
General, Organic, and Biological Chemistry
Practice Exam Questions
You may use a periodic table and a calculator only. Some of these questions may cover material
your instructor did not emphasize in your course. Just skip those and focus on the material that
was covered in your particular course.
Source: Test Bank, Timberlake, Structures of Life, 4e (Pearson)
Chapter 1: Chemistry and Measurements
1) The measurement 0.000 0043 m, expressed correctly using scientific notation, is
A) 4.3 × 10
-7
m.
B) 4.3 × 10
-6
m.
C) 4.3 × 10
6
m.
D) 0.43 × 10
-5
m.
E) 4.3 m.
2) Which of the following numbers contains the designated CORRECT number of significant
figures?
A) 0.04300 5 significant figures
B) 0.00302 2 significant figures
C) 156 000 3 significant figures
D) 1.04 2 significant figures
E) 3.0650 4 significant figures
3) Which of the answers for the following conversions contains the correct number of significant
figures?
A) 2.543 m ×
m 1
in 39.4
= 100.1942 in
B) 2 L ×
L 1
qt 1.06
= 2.12 qt
C) 24.95 min ×
min 60
hr 1
= 0.4158 hr
D) 12.0 ft ×
ft 1
in 12
×
in 1
cm 2.54
= 370 cm
E) 24.0 kg ×
kg 2.20
lb 1
= 11 lb
4) Which of the following measurements are NOT equivalent?
A) 25 mg = 0.025 g
B) 183 L = 0.183 kL
C) 150. msec = 0.150 sec
D) 84 cm = 8.4 mm
E) 24 dL = 2.4 L
5) Which of the following setups would convert centimeters to feet?
A) cm ×
cm 1
in. 2.54
×
in. 12
ft 1
B) cm ×
in. 1
cm 2.54
×
ft 1
in. 12
C) cm ×
cm 2.54
in. 1
×
in. 12
ft 1
D) cm ×
cm 2.54
in. 1
×
ft 1
in. 12
E) cm ×
cm 1
in. 2.54
×
in. 12
ft 1
6) A doctor's order is 0.125 g of ampicillin. The liquid suspension on hand contains 250 mg/5.0
mL. How many milliliters of the suspension are required?
A) 0.0025 mL
B) 3.0 mL
C) 2.5 mL
D) 6.3 mL
E) 0.0063 mL
7) A nugget of gold with a mass of 521 g is added to 50.0 mL of water. The water level rises to a
volume of 77.0 mL. What is the density of the gold?
A) 10.4 g/mL
B) 6.77 g/mL
C) 1.00 g/mL
D) 0.0518 g/mL
E) 19.3 g/mL
8) Which one of the following substances will float in gasoline, which has a density of 0.66
g/mL?
A) table salt (density = 2.16 g/mL)
B) balsa wood (density = 0.16 g/mL)
C) sugar (density = 1.59 g/mL)
D) aluminum (density = 2.70 g/mL)
E) mercury (density = 13.6 g/mL)
Match the type of measurement (#9-14 below) to the unit given, options A through E.
A) mass
B) volume
C) distance
D) temperature
E) density
9) milliliter_____
10) mm _____
11) gram _____
12) 125 K_____
13) kilometer_____
14) milligram_____
For #15-22, select the correct prefix to complete the equality.
A) 1000
B) 10
C) 1
D) 0.01
E) 1 × 10
12
F) 100
G) 0.001
H) 1 × 10
12
I) 1 × 10
-12
J) 0.1
15) 1 g = _____ kg
16) 1 m = _____ mm
17) 1 cm = _____ mm
18) 1 dL = __________ mL
19) 1 kg = _____ g
20) 1 pg = _____ g
21) 1 g = _____ pg
22) 1 mL = _____ cc
Chapter 2: Energy and Matter
23) When a solid is converted directly to a gas, the change of state is called
A) freezing.
B) melting.
C) boiling.
D) condensation.
E) sublimation.
24) In a gas, the distance between the particles is
A) very close relative to the size of the molecules.
B) close relative to the size of the molecules.
C) fixed relative to the size of the molecules.
D) small relative to the size of the molecules.
E) very large relative to the size of the molecules.
25) Helium is a(n)
A) compound.
B) heterogeneous mixture.
C) element.
D) homogeneous mixture.
E) electron.
26) Air is a(n)
A) compound.
B) heterogeneous mixture.
C) element.
D) homogeneous mixture.
E) None of the above.
