
11/21/2016
34(6)SixthInterimRevisionAnnouncement:61<61>MICROBIOLOGICALEXAMINATIONOFNONSTERILEPRODUCTS:MICROBIALENUMERA…
http://www.usppf.com/pf/pub/index.html 1/9
61 MICROBIOLOGICALEXAMINATIONOFNONSTERILEPRODU CTS:MICROBIAL
ENUMERATIONTESTS
INTRODUCTION
Thetestsdescribedhereafterwillallowquantitativeenumerationofmesophilicbacteriaandfungithat
maygrowunderaerobicconditions.
Thetestsaredesignedprimarilytodeterminewhetherasubstanceorpreparationcomplieswithan
establishedspecificationformicrobiologicalquality.Whenusedforsuchpurposes,followthe
instructionsgivenbelow,includingthenumberofsamplestobetaken,andinterprettheresultsas
statedbelow.
Themethodsarenotapplicabletoproductscontainingviablemicroorganismsasactiveingredients.
Alternativemicrobiologicalprocedures,includingautomatedmethods,maybeused,providedthat
theirequivalencetothePharmacopeialmethodhasbeendemonstrated.
GENERALPROCEDURES
Carryoutthedeterminationunderconditionsdesignedtoavoidextrinsicmicrobialcontaminationof
theproducttobeexamined.Theprecautionstakentoavoidcontaminationmustbesuchthattheydo
notaffectanymicroorganismsthataretoberevealedinthetest.
Iftheproducttobeexaminedhasantimicrobialactivity,thisis,insofaraspossible,removedor
neutralized.Ifinactivatorsareusedforthispurpose,theirefficacyandtheirabsenceoftoxicityfor
microorganismsmustbedemonstrated.
Ifsurfaceactivesubstancesareusedforsamplepreparation,theirabsenceoftoxicityfor
microorganismsandtheircompatibilitywithanyinactivatorsusedmustbedemonstrated.
ENUMERATIONME THODS
UsetheMembraneFiltrationmethodoroneofthePla teCountMethods,asdirected.TheMost
ProbableNumber(MPN)Methodisgenerallytheleastaccuratemethodformicrobialcounts;however,
forcertainproductgroupswithverylowbioburden,itmaybethemostappropriatemethod.
Thechoiceofamethodisbasedonfactorssuchasthenatureoftheproductandtherequiredlimitof
microorganisms.Themethodchosenmustallowtestingofasufficientsamplesizetojudgecompliance
