
Vol. 79 Friday,
No. 100 May 23, 2014
Part II
Department of Health and Human Services
Centers for Medicare & Medicaid Services
42 CFR Parts 417, 422, 423, et al.
Medicare Program; Contract Year 2015 Policy and Technical Changes to
the Medicare Advantage and the Medicare Prescription Drug Benefit
Programs; Final Rule
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00001 Fmt 4717 Sfmt 4717 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29844
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
DEPARTMENT OF HEALTH AND
HUMAN SERVICES
Centers for Medicare & Medicaid
Services
42 CFR Parts 417, 422, 423, and 424
[CMS–4159–F]
RIN 0938–AR37
Medicare Program; Contract Year 2015
Policy and Technical Changes to the
Medicare Advantage and the Medicare
Prescription Drug Benefit Programs
AGENCY
: Centers for Medicare &
Medicaid Services (CMS), HHS.
ACTION
: Final rule.
SUMMARY
: The final rule will revise the
Medicare Advantage (MA) program (Part
C) regulations and prescription drug
benefit program (Part D) regulations to
implement statutory requirements;
improve program efficiencies; and
clarify program requirements. The final
rule also includes several provisions
designed to improve payment accuracy.
DATES
: Effective Dates: These
regulations are effective on July 22, 2014
except for the amendment in instruction
27 to § 423.100, the amendment in
instruction 30 to § 423.501, and the
amendment in instruction 34 to
§ 423.505, which are effective on
January 1, 2016.
Applicability Dates: In the
SUPPLEMENTARY INFORMATION
section of
this final rule, we provide a table (Table
1) which lists key changes in this final
rule that have an applicability date
other than the effective date of this final
rule.
FOR FURTHER INFORMATION CONTACT
:
Christopher McClintick, (410) 786–
4682, Part C issues.
Marie Manteuffel, (410) 786–3447, Part
D issues.
Kristy Nishimoto, (206) 615–2367, Part
C and D enrollment and appeals
issues.
Whitney Johnson, (410) 786–0490, Part
C and D payment issues.
Joscelyn Lissone, (410) 786–5116, Part C
and D compliance issues.
Frank Whelan, (410) 786 1302, Part D
improper prescribing issues.
SUPPLEMENTARY INFORMATION
: Table 1
lists key changes that have an
applicability date other than 60 days
after the date of publication of this final
rule. The applicability dates are
discussed in the preamble for each of
these items.
T
ABLE
1—A
PPLICABILITY
D
ATE OF
K
EY
P
ROVISIONS
O
THER
T
HAN
60 D
AYS
A
FTER THE
D
ATE OF
P
UBLICATION OF THE
F
INAL
R
ULE
Preamble section Section title Applicability date
III.A.4 ........................ Reducing the Burden of the Compliance Program Training Requirements (§§ 422.503(b)(4)(vi)(C) and
§ 423.504(b)(4)(vi)(C)).
01/01/2016
III.A.7 ........................ Agent/Broker Compensation Requirements (§§ 422.2274 and 423.2274) ............................................... 01/01/2015
III.A.20 ...................... Enrollment Requirements for the Prescribers of Part D Covered Drugs (§ 423.120(c)(6)) ..................... 06/01/2015
III.A.24 ...................... Eligibility of Enrollment for Incarcerated Individuals (§§ 417.1, 417.460(b)(2)(i), 417.460(f)(1)(i)(A)
through (C), 422.74(d)(4)(i)(A), 422.74(d)(4)(v), 423.44(d)(5)(iii) and (iv)).
01/01/2015
Table of Contents
I. Executive Summary
A. Purpose
B. Summary of the Major Provisions
1. Modifying the Agent/Broker
Requirements, Specifically Agent/Broker
Compensation
2. Drug Categories or Classes of Clinical
Concern
3. Improving Payment Accuracy—
Implementing Overpayment Provisions
of Section 1128J (d) of the Social
Security Act (§§ 422.326 and 423.360).
4. Risk Adjustment Data Requirements
(§ 422.310)
C. Summary of Costs and Benefits
II. Background
A. General Overview and Regulatory
History
B. Issuance of a Notice of Proposed
Rulemaking
C. Public Comments Received in Response
to the CY 2015 Policy and Technical
Changes to the Medicare Advantage and
the Medicare Prescription Drug Benefit
Programs Proposed Rule
D. Provisions Not Finalized in this Final
Rule
III. Provisions of the Proposed Regulations
A. Clarifying Various Program
Participation Requirements
1. Closing Cost Contract Plans to New
Enrollment (§§ 422.2 and 22.503)
2. Authority to Impose Intermediate
Sanctions and Civil Money Penalties
(§§ 422.752, 423.752, 422.760 and
423.760)
3. Contract Termination Notification
Requirements and Contract Termination
Basis (§§ 422.510 and 423.509)
4. Reducing the Burden of the Compliance
Program Training Requirements
(§§ 422.503(b)(4)(vi)(C) and
423.504(b)(4)(vi)(C))
5. Procedures for Imposing Intermediate
Sanctions and Civil Money Penalties
Under Parts C and D (§§ 422.756 and
423.756)
6. Timely Access to Mail Order Services
(§ 423.120)
7. Agent/Broker Requirements, Particularly
Compensation (§§ 422.2274 and
423.2274)
8. Drug Categories or Classes of Clinical
Concern (§ 423.120(b)(2)(v))
9. Medication Therapy Management
Program (MTMP) under Part D
(§ 423.153(d))
a. Multiple Chronic Diseases
b. Multiple Part D Drugs
c. Annual Cost Threshold
10. Requirement for Applicants or their
Contracted First Tier, Downstream, or
Related Entities to Have Experience in
the Part D Program Providing Key Part D
Functions (§ 423.504(b))
11. Requirement for Applicants for Stand
Alone Part D Plan Sponsor Contracts to
Be Actively Engaged in the Business of
the Administration of Health Insurance
Benefits (§ 423.504(b)(9))
12. Limit Parent Organizations to One
Prescription Drug Plan (PDP) Sponsor
Contract Per PDP Region (§ 423.503)
13. Limit Stand-Alone Prescription Drug
Plan Sponsors to Offering No More Than
Two Plans Per PDP Region (§ 423.265)
14. Applicable Cost-Sharing for Transition
Supplies: Transition Process Under Part
D (§ 423.120(b)(3))
15. Interpreting the Non Interference
Provision (§ 423.10)
16. Pharmacy Price Concessions in
Negotiated Prices (§ 423.100)
17. Preferred Cost Sharing (§§ 423.100 and
423.120)
18. Prescription Drug Pricing Standards
and Maximum Allowable Cost
(§ 423.505(b)(21))
19. Any Willing Pharmacy Standard Terms
& Conditions (§ 423.120(a)(8))
20. Enrollment Requirements for
Prescribers of Part D Covered Drugs
(§ 423.120(c)(5) and (6))
21. Improper Prescribing Practices
(§§ 424.530 and 424.535)
a. Background and Program Integrity
Concerns
b. Drug Enforcement Administration (DEA)
Certification of Registration
c. Patterns or Practices of Prescribing
22. Broadening the Release of Part D Data
(§ 423.505)
23. Establish Authority to Directly Request
Information From First Tier,
Downstream, and Related Entities
(§§ 422.504(i)(2)(i) and 423.505(i)(2)(i))
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00002 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29845
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
24. Eligibility of Enrollment for
Incarcerated Individuals (§§ 417.1,
417.422, 417.460, 422.74, and 423.44)
a. Changes in Definition of Service Area for
Cost Plans (§§ 417.1 and 417.422(b))
b. Involuntary Disenrollment for
Incarcerated Individuals Enrolled in MA,
PDP and cost plans (§§ 417.460, 422.74,
and 423.44)
25. Rewards and Incentives Program
Regulations for Part C Enrollees
(§ 422.134)
B. Improving Payment Accuracy
1. Implementing Overpayment Provisions
of Section 1128J(d) of the Social Security
Act (§§ 422.326 and 423.360)
a. Terminology (§§ 422.326(a) and
423.360(a))
b. General Rules for Overpayments
(§ 422.326(b) through (c); § 423.360(b)
through (c))
c. Look-back Period for Reporting and
Returning Overpayments
2. Risk Adjustment Data Requirements
(§ 422.310)
3. RADV Appeals
a. Background
b. RADV Definitions
c. Publication of RADV Methodology
d. Proposal to Update RADV Appeals
Terminology (§ 422.311)
e. Proposal to Simplify the RADV Appeals
Process
(1) Issues Eligible for RADV Appeal
(2) Issues Not Eligible for RADV Appeals
(3) Manner and Timing of a Request for
RADV Appeal
(4) Reconsideration Stage
(5) Hearing Stage
(6) CMS Administrator Review Stage
f. Proposal to Expand Scope of RADV
Audits
g. Proposal to Clarify the RADV Medical
Record Review Determination Appeal
Burden of Proof Standard
h. Proposal to Change RADV Audit
Compliance Date
4. Recovery Audit Contractor (RAC)
Determination Appeals (Proposed Part
422 Subpart Z and Part 423 Subpart Z)
a. Background
b. Proposed RAC Appeals Process
(1) Reconsiderations (§§ 422.2605 and
423.2605)
(2) Hearing Official Determinations
(§§ 422.2610 and 423.2610)
(3) Administrator Review (§§ 422.2615 and
423.2615)
C. Implementing Other Technical Changes
1. Definition of a Part D Drug (§ 423.100)
a. Combination Products
b. Barbiturates and Benzodiazepines
c. Medical Foods
2. Special Part D Access Rules During
Disasters or Emergencies (§ 423.126)
3. Termination of a Contract Under Parts C
and D (§§ 422.510 and 423.509)
a. Cross-reference Change (§ 423.509(d))
b. Terminology Changes (§§ 422.510 and
423.509)
c. Technical Change to Align Paragraph
Headings (§ 422.510(b)(2))
d. Terminology Change
(§ 423.509(b)(2)(C)(ii))
4. Technical Changes Regarding
Intermediate Sanctions and Civil Money
Penalties (§§ 422.756 and 423.756)
a. Technical Changes to Intermediate
Sanctions Notice Receipt Provisions
(§§ 422.756(a)(2) and 423.756(a)(2))
b. Cross-reference Changes
(§§ 422.756(b)(4) and 423.756(b)(4))
c. Technical Changes (§§ 422.756(d) and
423.756(d))
d. Technical Changes to Align the Civil
Money Penalty Provision with the
Authorizing Statute (§§ 422.760(a)(3) and
423.760(a)(3))
e. Technical Changes to Align the Civil
Money Penalty Notice Receipt Provisions
(§§ 422.1020(a)(2), 423.1020(a)(2),
422.1016(b)(1), and 423.1016(b)(1))
IV. Collection of Information Requirements
A. ICRs Related to Improper Prescribing
Practices and Patterns (§ 424.535(a)(13)
and (14))
B. ICRs Related to Applicants or their
Contracted First Tier, Downstream, or
Related Entities to Have Experience in
the Part D Program Providing Key Part D
Functions (§ 423.504(b)(8)(i) through
(iii))
C. ICRs Related to Eligibility of Enrollment
for Incarcerated Individuals (§§ 417.460,
422.74, and 423.44)
D. ICRs Related to Rewards and Incentives
Program Regulations for Part C Enrollees
(§ 422.134)
E. ICR Related to Recovery Audit
Contractor Determinations (Part 422,
Subpart Z and Part 423, Subpart Z)
V. Regulatory Impact Analysis
A. Statement of Need
B. Overall Impact
C. Anticipated Effects
1. Effects of Closing Cost Contract Plans to
New Enrollment
2. Effects of the Authority to Impose
Intermediate Sanctions and Civil Money
Penalties
3. Effects of Contract Termination
Notification Requirements and Contract
Termination Basis
4. Effects of Reducing the Burden of the
Compliance Program Training
Requirements
5. Effects of the Procedures for Imposing
Intermediate Sanctions and Civil Money
Penalties under Parts C and D
6. Effects of Timely Access to Mail Order
Services
7. Effects of the Modification of the Agent/
Broker Compensation Requirements
8. Effects of Drug Categories or Classes of
Clinical Concern
9. Effects of the Medication Therapy
Management Program (MTMP) under
Part D
10. Effects of the Requirement for
Applicants or their Contracted First Tier,
Downstream, or Related Entities to Have
Experience in the Part D Program
Providing Key Part D Functions
11. Effects of Requirement for Applicants
for Stand Alone Part D Plan Sponsor
Contracts to Be Actively Engaged in the
Business of the Administration of Health
Insurance Benefits
12. Effects of Limit Parent Organizations to
One Prescription Drug Plan (PDP)
Sponsor Contract per PDP Region
13. Effects of Limit Stand-Alone
Prescription Drug Plan Sponsors to
Offering No More Than Two Plans per
PDP Region
14. Effects of Applicable Cost-Sharing for
Transition Supplies: Transition Process
Under Part D
15. Effects of Interpreting the Non-
Interference Provision
16. Effects of Pharmacy Price Concessions
in Negotiated Prices
17. Effects of Preferred Cost Sharing
18. Effects of Maximum Allowable Cost
Pricing Standard
19. Effects of Any Willing Pharmacy
Standard Terms & Conditions
20. Effects of Enrollment Requirements for
Prescribers of Part D Covered Drugs
21. Effects of Improper Prescribing
Practices and Patterns
22. Effects of Broadening the Release of
Part D Data
23. Effects of Establish Authority to
Directly Request Information From First
Tier, Downstream, and Related Entities
24. Effects of Eligibility of Enrollment for
Incarcerated Individuals
25. Effects of Rewards and Incentives
Program Regulations for Part C Enrollees
26. Effects of Improving Payment
Accuracy: Reporting Overpayments,
RADV Appeals, and LIS Cost Sharing
27. Effects of Part C and Part D RAC
Determination Appeals
28. Effects of the Technical Changes to the
Definition of a Part D Drug
29. Effects of the Special Part D Access
Rules During Disasters
30. Effects of Termination of a Contract
under Parts C and D
31. Effects of Technical Changes Regarding
Intermediate Sanctions and Civil Money
Penalties
D. Expected Benefits
1. Drug Categories or Classes of Clinical
Concern
2. Medication Therapy Management
Program under Part D
E. Alternatives Considered
1. Modifying the Agent/Broker
Compensation Requirements
2. Any Willing Pharmacy Standard Terms
and Conditions
3. Pharmacy Price Concessions in
Negotiated Prices
4. Special Part D Access Rules During
Disasters or Emergencies
5. Drug Categories or Classes of Clinical
Concern
6. Medication Therapy Management
Program (MTM) Under Part D
7. Requirement for Applicants or their
Contracted First Tier, Downstream, or
Related Entities to have Experience in
the Part D Program Providing Key Part D
Functions
F. Accounting Statement and Table
G. Conclusion
Regulations Text
Acronyms
ADS Automatic Dispensing System
AEP Annual Enrollment Period
AHFS American Hospital Formulary
Service
AHFS–DI American Hospital Formulary
Service–Drug Information
AHRQ Agency for Health Care Research
and Quality
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00003 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29846
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
ALJ Administrative Law Judge
ANOC Annual Notice of Change
AO Accrediting Organization
AOR Appointment of Representative
BBA Balanced Budget Act of 1997 (Pub. L.
105–33)
BBRA [Medicare, Medicaid and State Child
Health Insurance Program] Balanced
Budget Refinement Act of 1999 (Pub. L.
106–113)
BIPA [Medicare, Medicaid, and SCHIP]
Benefits Improvement Protection Act of
2000 (Pub. L. 106–554)
BLA Biologics License Application
CAHPS Consumer Assessment Health
Providers Survey
CAP Corrective Action Plan
CCIP Chronic Care Improvement Program
CC/MCC Complication/Comorbidity and
Major Complication/Comorbidity
CCS Certified Coding Specialist
CDC Centers for Disease Control
CHIP Children’s Health Insurance Programs
CMP Civil Money Penalty
CMR Comprehensive Medical Review
CMS Centers for Medicare & Medicaid
Services
CMS–HCC CMS Hierarchal Condition
Category
CTM Complaints Tracking Module
COB Coordination of Benefits
CORF Comprehensive Outpatient
Rehabilitation Facility
CPC Certified Professional Coder
CY Calendar year
DAB Departmental Appeals Board
DEA Drug Enforcement Administration
DIR Direct and Indirect Remuneration
DME Durable Medical Equipment
DMEPOS Durable Medical Equipment,
Prosthetic, Orthotics, and Supplies
D–SNPs Dual Eligible SNPs
DOL U.S. Department of Labor
DUA Data Use Agreement
DUM Drug Utilization Management
EAJR Expedited Access to Judicial Review
EGWP Employer Group/Union-Sponsored
Waiver Plan
EOB Explanation of Benefits
EOC Evidence of Coverage
ESRD End-Stage Renal Disease
FACA Federal Advisory Committee Act
FDA Food and Drug Administration
FEHBP Federal Employees Health Benefits
Plan
FFS Fee-For-Service
FIDE Fully-integrated Dual Eligible
FIDE SNPs Fully-integrated Dual Eligible
Special Needs Plans
FMV Fair Market Value
FY Fiscal year
GAO Government Accountability Office
HAC Hospital-Acquired Conditions
HCPP Health Care Prepayment Plans
HEDIS HealthCare Effectiveness Data and
Information Set
HHS [U.S. Department of] Health and
Human Services
HIPAA Health Insurance Portability and
Accountability Act of 1996 (Pub. L. 104–
191)
HMO Health Maintenance Organization
HOS Health Outcome Survey
HPMS Health Plan Management System
ICL Initial Coverage Limit
ICR Information Collection Requirement
ID Identification
IVC Initial Validation Contractor
LCD Local Coverage Determination
LEP Late Enrollment Penalty
LIS Low Income Subsidy
LPPO Local Preferred Provider
Organization
LTC Long Term Care
MA Medicare Advantage
MAAA Member of the American Academy
of Actuaries
MA–PD Medicare Advantage–Prescription
Drug Plan
MIPPA Medicare Improvements for Patients
and Providers Act of 2008 (Pub. L. 110–
275)
MOC Medicare Options Compare
MOOP Maximum Out-of-Pocket
MPDPF Medicare Prescription Drug Plan
Finder
MMA Medicare Prescription Drug,
Improvement, and Modernization Act of
2003 (Pub. L. 108–173)
MS–DRG Medicare Severity Diagnosis
Related Group
MSA Metropolitan Statistical Area
MSAs Medical Savings Accounts
MSP Medicare Secondary Payer
MTM Medication Therapy Management
MTMP Medication Therapy Management
Program
NAIC National Association of Insurance
Commissioners
NCD National Coverage Determination
NCPDP National Council for Prescription
Drug Programs
NCQA National Committee for Quality
Assurance
NDA New Drug Application
NDC National Drug Code
NGC National Guideline Clearinghouse
NIH National Institutes of Health
NOMNC Notice of Medicare Non-Coverage
NPI National Provider Identifier
NWS National Weather Service
OIG Office of Inspector General
OMB Office of Management and Budget
OPM Office of Personnel Management
OTC Over the Counter
Part C Medicare Advantage
Part D Medicare Prescription Drug Benefit
Program
PBM Pharmacy Benefit Manager
PDE Prescription Drug Event
PDP Prescription Drug Plan
PFFS Private Fee For Service Plan
POA Present on Admission (Indicator)
POS Point-of-Sale
PPO Preferred Provider Organization
PPS Prospective Payment System
P&T Pharmacy & Therapeutics
QIC Qualified Independent Contractor
QIO Quality Improvement Organization
QRS Quality Review Study
PACE Programs of All Inclusive Care for the
Elderly
RADV Risk Adjustment Data Validation
RAPS Risk Adjustment Payment System
RPPO Regional Preferred Provider
Organization
SCORM Sharable Content Object Reference
Model
SEP Special Election Period
SHIP State Health Insurance Assistance
Programs
SNF Skilled Nursing Facility
SNP Special Needs Plan
SPAP State Pharmaceutical Assistance
Programs
SSA Social Security Administration
SSI Supplemental Security Income
T&C Terms and Conditions
TPA Third Party Administrator
TrOOP True Out-Of-Pocket
U&C Usual and Customary
UPIN Uniform Provider Identification
Number
USP U.S. Pharmacopoeia
I. Executive Summary
A. Purpose
The purpose of this final rule is to
make revisions to the Medicare
Advantage (MA) program (Part C) and
Prescription Drug Benefit Program (Part
D) regulations based on our continued
experience in the administration of the
Part C and Part D programs and to
implement certain provisions of the
Affordable Care Act. This final rule is
necessary to—(1) clarify various
program participation requirements; (2)
improve payment accuracy; and (3)
make other clarifications and technical
changes.
B. Summary of the Major Provisions
1. Modifying the Agent/Broker
Requirements, Specifically Agent/
Broker Compensation
The former regulatory compensation
structure was comprised of a 6-year
cycle that ended December 31, 2013.
Under that structure, MA organizations
and Part D sponsors provided an initial
compensation payment to independent
agents for new enrollees (Year 1), and
paid a renewal rate (equal to 50 percent
of the initial year compensation) for
Years 2 through 6. MA organizations
and Part D sponsors had the option to
pay the 50 percent renewal rate for
CY2014 (year 1). This compensation
structure proved to be complicated to
implement and monitor, and also
created an incentive for agents to move
beneficiaries as long as the fair market
value (FMV) continued to increase each
year. To resolve these issues, we
proposed to revise the compensation
structure. Under our proposal, MA
organizations and Part D sponsors
would continue to have the discretion to
decide, on an annual basis, whether or
not to use independent agents. Also, for
new enrollments, MA organizations and
Part D sponsors could determine what
their initial rate would be, up to the
CMS designated FMV amount. For
renewals in Year 2 and subsequent
years, with no end date, the MA
organization or Part D sponsor could
pay up to 35 percent of the current FMV
amount for that year. We believed that
revising the existing compensation
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00004 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29847
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
structure to allow MA organizations or
Part D sponsors to pay up to 35 percent
of the FMV for year 2 and subsequent
years was appropriate based on a couple
of factors. First, we believed that a 2
tiered payment system (that is, initial
and renewal) would be significantly less
complicated than a 3-tiered system (that
is, initial, 50 percent renewal for years
2 through 6, and 25 percent residual for
years 7 and subsequent years), and
would reduce administrative burden
and confusion for plan sponsors.
