6-1
Experiment 6
Titration II – Acid Dissociation Constant
Introduction:
An acid/base titration can be monitored with an indicator or with a pH meter. In either case,
the goal is to determine the equivalence point of the titration. This is the point at which
enough titrant has been added to the analyte to just exactly neutralize the analyte. In this
experiment, knowledge of the equivalence point will be used to obtain information about the
acid dissociation constant, K
a,
of the acid being titrated.
When an indicator is used in a titration, the color change occurs at what is called the
endpoint. If the indicator has been properly selected, this point will be the same as the
equivalence point. When a pH meter is used, the pH of the solution is recorded as the titrant
is added. The pH versus the volume of titrant added can be plotted on what is called a
titration curve. In this case the equivalence point occurs at the point where very small
additions of titrant cause a very rapid rise in the pH. Graphically, it is also the point on the
curve where the slope, ΔpH/ΔV, changes from positive to negative (called the inflection
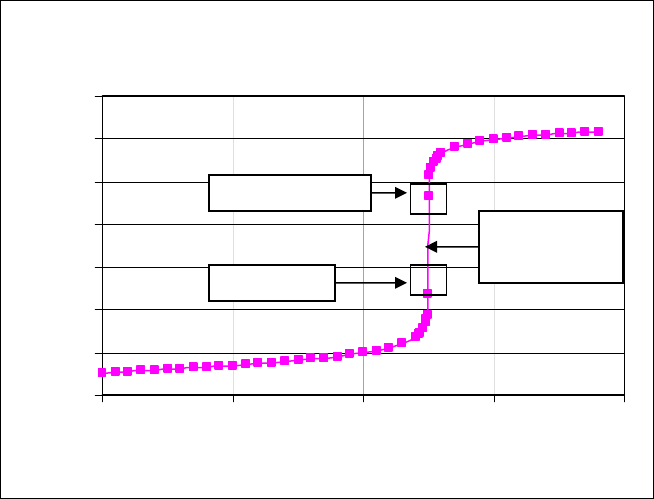
point.) Figure 1 is a titration curve for the titration of HCl by NaOH, a strong acid and strong
base, where 25.0 mL of 0.1 M HCl is titrated with 0.1 M NaOH.
NaOH titration of HCl
0
2
4
6
8
10
12
14
0 102030
mL NaOH added
p H
40
Phenol
p
hthalein
Equivalence
Point
Meth
y
l Red
Figure 1. Titration of 25.0 mL of 0.1M HCl by 0.1 M NaOH. Blocked areas on the curve
indicate the pH range in which phenolphthalein and methyl red change colors.
Note that the slope, ΔpH/ΔV, becomes large when the volume of NaOH added is at 25 mL, so
this is the equivalence point. Because of this rapid rise through a range of pH values when