withthespecification.Thesuitabilityofthechosenmethodmustbeestablished.
Changetoread:
GROWTHPROMOTIONTEST, SUITABILITYOFTHECOUNTINGMET HOD ANDNEGATIVE
CONTROLS
GeneralConsiderations
Theabilityofthetesttodetectmicroorganismsinthepresenceofproducttobetestedmustbe
established.
6
6
11/21/2016
34(6)SixthInterimRevisionAnnouncement:61<61>MICROBIOLOGICALEXAMINATIONOFNONSTERILEPRODUCTS:MICROBIALENUMERA…
http://www.usppf.com/pf/pub/index.html 2/9
Suitabilitymustbeconfirmedifachangeintestingperformanceorachangeintheproductthatmay
affecttheoutcomeofthetest,isintroduced.
PreparationofTestStrains
Usestandardizedstablesuspensionsofteststrainsorprepareasstatedbelow.Seedlotculture
maintenancetechniques(seedlotsystems)areusedsothattheviablemicroorganismsusedfor
inoculationarenotmorethan5passagesremovedfromtheoriginalmasterseedlot.Groweachofthe
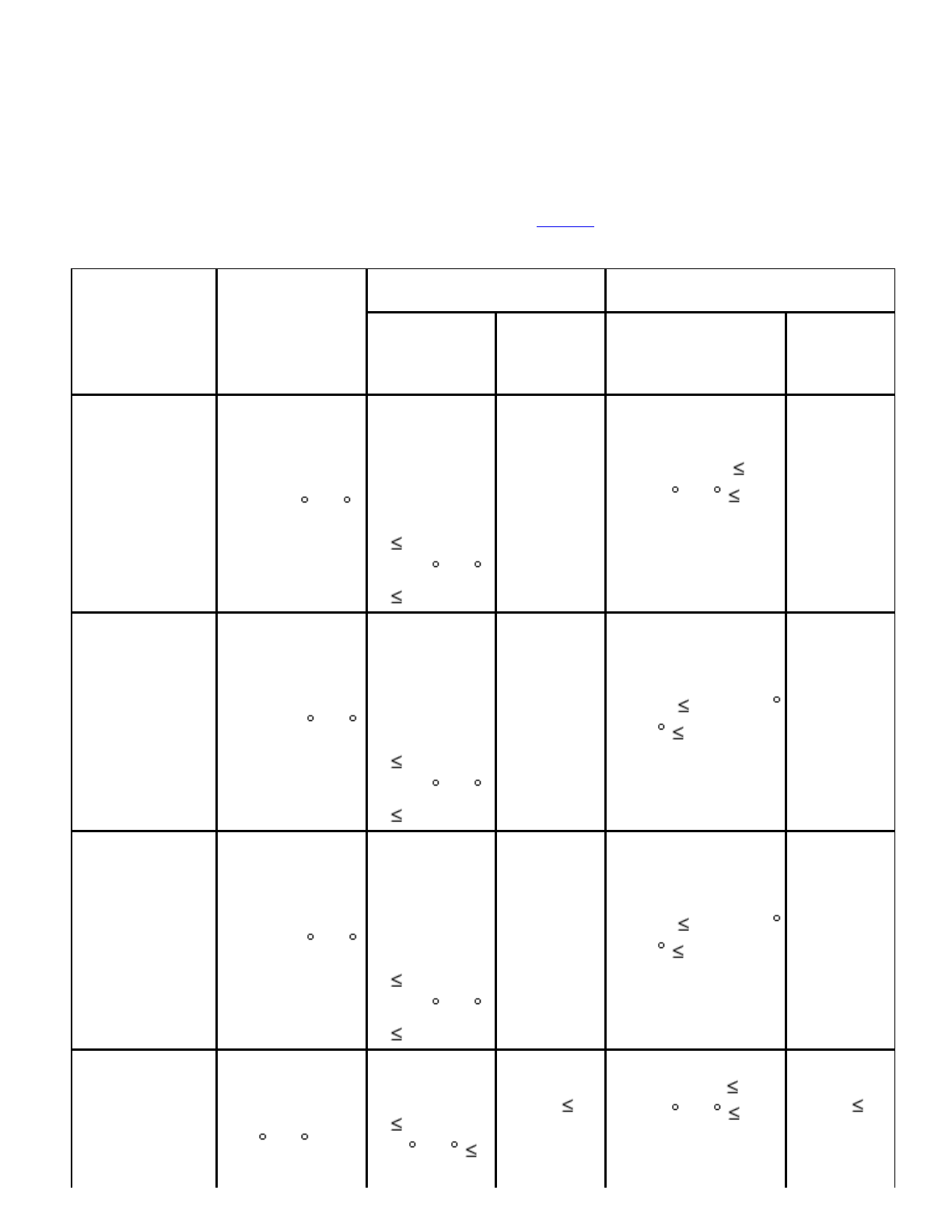
bacterialandfungalteststrainsseparatelyasdescribedinTable1.
Table1.PreparationandUseofTestMicroorganisms
Microorganism
Preparationof
Test
Strain
GrowthPromotion
SuitabilityofCountingMethodin
thePresenceofProduct
TotalAerobic
Microbial
Count
Total
Yeasts
andMolds
Count
TotalAerobic
MicrobialCount
Total
Yeasts
andMolds
Count
Staphylococcus
aureussuchas
ATCC6538,
NCIMB9518,
CIP4.83,or
NBRC13276
Soybean–
CaseinDigest
Agaror
Soybean–
CaseinDigest
Broth30 –35
18–24hours
Soybean–
Casein
DigestAgar
and
Soybean–
Casein
DigestBroth
100
cfu30 –35
3days
Soybean–Casein
DigestAgar/MPN
Soybean–Casein
DigestBroth 100
cfu30 –35 3
days
Pseudomonas
aeruginosa
suchasATCC
9027,NCIMB
8626,CIP
82.118,or
NBRC13275
Soybean–
CaseinDigest
Agaror
Soybean–
CaseinDigest
Broth30 –35
18–24hours
Soybean–
Casein
DigestAgar
and
Soybean–
Casein
DigestBroth
100
cfu30 –35
3days
Soybean–Casein
Digest
Agar/MPNSoybean–
CaseinDigest
Broth 100cfu30
–35 3days
Bacillus
subtilissuchas
ATCC6633,
NCIMB8054,
CIP52.62,or
NBRC3134
Soybean–
CaseinDigest
