CHAPTER 6 NOTES: The
Periodic Table
NOTES: 6.1-6.2
The Periodic Table –
Organizing the Elements

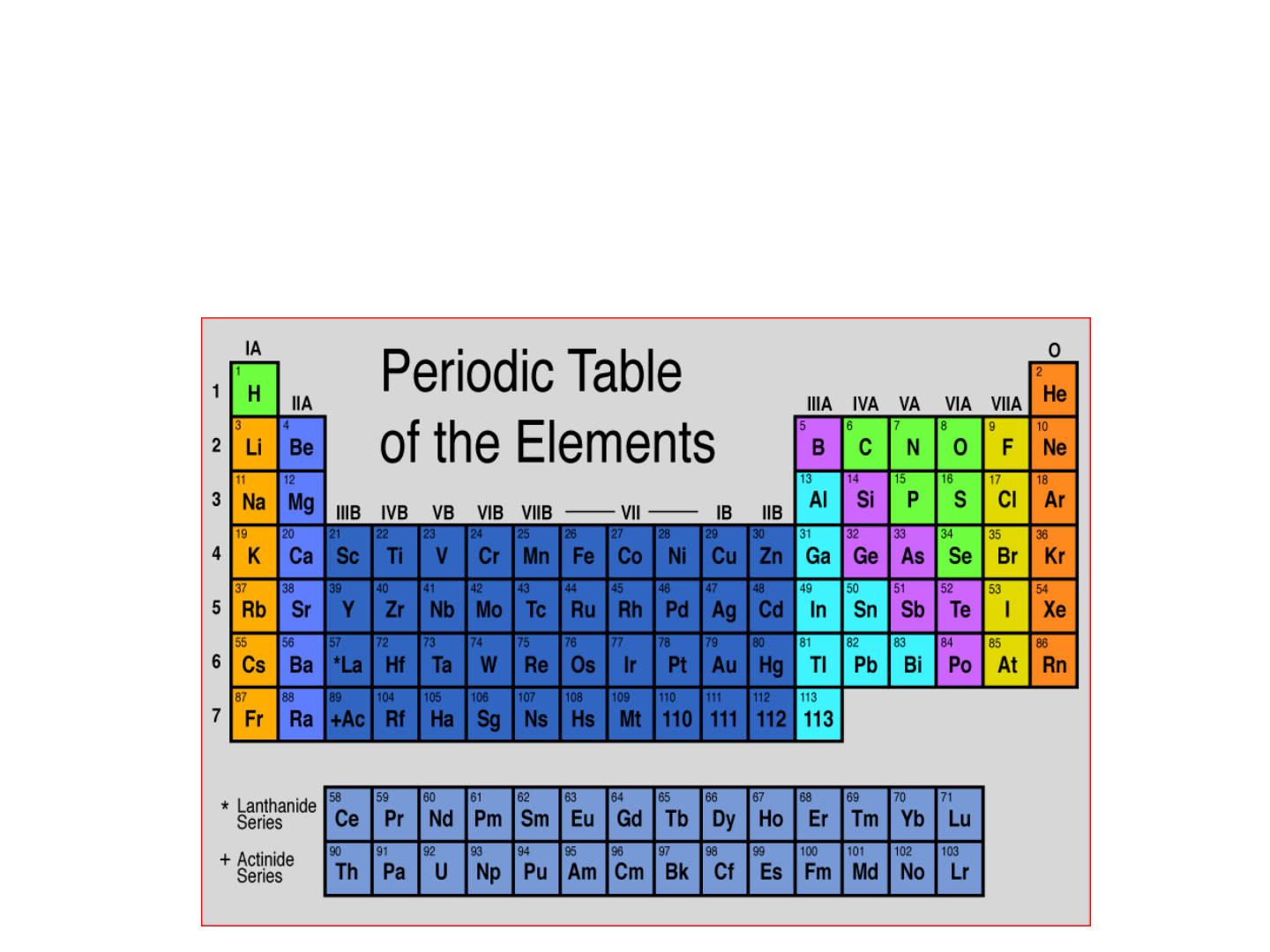
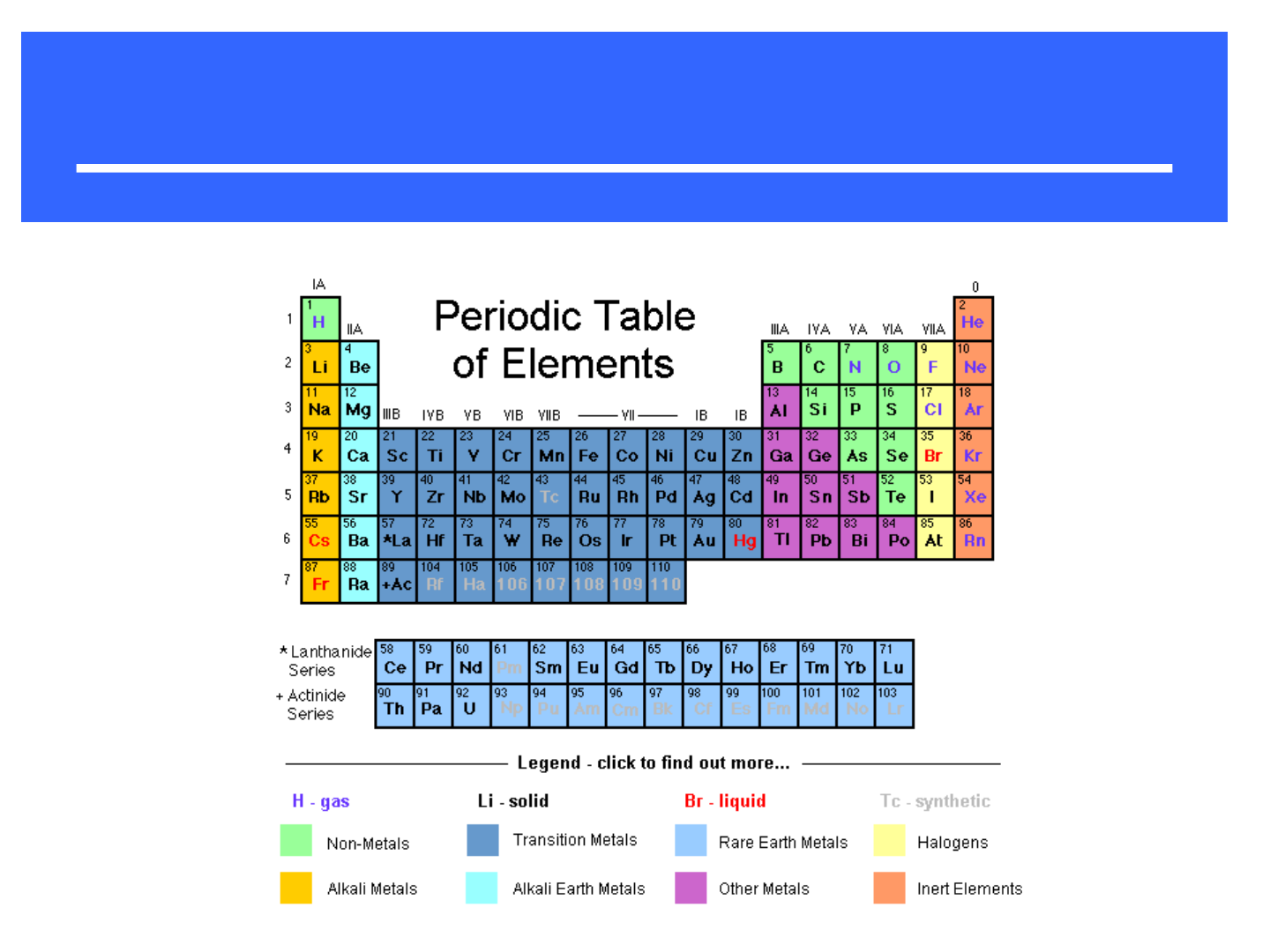
PERIODIC TABLE:
● Dmitri Mendeleev – mid
1800’s
-proposed a table for 70
elements based on increasing
mass and similar properties
● Henry Moseley – 1913
-determined the atomic number
of elements and arranged the
table in order of increasing
atomic number

