
Contains Nonbinding Recommendations
Content of Premarket Submissions for
Device Software Functions
______________________________________________________________________________
Guidance for Industry and
Food and Drug Administration Staff
Document issued on June 14, 2023.
The draft of this document was issued on November 4, 2021.
This document supersedes Guidance for the Content of Premarket
Submissions for Software Contained in Medical Devices, May 2005.
For questions about this document regarding CDRH-regulated devices, contact the Digital Health
Center of Excellence at di[email protected]. For questions about this document regarding
CBER regulated devices, contact the Office of Communication, Outreach, and Development
(OCOD) at 1-800-835-4709 or 240-402-8010, or by email at [email protected].
U.S. Department of Health and Human Services
Food and Drug Administration
Center for Devices and Radiological Health
Center for Biologics Evaluation and Research
Center for Drug Evaluation and Research
Office of Combination Products in the Office of the
Commissioner

Contains Nonbinding Recommendations
Preface
Public Comment
You may submit electronic comments and suggestions at any time for Agency consideration to
https://www.regulations.gov. Submit written comments to the Dockets Management Staff, Food
and Drug Administration, 5630 Fishers Lane, Room 1061, (HFA-305), Rockville, MD 20852.
Identify all comments with the docket number FDA-2021-D-0775. Comments may not be acted
upon by the Agency until the document is next revised or updated.
Additional Copies
CDRH
Additional copies are available from the Internet. You may also send an email request to CDRH-
GUI00000337 and complete title of the guidance in the request.
CBER
Additional copies are available from the Center for Biologics Evaluation and Research (CBER),
Office of Communication, Outreach, and Development (OCOD), 10903 New Hampshire Ave.,
Bldg. 71, Room 3128, Silver Spring, MD 20993-0002, or by calling 1-800-835-4709 or 240-402-
8010, by email, [email protected] or from the Internet at
https://www.fda.gov/vaccines-blood-biologics/guidance-compliance-regulatory-information-
biologics/biologics-guidances.
CDER
Additional copies are available from the Center for Drug Evaluation and Research (CDER),
Office of Communication, Division of Drug Information, 10001 New Hampshire Ave.,
Hillandale Bldg., 4th Floor, Silver Spring, MD 20993-0002, or by calling 855-543-3784 or 301-
796-3400, or by email, [email protected], or from the Internet at
https://www.fda.gov/drugs/guidance-compliance-regulatory-information/guidances-drugs.
Contains Nonbinding Recommendations
Table of Contents
I. Introduction ............................................................................................................................. 1
II. Background ............................................................................................................................. 2
III. Scope ....................................................................................................................................... 4
IV. Definitions............................................................................................................................... 6
V. Documentation Level .............................................................................................................. 8
VI. Recommended Documentation ............................................................................................... 9
Documentation Level Evaluation ................................................................................... 12
Software Description ...................................................................................................... 12
Risk Management File ................................................................................................... 15
Software Requirements Specification (SRS) ................................................................. 18
System and Software Architecture Diagram .................................................................. 20
Software Design Specification (SDS) ............................................................................ 22
(1) Basic Documentation Level ........................................................................................ 23
(2) Enhanced Documentation Level ................................................................................. 23
Software Development, Configuration Management, and Maintenance Practices ........ 23
(1) Basic Documentation Level ........................................................................................ 24
(2) Enhanced Documentation Level ................................................................................. 24
Software Testing as part of Verification and Validation................................................ 25
(1) Basic Documentation Level ........................................................................................ 25
(2) Enhanced Documentation Level ................................................................................. 26
Software Version History ............................................................................................... 26
Unresolved Software Anomalies .................................................................................... 27
VII. Additional Information - Regulatory Considerations for Software Functions ..................... 28
Appendix A: Documentation Level Examples ............................................................................. 28
Appendix B: System and Software Architecture Diagram Examples .......................................... 39

Contains Nonbinding Recommendations
1
Content of Premarket Submissions for
Device Software Functions
______________________________________________________________________________
Guidance for Industry and
Food and Drug Administration Staff
This guidance represents the current thinking of the Food and Drug Administration (FDA or
Agency) on this topic. It does not establish any rights for any person and is not binding on
FDA or the public. You can use an alternative approach if it satisfies the requirements of the
applicable statutes and regulations. To discuss an alternative approach, contact the FDA staff
or Office responsible for this guidance as listed on the title page.
I. Introduction
This guidance document is intended to provide information regarding the recommended
documentation for premarket submissions for FDA’s evaluation of the safety and effectiveness
of device software functions, which are software functions that meet the definition of a device
under section 201(h) of the Federal Food, Drug, and Cosmetic Act (FD&C Act).
1
This document
replaces FDA’s Guidance for the Content of Premarket Submissions for Software Contained in
Medical Devices issued on May 11, 2005, and updates FDA’s thinking related to the
documentation FDA recommends sponsors include for the review of device software functions in
premarket submissions.
The recommendations in this guidance are intended to facilitate FDA’s premarket review. This
guidance describes information that would be typically generated and documented
2
during
software development, verification, and validation. The least burdensome approach was applied
to identify the minimum amount of information that, based on our experience, would generally
be needed to support a premarket submission for a device that uses software. During premarket
review, FDA may request additional information that is needed to evaluate the submission. For
example, in order to demonstrate a reasonable assurance of safety and effectiveness for devices
1
The term “device” is defined in 201(h)(1) of the Federal Food, Drug, and Cosmetic (FD&C) Act to include an
“instrument, apparatus, implement, machine, contrivance, implant, in vitro reagent, or other similar or related article,
including any component, part, or accessory, which is ...intended for use in the diagnosis of disease or other
conditions, or in the cure, mitigation, treatment, or prevention of disease, in man ... or intended to affect the structure
or any function of the body of man...” and “does not include software functions excluded pursuant to section 520(o)”
of the FD&C Act.
2
As a reminder, manufacturers of device software must create and maintain software-related documentation in
accordance with the requirements of the Quality System (QS) Regulation (21 CFR 820.30 Subpart C – Design
Controls of the Quality System Regulation).

Contains Nonbinding Recommendations
2
that use software, documentation related to the requirements of the Quality System Regulation
(QSR) (21 CFR Part 820) is often a necessary part of the premarket submission. As part of QSR
design controls, a manufacturer must “establish and maintain procedures for validating the
device design,” which “shall include software validation and risk analysis, where appropriate”
(21 CFR 820.30(g)).
The documentation recommended in this guidance is based on FDA’s experience evaluating the
safety and effectiveness of device software. However, sponsors may use alternative approaches
and provide different documentation so long as their approach and documentation satisfy
premarket submission requirements in applicable statutory provisions and regulations. For the
current edition(s) of the FDA-recognized consensus standard(s) referenced in this document, see
the FDA Recognized Consensus Standards Database.
3
For more information regarding use of
consensus standards in regulatory submissions, please refer to the FDA guidance titled
Appropriate Use of Voluntary Consensus Standards in Premarket Submissions for Medical
Devices
4
and Standards Development and the Use of Standards in Regulatory Submissions
Reviewed in the Center for Biologics Evaluation and Research.
5
As stated above, this guidance identifies the software information FDA considers to generally be
necessary to support a premarket submission. The recommendations in this guidance are also
intended to facilitate FDA’s premarket review. FDA anticipates that the Agency and industry
will need up to 60 days after the publication of this guidance to operationalize the
recommendations discussed. However, CDRH intends to review any such information if
submitted at any time.
In general, FDA’s guidance documents do not establish legally enforceable responsibilities.
Instead, guidances describe the Agency’s current thinking on a topic and should be viewed only
as recommendations, unless specific regulatory or statutory requirements are cited. The use of
the word should in Agency guidances means that something is suggested or recommended, but
not required.
II. Background
The purpose of this guidance is to describe FDA’s thinking on the recommended documentation
sponsors should include in premarket submissions for FDA’s evaluation of the safety and
effectiveness of device software functions. This thinking recognizes recent changes to the FD&C
Act made by the 21st Century Cures Act (Cures Act), which amended section 520 of the FD&C
Act and excludes certain software functions from the device definition. It also considers the
rapidly evolving nature of digital health and recent FDA-recognized consensus standards related
to software. This guidance, as described in Section III (Scope), is intended to complement other
existing guidance documents that provide recommendations related to software, including the
3
Available at https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfStandards/search.cfm.
4
Available at https://www.fda.gov/regulatory-information/search-fda-guidance-documents/appropriate-use-
voluntary-consensus-standards-premarket-submissions-medical-devices.
5
Available at https://www.fda.gov/regulatory-information/search-fda-guidance-documents/standards-development-
and-use-standards-regulatory-submissions-reviewed-center-biologics-evaluation.

Contains Nonbinding Recommendations
3
guidance documents listed below. The following guidance documents represent a subset of FDA
guidances with digital health content
6
relevant to premarket software documentation activities.
Please note the list is not exhaustive and is subject to change:
· Multiple Function Device Products: Policy and Considerations
7
· Off-The-Shelf Software Use in Medical Devices
8
· Design Considerations and Premarket Submission Recommendations for Interoperable
Medical Devices
9
· General Principles of Software Validation
10
· Content of Premarket Submissions for Management of Cybersecurity in Medical
Devices
11
· Cybersecurity for Networked Medical Devices Containing Off-The-Shelf (OTS)
Software
12
· Applying Human Factors and Usability Engineering to Medical Devices
13
FDA encourages the consideration of these guidances when developing device software
functions and preparing premarket software documentation.
The emergence of consensus standards related to software has helped to improve the consistency
and quality of software development and documentation, particularly with respect to activities
such as risk assessment and management. When possible, FDA harmonized the terminology and
recommendations in this guidance with software-related consensus standards, such as the
following examples. The following standards are not intended to represent an exhaustive list and
are subject to change:
14
· ANSI/AAMI/ISO 14971: Medical devices - Applications of risk management to medical
devices
· ANSI/AAMI/IEC 62304: Medical Device Software - Software Life Cycle Processes
· ANSI/AAMI SW91: Classification of defects in health software
6
Available at https://www.fda.gov/medical-devices/digital-health-center-excellence/guidances-digital-health-
content.
7
Available at https://www.fda.gov/regulatory-information/search-fda-guidance-documents/multiple-function-
device-products-policy-and-considerations.
8
Available at https://www.fda.gov/regulatory-information/search-fda-guidance-documents/shelf-software-use-
medical-devices.
9
Available at https://www.fda.gov/regulatory-information/search-fda-guidance-documents/design-considerations-
and-premarket-submission-recommendations-interoperable-medical-devices.
10
Available at https://www.fda.gov/regulatory-information/search-fda-guidance-documents/general-principles-
software-validation.
11
Available at https://www.fda.gov/regulatory-information/search-fda-guidance-documents/content-premarket-
submissions-management-cybersecurity-medical-devices.
12
Available at https://www.fda.gov/regulatory-information/search-fda-guidance-documents/cybersecurity-
networked-medical-devices-containing-shelf-ots-software.
13
Available at https://www.fda.gov/regulatory-information/search-fda-guidance-documents/applying-human-
factors-and-usability-engineering-medical-devices.
14
The most up-to-date list of voluntary FDA-recognized consensus standards is available at
https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfStandards/search.cfm.