Agaror
Soybean–
CaseinDigest
Broth30 –35
18–24hours
Soybean–
Casein
DigestAgar
and
Soybean–
Casein
DigestBroth
100
cfu30 –35
3days
Soybean–Casein
Digest
Agar/MPNSoybean–
CaseinDigest
Broth 100cfu30
–35 3days
Candida
albicanssuch
asATCC
10231,NCPF
3179,IP48.72,
orNBRC1594
Sabouraud
DextroseAgar
orSabouraud
DextroseBroth
20 –25 2–3
days
Soybean–
Casein
DigestAgar
100cfu
30 –35
5days
Sabouraud
Dextrose
Agar
100cfu
Soybean–Casein
DigestAgar 100
cfu30 –35 5
daysMPN:not
applicable
Sabouraud
Dextrose
Agar
100cfu
11/21/2016
34(6)SixthInterimRevisionAnnouncement:61<61>MICROBIOLOGICALEXAMINATIONOFNONSTERILEPRODUCTS:MICROBIALENUMERA…
http://www.usppf.com/pf/pub/index.html 3/9
20 –25
5days
20 –25
5days
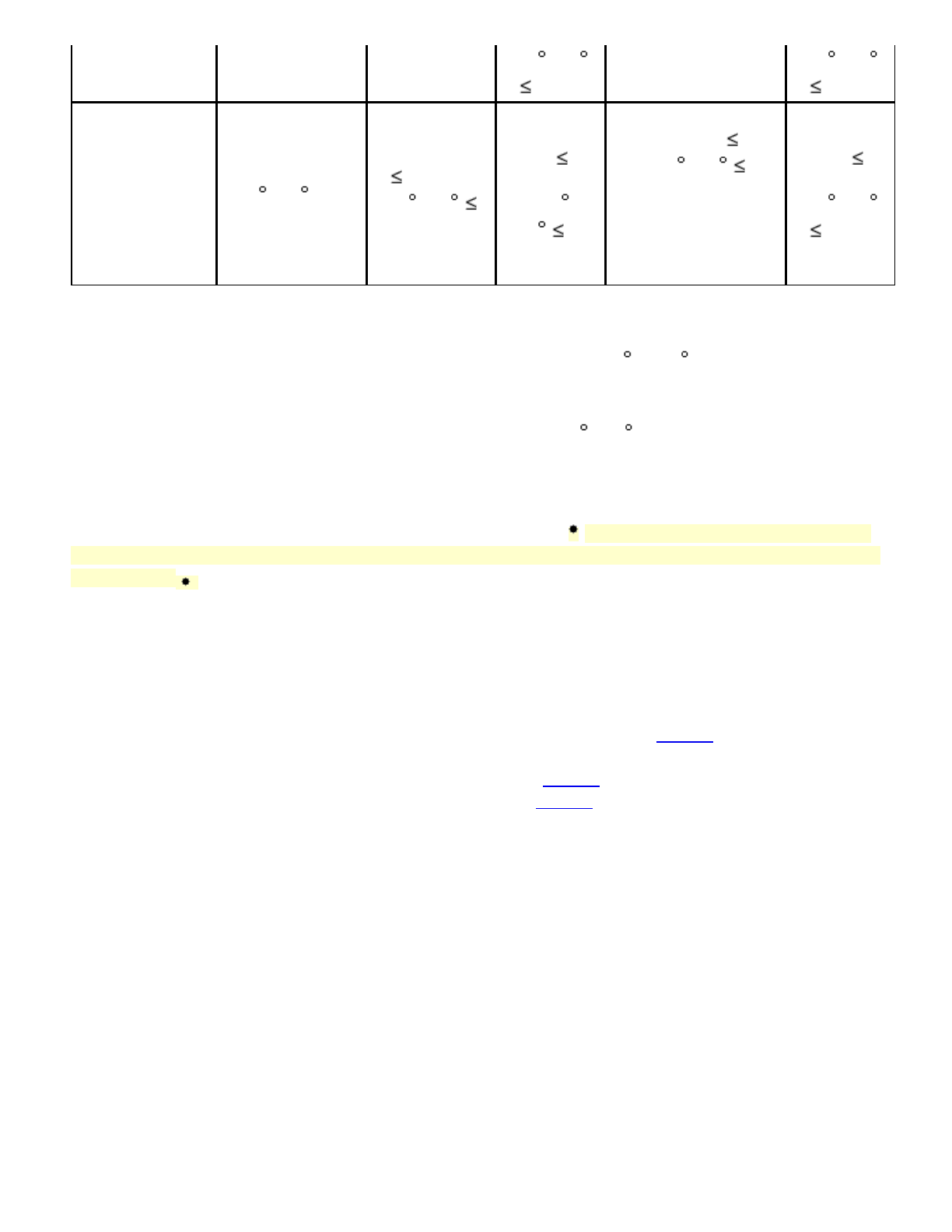
Aspergillus
nigersuchas
ATCC16404,
IMI149007,IP
1431.83,or
NBRC9455
Sabouraud
DextroseAgar
orPotato–
DextroseAgar
20 –25 5–7
days,oruntil
good
sporulationis
achieved
Soybean–
Casein
DigestAgar
100cfu
30 –35
5days
Sabouraud
Dextrose
Agar
100
cfu20 –
25 5
days
Soybean–Casein
DigestAgar 100
cfu30 –35 5
daysMPN:not
applicable
Sabouraud
Dextrose
Agar
100cfu
20 –25
5days
UseBufferedSodiumChloride–PeptoneSolutionpH7.0orPhosphateBufferSolutionpH7.2tomake
testsuspensions;tosuspendA.nigerspores,0.05%ofpolysorbate80maybeaddedtothebuffer.Use
thesuspensionswithin2hours,orwithin24hoursifstoredbetween2 and8 .Asanalternativeto
preparingandthendilutingafreshsuspensionofvegetativecellsofA.nigerorB.subtilis,astable
sporesuspensionispreparedandthenanappropriatevolumeofthesporesuspensionisusedfortest
inoculation.Thestablesporesuspensionmaybemaintainedat2 to8 foravalidatedperiodoftime.
NegativeControl
Toverifytestingconditions,anegativecontrolisperformedusingthechosendiluentinplaceofthe
testpreparation.Theremustbenogrowthofmicroorganisms. Anegativecontrolisalsoperformed
whentestingtheproductsasdescribedunderTestingofProducts.Afailednegativecontrolrequiresan
investigation.