Periodic Table
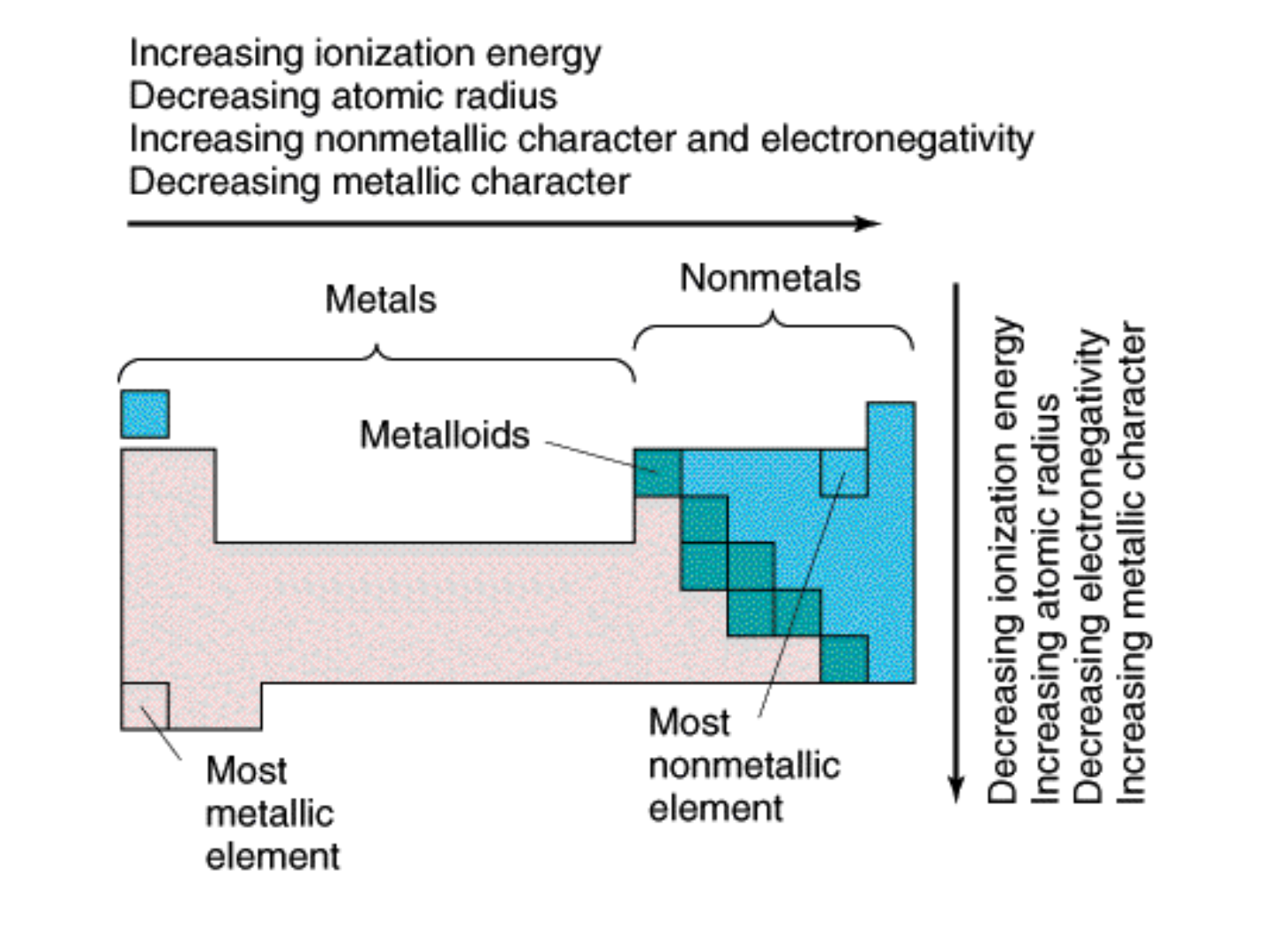
● Periodicity: (a.k.a. “PERIODIC LAW”)
-regular variations (or patterns) of properties
with increasing atomic weight; both chemical
and physical properties vary in a “periodic” way
(repeating pattern).
● Group: vertical column of elements (“family”)
● Period: horizontal row of elements
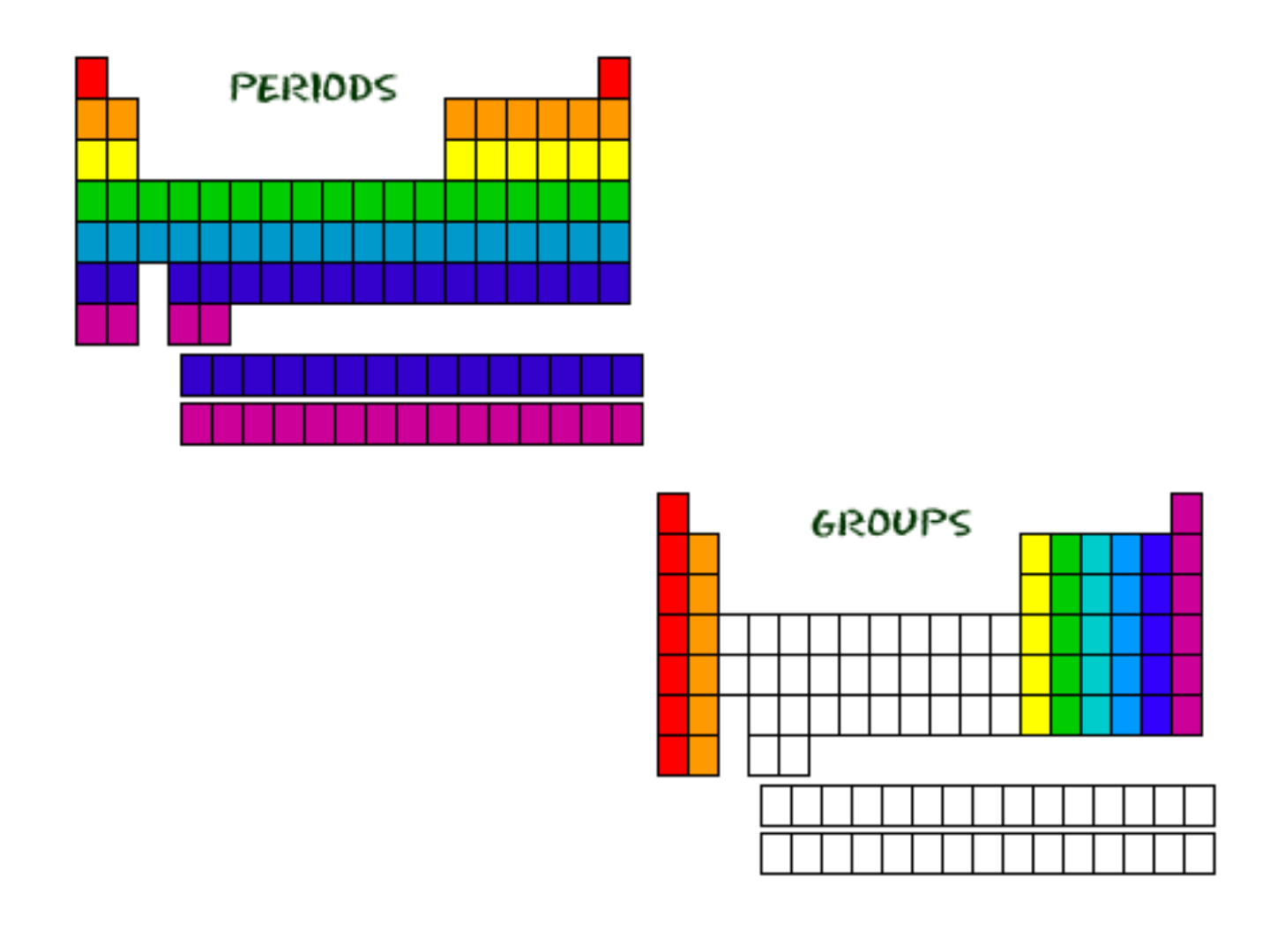
Periodic Groups and Trends
Groupings to know on the
Periodic Table
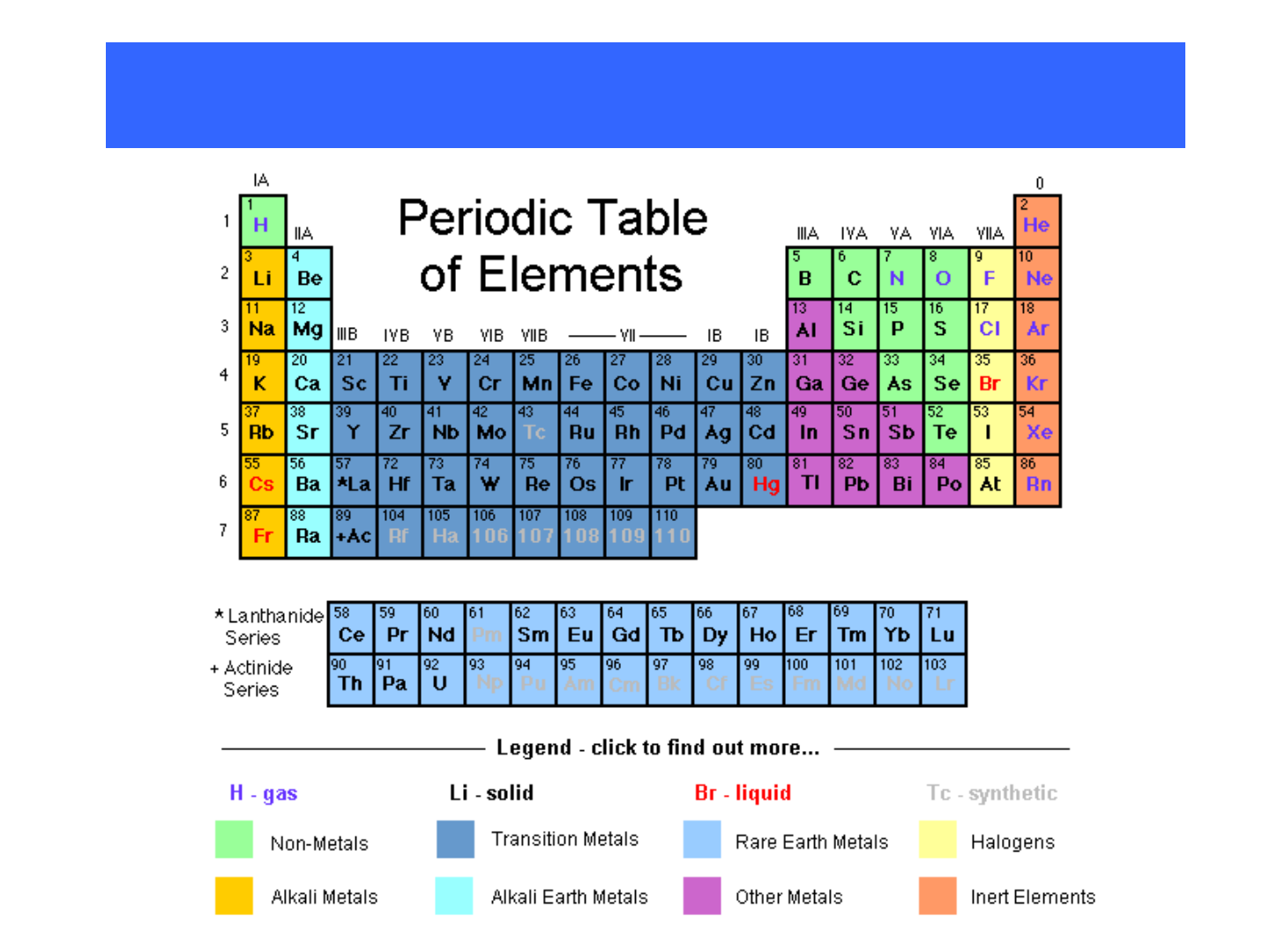
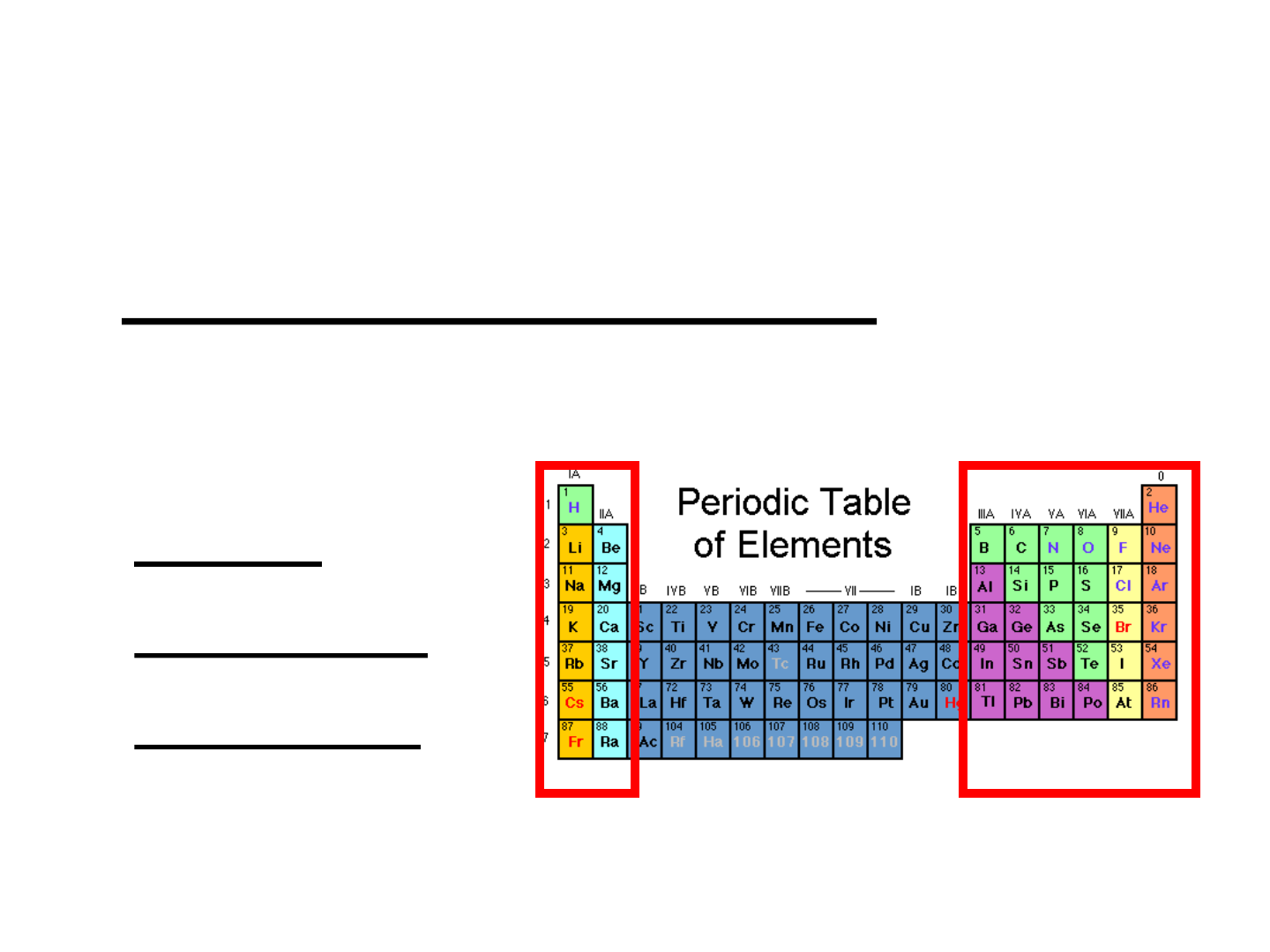
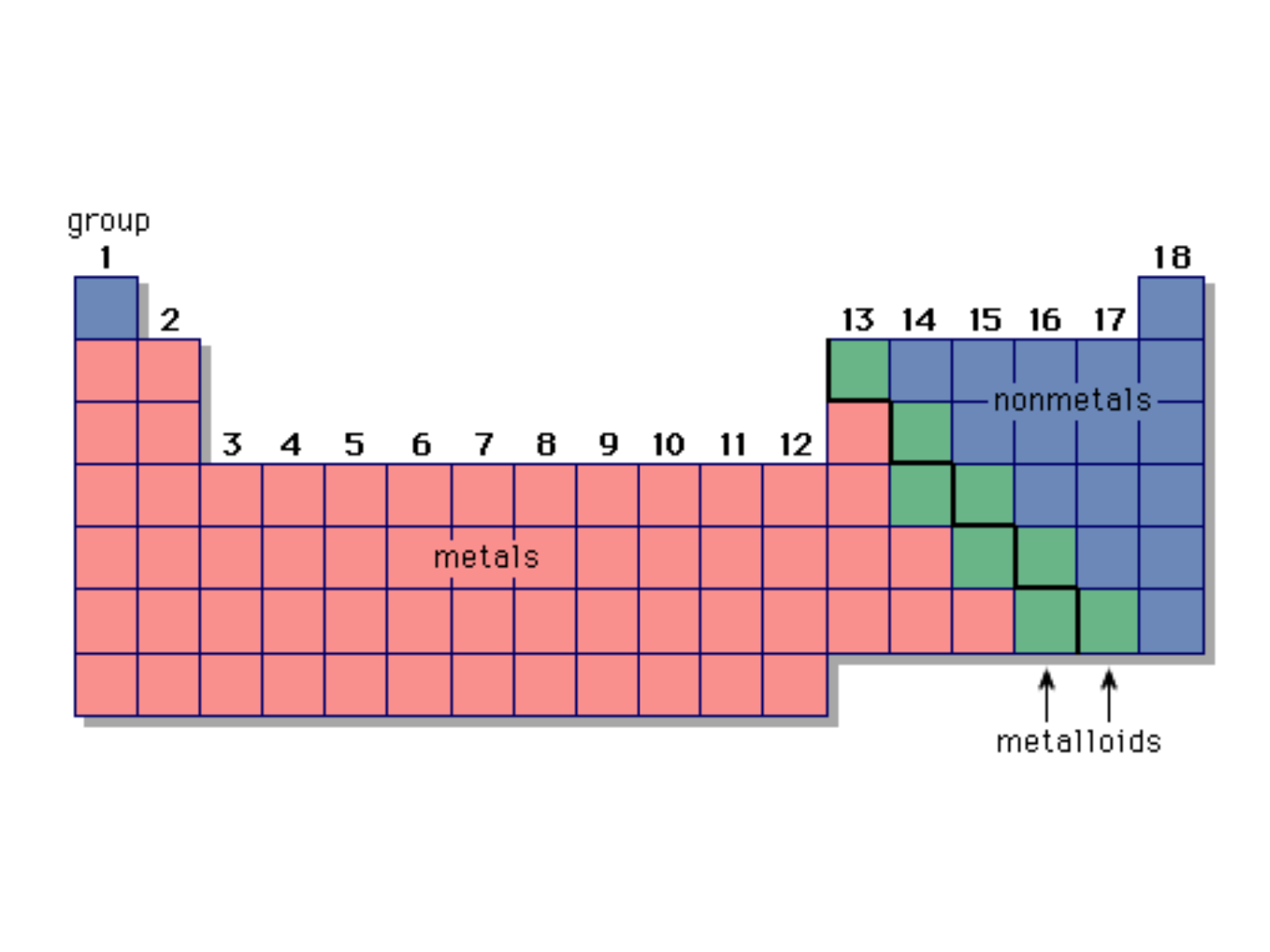
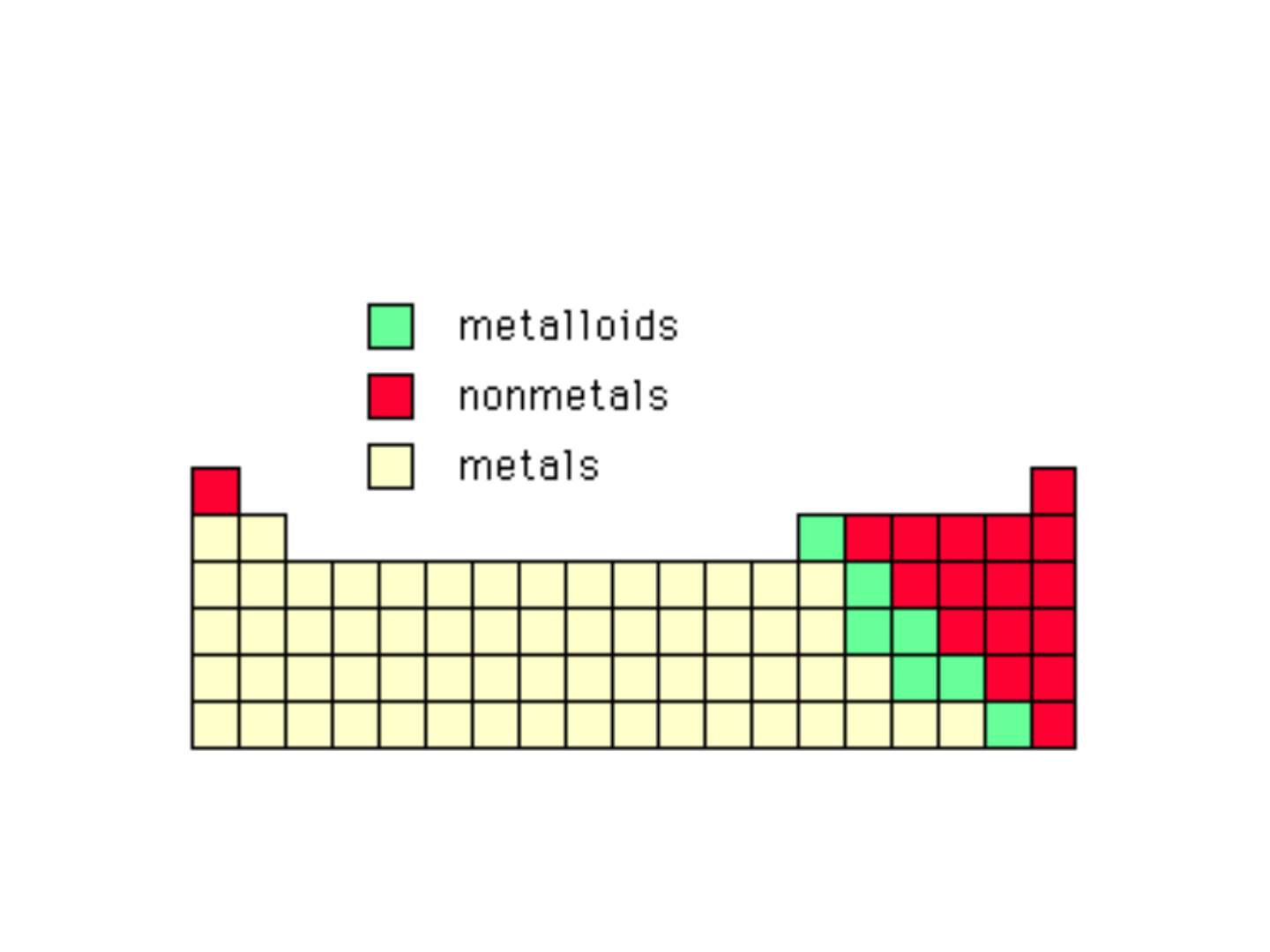
● Representative Elements: “Group A”
elements; columns 1A-8A; they
include:
-metals
-nonmetals
-metalloids