Second, our analysis determined that 35
percent was the renewal compensation
level at which the present value of
overall payments under a 2-tiered
system would be relatively equal to the
present value of overall payments under
a 3-tiered system (taking into account
the estimated life expectancy for several
beneficiary age cohorts). In addition to
revising the agent and broker
compensation structures, we proposed
to amend the training and testing
requirements as well as setting limits on
referral fees ($100) for agents and
brokers.
We received more than 140 comments
from agents, health plans, and trade
associations opposing the 35 percent
renewal rate, and instead suggesting that
CMS maintain the 50 percent renewal
rate. A number of commenters
expressed concerns that the proposed
reduction in compensation would
represent a significant decrease from the
current compensation limit, and a rate
set at 50 percent of FMV would be in
line with industry standard. They noted
that the higher compensation amount
would be particularly important for
stand-alone prescription drug plans, as
35 percent would be insufficient to
cover an agent’s costs associated with
the renewal transaction and could
discourage agents from assisting in the
annual evaluation of a Medicare
beneficiary’s options. Commenters also
stated that, compared to current
practice, the proposed 35 percent
renewal rate is a reduction since a
number of MA plans began offering a
renewal rate of 50 percent for 10 years
or more at the end of the 6-year cycle
(2013). The majority of commenters also
stated that agents play an important role
in educating beneficiaries about
Medicare and the proposed reduction in
the renewal rate could reduce the level
and quality of services provided to
beneficiaries, thereby resulting in less
information sharing and poorer plan
choices by beneficiaries. Many
commenters also stated that agents
spend a significant amount of time in
training, preparing, and educating
beneficiaries and that the compensation
is already low relative to the hours
spent. Some commenters also expressed
concern that the lower compensation
rate would discourage new agents from
entering the MA market. Many agents
stated they would have to stop selling
MA products and instead sell other
more profitable products. No plans
strongly supported the 35 percent
renewal rate. Therefore, we are
modifying the compensation renewal
rate from up to 35 percent to up to 50
percent. These changes will be
applicable for enrollments effective
January 2015. Because the proposed rate
is similar to previous regulatory
requirements, present CMS guidance,
and industry practice, we believe this
implementation timeframe is reasonable
and appropriate. We are not finalizing
the proposed changes to agent and
broker training and testing at this time.
We are finalizing limits on referral fees
for agents as proposed.
2. Drug Categories or Classes of Clinical
Concern
We are not finalizing any new criteria
and will maintain the existing six
protected classes.
3. Improving Payment Accuracy—
Implementing Overpayment Provisions
of Section 1128J(d) of the Social
Security Act (§§ 422.326 and 423.360)
These proposed regulatory provisions
codify the Affordable Care Act
requirement establishing section
1128J(d) of the Act that MA
organizations and Part D sponsors report
and return identified Medicare
overpayments.
We proposed to adopt the statutory
definition of overpayment for both Part
C and Part D, which means any funds
that an MA organization or Part D
sponsor has received or retained under
Title XVIII of the Act to which the MA
organization or Part D sponsor, after
applicable reconciliation, is not entitled
under such title. To reflect the unique
structure of Part C and Part D payments
to plan sponsors, we also propose to
define two terms included in the
statutory definition of overpayments:
‘‘funds’’ and ‘‘applicable
reconciliation.’’ We proposed to define
funds as payments an MA organization
or Part D sponsor has received that are
based on data that these organizations
submitted to CMS for payment
purposes. For Part C we proposed that
applicable reconciliation occurs on the
annual final risk adjustment data
submission deadline. For Part D, we
proposed that applicable reconciliation
occurs on the date that is the later of
either the annual deadline for
submitting prescription drug event
(PDE) data for the annual Part D
payment reconciliations referred to in
§ 423.343(c) and (d) or the annual
deadline for submitting DIR data.
In addition, we proposed to state in
regulation that an MA organization or
Part D sponsor has identified an
overpayment if it has actual knowledge
of the existence of the overpayment or
acts in reckless disregard or deliberate
ignorance of the existence of the
overpayment. An MA organization or
Part D sponsor must report and return
any identified overpayment it received
no later than 60 days after the date on
which it identified it received an
overpayment. The MA organization or
Part D sponsor must notify CMS, using
a notification process determined by
CMS, of the amount and reason for the
overpayment. Finally, we proposed a
look-back period with an exception for
overpayments resulting from fraud,
whereby MA organizations and Part D
sponsors would be held accountable for
reporting overpayments within the 6
most recent completed payment years
for which the applicable reconciliation
has been completed.
We received approximately 30
comments from organizations and
individuals. Generally, commenters
supported establishing separate
applicable reconciliation dates for Part
C and Part D. Many commenters
questioned when the 60-day period for
reporting and returning begins, and
what activities constitute reporting and
returning an overpayment to CMS,
including questions about estimating an
amount of overpayment. A number of
commenters also requested to clarify the
standards for ‘‘identifying’’ an
overpayment, including questions about
the meaning of reasonable diligence.
Finally, a few commenters
recommended that we impose the same
limitation on the look-back period for
all overpayments, even those relating to
fraud.
We are finalizing the provisions at
§§ 422.326 and 423.360, with the
following modifications. First, we add at
the end of paragraph § 422.326(d) the
phrase ‘‘unless otherwise directed by
CMS for the purpose of § 422.311.’’
Also, to increase clarity we revise
§§ 422.326(c) and 423.360(c) regarding
identified overpayments. Finally, we
strike the following sentence in the
proposed paragraphs on the 6-year look-
back period: ‘‘Overpayments resulting
from fraud are not subject to this
limitation of the lookback period.’’
4. Risk Adjustment Data Requirements
We proposed several amendments to
§ 422.310 to strengthen existing
regulations related to the accuracy of
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00005 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29848
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
risk adjustment data, including: (1) A
requirement that medical record
reviews, if used, be designed to
determine the accuracy of diagnoses
submitted under §§ 422.308(c)(1) and
422.310(g)(2); (2) a revision in the
deadlines for submission of risk
adjustment data; and (3) a limitation on
the type and purpose of late data
submissions. We also proposed a
restructuring of subparagraph (g)(2) as
part of the revisions. We received
approximately 25 comments from
organizations and individuals regarding
these proposals; many of the comments
were concerned and critical of the
proposals, highlighting vagueness and
the potential for operational instability.
For reasons discussed in more detail
below in section III.B.2 of the preamble,
we are not finalizing the proposed
amendment regarding the scope of
medical reviews and we are not
finalizing at this time the proposal to
change the date for final risk adjustment
data submission. We are finalizing as
proposed the restructuring of
§§ 422.310(g)(2) and the 422.310(g)(2)(ii)
provision to prohibit submission of
diagnoses for additional payment after
the final risk adjustment data
submission deadline.
C. Summary of Costs and Benefits
T
ABLE
2—S
UMMARY OF
C
OSTS AND
B
ENEFITS
Provision description Total costs Transfers
Modifying the agent/broker require-
ments, specifically agent/broker
compensation.
N/A ............... N/A
Improving Payment Accuracy ........... N/A ............... N/A
Eligibility of Enrollment for Incarcer-
ated Individuals.
................. We estimate that this change could save the MA program up to $27 million in 2015, in-
creasing to $103 million in 2024 (total of $650 million over this period), and could save
the Part D program (includes the Part D portion of MA PD plans) up to $46 million in
2015, increasing to $153 million in 2024 (total of $965 million over this period).
II. Background
A. General Overview and Regulatory
History
The Balanced Budget Act of 1997
(BBA) (Pub. L. 105–33) created a new
‘‘Part C’’ in the Medicare statute
(sections 1851 through 1859 of the
Social Security Act (the Act)) which
established what is now known as the
Medicare Advantage (MA) program. The
Medicare Prescription Drug,
Improvement, and Modernization Act of
2003 (MMA) (Pub. L. 108–173), enacted
on December 8, 2003, added a new ‘‘Part
D’’ to the Medicare statute (sections
1860D–1 through 42 of the Act) entitled
the Medicare Prescription Drug Benefit
Program (PDP), and made significant
changes to the existing Part C program,
which it named the Medicare Advantage
(MA) Program. The MMA directed that
important aspects of the Part D program
be similar to, and coordinated with,
regulations for the MA program.
Generally, the provisions enacted in the
MMA took effect January 1, 2006. The
final rules implementing the MMA for
the MA and Part D prescription drug
programs appeared in the Federal
Register on January 28, 2005 (70 FR
4588 through 4741 and 70 FR 4194
through 4585, respectively).
Since the inception of both Parts C
and D, we have periodically revised our
regulations either to implement
statutory directives or to incorporate
knowledge obtained through experience
with both programs. For instance, in the
September 18, 2008 and January 12,
2009 Federal Register (73 FR 54226 and
74 FR 1494, respectively), we issued
Part C and D regulations to implement
provisions in the Medicare
Improvement for Patients and Providers
Act (MIPPA) (Pub. L. 110–275). We
promulgated a separate interim final
rule in January 16, 2009 (74 FR 2881) to
address MIPPA provisions related to
Part D plan formularies. In the final rule
that appeared in the April 15, 2010
Federal Register (75 FR 19678), we
made changes to the Part C and D
regulations which strengthened various
program participation and exit
requirements; strengthened beneficiary
protections; ensured that plan offerings
to beneficiaries included meaningful
differences; improved plan payment
rules and processes; improved data
collection for oversight and quality
assessment; implemented new policies;
and clarified existing program policy.
In a final rule that appeared in the
April 15, 2011 Federal Register (76 FR
21432), we continued our process of
implementing improvements in policy
consistent with those included in the
April 2010 final rule, and also
implemented changes to the Part C and
Part D programs made by recent
legislative changes. The Patient
Protection and Affordable Care Act
(Pub. L. 111–148) was enacted on March
23, 2010, as passed by the Senate on
December 24, 2009, and the House on
March 21, 2010. The Health Care and
Education Reconciliation Act (Pub. L.
111–152), which was enacted on March
30, 2010, modified a number of
Medicare provisions in Pub. L. 111–148
and added several new provisions. The
Patient Protection and Affordable Care
Act (Pub. L. 111–148) and the Health
Care and Education Reconciliation Act
(Pub. L. 111–152) are collectively
referred to as the Affordable Care Act.
The Affordable Care Act included
significant reforms to both the private
health insurance industry and the
Medicare and Medicaid programs.
Provisions in the Affordable Care Act
concerning the Part C and D programs
largely focused on beneficiary
protections, MA payments, and
simplification of MA and Part D
program processes. These provisions
affected implementation of our policies
regarding beneficiary cost-sharing,
assessing bids for meaningful
differences, and ensuring that cost-
sharing structures in a plan are
transparent to beneficiaries and not
excessive. In the April 2011 final rule,
we revised regulations on a variety of
issues based on the Affordable Care Act
and our experience in administering the
MA and Part D programs. The rule
covered areas such as marketing,
including agent/broker training;
payments to MA organizations based on
quality ratings; standards for
determining if organizations are fiscally
sound; low income subsidy policy
under the Part D program; payment
rules for non-contract health care
providers; extending current network
adequacy standards to Medicare
medical savings account (MSA) plans
that employ a network of providers;
establishing limits on out-of-pocket
expenses for MA enrollees; and several
revisions to the special needs plan
requirements, including changes
concerning SNP approvals.
In a final rule that appeared in the
April 12, 2012 Federal Register (77 FR
22072 through 22175), we made several
changes to the Part C and Part D
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00006 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29849
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
programs required by statute, including
the Affordable Care Act, as well as made
improvements to both programs through
modifications reflecting experience we
have obtained administering the Part C
and Part D programs. Key provisions of
that final rule implemented changes
closing the Part D coverage gap, or
‘‘donut hole,’’ for Medicare beneficiaries
who do not already receive low-income
subsidies from us by establishing the
Medicare Coverage Gap Discount
Program. We also included provisions
providing new benefit flexibility for
fully-integrated dual eligible special
needs plans, clarifying coverage of
durable medical equipment, and
combatting possible fraudulent activity
by requiring Part D sponsors to include
an active and valid prescriber National
Provider Identifier on prescription drug
event records.
B. Issuance of a Notice of Proposed
Rulemaking
In the proposed rule titled ‘‘Contract
Year 2015 Policy and Technical
Changes to the Medicare Advantage and
the Medicare Prescription Drug Benefit
Programs,’’ which appeared in the
January 10, 2014 Federal Register (79
FR 1918), we proposed to revise the
Medicare Advantage (MA) program (Part
C) regulations and prescription drug
benefit program (Part D) regulations to
implement statutory requirements;
strengthen beneficiary protections;
exclude plans that perform poorly;
improve program efficiencies; and
clarify program requirements. The
proposed rule also included several
provisions designed to improve
payment accuracy.
C. Public Comments Received in
Response to the CY 2015 Policy and
Technical Changes to the Medicare
Advantage and the Medicare
Prescription Drug Benefit Programs
Proposed Rule
We received approximately 7,600
timely pieces of correspondence
containing multiple comments on the
CY 2014 proposed rule. While we are
finalizing several of the provisions from
the proposed rule, there are a number of
provisions from the proposed rule (for
example, enrollment eligibility criteria
for individuals not lawfully present in
the United States) that we intend to
address later and a few which we do not
intend to finalize. We also note that
some of the public comments were
outside of the scope of the proposed
rule. These out-of-scope public
comments are not addressed in this final
rule. Summaries of the public comments
that are within the scope of the
proposed rule and our responses to
those public comments are set forth in
the various sections of this final rule
under the appropriate heading.
However, we note that in this final rule
we are not addressing comments
received with respect to the provisions
of the proposed rule that we are not
finalizing at this time. Rather, we will
address them at a later time, in a
subsequent rulemaking document, as
appropriate.
D. Provisions Not Finalized in This
Final Rule
As noted previously, some of the
provisions of the proposed rule will be
addressed later and, therefore, are not
being finalized in this rule. Table 3 lists
the provisions that were proposed but
are not addressed at this time. We note
that several provisions that were
proposed are not being finalized in this
rule and are effectively being
withdrawn; those provisions are not
listed in Table 3.
T
ABLE
3—P
ROVISIONS
N
OT
F
INALIZED AT
T
HIS
T
IME
Proposed
rule section
Topic
Clarifying Various Program Participation Requirements
III.A.2 .......... Two-year Limitation on Submitting a New Bid in an Area Where an MA has been Required to Terminate a Low-enrollment MA
Plan (§ 422.504(a)(19)).
III.A.6 .......... Changes to Audit and Inspection Authority (§ 422.503(d)(2) and § 423.504(d)(2)).
III.A.9 .......... Collections of Premiums and Cost Sharing (§ 423.294).
III.A.10 ........ Enrollment Eligibility for Individuals Not Lawfully Present in the United States (§§ 417.2, 417.420, 417.422, 417.460, 422.1,
422.50, 422.74, 423.1, 423.30, and 423.44).
III.A.11 ........ Part D Notice of Changes (§ 423.128(g)).
III.A.12 ........ Separating the Annual Notice of Change (ANOC) from the Evidence of Coverage (EOC) (§ 422.111(a)(3) and § 423.128(a)(3)).
III.A.14 ........ Exceptions to Drug Categories or Classes of Clinical Concern (§ 423.120(b)(2)(vi)).
III.A.15 ........ Medication Therapy Management Program (MTMP) under Part D (§ 423.153(d)(1)(v)(A))—outreach strategies.
III.A.16 ........ Business Continuity for MA Organizations and PDP Sponsors (§ 422.504(o) and § 423.505(p)).
III.A.21 ........ Efficient Dispensing in Long Term Care Facilities and Other Changes (§ 423.154).
III.A.23 ........ Medicare Coverage Gap Discount Program and Employer Group Waiver Plans (§ 423.2325).
III.A.26 ........ Payments to PDP Plan Sponsors For Qualified Prescription Drug Coverage (§ 423.308) and Payments to Sponsors of Retiree
Prescription Drug Plans (§ 423.882).
III.A.32 ........ Transfer of TrOOP Between PDP Sponsors Due to Enrollment Changes during the Coverage Year (§ 423.464).
III.A.37 ........ Expand Quality Improvement Program Regulations (§ 422.152).
III.A.38 ........ Authorization of Expansion of Automatic or Passive Enrollment Non-Renewing Dual Eligible SNPs (D-SNPs) to another D-SNP to
Support Alignment Procedures (§ 422.60).
Improving Payment Accuracy
III.B.2 .......... Determination of Payments (§ 423.329).
III.B.3 .......... Reopening (§ 423.346).
III.B.4 .......... Payment Appeals (§ 423.350).
III.B.5 .......... Payment Processes for Part D Sponsors (§ 423.2320).
III.B.6 .......... Risk adjustment data requirements—proposal regarding annual deadline for MAO submission of final risk adjustment data
(§ 422.310(g)(2)(ii)).
Strengthening Beneficiary Protections
III.C.1 .......... Providing High Quality Health Care (§ 422.504(a)(3) and § 423.505(b)(27)).
III.C.2 .......... MA-PD Coordination Requirements for Drugs Covered Under Parts A, B, and D (§ 422.112).
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00007 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29850
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
T
ABLE
3—P
ROVISIONS
N
OT
F
INALIZED AT
T
HIS
T
IME
—Continued
Proposed
rule section
Topic
III.C.3 .......... Good Cause Processes (§ 417.460, § 422.74 and § 423.44).
III.C.4 .......... Definition of Organization Determination (§ 422.566).
III.C.5 .......... MA Organizations May Extend Adjudication Timeframes for Organization Determinations and Reconsiderations (§ 422.568,
§ 422.572, § 422.590, § 422.618, and § 422.619).
Strengthening Our Ability to Distinguish Stronger Applicants for Part C and D Program Participation and to Remove Consistently Poor
Performers
III.D.1 .......... Two-Year Prohibition When Organizations Terminate Their Contracts (§§ 422.502, 422.503, 422.506, 422.508, and 422.512).
III.D.2 .......... Withdrawal of Stand-Alone Prescription Drug Plan Bid Prior to Contract Execution (§ 423.503).
III.D.3 .......... Essential Operations Test Requirement for Part D (§§ 423.503(a) and (c), 423.504(b)(10), 423.505(b)(28), and 423.509).
III.D.4. ......... Termination of the Contracts of Medicare Advantage Organizations Offering PDP for Failure for 3 Consecutive Years to Achieve
3 Stars on Both Part C and Part D Summary Star Ratings in the Same Contract Year (§ 422.510).
Implementing Other Technical Changes
III.E.1 .......... Requirements for Urgently Needed Services (§ 422.113).
III.E.2 .......... Skilled Nursing Facility Stays (§§ 422.101 and 422.102).
III.E.3 .......... Agent and Broker Training and Testing Requirements (§§ 422.2274 and 423.2274).
III.E.4 .......... Deemed Approval of Marketing Materials (§ 422.2266 and § 423.2266).
III.E.5 .......... Cross-Reference Change in the Part C Disclosure Requirements (§ 422.111).
III.E.6 .......... Managing Disclosure and Recusal in P&T Conflicts of Interest: [Formulary] Development and Revision by a Pharmacy and Thera-
peutics Committee under PDP (§ 423.120(b)(1)).
III.E.8 .......... Thirty-Six-Month Coordination of Benefits (COB) Limit (§ 423.466(b)).
III.E.9 .......... Application and Calculation of Daily Cost-Sharing Rates (§ 423.153).
III.E.10 ........ Technical Change to Align Regulatory Requirements for Delivery of the Standardized Pharmacy Notice (§ 423.562).
III.E.12 ........ MA Organization Responsibilities in Disasters and Emergencies (§ 422.100).
III.E.14 ........ Technical Changes to Align Part C and Part D Contract Determination Appeal Provisions (§§ 422.641 and 422.644).
III.E.15 ........ Technical Changes to Align Parts C and D Appeal Provisions (§§ 422.660 and 423.650).
III.E.17 ........ Technical Change to the Restrictions on use of Information under Part D (§ 423.322).
III. Provisions of the Proposed
Regulations and Analysis of and
Responses to Public Comments
A. Clarifying Various Program
Participation Requirements
1. Closing Cost Contract Plans to New
Enrollment (§ 422.503(b)(4))
To ensure that our original intent is
realized and to eliminate the potential
for organizations to move enrollees from
one of their plans to another based on
financial or some other interest, we
proposed to revise paragraph
§ 422.503(b)(4)(vi)(G)(5) so that an
‘‘entity seeking to contract as an MA
organization must [n]ot accept, or share,
a corporate parent organization with an
entity that accepts, new enrollees under
a section 1876 reasonable cost contract
in any area in which it seeks to offer an
MA plan.’’
In making the proposed revision to
paragraph § 422.503(b), we also
proposed to add the definition of
‘‘parent organization’’ to § 422.2 of the
MA program definitions, specifying
that, ‘‘Parent organization means a legal
entity that owns one or more other
subsidiary legal entities.’’ Although the
MA program regulations do not
currently define the term ‘‘parent
organization,’’ our proposed definition
is consistent with the way the term is
currently used in the context of the MA
program, for example, when assessing
an organization’s business structure. We
requested comments on whether a
parent organization with less than a 100
percent interest in a subsidiary legal
entity should trigger the prohibition we
proposed with the amendment at
§ 422.503(b)(4).
During the public notice and
comment process, a handful of
commenters provided their input on our
proposal. Some of the respondents
included multiple comments. The
comments and our responses follow.
Comment: A commenter supported
the proposal, stating that it would
prevent possible shifting of sicker
enrollees to cost plans and should result
in Medicare savings.
Response: We thank the commenter
for the support.
Comment: A commenter stated that
there is no evidence of complaints about
the current situation and thus no change
in current policy is necessary.