GrowthPromotionoftheMedia
Testeachbatchofreadypreparedmediumandeachbatchofmediumpreparedeitherfrom
dehydratedmediumorfromtheingredientsdescribed.
Inoculateportions/platesofSoybean–CaseinDigestBrothandSoybean–CaseinDigestAgarwitha
smallnumber(notmorethan100cfu)ofthemicroorganismsindicatedinTable1,usingaseparate
portion/plateofmediumforeach.InoculateplatesofSabouraudDex troseAgarwithasmallnumber
(notmorethan100cfu)ofthemicroorganismsindicatedinTable1,usingaseparateplateofmedium
foreach.IncubateaccordingtotheconditionsdescribedinTable1.
Forsolidmedia,growthobtainedmustnotdifferbyafactorgreaterthan2fromthecalculatedvalue
forastandardizedinoculum.Forafreshlypreparedinoculum,growthofthemicroorganisms
comparabletothatpreviouslyobtainedwithapreviouslytestedandapprovedbatchofmediumoccurs.
Liquidmediaaresuitableifclearlyvisiblegrowthofthemicroorganismscomparabletothatpreviously
obtainedwithapreviouslytestedandapprovedbatchofmediumoccurs.
Suit abilityoftheCountingMethodinthePresenceofProduct
PREPARATIONOFTHESAMPLE
Themethodforsamplepreparationdependsonthephysicalcharacteristicsoftheproducttobe
tested.Ifnoneoftheproceduresdescribedbelowcanbedemonstratedtobesatisfactory,asuitable
alternativeproceduremustbedeveloped.
WaterSolubleProducts—Dissolveordilute(usuallya1in10dilutionisprepared)theproducttobe
examinedinBufferedSodiumChloride–PeptoneSolutionpH7.0,PhosphateBufferSol utionpH7.2,or
6

11/21/2016
34(6)SixthInterimRevisionAnnouncement:61<61>MICROBIOLOGICALEXAMINATIONOFNONSTERILEPRODUCTS:MICROBIALENUMERA…
http://www.usppf.com/pf/pub/index.html 4/9
Soybean–CaseinDigestBroth.Ifnecessary,adjusttoapHof6to8.Furtherdilutions,wherenecessary,
arepreparedwiththesamediluent.
NonfattyProductsInsolubleinWater—Suspendtheproducttobeexamined(usuallya1in10
dilutionisprepared)inB ufferedSodiumChloride–PeptoneSolutionpH7.0,PhosphateBufferSolutionpH
7.2,orSoybe an–Case inDigestBroth.Asurfaceactiveagentsuchas1gperLofpolysorbate80maybe
addedtoassistthesuspensionofpoorlywettablesubstances.Ifnecessary,adjusttoapHof6to8.
Furtherdilutions,wherenecessary,arepreparedwiththesamediluent.
FattyP roducts—Dissolveinisopropylmyristatesterilizedbyfiltration,ormixtheproducttobe
examinedwiththeminimumnecessaryquantityofsterilepolysorbate80oranothernoninhibitorysterile
surfaceactivereagentheated,ifnecessary,tonotmorethan40 or,inexceptionalcases,tonotmore
than45 .Mixcarefullyandifnecessarymaintainthetemperatureinawaterbath.Addasufficient
quantityoftheprewarmedchosendiluenttomakea1in10dilutionoftheoriginalproduct.Mix
carefully,whilemaintainingthetemperaturefortheshortesttimenecessaryfortheformationofan
emulsion.Furtherserial10folddilutionsmaybepreparedusingthechosendiluentcontainingasuitable
concentrationofsterilepolysorbate80oranothernoninhibitorysterilesurfaceactivereagent.
FluidsorSolidsinAerosolForm—Asepticallytransfertheproductintoamembranefilterapparatusor
asterilecontainerforfurthersampling.Useeitherthetotalcontentsoradefinednumberofmetered
dosesfromeachofthecontainerstested.
TransdermalPatches—Removetheprotectivecoversheets(“releaseliners”)ofthetransdermal
patchesandplacethem,adhesivesideupwards,onsterileglassorplastictrays.Covertheadhesive
surfacewithasuitablesterileporousmaterial(e.g.,sterilegauze)topreventthepatchesfromsticking
together,andtransferthepatchestoasuitablevolumeofthechosendiluentcontaininginactivators
suchaspolysorbate80and/orlecithin.Shakethepreparationvigorouslyforatleast30minutes.
INOCULATIONANDDILUTION
Addtothesamplepreparedasdirectedaboveandtoacontrol(withnotestmaterialincluded)a
sufficientvolumeofthemicrobialsuspensiontoobtainaninoculumofnotmorethanthan100cfu.The
volumeofthesuspensionoftheinoculumshouldnotexceed1%ofthevolumeofdilutedproduct.