the equivalence point is reached, a wide variety of indicators may be used to detect the
UCCS Chem 106 Laboratory Manual Experiment 6
6-2
endpoint visually. Either methyl red or phenolphthalein can be used for an HCl/NaOH
analysis, since both will exhibit color changes in the range of pH values at the equivalence
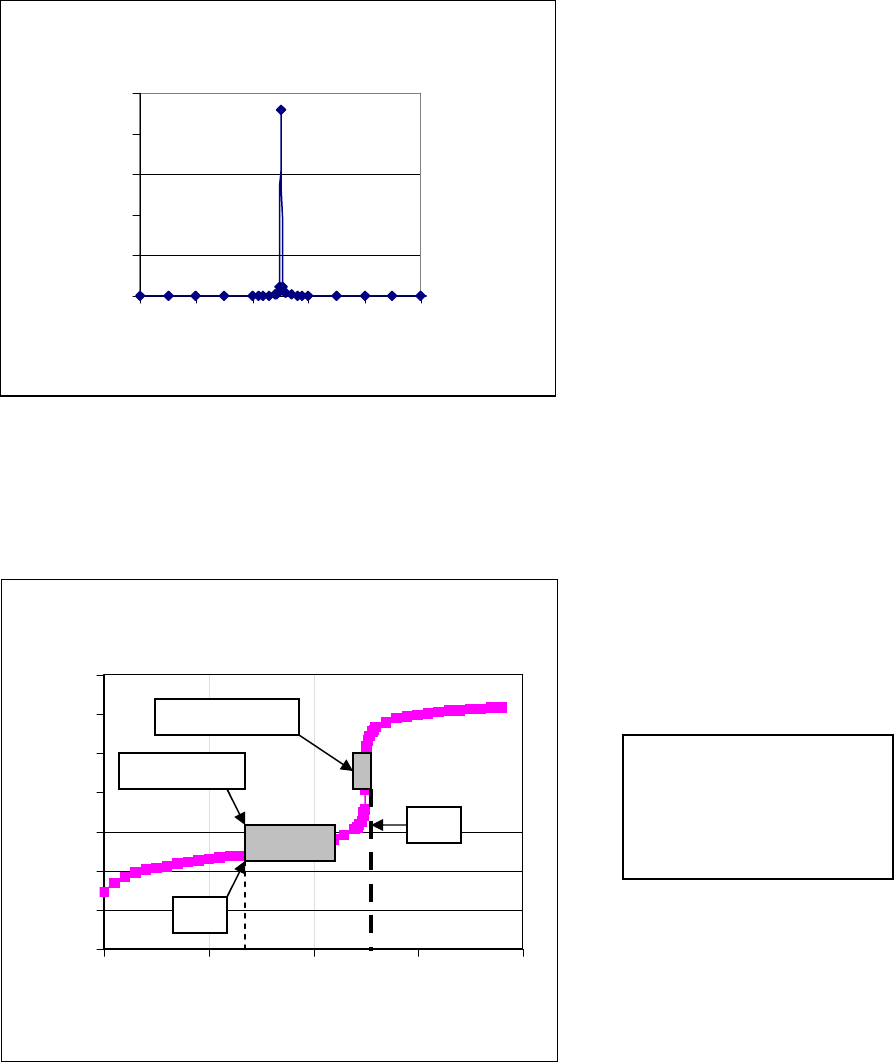
point. Also note that if the slope, ΔpH/ΔV, is plotted versus the volume of titrant added, the
inflection point will appear as a spike. This is the most precise method for determining the
equivalence point, and it is shown in Figure 2 below.
Slope vs. mL Titrant Added
0
50
100
150
200
250
20 22 24 26 28 30
mL NaoH
d(pH)/d(V)
Figure 2. Plot of slope, ΔpH/ΔV, vs. mL NaOH from 20 to 30 mL based on Figure 1 titration.
When a weak acid is titrated by a strong base, the fact that, in aqueous solution, the weak
acid dissociates into a hydrogen ion and the conjugate base of the acid changes the
appearance of the titration curve. The curve will look similar to Figure 3, which represents
the titration of 0.1 M acetic acid with 0.1 M NaOH.
NaOH titration of CH
3
COOH
0
2
4
6
8
10
12
14
0 10203040
mL NaOH added
p H
Meth
y
l red
B
A
A: Equivalence point
B: Half-equivalence point
pH at B = 4.74 = pK
a
Phenol
p
hthal
Figure 3. Titration of 25 mL of 0.1M acetic acid by 0.1M NaOH.
UCCS Chem 106 Laboratory Manual Experiment 6
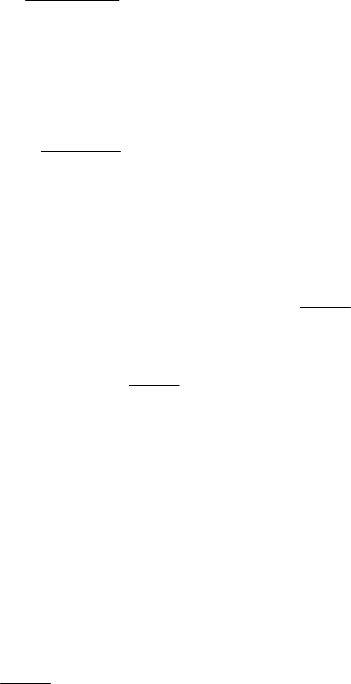
6-3
Several differences are readily apparent in the comparison of Figures 1 and 3. There are
variations in the initial pH, the rate of pH change, and the pH at the equivalence point. The
addition of a strong base to a weak acid creates a build up of the salt of the weak acid (in this
case, NaCH
3
COO) producing a buffering effect, which causes resistance to change in pH.