Response: The intention of our initial
rule was to ensure that situations not
arise in which an entity was able to
move an enrollee from one of its plans
to another plan in the same area based
on financial or other reasons that may
not be in the enrollee’s best interest. The
current regulations limit this possibility
to some extent, but, without the
proposed changes, would leave open the
possibility that legal entities controlled
by a shared parent organization could
move enrollees from one plan to
another, based on something other than
the enrollee’s best interest.
Comment: A commenter stated that
risk-adjusted payments for MA plans
eliminate any incentive for an entity to
move sicker enrollees from an MA plan
to a cost plan.
Response: While risk adjusted
payments do help to account for costs
associated with sicker enrollees, it may
still be advantageous for an organization
to move an enrollee from an MA plan
to a cost plan. Even with risk
adjustment, there are other reasons an
organization might want to move
enrollees from one plan to another to
include enrollment and other interests
based on the organization’s business
model.
Comment: A commenter stated that,
because cost plan cost-sharing and
premiums must be equal to the actuarial
value of Medicare fee-for-service cost-
sharing, cost plan enrollees with high
health care needs would have high
relative costs resulting in higher
premiums for the cost plan, thus
removing any incentive for moving
sicker enrollees from an entity’s MA
plan to the cost plan.
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00008 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29851
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
Response: MA plans also have
constraints with respect to cost-sharing
that affect premiums, and out-of-pocket
payments by enrollees. We believe, as a
result, that any difference in cost plan
and MA premiums or cost-sharing is
negligible and does little to remove the
incentives for organizations to move
enrollees from one of their plans to
another.
Comment: A couple of commenters
requested that, at minimum, the
provision not be applied to entities that
have both a cost plan and dual eligible
special needs plan (D–SNP). One of the
commenters states that: (1) cost plans
would likely have a premium and cost
sharing that would make it unattractive
for dual eligibles; and (2) the regulation
could eliminate D–SNPs that
‘‘participate in longstanding dual
eligible integrated plans,’’ and thus the
proposal ‘‘could have the effect of
hurting a major initiative of the
Administration.’’
Response: As we have addressed
elsewhere in the comments on this
issue, we do not believe that any
premium and cost-sharing differences in
cost plans and MA plans necessarily
reduce the incentives an organization
may have for moving an individual from
one of its plans to another. We believe
this is also the case for D–SNPs and,
that in the case of D–SNPs, which are
frequently made up of enrollees that are
sicker and frailer than the general
Medicare population, there may be even
greater incentive to move these
enrollees to a cost contract plan.
Comment: A commenter requested
that we not finalize the proposal
because cost plan enrollees will already
be subject to dwindling cost plan
enrollment options as a result of the cost
plan competition statute. The
commenter stated that if we do finalize
the proposal, we should grant an
exception and not require cost plans
affected by the cost plan competition
requirements to close to new
enrollment.
Response: It isn’t clear at this point
what kind of overlap there might be
between cost plans affected by the cost
plan competition requirements and
those cost plans that would have to stop
accepting enrollment because of sharing
a parent organization with an MA plan.
However, we do not believe that a
significant number of cost plans will be
affected by expanding the requirement
to include a shared parent organization,
as the requirement is largely prospective
and designed to prevent a situation that
we did not originally account for, but
which we believe could lead to
potential harm for enrollees.
Comment: A commenter stated that
‘‘the test should not only be whether
entities have the same parent but also
whether the two entities are affiliated,
including if one entity is the parent of
the other (rather than shares a parent).’’
Response: We agree with the
commenter with respect to the specific
example cited and have included
language in the final rule that will also
trigger a prohibition on new enrollment
in a cost plan in situations in which a
parent organization and its subsidiary
have a cost contract and MA plan in the
same service area. In addition to the
proposed language that MA
organizations ‘‘Not accept, or share a
corporate parent organization with an
entity that accepts, new enrollees under
a section 1876 reasonable cost contract
in any area in which it seeks to offer an
MA plan,’’ we are adding to § 422.503
(b)(4)(vi)(G)(5)(ii) that MA organizations
‘‘Not accept, as either the parent
organization owning a controlling
interest of or subsidiary of an entity that
accepts, new enrollees under a section
1876 reasonable cost contract in any
area in which it seeks to offer an MA
plan.’’ The language from the initial
proposal along with the additional
language will now be contained in
§ 422.503 (b)(4)(vi)(G)(5)(i) and (ii).
Comment: A few commenters stated
that CMS should define a parent
organization as an entity that ‘‘exercises
a controlling interest in the applicant.’’
Other commenters stated that we should
limit the definition of ‘‘parent
organization’’ to the context of this
provision only as our proposed
definition could create inconsistencies
in the Part C and D polices and
guidance or have ‘‘unanticipated
implications that are difficult to identify
at this time.’’ One of the commenters,
who asked us to limit the application of
the ‘‘parent organization’’ definition to
this provision only, stated that it would
support our proposal if we clarified that
the parent organization must have a
‘‘controlling interest’’ in the subsidiary
legal entities in question.
Response: In the proposed rule, we
specifically solicited comments on
whether the requirement should be
applied to a parent organization with
less than 100 percent interest in the
affected cost contract and MA plan. We
agree that a controlling interest is a
reasonable standard that is consistent
with our intention to prevent an
organization from having control over
both a cost contract and MA plan in the
same service area. We also agree that the
threshold for determining when the
prohibition should be applied is best
established in the context of this
provision and thus are not finalizing the
definition of ‘‘parent organization’’ in
§ 422.2 . Instead, we are including the
threshold for the prohibition in
modifications in
§ 422.503(b)(4)(vi)(G)(5)(i) and (ii).
These sections will now state that any
entity seeking to contract as an MA
organization—
• Not accept, or share a corporate
parent organization owning a
controlling interest in an entity that
accepts, new enrollees under a section
1876 reasonable cost contract in any
area in which it seeks to offer an MA
plan.
• Not accept, as either the parent
organization owning a controlling
interest of, or subsidiary of, an entity
that accepts, new enrollees under a
section 1876 reasonable cost contract in
any area in which it seeks to offer an
MA plan.
We are finalizing the provisions of the
proposed rule with the revisions and
additions discussed in this section
III.A.1 of this final rule.
2. Authority To Impose Intermediate
Sanctions and Civil Money Penalties
(§§ 422.752, 423.752, 422.760 and
423.760)
Sections 1857(a) and 1860D–12(b)(1)
of the Act provided the Secretary with
the authority to enter into contracts with
MA organizations, and Part D sponsors
(respectively). Section 1857(g)(1) of the
Act provided a list of contract violations
and the corresponding enforcement
responses (intermediate sanctions
(sanctions) and/or civil money penalties
(CMPs)) are listed under section
1857(g)(2) of the Act (section 1860D–
12(b)(3)(E) applied these provisions to
Part D contracts).
We proposed two changes to our
existing authority to impose sanctions
and CMPs based on section 6408 of the
Affordable Care Act (Pub. L. 111–148).
The provisions of section 6408 provided
CMS with the authority to impose
intermediate sanctions or CMPs for
violations of the Part C and D marketing
and enrollment requirements. As well
as, an organization that enrolls an
individual without prior consent
(except in certain limited
circumstances) or transfers an
individual to a new plan without prior
consent. Additionally, we proposed to
revise the language of these provisions
to clarify that either CMS or the OIG
may impose CMPs for the violations
listed at §§ 422.752(a) and 423.752(a),
except 422.752(a)(5) and 423.752(a)(5).
Comment: A commenter expressed
concern and stated that MA
organizations and Part D sponsors
should be given the opportunity to
refute marketing or other allegations of
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00009 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29852
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
non-compliance prior to sanctions and/
or CMPs being imposed.
Response: Enforcement actions are
only typically taken based on
substantiated, well documented
instances of non-compliance and in the
case of both a sanction and a CMP, even
after they are issued, MA organizations
and Part D sponsors are given an
opportunity to rebut or appeal CMS’
determination through a formal appeals
process.
Comment: A few commenters
requested clarification regarding the
new sanction authority, specifically the
language that would allow CMS to
impose intermediate sanctions on an
organization that enrolls an individual
without prior consent (except in certain
limited circumstances) or transfers an
individual to a new plan without prior
consent. The commenters requested that
CMS clarify that this would not apply
to organizations that perform facilitated
or auto-enrollment, passive enrollment,
seamless enrollment or requests from
Employer Group Waiver Plans (EGWPs).
Response: In the proposed rule, we
proposed to amend the regulation text at
§§ 422.752 and 423.752 by adding (a)(9),
and (a)(7), respectively, which read:
‘‘. . . Except as provided under § 423.34
of this chapter, enrolls an individual in
any plan under this part without the
prior consent of the individual or the
designee of the individual.’’ Section
423.34 specifically refers to enrollment
of individuals who receive the low
income subsidy (LIS) and are therefore
subject to facilitated or auto-enrollment.
Therefore, we believe that the proposed
regulation text already makes clear that
this provision would not apply to those
organizations that are performing
facilitated enrollment of LIS
beneficiaries. Additionally, passive
enrollment and use of the seamless
enrollment option are initiated or
approved by CMS, respectively.
Therefore, an organization who is
contacted by CMS to receive passive
enrollment would not be considered to
have performed enrollment without
prior consent. As for the seamless
enrollment option, as these proposals
must be submitted to and approved by
CMS, as long as organizations are
following CMS’ enrollment guidance in
Chapter 2, § 40.1.4, an organization,
again, would not be considered as
enrolling without prior consent and
would, therefore, not be considered for
a possible sanction. Finally, an
organization who is accepting group or
individual enrollment requests from
EGWPs must follow CMS’ enrollment
guidance in Chapter 2, § 40.1.6. As long
as CMS enrollment guidance is being
followed with respect to processing
these enrollments, CMS would not
consider MA and Part D organizations
in violation of the new requirement.
Comment: One commenter stated that
only one organization, either CMS or
OIG should have CMP authority and
that there should be no overlapping
authority. They went on to state that if
CMS proposed to allow overlapping
CMP authority that CMS agree that the
total amount of the CMPs issued not
exceed what either CMS or OIG could
impose separately.
Response: It is not CMS’ intent to
create overlapping CMP authority,
simply to clarify our existing CMP
authority. However, to the extent CMS
or OIG were planning on pursuing a
CMP, we have internal mechanisms in
place to ensure that the other entity
within the department is not
simultaneously pursuing a CMP for the
same or similar conduct. If we were to
determine that OIG was pursuing a CMP
for similar conduct, we would
coordinate with the OIG so that only
one CMP action would move forward.
Comment: One commenter requested
that CMS not finalize this provision
because they believe the current
division of authority to impose CMPs
should remain unchanged, with the
authority to CMP for certain violations
remaining with OIG, instead of adding
to CMS’ existing CMP authority, as this
approach ensures a natural division of
power and oversight expected from
government agencies.
Response: CMS has always had the
statutory authority to impose CMPs for
the violations currently designated as
belonging solely to the OIG in the
regulation. However, CMS agrees that
there are certain violations that should
be retained solely by OIG for purposes
of imposing CMPs, which is why the
proposed rule states that the authority to
impose CMPs for violations listed at
§§ 422.752(a)(5) and 423.752(a)(5),
involving misrepresentation or
falsification of information furnished to
CMS, an individual, or other entity, will
continue to reside solely with the OIG.
Comment: One commenter, in
addition to expressing support for our
proposal, stated that CMS should
authorize use of monies collected from
CMPs to allow states to contract with, or
grant funds to entities, provided that the
funds are used for CMS approved
projects to protect or improve SNF
services for residents.
Response: We thank the commenter
for their support and we will explore in
the future if such arrangements are
allowed within our current statutory
authority.
Comment: We received several
comments that supported the new
proposed sanction authority for
marketing and enrollment violations.
Response: We thank the commenters
for their support.
After careful consideration of all of
the comments we received, we are
finalizing these proposals without
modification.
3. Contract Termination Notification
Requirements and Contract Termination
Basis (§§ 422.510 and 423.509)
Sections 1857(c) and 1860D–
12(b)(3)(B) of the Act provided us with
the authority to terminate a Part C or D
sponsoring organization’s contract.
Sections 1857(h)(1)(B) and 1860D–
12(b)(3)(F) of the Act provided us with
the procedures necessary to facilitate
the termination of those contracts. We
proposed three revisions to our existing
regulations that relate to contract
termination.
First, we proposed to clarify the scope
of our authority to terminate Part C and
D contracts under §§ 422.510(a) and
423.509(a) by modifying the language at
§§ 422.510(a) and 423.509(a) to separate
the statutory bases for termination from
our examples of specific violations
which meet the standard for termination
established by the statute. We proposed
to effectuate this change by renumbering
the list of bases contained in
§§ 422.510(a) and 423.509(a).
Second, we proposed revisions to our
contract termination notification
procedures contained at §§ 422.510(b)(1)
and 423.509(b)(1). Current regulations
state that if CMS decides to terminate a
Part C or Part D sponsoring
organization’s contract, we must notify
the organization in writing 90 days
before the intended date of termination.
We proposed to shorten the notification
timeframe from 90 days to 45 days.
Additionally, in an effort to respond to
changes in the media and information
technology landscape, we proposed a
slight modification to the termination
notification provision for the general
public at §§ 422.510(b)(1)(iii) and
423.509(b)(1)(iii) which includes the
contracting organizations releasing a
press statement to news media serving
the affected community or county and
posting the press statement prominently
on the organization’s Web site instead of
publishing the notice in applicable
newspapers.
Finally, we proposed minor revisions
to the wording of our regulations at
§§ 422.510 and 423.509 to reflect the
authorizing language contained in
sections 1857(c)(2) and 1860D–12 of the
Act. Specifically, we proposed to
replace the word ‘‘fails’’ with ‘‘failed’’
so that it reads consistently throughout
§§ 422.510 and 423.509.
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00010 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29853
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
Comment: Several commenters
opposed our proposal to shorten the
notification period for contract
termination from 90 days to 45 days.
Commenters made several arguments
supporting their opposition to the
shortened notification timeframe, but
most stated that it is not enough time to
ensure members’ needs are adequately
addressed, specifically noting the
difficulty in effectively communicating
the change with their members and
ensuring their members were effectively
transitioned to a new plan. Other
commenters stated that the timeframe
was too short to provide adequate notice
to affected providers and vendors. Yet
another commenter stated that the
shortened timeframe did not allow
enough time for a plan to appeal the
termination. A final commenter noted
that the shortened timeframe would
increase costs to the contracting
organization if the termination period is
reduced.
Response: After carefully considering
the commenters’ concerns, we
respectfully disagree that these concerns
outweigh the need to protect
beneficiaries and have them moved
from a plan that is in such substantial
non-compliance with our regulations
that CMS would proceed with
termination. Plans that receive a notice
of termination from CMS are instructed
that they must provide notice to their
affected beneficiaries at least 30 days
prior to the effective date of the
termination. If CMS provides their
notice of termination to contracting
organizations 45 days before the
effective date of the termination, this
affords plans 15 days to issue their
notice to enrollees while still complying
with the existing 30-day beneficiary
notification requirements. While we do
request that terminated plans work with
the receiving plan to transition enrollee
data and records, it is not expected that
these tasks would be completed by the
effective date of the termination, but
would instead begin upon transfer of the
enrollees once the termination was
actually effective.
As for adequate notification to
affected vendors and providers, it is the
responsibility of the contracting
organization to design their contracts
with their providers and vendors in a
manner that recognizes possible
contract actions, such as termination,
that could be taken by CMS. For
example, all plans that have a contract
with CMS could ultimately be subject to
immediate termination if they are found
in such substantial non-compliance by
CMS that it poses an imminent and
serious risk to Medicare enrollees.
Therefore, most, if not all plans, likely
have clauses in their provider and
vendor contracts that allow them to
terminate these contracts expeditiously
with the affected entities in the event of
a contract termination by CMS.
We also do not agree that the
shortened timeframe in any way affects
a contracting organization’s ability to
appeal. Contracting organizations who
are subject to a contract termination in
§§ 422.510(b) or 423.509(b) must file
their request for a hearing within 15
days from the date of receipt of the
notice of termination. A timely filed
request for hearing effectively stays the
termination proceeding until a hearing
decision is reached. Consequently,
shortening the notice of termination
from 90 to 45 days should have no
impact on a contracting organization’s
ability to file an appeal of the contract
termination.
Finally, we do not agree that the
shortened notice timeframe to effectuate
a termination would result in increased
costs to an organization. We already
have the ability to prorate its payment
to an organization for terminations that
are effective in the middle of a month;
consequently we do not agree that
shortening the notification timeframe
would in any way change the CMS’s
current approach to payment or
recoupment of capitated payments in
these circumstances.
Comment: One commenter suggested
that CMS should have different
notification timeframes for termination.
They recommended that 90 day notice
be provided to all post-acute care (PAC)
providers as well as to beneficiaries in
PAC. They stated that 45 days for notice
may be sufficient for non-post-acute
care beneficiaries, but not for people in
a short stay setting. They also suggested
that MA plans that are serving full dual
eligible beneficiaries should be required
to provide 180 day notice to individuals
and providers.
Response: CMS’ proposal to shorten
the notification of termination from 90
days to 45 days affects the amount of
notice that CMS must give to an MA or
Part D organization prior to moving
forward with a termination action. The
timeframe in which that organization
must then notify their beneficiaries,
which is currently 30 days, is not being
changed in this proposal. While we
appreciate the commenter’s suggestion,
we believe that it would be incredibly
burdensome to organizations and
confusing to our beneficiaries to
implement such a striated notification
process for our beneficiaries during a
termination. Additionally, if we were to
adopt the commenter’s suggestion of a
90 day notice period for beneficiaries in
a PAC setting or 180 day notice for dual
eligible beneficiaries, this would require
that we give organizations even more
advance notice of our intent to
terminate than we do currently, which
is contrary to the ultimate goal of our
proposal, which is to remove
beneficiaries as quickly as possible from
a plan with such significant
noncompliance issues that CMS is
pursuing termination. Consequently, we
plan to proceed with our proposed
change.
Comment: Several commenters
supported CMS’ proposed revisions to
the contract termination authority
(§§ 422.510 and 423.509) and stated that
these measures will help enforce
consumer protections and enhance plan
accountability.
Response: We thank the commenters
for their support.
After consideration of the public
comment(s) received, we are finalizing
these proposals without modification.
We note that the amendatory instruction
to the regulation text in this final rule
more precisely describes the
redesignation of subparagraph (a)(4) of
§ 423.509 than that found in the
proposed rule.
4. Reducing the Burden of the
Compliance Program Training
Requirements (§§ 422.503(b)(4)(vi)(C)
and 423.504(b)(4)(vi)(C))
Section 1857(a) and section 1860D–
12(b)(1) of the Act provided the
Secretary with the authority to enter
into contracts with MA organizations
and Part D sponsors (respectively).
Sections 1860D–12(b)(3)(D)(i) and
1857(e)(1) of the Act, specify that these
contracts shall contain other terms and
conditions that the Secretary may find
necessary and appropriate. We first
established that all Part C and Part D
contracting organizations have the
necessary administrative and
management arrangements to have an
effective compliance program, as
reflected in §§ 422.503(b)(4)(vi) and
423.504(b)(4)(vi). We later established
that compliance plans for sponsoring
organizations must include training and
education and effective lines of
communication between the compliance
officer and the sponsoring
organization’s employees, managers,
and directors, as well as their first-tier,
downstream and related entities (FDRs).
We reiterated the importance of this
requirement in the October 22, 2009
proposed rule entitled, ‘‘Medicare
Program; Policy and Technical Changes
to the Medicare Advantage and the
Medicare Prescription Drug Benefit
Programs’’ (74 FR 53634). We were
concerned that these FDRs would
potentially have to participate in
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00011 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29854
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
(largely duplicative) training for each
organization with whom they contract.
We requested public comments on how
best to ensure that the training
requirement continued to be met while
not overly burdening the contracting
organization or its FDRs. In response,
we received numerous comments
suggesting that CMS develop its own
web-based trainings to lessen this
burden on sponsors and FDRs (75 FR
19688).
Consequently, we proposed in this
rule to require that all contracting
organizations accept a certificate of
completion of the CMS developed
training as satisfaction of this general
compliance program training
requirement. We proposed to modify the
regulation text by adding a new
§§ 422.503(b)(vi)(C)(3) and
423.504(b)(vi)(C)(4) to permit only this
CMS training for satisfaction of the
requirement to train first–tier,
downstream and related entities.
Comment: One commenter questioned
if there would be a fee associated with
the CMS mandated training.
Response: There is no fee to take the
CMS Standardized General Compliance
Program Training; it is provided free of
charge.
Comment: Multiple commenters
stated that Part C and Part D contracting
organizations should have the option of
using the CMS Standardized General
Compliance Program Training and
Education Module. The commenters
wrote that there should be flexibility in
meeting the proposed training
requirement, and that CMS should
consider allowing plan sponsors to
utilize their own training or the training
developed by established training
companies to meet the requirement.
Response: The CMS Standardized
General Compliance Program Training
and Education Module was created to
reduce the burden on sponsors and
FDRs. If we continue to allow sponsors
to modify or utilize their own training
in lieu of using the CMS Compliance
training, it will no longer ensure the
elimination of the prior duplication of
effort that so many FDRs stated was
creating a huge burden on their
operation. This is why CMS proposed
that only our training can be used, as it
is the only means to ensure that
duplication of effort is avoided for FDRs
who hold contracts with multiple Part C
and Part D contracting organizations.
Comment: One commenter raised
concerns over the significant amount of
time required to complete the current
CMS Compliance training and stated
that it may take time away from other
areas of training the organization has
deemed necessary through their own
internal risk assessments. They
suggested CMS consider modifying the
requirement to allow the longer training
initially and developing a shorter
‘‘refresher’’ version that could be taken
annually thereafter.
Response: We will not modify the
existing CMS Standardized General
Compliance Program Training at this
time. However, we recognize the
commenter’s concern and will take
under consideration the development of
a refresher training module for future
use.
Comment: A few commenters
recommended that CMS establish a
single centralized electronic location
where FDRs could obtain this training,
and that the centralized location would
also serve as a repository to hold
attestations of training completion
accessible to Part C and Part D
contracting organizations for
compliance oversight purposes.