Todemonstrateacceptablemicrobialrecoveryfromtheproduct,thelowestpossibledilutionfactorof
thepreparedsamplemustbeusedforthetest.Wherethisisnotpossibleduetoantimicrobialactivity
orpoorsolubility,furtherappropriateprotocolsmustbedeveloped.Ifinhibitionofgrowthbythesample
cannototherwisebeavoided,thealiquotofthemicrobialsuspensionmaybeaddedafterneutralization,
dilution,orfiltration.
NEUTRALIZATION/REMOVALOFANTIMICROBIALACTIVITY
Thenumberofmicroorganismsrecoveredfromthepreparedsampledilutedasdescribedin
InoculationandDilutionandincubatedfollowingtheproceduredescribedinRecoveryofMicroorganisms
inthePresence ofProduct,iscomparedtothenumberofmicroorganismsrecoveredfromthecontrol
preparation.
Ifgrowthisinhibited(reductionbyafactorgreaterthan2),thenmodifytheprocedureforthe
particularenumerationtesttoensurethevalidityoftheresults.Modificationoftheproceduremay
include,forexample,
1.Anincreaseinthevolumeofthediluentorculturemedium;
2.Incorporationofaspecificorgeneralneutralizingagentsintothediluent;
3.Membranefiltration;or
4.Acombinationoftheabovemeasures.

11/21/2016
34(6)SixthInterimRevisionAnnouncement:61<61>MICROBIOLOGICALEXAMINATIONOFNONSTERILEPRODUCTS:MICROBIALENUMERA…
http://www.usppf.com/pf/pub/index.html 5/9
NeutralizingAgents—Neutralizingagentsmaybeusedtoneutralizetheactivityofantimicrobial
agents(seeTable2).Theymaybeaddedtothechosendiluentorthemediumpreferablybefore
sterilization.Ifused,theirefficacyandtheirabsenceoftoxicityformicroorganismsmustbe
demonstratedbycarryingoutablankwithneutralizerandwithoutproduct.
Table2.CommonNeutralizingAgents/Methodsfor
InterferingSubstances
InterferingSubstance
PotentialNeutralizing
Agents/Method
Glutaraldehyde,mercurials Sodiumhydrogensulfite
(Sodiumbisulfite)
Phenolics,alcohol,aldehydes,sorbate Dilution
Aldehydes Glycine
Quaternaryammoniumcompounds(QACs),parahydroxybenzoates
(parabens),bisbiguanides
Lecithin
QACs,iodine,parabens Polysorbate
Mercurials Thioglycollate
Mercurials,halogens,aldehydes Thiosulfate
EDTA(edetate) MgorCaions
Ifnosuitableneutralizingmethodcanbefound,itcanbeassumedthatthefailuretoisolatethe
inoculatedorganismisattributabletothemicrobicidalactivityoftheproduct.Thisinformationservesto
indicatethatthearticleisnotlikelytobecontaminatedwiththegivenspeciesofthemicroorganism.
However,itispossiblethattheproductinhibitsonlysomeofthemicroorganismsspecifiedherein,but
doesnotinhibitothersnotincludedamongtheteststrainsorthoseforwhichthelatterarenot
representative.Then,performthetestwiththehighestdilutionfactorcompatiblewithmicrobialgrowth
andthespecificacceptancecriterion.
RECOVERYOFMICROORGANISMSINTHEPRESENCEOFPRODUCT
Foreachofthemicroorganismslisted,separatetestsareperformed.Onlymicroorganismsofthe
addedteststrainarecounted.
MembraneFiltration—Usemembranefiltershavinganominalporesizenotgreaterthan0.45µm.The
typeoffiltermaterialischoseninsuchawaythatthebacteriaretainingefficiencyisnotaffectedby
thecomponentsofthesampletobeinvestigated.Foreachofthemicroorganismslisted,onemembrane
filterisused.
TransferasuitablequantityofthesamplepreparedasdescribedunderPreparation oftheSample ,
InoculationandDilution,andNeutralization/Remo valofAntimicrobialActivity(preferablyrepresenting1
goftheproduct,orlessiflargenumbersofcfuareexpected)tothemembranefilter,filter
immediately,andrinsethemembranefilterwithanappropriatevolumeofdiluent.
Forthedeterminationoftotalaerobicmicrobialcount(TAMC),transferthemembranefiltertothe
surfaceoftheSoybean–CaseinDigestAgar.Forthedeterminationoftotalcombinedyeastsandmolds
count(TYMC),transferthemembranetothesurfaceoftheSabouraud DextroseAgar.Incubatethe
platesasindicatedinTable1.Performthecounting.
PlateCountMethods—Performplatecountmethodsatleastinduplicateforeachmedium,anduse
themeancountoftheresult.
PourPlateMethod—ForPetridishes9cmindiameter,addtothedish1mLofthesamplepreparedas
describedunderPreparationoftheSample ,Inoculationand Dilution,andNeutralization/Removalof
11/21/2016
34(6)SixthInterimRevisionAnnouncement:61<61>MICROBIOLOGICALEXAMINATIONOFNONSTERILEPRODUCTS:MICROBIALENUMERA…
http://www.usppf.com/pf/pub/index.html 6/9
AntimicrobialActivityand15to20mLofSoybean–CaseinDigestAgarorSabouraudDextroseAgar,
bothmediamaintainedatnotmorethan45 .IflargerPetridishesareused,theamountofagar
mediumisincreasedaccordingly.ForeachofthemicroorganismslistedinTable 1,atleasttwoPetri
dishesareused.