Contains Nonbinding Recommendations
4
The Agency encourages the consideration of these FDA-recognized consensus standards when
developing device software functions and preparing premarket software documentation. When
assessing the appropriate Documentation Level for the device and the overall recommended
documentation for inclusion in a premarket submission, please refer to Section V
(Documentation Level) and Section VI (Recommended Documentation) of this guidance.
Section 3308 of the Food and Drug Omnibus Reform Act of 2022, Title III of Division FF of the
Consolidated Appropriations Act, 2023, Pub. L. No. 117-328 (“FDORA”), enacted on December
29, 2022, added section 515C “Predetermined Change Control Plans for Devices” to the FD&C
Act (section 515C). Under section 515C, FDA can approve or clear a predetermined change
control plan (PCCP) for a device that describes planned changes that may be made to the device
and that would otherwise require a supplemental premarket approval application or premarket
notification. For example, section 515C provides that a supplemental premarket approval
application (section 515C(a)) or a premarket notification (section 515C(b)) is not required for a
change to a device if the change is consistent with a PCCP that is approved or cleared by FDA.
Section 515C also provides that FDA may require that a PCCP include labeling for safe and
effective use of a device as such device changes pursuant to such plan, notification requirements
if the device does not function as intended pursuant to such plan, and performance requirements
for changes made under the plan. If you are interested in proposing a PCCP in your marketing
submission, we encourage you to submit a Pre-Submission to engage in further discussion with
CDRH. See FDA’s guidance “Requests for Feedback and Meetings for Medical Device
Submissions: The Q-Submission Program” available at https://www.fda.gov/regulatory-
information/search-fda-guidance-documents/requests-feedback-and-meetings-medical-device-
submissions-q-submission-program.
III. Scope
For the purposes of this document, FDA refers to a software function that meets the definition of
a device as a device software function. For any given product, the term “function” is a distinct
purpose of the product, which could be the intended use or a subset of the intended use of the
product.
15
For example, a product with an intended use to analyze data has one function:
analysis. A product with an intended use to store, transfer, and analyze data has three functions:
(1) storage, (2) transfer, and (3) analysis. As this example illustrates, a product may contain
multiple functions.
This guidance is intended to cover device software functions. Examples include, but are not
limited to, firmware and other means for software-based control of medical devices, software
accessories to medical devices, and software only
16
function(s) that meet the definition of a
device.
15
For details, see “Multiple Function Device Products: Policy and Considerations,” available at
https://www.fda.gov/regulatory-information/search-fda-guidance-documents/multiple-function-device-products-
policy-and-considerations.
16
“Software only” functions include device software functions intended to be operated on commercial OTS
computing platforms.

Contains Nonbinding Recommendations
5
This guidance recommends the information to provide in a premarket submission that includes a
device software function(s). For the purposes of this guidance, the term premarket submission
includes, but is not limited to, premarket notification (510(k)) submission, De Novo
classification request, Premarket Approval (PMA) application, Investigational Device Exemption
(IDE), Humanitarian Device Exemption (HDE), or Biologics License Application (BLA).
Certain devices are subject to premarket review through a BLA under section 351 of the Public
Health Service Act.
This guidance does not apply to automated manufacturing and Quality System software
17
or
software that is not a device. For further information or to clarify the documentation
expectations, please contact the responsible FDA review division.
Generally, the recommendations in this guidance apply to the device constituent part of a
combination product
18
(such as drug-device and biologic-device combination products) when the
device constituent part
19
includes a device software function, including combination products
assigned to CDER and CBER regulated under drug or biological product market submission
types. For more information, contact the FDA review Division that will have the lead review for
the combination product.
20
Other FDA guidance documents may recommend additional software-related documentation for
inclusion in a premarket submission. For example, recommendations regarding cybersecurity
information to include in a device premarket submission can be found in the guidances “Content
of Premarket Submissions for Management of Cybersecurity in Medical Devices” and
“Cybersecurity for Networked Medical Devices Containing Off-The-Shelf (OTS) Software.”
21
Section II (Background) references other relevant guidance documents that supplement the
recommendations contained in this guidance.
17
As part of Quality System Regulation production and process controls, 21 CFR 820.70(i) states, “When computers
or automated data processing systems are used as part of production or the quality system, the manufacturer shall
validate computer software for its intended use according to an established protocol. All software changes shall be
validated before approval and issuance. These validation activities and results shall be documented.”
18
21 CFR 3.2(e).
19
21 CFR 4.2.
20
Sponsors may request in writing the participation of representatives of the Office of Combination Products (OCP)
in meetings regarding their combination products, or to have OCP otherwise engage on regulatory matters
concerning the product (section 503(g)(1)(A) of the FD&C Act). In addition, if you are uncertain whether your
product is a combination product or a constituent part of a combination product, or which center has primary
jurisdiction, you may request engagement by the Office of Combination Products. For more information on
requesting engagement on regulation of combination products, please see FDA’s guidance “Requesting FDA
Feedback on Combination Products,” available at https://www.fda.gov/regulatory-information/search-fda-guidance-
documents/requesting-fda-feedback-combination-products.
21
For example, as part of the software validation and risk analysis required by 21 CFR 820.30(g), software device
manufacturers may need to establish a cybersecurity risk management process that encompasses the total product
lifecycle in order to address cybersecurity risks and emerging vulnerabilities.

Contains Nonbinding Recommendations
6
Device software functions subject to specific special controls
22
may require additional software-
related documentation in a premarket submission. As applicable, please refer to the relevant
special controls for the device.
This guidance does not apply to the software-related documentation that may be needed to
evaluate postmarket software device issues, including corrections and removals.
23
While this guidance identifies the documentation sponsors should include in premarket
submissions, this guidance is not meant to provide recommendations regarding how device
software should be developed, verified, and validated. This guidance does not recommend the
use of any specific software life cycle model or development methodology (such as waterfall
model or other variations thereof, spiral model, Agile model, etc.). Sponsors should establish a
software life cycle model that is appropriate for their product and organization, and meets the
applicable regulatory requirements. The software life cycle model that is selected should cover
the software throughout its total product life cycle. Regardless of the software lifecycle model
being utilized, sponsors should ensure that the establishment of their design history file (DHF)
24
documentation is synchronized with their software development, verification, and validation
efforts. DHF documentation that is created retrospectively or following a prolonged period of
time after actual software development, verification, and validation efforts could raise concerns
regarding whether a developer has adequate control of their design process. For
recommendations on device software development, verification, and validation, please consult
software-related FDA-recognized voluntary consensus standards and other software-related FDA
guidance documents referenced in this guidance (e.g., “General Principles of Software
Validation”
25
).
IV. Definitions
The following terms are used for the purposes of this guidance:
Device Software Function - Software function that meets the device definition in section 201(h)
of the FD&C Act. As discussed above, the term “function” is a distinct purpose of the product,
which could be the intended use or a subset of the intended use of the product.
22
For more information regarding special controls, please see FDA’s Regulatory Controls webpage, available at
https://www.fda.gov/medical-devices/overview-device-regulation/regulatory-controls.
23
See 21 CFR Part 806.
24
Each manufacturer shall establish and maintain a DHF for each type of device. 21 CFR 820.30(j). A DHF is a
compilation of records which describes the design history of a finished device. The DHF shall contain or reference
the records necessary to demonstrate that the design was developed in accordance with the approved design plan and
the requirements of 21 CFR 820. See 21 CFR 820.30(j).
25
Available at https://www.fda.gov/regulatory-information/search-fda-guidance-documents/general-principles-
software-validation.

Contains Nonbinding Recommendations
7
Off-the-Shelf (OTS) Software
26
- A generally available software component used by a device
manufacturer for which the manufacturer cannot claim complete software life cycle control (e.g.,
operating system, printer/display libraries).
Serious Injury
27
- An injury or illness that:
1) Is life threatening,
2) Results in permanent impairment of a body function or permanent damage to a
body structure, or
3) Necessitates medical or surgical intervention to preclude permanent impairment
of a body function or permanent damage to a body structure. Permanent is defined
as irreversible impairment or damage to a body structure or function, excluding
trivial impairment or damage.
Software Verification and Software Validation - This guidance uses the terms “software
verification” and “software validation,” which are described in further detail below.
· For the purposes of this guidance, software verification is confirmation by objective
evidence that the output of a particular phase of development meets all the input
requirements for that phase. Software verification involves evaluating the consistency,
completeness, and correctness of the software and its supporting documentation, as it is
being developed, and provides support for a subsequent conclusion that software is
validated. Software testing is one of several verification activities intended to confirm
that the software development output meets its input requirements. Other verification
activities include source code evaluations (e.g., code inspections and walkthroughs),
document inspections, design reviews, technical evaluations (e.g., software architecture,
software detailed design, etc.) and traceability analyses (e.g., software requirements
specification to software design specification (and vice versa), source code to software
design specification (and vice versa), and test cases to source code and to software design
specification). For example, the input and output of the design phase are known as
Software Requirements Specification (SRS) and Software Design Specification (SDS),
respectively. In this case, software verification would involve confirming by objective
evidence (e.g., reviews, traceability analysis) that the software design as documented in
the SDS (i.e., output) correctly and completely implements all the requirements of the
SRS (i.e., input).
· For the purposes of this guidance, software validation refers to establishing, by objective
evidence, that the software specifications conform to user needs and intended uses, and
that the particular requirements implemented through software can be consistently
fulfilled. Software validation is a part of design validation of the finished device. It
involves checking for proper operation of the software in its actual or simulated use
environment, including integration into the final device where appropriate. Software
validation is highly dependent upon comprehensive software testing and other
26
Available at https://www.fda.gov/regulatory-information/search-fda-guidance-documents/shelf-software-use-
medical-devices.
27
Serious injury as defined in 21 CFR 803.3(w).

Contains Nonbinding Recommendations
8
verification tasks previously completed at each stage of the software development life
cycle. Planning, requirements, traceability, testing, risk assessment, design reviews,
change management, and many other aspects of good software engineering are important
activities that together help to support a conclusion that software is validated.
The above descriptions of software verification and software validation are consistent with
FDA’s thinking as described in the guidance “General Principles of Software Validation.”
V. Documentation Level
The recommended documentation for a premarket submission depends on the device’s risk to a
patient, a user of a device, or others in the environment of use. FDA intends to take a risk-based
approach to help determine the device’s Documentation Level, which is either Basic or
Enhanced. The purpose of the Documentation Level is to help identify the minimum amount of
information that would support a premarket submission that includes device software functions.
The Documentation Level of a device is based on the risks of its device software function(s) in
the context of the device’s intended use,
28
such that the documentation level reflects the device
as a whole.
For the purpose of this guidance:
Enhanced Documentation should be provided for any premarket submission that
includes device software function(s) where a failure or flaw of any device software
function(s) could present a hazardous situation with a probable
29
risk of death or serious
injury,
30
either to a patient, user of the device, or others in the environment of use. These
risks should be assessed prior to implementation of risk control measures. Sponsors
should consider the risks in the context of the device’s intended use (e.g., impacts to
safety, treatment, and/or diagnosis), and other relevant considerations.
Basic Documentation should be provided for any premarket submission that includes
device software function(s) where Enhanced Documentation does not apply.
When determining the Documentation Level, sponsors should consider all known or foreseeable
software hazards and hazardous situations associated with the device, including those resulting
from reasonably foreseeable misuse, whether intentional or unintentional, prior to the
implementation of risk control measures. This also includes the likelihood that device
functionality is intentionally or unintentionally compromised by inadequate device cybersecurity.
28
See 21 CFR 801.4 (“…[I]ntended uses…refer to the objective intent of the persons legally responsible for the
labeling of an article. The intent may be shown by such persons’ expressions, the design or composition of the
article, or by the circumstances surrounding the distribution of the article.”).
29
The term “probable” is intended to exclude the consideration of purely hypothetical risks.
30
See 21 CFR 803.3(w).