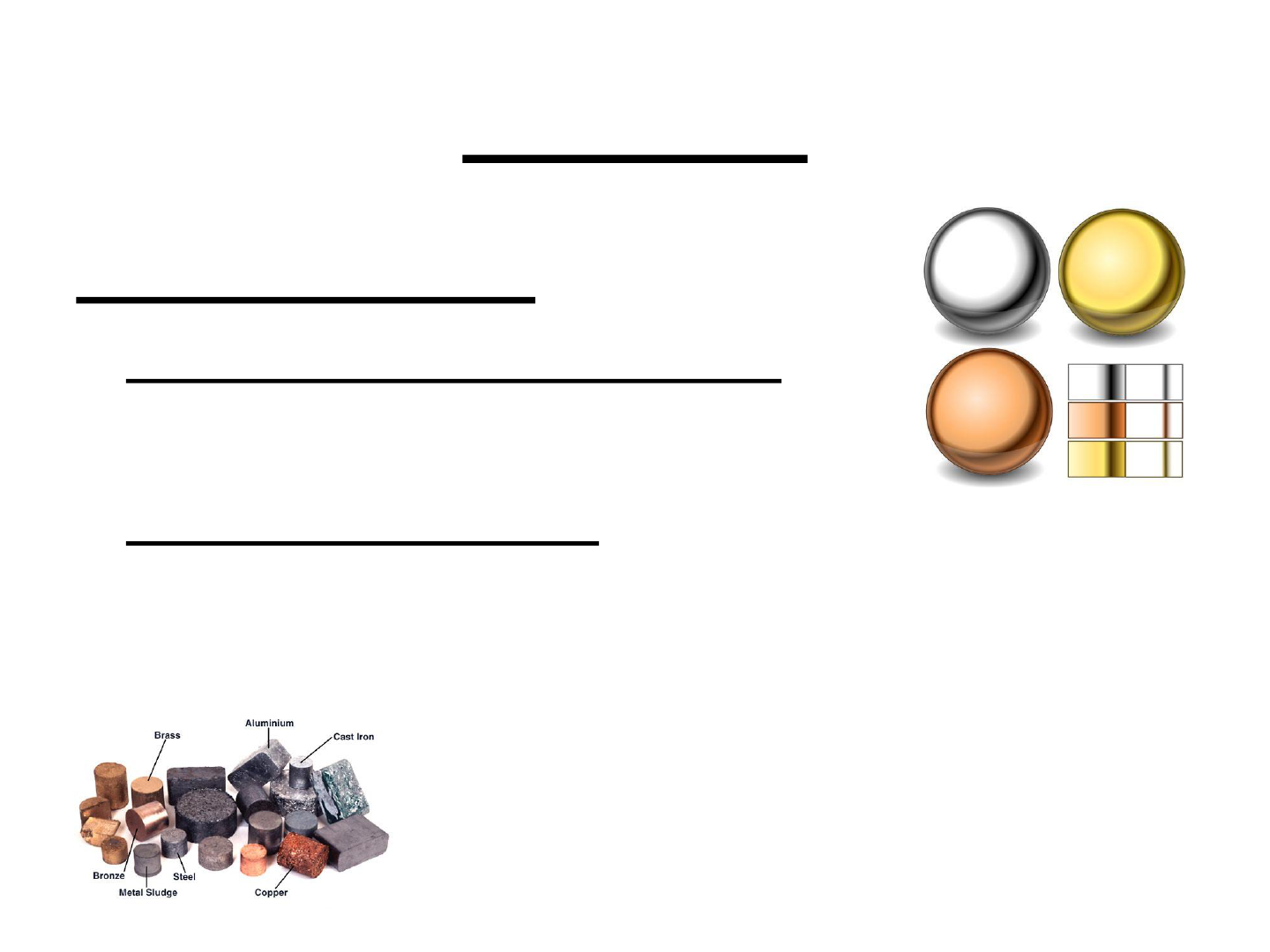
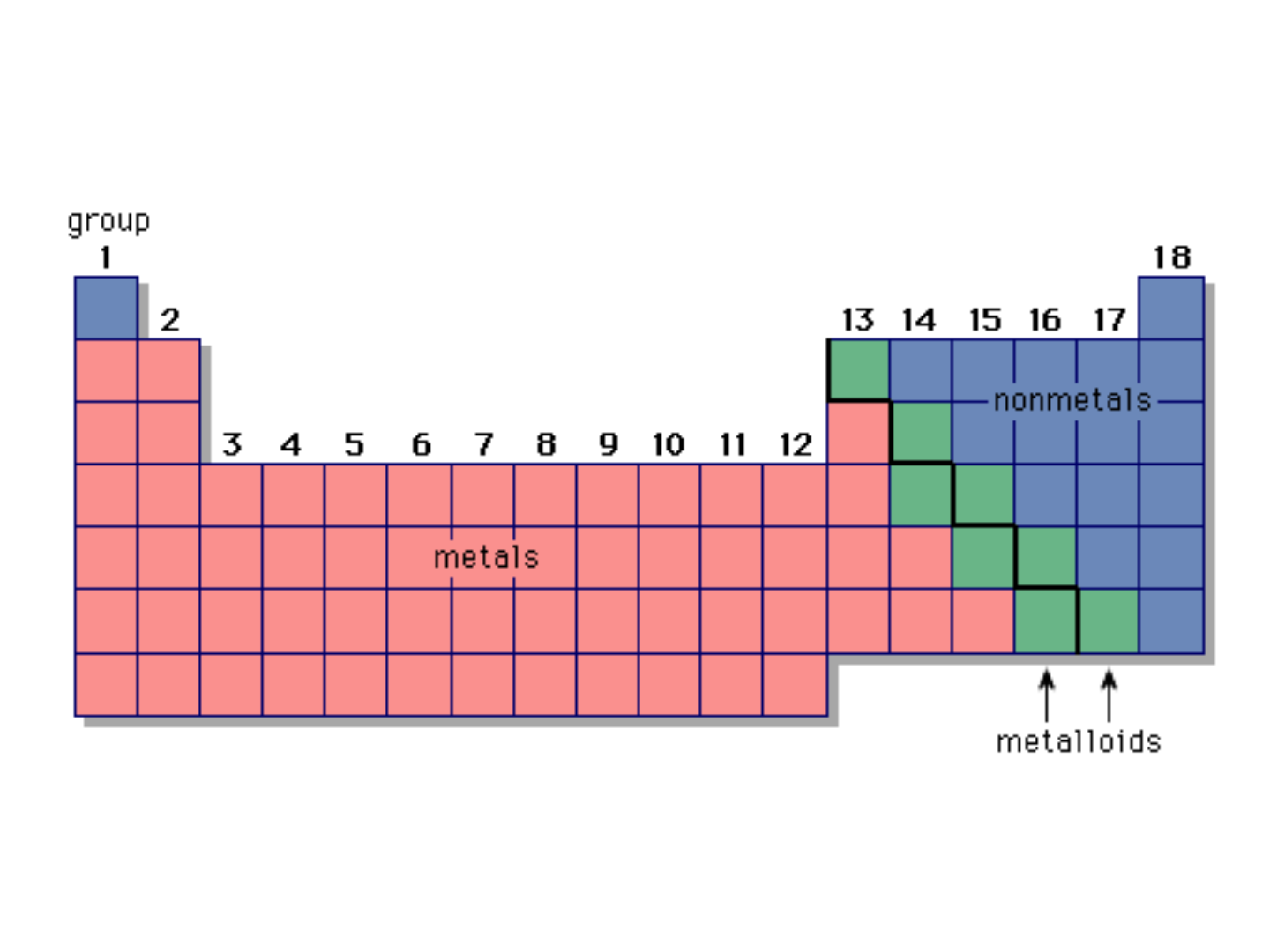
METALS:
Characteristics:
● high electrical conductivity
● high luster
● ductile & malleable
● on the left side of the periodic table
(except hydrogen)
NONMETALS:
Characteristics:
● in the upper-right corner of the periodic
table
● nonlustrous
● poor conductors of electricity
● some (O, Cl) are gases at room temp.
● others (S) are brittle solids
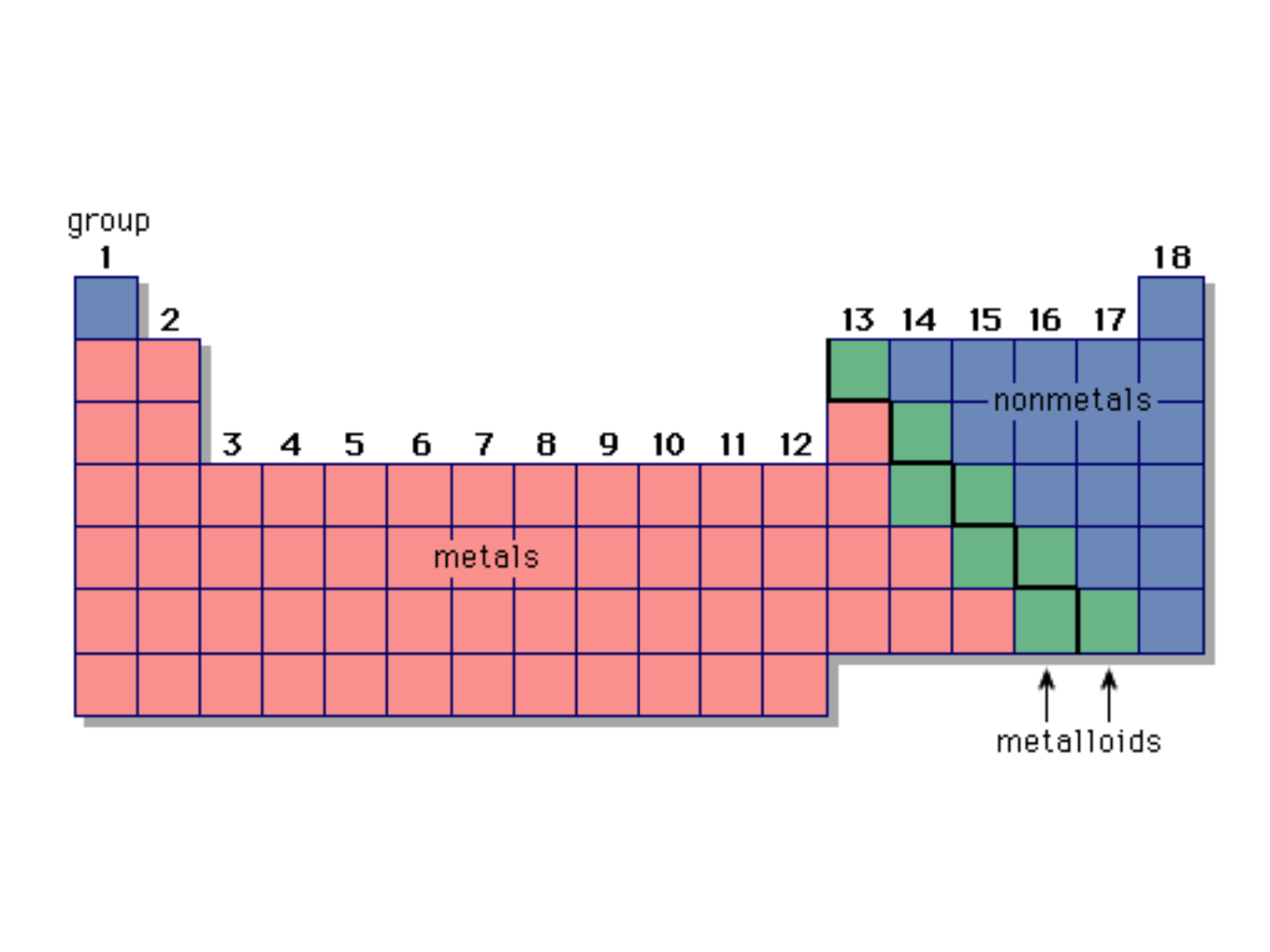
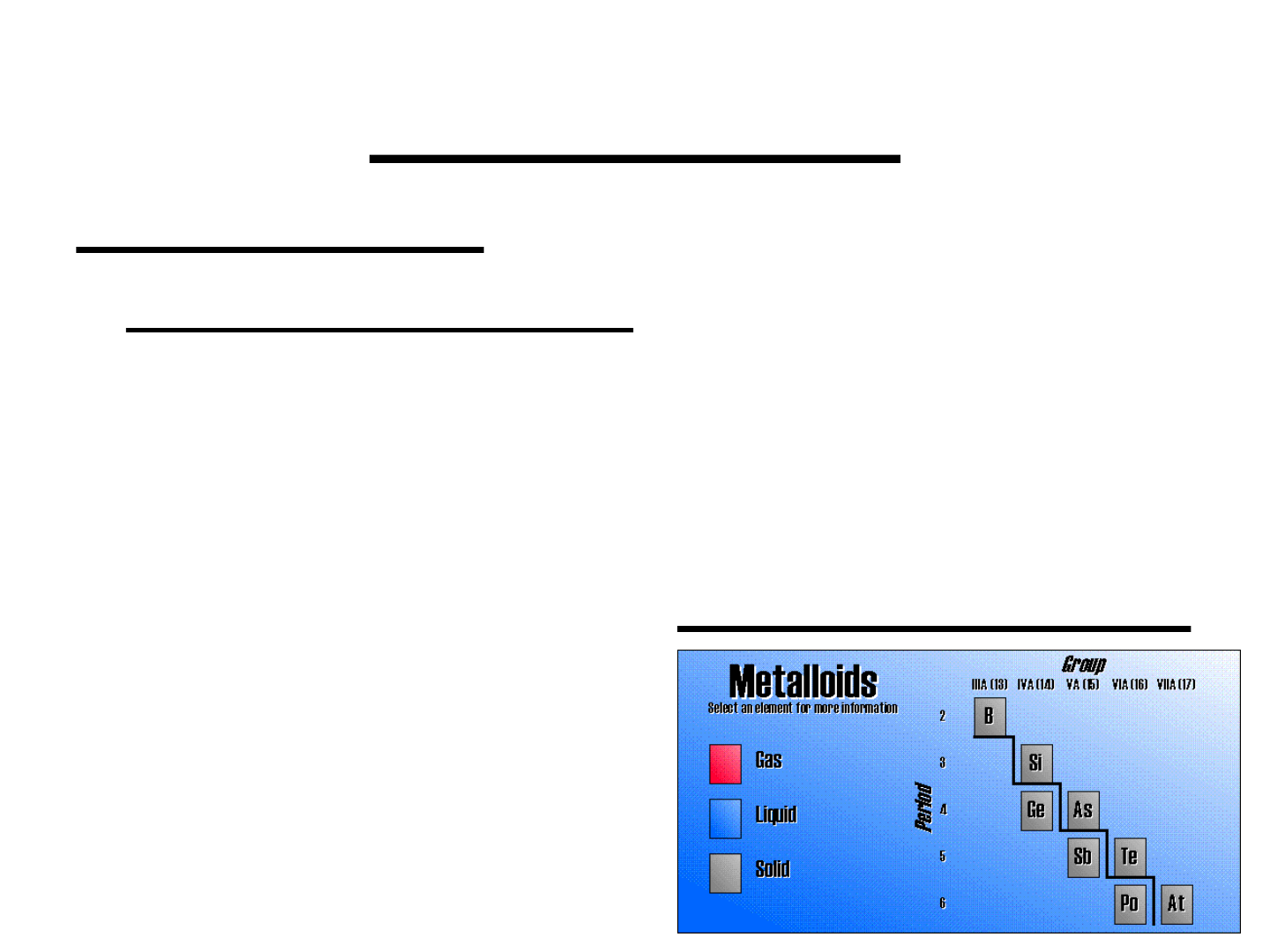
METALLOIDS:
Characteristics:
● on the stair-step line that divides the
metals from the nonmetals
● exhibit properties that are intermediate
between those of metals and nonmetals
● important metalloids: silicon, germanium