IncubatetheplatesasindicatedinTable1.Takethearithmeticmeanofthecountspermedium,and
calculatethenumberofcfuintheoriginalinoculum.
Sur faceSpreadMethod—ForPetridishes9cmindiameter,add15to20mLofSoybean–CaseinDigest
AgarorSabouraudDextroseAgaratabout45 toeachPetridish,andallowtosolidify.IflargerPetri
dishesareused,thevolumeoftheagarisincreasedaccordingly.Drytheplates,forexample,ina
laminarairflowcabinetorinanincubator.ForeachofthemicroorganismslistedinTable1,atleasttwo
Petridishesareused.Spreadameasuredvolumeofnotlessthan0.1mLofthesample,preparedas
directedunderPreparationo ftheSample,Ino culationandDilution,andNeutralization/Removalof
AntimicrobialActivityoverthesurfaceofthemedium.IncubateandcountasdirectedforPo urPlate
Method.
MostProba bleNumber(MPN)Method—TheprecisionandaccuracyoftheMPNMethodislessthan
thatoftheMembraneFiltrationmethodorthePlateCountMetho d.Unreliableresultsareobtained
particularlyfortheenumerationofmolds.Forthesereasons,theMPNMethodisreservedforthe
enumerationofTAMCinsituationswherenoothermethodisavailable.Iftheuseofthemethodis
justified,proceedasfollows.
Prepareaseriesofatleastthreeserial10folddilutionsoftheproductasdescribedforPreparationof
theSa mple,Inoculatio nandDilution, andNeutr alization/RemovalofAntimicrobialActivity.Fromeach
levelofdilution,threealiquotsof1gor1mLareusedtoinoculatethreetubeswith9to10mLof
Soybean–CaseinDigestBroth.Ifnecessaryasurfaceactiveagentsuchaspolysorbate80,oran
inactivatorofantimicrobialagentsmaybeaddedtothemedium.Thus,ifthreelevelsofdilutionare
prepared,ninetubesareinoculated.
Incubatealltubesat30 to35 fornotmorethan3days.Ifreadingoftheresultsisdifficultor
uncertainowingtothenatureoftheproducttobeexamined,subcultureinthesamebrothorin
Soybean–CaseinDigestAgarfor1to2daysatthesametemperature,andusetheseresults.From
Table3,determinethemostprobablenumberofmicroorganismspergormLoftheproducttobe
examined.
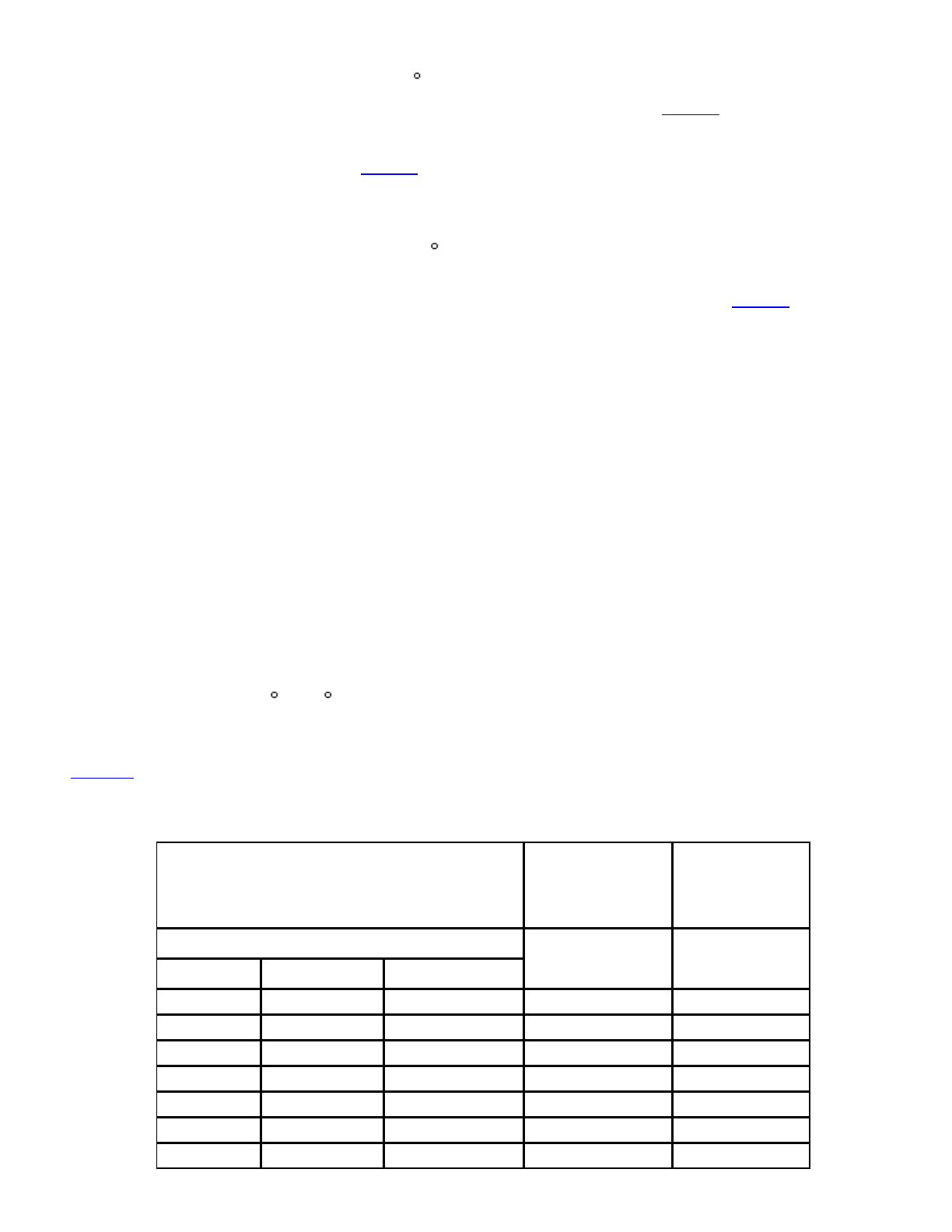
Table3.MostProbableNumberVa luesofMicroo rganisms
Observed
Combinations
ofNumbersofTubes
ShowingGrowthinEachSet
MPNpergor
permLof
Product
95%
Confidence
Limits
NumberofgormLofProductperTube
0.1 0.01 0.001
0 0 0 <3 0–9.4
0 0 1 3 0.1–9.5
0 1 0 3 0.1–10
0 1 1 6.1 1.2–17