Contains Nonbinding Recommendations
9
It is the sponsor’s responsibility to proactively and comprehensively consider risks as part of the
device’s risk assessment.
31
While devices within the scope of this guidance should be individually assessed to determine the
appropriate Documentation levels, there are certain categories of devices for which we
recommend that Enhanced Documentation be provided in a premarket submission. Specifically,
we recommend that Enhanced Documentation should be provided in a premarket submission for
devices intended to test blood donations for transfusion-transmitted infections, devices used to
determine blood donor and recipient compatibility, automated blood cell separator devices
intended for collection of blood and blood components for transfusion or further manufacturing
use, and blood establishment computer software (BECS).
32
There are other categories of device for which we generally recommend Enhanced
Documentation be provided in a premarket submission. FDA believes that given the nature of
these products and their intended uses, these products may have unique risks that require further
documentation to ensure that FDA is able to evaluate the safety and effectiveness of the device.
These include devices that are a constituent part of a combination product (i.e., drug/device,
biologic/device, or drug/device/biologic)
33
and Class III devices. In the course of evaluating the
appropriate Documentation Level for a device that is a constituent part of a combination product
or a Class III device, a sponsor may determine that an Enhanced Documentation level does not
apply. In such cases, the sponsor should provide an appropriately detailed rationale as to why
Basic Documentation instead of Enhanced Documentation is appropriate for the premarket
submission. As previously discussed, during the course of submission review, FDA may request
additional information if needed to evaluate the safety and effectiveness of the device.
Sponsors may submit a Pre-Submission to obtain FDA feedback about a device’s Documentation
Level and recommended documentation prior to a premarket submission, as described in the
guidance “Requests for Feedback and Meetings for Medical Device Submissions: The Q-
Submission Program.”
34
For additional information on how to engage with a particular Center
with regard to a combination product, including best practices for doing so, please see the final
guidance “Requesting FDA Feedback on Combination Products.”
35
For additional information and examples of devices that demonstrate the implementation of the
Documentation Level risk-based approach, please refer to Appendix A of this guidance.
VI. Recommended Documentation
This section reflects FDA’s recommendations for information to be included in premarket
submissions for Basic and Enhanced Documentation Levels. This recommended information
31
For information on risk assessment refer to Section VI.C of this guidance.
32
See Appendix A for rationale on enhanced documentation level for BECS.
33
21 CFR 3.2(e).
34
Available at https://www.fda.gov/regulatory-information/search-fda-guidance-documents/requests-feedback-and-
meetings-medical-device-submissions-q-submission-program.
35
Available at https://www.fda.gov/regulatory-information/search-fda-guidance-documents/requesting-fda-
feedback-combination-products.

Contains Nonbinding Recommendations
10
should demonstrate that planning, requirements, risk assessment, design reviews, traceability,
change management, testing plans and results, and other aspects of good software engineering
for device software functions were employed, to support a conclusion that the device software
function was appropriately designed, verified, and validated.
If the device is a multiple function device product and includes software function(s) that are
considered “other functions,” as that term is used in the guidance “Multiple Function Device
Product: Policy and Considerations,” the recommendations described in the aforementioned
guidance should be considered when preparing the software documentation for a premarket
submission.
Table 1 below provides an outline of the recommended documentation for each software
documentation element and corresponding Documentation Level. Please refer to subsections A-J
in this section of the guidance (VI) for more detail.
Table 1. Outline of Recommended Documentation
Software
Documentation
Elements
Basic Documentation Level
Enhanced Documentation Level
Documentation
Level Evaluation
(Section VI.A)
A statement indicating the Documentation Level and a description of the
rationale for that level.
Software
Description
(Section VI.B)
Software description, including overview of significant software features,
functions, analyses, inputs, outputs, and hardware platforms.
Risk
Management File
(Section VI.C)
Risk management plan, risk assessment demonstrating that risks have been
appropriately mitigated, and risk management report.
Software
Requirements
Specification
(SRS)
(Section VI.D)
SRS documentation, describing the needs or expectations for a system or
software, presented in an organized format, at the software system level or
subsystem level, as appropriate, and with sufficient information to
understand the traceability of the information with respect to the other
software documentation elements (e.g., risk management file, software
design specification, system and software architecture design chart,
software testing).
Contains Nonbinding Recommendations
11
Software
Documentation
Elements
Basic Documentation Level
Enhanced Documentation Level
System and
Software
Architecture
Design
(Section VI.E)
Detailed diagrams of the modules, layers, and interfaces that comprise the
device, their relationships, the data inputs/outputs and flow of data, and
how users or external products (including information technology (IT)
infrastructure and peripherals) interact with the system and software.
Software Design
Specification
(SDS)
(Section VI.F)
FDA is not recommending the
SDS as part of the premarket
submission. Sponsor should
document this information on
the design via the DHF for the
device. During premarket
review, FDA may request
additional information, if
needed, to evaluate the safety
and effectiveness of the device.
SDS documentation, including sufficient
information that would allow FDA to
understand the technical design details
of how the software functions, how the
software design completely and
correctly implements all the
requirements of the SRS, and how the
software design traces to the SRS in
terms of intended use, functionality,
safety, and effectiveness.
Software
Development,
Configuration
Management,
and Maintenance
Practices
(Section VI.G)
A summary of the life cycle
development plan and a
summary of configuration
management and maintenance
activities;
OR
A Declaration of Conformity
36
to the FDA-recognized version
of IEC 62304, including
subclauses 5.1.1-5.1.3, 5.1.6-
5.1.9, clause 6 (Software
maintenance process), and
clause 8 (Software configuration
management process), among
others as applicable.
Basic Documentation Level, PLUS
complete configuration management and
maintenance plan document(s);
OR
A Declaration of Conformity
37
to the
FDA-recognized version of IEC 62304,
including subclause 5.1 (Software
development planning), clause 6
(software maintenance process), and
clause 8 (software configuration
management process), among others as
applicable.
36
For more information, please see Appropriate Use of Voluntary Consensus Standards in Premarket Submissions
for Medical Devices, available at https://www.fda.gov/regulatory-information/search-fda-guidance-
documents/appropriate-use-voluntary-consensus-standards-premarket-submissions-medical-devices.
37
For more information, please see Appropriate Use of Voluntary Consensus Standards in Premarket Submissions
for Medical Devices, available at https://www.fda.gov/regulatory-information/search-fda-guidance-
documents/appropriate-use-voluntary-consensus-standards-premarket-submissions-medical-devices.

Contains Nonbinding Recommendations
12
Software
Documentation
Elements
Basic Documentation Level
Enhanced Documentation Level
Software Testing
as Part of
Verification and
Validation
(Section VI.H)
A summary description of the
testing activities at the unit,
integration and system levels;
AND
System level test protocol
including expected results,
observed results, pass/fail
determination, and system level
test report.
Basic Documentation Level, PLUS
unit and integration level test protocols
including expected results, observed
results, pass/fail determination, and unit
and integration level test reports.
Software Version
History
(Section VI.I)
A history of tested software versions including the date, version number,
and a brief description of all changes relative to the previously tested
software version.
Unresolved
Software
Anomalies
(Section VI.J)
List of remaining unresolved software anomalies with an evaluation of the
impact of each unresolved software anomaly on the device’s safety and
effectiveness.
Documentation Level Evaluation
A statement indicating the Documentation Level for the device and a description of the rationale
for such Documentation Level. The rationale should account for the device’s intended use, and
include references, where appropriate, from the submission documentation (such as Risk
Management File, Software Description, etc.) to support the indicated Documentation Level.
Software Description
An overview of significant software features and functions, including images, flow charts, and
state diagrams as needed to adequately explain the software functionality
38
should be provided. If
the premarket submission is for a modified device, provide the document number of the previous
submission and highlight pertinent software changes (e.g., changes that affect safety and
effectiveness) since the last FDA approval or clearance.
38
FDA may request additional architecture diagrams to address the cybersecurity risks associated with a device. For
more information, please refer to the guidance document, “Content of Premarket Submissions for Management of
Cybersecurity in Medical Devices.”

Contains Nonbinding Recommendations
13
Consider and, as applicable, provide information to address the questions below when preparing
the software description. However, FDA recognizes that these questions and examples may not
capture all the unique aspects of device software and encourages the inclusion of additional
information that will further FDA’s understanding of the device’s functionality to facilitate the
review of a submission.
If the device is a multiple function device product and includes software function(s) that are
considered “other functions,” as that term is used in the guidance “Multiple Function Device
Products: Policy and Considerations,” the recommendations described in the aforementioned
guidance should be considered when preparing the software description information.
· Software Operation
o What is the role of the software as it relates to the intended use of the device?
Examples: software-only device, software that controls the device hardware,
software application that accompanies the device for data processing.
o Who is the intended user of the software? Examples: the patient, a caregiver, a
healthcare professional, or a combination thereof
o What is the intended patient population?
§ Does the software function focus on a specific disease, condition, patient
characteristic or demographic?
§ Does the software provide information that is directly applicable to a
specific disease or condition?
o If the software performs analysis of signals/patterns or images, what is the
analysis methodology? Examples: rule-based calculations, online test
administration, artificial intelligence and machine learning (AI/ML), neural
networks, fixed (locked) or adaptive (iterative, continuously learning) algorithms.
o If the device software function uses ML models trained through ML methods (i.e.,
is AI/ML-enabled, such as AI, ML, adaptive models, natural language processing
(NLP), neural networks, and related approaches):
§ What methods, models, frameworks, and/or platforms were used?
§ What data (populations, samples) informed the model(s)? How, when, and
where was the data collected?
§ What steps were taken to identify and address potential biases and
limitations of the model(s)?
§ What materials, mechanisms, and/or approaches are used to provide
transparency about the model’s development, performance, and
limitation(s)?

Contains Nonbinding Recommendations
14
· Software Specifics
39
o What hardware platforms are used?
o What software platforms are used?
§ If applicable, what hosting environments are used (e.g., hospital networks,
cloud infrastructures) and for what functions (e.g., processing, storage)?
o Does the device use OTS software?
40
o What is the final release version (i.e., version intended to be released to end
users)? If this version is different from the documentation’s version, explain the
differences.
· Software Inputs and Outputs
o What are the inputs and their format? Examples: signals, images (specify
modality), measurements (specify units), reports, questionnaires, other device
data/results.
o Who or what provides the inputs? Examples: patients, caregivers, healthcare
professionals, technicians, sensors/attachments, signal acquisition systems, in
vitro diagnostic devices, other medical devices, other non-medical products or
software.
o What are the outputs and their format? Examples: diagnostic information,
treatment information, control signals for device hardware, images (specify
modality), measurements (specify units), alarms, alerts, or reports.
o Who or what receives the outputs? Examples: patients, caregivers, healthcare
professionals, technicians, health records, device hardware, other medical
devices, interoperable systems.
o Does the software impact or replace any action(s) otherwise performed manually
by a health care professional, patient, caregiver, or other operator? What are the
clinical workflow steps and assumptions (from beginning to end state)?
Examples: automates steps, triages patients, provides a definite diagnosis or
suggests likely diagnosis for further confirmation by physician, performs or
recommends specific treatment, identifies a region of interest for further review.
39
One example approach for providing some of the identified software specifics is by providing a Software Bill of
Materials (SBOM) that lists and provide details on software components including, but not limited to, commercial,
open source, OTS, and manufacturer-developed software components. For some devices, information related to an
SBOM may be required part of a premarket submission.
40
If a device uses OTS software, FDA may request additional information in premarket submissions. For more
information, please refer to the guidance document “Guidance for Off-The-Shelf Software Use in Medical Devices.”

