
Page 1 of 5
EPIC UPGRADE NOVEMBER 2017
RESEARCHER NOTES FOR STUDY-RELATED IMAGING
Use of the current paper Radiology Research Order forms will be discontinued in conjunction with EPIC go-live.
After EPIC go-live, there will no longer be separate research orderables for imaging exams. After EPIC go-live, all
Radiology research orders must be placed electronically in eStar by the research team and MUST be
linked/associated to the trial. These “linked” research imaging orders will be supplemented with Clinical Trial
Imaging Instructions (CTII) forms as needed.
The Clinical Trial Imaging Instructions (CTII) forms will only be created by Radiology for trials that involve an imaging
component with sponsor-paid imaging and/or special imaging guidelines.
The Clinical Trial Imaging Instructions (CTII) forms will contain the same pertinent information as the former Radiology
research “order” forms: scheduling guidelines; exam types, technical parameters.
RECOMMENDED imaging exams to be ordered by clinician are listed on Clinical Trial Imaging Instructions (CTII) forms.
These recommendations are based on Radiology review of the study documents provided by the research team. If
the desired imaging exam is not listed under the “Recommended Imaging Exams” section of the Clinical Trials Imaging
Instructions (CTII) form, that would indicate that Radiology is not aware that the exam is included in the trial. Proceed
with ordering and scheduling the desired exam. Exam will then be performed according to departmental guidelines.
Researcher should contact the Clinical Trials Radiology Support Core to ensure that any changes to study protocol and
imaging exams are fully reviewed by Radiology staff.
Located at the top of each Clinical Trial Imaging Instructions (CTII) form is a three or four digit number used to identify
the trial within the Radiology department computer systems. This number will be sent to the RIS system (Imagecast)
automatically from EPIC when patient is “arrived” by Radiology PSRs for imaging exam.
Please note that EPIC does not allow for ordering of special research exams. Exams must be ordered using a list of
standard orderables developed by the EPIC team. Researchers must pay close attention to Clinical Trial Imaging
Instructions (CTII) regarding any imaging exams that require additional order “comments” to be entered in eStar by
the researcher. (Example: There is no option to order a PET scan with investigational tracer. You must enter an order
for routine PET scan, and add “comment” to order describing what investigational tracer should be used for the
exam.) Linking the order and visit to the research study will help ensure that imaging is acquired per study protocol.
Many trials have imaging exams that are designated as “no-read” (no VUMC Radiologist dictation/report), or a
mixture of “read” and “no-read” imaging exams (some exams are dictated, some are not). Clinical Trial Imaging
Instructions (CTII) forms will indicate if your trial involves “no-read” or “mixed-read” imaging exams.
Any currently scheduled Starpanel imaging exams (both Standard of Care and research) are being converted to
electronic eStar orders by Radiology staff prior to EPIC go-live.
Prior to EPIC go-live: For current/active trials, any Clinical Trial Imaging Instructions (CTII) forms will be emailed to
study coordinators and accessible via a hyperlink within eStar.
After EPIC go-live: For new trials, after Radiology review has been completed, Clinical Trial Imaging Instructions (CTII)
forms will be emailed to study coordinators and accessible via a hyperlink within eStar prior to patient accrual.
NOTE: These changes do NOT in any way affect the current Clinical Trials Radiology Support Core “service request”
workflow via iLab.
Page 2 of 5
New workflow for ordering study-related imaging exams
1) Prepare for the study-related imaging encounter in eStar and place order
o Ensure the patient is associated to the research study with an active status in eStar
(Active statuses include: In Screening Consented; On Study; Long-term Follow-up, and when
scheduling is required prior to consenting, Scheduling-Pending Consent.)
o Check Clinical Trial Imaging Instructions (CTII) for ordering and scheduling guidelines
o Place clinical trial-related imaging orders in eStar
o If directed by Clinical Trials Imaging Instructions (CTII) form, please enter any necessary order
“comments” (IND contrast or tracer indication, extremity region of interest, etc.)
o Link imaging orders to the research study in eStar before signing the order. Imaging payor will
be based on Billing Protocol information in eStar and eSMART/OnCore.
o Link all research related orders to research study regardless of payor
o A credentialed provider must electronically sign order(s) before they are activated/scheduled
(i.e., a second signature if the order was entered by a non-credentialed staff member)
o Research flask/link icon appears next to order and the encounter in eStar
2) Ensure the Patient Services Representative (PSR) links the imaging visit/encounter to the patient
timeline in eStar and schedule imaging exam(s)
o Contact scheduling.
o Scheduler will see research flask/link icon next to order.
o Scheduler can access CTII link via study info in eStar as needed.
o It is the study team’s responsibility to ensure the encounter/visit is linked.
o You must provide the visit name to the PSR so they can link the visit accurately.