Commenters suggested it be searchable
or that CMS provide updates, one
suggesting daily reports be pushed to
each MA organization and Part D
sponsor so that they could track
compliance with the training
requirement. One other commenter
suggested that the training be provided
in a Sharable Content Object Reference
Model (SCORM) format for
downloading into various organizations’
systems.
Response: The training is in a
centralized location on the Medicare
Learning Network. All who take the
training will be able to print out a
certificate of completion to prove they
have completed the training. It is the
responsibility of Part C and Part D
contracting organizations to determine
how to best retrieve and catalog this
information from their FDRs. CMS is
unable, at this time, to provide the
capacity for a publicly searchable
database of users who have completed
that training or a system that would
allow reports to be sent to the various
contracting organizations regarding the
training status of various FDR
organizations. We will consider and
determine if our training module could
be available for download into the
format suggested by commenters, but we
would need to ensure that the content
could not be modified to ensure the
integrity and completeness of the
training module.
Comment: One commenter suggested
CMS leverage the existing Compliance
Training, Education & Outreach (CTEO)
site to support this initiative and to
interactively execute the training and
collect and track the required
attestations.
Response: When we developed the
Standardized General Compliance
Program Training, the CTEO Web site
was not yet in existence. We will take
the commenter’s suggestion under
consideration and further explore that
Web site’s capability to determine if it
actually exceeds the current capability
of the Medicare Learning Network,
where the training is currently housed.
Comment: A few commenters
recommended maintaining the current
policy of allowing flexibility in how the
training requirement is met. These
commenters stated the current training
requirements meet their needs because
it allows options and reduces the
burden on various sectors of the
industry. They stated that various
organizations had already invested
resources to become compliant and to
develop efficient means of both
delivering and tracking the training. The
flexibility in the current regulations
allows plan sponsors to work in concert
with FDRs to develop effective training
for those specific entities and their
existing learning models.
Response: We recognize that the
current compliance program training
requirement does meet the needs of
some contracting organizations.
However, based on public feedback
received previously, as well as in
response to this proposed change, we
continue to believe that the proposed
approach is most efficient and effective
for the majority of FDRs and contracting
organizations.
Comment: Many commenters
requested clarification regarding who is
required to take the training: Providers,
brokers, FDRs, and/or internal
employees.
Response: The compliance and fraud,
waste, and abuse (FWA) training and
education requirement applies to all
delegated entities (which may include
agents/brokers) whom the Part C or Part
D contracting organization qualifies as
an FDR using the definition at 42 CFR
§§ 422.500(b) and 423.500. Whether a
Part C or Part D contracting organization
identifies a certain entity or individual
provider as an FDR depends on the
contractual relationship and/or written
agreement between the entity/
individual and the contracting
organization. The compliance and FWA
training is not intended to be mandatory
for the employees of those contracting
organizations.
Comment: Several commenters
wanted to know if this training would
satisfy the FWA and Compliance
training requirements.
Response: There is both a FWA and
a Compliance training module available
on the Medicare Learning Network,
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00012 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29855
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
FDRs must take both modules in order
to satisfy the entire training
requirement.
Comment: A few comments requested
clarification regarding who was deemed
for purposes of the FWA training
requirement (for example, is it just the
provider participating in Medicare FFS
or also all of the employees that work
in his office, similarly with a hospital
participating in Medicare, does it extend
to their employees). Commenters also
requested if CMS was exploring
deeming status for providers in the Part
D program.
Response: This question is outside of
the scope of this regulation. We did not
propose any changes to the FWA
training module or the associated
deeming requirements. Therefore, we
are unable to address your question at
this time.
Comment: Several commenters had
questions regarding the one-pager that
contracting organizations can provide
with organization specific information,
and requested whether this meets the
requirements for distributing their codes
of conduct (COC) or standards of
conduct (SOC) located in Chapter 9 of
Pub. 100–18, Medicare Prescription
Drug Manual, and Chapter 21 of Pub.
100–16 of the Medicare Advantage
Manual. Some commenters inquired if
this new proposal could be construed to
forbid them from distributing their
COC/SOC to their FDRs.
Response: We intend that the
standardized FWA and Compliance
Training modules will cover the basic
training requirements. We recognize
that each contracting organization has
specific information that must be shared
with their FDRs regarding the
organization’s specific operations. The
one-pager was suggested for
organizations to communicate unique
information that is usually shared in
FWA/Compliance such as relevant
organization contact information (for
example, Web site address, hotline/
ethics phone numbers) the Compliance
Officer’s contact information, the
Compliance Department staff, and
possibly even online access to the COC/
SOC or disciplinary policies. Our
experience has shown that many
contracting organizations issue their
COC/SOC electronically (internally and
externally) and/or create Web sites
designated for their FDRs to locate the
information mentioned previously.
Contracting organizations must continue
to distribute their COC/SOC to all of
their employees, FDRs, board members,
etc. Nothing is this regulation should be
interpreted to preclude organizations
from satisfying the seven elements of
the compliance program requirements.
Comment: The commenters suggested
that feedback should be solicited from
the plans to assist with improving the
content of the training, specifically
including more examples that are
relevant to FDRs, as commenters
mentioned the modules examples are
often organization-centric.
Response: We always welcome
feedback from contracting organizations
and FDRs with respect to improving our
training products. Organizations,
entities or individuals who have
suggestions should submit them to the
following mailbox: Parts_C_and _D_CP_
Comment: Some commenters stated
that CMS should consider how it can
implement this proposal in a way that
reduces administrative burdens on
contracting organizations and FDRs, as
new processes to collect and track
attestations may be difficult and time
consuming. Many suggested that a
January 1, 2015 effective date was an
insufficient amount of time to set up
such elaborate processes and
recommended that these provisions be
effective no earlier than January 1, 2016.
Response: CMS recognizes the
administrative burden imposed on the
contracting organizations and their
FDRs. The primary goal of this proposal
is to reduce that administrative burden
by instituting a uniform compliance
training module and we believe that
contracting organizations are in the best
position to determine the most effective
way to collect and track compliance
amongst their FDRs. However, we
recognize that setting up these new
processes may take time, along with
potentially updating contracts to reflect
the new requirements. Therefore, we
will delay the implementation of this
provision to January 1, 2016.
Comment: The largest number of
commenters represented FDRs that
wrote in support of the proposed
compliance training program
requirements and use of the CMS
Standardized General Compliance
Program Training, agreeing that it would
greatly reduce burden on FDRs.
Response: We thank the commenters
for their support.
After careful consideration of all of
the comments received, we are
finalizing this proposal with the one
modification discussed previously, with
a delayed applicability date of January
1, 2016.
5. Procedures for Imposing Intermediate
Sanctions and Civil Money Penalties
Under Parts C and D (§§ 422.756 and
423.756)
Sections 1857(g) and 1860D–
12(b)(3)(E) of the Act provide the
Secretary the ability to impose
intermediate sanctions on MA
organizations and PDP sponsors.
Intermediate sanctions consist of
suspension of enrollment, suspension of
marketing and suspension of payment.
Current regulations governing
intermediate sanctions are contained in
subparts O of part 422 and part 423.
Sections 422.756 and 423.756 provide
specific procedures for imposing
intermediate sanctions and include
provisions which address: The duration
of the sanction; and the standard that
we apply when determining if a
sanction should be lifted.
We proposed to expand the potential
applicability of the test period
requirement to three types of
intermediate sanctions by modifying the
existing rules to clarify that CMS may
require a test period for a sponsoring
organization that has had any of the
three types of intermediate sanctions
imposed: Marketing, enrollment and/or
payment. Second, we proposed to
clarify the enrollment parameters for
sanctioned sponsoring organizations
offering Part D plans to include
language specifying that a sanctioned
plan is not available to receive
automatically assigned beneficiaries for
the entire duration or a portion of the
testing period. We proposed to modify
the regulation text at §§ 422.756 and
423.756 to reflect these changes.
Comment: One commenter questioned
clarification on what CMS considers a
contract violation of marketing
requirements and requested if violations
would be based solely on allegations of
misconduct.
Response: Marketing standards for
MA organizations and Part D sponsors
are codified in subpart V of parts 422
and 423. The current Medicare
Marketing guidelines are located in
Chapter 3 of Pub. 100–16, Medicare
Managed Care Manual, and Chapter 3
of Pub.100–18, The Medicare
Prescription Drug Manual, which
should provide sponsors with guidance
regarding current marketing
requirements. With respect to contract
violations being based on
unsubstantiated allegations of wrong-
doing, enforcement actions are only
taken based on substantiated, well
documented instances of non-
compliance. Additionally, MA
organizations and Part D sponsors that
are sanctioned are given an opportunity
to rebut or appeal our determination
through a formal appeals process.
Comment: One commenter suggested
the prohibition on auto-enrollment into
plans under a test period should also
apply to passive enrollment.
Specifically, the commenter stated that
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00013 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29856
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
Medicare-Medicaid eligible individuals
should not be passively enrolled into an
MA or an MA Special Needs Plan (SNP)
that is under sanction or under sanction
and in a test period as part of a
demonstration or a state developed
integrated plan product.
Response: Plans that are under a
sanction are not eligible to receive
enrollments. However, we have the
discretion to require a sanctioned plan
to market or accept enrollments for a
limited period to assist in making a
determination as to whether the bases
for imposing the sanction have been
fully corrected and are not likely to
recur. As stated previously, sanctioned
sponsoring organizations offering a Part
D benefit would not be eligible to
receive automatically assigned
beneficiaries during the test period.
During a ‘‘test period’’ the sanction(s)
remain in effect.
Comment: One commenter requested
that we extend the proposal to also not
allow passive enrollment into plans that
are coming off of sanction or are
currently in a test period until we have
determined they are ready.
Response: CMS has determined that it
is legally permissible to provide for
enrollment in an MA or Part D plan
under a passive enrollment request
process in specific, limited
circumstances generally associated with
either immediate plan terminations or
in other situation where CMS
determines that remaining enrolled in
the plan would pose potential harm to
members. We determine when passive
enrollment is appropriate. In evaluating
whether such CMS-directed enrollee
movements are appropriate, a key factor
is the determination as to whether the
receiving plan is essentially equivalent
to (or better than) the current plan from
an overall perspective.
Therefore, in situations where passive
enrollment is determined permissible,
like an immediate plan termination,
CMS would factor in a number of
criteria, including the receiving plan’s
current premium, benefit and formulary
structure, as well as plan past
performance. In any event, our goal
would be to ensure that those affected
members suffered as little disruption as
possible during their transition. Plans
that were under sanction at the time of
a passive enrollment would not be
considered a viable option for affected
enrollees and it is unlikely that sponsors
under a test period would either.
However, if a sponsor who was removed
from sanction and was under a test
period met several other criteria for
receiving passive enrollment (that is,
plan’s benefit and formulary structure
was largely the same and their premium
was not significantly higher), we may
consider them among the group of
available plans to receive passive
enrollment.
Comment: A few commenters
requested clarification regarding the
new sanction authority, specifically the
language that would allow CMS to
impose intermediate sanctions on an
organization that enrolls an individual
without prior consent (except in certain
limited circumstances) or transfers an
individual to a new plan without prior
consent. The commenters asked that
CMS clarify that this would not apply
to organizations that perform facilitated
or auto-enrollment, passive enrollment,
seamless enrollment or group or
individual enrollment requests from
EGWPs.
Response: In the proposed rule, we
proposed to amend the regulation text at
§§ 422.752 and 423.752 by adding
subparagraph (a)(9), which reads:
‘‘. . .Except as provided under § 423.34
of this chapter, enrolls an individual in
any plan under this part without the
prior consent of the individual or the
designee of the individual.’’ Section
423.34 specifically refers to enrollment
of individuals who receive the low
income subsidy (LIS) and are therefore
subject to facilitated or auto-enrollment.
Therefore, we believe that the proposed
regulation text already makes clear that
this provision would not apply to those
organizations that are performing
facilitated enrollment of LIS
beneficiaries. Additionally, passive
enrollment and use of the seamless
enrollment option are initiated or
approved by CMS, respectively.
Therefore, an organization who is
contacted by CMS to receive passive
enrollment would not be considered to
have performed enrollment without
prior consent. As for the seamless
enrollment option, as these proposals
must be submitted to and approved by
CMS, as long as organizations are
following CMS’ enrollment guidance in
Chapter 2, § 40.1.4, and have received
CMS’ approval, an organization again
would not be considered as enrolling
without prior consent and would
therefore not be considered for a
possible sanction. Finally an
organization who is accepting
enrollment requests for an employer or
union sponsored plan using the group
enrollment mechanism must follow
CMS’ enrollment guidance in Chapter 2,
§ 40.1.6.1. As long as CMS enrollment
guidance is being followed with respect
to processing these enrollments, CMS
would not consider MA and Part D
organizations in violation of the new
requirement. However, we expect that
requests for enrollment into an
employer or union sponsored plan
outside of the group enrollment process
(that is, beneficiary initiated enrollment
requests) follow all requirements,
including prior consent, applicable to
any other individual enrollment request.
Comment: Several commenters
expressed support for our proposal to
expand the use of the ‘‘test period’’
requirement to all intermediate
sanctions, and support the proposal that
previously sanctioned below-the-
benchmark Part D plans not be allowed
to receive or process auto-enrollments
or reassignments until they are
determined to be ready by CMS.
Response: We thank the commenters
for their support.
After careful consideration of the
comments received, we are finalizing
these proposals without modification.
We inadvertently failed to include
proposed regulation text for § 423.756
that corresponds to this proposal. In this
final rule, we finalize amendments to
§§ 422.756 and 423.756 that are
virtually identical to implement this
proposal.
6. Timely Access to Mail Order Services
(§ 423.120)
Section 1860D–12(b)(3) of the Act
authorizes the Secretary to include
contract terms for Part D sponsors, not
inconsistent with the Part C and D
statutes, as necessary and appropriate.
Section 423.120(a)(3) specifies that a
Part D sponsor’s contracted network
may include non-retail pharmacies,
including mail order pharmacies, so
long as the network access requirements
are met. Part D plans are increasingly
entering into contracts with mail order
pharmacies to offer beneficiaries an
alternative way to fill prescriptions
under the Part D benefit, often at much
lower cost sharing than is available at
network retail pharmacies. While mail
order pharmacies make up a relatively
small percentage of total prescriptions
filled under the Part D program, we are
committed to ensuring consistent and
reliable beneficiary access to
medications, regardless of what type of
pharmacy fills the prescriptions.
Section 1860D–4 of the Act describes
the various beneficiary protections in
place in the Part D program. For mail
order pharmacies, the industry standard
for delivery times appears to range from
7 to 10 business days from the date the
prescription was received, and Part D
sponsors’ marketing materials often
specify this time frame to beneficiaries.
Beneficiaries generally choose to fill
prescriptions through a mail order
pharmacy, for lower cost sharing, when
it is feasible to wait 7 to 10 days to
receive their medications. However, if
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00014 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29857
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
this time frame is disrupted,
beneficiaries may experience gaps in
therapy.
When issues with filling a
prescription arise in a retail setting, the
beneficiary often is notified of the
problem in real time, or within hours of
discovery. When issues arise in a mail
order setting, the delays in finding,
communicating, and making the
appropriate contacts to resolve the
problem may add days onto the ultimate
delivery date, resulting in a potentially
more significant concern for mail order
beneficiaries if these delays result in
gaps in therapy. For this reason, we
proposed to establish fulfillment
requirements for mail order pharmacies
as well as home delivery services
offered by retail pharmacies, to set
consistent expectations for beneficiary
access to drugs in this growing segment.
Many beneficiaries may be very well
served by this type of pharmacy access,
but only if they can rely upon efficient
processing and turnaround times. Mail
order pharmacies contracted by Part D
sponsors can reasonably be expected to
meet minimum performance standards
for order fulfillment, including
convenient order turnaround times, as a
beneficiary protection and as a
component of providing good customer
service. Clearly stating in beneficiary
materials the expected turnaround time
for delivery allows the beneficiary to
better control when they need to reorder
to ensure no gaps in medication supply.
Clarity in expected turnaround times
also can prevent needing to address
customer inquiries into the status of a
pending order, setting parameters for
when an order is or is not delayed and
what options become available at that
point. We believe that established
companies that have been providing
these services for years have generally
been meeting these standards in practice
already, and that the proposed
turnaround times are in line with
current practices followed by mail order
pharmacies today.
Therefore, we proposed to amend
§ 423.120(a)(3) to specify mail order
fulfillment requirements in line with
what we have observed in other
markets: 5 business days (from when the
pharmacy receives the prescription
order to when it is shipped) for those
prescriptions requiring intervention
beyond filling (such as clarifying
illegible orders, resolving third party
rejections, and coordinating with
multiple providers as part of drug
utilization management); and 3 business
days (from when the pharmacy receives
the prescription order to when it is
shipped) for those prescriptions not
requiring intervention. We recognize
that some prescription orders may
require clarification or additional steps
to be taken by the provider or
beneficiary that would extend beyond
the proposed period of 5 days. We
believe that such cases represent a
minority of mail order prescriptions,
and as such we would anticipate that
more than 99 percent of all mail order
prescriptions processed are filled in
compliance with either the 3- or 5-day
standard. We believed our proposed
standards are in alignment with
fulfillment requirements already in
place in the market and as such do not
create a new burden or new standard for
mail order pharmacies to meet. We
solicited comments not only on the
proposed time frames, but also on
whether there are instances (in addition
to those discussed previously) in which
the proposed 5-day time frame should
apply. We received the following
comments and our response follows:
Comment: A few commenters
questioned why we proposed
turnaround times of 3 and 5 days if we
list in preamble that standard
turnaround times are 7 to 10 days for
delivery.
Response: The preamble discussion
surrounding delivery of prescriptions
within 7 to 10 days is from the
perspective of the beneficiary; listing
the total time from when a medication
is ordered to the time it is delivered.
Importantly, this includes shipping
time. The proposed fulfillment
standards were specific to mail order
pharmacy processing times and did not
include actual time in shipping. In other
words, the 3 to 5-day turnaround time
only refers to the timeframe from when
the pharmacy receives the order until
the pharmacy ships the order.
Comment: Many commenters
expressed concerns that 5 business days
is too short of a time frame to require
mail order pharmacies to resolve some
issues when they arise (such as
manufacturer drug shortages), many of
which are outside the control of the
pharmacy. Many commenters noted
unique timeline concerns specific to
specialty medications, such as cold
chain shipping and needing to contact
the beneficiary to coordinate delivery.
Multiple commenters suggested that
additional leeway is also needed to
accommodate issues such as natural
disasters. Multiple commenters
suggested that mail order pharmacies
should contact beneficiaries as a good
customer service practice when any
delay in filling will prevent an order
from shipping within 5 days. Many
commenters noted that they currently
would be able to meet a 3-day
turnaround standard for filling orders
not requiring follow up contact with the
beneficiary or prescriber.
Response: We recognize that some
interventions may require more than 5
business days to resolve. In those cases,
we agree with the suggestion from
multiple commenters that mail order
pharmacies should contact beneficiaries
as a good customer service practice
when any delay in filling will prevent
an order from shipping within 5 days.
However, in light of the comments
received regarding a variety of situations
that we had not considered, including
some outside of the pharmacy’s control
that could create delays longer than 5
days, we are not finalizing the proposal
to establish fulfillment standards for
mail order. Instead, we will continue
analysis on mail order fulfillment time
frames, including evaluating the impact
of the implementation of the auto-ship
beneficiary consent policy finalized in
the 2014 Call Letter. In addition, Part D
sponsors are expected to follow best
practices by making clear their expected
delivery turnaround times in their
beneficiary materials, consistently
meeting such delivery time frames, and
having contingency plans for when they
cannot, such as allowing retail access at
mail order cost sharing levels if
necessary. The volume of complaints
that we receive related to mail order
delivery suggests that beneficiary
expectations are not consistently being
met. We will increase our monitoring of
mail order pharmacies, and will
consider the need to establish standards
and requirements in the future. Based
on the comments submitted, additional
consideration may be necessary
surrounding specialty medications and
their delivery, especially when there are
cold chain or other shipping
considerations. We reviewed the
information provided on how specialty
pharmacy differs from other mail order
deliveries, and agree that additional
consideration should be given to these
pharmacies and medications in any
future guidance. Additionally, we will
clarify existing guidance about
exception processes and coverage
denials to ensure that mail order
pharmacies provide beneficiaries notice
of non-fulfillment of a prescription as
expeditiously as possible. Current
guidance on disaster responses and drug
shortages still apply, and we encourage
sponsors to communicate with their
enrollees when unique situations like
these arise.
Comment: A few commenters
suggested that mail order turnaround
times are best left to state Boards of
Pharmacy to monitor, instead of being
set in regulation.
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00015 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29858
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
Response: We proposed specifying
parameters for timely mail order
fulfillment, consistent with the
authority given to the Secretary to
specify additional contract terms not
inconsistent with the Part D statute.
However, we had not considered the
potential conflict or duplication with
state-based requirements and appreciate
the comments. We will take this under
consideration as we consider
establishing requirements for Part D
sponsors offering a mail order benefit in
the future.
Comment: Some commenters wrote
that turnaround times would be better
defined in guidance or incorporated in
star ratings or other quality metrics.
Response: We appreciate the
suggestion. As we will not be finalizing
the proposed fulfillment standards in
this final rule, we are exploring
alternatives for ensuring consistent and
predictable access to medications for
beneficiaries in a plan offering a mail
order benefit. As part of this effort, we
are currently developing a study of how
mail order benefits are used within the
Part D benefit. The comments received
on the proposed rule and the results of
this study will be considered when
determining whether fulfilment
standards should be included in future
star ratings measures, as well as used to
inform the need for future guidance or
rulemaking. Additionally, we will
increase our monitoring and analysis of
mail order-related complaints in the
CTM and explore setting a threshold for
the volume or severity of complaints
triggering a review by CMS. We remain
very concerned by the high level of
complaints received relating to mail
order, and take seriously the issues
raised by beneficiaries. We are also
exploring how fulfillment of plan-
designated turnaround times listed in
marketing or other beneficiary materials
could be included within the audit
framework.