Contains Nonbinding Recommendations
15
o Is the device designed to be interoperable?
41
In other words, does the device
transmit, exchange, and/or use information through an electronic interface (e.g.,
network, wireless) with another medical/non-medical product, system, or device?
If yes, what other products does the device interface with, and what methods,
standards, and specifications are used to interact and/or communicate with other
medical/non-medical product, system, or device? Are the medical/non-medical
products, systems, or devices networked?
If any of the information requested above is included in another document, such as the Software
Requirements Specification (SRS), an annotation and a reference to the document in the
submission where this information is located should be provided.
Risk Management File
The risk management file should be provided as part of the premarket submission and include
the following documentation. FDA recommends sponsors refer to an FDA-recognized version of
ISO 14971 for additional information on the development and application of a risk management
file.
(1) Risk Management Plan
FDA recommends sponsors submit a risk management plan to support the effectiveness of the
risk management activities and processes for a particular medical device.
42
In FDA’s review of
the risk management plan, the Agency intends to primarily focus on:
· Individual risk acceptability criteria including the need for risk reduction (control).
· Method to evaluate the acceptability of the overall residual risk for all residual risks after
all risk control measures have been implemented and verified.
The risk acceptability criteria should be based on the sponsor’s process for determining
acceptable risk. The acceptability criteria should be documented in the risk management plan
before an initial risk evaluation is performed for the device software under review.
It should be clear in the risk management plan how the sponsor plans to evaluate the overall
residual risk against the benefits of the intended use of the device.
41
More information on interoperable medical devices is available at: https://www.fda.gov/regulatory-
information/search-fda-guidance-documents/design-considerations-and-pre-market-submission-recommendations-
interoperable-medical-devices.
42
For combination products that include device software functions, a 14971-based risk management framework that
incorporates relevant considerations from International Council for Harmonisation of Technical Requirements for
Pharmaceuticals for Human Use (ICH) Q9 Quality Risk Management is recommended. For further information, see,
for example, AAMI TIR 105 Risk Management Guidance for Combination Products.

Contains Nonbinding Recommendations
16
(2) Risk Assessment
A risk assessment that includes a risk analysis, risk evaluation, risk control and a benefit-risk
analysis (where applicable) should be provided for all device software. For software that is part
of a system, a risk assessment should be performed on the system comprising the software and
its whole hardware environment. If this information is covered in the system risk assessment
documentation, this should be noted in the software documentation with reference to the
particular section of the premarket submission.
For multiple function device products, the risk assessment should include the results of a risk-
based analysis of any potential adverse impact or labeled positive impact of “other function(s),”
as that term is used in the guidance “Multiple Function Device Product: Policy and
Considerations,” to the safety or effectiveness of the device function(s)-under-review.
A risk assessment should document the following items (e.g., in tabular format):
· Risk Analysis
o Identification of known or foreseeable hazards
43
(and their causes) associated with
the device based on the intended use; reasonably foreseeable misuse whether
intentional or unintentional; and the impacts to safety, treatment, and/or diagnosis.
For each identified hazard, the sponsor should consider the reasonably foreseeable
sequences or combinations of events that can result in a hazardous situation,
44
and
should identify and document the resulting hazardous situation(s).
o Estimation of the risk of each hazard and hazardous situation.
45
o Severity of the harm
46
resulting from the hazardous situation.
· Initial Risk Evaluation of the Hazardous Situation
o This includes assessment of acceptability (e.g., acceptable, not acceptable) and
need for risk reduction (control) measures as defined in the risk management plan.
· Risk Control Measures
o This should include the following risk control measures listed in descending order
from highest to lowest priority:
43
For the purposes of this guidance, hazard refers to a potential source of harm.
44
For the purposes of this guidance, hazardous situation refers to a circumstance in which people, property or the
environment is/are exposed to one or more hazards.
45
It is often difficult to adequately estimate the probability of software failures that could contribute to a hazardous
situation. Applying unrealistically low probability estimates to software failures could result in unrealistic risk
evaluation and subsequently lead to inappropriate risk control measures. As a result, in some instances it may be
prudent to focus on the identification of potential software functionality and failures that could result in hazardous
situations instead of estimating probability. In such cases, considering a worse case probability is appropriate, the
probability for the software failure occurring should be set to 1.
46
For the purposes of this guidance, harm refers to injury or damage to the health of people, or damage to property
or the environment.

Contains Nonbinding Recommendations
17
§ Design (e.g., eliminating or reducing unnecessary features, modifying the
software architecture to prevent hazardous situations, modifying the user
interface to prevent usability errors)
§ Protective measures (e.g., defensive programming checks that detect
unexpected faults followed by automatic intervention to halt the delivery of
results or therapy, alarms allowing user intervention to prevent a hazardous
situation)
§ Information for safety (e.g., written warnings, on-screen warnings, training)
o There should be verification of the implementation of the risk control measures
and verification of the effectiveness of the implemented risk control measures
(i.e., the implemented risk control measure reduces risk). This can be
accomplished by tracing the identified hazard to the verified specific risk control
measures (e.g., a requirement ID in the SRS and SDS, a test name and identifier
in the testing documentation that shows pass/fail results, a user manual name and
identifier, a training manual name and identifier). For example, given an
identified hazard (e.g., HAZ-XXX
47
), the following could be listed for
traceability: a design related risk control measure that is documented in a software
requirement specification (e.g., SRS-XXX) and software design specification
(e.g., SDS-XXX), and tested as part of a unit test case (e.g., UT-XXX),
integration test case (e.g., INT-XXX) and system test case (e.g., SYS-XXX). FDA
recognizes that there may be instances where a specific hazard traces to several
requirements/specifications and tests, thereby making the presentation of
information cumbersome in the risk assessment document. Therefore, sponsors
may choose to present this traceability in a separate document linking together
software requirements specifications, software design specifications, testing and
identified hazards derived from the risk assessment.
o There should be an assessment of whether risk control measures introduce new
hazards or hazardous situations or impact the initial risk evaluation.
o Document any risk control measures employed to mitigate increased risk or
adverse effect on performance due to the combination of “other function(s),” as
that term is used in the guidance “Multiple Function Device Products: Policy and
Considerations,” with the device function-under-review.
· Residual Risk Evaluation of the Hazardous Situation after implemented Risk Control
Measures.
o This includes assessment of acceptability (e.g., acceptable, not acceptable) as
defined in the risk management plan.
47
“XXX” denotes a unique number identifier for a specific hazard, software requirement specification, software
design specification, unit test case, integration test case or system test case.

Contains Nonbinding Recommendations
18
· Benefit-Risk
o If a residual risk is deemed not acceptable according to the acceptability criteria in
the risk management plan and further risk control is not possible, the sponsor
should provide documented benefit-risk analysis to demonstrate that the benefits
of the intended use outweigh the residual risk, which may be referenced in other
benefit-risk assessment documentation.
(3) Risk Management Report
A risk management report should be provided to:
· Show how the risk management plan has been appropriately implemented.
· Demonstrate that the risk management file has been assessed by the appropriate
personnel and the overall residual risk is acceptable.
· Demonstrate appropriate methods are established for the collection and assessment of
relevant production and post-production information.
Software Requirements Specification (SRS)
The SRS documents the requirements
48
for the software which typically specifies inputs and
outputs, functions that the software will perform, hardware,
49
performance,
50
interfaces,
51
user
interaction, error definition and handling, intended operating environment, safety related
requirements derived from a risk assessment (Refer to Section VI.C Risk Management File) and
all ranges, limits, defaults, and specific values that the software will accept. For additional details
on what should be included in the software requirements specification, refer to the guidance,
“General Principles of Software Validation.”
48
The term “requirements” is used in this section as part of the term “Software Requirements Specification,” and
does not refer to a regulatory requirement.
49
Hardware requirements generally include, but are not limited to, requirements related to: microprocessors,
memory devices, sensors, energy sources, safety features, and communications.
50
Software performance and functional requirements generally include, but are not limited to, requirements related
to algorithms or control characteristics for therapy, diagnosis, monitoring, alarms, analysis, and interpretation with
full text references or supporting clinical data, if necessary. Software performance and functional requirements may
also include: device limitations due to software, internal software tests and checks, error and interrupt handling, fault
detection, tolerance, and recovery characteristics, safety requirements, and timing and memory requirements.
51
Interface requirements (e.g., external, user, internal) generally include, but are not limited to, both communication
between system components and communication with the user such as: printers, monitors, keyboard, mouse, cloud
servers, peripheral medical devices, mobile technology platforms.

Contains Nonbinding Recommendations
19
The QSR requires “a mechanism for addressing incomplete, ambiguous, or conflicting
requirements.”
52
Each requirement identified in the software requirements specification should
be traceable and we recommend that the SRS be evaluated for accuracy, completeness,
consistency, testability, correctness, and clarity.
A singular SRS document or set of multiple SRS documents should be provided. The
documentation should include a description of the software requirement identification and
tracking methodology used to support the traceability of the requirements. For example, in a
device that includes multiple functions, components, and/or accessories, where each has its own
individual SRS document and associated software device functions, the set of individual SRS
documents would comprise the complete SRS.
FDA acknowledges that modern development practices may employ incremental or evolutionary
software development practices. Additional forms of software requirements might be included in
the submission, such as well elaborated stories, use cases, textual descriptions, screen mockups,
and data flows.
In order to facilitate a timely premarket review, the following recommendations should be
considered in preparing SRS documentation:
· Format the SRS to be well-organized, easily navigable and readable with the labeling
and/or grouping of requirements (such as by modules or units of a function).
· Note any relevant traceability between requirements listed in the SRS and information
related to those requirements in other software documentation (such as the SDS, System
and Software Architecture Diagram, etc.).
· If the premarket submission involves a modification to an existing approved or cleared
device, highlight all pertinent differences in software requirements.
· Identify the requirements the sponsor believes are most critical (i.e., could have the most
significant impact) to the device’s safety and effectiveness. These requirements could be
highlighted within the SRS document and/or consolidated in a supplemental document
that includes these requirements of interest in a summarized format. This technique may
help facilitate the presentation of those requirements that most critically affect clinical
functionality or performance specifications that are directly associated with the intended
use of the device, or would otherwise impact the device’s safety and effectiveness.
52
See 21 CFR 820.30(c) (“Design input. Each manufacturer shall establish and maintain procedures to ensure that
the design requirements relating to a device are appropriate and address the intended use of the device, including the
needs of the user and patient. The procedures shall include a mechanism for addressing incomplete, ambiguous, or
conflicting requirements. The design input requirements shall be documented and shall be reviewed and approved
by a designated individual(s). The approval, including the date and signature of the individual(s) approving the
requirements, shall be documented.”).