0 2 0 6.2 1.2–17
0 3 0 9.4 3.5–35
1 0 0 3.6 0.2–17

11/21/2016
34(6)SixthInterimRevisionAnnouncement:61<61>MICROBIOLOGICALEXAMINATIONOFNONSTERILEPRODUCTS:MICROBIALENUMERA…
http://www.usppf.com/pf/pub/index.html 7/9
Observed
Combinations
ofNumbersofTubes
ShowingGrowthinEachSet
MPNpergor
permLof
Product
95%
Confidence
Limits
NumberofgormLofProductperTube
0.1 0.01 0.001
1 0 1 7.2 1.2–17
1 0 2 11 4–35
1 1 0 7.4 1.3–20
1 1 1 11 4–35
1 2 0 11 4–35
1 2 1 15 5–38
1 3 0 16 5–38
2 0 0 9.2 1.5–35
2 0 1 14 4–35
2 0 2 20 5–38
2 1 0 15 4–38
2 1 1 20 5–38
2 1 2 27 9–94
2 2 0 21 5–40
2 2 1 28 9–94
2 2 2 35 9–94
2 3 0 29 9–94
2 3 1 36 9–94
3 0 0 23 5–94
3 0 1 38 9–104
3 0 2 64 16–181
3 1 0 43 9–181
3 1 1 75 17–199
3 1 2 120 30–360
3 1 3 160 30–380
3 2 0 93 18–360
3 2 1 150 30–380
3 2 2 210 30–400
3 2 3 290 90–990
3 3 0 240 40–990
3 3 1 460 90–1980
3 3 2 1100 200–4000
3 3 3 >1100
RESULTSANDINTERPRETATION
WhenverifyingthesuitabilityoftheMembraneFiltr ationmethodorthePl ateCountMethod,amean
countofanyofthetestorganismsnotdifferingbyafactorgreaterthan2fromthevalueofthecontrol
definedinInoculationa ndDilutio nintheabsenceofproductmustbeobtained.Whenverifyingthe

11/21/2016
34(6)SixthInterimRevisionAnnouncement:61<61>MICROBIOLOGICALEXAMINATIONOFNONSTERILEPRODUCTS:MICROBIALENUMERA…
http://www.usppf.com/pf/pub/index.html 8/9
suitabilityoftheMPNMethod,thecalculatedvaluefromtheinoculummustbewithin95%confidence
limitsoftheresultsobtainedwiththecontrol.
Iftheabovecriteriacannotbemetforoneofmoreoftheorganismstestedwithanyofthedescribed
methods,themethodandtestconditionsthatcomeclosesttothecriteriaareusedtotesttheproduct.
TESTINGOFP RODUCTS
AmountUsedfortheTest
Unlessotherwisedirected,use10gor10mLoftheproducttobeexaminedtakenwiththe
precautionsreferredtoabove.Forfluidsorsolidsinaerosolform,sample10containers.For
transdermalpatches,sample10patches.
Theamounttobetestedmaybereducedforactivesubstancesthatwillbeformulatedinthefollowing
conditions:theamountperdosageunit(e.g.,tablet,capsule,injection)islessthanorequalto1mg,or
theamountpergormL(forpreparationsnotpresentedindoseunits)islessthan1mg.Inthesecases,
theamountofsampletobetestedisnotlessthantheamountpresentin10dosageunitsor10gor10
mLoftheproduct.