Comment: One commenter wrote in
with concerns that the methodology
used in two CMS studies cited in
another provision were problematic and
stated that no regulation proposals
relating to mail order should be
finalized until corrected and
reexamined.
Response: The studies noted by the
commenter were not used when
designing the proposal specific to timely
delivery of mail order prescriptions.
Comment: Some commenters
suggested that the policy announced in
the CMS 2014 Call Letter that
pharmacies obtain beneficiary consent
prior to shipping any medications that
the beneficiary did not affirmatively
order directly affects the timeline for
order fulfilment and any defined
turnaround times for delivery should be
adjusted accordingly.
Response: We recognize that the CMS
2014 Call Letter auto-ship policy
necessitates an increased level of
coordination with the beneficiary for
some pharmacies, when filling
prescriptions that the beneficiary did
not directly request (such as new orders
submitted directly by the provider or
refills prompted by an automatic
delivery program). We will not be
finalizing the proposed fulfillment
standards in this final rule, but
encourage all plan sponsors to consider
the need for coordination with the
beneficiary when establishing and
marketing average turnaround time
estimates for their members.
Comment: A few commenters
suggested that beneficiaries should be
allowed to fill their medications at the
retail pharmacy of their choice, at the
same cost sharing level as mail order, if
a mail order pharmacy encounters any
delays, or delays extending beyond 5
days.
Response: While this was not a part
of our proposal, and we will not be
finalizing any new requirements at this
time, we do agree with commenters that
this would be an important beneficiary
protection. We believe that best
practices for addressing a lost or
delayed order would include plan
sponsors providing clear and timely
guidance to the beneficiary in the event
of a lost or delayed order, including a
list of options for obtaining a
medication. Part D sponsors should
have contingencies in place when issues
are encountered that lead to a delay and
potentially a gap in therapy. This could
include offering beneficiaries the ability
to fill a delayed mail order prescription
at a retail pharmacy and pay no more
than what they would have been
charged by a mail order pharmacy. The
need to prevent gaps in therapy for
beneficiaries relying on mail order
pharmacies remains a significant
concern to us.
In summary, we are not finalizing any
fulfillment standards for mail order
prescriptions, in light of the concerns
raised. We will use the information
gained from our mail order study and
from the public comments submitted to
explore the need for additional guidance
or rulemaking in the future. The need to
ensure consistent access to and prevent
gaps in therapy for enrollees relying on
mail order for their medications
continues to be a significant concern.
We additionally solicited comments
on whether we should establish
additional requirements for beneficiary
materials relating to mail order services,
such as: Clear definitions of processing
time and delivery time; how to access
customer support; how to submit a
complaint via 1 800 MEDICARE; and
beneficiary options for accessing
medications when a delivery is lost or
delayed. We received the following
comments and our response follows:
Comment: Many commenters stated
that additional requirements for
beneficiary materials would enhance
mail order services and that this would
be a positive change for beneficiaries.
These commenters noted that clear
definitions of requirements are needed
to resolve issues, ensure consistent
access, and ensure no gaps in therapy.
Response: We appreciate the
comments. We intend to conduct a
study of mail order benefits offered by
Part D sponsors and will use this, and
the information received from public
comments, to inform changes to
beneficiary materials relating to mail
order. At a minimum, we expect
sponsors offering mail order services to
follow best practices by clearly listing
estimated delivery times in their
marketing and beneficiary materials. In
the event of a failure to meet plan-
designated timeframes for delivery, as a
best practice sponsors should be
prepared to take the steps necessary to
provide their enrollee the medication in
a timely manner in order to avoid gaps
in therapy. This could include offering
enrollees the option to obtain delayed
medications at a retail pharmacy at the
same cost sharing level as mail order.
We also welcomed comments on any
other requirements we should consider
for mail order or other home delivery
options. For example, also potentially
affecting consistent access to medication
is the use of mail order to fill initial
prescriptions of new drugs or to fill 30-
day supplies of chronically used
medications. The need to order a refill
early, allowing sufficient time for
processing and delivery, can result in
refill-too-soon edits based upon retail 30
day standards. Resolving inappropriate
or inapplicable edits increases burden
on the beneficiary and the mail order
pharmacy and essentially creates a
disincentive for beneficiaries who are
planning ahead and attempting to order
early enough to ensure uninterrupted
supplies of chronic medications. In
general, we believe that filling initial
prescriptions or routine 30-day supplies
at mail order is not good practice. We
recognize that there may be a small
minority of beneficiaries who
successfully depend solely upon mail
order or other home delivery options for
access to prescription drugs due to
particular circumstances of geography
or mobility. We have no reason to
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00016 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29859
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
discourage their continued use of these
services. However, due to the
difficulties reported to CMS with
consistently and effectively filling short
time frame supplies through mail order,
we do not believe that Medicare
beneficiaries in general should be
incentivized through lower cost sharing
to utilize mail order pharmacies for
initial prescriptions or 30-day supplies.
We received the following comments
and our response follows:
Comment: A few commenters agreed
that mail order is not an appropriate
venue for filling 30 day supplies of
medications.
Response: We appreciate the
comments and will explore how often
mail order is used for short days’
supplies of medications as a part of the
current study on mail order benefits.
Comment: Some commenters noted
that specialty pharmacies often dispense
medications by mail order in an amount
lasting 1 month or less.
Response: We agree with the
comments that noted some specialty
medications may be best supplied,
when supplied by mail order, in
quantities less than a 3 month supply,
due to frequent dose titrations, financial
concerns, or applicable controlled
substance laws.
We did not propose any specific
regulatory requirements to mail order
for 30-day supplies or less. We are
currently analyzing the types of
prescriptions filled by mail order
pharmacies and will use the information
gained from this to explore the need for
future guidance or rulemaking that
could help ensure consistent timely
access for Part D beneficiaries opting to
use mail order for both short and
extended days’ supplies.
7. Agent/Broker Compensation
Requirements (§§ 422.2274 and
423.2274)
Section 103(b)(1)(B) of MIPPA revised
the Act to charge the Secretary with
establishing guidelines to ’’ensure that
the use of compensation creates
incentives for agents and brokers to
enroll individuals in the MA plan that
is intended to best meet their health
care needs.’’ Section 103(b)(2) of MIPPA
revised the Act to apply these same
guidelines to Part D sponsors. Our
program experience indicates that some
agents may encourage beneficiaries to
enroll in plans that offer higher
commissions without regard to whether
plan benefits meet the beneficiaries’
health needs. In recognition that agents
and brokers play a significant role in
providing guidance and advice to
beneficiaries and are in a unique
position to influence beneficiary choice,
we had proposed, prior to the enactment
of MIPPA, a rule to regulate agent and
broker compensation. To implement the
MIPAA provisions and relying in part
on comments in response to our
previously proposed rule, we adopted
an interim final rule on September 18,
2008, entitled ‘‘Medicare Program;
Medicare Advantage and Prescription
Drug Benefit Programs: Final Marketing
Provisions’’ (73 FR 554226), which,
among other things, established the
current compensation structure for
agents and brokers as it applies to Parts
C and D. That rule remains significantly
in place at §§ 422.2274 and 423.2274,
and our experience since then indicates
that revision of the compensation
requirements is necessary to ensure that
we continue to meet our statutory
mandate.
The current compensation structure is
comprised of a 6-year compensation
cycle that began in Contract Year (CY)
2009. MA organizations and Part D
sponsors were to provide an initial
compensation payment to independent
agents for new enrollees (Year 1) and
pay a renewal rate (equal to 50 percent
of the initial year compensation) to
independent agents for Years 2 through
6. These rates were to be adjusted
annually based on changes to the MA
payment rates or Part D parameters as
established by CMS. We later amended
the regulations to allow MA
organizations and Part D sponsors to
compensate independent agents and
brokers annually using an amount at or
below the Fair Market Value (FMV).
(See the final rule with comment period
entitled, ‘‘Medicare Program; Changes to
the Medicare Advantage and the
Medicare Prescription Drug Benefit
Programs for Contract Year 2013 and
Other Changes’’ (77 FR 22072)
published in the April 12, 2012 Federal
Register.)
The first 6-year cycle ended at the end
of CY 2013, on December 31, 2013. The
first year, CY 2009, was considered to be
the first renewal year for those already
enrolled, effectively making CY 2009
the second full year of compensation.
Because our regulations were silent
regarding compensation amounts for
Year 7 and beyond, we stated in our
Final Call Letter for Contract Year 2014,
issued on April 1, 2013, that MA
organizations and Part D sponsors
could, at their discretion, pay agents
and brokers the renewal amount for
Year 7 and beyond. However, this
subregulatory guidance was intended to
be a temporary measure, pending final
changes to our regulations.
Under the current structure, MA
organizations and Part D sponsors pay
an initial rate for the first year, and then
a renewal payment of 50 percent of the
initial compensation paid to the agent
for years 2 through 6. This structure has
proven to be complicated to implement
and monitor as it requires the MA
organization or Part D sponsor to track
the compensation paid for every
enrollee’s initial enrollment, and
calculate the renewal rate based on that
initial payment. In our NPRM, dated
January 10, 2014, we provided a
detailed example of the complexities of
the current compensation structure.
Summarizing the current complexities,
every MA organization or Part D
sponsor has to know, at any given time,
the amount of the initial compensation
for each plan year—going back as far as
2009—in which the member enrolled in
order to pay the correct compensation
amount to the agent for the current
contract year. For new members, MA
organizations and Part D sponsors must
first review CMS’ reports to determine
whether an initial or renewal payment
should be made, and then combine that
information with the FMV, or, if
applicable, the plan’s compensation set
at less than the FMV, for each plan year
to ensure the correct payments are made
to agents.
In addition to its complexity, we
remain concerned that the current
structure creates an incentive for agents
and brokers to move enrollees from a
plan of one parent organization to a plan
of another parent organization, even for
like plan-type changes. In our NPRM,
we discussed and expanded upon our
example of how the current system
results in different payments when a
beneficiary moves from one like plan to
another like plan in different
organizations. In these cases, the new
parent organization would pay the agent
50 percent of the current initial rate of
the new parent organization; not 50
percent of the original initial rate paid
by the other parent organization. Thus,
in cases where the FMV has increased,
or the other parent organization pays a
higher commission, an incentive exists
for the agent to move beneficiaries from
one parent organization to another. (See
§§ 422.2274(a)(3) and 423.2274(a)(3)).
Since 2008, we have received
inquiries from MA organizations and
Part D sponsors regarding the correct
calculation of agent/broker
compensation, and found it necessary to
take compliance actions against MA
organizations and Part D sponsors for
failure to comply with the
compensation requirements. To the
extent that there is confusion about the
required levels of compensation or the
timing of compensation, there could be
an uneven playing field for MA
organizations and Part D sponsors
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00017 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29860
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
operating in the same geographic area.
In addition, CMS’ audit findings and
monitoring efforts have shown that MA
organizations and Part D sponsors are
having difficulty correctly administering
the compensation requirements.
Therefore, we proposed simpler agent/
broker compensation regulations to
better ensure that plan payments are
correct and establish a level playing
field that will further limit incentives
for agents and brokers to move enrollees
for financial gain.
We proposed to revise the existing
compensation structure for agents and
brokers so that, for new enrollments,
MA organizations and Part D sponsors
could make an initial payment that is no
greater than the FMV amount for
renewals in Year 2 and beyond, the MA
organization or Part D sponsor could
pay up to 35 percent of the FMV amount
for the renewal year, resulting in
renewal year payment changes each
year if the MA organization or Part D
sponsor chooses to pay 35 percent of the
current FMV (that is, the renewal year
FMV threshold). As is currently the
case, we would interpret the FMV
threshold in our annual guidance to MA
organizations and Part D sponsors. This
flexibility would enable MA
organizations and Part D sponsors to
better react to changes in the
marketplace and adjust their
compensation structures accordingly.
When we proposed the 35 percent
renewal rate, we also discussed several
different alternatives, including
prohibiting compensation payments
entirely beyond year 6, permitting MA
organizations and Part D sponsors to
pay a residual payment for year 7 and
subsequent years, and permitting
existing renewal payments to extend
past year 7. We also evaluated different
renewal amounts, including a 50
percent renewal payment for years 2
through 6 with a continuing 25 percent
residual payment for years 7 and
beyond. The evaluation took into
account different beneficiary ages for an
initial enrollment, as well as life
expectancy. In the analysis, a renewal
payment of 35 percent was similar in
payout to the combination of a 50
percent payment for years 2 through 6
and a residual payment of 25 percent for
year 7 and beyond.
In our NPRM, we stated that we
believed that revising the existing
compensation structure to allow MA
organizations or Part D sponsors to pay
up to 35 percent of the FMV for year 2
and beyond was appropriate based on
several factors. First, we stated that a
two-tiered (initial and renewal) payment
system would be significantly less
complicated than a three-tiered system
(initial, 50 percent renewal for years 2
through 6, and 25 percent residual for
years 7 and beyond), and would reduce
administrative burden and confusion for
plan sponsors. Second, our analysis
determined that 35 percent is the
renewal compensation level at which
the present value of overall payments
under a two-tiered system would be
relatively equal to the present value of
overall payments under a three-tiered
system (taking into account the
estimated mortality rates for several
beneficiary age cohorts). This analysis
was based on the existing commission
structure basing renewal commissions
on the starting year initial commission
amount and not the current year FMV
amount.
In order to implement the changes in
the identical Part C and Part D
regulations at §§ 422.2274 and 423.2274,
our NPRM first proposed to revise the
introductory language for each section
and then define ‘‘compensation’’ in
paragraph (a)(1) and to restate the fair
market value limit on compensation for
the initial year as paragraph (b)(1)(i).
Second, we proposed to combine the
current (a)(1)(i)(B), which addresses
payments for renewals, and (a)(1)(iii),
which addresses the length of time that
renewals should be paid, and designate
the revisions as a new (b)(1)(ii). Thus,
the proposed new paragraph (b)(1)(ii)
would state that plans may pay up to 35
percent of the current FMV and that
renewal payments may be made for the
second year of enrollment and beyond.
In addition, we proposed to modify
paragraph (a)(3) to remove the 6-year
cap on the compensation cycle.
Currently, paragraph (a)(3) refers to
policies that are replaced with a like
plan during the first year or the
subsequent 5 renewal years. Since we
proposed to eliminate the 6-year cycle,
our revised paragraph (b)(2) deletes the
reference to the initial year and the 5
renewal years. By tying renewal
compensation to the FMV for the
renewal year, rather than to the initial
year of enrollment, our proposal reduces
the financial incentives for an agent or
broker to encourage Medicare
beneficiaries to change plans, especially
from one parent organization to another
parent organization. As with the current
regulation, we proposed in paragraph
(b)(2)(iii) that a change in enrollment to
a new plan type be payable under the
same rules that apply to an initial
enrollment, regardless of whether the
change is to an unlike plan type in the
same parent organization or an unlike
plan type in another parent
organization. Note that, as with the
current rule, our proposal only
addresses compensation paid to
independent agents and does not
address compensation payable by an
MA organization or Part D sponsor to its
employees who perform services similar
to agents and brokers.
We welcomed comments on both the
amount of the renewal payment, as well
as the proposed indefinite time frame,
which are discussed in depth as follows.
In summary, we received a number of
comments supporting our efforts to
simplify agent/broker compensation
calculation. These comments were
primarily from plans and industry trade
groups. We will be finalizing the rule to
implement a two-tiered (initial and
renewal) payment system using the
FMV in the current year for renewal
calculations.
We received numerous comments
from agents, brokers, plans and trade
associations overwhelmingly opposing
the 35 percent renewal rate. Based on
the comments received, we will finalize
the amendment to the regulations with
a cap of 50 percent of the current FMV
for renewals.
In response to the comments received,
we also determined that some
clarifications were necessary. For
renewals, the payment is based on the
current FMV and not the initial
enrollment year FMV. For example,
assume a beneficiary enrolls in an MA
plan in CY 2013. The plan pays the
initial FMV for CY 2013, which is $413.
In CY 2015, assume the FMV is $420.
The plan chooses to pay 50 percent of
the FMV for renewals. The maximum
renewal payment for this member for
CY 2015 would be $210 ($420 * .50)
instead of $207 ($413 * .50). For all
enrollments, MA organizations and Part
D Sponsors should calculate the
renewal rate based on the FMV of the
enrollment year. We are also clarifying
that our proposed and final regulations
do not require an indefinite payment of
50 percent of the FMV. The final rule
would permit up to 50 percent of the
current FMV to be paid by an MA
organization or Part D sponsor. CMS
currently requires that plans inform
CMS as to whether they are using
independent agents. Contracts between
MA organizations and Part D Sponsors,
on one hand, and their independent
agents and/or downstream entities on
the other hand, such as Field Marketing
Organizations, are not exhaustively
regulated by CMS. Therefore, MA
organizations and Part D sponsors may
decide the duration of their contract
with agents, number of applicable
renewals, and the actual rate for
renewals for each year, subject to the
limits in this final rule.
Current regulations at
§§ 422.2274(a)(4) and 423.2274(a)(4),
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00018 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29861
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
which we proposed to redesignate as
part of paragraph (b), address the timing
of plan payments, as well as
recoupment of payments when a
beneficiary disenrolls from a plan.
Specifically, current paragraph (a)(4)
states that compensation may only be
paid for the beneficiary’s months of
enrollment during the year (January
through December). Under our proposal,
the new subparagraph (a) would more
clearly define a plan year for purposes
of compensation. The annual
compensation amount covers January 1
through December 31 of each year. Our
proposal also clarified that the payment
made to an agent must be for January 1
through December 31 of the year and
may not span calendar years. For
example, a renewal payment cannot be
made for the period of November 1,
2013 through October 31, 2014. These
proposed revisions represented
clarifications rather than new proposals
and were necessary based on our
findings that some plans have been
paying compensation based on a rolling
year cycle, rather than a calendar year
cycle. Therefore, we are implementing
the provision defining ‘‘plan year’’ and,
at subparagraph (b)(3)(i), limiting
payments to the months of enrollment
during the calendar year, as proposed.
Comments concerning this provision are
discussed later in this section.
Currently, regulations at
§ 422.2274(a)(4)(i) permit payments to
be made at one time or in installments
and at any time. In order to reduce the
number of payments that need to be
recouped based on changes made during
the annual coordinated election period
(AEP), which runs from October 15
through December 7, CMS proposed, in
new subparagraph (b)(3)(ii), changing
the timing of payments to require that
payments may not be made until
January 1 of the enrollment year and
must be paid in full by December 31 of
the enrollment year. We stated that this
proposal was appropriate given that the
beneficiary’s final application during
the AEP becomes the effective
enrollment. This would reduce the
number of recoupments required when
an enrollee signed more than one
application during the AEP. We
received several comments opposing the
requirement that MA organizations and
Part D plans may not make AEP
payments until January 1 of the
following year, but do not find these
arguments sufficiently compelling to
outweigh the simplification that would
be gained by establishing the January
deadline. We also received comments
regarding our proposed requirement that
payments be completed by December
31. MA organizations and industry
associations stated that accurate
payments, especially for enrollments
effective on December 1, would be
difficult to operationalize by the end of
the year. However, we would expect
enrollment requests for a December 1
effective date to be relatively low, as
only individuals newly eligible to
Medicare Advantage and those with a
special election period would be able to
enroll for that date. Moreover,
organizations and sponsors are already
required to process most post-
enrollment activities within two weeks.
Therefore, we continue to believe that
the December 31 deadline is in the best
interest of the program and are
finalizing subparagraph (b)(3)(ii) as
proposed.
Current regulations at
§§ 422.2274(4)(ii)(A) and
423.2274(4)(ii)(A) require MA
organizations and Part D sponsors to
recoup compensation paid to agents
when a beneficiary disenrolls from a
plan within the first 3 months of
enrollment. However, in sub-regulatory
guidance, we have recognized several
circumstances (for example, death of the
beneficiary, the beneficiary moves out of
the service area, the beneficiary
becomes eligible to receive LIS, or the
beneficiary loses Medicaid benefits) in
which plans should not recoup
compensation, even though the
beneficiary was enrolled in the plan for
less than 3 months. In such
circumstances, since the disenrollment
decision could not be based on agent or
broker behavior, we believe it to be
appropriate and in the best interest of
the Medicare program for the agent to
receive the compensation based on the
number of months that beneficiary was
enrolled in the plan. While the plan
would not recoup the compensation for
those months, it would recoup any
compensation paid for the months after
the date of disenrollment.
CMS proposed to combine current
paragraphs (a)(4)(ii)(A) and (a)(4)(ii)(B)
into a revised paragraph (b)(3)(iii),
which included new text to require
plans to recover compensation for only
the months that the beneficiary is not
enrolled, unless the disenrollment took
place within the first 3 months. In our
proposed rule, paragraph (b)(3)(iii)
would require recoupment of all
compensation in cases where the
disenrollment was the result of agent or
broker behavior. We received few but
compelling comments on this proposal,
which stated that it would be extremely
difficult for MA organizations and Part
D Sponsors to determine whether the
disenrollment was a result of agent
behavior, potentially resulting in
compensation either being
inappropriately recouped or not
recouped when necessary. Based on
these comments, we are not finalizing
our proposal for subparagraph (b)(3)(iii)
but are finalizing regulation text to state
that the entire compensation must
recouped if a disenrollment occurs
during the first 3 months unless CMS
determines that recoupment is not in
the best interest of the Medicare
program. We intend for this standard to
be applied as we have implemented this
aspect of the current regulation in past,
with certain circumstances (for
example, death of the beneficiary, the
beneficiary moves out of the service
area, the beneficiary becomes eligible to
receive LIS, or the beneficiary loses
Medicaid benefits) not triggering the
recoupment requirement. We will
continue to provide exceptions to the
requirement in sub-regulatory guidance
by applying the standard we are
finalizing today.