Contains Nonbinding Recommendations
20
· If any of the information requested above is included in another document, an annotation
and a reference to the document in the submission where this information is located
should be provided.
Documentation of requirements included in the premarket submission for the device function-
under-review should include adequate detail to describe any expected relationship, utility,
reliance, or interoperability with any “other function,” as that term is used in the guidance
“Multiple Function Device Products: Policy and Considerations.”
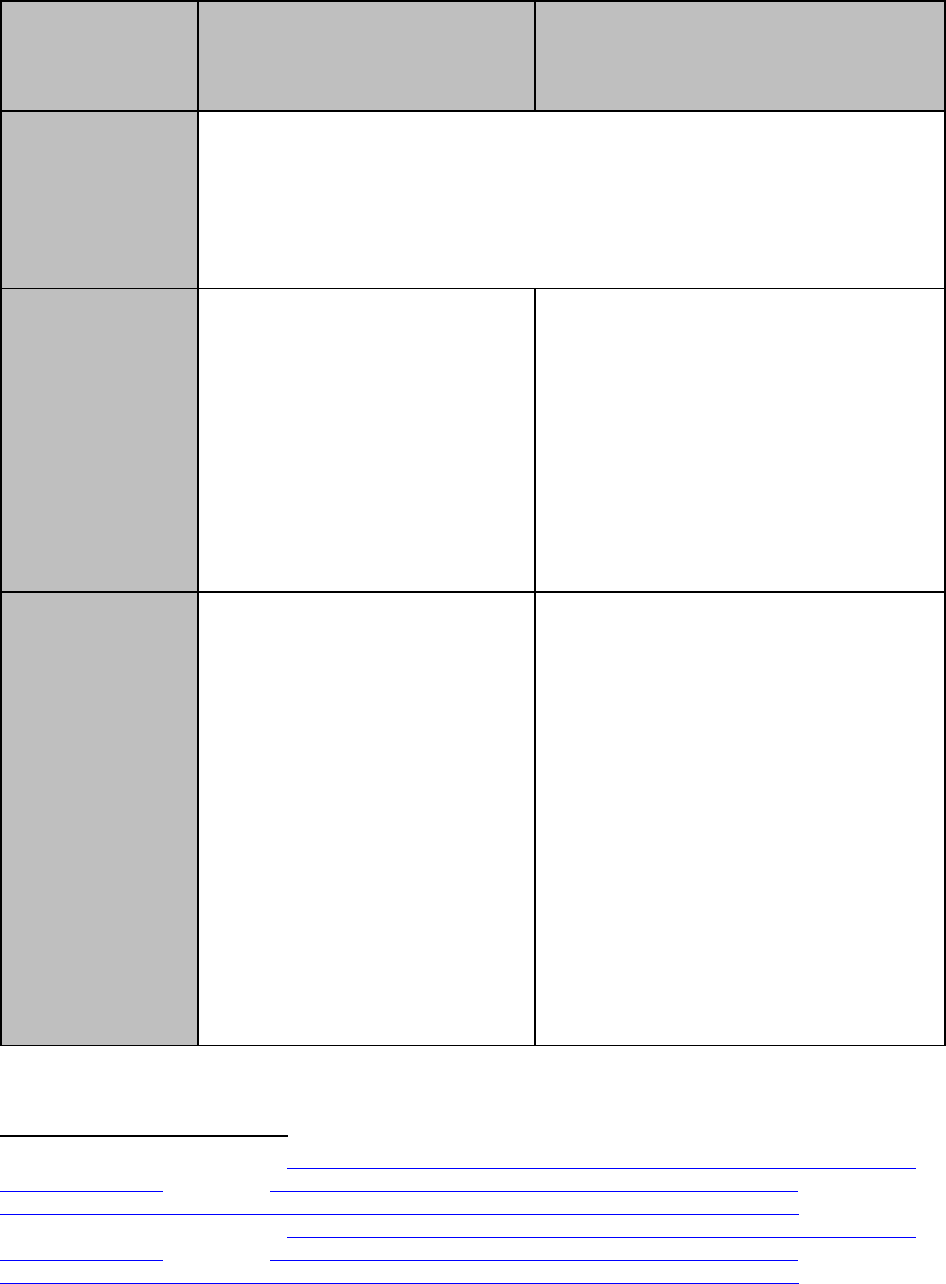
System and Software Architecture Diagram
The purpose of the system and software architecture diagram is to present a roadmap of the
device design to facilitate a clear understanding of:
· The modules and layers that make up the system and software;
· The relationships among the modules and layers;
· The data inputs/outputs and flow of data among the modules and layers; and
· How users or external products, including IT infrastructure and peripherals (e.g.,
wirelessly connected medical devices) interact with the system and software.
For purposes of the system and software architecture diagram, this guidance considers a module
to be a discrete unit or architectural item within the system or software. A module could
represent, for example, a finished hardware device within a system of hardware and software
products, a hardware component within a finished hardware device, a finished software product
within a system of software products, or a software function within a finished software product.
A module is not specifically meant to describe code-level software functions, although such
code-level software functions could be considered modules if appropriate. The sponsor should
determine what constitutes a module in the context of its system and software.
Sponsors should provide the appropriate level of detail in the system and software architecture
diagram to convey the information in a manner that can facilitate an efficient premarket review,
including descriptive text (in the diagram or in an accompanying document) to explain the
architecture diagram where appropriate. A system and software architecture diagram that is not
appropriately tailored (e.g., too high-level, too detailed, or overly confusing) or that is illegible
(e.g., cropped diagrams, inadequate font size, unreadable without high magnification) could lead
to requests for additional information from the FDA review division. To the extent appropriate,
the system and software architecture diagram can be communicated in one or more diagrams and
in one or more formats, and may convey different dimensions of the system and software. The
system and software architecture diagram may comprise multiple static diagrams (e.g., those
illustrated in Appendix B), dynamic diagrams (e.g., state diagrams), cybersecurity architecture
diagrams, and others as needed for adequate detail. If more than one diagram is used, the sponsor
Contains Nonbinding Recommendations
21
should provide a high-level diagram that communicates the overview and points to the other
diagrams that provide additional detail. The relationship between diagrams should also be clearly
communicated. In general, the sponsor should take note of the following visual, language, and
reference considerations when developing an effective system and software architecture diagram:
· Visual Considerations
o The diagram and the means for communicating information in the diagram should
be visually consistent (e.g., a solid arrow should convey a specific meaning as
compared to a dashed arrow; icons should be used consistently; lines that intersect
should clearly communicate whether the intersection is a crossing or connection)
and the meaning ascribed should be clearly communicated (e.g., through the use
of standard symbols and notation).
o The level of detail provided in the diagram should be consistent throughout unless
areas of less detail are clearly explained (e.g., in the case of functions, within the
system or software, that are not under review).
o Modules should be grouped in a logical and obvious manner.
o Use of color or other visual means (e.g., dashed boxes within solid boxes) should
be used to convey layering within the system, software, or module.
o Visual clutter should be avoided, and the diagram should be scaled according to
the complexity and amount of information presented.
· Language Considerations
o Annotations should be used to provide additional information about a particular
module or data element (e.g., a plain language explanation of the module purpose
or a pointer to a document or requirement within the premarket submission).
o Use of terminology and naming conventions should be consistent within the
diagram and the remainder of the premarket submission materials.
o Use of acronyms, jargon, or terms that are not defined in the diagram itself should
be avoided.
· Reference Considerations
o The diagram should reference other documents in the submission (e.g., Software
Description, Software Requirements Specification), as appropriate.
o For submissions related to modification(s) to a previously cleared or approved
device, the diagram should identify and reference the modules that are affected by
the modification(s).
The above considerations are intended to serve as a guide and may not apply in every case or
may apply differently for different diagrams. When developing the system and software

Contains Nonbinding Recommendations
22
architecture diagram, sponsors are encouraged to leverage industry best practices within and
outside the medical device industry. OTS modeling languages or platforms may be used to
develop the system and software architecture diagram. Any modeling language- or platform-
specific terminology should be defined or supported by reference to a publicly-available
consensus standard or specification. The resulting architecture diagram should be provided in a
form that aligns with eCopy guidelines recommended in the guidance “eCopy Program for
Medical Device Submissions.”
53
For multiple function device products, the system and software architecture diagram should
clearly delineate between the device functions-under-review and the “other functions,” as that
term is used in the guidance “Multiple Function Device Products: Policy and Considerations.”
The system and software architecture diagram should include adequate detail to understand how
or if the “other function(s)” interact with or impact the device function-under-review.
Example system and software architecture diagrams are provided in Appendix B of this
guidance, illustrating approaches to effectively convey the recommended information to
facilitate an efficient premarket review. These simplified examples demonstrate how the
considerations described in this section can be implemented into a system and software
architecture diagram. The modules in the example are intended for illustration purposes only and
are not intended to document or represent a comprehensive or complete system and software
architecture diagram for a specific medical device or system. The illustrated approach does not
prescribe any specific modeling languages or platforms to allow for flexibility in development
and documentation. The approaches illustrated can be applied to any system and software
architecture diagram.
Software Design Specification (SDS)
The Software Design Specification (SDS) may contain both a high level summary of the design
and detailed design information. In terms of the relationship between the Software Requirement
Specification (SRS) and the SDS, the SRS describes what the software function will do and the
SDS describes how the requirements in the SRS are implemented. The information presented in
the SDS should be sufficient to ensure that the work performed by the software engineers who
created the device software function was clear and unambiguous, with minimal ad hoc design
decisions. The use of minimal ad hoc design decisions reflects the general principle that the
creation of SDS should occur as a prospective activity rather than documented retrospectively
after the software design has been implemented by ad hoc design methods. Documentation of
specifications included in the premarket submission for the device function-under-review should
include adequate detail to describe any expected relationship, utility, reliance, or interoperability
with any “other function,” as that term is used in the guidance “Multiple Function Device
Products: Policy and Considerations.”
53
Available at https://www.fda.gov/regulatory-information/search-fda-guidance-documents/ecopy-program-
medical-device-submissions.

Contains Nonbinding Recommendations
23
(1) Basic Documentation Level
FDA is not recommending the SDS as part of the premarket submission. Sponsors should
document this information on the design internally via the DHF for the device. During premarket
review, FDA may request additional information on the design, if needed, to evaluate the safety
and effectiveness of the device.
(2) Enhanced Documentation Level
A singular SDS document or set of SDS documents that provide the technical design details of
how the software functions, how the software design completely and correctly implements all the
requirements of the SRS and how the software design traces to the SRS in terms of intended use,
functionality, safety, and effectiveness. The software functional units or modules along with the
interfaces among them identified in the architectural (i.e., high-level) design should be
documented with the corresponding detailed (i.e., low-level) design information in the SDS. The
information provided for review should be sufficient to ensure that the work performed in
developing the software functional units or modules and their interfaces was clear and
unambiguous, with minimal ad hoc design decisions. For example, the creation of the SDS is
expected to have occurred as a prospective activity where the SDS was used to guide the design,
development and testing of the software rather than documented retrospectively after the
software design has been implemented by ad hoc design methods.
For additional details on what should be included in the software design specification, refer to
the guidance, “General Principles of Software Validation.”
Software Development, Configuration Management, and
Maintenance Practices
One way a sponsor could address this documentation element of a premarket submission is to
provide the recommended information on related software development, configuration
management, and maintenance practices and procedures. In another approach, a sponsor could
provide a Declaration of Conformity to specific clauses of the FDA-recognized version of
ANSI/AAMI/IEC 62304 Medical Device Software - Software Life Cycle Processes or the FDA-
recognized version of IEC 62304 Medical device software - Software life cycle processes
(hereafter referred to collectively as ANSI/AAMI/IEC 62304). A sponsor could also provide a
Declaration of Conformity to the complete ANSI/AAMI/IEC 62304 standard. However, a
Declaration of Conformity to the complete ANSI/AAMI/IEC 62304 standard is not needed due
to known differences in categorization of device software functions and other recommended
documentation.