27) Which of the following is a chemical change?
A) cutting a rope
B) bending a steel rod
C) making a snowman
D) burning sugar
E) melting gold
28) Which of the following is a physical change?
A) baking a cake
B) dry ice subliming
C) fermenting grapes to produce wine
D) digesting a meal
E) a tomato ripening
29) How many calories are required to raise the temperature of a 150. g sample of gold from 25
°C to 175 °C? The specific heat of gold is 0.0308 cal/g °C.
A) 4.62 cal
B) 116 cal
C) 22500 cal
D) 693 cal
E) 130 cal
For #30-38 below, identify each of the following transformations as a physical (A) or chemical
(B) change
A) physical
B) chemical
30) water evaporating
31) silver tarnishing
32) cutting the grass
33) a nail rusting
34) baking a cake
35) placing photographs in a scrapbook
36) formation of green leaves on a plant
37) burning leaves
38) melting ice
!
Chapter 3: Atoms and Elements
39) Which of the following elements is a metal?
A) nitrogen
B) fluorine
C) argon
D) strontium
E) phosphorus

40) Which of the following elements is a noble gas?
A) oxygen
B) chlorine
C) bromine
D) argon
E) nitrogen
41) The element in this list with chemical properties similar to magnesium is
A) sodium.
B) boron.
C) carbon.
D) strontium.
E) chlorine.
42) Which of the following descriptions of a subatomic particle is correct?
A) A proton has a positive charge and a mass of approximately 1 amu.
B) An electron has a negative charge and a mass of approximately 1 amu.
C) A neutron has no charge and its mass is negligible.
D) A proton has a positive charge and a negligible mass.
E) A neutron has a positive charge and a mass of approximately 1 amu.
43) The mass number of an atom can be calculated from
A) the number of electrons.
B) the number of protons plus neutrons.
C) the number of protons.
D) the number of electrons plus protons.
E) the number of neutrons.
44) The correct symbol for the isotope of potassium with 22 neutrons is
A) K.
B) K.
C) P.
D) P.
E) K.
45) Which of the following gives the correct numbers of protons, neutrons, and electrons in a
neutral atom of Sn?
A) 118 protons, 50 neutrons, 118 electrons
B) 118 protons, 118 neutrons, 50 electrons
C) 50 protons, 68 neutrons, 50 electrons
D) 68 protons, 68 neutrons, 50 electrons
E) 50 protons, 50 neutrons, 50 electrons
46) Isotopes are atoms of the same element that have
A) different atomic numbers.
B) the same atomic numbers but different numbers of protons.
C) the same atomic numbers but different numbers of electrons.
D) the same atomic number but different numbers of neutrons.
E) the same atomic mass but different numbers of protons.
47) A sample of chlorine has two naturally occurring isotopes. The isotope Cl-35 (mass 35.0
amu) makes up 75.8% of the sample, and the isotope Cl-37 (mass = 37.0 amu) makes up 24.3%
of the sample. What is the average atomic mass for chlorine?
A) 36.0 amu
B) 35 amu
C) 36.6 amu
D) 35.5 amu
E) 35.521 amu
48) A sample of silicon has three naturally occurring isotopes: Si-28 (mass 28.0 amu); Si-29
(mass 29.0 amu) and Si-30 (mass = 30.0 amu). If the average atomic mass of silicon is 28.1
amu, which isotope is the most abundant?
A) Si-28
B) Si-29
C) Si-30
D) All isotopes have the same natural abundance.
49) The number of electrons in the outer energy level of a neutral atom of boron (atomic number
5) is
A) 2.
B) 3.
C) 5.
D) 8.
E) 10.
50) What is the electron configuration for potassium (atomic number 19)?
A) 1s
2
2s
2
2p
6
3s
2
3p
7
B) 1s
2
2s
2
2p
6
3s
2
3p
5
3d
2
C) 1s
2
2s
2
2p
8
3s
2
3p
5
D) 1s
2
2s
2
2p
6
3s
2
3p
6
4s
1
E) 1s
2
2s
2
2p
6
3s
2
3p
5
4s
1
51) The number of dots in the electron dot structure of carbon is
A) one.
B) two.
C) three.
D) four.
E) five.

Classify the following elements (#52-57 below) with options A through F.
A) noble gas
B) alkali metal
C) nonmetal
D) alkaline earth metal
E) transition element
F) halogen
52) sodium
53) argon
54) bromine
55) copper
56) magnesium
57) phosphorus
Chapter 4: Nuclear Chemistry
58) What is the nuclear symbol for a radioactive isotope of copper with a mass number of 60?