3) Patient arrives for imaging appointment
o Clinical trial imaging exams will now be identified in the RIS system (Imagecast) by a three or
four digit research “modifier” number. This “modifier” number will match the CTII identification
number.
o Radiology PSR team will “arrive” the patient in EPIC (this will automatically arrive in Imagecast)
o Radiology will see CTII# associated with clinical trial in Imagecast when patient is “arrived”
o Radiology staff can hover over the RIS/Imagecast research modifier number to identify name of
clinical trial (or open RIS/Imagecast entry for full view)
o Any information entered in the order “comments” field by researcher will appear in the
comment section of the Imagecast entry.
o Radiology staff will see research flask/link icon next to the eStar order.
o To ensure any study-specific imaging guidelines are followed, technologists will review CTII
form via eStar hyperlink and/or use departmental research records regarding special imaging
acquisition guidelines for the trial.
Page 3 of 5
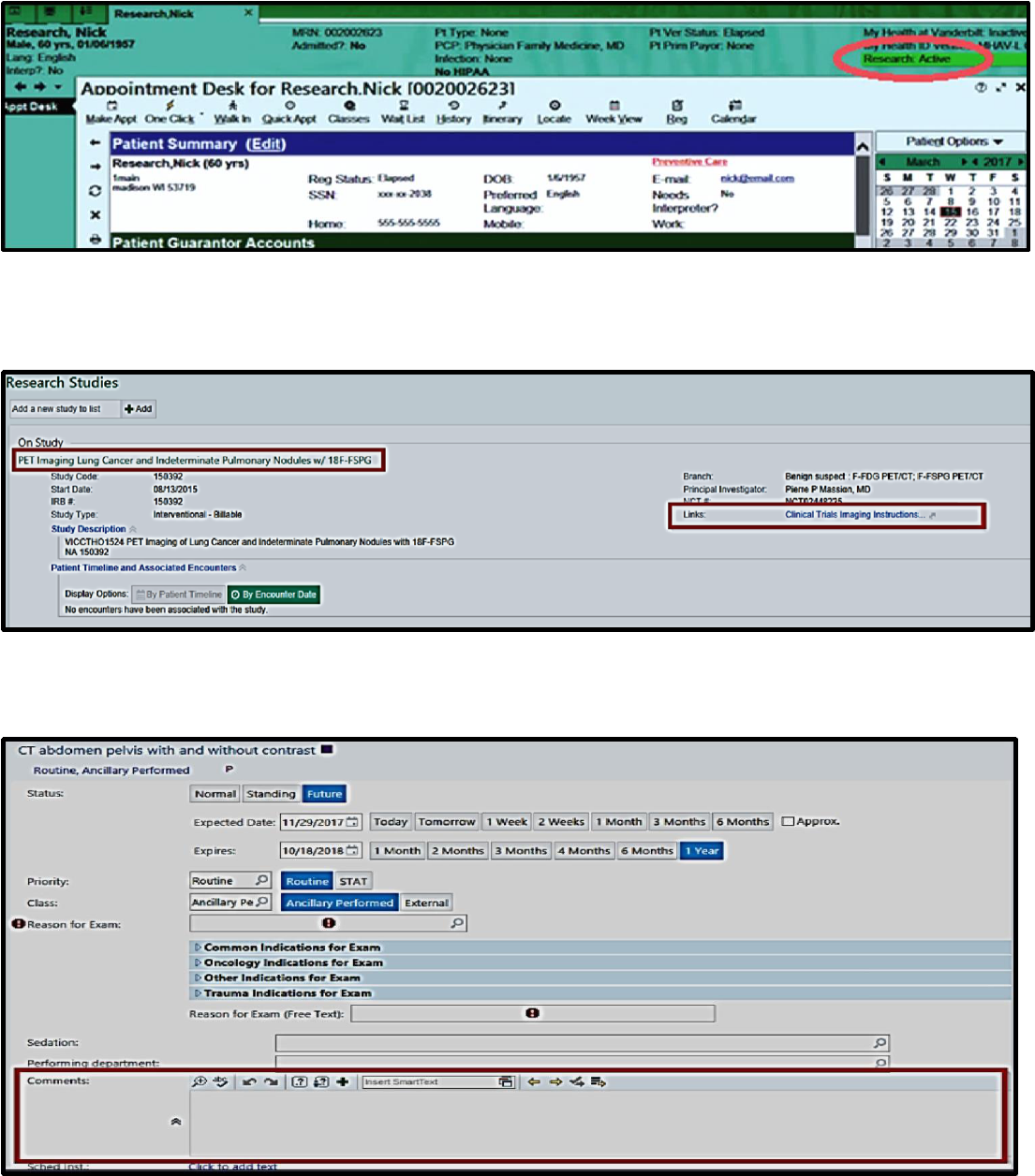
To access the Clinical Trial Imaging Instructions (CTII) form in eStar, click on the “Research: Active” section in
patient banner:
When eStar study information tool opens, please confirm the name of the clinical trial and click on the Clinical
Trials Imaging Instructions (CTII) form hyperlink:
The eStar order “comments” field:

Page 4 of 5
Example of new Clinical Trials Imaging Instructions form:
Prior to EPIC go-live: Clinical Trials Imaging Instructions (CTII) forms for current/active trials will be emailed to
study coordinators prior to go-live for all current/active trials, and will be available via hyperlink in eStar study
tool.