PERIODIC GROUPS
● alkali metals
● alkaline earth metals
● transition metals
● lanthanides
● actinides
● halogens
● noble gases
“inner” transition
metals

THE METALS!!

Alkaline Earth Metals
● Group 2 on periodic table
● Abundant metals in the earth
● Not as reactive as alkali metals
● examples: magnesium, strontium

Transition Metals
● Groups 3 - 12 on periodic table
● Important for living organisms (i.e. as
minerals)
● examples: iron, zinc, manganese
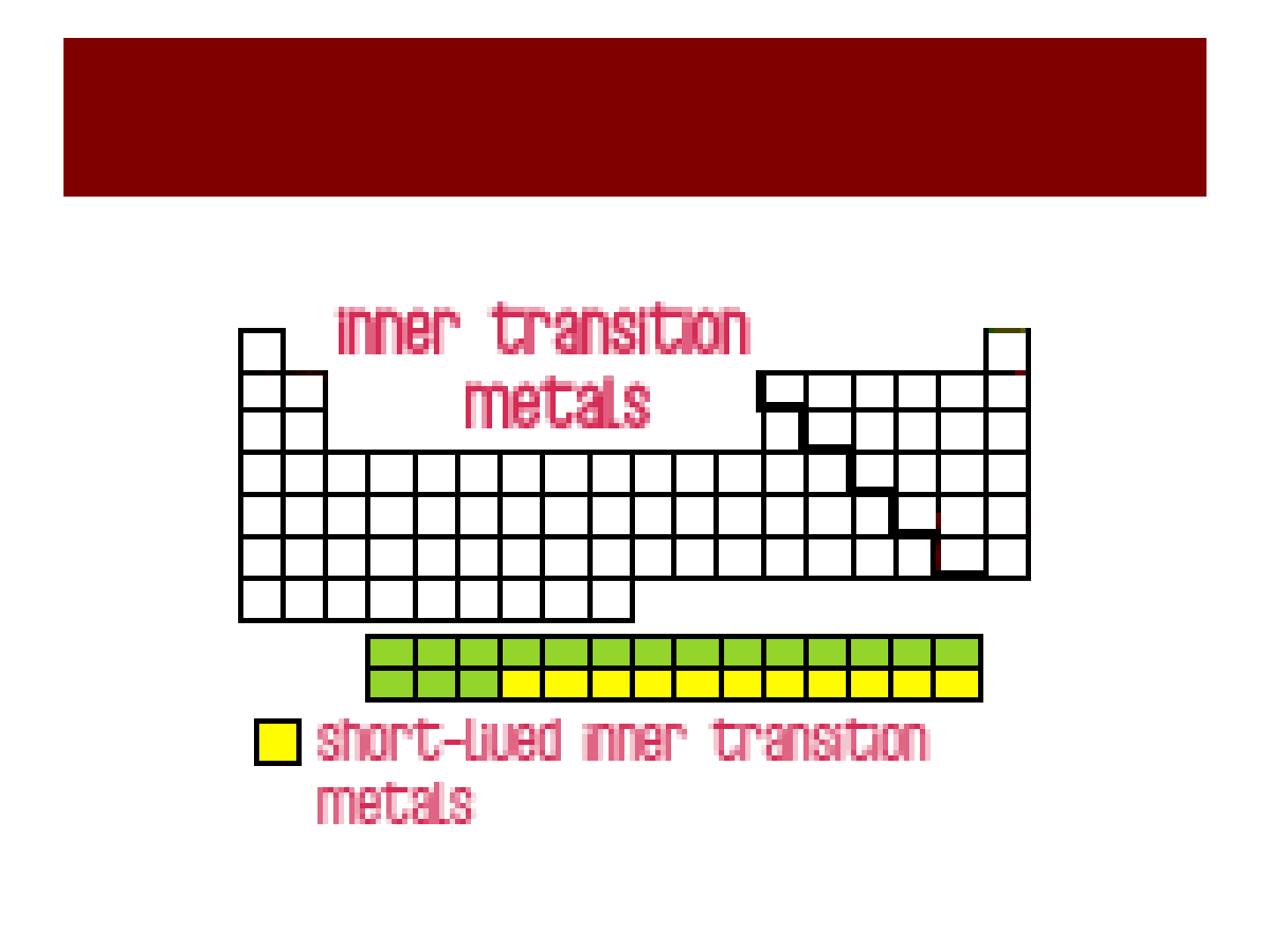
Inner Transition Metals!!
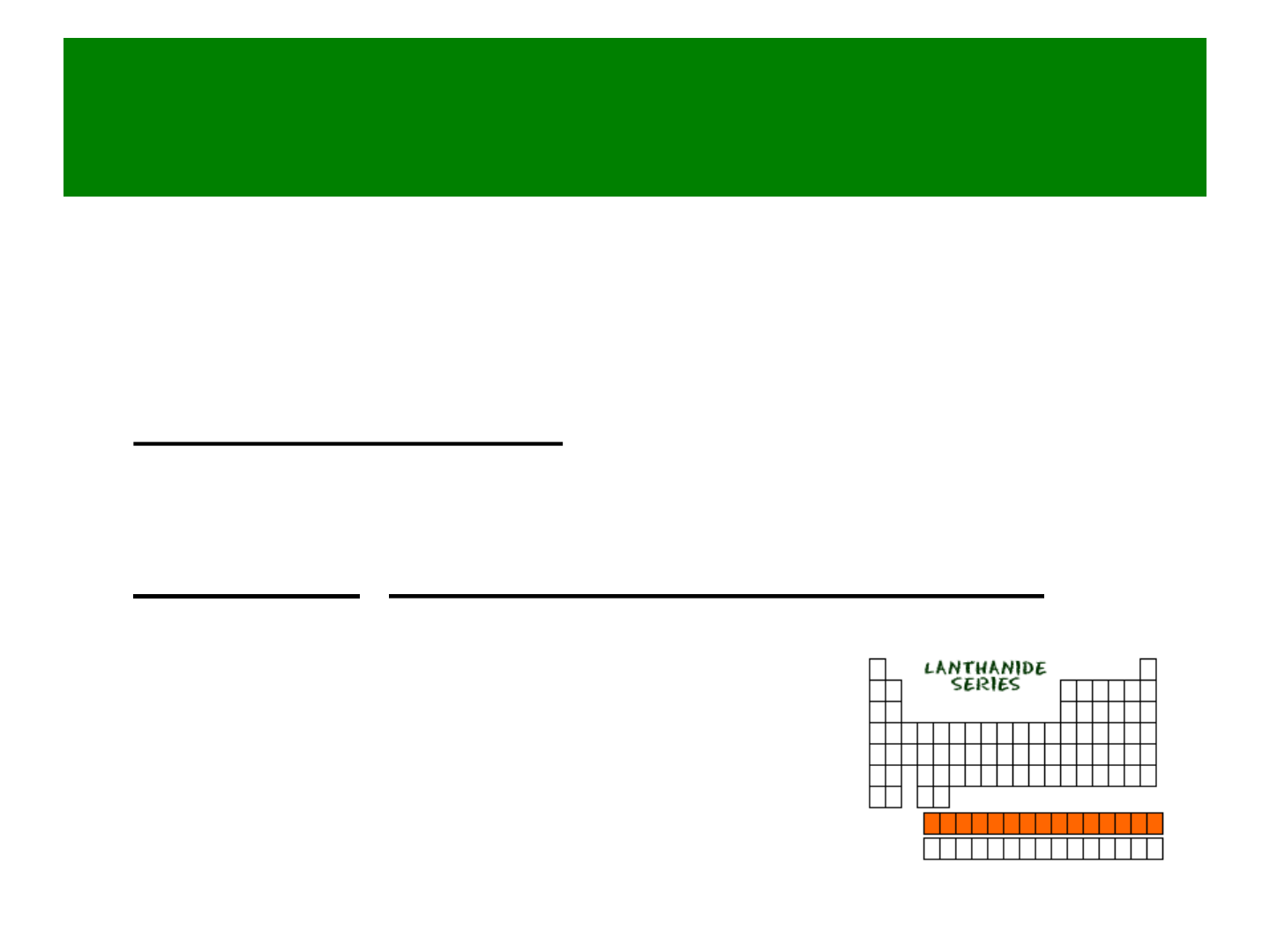
Lanthanides
● Part of the “inner transition metals”
● Soft silvery metals
● elements # 57 - 70
● examples: cerium, europium, ytterbium
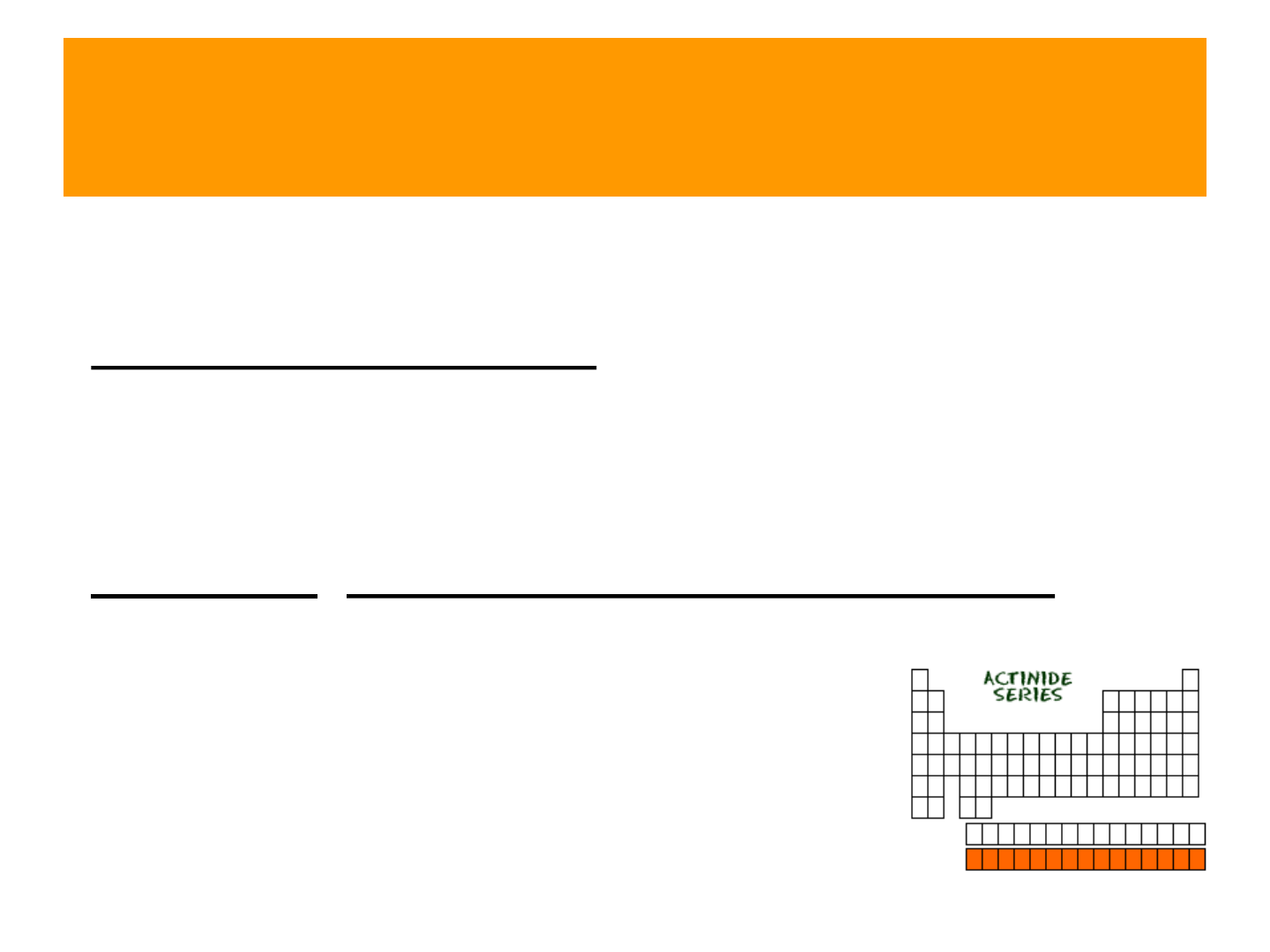
Actinides
● Radioactive elements
● Part of the “inner transition metals”
● elements # 89 – 102
● examples: uranium, plutonium, berkelium
THE NONMETALS!!
Halogens
● Group 17 on periodic table
● “Salt former” – combines with groups 1 and