A) Cu
B) Cu
C) 29Cu
D) Cu
E) Cu
59) The nuclear symbol of helium, He, is also the symbol for designating a(n)
A) proton.
B) neutron.
C) gamma ray.
D) beta particle.
E) alpha particle.
60) The symbol e is a symbol used for a(n)
A) proton.
B) neutron.
C) gamma ray.
D) beta particle.
E) alpha particle.

61) What is missing in the nuclear reaction shown below?
B + He → N + _____
A) gamma radiation
B) a positron
C) a neutron
D) an alpha particle
E) a beta particle
62) What is the radioactive particle released in the following nuclear equation?
W → Hf + ?
A) alpha particle
B) beta particle
C) gamma ray
D) proton
E) neutron
63) A sample of cerium-141 for a diagnostic test was dissolved in saline solution to an activity of
4.5 millicuries/mL. If the patient undergoing the test needs a dose of 10. millicuries, how much
of the solution should be injected into the patient?
A) 45 mL
B) .45 mL
C) 2.2 mL
D) 22 mL
E) 4.5 mL
64) The half-life of a radioisotope is
A) one-half of the time it takes for the radioisotope to completely decay to a nonradioactive
isotope.
B) the time it takes for the radioisotope to become an isotope with one-half of the atomic weight
of the original radioisotope.
C) the time it takes for the radioisotope to become an isotope with one-half the atomic number of
the original radioisotope.
D) the time it takes for the radioisotope to lose one-half of its neutrons.
E) the time it takes for one-half of the sample to decay.
65) Krypton-79 has a half-life of 35 hours. How many half-lives have passed after 105 hours?
A) 1 half-life
B) 2 half-lives
C) 3 half-lives
D) 4 half-lives
E) 5 half-lives
66) The half-life of bromine-74 is 25 min. How much of a 4.0 mg sample is still active after 75
min?
A) 0.50 mg
B) 1.0 mg
C) 2.0 mg
D) 0.25 mg
E) 4.0 mg
Chapter 5: Compounds and their Bonds
67) How many electrons will aluminum gain or lose when it forms an ion?
A) lose 1
B) gain 5
C) lose 2
D) lose 3
E) gain 1
68) What is the symbol for the ion with 19 protons and 18 electrons?
A) F
+
B) F
-
C) Ar
+
D) K
-
E) K
+
69) The correct formula for a compound formed from the elements Al and O is
A) AlO.
B) Al
2
O.
C) Al
3
O
2
.
D) AlO
3
.
E) Al
2
O
3
.
70) The compound MgCl
2
is named
A) magnesium chlorine.
B) magnesium dichloride.
C) magnesium (II) chloride.
D) magnesium chloride.
E) dimagnesium chloride.
71) Fe
2
(SO
4
)
3
is called
A) iron sulfate.
B) iron (II) sulfate.
C) iron (III) sulfate.
D) diiron trisulfate.
E) iron trisulfate.
72) Which of the following elements does NOT exist as a diatomic molecule?
A) hydrogen
B) nitrogen
C) chlorine
D) oxygen
E) carbon
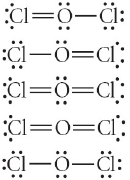
73) Choose the best electron-dot structure for OCl
2
.
A)
B)
C)
D)
E)
74) The correct name of the compound NCl
3
is
A) nitrogen chloride.
B) trinitrogen chloride
C) nitrogen(III) chloride.
D) nickel chloride.
E) nitrogen trichloride.
75) Which of the following substances contains a nonpolar covalent bond?
A) H
2
O
B) NaCl
C) NH
3
D) M
g
F
2
E) N
2
76) Which of the following compounds contains a polar covalent bond?
A) NaF
B) HCl
C) Br
2
D) MgO
E) O
2
77) Which of the following compounds contains an ionic bond?
A) NH
3
B) H
2
O
C) CaO
D) H
2
E) CH
4
78) If the electronegativity difference between elements X and Y is 2.1, the bond between the
elements X-Y is
A) ionic.
B) nonpolar ionic.
C) nonpolar covalent.
D) polar covalent.
E) impossible.
79) The shape of the ammonia molecule (NH
3
) is
A) linear.
B) square.
C) pyramidal.
D) hexagonal.
E) octagonal.
80) The shape of the carbon dioxide (CO
2
) is
A) linear.