Contains Nonbinding Recommendations
24
(1) Basic Documentation Level
A summary of the processes and procedures that are in place to manage the software life cycle
development, software configuration and change management, and software maintenance
activities should be provided. This summary information should include an adequate description
of:
· Processes and procedures used in software development, verification, and validation.
· Software coding standards, methods, and tools used in software development.
· Main deliverables of the typical activities and tasks involved in software development,
verification, and validation.
· Processes, procedures, and tools used to link user needs, system requirements, software
requirements, software design specifications, software testing and implemented risk
control measures (i.e., traceability).
· Processes and procedures used in software configuration and change management.
· Processes and procedures used in software maintenance that includes risk assessment of
software changes, initial testing that evaluates the correctness of the implemented
software change(s), and regression analysis and testing.
Alternatively, to address the recommendations for this documentation element, a sponsor may
provide a Declaration of Conformity to the FDA-recognized version of ANSI/AAMI/IEC 62304
Medical Device Software - Software Life Cycle Processes, including subclauses 5.1.1 (“Software
development plan”), 5.1.2 (“Keep software development plan updated”), 5.1.3 (“Software
development plan reference to system design and development”), 5.1.6 (“Software verification
planning”), 5.1.7 (“Software risk management planning”), 5.1.8 (“Documentation planning”),
5.1.9 (“Software configuration management planning”), clause 6 (“Software maintenance
process”), and clause 8 (“Software configuration management process”),
54
among others as
applicable.
(2) Enhanced Documentation Level
Documents implementing the configuration management and maintenance plans should be
provided in addition to the summary documentation requested for the Basic Documentation
Level, as described above.
Alternatively, to address the recommendations for this documentation element, a sponsor may
provide a Declaration of Conformity to the FDA-recognized version of ANSI/AAMI/IEC 62304
54
References to clauses and subclauses based on ANSI/AAMI/IEC 62304:2006 & A1:2016.

Contains Nonbinding Recommendations
25
Medical Device Software – Software Life Cycle Processes, including subclause 5.1 (Software
development planning), clause 6 (“Software maintenance process”), and clause 8 (“Software
configuration management process”),
55
among others as applicable.
Software Testing as part of Verification and Validation
Refer to Section IV (Definitions) for important information pertaining to FDA’s thinking on
verification and validation, as it relates to this guidance. Additionally, please refer to guidance
“General Principles of Software Validation” for additional details regarding FDA’s thinking
regarding software testing, particularly unit level (module or component) testing, integration
level (internal and external interfaces) testing, and system level (functional) testing.
The recommendations in this guidance do not address other forms of testing, such as non-clinical
bench, animal, or clinical testing. For more information regarding other forms of device
performance testing, sponsors should refer to relevant special controls,
56
FDA-recognized
voluntary consensus standards,
57
and device-specific guidances. If the premarket submission
leverages information from other device performance testing section(s) to address software
verification and validation, the sponsor is encouraged to appropriately reference the other
performance testing material to facilitate the navigation between submission sections, reduce
instances of duplication, and improve readability.
(1) Basic Documentation Level
The following software testing documentation should be provided:
· A summary description of the testing activities at the unit, integration, and system levels.
The summary description should include the software version tested and the overall
pass/fail test results for all test protocols (i.e., collection of test procedures for specific
software functionality) executed. If the device is a modified version of a previously
cleared or approved device, provide a summary of the modifications compared with the
previous cleared or approved version along with a summary description of the additional
testing activities performed at the unit, integration, and system levels as compared with
the previously cleared or approved version.
· Any intentional changes made in response to failed tests and documentation of test results
demonstrating that the intentional changes were implemented correctly.
· A regression analysis and regression testing with pass/fail test results (where applicable)
to account for unintended effects of a software change.
55
References to clauses and subclauses based on ANSI/AAMI/IEC 62304:2006 & A1:2016.
56
For more information regarding special controls, please see FDA’s Regulatory Controls webpage, available at
https://www.fda.gov/medical-devices/overview-device-regulation/regulatory-controls.
57
The most up-to-date list of voluntary FDA- recognized consensus standards is available at
https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfStandards/search.cfm.

Contains Nonbinding Recommendations
26
o Regression analysis is a documented evaluation of the impact of a software
change based on review of the relevant documentation (e.g., software
requirements specification, software design specification, source code, test plans,
test cases, test scripts, etc.) to determine whether regression testing is needed. If
regression testing is needed as determined by the regression analysis, the sponsor
should identify the necessary regression tests to be run. Regression testing is the
rerunning of test cases that a program has previously executed correctly and
comparing the current result to the previous result in order to detect unintended
effects of a software change.
· System level test protocol including expected results derived from software requirements,
actual results that are observed and recorded, objective pass/fail determination (i.e.,
actual results are acceptably equivalent to expected results) and a system level test report.
The system level test report should demonstrate that the protocol has been acceptably
executed with passing test results and any unresolved anomalies have been acceptably
deferred based on a risk assessment for the candidate release version.
(2) Enhanced Documentation Level
In addition to the documentation requested for the Basic Documentation Level, all unit and
integration level test protocols and reports should be provided, including expected results derived
from software requirements and design, actual results that are observed and recorded, and
objective pass/fail determination (i.e., actual results are acceptably equivalent to expected
results). The unit and integration level test reports should demonstrate that the protocols have
been acceptably executed with passing testing results and any unresolved anomalies have been
acceptably deferred based on a risk assessment for the candidate release version.
Software Version History
The documentation should include the history of software versions that were tested and
documented at the unit, integration, and system levels as part of verification and validation
activities, beginning with the version that became subject to the design controls, as described in
21 CFR 820.30. This typically takes the form of a line-item tabulation including the date, version
number that was tested (including, if applicable, bench, animal, and clinical testing) and a brief
description of all changes in the version relative to the previously tested version.
The last entry in a line-item tabulation should be the final version to be incorporated in the
released device. This entry should also include any differences between the tested version of
software and the released version, along with an assessment of the potential effect of the
differences on the safety and effectiveness of the device.
If the software version history includes a version(s) that corresponds to a previously released
cleared or approved version of the software, the sponsor should highlight in the version history
document each prior released cleared or approved version and the premarket submission
number(s) associated with that release.

Contains Nonbinding Recommendations
27
If the device is a multiple function device product and includes software function(s) that are
considered “other functions,” as that term is used in the guidance “Multiple Function Device
Products: Policy and Considerations,” the recommendations described in the aforementioned
guidance should be considered when preparing the software version history.
Unresolved Software Anomalies
An anomaly is any condition that deviates from the expected behavior based on user needs,
requirements, specifications, design documents, or standards. Anomalies may be found during
the review, test, analysis, compilation, or use of the software (whether before or after release, or
whether inside a sponsor’s organization or outside it) or at other times. An unresolved software
anomaly is a defect that still resides in the software because a sponsor deemed it appropriate not
to correct or fix the anomaly, according to a risk-based rationale about its impact to the device’s
safety and effectiveness.
A list of unresolved anomalies should document the following items (e.g., in tabular format) for
each unresolved anomaly present in the software:
· A description of the anomaly;
· Identification of how the anomaly was discovered and, where possible, identification of
the root cause(s) of the anomaly;
· Evaluation of the impact of the anomaly on the device’s safety and effectiveness,
including operator usage and human factors considerations;
· Outcome of the evaluation; and
· Risk-based rationale for not correcting or fixing the anomaly in alignment with the
sponsor’s risk management plan or procedure(s).
Additionally, the Agency recommends considering the utilization of a defect classification
system, or taxonomy, for each anomaly, such as ANSI/AAMI SW91’s Classification of defects
in health software.
58
Regardless of the defect classification system used, the sponsor should
evaluate the impact of an unresolved anomaly on the device’s safety and effectiveness based on
the software’s intended use.
Where appropriate, a sponsor should communicate to end users any mitigations or possible
work-arounds for unresolved anomalies to assist in the proper operation of the device to fulfill its
intended use. FDA recommends that any planned or already distributed communication
58
As stated in ANSI/AAMI SW91, a defect classification system, or taxonomy, is used to classify or categorize the
types of defects that might exist in software. A defect classification system is neutral with respect to programming
language, methodology, product, intended use, risk (severity of anomalies), and regulatory status.