After EPIC go-live: After completion of Radiology review, Clinical Trials Imaging Instructions (CTII) forms for
pending or new trials will be emailed to all study coordinators prior to patient accrual, and will be available via
hyperlink in eStar study tool.
Page 5 of 5
Radiology staff point-of-view
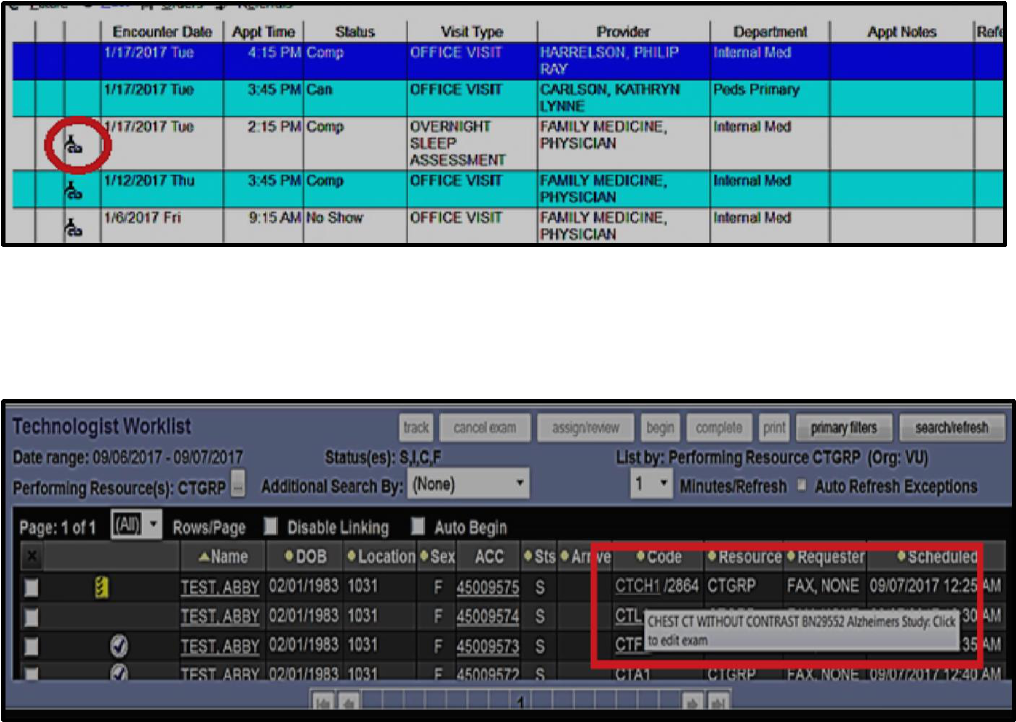
Exams associated with research studies will be identified in eStar with research flask/link icon:
Below is the RIS system (Imagecast) view of imaging exam linked to research study:
Radiology staff will see an imaging exam code followed by three or four digit “modifier” in
Imagecast. Please note that the Imagecast “modifier” is not a billing modifier, it is only
designed to be a research flag for Radiology staff. Radiology staff can hover over the research
“modifier” number in the Imagecast worklist to show the full name of the clinical trial, or open
full view of Imagecast entry.
Radiology staff can access the Clinical Trials Imaging Instructions (CTII) form via the hyperlink
in eStar study page/tool to see any technical instructions for each trial, and/or use
departmental research records regarding special imaging acquisition guidelines for the trial.