Also, the pH of the equivalence point corresponds to the pH of the conjugate base,
CH
3
COO
−
, which hydrolyzes in water. Notice that the endpoint of the methyl red does not
occur at the equivalence point, and therefore it could not be used in the titration. The
phenolphthalein is appropriate for this specific titration. All these effects are related to the
strength (or degree of dissociation) of the acid being titrated. Because of the incomplete
dissociation of the acid, the reaction is in equilibrium, with an acid dissociation constant, K
a,
which is specific to that acid.
For the dissociation of any weak acid, HA:
HA(aq) → H
+
(aq) + A
–
(aq)
there is an acid dissociation constant, K
a
:
[HA]
]][A[H
a
−+
=K
This can be rearranged to solve for [H
+
]:
][A
[HA]
]H[
a
−
+
=
K
Using the definition of pH, this equation can be rearranged as follows:
⎟
⎟
⎠
⎞
⎜
⎜
⎝
⎛
−−=−=
−
+
][A
[HA]
loglog]Olog[HpH
a3
K
, or
⎟
⎟
⎠
⎞
⎜
⎜
⎝
⎛
+=
[HA]
][A
log ppH
-
a
K
This last expression is known as the Henderson-Hasselbach equation. It can be used to
calculate the pK
a
(and thus K
a
) of an acid. At the equivalence point, the volume of base added
is just enough to exactly neutralize all of the acid. At one-half of this volume of added base,
called the half-equivalence point, enough has been added to neutralize half of the acid. Since
half of the acid reacted to form A
–
, the concentrations of A
–
and HA at the half-equivalence
point are the same. Therefore, at the half-equivalence point, the pH is equal to the pK
a
.
Since
a
-
ppH that followsit , 0 log(1)
[HA]
][A
log K===
⎟
⎟
⎠
⎞
⎜
⎜
⎝
⎛
.
UCCS Chem 106 Laboratory Manual Experiment 6
6-4
A plot of the titration curve allows the equivalence point to be determined. At exactly one-
half the volume of the equivalence point, the measured pH is equal to pK
a
as illustrated in
Figure 3.
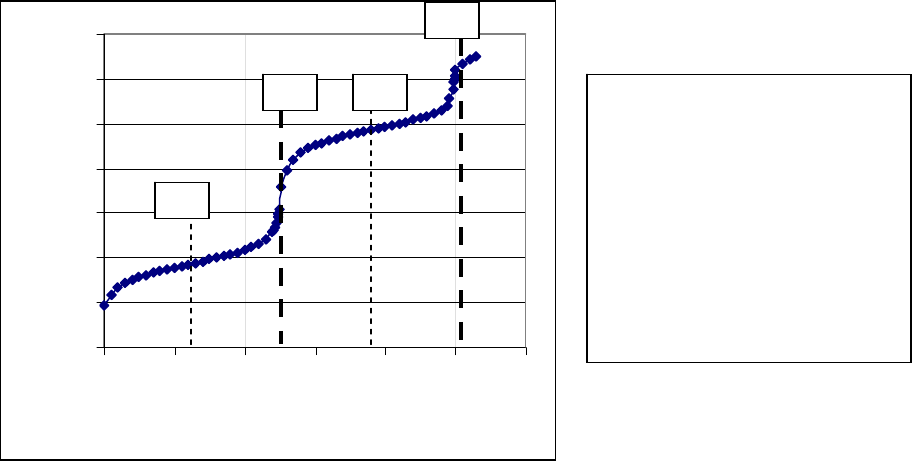
For polyprotic acids there are multiple dissociation steps and equivalence points, one for each
acidic hydrogen present. The dissociation reactions of a weak polyprotic acid, H
3
A, are
shown below along with the neutralization reactions that occur in a titration by a strong base.