Formaterialsusedasactivesubstanceswherethesamplequantityislimitedorbatchsizeis
extremelysmall(i.e.,lessthan1000mLor1000g),theamounttestedshallbe1%ofthebatchunless
alesseramountisprescribedorjustifiedandauthorized.
Forproductswherethetotalnumberofentitiesinabatchislessthan200(e.g.,samplesusedin
clinicaltrials),thesamplesizemaybereducedtotwounits,oroneunitifthesizeislessthan100.
Selectthesample(s)atrandomfromthebulkmaterialorfromtheavailablecontainersofthe
preparation.Toobtaintherequiredquantity,mixthecontentsofasufficientnumberofcontainersto
providethesample.
ExaminationoftheProduct
MEMBRANEFILTRATION
Useafiltrationapparatusdesignedtoallowthetransferofthefiltertothemedium.Preparethe
sampleusingamethodthathasbeenshowntobesuitableasdescribedinGrowthPromotionTestand
SuitabilityoftheCountingMethod,transfertheappropriateamounttoeachoftwomembranefilters,
andfilterimmediately.Washeachfilterfollowingtheprocedureshowntobesuitable.
ForthedeterminationofTAMC,transferoneofthemembranefilterstothesurfaceofSoybean–Casein
DigestAgar.ForthedeterminationofTYMC,transfertheothermembranetothesurfaceofSabouraud
DextroseAgar.IncubatetheplateofSoybean–CaseinDigestAgarat30 to35 for3to5daysandthe
plateofSabouraudDextroseAgarat20 to25 for5to7days.Calculatethenumberofcfupergorper
mLofproduct.
Whenexaminingtransdermalpatches,separatelyfilter10%ofthevolumeofthepreparation
describedforPrepa rationof theSamplethrougheachoftwosterilefiltermembranes.Transferone
membranetoSoybean–CaseinDigestAgarforTAMCandtheothermembranetoSabour audDex trose
AgarforTYMC.
PLATECOUNTMETHODS
PourPlateMethod—Preparethesampleusingamethodthathasbeenshowntobesuitableas
describedinGrowthPromotionTestandSuitabilityoftheCountingMethod.Prepareforeachmediumat

11/21/2016
34(6)SixthInterimRevisionAnnouncement:61<61>MICROBIOLOGICALEXAMINATIONOFNONSTERILEPRODUCTS:MICROBIALENUMERA…
http://www.usppf.com/pf/pub/index.html 9/9
leasttwoPetridishesforeachlevelofdilution.IncubatetheplatesofSoybean–CaseinDigestAgarat30
to35 for3to5daysandtheplatesofSabouraudDextroseAga rat20 to25 for5to7days.Select
theplatescorrespondingtoagivendilutionandshowingthehighestnumberofcolonieslessthan250
forTAMCand50forTYMC.Takethearithmeticmeanperculturemediumofthecounts,andcalculate
thenumberofcfupergorpermLofproduct.
Surface SpreadMethod—Preparethesampleusingamethodthathasbeenshowntobesuitableas
describedinGrowthPromotionTestandSuitabilityoftheCountingMethod.PrepareatleasttwoPetri
dishesforeachmediumandeachlevelofdilution.Forincubationandcalculationofthenumberofcfu,
proceedasdirectedforthePourPlateMethod.
MOSTPROBABLENUMBERMETHOD
Prepareanddilutethesampleusingamethodthathasbeenshowntobesuitableasdecribedin
GrowthP rom otionTestandSuitability oftheCountingMethod.Incubatealltubesfor3to5daysat30
to35 .Subcultureifnecessary,usingtheprocedureshowntobesuitable.Recordforeachlevelof
dilutionthenumberoftubesshowingmicrobialgrowth.Determinethemostprobablenumberof
microorganismspergormLoftheproducttobeexaminedfromTable3.
InterpretationoftheResults
Thetotalaerobicmicrobialcount(TAMC)isconsideredtobeequaltothenumberofcfufoundusing
Soybean–CaseinDigestAgar;ifcoloniesoffungiaredetectedonthismedium,theyarecountedaspart
ofTAMC.Thetotalcombinedyeastsandmoldscount(TYMC)isconsideredtobeequaltothenumberof
cfufoundusingSabouraudDextroseAgar;ifcoloniesofbacteriaaredetectedonthismedium,theyare
countedaspartofTYMC.WhentheTYMCisexpectedtoexceedtheacceptancecriterionduetothe
bacterialgrowth,SabouraudDextroseAga rcontainingantibioticsmaybeused.Ifthecountiscarried
outbytheMPNMethod,thecalculatedvalueisTAMC.
Whenanacceptancecriterionformicrobiologicalqualityisprescribed,itisinterpretedasfollows:
10
1
cfu:maximumacceptablecount=20;
10
2
cfu:maximumacceptablecount=200;
10
3
cfu:maximumacceptablecount=2000;
andsoforth.
TherecommendedsolutionsandmediaaredescribedinMicrobiologicalExaminationofNonsterile
Products:TestsforSpecifiedMicroorganisms 62 .
(EntireChapterandrevisionsmarkedforIRA—OfficialMay1,2009)