We also proposed, to be codified at
§§ 422.2274(h) and 423.2274(h), to
codify existing sub-regulatory guidance
regarding referral (finder’s) fees. We
released a memorandum on October 19,
2011 addressing excessive referral fees,
noting that referral fees should not
exceed $100. We have long been
concerned that some MA organizations
or Part D sponsors can offer the entire
amount of compensation an agent or
broker receives through only a referral
while referral fees paid to others are part
of the total compensation. This creates
an uneven playing field within the
marketplace and a clear financial
incentive for the referring agent to steer
beneficiaries to MA organizations or
Part D sponsors that offer the higher
amount, without regard for whether
plan benefits meet the beneficiaries’
health care needs. Therefore, we
proposed to limit the amount that can
be paid as a referral fee to independent,
captive, and employed agents and
brokers, regardless of who completes the
enrollment form, to a reasonable
amount, as determined by CMS, which
is currently $100, for CY 2013 and CY
2014. The entire proposal concerning
agent and broker compensation was
discussed in the context of our concern
that agents and brokers not be
influenced by payments from MA
organizations and Part D sponsors to
steer beneficiaries to plans that do not
meet the beneficiaries’ needs. We note
that this proposal was clearly identified
in the preamble, 79 FR 1936, but the
proposed regulation text, 79 FR 2060
and 2071, mistakenly included language
discussing enrollee behavior and the
value of health-related activities.
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00019 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29862
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
Furthermore, under §§ 422.2274(h)(2)
and 423.2274(h)(2), CMS proposed that
that referral fees paid to independent
agents and brokers must be part of total
compensation not to exceed the FMV for
that calendar year. Although a few
comments were received concerning our
proposals on referral fees, we are
implementing this proposal
substantively as described in the
preamble. However, we believe that use
of the phrase ‘‘. . . while not exceeding
the value of the health-related service or
activity itself’’ was an error in the
proposed regulation text. Therefore, we
are finalizing text at subparagraph (h)(1)
by removing that error and more clearly
providing that CMS will set an annual
threshold for finder fees based on a
determination about amounts that
would improperly incentivize agents
and brokers to steer beneficiaries. We
are finalizing subparagraph (h)(2) as
proposed. Comment details and our
responses may be found as follows.
We are finalizing the regulations with
additional regulation text for a technical
correction. One entity commented that
the proposal eliminated
§§ 422.2274(a)(1)(iv) and
423.2274(a)(1)(iv). Our proposal was not
to remove these provisions concerning
the applicability of compensation to
third party entities and the regulation
text should have included the substance
of current subparagraph (a)(1)(iv). We
have inserted the text from the
regulation prior to the proposal at
§§ 422.2274(b)(1)(iii) and
423.2274(b)(1)(iii) of this final rule.
Finally, we are not finalizing the
change to the introductory language to
§§ 422.2274 and 423.2274 in favor of
deleting the existing introductory
language (which forms the substantive
basis for the new paragraph (a)
definitions); the introductory language
we proposed seems unnecessary to
establish the scope of each regulation.
Comment: We received more than 140
comments concerning the level of
renewal payments, proposed at 35
percent. A few of the comments
appreciated the simplification and
briefly discussed the 35 percent but
neither strongly supported the amount
or strongly opposed the amount. A few
commenters believed renewal
compensation should increase. The vast
majority (over 95 percent) of the
comments did not support the proposed
renewal rate of 35 percent for years two
and beyond with a few clearly stating
that the renewal rate should be 50
percent. Commenters included agents,
brokers, plans, and industry trade
associations. One major trade
association representing 37 plans stated
that 35 percent was overly restrictive
and 50 percent is in line with industry
standards, especially concerning PDPs
where the 35 percent renewal would not
cover the agent’s costs to ensure
members are in the best plans for them.
The commenters provided various
reasons why the 35 percent should not
be implemented. The majority of
commenters stated that agents play an
important role in educating
beneficiaries and the reduced level of
compensation would result in a negative
impact on beneficiaries, as it would
reduce the level and quality of services
provided to beneficiaries, resulting in
less information and poor plan choices
made by beneficiaries and would also
result in agents leaving the MA
marketplace. Many commenters stated
that agents spend a significant amount
of time in training, preparing, and
testing in order to properly educate
beneficiaries about plan choices. A
number of commenters stated that their
overhead costs (travel, postage, facility
costs) were significant and a reduction
in compensation would affect this
aspect of their business. Commenters
also stated that the lower compensation
would discourage new agents from
entering the MA market.
Response: Based on the comments
received, we are modifying our
proposed regulations to permit the
renewal payment to be up to 50 percent
of FMV. MA organizations and Part D
sponsors may still determine how much
will be paid, up to 50 percent of the
current FMV, and retain the authority to
specify the details of their contracts
with agents, including how many years
renewal payments will be made. We
believe that this increased percentage
meets the statutory standard of
‘‘ensur[ing] that the use of
compensation creates incentives for
agents and brokers to enroll individuals
in the MA plan that is intended to best
meet their health care needs.’’
Comment: We received one comment
from an individual who misunderstood
our proposal. It appears that the
commenter thought our proposal would
allow two different payment options.
Response: We have reviewed this
comment and are not taking action
based on an incorrect understanding of
the proposal. Our proposal actually
discussed two options that we
considered for the renewal
compensation.
Comment: We received two comments
from individuals who suggested
alternative agent payment strategies.
One commenter suggested modifying
Medicare.gov to track agents for
enrollments processed through the Web
site for payment by plans. The
commenter also proposed paying agents
on a monthly basis, coinciding with the
months the beneficiary is in a plan,
eliminating the need to commission
reversals. Another commenter proposed
that plans submit compensation
schedules to CMS for review and
approval.
Response: These recommendations
entail significant changes with
numerous operational implications.
Therefore, we are not implementing the
suggestions from these comments at this
time. With respect to the comment
regarding the frequency of payments, we
did not propose to modify the existing
regulatory permission for MA
organizations and Part D sponsors to
determine whether payments would be
made at one time or in installments;
therefore the comment is outside the
scope of this proposed rule.
Comment: We received a few
comments regarding the requirement
that payments be made between January
1 and December 31 of the enrollment
year. One commenter supported the
proposal. A few commenters did not
support the January 1 date because
agents would have to wait 3 to 4 months
for compensation for those enrolling
during the AEP. One of these
commenters also noted that getting the
commission assures agents that the
beneficiary was enrolled. A few plans
were concerned about timely payment
of December 1 effective enrollees.
Response: Our proposal is aimed at
simplifying compensation while
ensuring an even playing field. As
explained previously, using a January 1
through December 31 payment
timeframe limits the recoupment of
payments made when a beneficiary
makes more than one election during
the AEP. Therefore, we are
implementing this provision as
proposed.
Comment: We received one comment
stating that regulating the payment of
only independent agents was unfair and
that employed agents should also be
regulated.
Response: We have reviewed this
comment and have determined that the
regulation of only independent agents is
still appropriate. Our initial regulations
were promulgated to ensure that agents/
brokers do not steer beneficiaries into
plans due to the agent’s/broker’s
financial or other interest; we continue
to be concerned about such steerage on
the part of independent agents, since
they often sell multiple products, with
varying levels of compensation. In
contrast, employed agents work for only
one company and therefore do not have
an incentive to move a member into a
plan offered by a different organization
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00020 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29863
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
or sponsor in exchange for a higher
commission.
Comment: We received a comment
from a trade association recommending
that CMS consider changes from cost
plans to MA plans as ‘‘like’’ plan
changes, rather than ‘‘unlike’’ plan
changes for compensation purposes.
The commenter stated beneficiaries
evaluate cost plans similar to MA plans
and that treating these as unlike plan
types encourages churning.
Response: We have received this
comment and declined to implement
such this change from our proposal; we
believe that this is outside the scope of
the proposed rule. Our proposal did not
address what constitutes ‘‘like’’ and
‘‘unlike’’ plan types, but instead simply
referenced ‘‘like’’ and ‘‘unlike’’ plan
types, using the existing regulation
standards on this point, because CMS
re-designated and revised certain
portions of the existing regulation for
simplification.
Comment: We received a few
comments regarding referral fees. One
commenter recommended that the
referral fee for enrollments be limited to
FMV instead of $100. Other commenters
requested that CMS not allow referral
fees to be paid.
Response: We reviewed these
comments and are finalizing our
proposal as described in the preamble to
the proposed rule, with the changes to
the regulation text at subparagraph
(h)(1) as explained previously. Referral
fees are applicable to employed, captive,
and independent agents, and permitting
the referral fee to be as high as the Fair
Market Value (FMV) would increase the
potential for steerage among different
types of agents and thus plans. The $100
cap, which is required to be part of the
total compensation, is an added
protection to ensure financial interests
of agents do result in misleading
beneficiaries. Our proposal did not
address whether referral fees should be
permitted, only whether such fees
should be capped and, if so, at what
level. We do not believe that it is
appropriate to prohibit or eliminate
referral fees without additional rule-
making that is specific on that question.
Comment: One plan requested
clarification as to whether the renewal
rate of the ‘‘current’’ FMV meant the
year in which the renewal commission
is being paid.
Response: We intend, for purposes of
renewal rates, that the ‘‘current’’ FMV
be the FMV for the enrollment year. For
example, an agent would be paid 50
percent of Contract Year (CY) 2015’s
FMV for a renewal member who is
enrolled in CY 2015.
Comment: One plan requested
clarification as to whether CMS would
require payments to be retroactive or if
the existing regulations would continue
until member’s current 6-year cycle
ended. One trade organization wanted
to know whether the requirements will
be effective for January 1, 2015
enrollments and how the new
regulations will affect members
currently in the existing 6-year cycle.
Response: As part of this final rule,
the new compensation requirements
will be implemented for all members for
CY2015. One of CMS’ intentions was to
simplify the regulations and create an
even playing field. We would not be
able to accomplish these goals if we
were to wait to implement these new
requirements until all members finish
their current 6-year cycle. However, we
note that the final provides flexibility to
MA organizations and Part D sponsors
so long as payments are within the
thresholds established in the rule. To
the extent that an MA organization or
Part D sponsor wishes to continue
payment using a cycle system,
negotiates that payment structure with
its agents and brokers, and that cycle
system complies with the limits and
requirements of this final rule, the MA
organization or Part D sponsor may do
so.
Comment: We received a few
comments concerning recoupment of
compensation when a member
disenrolls within the first three months
of enrollment. One plan requested a
better definition of ‘‘broker behavior.’’
One trade association stated that there
would be significant challenges in
determining whether disenrollments
were due to independent agent/broker
conduct. The trade association is
concerned that plans could face
significant disputes with agents/brokers
about these decisions.
Response: We have reviewed these
comments and determined that the
current situation should remain
unchanged based on these concerns that
our proposed revisions would hamper
MA organizations’ and Part D sponsors’
ability to determine which enrollments
should be fully recouped, with the
result that compensation is either
inappropriately recouped or not
recouped when necessary. Therefore,
we are finalizing the regulation to
require full recoupment of
compensation when a member
disenrolls within the first three months
unless CMS determines that the
recoupment is not in the best interests
of the Medicare program. CMS will
apply this standard and specify
exceptions in sub-regulatory guidance.
Our current guidance is consistent with
this standard and will remain
applicable.
Comment: We received a few
comments regarding the implementation
date of the regulations. One trade
association stated that it typically took
nine months to make systems changes to
accommodate new requirements.
Response: We understand that
systems changes may take time to
implement. Because of necessary
industry systems changes, and because
the rule provides for a payment
structure applicable by calendar year,
these compensation changes do not take
effect until enrollments effective
January 2015. Therefore, organizations
and sponsors will have approximately
seven months to make such changes.
Other than simplifying how FMV
applies to renewal rates, the new
compensation structure is similar to
industry practice and present guidance.
Therefore, we did not make any changes
to this section of the regulation.
Comment: One trade organization
commented that many MA
organizations and Part D sponsors
currently operate on a ‘‘rolling year’’
basis, such that, if an enrollment is
effective February 1, the compensation
covers the period starting on February 1
and continuing through January 31 of
the following year. The association said
that these were well-established
processes and a change could disrupt
systems and require a significant re-
design effort.
Response: Our position has always
been that organizations and sponsors
were required under the existing rules
to pay compensation on a calendar year
basis, not a ‘‘rolling’’ year basis. When
we encountered situations where
organizations and sponsors have not
implemented these requirements
correctly, we have required the
organization to adjust its processes to
comply and they have done so in a
timely manner. We decided to clarify
this requirement in our regulations to
ensure that all plans fully understand
the CMS definition of an enrollment
year. Therefore, we will not be making
any modification based on this
comment.
Comment: One trade association
stated that the NPRM appears to have
eliminated the current provisions at
§§ 422.2274(a)(1)(iv) and
423.2274(a)(1)(iv), which address
compensation requirements for Third
Party Entities.
Response: We thank the commenter
for this observation. These provisions
were inadvertently eliminated from the
current provisions. We have revised the
regulation text accordingly.
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00021 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29864
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
Comment: We received one comment
from a trade association that was
concerned about CMS’ requirement to
recover commissions if an enrollee
disenrolls in the middle of the year.
They suggested that CMS require MA
organizations and Part D sponsors to
take ‘‘commercially reasonable efforts’’
to recover funds.
Response: The requirement to recover
funds when a member disenrolls mid-
year remains the same; we did not
propose to change this requirement.
Organizations and sponsors have the
ability to make payments yearly,
quarterly, monthly, or in other
frequencies. Therefore, they could pay
monthly, rather than on a yearly or
quarterly basis, and thereby limit the
need to recoup funds for disenrollments
that occur at mid-year. Therefore, we
will not be making any changes to the
regulation.
Comment: We received a few
comments recommending that CMS
provide a mandatory plan comparison
form. Agents/Brokers would be required
to fill this out and provide to the
beneficiary for review.
Response: These comments are
outside the scope of our proposed rule,
but we will consider this suggestion for
future changes.
Comment: We received a few
comments from beneficiary advocacy
groups stating that MA organizations
and Part D sponsors slow down,
artificially delay, or dispute the
payment of compensation, which
ultimately encourages agents and
brokers to take their business to another
plan.
Response: These comments are
outside the scope of our proposed rule,
but we believe that our new requirement
that compensation be paid within the
enrollment year will address some of
these issues.
After consideration of the public
comments received, we are finalizing
our proposal at §§ 422.2274(a), (b) and
(h) and 423.2274(a), (b), and (h) with the
following modifications as previously
discussed:
• Deleting the introductory text to the
regulation section.
• Raising the renewal compensation
rate from 35 percent to (up to) 50
percent of the current fair market value
cut-off amounts published annually by
CMS.
• Removing the proposed recoupment
standard for rapid disenrollments by
reverting to the status quo where
subregulatory guidance describes
activities not triggering recoupments
(rather than requiring recoupment based
on ‘‘agent or broker behavior’’;
implementing a standard based on the
best interests of the Medicare program
to identify disenrollments that do not
require recoupment.
• Incorporating existing regulation
text about compensation to Field
Marketing Organizations.
• Clarifying the CMS standard for
applying the limit on referral fees.
8. Drug Categories or Classes of Clinical
Concern (§ 423.120(b)(2)(v))
Section 3307 of the Affordable Care
Act amended section 1860D–4(b)(3)(G)
of the Act by replacing the specific
criteria established under MIPPA in
2008 to identify categories or classes of
Part D drugs for which all Part D drugs
therein shall be included on Part D
sponsor formularies. The specified
criteria were replaced with the
requirement that the Secretary establish
criteria through notice and comment
rulemaking to identify drug categories
or classes of clinical concern. In
addition, section 3307 of the Affordable
Care Act requires the Secretary to
engage in rulemaking to establish
exceptions that permit a Part D sponsor
to exclude from its formulary a
particular Part D drug that is otherwise
required to be included in the formulary
in a drug category or class of clinical
concern (or otherwise limit access to
such a drug, including through prior
authorization or utilization
management). The Affordable Care Act
amendments to section 1860D–
4(b)(3)(G) of the Act specified that until
such time as the Secretary establishes
through rulemaking the criteria to
identify drug categories or classes of
clinical concern through rulemaking,
the following categories or classes shall
be identified as categories or classes of
clinical concern: anticonvulsants,
antidepressants, antineoplastics,
antipsychotics, antiretrovirals, and
immunosuppressants for the treatment
of transplant rejection. We proposed to
implement the Affordable Care Act
requirements set forth in section 1860D–
4(b)(3)(G) of the Act by revising
§ 423.120(b)(2)(v) and (vi) to specify: (1)
the criteria the Secretary will use to
identify drug categories or classes of
clinical concern; and (2) exceptions that
permit Part D sponsors to exclude a
particular Part D drug from within a
category or class of clinical concern that
is otherwise required to be included in
the formulary (or to otherwise limit
access to such a drug, including through
utilization management or prior
authorization restrictions). We also
proposed to specify which drug
categories or classes met the proposed
criteria and explained the process we
used for making these determinations.
We proposed to modify
§ 423.120(b)(2)(v) to require that, unless
an exception applies, all Part D drugs
within a drug category or class be
included on the formulary if the drug
category or class of drugs for a typical
individual with a disease or condition
treated by the drugs in the category or
class meets both of the following
criteria, as determined by CMS—
• Hospitalization, persistent or
significant disability or incapacity, or
death likely will result if initial
administration (including self-
administration) of a drug in the category
or class does not occur within 7 days of
the date the prescription for the drug
was presented to the pharmacy to be
filled; and
• More specific CMS formulary
requirements will not suffice to meet the
universe of clinical drug-and-disease-
specific applications due to the
diversity of disease or condition
manifestations and associated
specificity or variability of drug
therapies necessary to treat such
manifestations.
We were concerned that requiring
essentially open coverage of certain
categories and classes of drugs presents
both patient welfare concerns and
financial disadvantages for the Part D
program as a result of increased drug
prices and overutilization. We also
believed that criteria for identifying
drug categories and classes of clinical
concern should identify only those drug
categories or classes for which access
cannot be adequately ensured by
beneficiary protections that otherwise
apply. Consequently, as we took the
opportunity to propose to codify criteria
for identifying categories or classes of
drugs that are of clinical concern, we
believed that the requirements of
section 3307 of the Affordable Care Act
should be implemented taking into
consideration the other protections
available to beneficiaries. Otherwise, we
believed section 3307 of the Affordable
Care Act would establish duplicative,
and thus unnecessary, protections that
would serve only to increase Part D
costs—without any added benefit and
with the possibility of added harm from
misuse. Therefore, in considering
whether additional protections continue
to be needed under this section, we
needed to take the other beneficiary
access protections into account. We
detailed five such protections:
formulary transparency, formulary
requirements, reassignment formulary
coverage notices, transition supplies
and notices, and the coverage
determination and appeals processes.
Taken together, we believed these
requirements were comprehensive
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00022 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29865
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
enough that additional access
safeguards would be needed only in
those situations where a Part D
beneficiary’s clinical needs cannot be
more efficiently met.
We received the following comments,
and our response follows:
Comment: We received strong support
for our entire proposal from some
commenters who agreed, as they stated,
with ‘‘all of the reasons’’ underlying the
proposal, but we received no supportive
comments explicitly directed toward
our proposed criteria for identifying
categories and classes of clinical
concern. However, we did receive
significant opposition to our proposed
criteria. Several commenters generally
stated that the criteria themselves were
flawed, much less their application to
the drug categories and classes in our
analysis. Although the statute did not
provide individual criteria, some
commenters stated that the criteria were
more restrictive than statutory intent
and insufficiently accounted for patient
complexity. Other commenters stated
that application of overly restrictive
criteria set a dangerous precedent, and
other commenters raised related
concerns that other categories and
classes of clinical concern could be
eliminated in the future or that they
could be incorrectly applied to other
disease states whose guidelines indicate
the use of these drugs. For example,
many commenters expressed concern
that if immunosuppressants for
transplant rejection no longer received
the additional protections under section
3307, patients with multiple sclerosis
who use immunosuppressants may face
access issues. Additionally, although it
was mentioned as a source of savings in
our RIA, some commenters opposed the
idea that future drugs in a particular
category or class, representing advances
in therapy, may not be covered,
believing this jeopardized beneficiary
health. Indeed, many commenters stated
that the application of these criteria
would be life-threatening.
Response: We thank the commenters
for bringing their concerns to our
attention. We attempted to embrace the
principle of balancing access and cost
through optimal formulary management
inherent in the design of the Part D
benefit in proposing to establish criteria
pursuant to section 3307 of the
Affordable Care Act. However, based on
the comments received, we have
concluded that our proposed criteria did
not strike the balance among beneficiary
access, quality assurance, cost-
containment, and patient welfare that
we were striving to achieve. Thus, we
are not finalizing our proposal to
establish new criteria for the categories
and classes of clinical concern.
Accordingly, we are maintaining the
existing six categories and classes of
clinical concern listed in the statute and
are amending the regulation at
§ 423.120(b)(2)(v) to reflect that the
categories and classes of clinical
concern will be as specified in section
1860D–4(b)(3)(G)(iv) of the Act until we
undertake rulemaking to specify criteria
to identify the categories and classes of
clinical concern.
During our annual formulary review
and approval process, regardless of a
drug’s placement in a category or class
of clinical concern, to the extent that a
treatment guideline speaks to a specific
category or class of drugs, we look for
representation from that category or
class of drugs on the formulary.
Moreover, if the treatment guidelines
address specific drugs, we would review
formularies to ensure inclusion of those
specific drugs. Thus, although a
category or class of clinical concern is
immunosuppressants for transplant
rejection, to the extent that the
treatment guidelines for multiple
sclerosis indicate the use of
immunosuppressants, we still would
look for representation of these drugs on
the formulary during our treatment
guidelines review for multiple sclerosis.
After consideration of the public
comments we received, we are
finalizing a technical change to
§ 423.120(b)(2)(v) to reflect the existing
categories and classes of clinical
concern. Because the existing regulation
at § 423.120(b)(2)(v) is obsolete in light
of the Affordable Care Act, we are
making a technical change to specify
that until such time as we undertake
rulemaking to establish criteria to
identify, as appropriate, categories and
classes of drugs for which we determine
are of clinical concern, the categories
and classes of clinical concern shall be
as specified in section 1860D–
4(b)(3)(G)(iv) of the Act.