Contains Nonbinding Recommendations
28
(customer notification, labeling, etc.) to end users regarding unresolved anomalies is referenced
in the premarket submission.
VII. Additional Information Regulatory Considerations for
Software Functions
Section 3060(a) of the Cures Act amended section 520 of the FD&C Act on December 13, 2016,
removing certain software functions from the definition of device in section 201(h) of the FD&C
Act. Sponsors should consider the following reference materials to learn more about FDA’s
regulatory considerations for software functions:
· Changes to Existing Medical Software Policies Resulting from Section 3060 of the 21st
Century Cures Act
59
· General Wellness: Policy for Low Risk Devices
60
· Policy for Device Software Functions and Mobile Medical Applications
61
· Medical Device Data Systems, Medical Image Storage Devices, and Medical Image
Communications Devices
62
· How to Determine if Your Product is a Medical Device
63
· Clinical Decision Support Software
64
Appendix A: Documentation Level Examples
The following list of example devices is intended to demonstrate the implementation of the
Documentation Level risk-based approach. Please note that these generalized examples do not
necessarily account for every possible detail, risk, or consideration a sponsor should evaluate,
and should not be taken to mean that the devices described do or do not require a certain
Documentation Level. These examples do not define the appropriate Documentation Level for a
particular device type. As such, the Documentation Level should be uniquely considered for each
particular device or device modification and in consideration of the device’s intended use. The
rationales in the examples below are abbreviated and FDA encourages sponsors to provide a
detailed assessment that accounts for the specifics of their device (such as the device’s risk
assessment,
65
software description, etc.) when addressing the recommendations in Sections V
and VI.A of this guidance.
59
Available at https://www.fda.gov/regulatory-information/search-fda-guidance-documents/changes-existing-
medical-software-policies-resulting-section-3060-21st-century-cures-act.
60
Available at https://www.fda.gov/regulatory-information/search-fda-guidance-documents/general-wellness-
policy-low-risk-devices.
61
Available at https://www.fda.gov/regulatory-information/search-fda-guidance-documents/policy-device-software-
functions-and-mobile-medical-applications.
62
Available at https://www.fda.gov/regulatory-information/search-fda-guidance-documents/medical-device-data-
systems-medical-image-storage-devices-and-medical-image-communications-devices.
63
Available at https://www.fda.gov/medical-devices/classify-your-medical-device/how-determine-if-your-product-
medical-device.
64
Available at https://www.fda.gov/regulatory-information/search-fda-guidance-documents/clinical-decision-
support-software.
65
For more information on risk assessment refer to Section VI.C of this guidance.
Contains Nonbinding Recommendations
29
1. A hardware-only non-patient-matched hip prosthesis.
Description: The device is a non-patient-matched hip prosthesis that contains no
firmware or other means for software-based control.
Rationale: The device does not contain software.
Outcome: No Documentation Level
2. A non-contact infrared thermometer intended for intermittent measurement of body
temperature from the forehead.
Description: The device is intended to measure body temperature from the
forehead using an infrared sensor. The device is a hand-held, battery powered,
reusable device for home and professional healthcare facility use.
Rationale: In general, a failure or latent flaw of the device software function(s)
would not present a hazardous situation with a probable risk of death or serious
injury to either a patient, user of the device, or others in the environment of use,
prior to the implementation of risk control measures.
Outcome: Basic Documentation Level
3. A non-invasive blood pressure monitor with inflatable cuff.
Description: The device is intended to measure a person’s systolic and diastolic
blood pressure in units of mmHg through software-controlled inflation and
deflation of the cuff. The device is a battery powered, reusable device for home
and professional healthcare facility use.
Rationale: In general, a failure or latent flaw of the device software function(s)
would not present a hazardous situation with a probable risk of death or serious
injury to either a patient, user of the device, or others in the environment of use,
prior to the implementation of risk control measures.
Outcome: Basic Documentation Level
4. A device software function for optical camera-based measurement of pulse rate and
breathing rate.
Description: The device is intended for non-contact, periodic, spot measurement
of pulse rate and breathing rate when the subject is at rest. The software analyzes
compatible video signals of subjects in single occupancy rooms of subjects that do
not require continuous vital signs monitoring or critical care. The device is
Contains Nonbinding Recommendations
30
intended for use by trained staff and is not intended to be the sole method of
checking the physical health of a subject.
Rationale: A failure or latent flaw of the device software function(s) would not
present a hazardous situation with a probable risk of death or serious injury to
either a patient, user of the device, or others in the environment of use, prior to the
implementation of risk control measures.
Outcome: Basic Documentation Level
5. A computerized behavioral therapy device to treat psychiatric disorders.
Description: The device is a prescription software device intended to provide a
computerized version of condition-specific cognitive behavioral therapy as an
adjunct to clinician supervised outpatient treatment to patients who have been
previously diagnosed with a psychiatric condition. It is intended to provide
patients access to therapy tools used during treatment sessions to improve
recognized treatment outcomes. It is not intended to substitute for routine in-
person therapy sessions.
Rationale: A failure or latent flaw of the device software function(s) would not
present a hazardous situation with a probable risk of death or serious injury to
either a patient, user of the device, or others in the environment of use, prior to the
implementation of risk control measures.
Outcome: Basic Documentation Level
6. A traumatic brain injury (TBI) eye movement assessment aid.
Description: The device is a prescription device that is intended to track a
patient’s eye movements using a commercial OTS mobile phone and camera and
analyze the tracked eye movements to aid in the assessment of mild TBI,
commonly known as “concussion.” The device provides a positive or negative
indicator about the presence of eye movements that are consistent with mild TBI.
Rationale: A failure or latent flaw of the device software function(s) would not
present a hazardous situation with a probable risk of death or serious injury to
either a patient, user of the device, or others in the environment of use, prior to the
implementation of risk control measures. The device is intended for use as an aid
in the assessment of mild (non-severe) injury and is not intended as a standalone
diagnostic.
Outcome: Basic Documentation Level
7. An implantable sensor for measuring pulmonary artery (PA) pressure.
Contains Nonbinding Recommendations
31
Description: The device is a system comprising a permanent implant that is
placed in the PA and an external software-controlled reader that retrieves and
transmits PA pressure measurements. The PA hemodynamic data are used by the
physician for heart failure management and with the goal of reducing heart failure
hospitalizations. There are no device software functions located on the implant.
Rationale: While the device is a class III device, a failure or latent flaw of the
device software function(s) would not present a hazardous situation with a
probable risk of death or serious injury to either a patient, user of the device, or
others in the environment of use, prior to the implementation of risk control
measures. Risks of death or serious injury to the patient in this scenario are due to
the implantable nature of the device and failure of the hardware or implant
procedure itself, not the device software function(s).
Outcome: Basic Documentation Level
8. Over-the-counter (OTC) application for identification of irregular heart rhythms.
Description: The software application is intended for analysis of
photoplethysmograh data, identification of episodes of irregular heart rhythms
suggestive of Atrial Fibrillation (AF), and providing notification to the user of the
irregular rhythm episodes. It is intended to opportunistically provide a notification
of possible AF and the absence of a notification is not intended to indicate that no
disease is present. It is intended for OTC use and it is not intended to replace
traditional methods of diagnosis or treatment.
Rationale: A failure or latent flaw of the device software function(s) would not
present a hazardous situation with a probable risk of death or serious injury to
either a patient, user of the device, or others in the environment of use, prior to the
implementation of risk control measures.
Outcome: Basic Documentation Level
9. An in vitro nucleic acid test for the qualitative detection of Human Papillomavirus (HPV)
DNA in human cervical specimens.
Description: The test is intended for routine cervical cancer screening as per
professional medical guidelines, including triage of ASC-US (atypical squamous
cells of undetermined significance) cytology, co-testing (or adjunctive screen)
with cytology, and HPV primary screening of women to assess the risk for
cervical precancer and cancer. Patients should be followed-up in accordance with
professional medical guidelines, results from prior screening, medical history, and
other risk factors.
Contains Nonbinding Recommendations
32
Rationale: While the test is classified as a class III device, a failure or latent flaw
of the device software function(s) would not present a probable risk of death or
serious injury to either a patient, user of the device, or others in the environment
of use prior to the implementation of risk control measures. There are several
alternatives for the detection of cervical cancer precursors including testing by
cytology alone, co-testing with HPV alongside or as a follow-up to cytology or
HPV testing as a first line screening test for cervical cancer. The patient’s age,
medical history, and thorough physical examination will provide further
information on the risk of cervical disease, as well as the need for referral to
colposcopy. The test should only be used in conjunction with this clinical
information in accordance with professional clinical patient management
guidelines.
Outcome: Basic Documentation Level
10. An in vitro nucleic acid test for the qualitative detection and differentiation of influenza
A virus, influenza B virus, and respiratory syncytial virus (RSV) RNA in nasopharyngeal
swabs from human patients with signs and symptoms of respiratory tract infection in
conjunction with clinical and epidemiological risk factors.
Description: The test is intended for use as an aid in the differential diagnosis of
influenza A, influenza B, and RSV viral infections in humans. Negative results do
not preclude influenza virus or RSV infection and should not be used as the sole
basis for treatment or other patient management decisions.
Rationale: A failure or latent flaw of the device software function(s) would not
present a hazardous situation with a probable risk of death or serious injury to
either a patient, user of the device, or others in the environment of use prior to the
implementation of risk control measures.
Outcome: Basic Documentation Level
11. A device software function on a commercial OTS head-mounted display (e.g., augmented
reality/virtual reality/mixed reality (AR/VR/MR)) that superimposes pre-surgical images
on a patient’s body.
Description: The device is intended to provide real-time superimposition of
medical images on the patient during a surgical procedure, but is neither intended
to directly guide surgical planning or procedures nor be worn by the lead surgeon.
Rationale: A failure or latent flaw of the device software function(s) would not
present a hazardous situation with a probable risk of death or serious injury to
either a patient, user of the device, or others in the environment of use prior to the
implementation of risk control measures, since the device is neither intended to
directly guide surgical planning or procedures nor be worn by the lead surgeon.
Contains Nonbinding Recommendations
33
Outcome: Basic Documentation Level
12. A laser system for the treatment of acne vulgaris.
Description: The device is a laser system used in dermatology offices used to
treat acne vulgaris through heating of dermal tissue. The device software only
operates the laser engine. Delivery of therapy requires the operator to depress a
physical switch on the handpiece.
Rationale: A failure or latent flaw of the device software functions(s), such as not
delivering the laser energy when directed, would not present a hazardous situation
with a probable risk of death or serious injury to the patient prior to the
implementation of risk control measures.
Outcome: Basic Documentation Level
13. A radiological display device.
Description: The device is intended for displaying clinical radiology images for
review, analysis, and diagnosis by trained medical practitioners. The software
contained in the device is limited to the following functionalities: display controls,
ambient light sensing, luminance calibration tools, and quality-control software.
Rationale: A failure or latent flaw of the device software function(s), such as
inadequate quality of displayed images, would not present a hazardous situation
with a probable risk of death or serious injury to either a patient, user of the
device, or others in the environment of use, prior to the implementation of risk
control measures.
Outcome: Basic Documentation Level
14. An electric breast pump.
Description: The device is intended to be used by lactating women to express and
collect milk from their breasts. The device uses a DC-powered motor driving the
vacuum pump which is controlled electronically to provide a range of user-
selected suction levels at specified cycle frequencies. The device display provides
the user with information on the pumping mode, timer, battery level, and suction.
Rationale: A failure or latent flaw of the device software function(s) (e.g.,
software fails to properly control the suction level) would not present a hazardous
situation with a probable risk of death or serious injury to either a patient, user of
the device, or others in the environment of use prior to the implementation of risk
control measures.
Outcome: Basic Documentation Level
Contains Nonbinding Recommendations
34
15. An implantable cardiac pacemaker used to treat bradycardia.
Description: The device is an implanted, programmable dual-chamber pulse
generator intended to provide rate-adaptive bradycardia therapy as well as other
therapeutic and diagnostic functionality. It senses the heart’s electrical activity
and generates electrical impulses to cause the heart to contract or beat according
to the programmed patient’s needs. It communicates with a programmer and the
patient’s home monitoring device.
Rationale: A failure or latent flaw of the device software function(s), such as
failure to pace or a latent flaw leading to incorrect sensing of an ectopic beat,
would lead to a hazardous situation that would present a probable risk of death or
serious injury to the patient prior to the implementation of risk control measures.
Outcome: Enhanced Documentation Level
16. A facility use continuous ventilator.
Description: The device is intended to provide continuous ventilation for adult,
pediatric, and neonatal patients who require invasive or noninvasive respiratory
support. It allows clinicians to set ventilator control parameters, set alarm limits,
and view monitored values and waveforms for patient management. It includes
respiratory monitoring as well as both mandatory and spontaneous ventilation
modes. It is intended for use in professional healthcare facilities.
Rationale: A failure or latent flaw of the device software function(s), such as
failure to provide appropriately timed ventilation, would present a hazardous
situation with a probable risk of death or serious injury to a patient, prior to the
implementation of risk control measures.
Outcome: Enhanced Documentation Level
17. A multi-parameter patient monitor for use in a professional healthcare facility.
Description: The device is a multi-parameter monitor intended for use on adult,
pediatric, and neonatal patients in a professional healthcare facility. It is used for
monitoring of various hemodynamic and respiratory vital signs and parameters,
including central venous oxygen saturation, electrocardiogram (ECG), arrhythmia
detection, invasive and non-invasive blood pressure, temperature, cardiac output,
hemoglobin concentration, pulse oximetry, spirometry, airway gases, and gas
exchange. It is designed to detect alarm conditions and generate alarm signals. It
can be connected to the hospital network and other monitors to allow for remote
viewing and management of patients.
Contains Nonbinding Recommendations
35
Rationale: A failure or latent flaw of the device software function(s), such as
incorrect calculation of parameters or an exploited cybersecurity vulnerability that
compromises its ability to provide life-threatening arrhythmia detection and
alarms, would present a hazardous situation with a probable risk of death or
serious injury to a patient, prior to the implementation of risk control measures.
Outcome: Enhanced Documentation Level
18. A Blood Establishment Computer Software (BECS) or BECS accessory.
Description: The device is used in the manufacture of blood and blood
components to assist in the prevention of disease in humans by identifying
ineligible donors, by preventing the release of unsuitable blood and blood
components for transfusion or for further manufacturing into products for human
treatment or diagnosis, by performing compatibility testing between donor and
recipient, or by performing positive identification of patients and blood
components at the point of transfusion to prevent transfusion reactions. A BECS
accessory is a device intended for use with BECS to augment the performance of
the BECS or to expand or modify its indications for use.
Rationale: A failure or latent flaw of the device software function(s), such as a
failure to prevent the release of unsuitable blood and blood components, would
present a hazardous situation with a probable risk of death or serious injury to a
patient, prior to the implementation of risk control measures.
Outcome: Enhanced Documentation Level
19. A qualitative in vitro nucleic acid screening test for the direct detection of Babesia DNA
and RNA in whole blood samples from individual human donors.
Description: The device is a screening test that includes device software for
detection of Babesia DNA and RNA in whole blood samples to prevent the
release of unsuitable blood and blood components.
Rationale: A failure or latent flaw of the device software function(s), such as an
inaccurate result in the identification of a transfusion-transmitted infection, would
present a hazardous situation with a probable risk of death or serious injury to a
patient, or serious injury, prior to the implementation of risk control measures.
Outcome: Enhanced Documentation Level
20. An infusion pump intended for use in a health care facility to pump fluids and
medications into a patient.
Description: The device is intended for use on adults, pediatrics, and neonates for
the intermittent or continuous delivery of fluids, medications, blood, and blood
Contains Nonbinding Recommendations
36
products through clinically accepted routes of administration (intravenous, intra-
arterial, subcutaneous, epidural, and enteral). It is intended for use by trained
health care professionals in healthcare facilities.
Rationale: A failure or latent flaw of the device software function(s), such as
providing the incorrect flow rate or failing to deliver fluids/medications, would
present a hazardous situation with a probable risk of death or serious injury to
either a patient, user of the device, or others in the environment of use, prior to the
implementation of risk control measures.
Outcome: Enhanced Documentation Level
21. An in vitro nucleic acid test for the quantitative measurement of Cytomegalovirus (CMV)
DNA in human plasma or whole blood.
Description: The test is intended for use as an aid in the management of CMV in
solid organ transplant patients and in hematopoietic stem cell transplant patients.
In patients receiving anti-CMV therapy, DNA measurements can be used to
assess viral response to treatment. The results from the test must be interpreted
within the context of all relevant clinical and laboratory findings and is not
intended for use as a screening test for blood or blood products
Rationale: The test is classified as a class III device. Furthermore, a failure or
latent flaw of the device software function(s), such as failure to provide correct
test results, would present a hazardous situation with a probable risk of death or
serious injury to the patient prior to the implementation of risk control measures.
Outcome: Enhanced Documentation Level
22. A device software function that provides a sepsis alarm to a healthcare provider in a
critical care environment.
Description: Software intended to analyze patient demographics, vital signs, and
lab results from an electronic medical record to provide asepsis alarm identifying
patients with sepsis or at risk of developing sepsis earlier than a healthcare
provider would otherwise. The patient clinical data used as an input for the
software is part of ongoing or active monitoring of the patient’s current health
state in a critical care environment.
Rationale: A failure or latent flaw of the device software function(s), such as
failure to provide a sepsis alarm) would present a hazardous situation with a
probable risk of death or serious injury to either a patient, user of the device, or
others in the environment of use prior to the implementation of risk control
measures.
Outcome: Enhanced Documentation Level
Contains Nonbinding Recommendations
37
23. A continuous glucose monitoring system.
Description: The device is intended to provide real time, continuous glucose
monitoring for the management of diabetes in persons 2 years of age and older. It
is intended to replace fingerstick blood glucose testing for diabetes treatment
decisions. It aids in the detection of episodes of hyperglycemia and hypoglycemia
and facilitates both acute and chronic therapy adjustments.
Rationale: A failure or latent flaw of the device software function(s), such as
failure to provide correct blood glucose measurement or detection of
hypoglycemia, would present a hazardous situation with a probable risk of death
or serious injury to either a patient, user of the device, or others in the
environment of use prior to the implementation of risk control measures.
Outcome: Enhanced Documentation Level
24. Powered Lower Extremity Exoskeleton.
Description: A powered lower extremity exoskeleton is a prescription device that
is composed of an external, powered, motorized orthosis that is placed over a
person's paralyzed or weakened limbs for medical purposes.
Rationale: A failure or latent flaw of the device software function(s), such as loss
of remote control or movement signal processing, could present a hazardous
situation with a probable risk of death or serious injury to the patient prior to the
implementation of risk control measures.
Outcome: Enhanced Documentation Level
25. A retinal diagnostic software device.
Description: The device is limited to prescription use and incorporates an
AI/ML-enabled algorithm that is intended to evaluate images for diagnostic
screening to identify retinal diseases or conditions.
Rationale: A failure or latent flaw of the device software function(s), such as a
diagnostic algorithm failure that provides a false result, could present a hazardous
situation with a probable risk of death or serious injury to the patient prior to the
implementation of risk control measures.
Outcome: Enhanced Documentation Level
26. A radiation therapy treatment system (e.g., Linear accelerator).