B) square.
C) pyramidal.
D) hexagonal.
E) bent.
81) The strongest interactions between molecules of ammonia ( NH
3
) are
A) ionic bonds.
B) hydrogen bonds.
C) polar covalent.
D) dipole-dipole.
E) dispersion forces.
Match the chemical name (#82-86) with the correct formula A through E.
A) MgSO
4
B) Mg(HSO
4
)
2
C) MgS
D) MgSO
3
E) Mg(HSO
3
)
2
82) magnesium sulfate
83) magnesium hydrogen sulfate
84) magnesium sulfide
85) magnesium sulfite
86) magnesium hydrogen sulfite
Identify each of the following molecules (#87-92) as polar (A) or nonpolar (B).
87) carbon tetrachloride
88) water
89) carbon dioxide
90) hydrogen sulfide
91) hydrogen fluoride
92) carbon monoxide
Chapter 6: Chemical Reactions and Quantities
93) Which of the following gives the balanced equation for this reaction?
K
3
PO
4
+ Ca(NO
3
)
2
→ Ca
3
(PO
4
)
2
+ KNO
3
A) KPO
4
+ CaNO
3
+ KNO
3
B) K
3
PO
4
+ Ca(NO
3
)
2
→ Ca
3
(PO
4
)
2
+ 3KNO
3
C) 2K
3
PO
4
+ Ca(NO
3
)
2
→ Ca
3
(PO
4
)
2
+ 6KNO
3
D) 2K
3
PO
4
+ 3Ca(NO
3
)
2
→ Ca
3
(PO
4
)
2
+ 6KNO
3
E) K
3
PO
4
+ Ca(NO
3
)
2
→ Ca
3
(PO
4
)
2
+ KNO
3
94) The following reaction takes place when an electric current is passed through water. It is an
example of a __________ reaction.
2 H
2
O → 2H
2
+ O
2
A) combination
B) single replacement
C) dehydration
D) decomposition
E) double replacement
95) Which of the following is an oxidation-reduction reaction?
A) CaCl
2
+ Na
2
SO
4
→ CaSO
4
+ 2NaCl
B) KOH + HNO
3
→ H
2
O + KNO
3
C) N
2
+ O
2
→ 2NO
D) AgNO
3
+ NaCl → AgCl + NaNO
3
E) Al
4
(SO
4
)
3
+ 6KOH → 2 Al(OH)
3
+ 3K
2
SO
4
96) What is oxidized and what is reduced in the following reaction?
2Al(s) + 3Br
2
(g) → 2AlBr
3
(s)
A) Al is oxidized and Br
2
is reduced.
B) AlBr
3
is reduced and Br
2
is oxidized.
C) Al is reduced and Br
2
is oxidized.
D) AlBr
3
is reduced and Al is oxidized.
E) AlBr
3
is oxidized and Al is reduced.
97) Which of the following describes an oxidation reaction?
A) loss of electrons or loss of oxygen
B) loss of electrons or gain of oxygen
C) loss of electrons or gain of hydrogen
D) gain of electrons or gain of oxygen
E) gain of electrons or loss of H

98) How many molecules of water, H
2
O, are present in 75.0 g of H
2
O?
A) 75.0 molecules
B) 4.17 molecules
C) 7.53 × 10
24
molecules
D) 2.51 × 10
24
molecules
E) 5.02 × 10
24
molecules
99) Avogadro's number is the number of
A) particles in 1 mole of a substance.
B) amu in 1 mole of a substance.
C) grams in 1 mole of a substance.
D) moles in 6.02 × 10
23
grams of an element.
E) moles in 6.02 × 10
23
amu of an element.
100) One mole of helium gas weighs
A) 1.00 g.
B) 2.00 g.
C) 3.00 g.
D) 4.00 g.
E) 8.00 g.
101) What is the molar mass of Mg
3
(PO
4
)
2
, a substance formerly used in medicine as an
antacid?
A) 71.3 g
B) 118.3 g
C) 150.3 g
D) 214.3 g
E) 262.9 g
102) Given the following equation, what is the correct form of the conversion factor needed to
convert the number of moles of O
2
to the number of moles of Fe
2
O
3
produced?
4Fe(s) + 3O
2
(g) → 2Fe
2
O
3
(s)
A)
O
2
of moles 3
F
e
of moles 4
B)
O
3
Fe
2
of moles 3
F
e
of moles 4
C)
O
3
Fe
2
of moles 3
O
2
of moles 3
D)
Fe
of moles 4
O
3
F
e2
of moles 2
E)
O
2
of moles 3
O
3
F
e2
of moles 2
For questions #103-105, consider the following equation.