2 to form salts (ionic bonds)
● examples: fluorine, bromine, iodine
Noble Gases
● Group 18 on periodic table
● Relatively inert, or nonreactive
● Gases at room temperature
● examples: helium, argon, radon
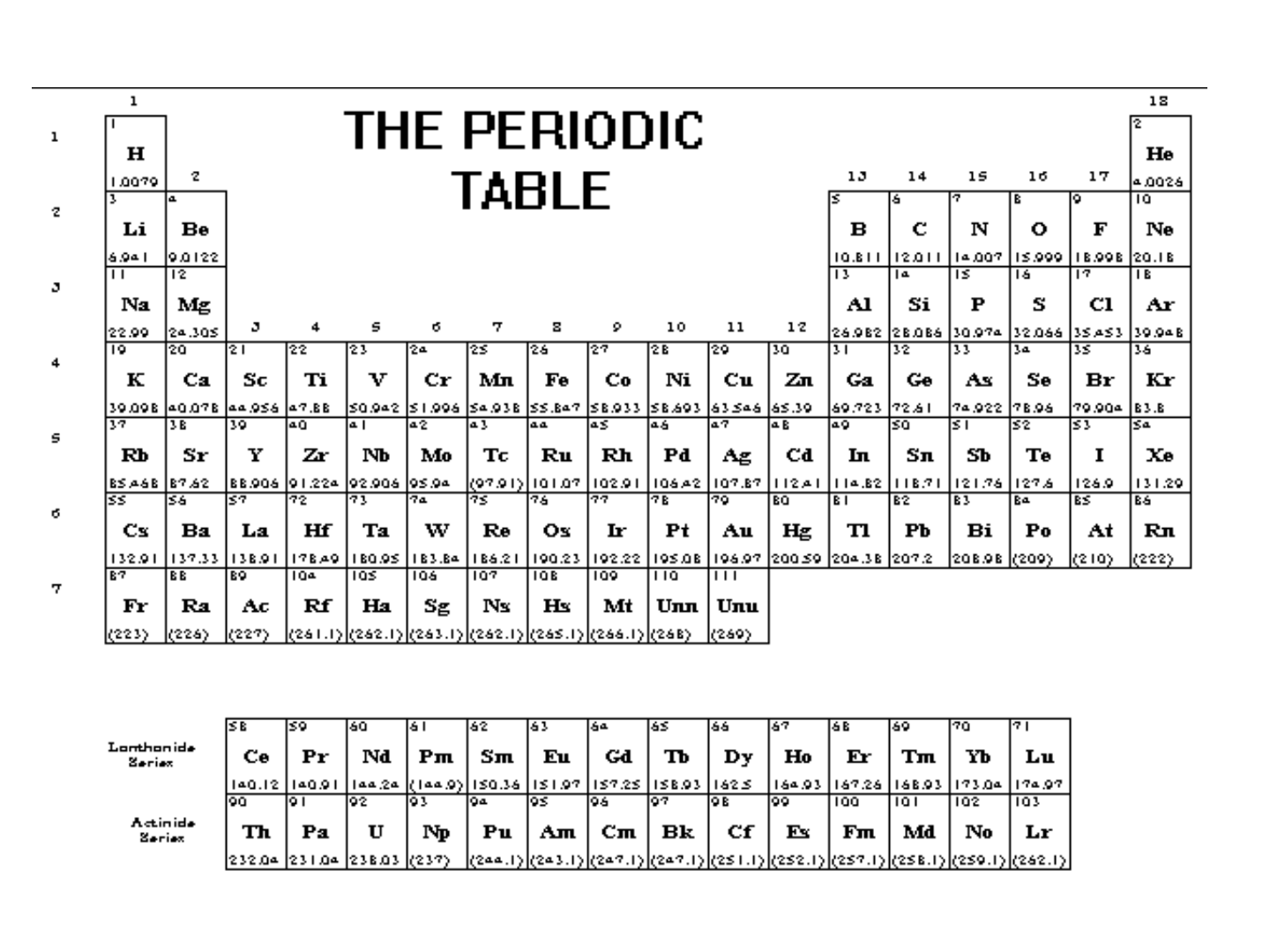
Activity: use a black and white copy of the periodic table.
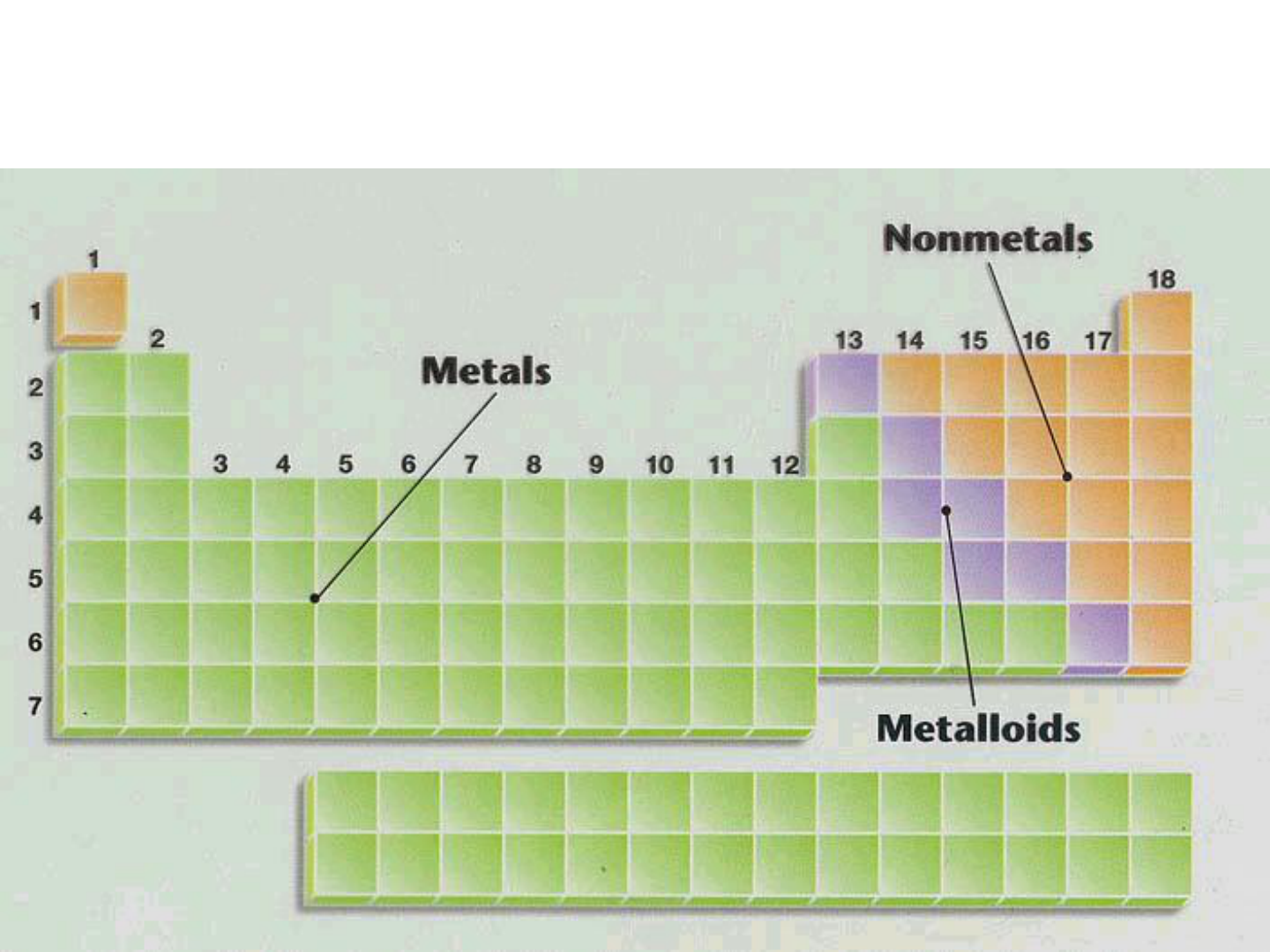
On one side, color and label the metals, nonmetals, and metalloids.
Another name for “metalloid” is “semi-metal”.
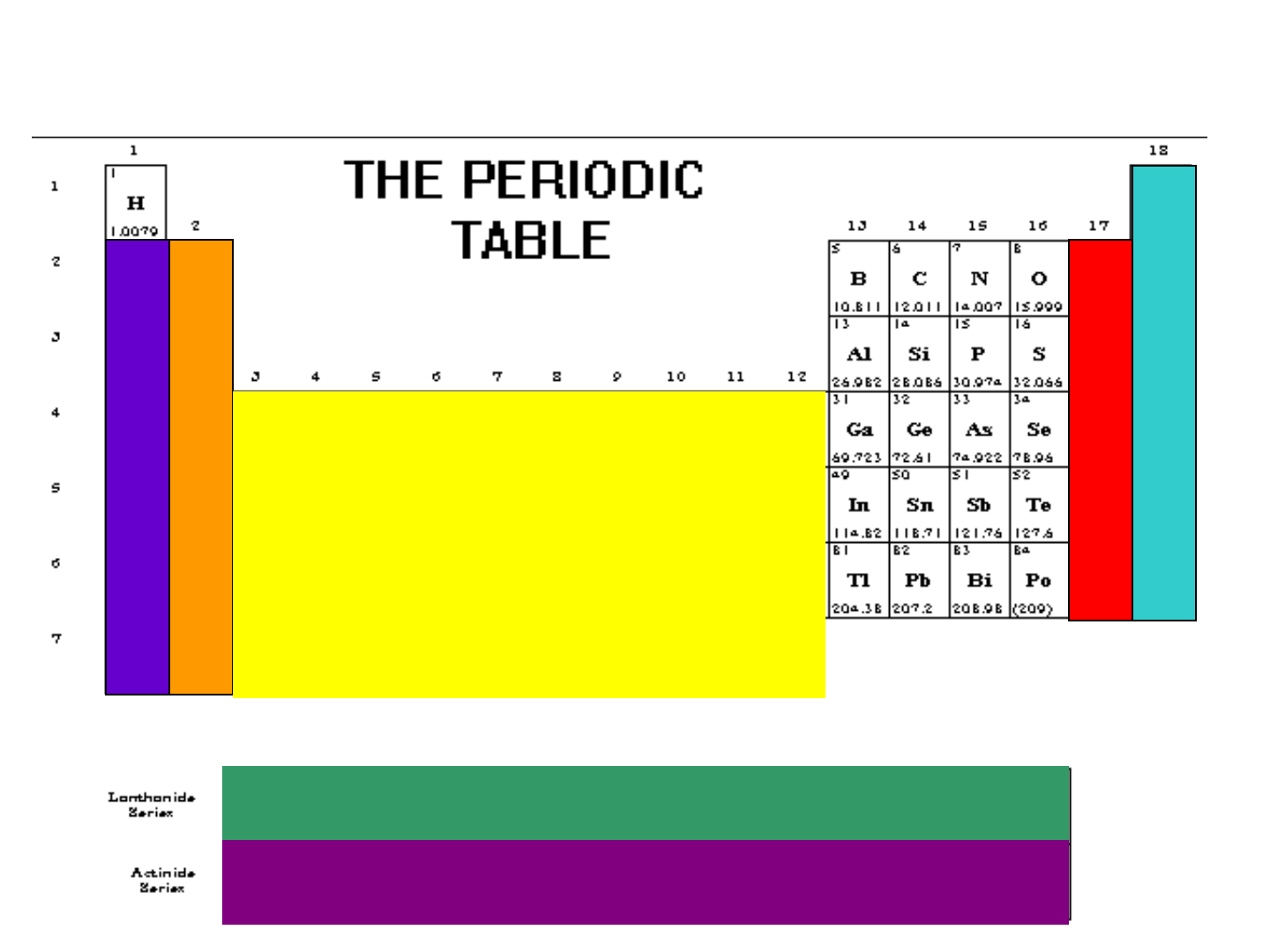
Transition metals
alkali metals
alkaline earth metals
halogens
noble gases
lanthanides
actinides
Color and label the groups/families of elements on the other side of
your paper. Remember to create a legend.
Chemical Properties & Families
● Chemical properties of elements are
based on their “VALENCE ELECTRONS”
● Families are groups of elements that have
similar VALENCE ELECTRONS
● VALENCE ELECTRONS = outermost
electrons in an atom
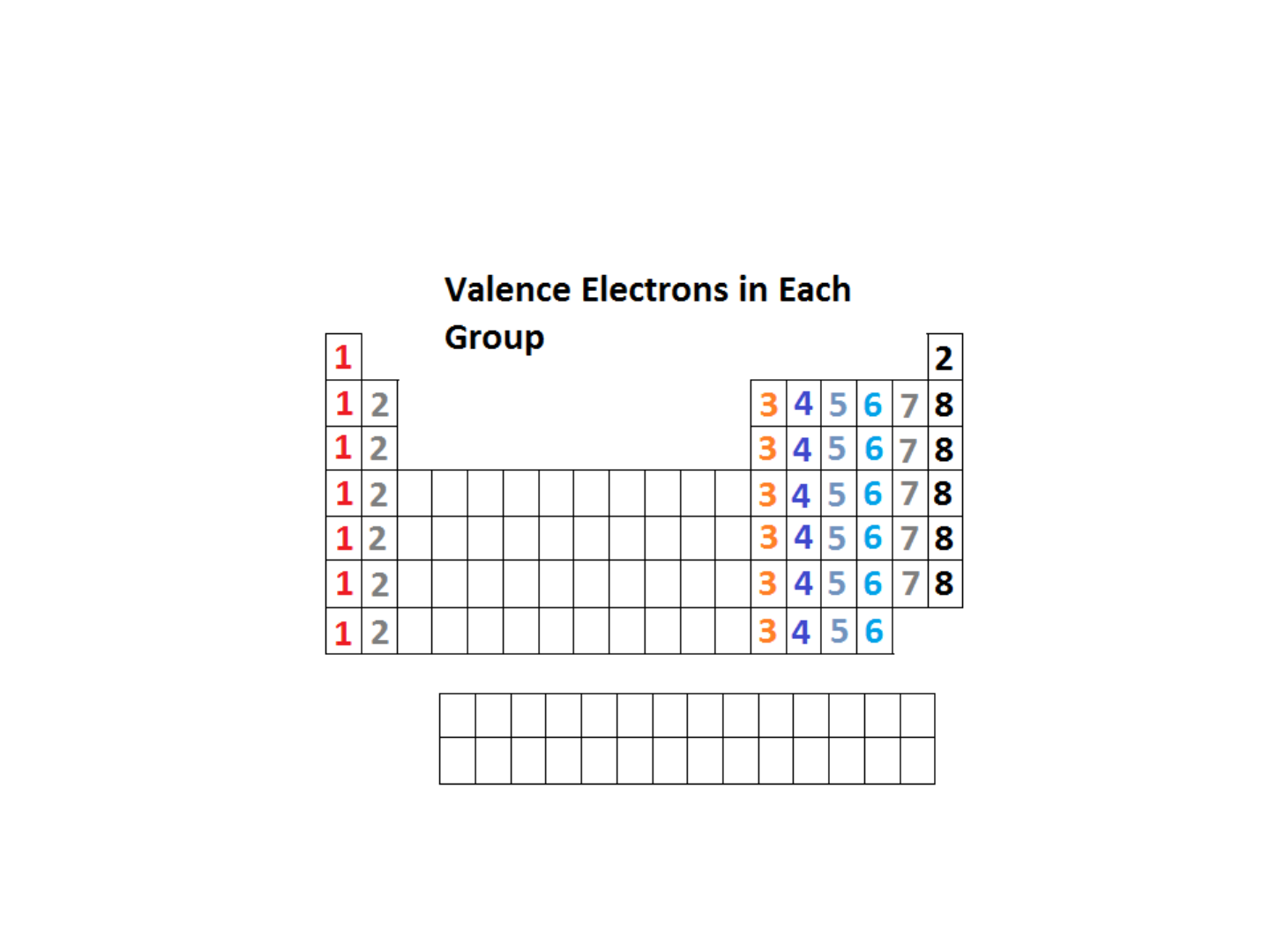
VALENCE ELECTRONS:

