moves to
counteract the change.
b) (i) Dynamic means that the reactions are still continuing. Equilibrium means that the total
amounts of the various things present are constant. This is because the forward and back
reactions are happening at the same rates.
(ii) An increase in pressure will produce more N
2
O
4
. According to Le Chatelier, the
equilibrium moves to reduce the pressure again by producing fewer molecules.
(iii) A decrease in temperature will produce more N
2
O
4
. According to Le Chatelier, the
equilibrium moves to increase the temperature again by favouring the exothermic change.
2. a) Less carbon monoxide and hydrogen would be produced. (A high pressure favours the reaction
producing fewer molecules.)
b) Raising the temperature moves the equilibrium in the direction which would tend to reduce it
again, and so a high temperature favours the endothermic change (in this case, the forward
reaction).
c) The reaction would be too slow in the absence of the catalyst.
d) Increasing the proportion of steam will favour the forward reaction (Le Chatelier’s Principle).
Steam is also much cheaper than methane and an excess will help ensure the maximum
conversion of the methane into products.
3. a) Adding extra hydrogen ions favours the back reaction as the equilibrium shifts to reduce their
concentration again (Le Chatelier’s Principle). This produces more HLit, which is red.
b) The hydroxide ions react with the hydrogen ions in the equilibrium, reducing their concentration.
The equilibrium moves to replace them again, producing extra Lit
-
ions which are blue.
4. a) A high pressure favours the reaction producing fewer molecules (Le Chatelier’s Principle - fewer
molecules produce a lower pressure) - in this case, the production of ammonia.
b) Very high pressures are expensive to produce for two reasons: all the reaction vessels and
pipework have to be built much more strongly, and it takes a lot more energy to drive the
compressors producing the very high pressures. The extra ammonia produced isn’t worth the
extra cost.
c) The forward reaction producing ammonia is exothermic. This is favoured by a low temperature
according to Le Chatelier’s Principle (the equilibrium moves in a direction to increase the
temperature again).
d) At low temperatures the reaction is extremely slow even in the presence of a catalyst. 450°C is
chosen because it gives a reasonable percentage of ammonia in the equilibrium mixture
reasonably quickly.

End of Section B Questions
1. a) (i) barium sulfate (1)
(ii) Ba
2+
(aq) + SO
4
2-
(aq) ----> BaSO
4
(s) (1)
b) Add sodium hydroxide solution (1) and look for blue precipitate (1).
c) (i) Brown precipitate (1). Solution fades from blue to colourless (1).
(ii) zinc (1). Loss of electrons (1).
d) (i) cathode (or negative) (1)
(ii) Cu
2+
(aq) + 2e
-
----> Cu(s) (1)
(iii) reduction (1). Gain of electrons (1).
2. a) (i) solid copper(II) oxide (or hydroxide or carbonate) (1)
(ii) sodium hydroxide (or carbonate) solution (1)
(iii) silver nitrate solution (1)
b) Add a measured volume of potassium hydroxide solution to a flask using a pipette (or
measuring cylinder) (1). Add a named indicator (1). Add acid from a burette (1) until a correctly
stated colour change (1). Note the volume of acid added (1). Mix the same volumes of acid and
potassium hydroxide solution without the indicator (1).
3. a) (i) A = chlorine (1)
(ii) B = hydrogen (1) (Allow one mark overall if reversed)
b) (i) bleaches (1) damp litmus paper (1)
(ii) pops (1) with lighted splint (1) (Consequential marking from part (a). e.g. If A
was mis-named as oxygen, allow a correct test for oxygen.)
c) (i) red / brown (1) solution (1) instead of colourless gas
(ii) no change (1)
d) copper(II) (1) bromide (1)
4. a) (i) Cl
2
(g) + 2I
-
(aq) ----> 2Cl
-
(aq) + I
2
(s) (1)
(ii) Colourless solution + green gas (1) gives dark grey precipitate (1) (allow red/brown
solution).
(iii) Oxidising agent (1) because it removes electrons (1) from the iodide ions.
b) (i) iron(II) (1) chloride (1)
(ii) I = iron(II) hydroxide (1). K = iron(III) hydroxide (1)
(iii) iron(III) chloride (1)
(iv) Fe
2+
(aq) + 2OH
-
(aq) ----> Fe(OH)
2
(s) (1)
5. a) Any sensible physical property (1) - e.g. solid rather than gas.
b) Correct reagent (e.g. water or oxygen or chlorine) (1). Name products (1). Correctly balanced
equation (2)
c) (i) Dots-and-crosses diagrams showing all the electrons: Na 2,8,1; K 2,8,8,1 (2 x 1 = 2)
(ii) Loss of outer electron (1)
(iii) Potassium's electron is further from nucleus (1) and so experiences less attraction (1).
Some mention of increased nuclear charge being offset by extra screening (1).
d) A molecule consisting of a single atom (1). Argon's outer level contains 8 electrons (1). No
room for sharing another electron to make the covalent bond needed to join other atoms to it. (1)
6. a) redox (1)
b) thermal decomposition (1)
c) neutralisation (1)
d) precipitation (1)
e) redox (1)
f) redox (1)
7. a) ΔH shows the amount of heat evolved or absorbed during the reaction (1). The positive sign
shows heat is absorbed (1).
b) (i) increases
(ii) decreases
c) (i) N
2
(g) + O
2
(g) ----> 2NO(g) (1)
(ii) acid rain (1). Any valid consequence of acid rain (e.g. death of trees, loss of life in
lakes, corrosion of iron or limestone) (1)
(iii) sulfur dioxide (1)