2Mg + O
2
→ 2MgO
103) The number of moles of oxygen gas needed to react with 4.0 moles of Mg is
A) 1.0 mole.
B) 2.0 moles.
C) 3.0 moles.
D) 4.0 moles.
E) 6.0 moles.
104) The number of moles of MgO produced when 0.20 mole of O
2
reacts completely is
A) 0.10 mole.
B) 0.20 mole.
C) 0.40 mole.
D) 0.60 mole.
E) 0.80 mole.
105) How many moles of magnesium are needed to react with 0.50 mole of O
2
?
A) 0.50 mole
B) 1.0 moles
C) 2.0 moles
D) 3.0 moles
E) 4.0 moles
106) When 4 moles of aluminum are allowed to react with an excess of chlorine gas, Cl
2
, how
many moles of aluminum chloride are produced?
A) 1 mole
B) 2 moles
C) 3 moles
D) 4 moles
E) 5 moles
107) How many grams of NO are required to produce 145 g of N
2
in the following reaction?
4NH
3
(g) + 6NO(g) → 5N
2
(g) + 6H
2
O(l)
A) 186 g
B) 155 g
C) 125 g
D) 129 g
E) 145 g
108) The __________ is the minimum energy needed for a chemical reaction to begin.
A) reaction energy
B) activation energy
C) energy of reactants
D) energy of products
E) heat of reaction
109) A reaction that releases energy as it occurs is classified as a(n) __________.
A) endothermic reaction
B) exothermic reaction
C) oxidation-reduction reaction
D) catalyzed reaction
E) decomposition reaction
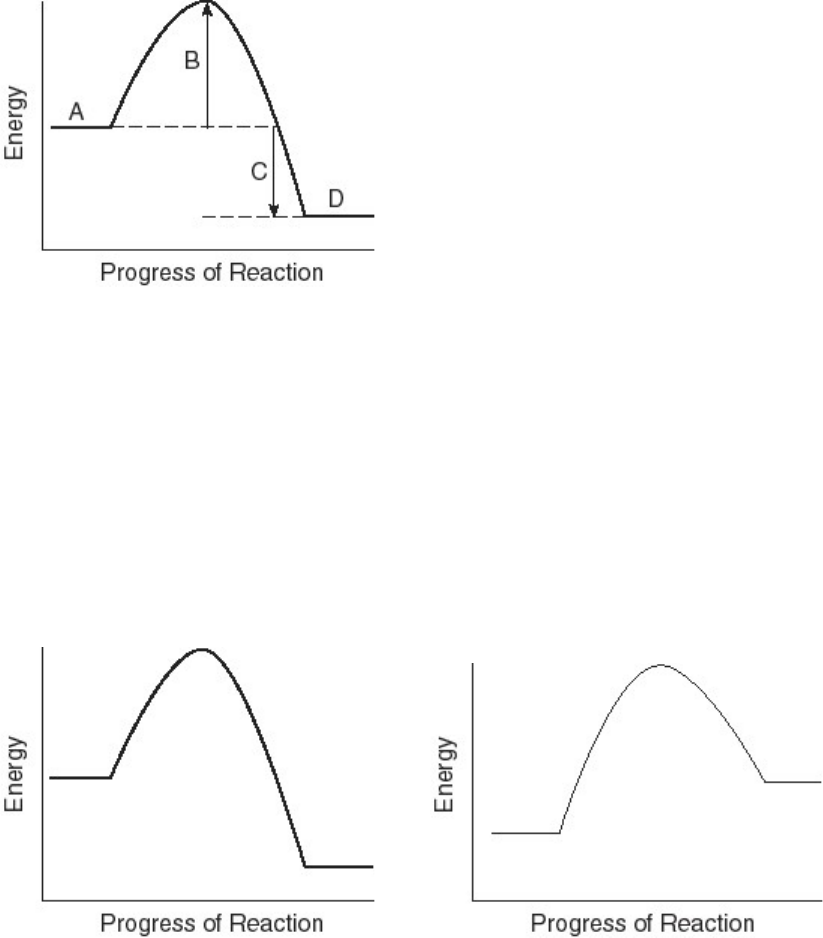
Identify the energy (A thorugh D) associated with each of the labeled parts (#110-114) of the
following diagram.