VALENCE ELECTRONS:
NOTES: 6.3 – Periodic Trends

PERIODIC TABLE:
RECALL…
● Periodicity: regular variations (or patterns) of
properties with increasing atomic weight; both
chemical and physical properties vary in a
periodic (repeating pattern).
● Group: vertical column of elements (“family”)
● Period: horizontal row of elements

PERIODIC PROPERTIES:
● Atomic radius
● Ionization energy
● Ionic size / radius
● Electronegativity

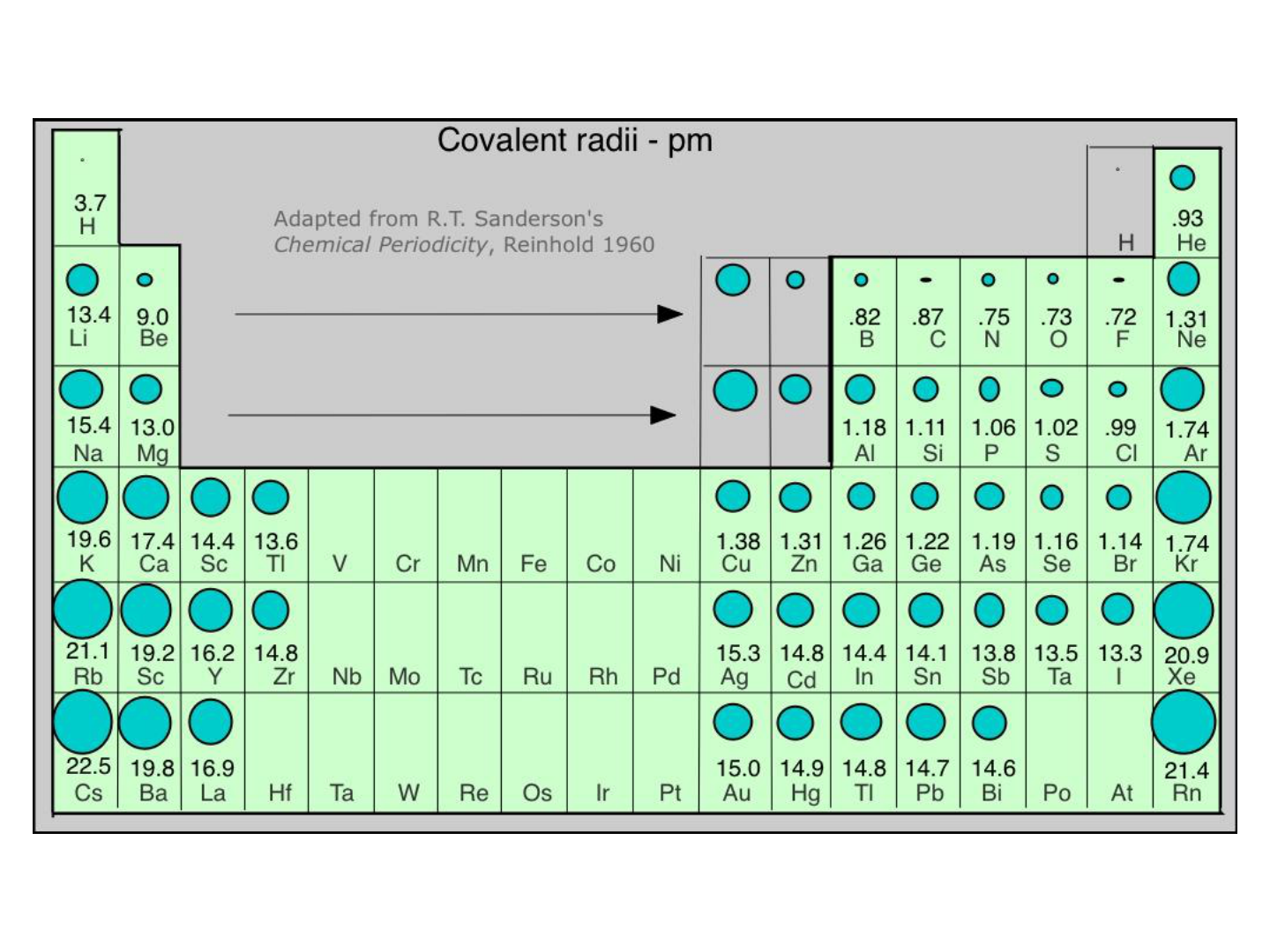
ATOMIC RADIUS:
● GROUP TREND: increases as you move
down a group
● WHY???
-electrons are added to higher energy levels
(farther away from the nucleus).

ATOMIC RADIUS:
● PERIODIC TREND: decreases as you
move L to R across a period
● WHY???
-As the # of protons in the nucleus
increases, the positive charge increases
and as a result, the “pull” on the electrons
increases.