9. Medication Therapy Management
Program (MTM) Under Part D
(§ 423.153(d))
Section 1860D–4(c)(2) of the Act
provides that Part D sponsors, in
offering Medication Therapy
Management (MTM) programs, must
target individuals who: (1) have
multiple chronic diseases (such as
diabetes, asthma, hypertension,
hyperlipidemia, and congestive heart
failure); (2) are taking multiple covered
Part D drugs; and (3) are identified as
likely to incur annual costs for covered
Part D drugs that exceed a level
specified by the Secretary. At the start
of the Part D program, we believed that
25 percent of enrollees would qualify
for MTM services. However, analysis
revealed that MTM program enrollment
was well below that level. In the 2010
Call Letter and subsequent regulation,
we modified the criteria to reduce the
variability in eligibility and level of
service and to improve access to MTM
services, again targeting 25 percent of
enrollees. Despite these changes, MTM
program participation remains very low.
Moreover, additional evidence that the
program improves quality and generates
medical savings supports the belief that
more than 25 percent of enrollees will
benefit from MTM services.
We continue to see restrictive criteria,
such as plan sponsors specifying a
narrow list of chronic diseases or Part D
drugs coupled with requiring a higher
minimum number of covered drugs (for
example, eight drugs versus two) for
eligibility. As a result, access to MTM
services remains very low with MTM
program eligibility rates at less than 8
percent in 2011. In the proposed rule,
we cited a number of studies which
discussed the following: there may be
racial disparities in meeting the
eligibility criteria, the current eligibility
criteria and variability are restricting
access to MTM services, and MTM
enrollees with certain chronic diseases,
particularly those who received annual
comprehensive medication reviews
(CMRs), experienced significant
improvements in drug therapy outcomes
when compared to beneficiaries who
did not receive any MTM services, and
cost savings.
We believe the studies support the
necessity to reduce variability and racial
disparity in eligibility criteria among
plans and improve access to beneficial
MTM services. We proposed changes to
the eligibility requirements regarding
multiple chronic diseases, multiple Part
D drugs, and the annual cost threshold.
a. Multiple Chronic Diseases
Under the statute, one of the three
criteria that are used to target
beneficiaries for MTM services is
whether a Part D beneficiary has
multiple chronic diseases such as
diabetes, asthma, hypertension,
hyperlipidemia, and congestive heart
failure. We previously interpreted this
language to allow sponsors to define
‘‘multiple chronic diseases’’ with three
chronic diseases being the maximum
number a plan sponsor may require for
targeted enrollment. Further, sponsors
are allowed to target beneficiaries with
select chronic diseases, but must
include at least five of the nine core
chronic diseases in their criteria. This
list of core chronic diseases, as updated
in the 2013 Call Letter (available at
http://www.cms.gov/Medicare/Health-
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00023 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29866
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
Plans/MedicareAdvtgSpecRateStats/
Downloads/Announcement2013.pdf),
includes hypertension, congestive heart
failure, diabetes, dyslipidemia,
respiratory disease, bone disease—
arthritis, mental health, Alzheimer’s
disease, and end stage renal disease. We
proposed to revise our interpretation of
‘‘multiple chronic diseases’’ to require
that sponsors must target enrollees
having two or more chronic diseases for
MTM services. We also proposed to
require that at least one of the chronic
diseases that a beneficiary has in order
to satisfy the eligibility criteria must be
one of the list of core chronic diseases.
In addition, we proposed to redefine the
core diseases by combining
hypertension and congestive heart
failure under the umbrella of
‘‘cardiovascular disease,’’ which would
also encompass congestive heart failure,
acute myocardial infarction, cerebral
hemorrhage and effects of stroke,
vascular disease, specified heart
arrhythmias, and hypertensive heart
disease. The proposed list of core
chronic diseases became cardiovascular
disease, diabetes, dyslipidemia,
respiratory disease, bone disease—
arthritis, mental health, Alzheimer’s
disease, and end stage renal disease.
b. Multiple Part D Drugs
The second of the three statutory
criteria for identifying targeted
beneficiaries is whether a Part D
beneficiary is taking multiple covered
Part D drugs. We proposed to revise our
interpretation of ‘‘multiple Part D
drugs’’ to require that sponsors must
target enrollees taking two or more Part
D covered drugs for MTM services. We
also proposed to restrict the flexibility
previously available to sponsors by
requiring that they consider any Part D
covered drug. In the proposed rule, we
cited literature that supported the idea
that patients with multiple diseases and
taking at least two drugs are more likely
to have drug therapy problems and need
MTM.
c. Annual Cost Threshold
The final statutory requirement for
targeting Part D beneficiaries for MTM
services is that the beneficiary be
identified as likely to incur costs for
covered Part D drugs that exceed a level
specified by the Secretary. The Congress
did not impose any specific
requirements with respect to the cost
threshold at the time the MTM criteria
were passed in to law, nor has it
addressed this threshold in any of the
subsequent amendments to section
1860D–4(c)(2) of the Act. We previously
codified a $3,000 threshold, as updated
annually by the annual percentage
increase in the average per capita
aggregate expenditures for Part D drugs
for Part D eligible individuals under
§ 423.104(d)(5)(iv) in the April 2010
final rule entitled, ‘‘Policy and
Technical Changes to the Medicare
Advantage and Medicare Prescription
Drug Benefit Programs’’ (75 FR 19818).
The threshold is currently $3,017 in
2014. However, we are concerned that
there are a number of beneficiaries who
need MTM, but are not currently
eligible because they do not meet the
current cost threshold of $3,017, despite
the increased likelihood of having drug
therapy problems as a result of having
multiple chronic diseases and taking
multiple medications. Moreover, the
current cost threshold may have the
unintended consequence of causing
beneficiaries to no longer qualify for
MTM services in the next plan year
(whether remaining in the same plan or
enrolling into a new plan) if they fall
below the cost threshold as a result of
their enrollment in plans that employ
cost avoidant strategies, such as
aggressive use of generics, or in MTM
programs that center on therapeutic
interchange. Consistent with our
proposal that sponsors must target
enrollees taking two or more Part D
covered drugs for MTM services and
taking into account that one or more of
these Part D drugs are likely to be
generics, we proposed setting the
annual amount in Part D drug costs at
an amount that represents the
intersection of multiple conditions and
multiple drugs. Specifically, we
proposed setting the threshold at $620
which is the estimated annual total drug
cost for a beneficiary filling two generic
prescriptions, based on an analysis of
prescription drug event (PDE) data.
We are not finalizing these proposals.
We will engage in new notice and
comment rulemaking on this issue as
warranted in the future.
We received a large number of
comments related to our proposal to
revise § 423.153(d)(2)(i) through (iii) to
expand MTM program eligibility and
our response follows.
Comment: Many commenters were
supportive of MTM in general and CMS’
goals. These commenters were
supportive of the proposed changes to
expand access to MTM services, shared
CMS’ concerns regarding restrictive and
variable eligibility criteria established
by some sponsors, and endorsed the
proposals to revise the eligibility criteria
to increase uniformity. This included
support for and clarifying questions
regarding the revised definitions for
‘‘multiple chronic diseases,’’ with the
addition of ‘‘cardiovascular disease’’ to
the list of core diseases, and ‘‘multiple
Part D drugs.’’ Some commenters stated
that CMS should post MTM eligibility
rates on the CMS Web site or make plan-
reported data more available for
research. Other commenters, who
supported the proposed changes to
expand access to MTM, provided
information on return on investment,
outcomes, or individual experiences in
improving quality and lowering costs
through MTM provided by community
pharmacists who have close
relationships with the beneficiaries and
local prescribers. A large number of
commenters also stated that, to date,
variability in plan offerings and limited
compensation has made the provision of
MTM in the community setting difficult
in a consistent, scalable and timely
manner.
A significant number of commenters
also were strongly opposed to the broad
expansion of eligibility. They
questioned the effectiveness of
expansion under the current
infrastructure as delivered by drug plans
with limited incentives and a lack of
care coordination, and they commented
that the clinical evidence did not
support the proposed changes. We
received many comments that the
proposed changes would significantly
increase costs (both administrative and
beneficiary premiums), reduce the
quality of programs delivered to
beneficiaries who most need MTM, and
could overwhelm limited resources.
Many commenters requested that the
proposed changes be withdrawn, and
some commenters offered alternative
eligibility criteria for CMS to consider in
the future. These included: delay the
proposed changes or implement the
changes incrementally, alternative
criteria for the minimum thresholds for
eligibility, alternative eligibility criteria
based on risk factors, and requiring
MTM at transition of care.
Response: We thank these
commenters for their thoughtful and
supportive comments. MTM has been
shown to improve drug therapy
outcomes and lower costs, and we agree
that the use of community-based
resources for providing MTM services
shows promise in improving access and
quality. We still have concerns that
many sponsors are applying restrictive
criteria to narrow the pool of targeted
beneficiaries for MTM rather than
optimizing the eligibility criteria to offer
MTM to beneficiaries who will most
benefit from these services. These
programs are not living up to our
expectations. As we discussed in the
regulatory impact analysis for the
proposed rule (79 FR 2036), we estimate
that only 2.5 million beneficiaries (8
percent) are eligible for MTM services,
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00024 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29867
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
13 percent opt-out of the MTM program,
and 10 percent of participating
beneficiaries receive an annual CMR.
That means that less than 220,000 Part
D enrollees receive CMRs, which
studies have shown is a crucial element
of MTM to improve drug therapy
outcomes and lower costs. Not enough
is being done by sponsors to provide
sufficient access to MTM services and
engage beneficiaries and providers in
this process. We will consider publicly
posting the MTM program eligibility
rates for each Part D contract, similar to
how we display MTM program CMR
rates, and explore ways to make the
plan-reported data available for public
use.
Despite the persuasive comments
from those who support the proposed
changes in eligibility criteria, we also
take into account the comments that the
timeline for implementing the proposed
changes may be too aggressive and
could negatively affect existing MTM
programs. While our goal was to
increase eligibility and access to MTM,
we do not want to do it at the expense
of sacrificing any quality with existing
programs. Therefore, we are not
finalizing our proposed changes to the
eligibility criteria. But, we will continue
to evaluate information on MTM
programs and monitor sponsors’
compliance in accordance with the
MTM requirements established by
§ 423.153, with the goal of proposing
other revisions to criteria in future
rulemaking that will help expand the
program. We believe that Part D
sponsors can target more beneficiaries
for MTM under the existing criteria. We
plan to closely scrutinize sponsors that
may be abusing the flexibility provided
to them in establishing the eligibility
criteria, which may have contributed to
the racial disparity, variability, and
beneficiary confusion with respect to
MTM eligibility that we identified in the
proposed rule. We will consider the
commenters’ suggestions for alternative
criteria and may consider revisions to
MTM eligibility criteria for future
rulemaking. We may also consider
changes to the definitions for ‘‘multiple
chronic diseases,’’ including the core
chronic diseases, and ‘‘multiple Part D
drugs’’ in the future.
10. Requirement for Applicants or Their
Contracted First Tier, Downstream, or
Related Entities To Have Experience in
the Part D Program Providing Key Part
D Functions (§ 423.504(b))
Since its establishment in 2006, the
Medicare Part D program has matured
into a generally stable, well-functioning
program, and the Part D sponsors (as
well as their first tier, downstream, and
related entities (FDRs)) with which CMS
contracts have developed vast expertise
in the operational complexities of the
program. While we will continue to fine
tune the program through rulemaking,
guidance, and additional oversight
procedures, we believe the program has
largely entered a mature stage. Despite
this progress, we still find ourselves
spending a disproportionate amount of
resources and attention on the
operations of new Part D sponsors
where neither the new sponsor nor its
supporting FDRs have experience with
Part D.
To address this problem, pursuant to
our authority at section 1860D–
12(b)(3)(D) of the Act to adopt
additional contract terms, not
inconsistent with the Part C and D
statutes, that are necessary and
appropriate to administer the Part D
program, we proposed to adopt
provisions that would require any entity
seeking to contract as a Part D plan
sponsor (as a stand-alone prescription
drug plan sponsor or as a MA
organization offering Part D benefits) to
have arrangements in place such that
either the applicant or one of its
contracted FDRs has one full benefit
year serving as a Part D plan sponsor, or
at least one full benefit year of
experience performing key Part D
functions for another Part D plan
sponsor. The applicant or a contracted
FDR will be required to have obtained
that experience within the 2 years
preceding the Part D sponsor
qualification application submission.
Under this proposal, the experience
requirement would be met by an entity
seeking to contract as a Part D plan
sponsor if its parent or another
subsidiary of that parent already holds
a Part D sponsor contract that has been
in effect for at least one year at the time
of the application submission.
Given the wealth of available Part D
expertise that now exists, it is justifiable
for us to require that new applicants to
the program bring with them Part D
experience so that we can better protect
Part D enrollees and minimize
unnecessary expenditures of resources
by us in correcting avoidable problems.
When neither a Part D sponsor, nor its
FDRs providing key Part D functions,
has any experience delivering Part D
benefits, the consequences can be
disastrous for beneficiaries and highly
disruptive for the program and CMS.
While there are many operational
functions that must run smoothly for a
Part D plan to be successful (for
example, pharmacy network
development/maintenance, enrollment
processing, prescription drug discount
negotiation, and provision of customer
service), we proposed to require Part D
experience in only three critical areas in
which beneficiaries are particularly
vulnerable should the sponsor
demonstrate significant non-
compliance. The three areas for which
we proposed to require prior experience
in Part D at the time of application to
become a new Part D sponsor are—
• (1) Authorization, adjudication and
processing of pharmacy claims at the
point of sale;
• (2) Administration and tracking of
enrollees’ drug benefits in real time,
including automated coordination of
benefits with other payers; and
• (3) Operation of an enrollee appeals
and grievance process.
It is in these three areas where—in
our view, based on our experience with
Part D—enrollee health is placed at the
most significant risk by Part D sponsor
compliance failures.
Under our proposal, multiple separate
organizations could together combine
their experience to meet the prior
qualification requirements for the three
key Part D functions. That is, no one
single entity would need to have prior
experience in all three areas. Rather, the
requirement would be for the Part D
applicant in combination with its FDRs,
if any, to have Part D experience
covering the three key functions.
Our proposal also does not prohibit
additional organizations from gaining
Part D experience in the selected key
functional areas. Should an organization
wish to become a new Part D FDR for
one or more of the key functions, this
‘‘novice’’ entity could provide the
service for just one of the hundreds of
existing Part D sponsors. After a period
of one year, the novice entity would
then be qualified to provide its services
to existing Part D sponsors as well as
partner with new Part D applicants. In
somewhat the opposite scenario, a new
Part D sponsor contracting with
experienced FDRs will have the
opportunity to gain its experience in the
key Part D functions by working closely
with its FDRs, developing in house
expertise, and providing oversight. After
a period of one or more years, if desired,
the Part D sponsor itself could
conceivably take responsibility for
carrying out one or more of the key Part
D functions.
While our proposal did not require
the Part D experience to be current at
the time of an application to become a
Part D sponsor, we proposed that the
experience be recent (that is, within the
past 2 years) and have lasted for at least
one full benefit year. We believe that
any experience older than 2 years would
be out of date and would not represent
experience with the current state of the
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00025 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29868
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
Part D program. As for our proposed
requirement that the experience be for at
least a term of one full benefit year, this
approach is appropriate because
operating the benefit involves cyclical
activities, some of which take place only
one time per year, and thus an
organization can only gain full
experience by operating its Part D
functional area for an entire benefit
year.
We intend to implement this proposal
through our existing Part D contract
qualification application process, and
we proposed to amend § 423.504(b)
accordingly. Applicants with existing
Part D contracts or whose parents or
other subsidiaries of the same parent
hold Part D contracts will not be
required to submit evidence of their Part
D experience.
We received the following comments
and our response follows:
Comment: We received strong
statements of support from many
commenters. We received only one
suggestion of not finalizing the policy,
but the commenter did not provide any
details or rationale to support its
comment.
Response: We appreciate the
widespread support for this proposal.
Comment: We received one
recommendation to consider a less
stringent standard for employer groups
seeking to act as Employer Group
Waiver Plan (EGWP) sponsors.
Response: We expect all sponsors,
including EGWP sponsors, to meet our
experience and capability requirements.
We have an obligation to ensure that all
beneficiaries receive their benefits from
experienced Part D sponsors.
Comment: One commenter that
supported the policy suggested that
CMS should also address the problem of
applicants not having the skills or
capacity to even oversee their
experienced FDRs.
Response: We share the concern that
applicants may not have experience
overseeing FDRs, which is why, in
addition to the current requirements
and standards in place for
administration and management, we are
finalizing at section A.III.11. of this final
rule our proposed requirement that new
PDP sponsor applicants have
immediately prior to the date of the
application submission 2 years’
experience administering health
insurance benefits directly or 5 years’
experience providing certain
prescription drug benefit management
services to a health insurer . We also
have procedures and mechanisms in
place to monitor a Part D sponsor’s
administration and management of its
contract, including the option of
conducting an audit of a sponsor’s
operations prior to the start of the
contract year to confirm that it is
prepared to oversee the delivery of Part
D benefits to its members.
Given the near universal support for
this proposal we are finalizing this
provision without modification.
11. Requirement for Applicants for
Stand-Alone Part D Plan Sponsor
Contracts to Be Actively Engaged in the
Business of the Administration of
Health Insurance Benefits
(§ 423.504(b)(9))
The Medicare prescription drug
benefit program has matured into a
generally stable, well-functioning
program, and the Part D sponsors with
which CMS contracts have developed
vast expertise in the operational
complexities of the program. The market
for stand-alone Part D Prescription Drug
Plans (PDPs) has also matured
significantly since the program’s
inception and what was once a novel
product is now available to residents of
every state from multiple sponsors who
offer several plan options. Over the
same period, we have noticed that the
Part D program has in some cases
attracted sponsors wishing to offer
stand-alone PDPs who have no prior
experience in the delivery of health or
prescription drug insurance benefits,
often to the detriment of the Part D
program and the Medicare beneficiaries
who elect plans offered by these
sponsors.
To address this problem, we
proposed, pursuant to our authority at
section 1860D–12(b)(3)(D) of the Act to
adopt additional contract terms that are
necessary and appropriate to administer
the Part D program, regulatory
provisions that would require any entity
seeking to contract as a stand-alone PDP
sponsor, to have either actively
provided health insurance or health
benefits coverage for 2 continuous years
immediately prior to submitting a
contract qualification application, or
provided certain prescription drug
benefit management services to a
company providing health insurance or
health benefits coverage for 5
continuous years immediately prior to
submitting an application. This
requirement would not apply to an
entity seeking to contract as the sponsor
of a stand-alone PDP if its parent or
another subsidiary of itself or its parent
possesses the requisite experience.
This proposal may appear similar to
the immediately-preceding proposal
(section III.A.10. of this final rule)
requiring, at § 423.504(b)(8), that new
Part D sponsors engage first tier,
downstream, and related entities with
prior Part D experience. However, the
proposed change we are discussing in
this section, which we proposed to
codify at § 423.504(b)(9), would apply
only to entities seeking to contract as a
Part D sponsor of a stand-alone PDP,
whereas the proposed requirement at
§ 423.504(b)(8) would apply to all new
Part D sponsors, including those seeking
to contract as MA organizations offering
Part D through an MA–PD plan. We
proposed both requirements because the
problems encountered by new PDP
sponsors with no experience in the
health insurance market are distinct
from those encountered by new PDP
sponsors and MA organizations that use
PBMs with no experience in the Part D
market. New PDPs with no prior health
insurance or health benefits experience
have demonstrated significant problems
even when using experienced PBMs.
While relatively few sponsors fit this
profile each year, they have caused
disproportionate problems for
beneficiaries and CMS. Time and again,
these sponsors fail our past Medicare
contract performance and audit tests or
receive low quality scores (that is, star
ratings) because they lack the ability to
administer even the most basic elements
of a health or drug benefit program, let
alone one as complex as Medicare Part
D.
When the sponsor is a novice not only
to Medicare Part D, but also to virtually
every aspect of health benefits
administration, there is no assurance
that the entity will be able to administer
or oversee the most basic elements of
health benefits coverage, such as
processing claims, administering a
coverage determination and appeals
process, enrolling beneficiaries, or
administering the benefit as approved.
To entrust inexperienced applicants
with responsibility for correctly
operating a program for which even
experienced health insurers have had to
develop new expertise has proven to be
unacceptably risky. We proposed that
new applicants have 2-years of
experience providing health insurance
or health benefits coverage (that is,
operating as risk-bearing entities
licensed in the states where they offer
benefits) prior to applying as stand-
alone Part D Sponsors because we
believe that this provides sufficient time
to demonstrate the applicant’s ability to
operate a health plan. We believe that
requiring 2-years of experience as a risk
bearing entity offering health insurance
or health benefits coverage ensures that
new sponsors of stand-alone PDPs have
minimal experience operating a health
benefits program without unduly
limiting new entrants to the
marketplace.
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00026 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29869
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
We recognize that a number of PBMs
and Third Party Administrators with
experience administering prescription
drug benefits have entered the stand-
alone PDP market and have adapted to
providing the Part D benefit despite
their lack of previous experience as
health insurers. Therefore, we proposed
that organizations applying to contract
as stand-alone PDP sponsors that do not
have experience as a risk-bearing entity
providing health insurance or health
benefits coverage would, in the
alternative, be eligible to hold a PDP
contract if they had 5-continuous years
of experience performing services on
behalf of an insurer in the delivery of
benefits in any health insurance market
in the three key areas indicated in this
section III.A.10. of this final rule. The
three areas that we proposed as meeting
the experience requirements are: (1)
Adjudication and processing of
pharmacy claims at the point of sale; (2)
administration and tracking of enrollees’
drug benefits in real time, including
automated coordination of benefits with
other payers; and (3) operation of an
enrollee appeals and grievance process.
Our reasons for selecting these three
areas as meeting the experience
requirements are described in more
detail in the section of this rulemaking
notice relating to the proposed
requirement at § 423.504(b)(8) that new
Part D sponsors employ experienced
FDRs for these functions. We proposed
a longer experience requirement for
these entities because entities offering
these services face fewer barriers to
entry in the marketplace and are not as
tightly regulated as risk bearing entities.