H
3
A(aq) → H
+
(aq) + H
2
A
–
(aq) OH
−
+ H
3
A → H
2
O + H
2
A
–
H
2
A
–
(aq) → H
+
(aq) + HA
2–
(aq) OH
−
+ H
2
A
–
→ H
2
O + HA
2–
HA
2–
(aq) → H
+
(aq) + A
3–
(aq) OH
−
+ HA
2–
→ H
2
O + A
3–
Each step has a separate dissociation constant: K
a1,
K
a2,
and K
a3
. A titration curve for a
triprotic acid is thus expected to have three equivalence points, but often only shows two. A
titration curve for a diprotic acid, H
2
A, would show two equivalence points, one in which
OH
−
neutralizes H
2
A and a second in which OH
−
neutralizes HA
−
. The pK
a
is obtained in the
same way as for a monoprotic acid, but in this case at least two half-equivalence points are
present. One half-equivalence point occurs at one-half the volume of the first equivalence
point, at which pH = pK
a1
. The second occurs at the volume that is at the midpoint between
the first and second equivalence points, and at that point, pH = pK
a2
.
0
2
4
6
8
10
12
14
0 102030405060
mL NaOH
p H
D
C
B
A A: First equivalence point
B: Second equivalence point
C: First half equivalence point
D: Second half equivalence point
pH at C = 3.73 = pK
a1
pH at D = 9.68 = pK
a2
Figure 4. Titration curve of weak diprotic acid by NaOH(aq).
Pre-Lab Notebook: Provide a title, purpose, CH
3
COOH / NaOH reaction, brief summary of
the procedure, and table of reagents (NaOH and CH
3
COOH).
UCCS Chem 106 Laboratory Manual Experiment 6
6-5
Equipment:
50 mL Buret Vernier pH Probe
250 mL Beakers (2) Vernier LabPro
25.00 mL Volumetric Pipet TI-84 Calculator
25.00 mL Pipet and Bulb Stir Bar and Stir Plate
DI water
Ring stand, buret clamp, clamp for pH probe
In-Lab Procedure:
Note: Work in pairs.
Use the standardized NaOH solution prepared during the last lab period. Set up the Vernier
system in DATAMATE according to the Vernier tutorial with the pH probe connected to
channel 1. Calibrate the electrode as described in Experiment 4.
Part A: Titration of Acetic Acid with NaOH
Phenolphthalein:
1. Fill the buret with 0.1M NaOH. Pipet 25.00 mL of 0.1 M CH
3
COOH into a 250 mL
beaker and add 3–4 drops of phenolphthalein indicator. Place the beaker on a white
paper towel to best observe color changes.
2. Titrate the solution by adding the NaOH titrant in 1–2 mL increments. Carefully swirl
the beaker with each addition.
3. The colored form of the phenolphthalein will begin to stay for a while and then
disappear. At this point add the NaOH dropwise until the acetic acid is a very light
color. This is the endpoint for phenolphthalein.
4. Measure and record the pH of the solution in the beaker at this end point. Then rinse
the pH probe with DI water and replace the probe tip into its vial.
5. Record any color change observed during the titration in your laboratory notebook.
The pH and added NaOH volume at that indicator’s endpoint should be used to
estimate the target point when conducting the following procedure.
Making a Titration Curve:
1. Rinse and thoroughly dry the two 250 mL beakers. Refill the buret to the 0.00 mL
mark with 0.1 M NaOH.
2. Pipet 25.00 mL of 0.1 M CH
3
COOH into a 250 mL beaker. Place a stir bar in the
beaker, and place the beaker on a stir plate.
3. Set up a ring stand with a buret clamp and another clamp to hold the pH probe. Stir
the solution on a low setting throughout the titration. Ensure that the probe tip is
completely immersed in the acetic acid.