IONS:
● ION: an atom or group or atoms that
has a positive or negative charge
● recall…an atom is electrically neutral
because it has equal # of protons (+)
& # of electrons (-)
● positive & negative ions form when
electrons are transferred between
atoms!
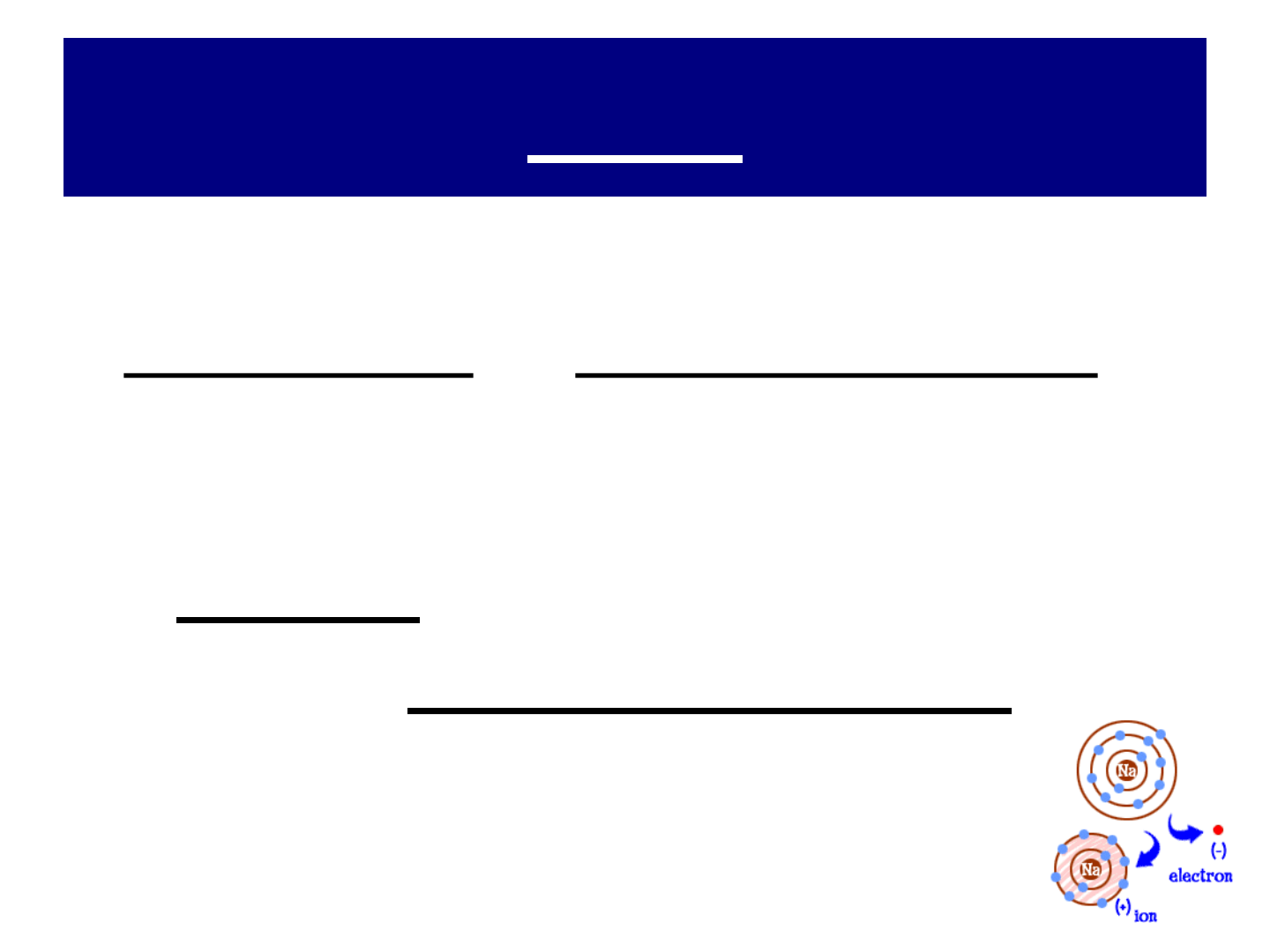
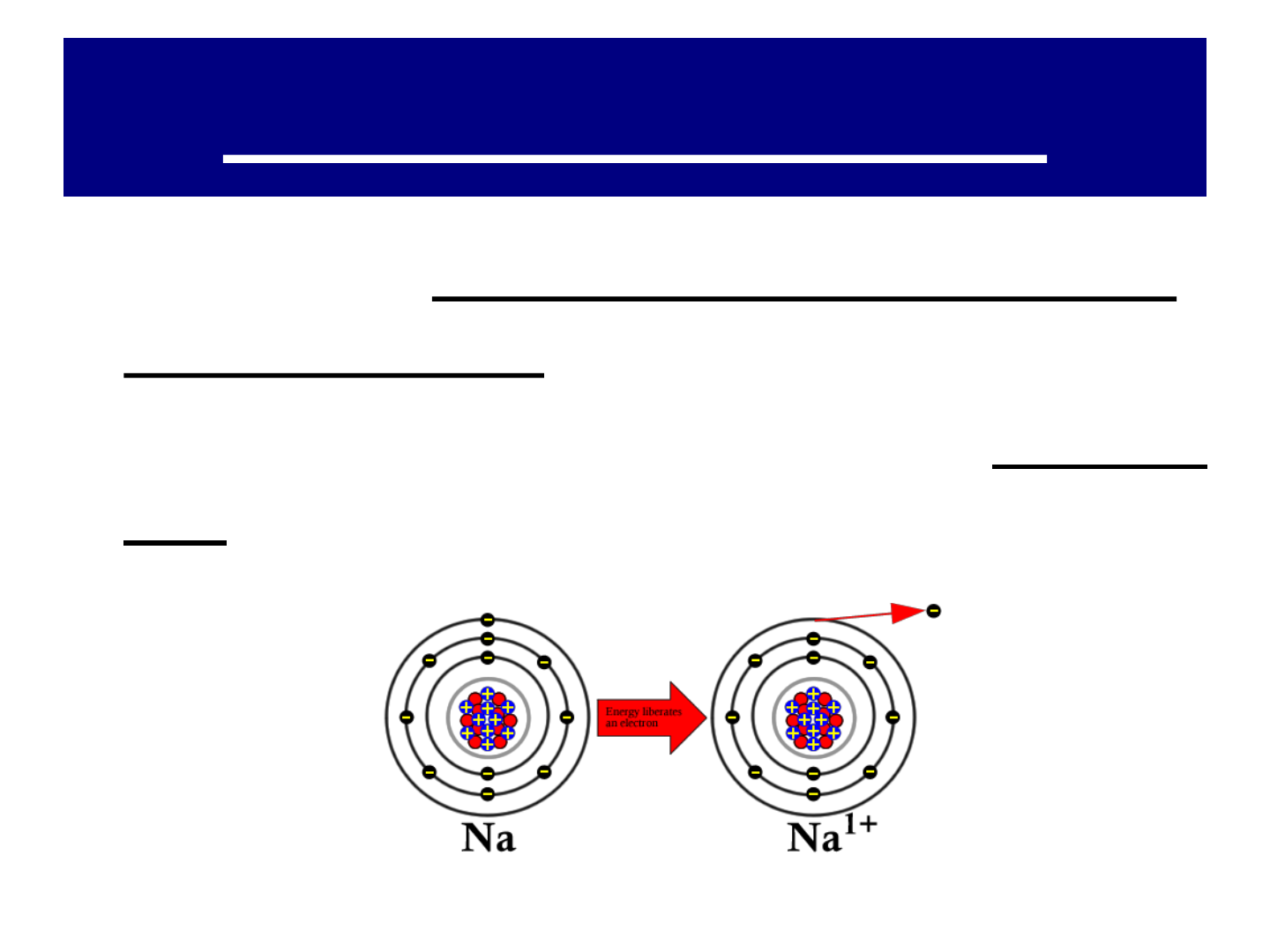
IONS:
● atoms of METALS tend to form
positive ions by losing 1 or more e
-
from their valence (outermost) shell
● an ion with a positive charge is called
a CATION.
● example: SODIUM (Na Na
+
)
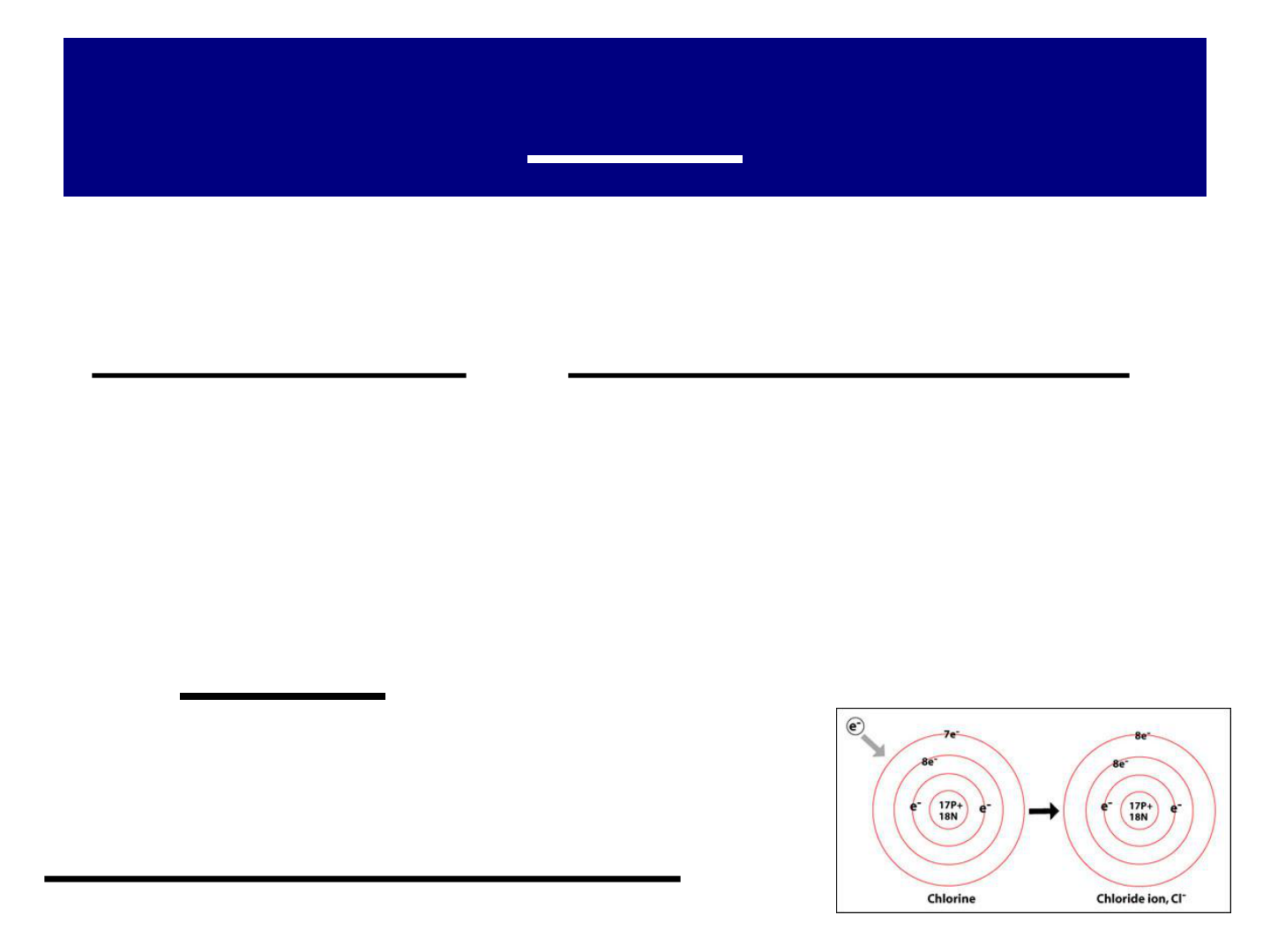
IONS:
● atoms of NONMETALS tend to form
negative ions by gaining 1 or more e
-
(& thus filling their outermost energy
level)
● an ion with a negative charge is called
an ANION.
● example:
CHLORINE (Cl Cl
-
)

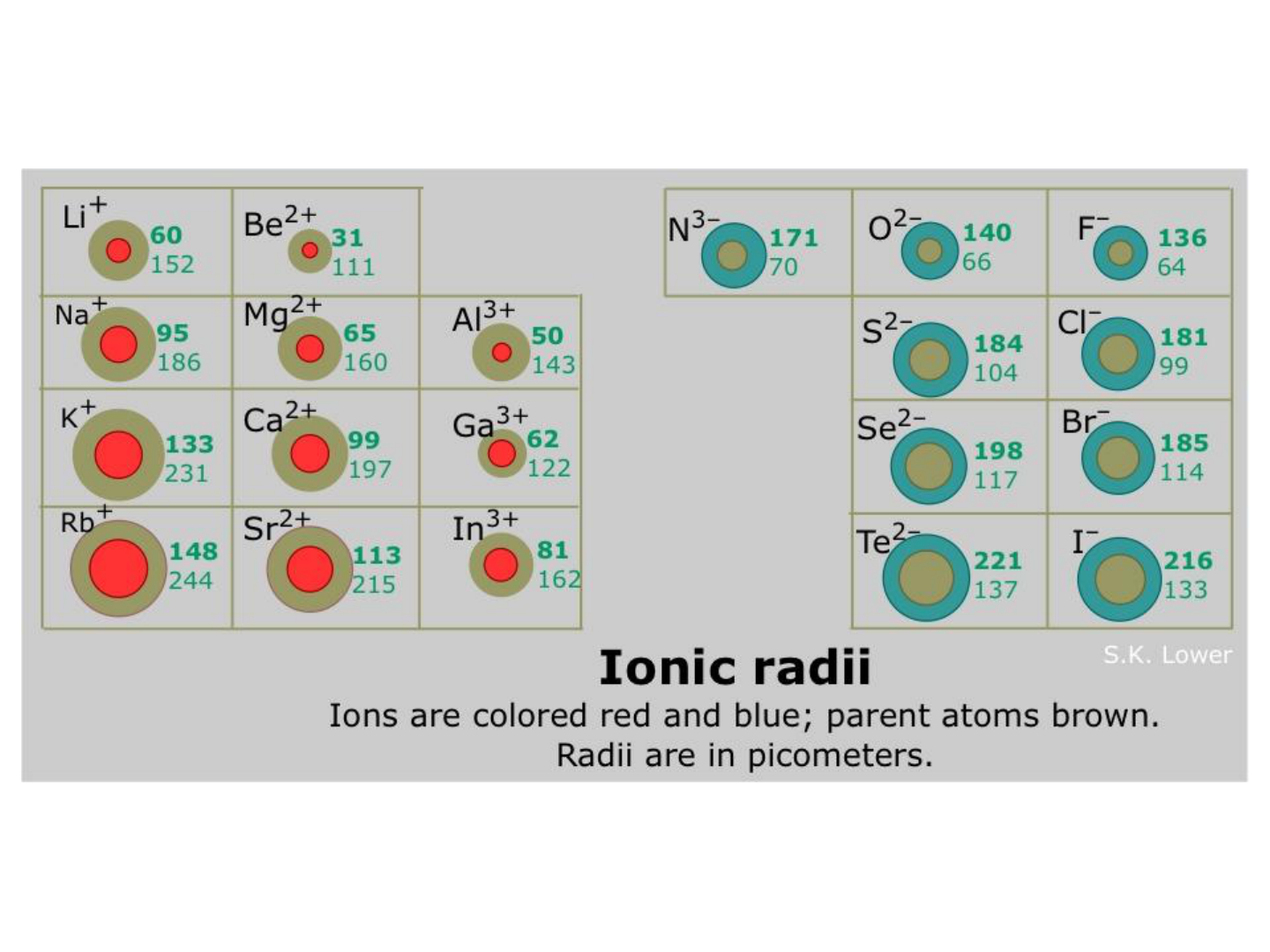
● ANIONS (negative ions) have full
valence shells: they are LARGER
than their neutral ATOM.
● CATIONS (positive ions) have empty
valence shells: they are SMALLER
than their neutral ATOM.
● BUT, the size of one ion compared to
the next is the same pattern as
ATOMIC RADIUS.
IONIC RADIUS:
IONIZATION ENERGY:
● Definition: energy required to remove
outer electrons
results in the formation of a positive
ion!

IONIZATION ENERGY:
● GROUP TREND: decreases as you
move down a group
● WHY???
-Electrons are in higher energy levels as
you move down a group; they are farther
away from the positive “pull” of the
nucleus and therefore easier to remove.

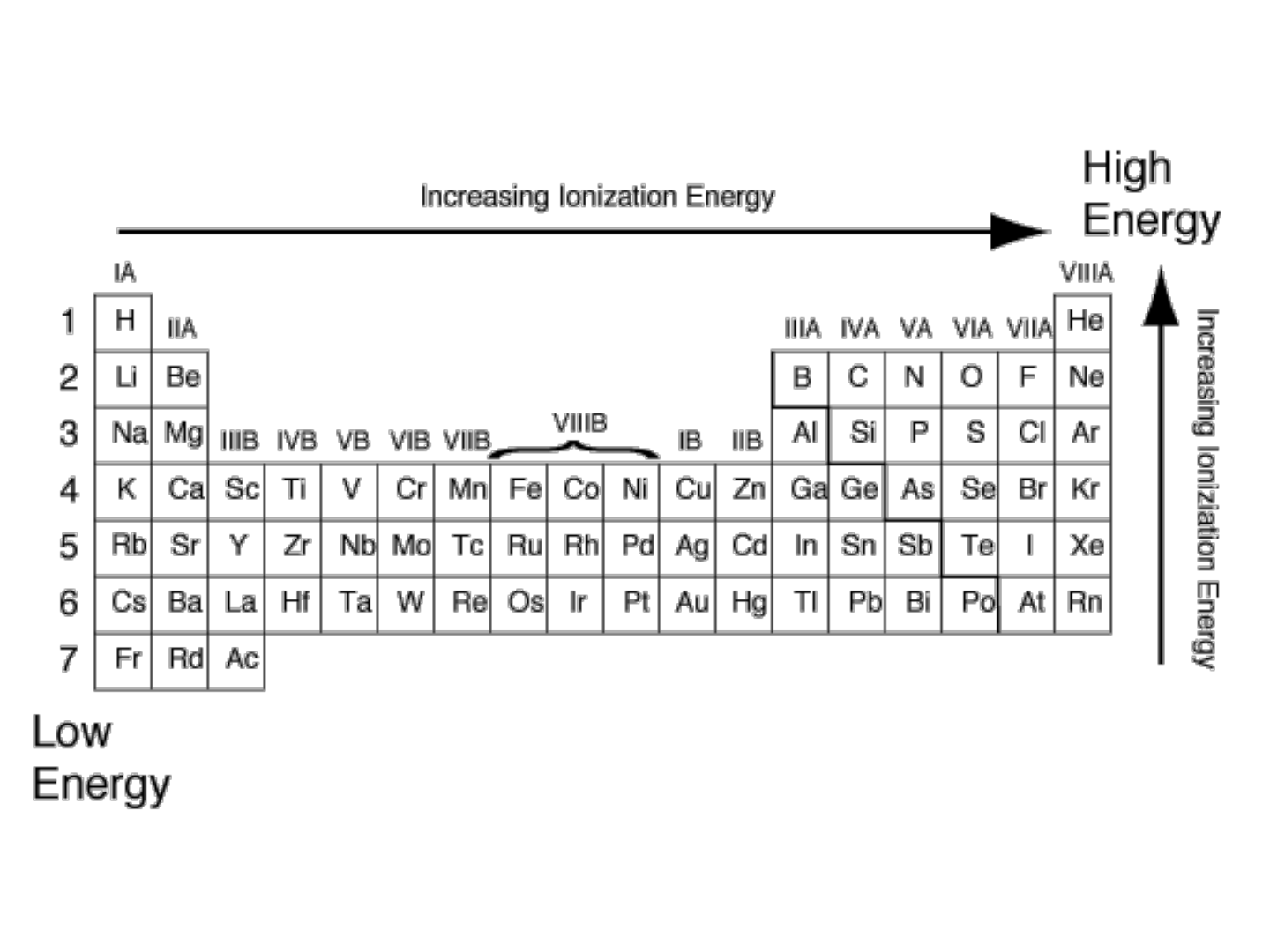
IONIZATION ENERGY:
● PERIODIC TREND: increases as you
move from L to R across a period
● WHY???
-The increasing charge in the nucleus as
you move across a period exerts greater
“pull” on the electrons; it requires more
energy to remove an electron.