A) heat of reaction
B) activation energy
C) energy of reactants
D) energy of products
110) Region A
111) Region B
112) Region C
113) Region D
Identify the letter of the diagram corresponding to the given type of reaction.
A B
114) endothermic reaction (A or B)
115) exothermic reaction (A or B)
Chapter 7: Gases
This chapter is not covered by many instructors. Wrong answers to questions on gases on the
exam will not be counted against you.
Chapter 8: Solutions
116) Which of the following molecules can form hydrogen bonds?
A) CH
4
!
B) NaH
C) NH
3
D) BH
3
E) HI
117) A solution is prepared by dissolving 2 g of KCl in 100 g of H
2
O. In this solution, H
2
O is
the
A) solute.
B) solvent.
C) solution.
D) solid.
E) ionic compound.
118) Oil does not dissolve in water because
A) oil is polar.
B) oil is nonpolar.
C) water is nonpolar.
D) water is saturated.
E) oil is hydrated.
119) When KCl dissolves in water
A) the Cl
-
ions are attracted to dissolved K
+
ions.
B) the Cl
-
ions are attracted to the partially negative oxygen atoms of the water molecule.
C) the K
+
ions are attracted to Cl
-
ions on the KCl crystal.
D) the K
+
ions are attracted to the partially negative oxygen atoms of the water molecule.
E) the K
+
ions are attracted to the partially positive hydrogen atoms of the water molecule.
120) Which one of the following compounds will NOT be soluble in water?
A) NaOH
B) PbS
C) K
2
SO
4
D) LiNO
3
E) MgCl
2
121) A hydrogen bond is
A) an attraction between a hydrogen atom attached to N, O, or F and an N, O, or F atom on
another molecule.
B) a covalent bond between H and O.
C) an ionic bond between H and another atom.
D) a bond that is stronger than a covalent bond.
E) the polar O-H bond in water.
122) What is the concentration, in mass percent, of a solution prepared from 50.0 g NaCl and
150.0 g of water?
A) 0.250%
B) 33.3%
C) 40.0%
D) 25.0%
E) 3.00%
123) Rubbing alcohol is 70.% isopropyl alcohol by volume. How many mL of isopropyl alcohol
are in a 1 pint (473 mL) container?
A) 70. mL
B) 0.15 mL
C) 680 mL
D) 470 mL
E) 330 mL
124) What is the concentration, in m/v percent, of a solution prepared from 50. g NaCl and 2.5 L
of water?
A) 5.0%
B) 2.0%
C) 0.020%
D) 0.050%
E) 20.%
125) How many grams of glucose are needed to prepare 400. mL of a 2.0%(m/v) glucose
solution?
A) 800. g
B) 0.0050 g
C) 8.0 g
D) 2.0 g
E) 200. g
126) A patient needs to receive 85 grams of glucose every 12 hours. What volume of a
5.0%(m/v) glucose solution needs to be administered to the patient each 12 hours?
A) 1700 mL
B) 60 mL
C) 6000 mL
D) 17 mL
E) 204 mL
127) How many milliliters of a 25% (m/v) NaOH solution would contain 75 g of NaOH?
A) 25 mL
B) 75 mL
C) 33 mL
D) 19 mL
E) 3.0 × 10
2
mL
128) What is the molarity of a solution that contains 17 g of NH
3
in 0.50 L of solution?
A) 34 M
B) 2.0 M
C) 0.50 M
D) 0.029 M
E) 1.0 M
129) When 200. mL of water are added to 100. mL of 12% KCl solution the final concentration
of KCl is (Assume the volumes add.)
A) 12%.
B) 4.0%.
C) 36%.
D) 6.0%.
E) 8.0%.
130) How many moles of CaCl
2
are in 250 mL of a 3.0 M of CaCl
2
solution?
A) 750 moles
B) 1.3 moles
C) 83 moles
D) 0.75 mole
E) 3.0 moles
131) What volume of a 1.5 M KOH solution is needed to provide 3.0 moles of KOH?
A) 3.0 L
B) 0.50 L
C) 2.0 L
D) 4.5 L
E) 0.22 L
132) During the process of diluting a solution to a lower concentration,
A) the amount of solute does not change.
B) the amount of solvent does not change.
C) there is more solute in the concentrated solution.
D) the volume of the solution does not change.
E) water is removed from the concentrated solution.