4. Set up the Vernier system to take a pH reading after each volume of NaOH is added.
On the main screen select “1” for SETUP. Select “6” for TIME GRAPH, “3” for EVENTS
WITH ENTRY
, and then “1” for OK. At the main screen press “2” to START data
collection.
5. When the pH stabilizes (and before adding any NaOH), press
ENTER and type in
“0” as the buret volume in mL. Then add the NaOH in increments of 1-2 mL
(recording the pH by pressing ENTER at each new total volume of NaOH added and
UCCS Chem 106 Laboratory Manual Experiment 6
6-6
then entering the buret reading). Continue until the volume added and pH approach
the values at the endpoint you determined with the phenolphthalein. When the pH
nears this value, add the NaOH in 0.5 and finally 0.2 mL increments. When the pH
passes the equivalence point, add the NaOH again in 1-2 mL increments until you
have added 35 mL.
6. You should have taken approximately 30 data points. Press STO> when you have
finished collecting data.
7. Transfer your data to the Logger Pro program and transfer the pH/volume data to an
Excel file for later analysis. Store the volumes in the A column and the pH values in
the B column of the spreadsheet, beginning in cells A1 and B1.
Part B: Titration of an Unknown Polyprotic Acid with NaOH
1. Transfer 15.00 mL of an unknown 0.1 M acid solution to a 250 mL beaker with a
15.00 mL volumetric pipet you have pre-rinsed with a small amount of the solution.
2. Fill a buret to the 0.00 mL mark with standardized NaOH.
3. Rinse the pH meter with DI water. Set up a titration as you did for Part A using the
Vernier equipment to measure the pH in the EVENTS WITH ENTRY mode
4. Titrate the unknown acid with NaOH using 1 mL increments of the base except near
the first endpoint (as indicated by the more rapid rise in pH) where the increments
should be 0.2 mL. After the first endpoint, increase the increments to 1 mL until the
second end point is approached. Continue the titration until the second end point has
been passed by about 5 mL of added NaOH
5. Transfer this data into Logger Pro as before and save it to an Excel file (volume in
column A and pH in column B) under a different name for later analysis.
For Next Lab: Prepare both the saturated Ca(OH)
2
and the saturated Ca(OH)
2
with
CaCl
2
⋅H
2
O solutions according to the directions given in Experiment #7. Cover with Parafilm
for use during next lab.
Treatment of Data:
Part A: Titration of Acetic Acid with NaOH
1. Using the saved data in your Excel file prepare a plot of pH vs. Volume of NaOH
added to observe the equivalence point and half equivalence point.
2. To verify the equivalence point, determine the inflection point by calculating the
change in pH per change in volume, ΔpH/ΔV, for each recorded volume, as shown in
Figure 2 for the NaOH/HCl titration. Plot ΔpH/ΔV vs. the volume of titrant added. To
perform this, check that your volume and pH values are in columns A and B on your
Excel spreadsheet. In cell C1, type: =(A1+A2)/2. Click on cell C1 and drag down to
the C cell that is on the same row as the last filled cell in columns A and B. Column C
now contains the average volume of titrant between two readings.
3. In cell D1, type: =(B2-B1)/(A2-A1) which will calculate the ΔpH/ΔV values.
Highlight cell D1 and drag to fill the remaining cells as you did with column C.
Column D now contains the values of ΔpH/ΔV for each volume reading. The
inflection point is found by plotting ΔpH/ΔV versus the volumes in column C.
4. Once the volume at the equivalence point is known, the volume of the half-
UCCS Chem 106 Laboratory Manual Experiment 6
6-7
equivalence point can be found. Using the titration curve, the pH at this volume can
be determined as shown in Figure 3. Thus pK
a
and hence K
a
for acetic acid can be
calculated. Compare this to the literature value (1.8 × 10
−5
).
Part B: Titration of an Unknown Acid with NaOH
1. To find the inflection points and half-inflection points for the unknown acid, use the
same method as above. Refer to Figure 4 to see how the values of pK
a1
and pK
a2
are
related to the pH values at the two half-inflection points.