Contains Nonbinding Recommendations
38
Description: The device is intended to noninvasively deliver a focal dose of
radiation to a specified volume of a patient's anatomy while sparing the
surrounding normal tissues and structures.
Rationale: A failure or latent flaw of the device software function(s), such as
under- or overdose to a target volume or delivery to the wrong volume, could
present a hazardous situation with a probable risk of death or serious injury to the
patient prior to the implementation of risk control measures.
Outcome: Enhanced Documentation Level
27. A drug-device combination, with the device constituent part detecting ingestion of the
drug component to prevent treatment failure.
Description: The product is a combination product comprised of two regulated
components (drug and device). The “primary mode of action”
66
is the drug
component, provided as a tablet. The device constituent part detects ingestion of
the tablet component to monitor adherence to the drug regimen. A missed dose
greatly increases the likelihood of treatment failure for a life-threatening
condition. The device constituent part includes hardware (sensors) and software
(signal processing).
Rationale: A failure or latent flaw of the device software function(s), such as a
false detection of tablet ingestion, would present a hazardous situation with a
probable risk of death or serious injury to either a patient (through worsening of
the life-threatening disease), user of the device, or others in the environment of
use, prior to the implementation of risk control measures.
Outcome: Enhanced Documentation Level
66
Section 503(g)(1)(C) of the FD&C Act states that the term “primary mode of action” means “the single mode of
action of a combination product expected to make the greatest contribution to the overall intended therapeutic
effects of the combination product.”
Contains Nonbinding Recommendations
39
Appendix B: System and Software Architecture Diagram
Examples
The three example diagrams below are simplified for the purpose of demonstrating how the
considerations described in Section VI.E (System and Software Architecture Diagram) could be
implemented into diagrams that facilitate a clear understanding of the system and software. Each
diagram is supported by descriptive text and a brief discussion on notable features. The examples
are intended for illustration purposes only and do not describe a complete and comprehensive
system and software architecture diagram.
The illustrative diagrams are based on the following distinct example devices:
1. A hand-held diagnostic device
2. An implantable therapeutic device with patient- and provider-facing applications
3. A cloud-based device algorithm for analyzing previously captured medical images
The diagrams are largely static diagrams with high-level identification of interfaces between
system and software components. The use of any specific design or formatting features is only
provided as a suggestion and does not preclude the use of alternative approaches and/or OTS
modeling languages or platforms.
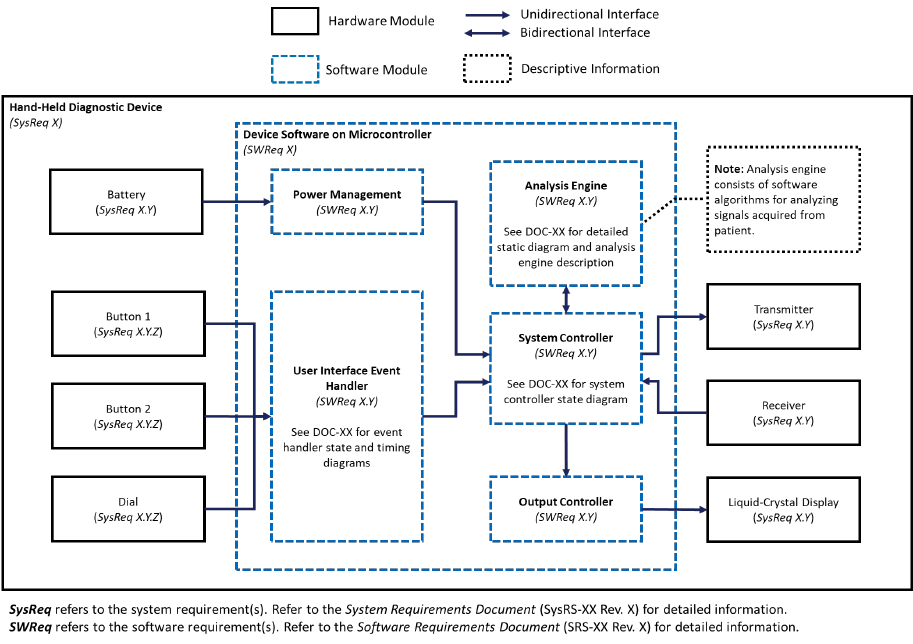
Contains Nonbinding Recommendations
40
Figure 1: Example System and Software Architecture Diagram – Hand-Held Diagnostic
Device
Figure 1 depicts a static, high-level system and software architecture diagram of the modules in a
fictional hand-held diagnostic device. A legend is provided to describe visual features used to
identify different components. References are provided to documents containing more
information, including other static diagrams, dynamic diagrams, and detailed descriptions. An
annotation is provided to improve clarity on the purpose of one module. Text is provided with
adequate clarity and font size for readability.
Contains Nonbinding Recommendations
41
Figure 2: Example System and Software Architecture Diagram – Implantable Therapeutic
Device with Patient- and Provider-Facing Applications
Figure 2 depicts a static, high-level system and software architecture diagram of the modules in a
fictional implantable therapeutic device. The example depicts the implementation of device
software functions on OTS platforms, including a cloud computing platform and a smartphone
application.
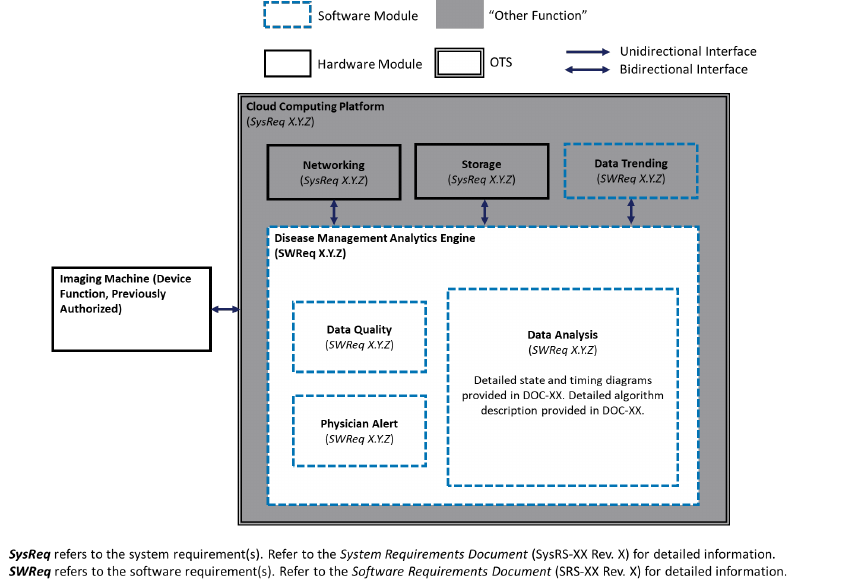
Contains Nonbinding Recommendations
42
Figure 3: Example System and Software Architecture Diagram - Cloud-based Device
Algorithm for Analyzing Previously Captured Medical Images
Figure 3 depicts a static, high-level system and software architecture diagram of the modules in a
fictional cloud-based algorithm for analyzing previously captured medical images. The example
shows how a software-only function can be described from a platform-level perspective.