Chapter 9: Reaction Rates and Chemical Equilibrium
133) In any chemical reaction, the rate of the reaction can be increased by
A) decreasing the temperature.
B) changing the size of the container.
C) adding water to the reaction.
D) adding product molecules to the reaction mixture.
E) increasing the concentrations of the reactants.
134) A catalyst is
A) a reactant in a chemical reaction.
B) a product in a chemical reaction.
C) a substance that speeds up a reaction without being consumed in the reaction.
D) a substance that increases the energy of the products.
E) a substance that decreases the energy of the products.
135) The activation energy of a chemical reaction is the energy that
A) must be removed from the mixture.
B) must be released from the mixture.
C) initiates the reaction.
D) activates the catalyst.
E) is the difference in the energies of the starting materials and products.
136) In a catalyzed chemical reaction, one function of a catalyst is to
A) increase the number of successful reactant collisions.
B) decrease the concentration of reactants.
C) change the equilibrium concentrations of the products and reactants.
D) increase the energy given off during the reaction.
E) increase the temperature at which the reaction is carried out.
Indicate the effect of each change (#138) upon the rate of a reaction (increases, A or decreases,
B).
137) adding a catalyst
138) removing some reactant
139) the temperature is doubled.
140) The concentration of a reactant is decreased.
141) More collisions between molecules occur.
Chapter 10: Acids and Bases
142) According to the Arrhenius concept, if NaOH were dissolved in water, it would act as
A) a base.
B) an acid.
C) a source of hydronium ions.
D) a source of H
-
ions.
E) a proton donor.
143) According to the Arrhenius concept, if HNO
3
were dissolved in water, it would act as
A) a base.
B) an acid.
C) a source of hydroxide ions.
D) a source of H
-
ions.
E) a proton acceptor.
144) The name given to an aqueous solution of HBr is
A) hydrogen bromide.
B) hydrobromic acid.
C) bromic acid.
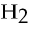
D) bromous acid.
E) hypobromous acid.
145) The name given to an aqueous solution of HNO
3
is
A) nitric acid.
B) nitrous acid.
C) hydrogen nitrate.
D) hydronitrogen acid.
E) hyponitric acid.
146) The conjugate base of HClO
3
is
A) HClO
2
.
B) ClO
3
-
.
C) Cl(OH)
2
.
D) ClO
3
.
E) HClO.
147) The conjugate acid of HSO
4
-
is
A) SO
4
2-
.
B) HSO
4
.
C) H
2
SO
4
.
D) H
2
SO
4
-
.
E) HSO
3
-
.
148) Which of the following statements correctly describes the hydronium-hydroxide balance in
the given solution?
A) In acids, [OH
-
] is greater than [H
3
O
+
].
B) In bases, [OH
-
] = [H
3
O
+
].
C) In neutral solutions, [H
3
O
+
] = [ O].
D) In bases, [OH
-
] is greater than [H
3
O
+
].
E) In bases, [OH
-
] is less than [H
3
O
+
].
149) What is the [H
3
O
+
] in a solution with [OH
-
] = 1 × 10
-12
M?
A) 1 × 10
-12
M
B) 1 × 10
2
M
C) 1 × 10
-7
M
D) 1 × 10
-8
M
E) 1 × 10
-2
M
150) What is the [OH
-
] in a solution that has a [H
3
O
+
] = 1 × 10
-6
M?
A) 1 × 10
-2
M
B) 1 × 10
-6
M
C) 1 × 10
-8
M
D) 1 × 10
-10
M
E) 1 × 10
-12
M
151) What is the pH of a solution with [OH
-
] = 1 × 10
-4
M?
A) 10.0
B) -10.0
C) 4.0
D) -4.0
E) 1.0 × 10
-10
152) The [H
3
O
+
] of a solution with pH = 2 is
A) 10 M.
B) -10 M.
C) 1 × 10
2
M.
D) 1 × 10
-2
M.
E) 1 × 10
-12
M.
153) Which of the following is the correctly balanced equation for the complete neutralization of
H
3
PO
4
with Ca(OH)
2
?
A) H
3
PO
4
+ Ca(OH)
2
→ CaHPO
4
+ 2H
2
O
B) 3H
3
PO
4
+ Ca(OH)
2
→ Ca
3
(PO
4
)
2
+ 5H
2
O
C) H
3
PO
4
+ Ca(OH)
2
→ Ca
3
(PO
4
)
2
+ H
2
O
D) 2H
3
PO
4
+ 3Ca(OH)
2
→ Ca
3
(PO
4
)
2
+ 6H
2
O
E) 4H
3
PO
4
+ 6Ca(OH)
2
→ 2Ca
3
(PO
4
)
2
+ 12H
3
O
154) The neutralization reaction between Al(OH)
3
and HNO
3
produces the salt with the formula
A) H
2
O.