2. Calculate K
a1
and K
a2
from pK
a1
and pK
a2
and identify the unknown by comparison
with the K
a1
and K
a2
values for polyprotic acids listed in the table below.
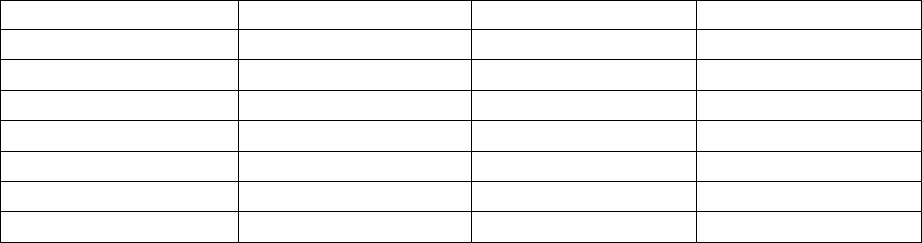
Acid K
a1
K
a2
K
a3
Boric, H
3
BO
3
7.3 × 10
-10
1.8 × 10
-13
1.6 × 10
-14
Carbonic, H
2
CO
3
4.2 × 10
-7
4.8 × 10
-11
Citric, H
3
C
6
H
5
O
7
7.4 × 10
-3
1.7 × 10
-5
4.0 × 10
-7
Hydrosulfuric, H
2
S
1 × 10
-7
1 × 10
-19
Oxalic, H
2
C
2
O
4
5.9 × 10
-2
6.4 × 10
-5
Phosphoric, H
3
PO
4
7.5 × 10
-3
6.2 × 10
-8
3.6 × 10
-13
Phosphorous, H
3
PO
3
1.6 × 10
-2
7.0 × 10
-7
Lab Report Outline for Titration II
Graded Sections of Report Percent of Grade
Pre-Lab 10
In-Lab 10
Post-Lab
Part A: Titration of Acetic Acid with NaOH
1. Attach an Excel plot of pH vs. volume of NaOH added. 10
2. Attach a plot of ΔpH/ΔV versus the average volume of NaOH added. 10
3. State “The volume at the half-equivalence point is __________. The pH at the half-
equivalence point is ___________. Thus K
a
for acetic acid is ________.” 15
Part B: Titration of an Unknown Polyprotic Acid with NaOH
1. Attach an Excel plot of pH vs. volume of NaOH added. 10
2. Attach a plot of ΔpH/ΔV versus the average volume of NaOH added. 10
3. Complete a table as shown below. In the entry “K
a1
and K
a2
from Text table,” enter the
values from the table that correspond most closely to your experimental value. Then state:
“The unknown polyprotic acid is _______________________.” 15
UCCS Chem 106 Laboratory Manual Experiment 6
6-8
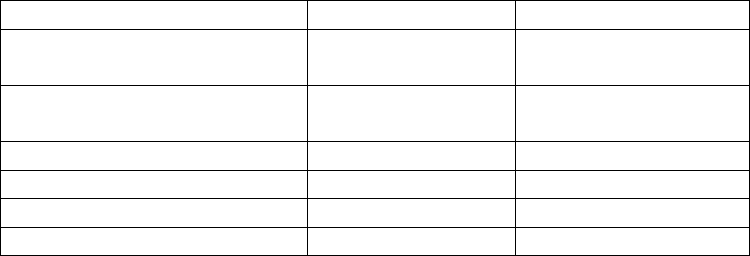
First Second
Equivalence point volume
(mL NaOH)
Half-equivalence point
volume (mL NaOH)
Equivalence point pH
Half-equivalence point pH
K
a1
and K
a2
experimental
K
a1
and K
a2
from Text table
4. Was the equivalence point determined from the end point of the titration with
phenolphthalein the same as the one determined by the plot of ΔpH/ΔV versus the average
volume of NaOH added? Discuss any difference. 5
5. Explain why methyl red could not be used as an indicator for the NaOH titration of acetic
acid. 5
UCCS Chem 106 Laboratory Manual Experiment 6