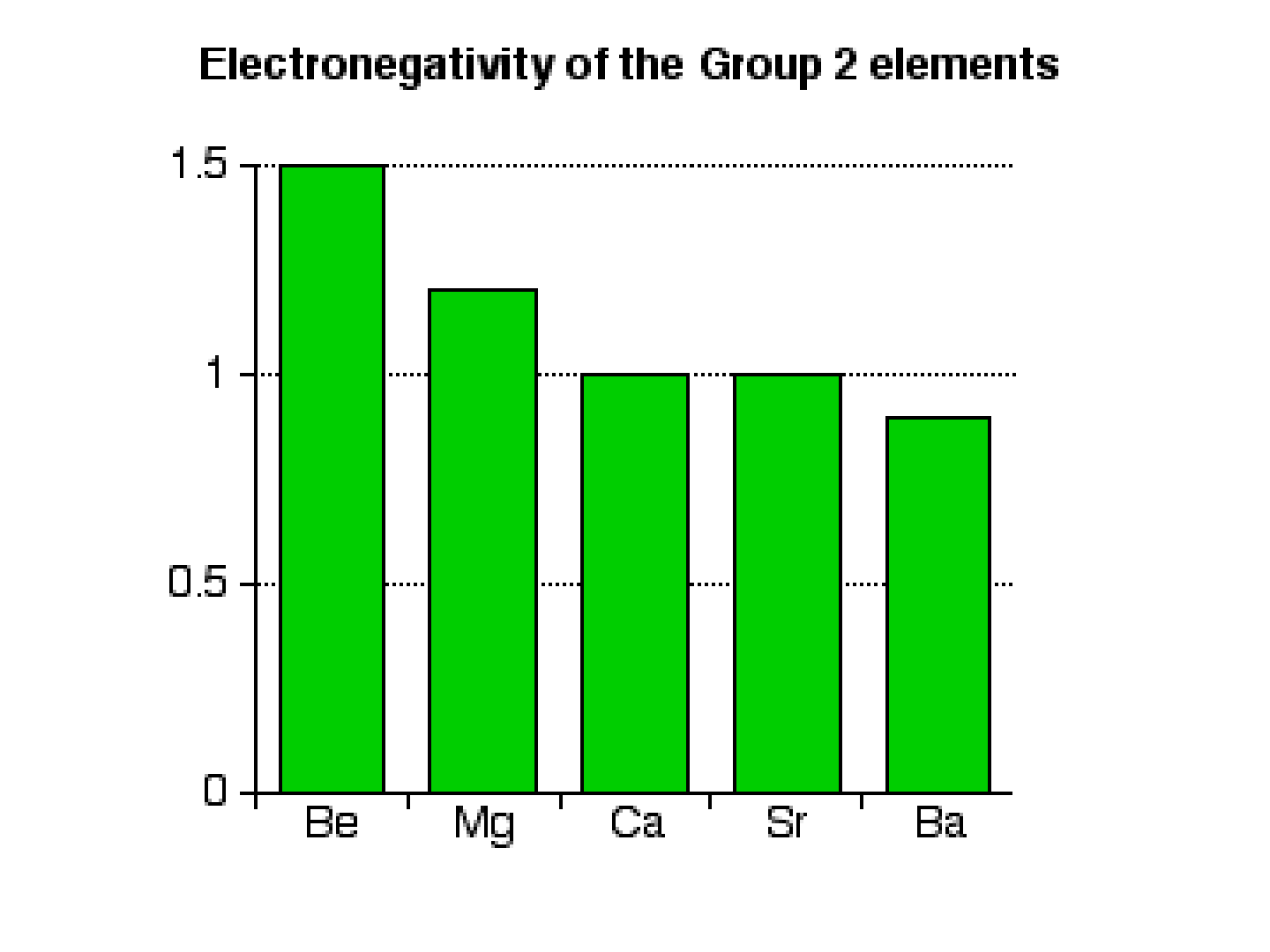
ELECTRONEGATIVITY:
● Definition: the tendency of an atom to
attract electrons of another atom
ELECTRONEGATIVITY:
● GROUP TREND: decreases as you move
down a group
● WHY???
-higher energy levels means the electrons
are farther away from the nucleus;
-greater distance = decreased attraction

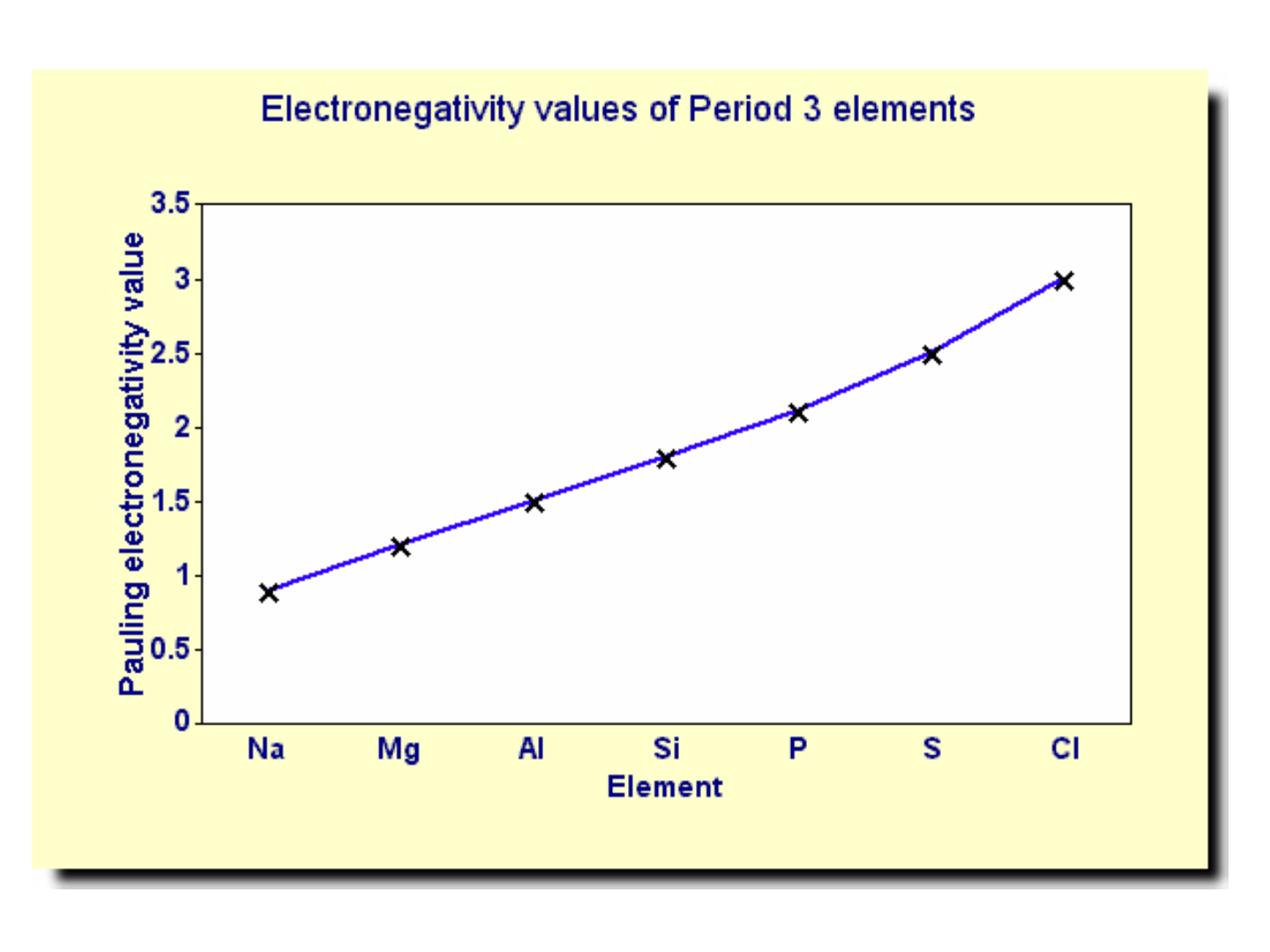
ELECTRONEGATIVITY:
● PERIODIC TREND: increases as you move
across a period (noble gases excluded!)
● WHY???
-nuclear charge is increasing;
-atomic radius is decreasing
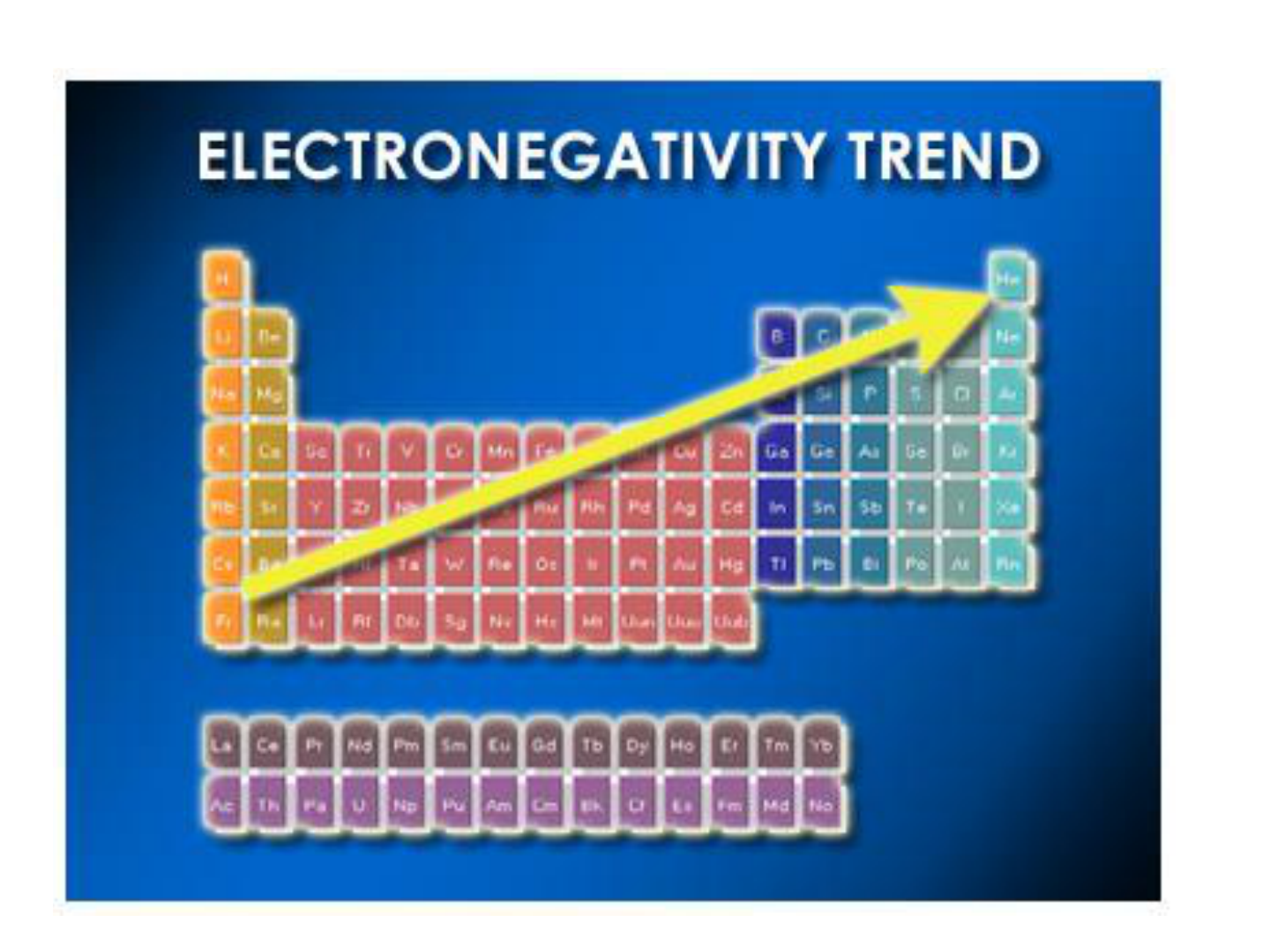

ELECTRONEGATIVITY:
● Most electronegative element:
FLUORINE (4.0)
● Least electronegative element:
CESIUM (0.7)
**see table 6.2 on page 181 for all values!