B) AlNO
3
.
C) AlH
2
.
D) Al(NO
3
)
3
.
E) NO
3
OH.
155) Which of the following is a neutralization reaction?
A) KCl + NaNO
3
→ KNO
3
+ NaCl
B) HNO
3
+ KOH → H
2
O + KNO
3
C) H
2
O + SO
3
→ H
2
SO
4
D) 4Na + O
2
→ 2Na
2
O
E) 2NO
2
→ 2NO + O
2
156) The function of a buffer is to
A) change color at the end point of a titration.
B) maintain the pH of a solution.
C) be a strong base.
D) maintain a neutral pH.
E) act as a strong acid.
157) In a buffer system of HF and its salt, NaF,
A) the HF neutralizes added acid.
B) the HF neutralizes added base.
C) the HF is not necessary.
D) the F
-
neutralizes added H
2
O.
E) the F
-
neutralizes added base.
158) Which of the following is a buffer system?
A) NaCl and NaNO
3
B) HCl and NaOH
C) H
2
CO
3
and KHCO
3
D) NaCl and NaOH
E) H
2
O and HCl
Matching Questions
Identify each of the following compounds as an acid (A), a base (B), or neither (C).
159) HCl
160) NaOH
161) NH
3
162) H
2
SO
4
163) CO
3
2-
164) NaCl
165) CN
-
166) H
2
CO
3
In the following solutions, is the [OH-] greater than, less than, or equal to the [H
3
O
+
]?
A) greater than
B) equal to
C) less than
167) acid
168) base
169) [H
3
O
+
] = 1.0 × 10
-6
M
170) [H
3
O
+
] = 1.0 × 10
-10
M
171) [H
3
O
+
] = 1.0 × 10
-7
M
172) pH = 2
173) pH = 9
Identify the following as acids (A), bases (B), or neutral (C) solutions.
174) has a sour taste
175) has a pH = 4.5
176) turns blue litmus paper red
177) contains more hydronium ions than hydroxide ions
178) H
2
O
179) [H
3
O
+
] = 3.4 × 10
-5
M
180) [OH
-
] = 2.8 × 10
-2
M
181) Ca(OH)
2
182) pH =9.0
183) [H
3
O
+
] = 1.0 × 10
-7
M
Answers:
1) B
2) C
3) C
4) C
5) C
6) C
7) E
8) B
9) B
10) C
11) A
12) D
13) C
14) A
15) G
16) A
17) B
18) F
19) A
20) I
21) E
22) C
23) E
24) E
25) C
26) D
27) D
28) B
29) D
30) A
31) B
32) A
33) B
34) B
35) A
36) B
37) B
38) A
39) D
40) D
41) D
42) A
43) B
44) A
45) C
46) D
47) D
48) A
49) B
50) D
51) D
52) B
53) A
54) F
55) E
56) D
57) C
58) A
59) E
60) D
61) C
62) A
63) C
64) E
65) C
66) A
67) D
68) E
69) E
70) D
71) C
72) E
73) E
74) E
75) E
76) B
77) C
78) A
79) C
80) A
81) B
82) A
83) B
84) C
85) D
86) E
87) B
88) A
89) B
90) A
91) A
92) A
93) D
94) D
95) C
96) A
97) B
98) D
99) A
100) D
101) E
102) E
103) B
104) C
105) B
106) D
107) A
108) B
109) B
110) C
111) B
112) A
113) D
114) B
115) A
116) C
117) B
118) B
119) D
120) B
121) A
122) D
123) E
124) B
125) C
126) A
127) E
128) B
129) B
130) D
131) C
132) A
133) E
134) C
135) C
136) A
137) A
138) B
139) A
140) B
141) A
142) A
143) B
144) B
145) A
146) B
147) C
148) D
149) E
150) C
151) A
152) D
153) D
154) D
155) B
156) B
157) B
158) C
159) A
160) B
161) B
162) A
163) B
164) C
165) B
166) A
167) C
168) A
169) C
170) A
171) B
172) C
173) A
174) A
175) A
176) A
177) A
178) C
179) A
180) B
181) B
182) B
183) C
!
!