Therefore, we believe that entities that
seek to qualify on the basis of their
experience as PBMs or Third Party
Administrators should be required to
have provided services in these key
areas for 5-continuous years, rather than
merely 2.
We intend to implement this proposal
through our existing Part D contract
qualification application process, and
we proposed to amend § 423.504(b)
accordingly.
We received the following comments
and our response follows:
Comment: We received strong
statements of support from several
commenters.
Response: We appreciate the support
for this proposal.
Comment: We received one
recommendation to consider a less
stringent standard for employer groups
seeking to act as EGWP sponsors.
Response: We are not persuaded by
this comment because, in general, we
expect that all sponsors, including
EGWP sponsors, meet all of our
experience and capability requirements.
EGWP sponsors perform the same core
functions as sponsors of individual
market PDPs, including claims
processing, formulary administration,
operation of an appeals and grievance
process, and coordination of benefits.
Therefore, the same concerns that led us
to adopt the requirement that new PDP
sponsors have experience in these areas
applies to EGWP sponsors as well as
sponsors of individual market plans.
Given the universal support for this
proposal, we are finalizing this
provision without modification.
12. Limit Parent Organizations to One
Prescription Drug Plan (PDP) Sponsor
Contract per PDP Region (§ 423.503)
Each year, we accept and review
applications from organizations seeking
to qualify to offer stand-alone
prescription drug plans in one or more
PDP regions. With limited exceptions
(for example, poor past contract
performance, limited Part D experience),
we approve all applications submitted
by organizations that demonstrate that
they meet all Part D application
requirements. We proposed, under our
authority at section 1860D–12(b)(3)(D)
of the Act to adopt additional contract
terms, not inconsistent with the Part C
and D statutes, that are necessary and
appropriate to administer the Part D
program, to add as a basis upon which
we may deny a PDP sponsor application
the fact that the applicant is applying
for qualification in a PDP Region where
another subsidiary of the applicant’s
parent organization already holds a PDP
sponsor contract. In our description of
this proposal, the term ‘‘parent
organization’’ refers to an entity that
controls a subsidiary through ownership
of more than 50 percent of the
subsidiary’s shares.
Section 1860D–12(b)(1) of the Act
provides that PDP sponsors may offer
multiple plan benefit packages (referred
to as PBPs or plans) under one PDP
sponsor contract. Therefore, parent
organizations need only one PDP
sponsor contract to offer the full range
of the possible plan options in a
particular PDP Region. Additionally,
informal communications made by past
requestors of duplicate contracts
indicated that the purpose has been to
either a) segregate low income
beneficiaries into their own contract, or
b) corral the experience of a particular
low-performing plan into its own CMS
contract so as not to taint the
performance rating of the better
performing plan offering, as
performance ratings are calculated at the
contract level. We oppose the
inefficiencies of duplicate contracts and
the gaming duplicate contracts can
support. That said, we welcomed
comments from industry, advocates, and
others as to circumstances for our
consideration under which duplicate
contracts may be beneficial.
One of the fundamental principles of
the Part D program is that the selection
of plans made available to beneficiaries
is the product of true competition
among PDP sponsors. Two subsidiaries
of the same parent organizations
offering plans in the same PDP region
are not truly competitors, as decisions
concerning their operations are
ultimately controlled by a single entity,
or parent organization. Also, we only
approve those PDP offerings that meet
the meaningful differences test stated at
§ 423.265(b)(2), and we apply that test at
the parent organization level. A parent
organization would not gain an
opportunity to offer more plan benefit
packages under two or more contracts it
controlled through its subsidiaries than
it would under one contract because we
would, as part of our bid review,
evaluate whether all the plans proposed
by the same parent organization met the
meaningful differences test.
The proposed limitation on the
number of PDP sponsor contracts a
parent may control in a PDP Region is
also necessary to preserve the integrity
of CMS’ star ratings. CMS assigns star
ratings at the contract level, and they are
intended to reflect all aspects of the PDP
operations controlled by a unique
contracting entity. However, that
principle is compromised when a parent
organization to one of the contracting
entities is permitted to control, through
other subsidiaries, more than one PDP
contract. Allowing a parent organization
to effectively administer two or more
PDP sponsor contracts would allow it
potentially to artificially inflate the star
ratings on one contract by excluding the
poor performance under its other
contract from the rating calculation. In
that instance, some beneficiaries could
make a plan election without complete
information about the performance of
the organization ultimately responsible
for the quality of services they would
receive by enrolling in that plan.
Based on our experience in
administering the Part D prescription
drug benefit program we do not believe
that there is a compelling justification
for parent organizations to administer
two PDP sponsor contracts in the same
PDP region. Moreover, such
arrangements impede our ability to
efficiently administer the Part D
program and provide a means by which
the integrity and reliability of our star
ratings system can be compromised.
Therefore, we proposed to amend
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00027 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29870
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
§ 423.503(a) by adding a paragraph (3)
stating that CMS will not approve an
application when it would result in the
applicant’s parent organization holding
more than one PDP sponsor contract in
the PDP region for which the applicant
is seeking qualification as a PDP
sponsor. We anticipate that we would
most frequently use this authority to
deny an application in instances where
the applicant’s parent organization
already controls a PDP sponsor contract,
either directly by acting as a PDP
sponsor itself (in instances when the
parent is licensed as a risk-bearing
entity) or through its ownership of a
subsidiary that qualifies as a PDP
sponsor and is a party to a stand-alone
PDP sponsor contract. In the less likely
situation where two or more
subsidiaries of the same parent
organization each submit applications
in the same year for PDP regions where
the parent organization controls no PDP
sponsor contracts, we would request
that the parent withdraw all but one of
the applications. In the absence of a
withdrawal election, we will deny all of
the parent organization’s applications.
We received the following comments
and the response follows:
Comment: The comments of several
beneficiary advocacy organizations
contained expressions of support for our
proposal, citing in particular the role it
will play in preserving the integrity of
CMS’ star ratings system.
Response: We appreciate the
expressions of support for our proposal.
Comment: Several commenters stated
their opposition to our proposal on the
basis that it would limit their business
opportunities and reduce competition in
the Part D market by reducing the
number of plan sponsors participating
in a given PDP region.
Response: We note that the
commenters did not describe or provide
examples of the nature of the business
opportunities that Part D sponsors and
their parent organizations would be
denied should this provision go into
effect. Also, we believe that to properly
assess the level of competition in the
Part D market, it is important to
consider not just the number of plan
sponsors offering benefits, but also
whether all of those sponsors truly have
incentives to compete against one
another. As we noted in our preamble
discussion to the proposed rule,
additional plan sponsors controlled by
entities that already participate in the
Part D market do not promote improved
plan options since subsidiaries of the
same parent cannot be said to be truly
in competition with each other. In a
truly competitive market, multiple
entities develop and promote products
to capture as large a share of that market
as possible at the expense of other
market participants. It is our experience
that two or more subsidiaries of the
same parent organization are ultimately
accountable to the same set of
shareholders and are administered by
the same senior management team. In
such an arrangement, we believe there
is little incentive for the parent
organization to manage one PDP
contract in a way that would attempt to
take enrollees away from, or prevent
beneficiaries from electing, plans
offered by the related entity operating a
second contract. We also note that none
of the commenters provided an
explanation as to how related entities
would truly compete in the same PDP
region.
Comment: Several plan sponsors that
have recently acquired other plan
sponsor contracts expressed their
concern that the new policy would
jeopardize their right to maintain two or
more contracts during a transition
period following the acquisition.
Response: We assure the commenters
that our proposal has no effect on our
application of the regulatory provision
at § 423.272(b)(3), which provides
acquiring organizations an exemption
from the meaningful differences
standard normally applied to a
sponsor’s (or its parent organization’s)
bids for a 2-year period following the
acquisition of or merger with another
Part D sponsor. We have allowed
acquiring sponsors to maintain the
separate acquired contract during the
authorized 2-year period, and we will
continue to apply that policy after the
adoption of this provision.
Comment: Some plan sponsors that
currently hold more than one PDP
sponsor contract in a PDP region
commented that they were concerned
that the proposed provision would
require them to consolidate their
operations into one contract.
Response: We note that the proposal
only addressed our intention to deny
applications for new contracts
submitted by entities related to
organizations that already hold a PDP
sponsor contract in a particular region.
As we discussed in the preamble to the
proposed regulation, we will continue
to encourage such organizations to
consolidate their contracts, but we are
not requiring organizations to take such
action at this time.
Comment: One commenter requested
that CMS revise the proposal to allow a
parent organization to hold two
contracts in the same PDP region if one
of those contracts is maintained solely
for the purpose of offering employer
group waiver plans (EGWPs). The
commenter explained that because
EGWPs operate differently than
individual market plans (for example,
different enrollment processes, the need
to coordinate with non-Part D
supplemental coverage), it may reduce
the complexity of a parent
organization’s Part D operations if it is
permitted to keep its EGWP business
under a separate contract. Moreover,
since EGWP plans are not offered to
individual beneficiaries, these contracts
would not be subject to the same
incentives that might encourage
sponsors to game their star rating
performance to attract enrollments.
Response: We do not believe the
commenter’s arguments support special
treatment under our proposal for
organizations offering EGWPs. While it
is true that CMS affords EGWPs,
through the application of our statutory
waiver authority, flexibility in meeting
Part D requirements, the resulting
differences in requirements are not so
significant that a separate EGWP-only
contract is necessary for an organization
to administer such plans successfully.
In fact, the resulting differences do not
represent conflicting requirements that
might create the need for a separate
contract held by a different legal entity
to administer EGWPs. Rather, the EGWP
requirements are a result of our
completely waiving certain
requirements (for example, pharmacy
access standards, prior approval of
marketing materials) or modifying other
requirements (for example, enrollment
limited to employer group members),
and a single plan sponsor can meet
these if it is already offering individual
market PDPs. In fact, it is common for
a PDP sponsor to sign a stand-alone PDP
contract with CMS that includes an
EGWP addendum through which the
single entity offers both individual
market plans and EGWPs (that is, ‘‘800
series’’ plans). Our experience in
administering the Part D program
indicates that a properly managed single
legal entity is capable of complying with
multiple sets of Part D requirements.
Also, while sponsors may not have the
same incentives to game the star rating
system to attract EGWP enrollments as
they do to attract individual
beneficiaries, that fact alone would not
support allowing sponsors to maintain
separate EGWP contracts. We believe
the single contract rule is necessary to
maintain the integrity of the star ratings
that are reported to the public. As we
stated earlier in the preamble discussion
of our proposal, star ratings are intended
to reflect all aspects of the PDP
operations controlled by a unique
contracting entity, including the
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00028 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29871
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
administration of EGWP products.
Allowing a parent organization to
maintain a separate EGWP contract
would mean that the star ratings
associated with each of its PDP
contracts contract would provide an
incomplete picture of the organization’s
performance. We believe that all
members of the public, including those
who make plan elections on behalf of
employer group members as well as
individual beneficiaries, benefit from
star ratings information that clearly
indicates the quality of all Part D
operations under one organization’s
control.
After consideration of the public
comments we received, we are
finalizing our proposal without
modification, with the exception of a
technical edit which changes the
proposed phrase ‘‘may not approve’’ to
‘‘does not approve’’ to clarify that CMS
will deny all applications that meet the
criteria stated in the provision.
13. Limit Stand-Alone Prescription Drug
Plan Sponsors To Offering No More
Than Two Plans per PDP Region
(§ 423.265)
Under our authority at section 1860D–
11(d) of the Act, we conduct
negotiations with stand-alone
prescription drug plan (PDP) sponsors
concerning our approval of the bids they
submit each year. As the Part D program
has evolved, we have adopted
regulations designed to authorize us to
use that negotiating authority to ensure
that the number of plans offered in a
given PDP region reflects a balance
between sponsors’ interest in providing
options tailored to meet the needs of a
diverse Medicare population and the
need to avoid creating undue confusion
for beneficiaries as they consider
various plan offerings. We continued
with this proposal our process of
updating our bid review authority to
reflect the evolution of the Part D
program by proposing to limit to two the
number of plans stand-alone PDP
sponsors may offer in each PDP region.
PDP sponsors must offer throughout a
PDP region at least one basic plan that
consists of: standard deductible and cost
sharing amounts (or actuarial
equivalents); an initial coverage limit
based on a set dollar amount of claims
paid on the beneficiary’s behalf during
the plan year; a coverage gap during
which a beneficiary pays more of his
drug costs; and finally, catastrophic
coverage that applies once a
beneficiary’s out-of-pocket expenditures
for the year have reached a certain
threshold. Prior to our adopting
regulations requiring meaningful
differences among each PDP sponsor’s
plan offerings in a PDP Region, CMS
guidance allowed sponsors that offered
a basic plan to offer in the same region
additional basic plans, as long as they
were actuarially equivalent to the basic
plan structure described in the statute.
These sponsors could also offer
enhanced alternative plans that provide
additional value to beneficiaries in the
form of reduced deductibles, reduced
copays, coverage of some or all drugs
while the beneficiary is in the gap
portion of the benefit, or some
combination of those features.
As we have gained experience with
the Part D program, we have made
consistent efforts to ensure that the
number and type of plan benefit
packages PDP sponsors may market to
beneficiaries are no more numerous
than necessary to afford beneficiaries
choices from among meaningfully
different plan options. In addition to
setting differential out-of-pocket-cost
(OOPC) targets each year to ensure
contracting organizations submit bids
that clearly offer differences in value to
beneficiaries, we issued regulations in
2010 that established at § 423.265(b)(2)
our authority to deny bids that are not
meaningfully different from other bids
submitted by the same organization in
the same service area. Our application
of this authority has effectively
eliminated PDP sponsors’ ability to offer
more than one basic plan in a PDP
region since all basic plan benefit
packages must be actuarially equivalent
to the standard benefit structure
discussed in the statute. That regulation
also effectively limited to two the
number of enhanced alternative plans
that we can approve for a single PDP
sponsor in a PDP region. As part of the
same 2010 rulemaking, we also
established at § 423.507(b)(1)(iii) our
authority to terminate existing plan
benefit packages that do not attract a
number of enrollees sufficient to
demonstrate their value in the Medicare
marketplace. Both of these authorities
have been effective tools in encouraging
the development of a variety of plan
offerings that provide meaningful
choices to beneficiaries without creating
undue confusion for beneficiaries.
We believe that the progressive
closure of the coverage gap provided for
in the Affordable Care Act affords us
another opportunity to promote even
greater clarity in the set of stand-alone
PDP plan options from which
beneficiaries may make an election.
Under the statute, beginning in 2011,
applicable beneficiaries enjoy discounts
of 50 percent off negotiated prices on
covered brand name drugs when
purchased while in the coverage gap
portion of the benefit. Also, since 2011,
the required coverage in the gap has
increased and will continue to do so
gradually until 2020, when the
combination of required coverage and
manufacturer discounts covers 75
percent on average for both brand-name
and generic drugs. This ‘‘closing’’ of the
coverage gap effectively will leave the
beneficiary with only a 25 percent cost
share on average across the entire
benefit (or its actuarial equivalent)
before the catastrophic threshold.
Our experience in applying the
meaningful differences standard
indicates that, as the Part D coverage
gap is closed, it will become
increasingly difficult for a PDP sponsor
to qualify to offer more than two plans
in the same service area and still meet
the meaningful differences test. Since
we began applying the meaningful
differences standard to our bid reviews,
we have generally approved two types
of enhanced alternative plans. The first
type of plan offers beneficiaries, in
exchange for a higher premium than
that charged for basic plan coverage,
significant reductions in the cost
sharing and deductible amounts
associated with the basic Part D benefit.
The second type offers even greater cost
sharing and deductible reductions as
well as coverage for many drugs in the
gap. Since coverage of Part D drugs in
the gap is the distinguishing feature
between the two types of enhanced
alternative plans currently available,
closing the coverage gap also means that
sponsors can no longer rely on it to
establish that their proposed second
enhanced alternative plan is
meaningfully different than their first.
Despite these developments, many
sponsors continue to submit three bids
per region each year. We believe that
plan sponsors and beneficiaries, as well
as the taxpayers, would be better served
by a more streamlined bid submission
process that limited sponsors to
submitting two PDP bids (one basic and
one enhanced) per PDP region each
year. This limitation would provide a
consistent bidding framework for all
sponsors, allowing them to focus on
quality, rather than quantity, in
development of their bids. It would also
reduce some of the sponsors’
administrative costs associated with
preparing, marketing, and administering
a third benefit package. It may also help
ensure that beneficiaries can choose
from a less confusing number of plans
that represent the best value each
sponsor can offer.
While the incremental closure of the
coverage gap continues until 2020, we
believe that the observed enrollment
trends in these plans demonstrate the
reduction in beneficiaries’ coverage gap
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00029 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29872
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
costs that has occurred already has
moved the stand alone PDP plan market
in a way that warrants the imposition of
the two plan limit as soon as possible.
In addition, in many cases one of the
two enhanced plans offers the minimum
level of supplemental coverage required
to meet our meaningful differences tests.
We refer to these as ‘‘low value
enhanced plans’’ to distinguish them
from second enhanced plans with
substantially more supplemental
coverage. In some cases, the premiums
for these low value enhanced plans have
been less than the premiums for the
sponsors’ basic plans due to favorable
risk selection. This occurs because
many of the beneficiaries with more
serious health issues and higher
utilization of prescription drugs are in
the low-income subsidy (LIS) eligible
population, which will not receive the
full LIS subsidies in plans with
supplemental coverage. For this reason
we neither auto-assign the LIS eligible
population into such plans, nor will this
population generally affirmatively
enroll in such plans. Thus, continuing
to permit multiple enhanced plans,
particularly low value enhanced plans,
facilitates risk segmentation. This can
increase costs for the Part D program
and the taxpayers overall. During the
most recently completed CY 2014 bid
review cycle, we continued to encounter
bids submitted by sponsors for low
value enhanced plans with premiums
lower than the premiums for their basic
plans. We believed it was urgent that we
adopt the proposed policy as soon as
possible so that we could bring an end
to this bidding practice. We solicited
comments on whether there is any real
need for more than two standalone plan
options per PDP sponsor.
Therefore, we proposed to amend the
Part D regulations at § 423.265 to add a
revised subsection (b)(3), which would
state that ‘‘CMS shall not accept more
than one basic bid and one enhanced
bid for a coverage year from a single
PDP sponsor in the same PDP region.’’
We would adopt this provision under
our authority at section 1860D–11(d) of
the Act. In instances where a parent
organization owns a controlling interest
in more than one subsidiary that
operates as a PDP sponsor in a single
PDP region, we would apply subsection
(b)(3) at the parent organization level.
That is, in the same way that we
currently apply the meaningful
differences test, a parent organization
with two subsidiary PDP sponsors could
offer no more than one plan under each
sponsors’ contract.
In addition to proposing to limit PDP
sponsors to submitting one basic and
one enhanced bid per coverage year, we
also stated that we were considering
several regulatory proposals for limiting
the type of coverage offered in those two
plans to reduce or eliminate the risk
segmentation described previously. We
believe that risk segmentation is not
consistent with the policy goal, based
on our interpretation of current law, of
obtaining the best value for the
government and the taxpayer. We
believe the Congress intended sponsors
to compete in the Part D market by
offering their best bids for basic plans,
in order to attract the greatest
enrollment through the lowest
premiums, and that this competition
would maintain downward pressure on
Part D bids and government subsidies.
We do not believe that the Congress
intended that instead sponsors would
offer their best bids for a segment of the
market that represents individuals who
are low utilizers of prescription drugs
due to better health and who can afford
unsubsidized supplemental premiums
due to better socioeconomic status.
When many healthy individuals are not
included in the basic plans, the cost of
the basic plans is increased, and this in
turn increases low-income premium
subsidies. Therefore, permitting risk
segmentation does not generate the best
value for the Part D program as a whole.
To reduce or eliminate risk
segmentation, we stated that we were
considering three options, including a
proposal, based on a reinterpretation of
section 1860D–11(b) and (c) of the Act,
that enhanced alternative coverage be
redefined to consist of supplemental
coverage added to the sponsor’s one
basic benefits offering (for an additional
premium). This could be thought of as
basic benefits plus a supplemental
benefit rider. We solicited comments on
this approach and on our belief that this
approach would be the most effective
strategy for eliminating risk
segmentation and providing the best
value for the government and the
taxpayer. We received the following
comments and our response follows:
Comment: Several commenters
expressed support for our proposal to
limit sponsors to offering no more than
two plans per PDP region. They agree
that beneficiaries can be overwhelmed
by the number of plan choices, which
can cause them to avoid even
considering exploring during the annual
election period plan options that might
better meet their needs.
Response: We appreciate the
expressions of support for our proposal.
Comment: A number of commenters
stated their opposition to the proposal
to limit sponsors to offer no more than
two plans per PDP region. Among the
assertions made by the commenters was
that the regulatory authority already in
place will produce shortly the
improvements in bid submissions that
CMS seeks and that no further authority
is required. The commenters stated that
CMS’ application of the meaningful
differences standard to its review of bids
and its authority to non-renew plans
that do not meet minimum enrollment
standards already place effective limits
on a given sponsor’s number of plan
offerings. They stated that the continued
application of those authorities,
combined with the upcoming closure of
the coverage gap, will eventually reduce
the room for plan variation to the point
of effectively creating the two-plan limit
that CMS sought with its proposal.
Commenters also expressed opposition
to our three options for preventing risk
segmentation in plan bidding, with the
option requiring enhanced benefits to be
offered as a supplement to a sponsor’s
basic plan benefit package being
particularly disfavored. Commenters
that addressed the ‘‘supplement’’ option
stated that such a bidding structure
would result in less generous enhanced
benefits because there would be less
opportunity to spread the costs
associated with such benefits. They also
stated that such a bidding structure
would limit formulary options available
to beneficiaries because sponsors would
have to offer the same formulary for
both the sponsor’s basic benefit plan
and its enhanced/supplemental option.
Response: We appreciate the
comments. We believe that the
commenters overstate the effectiveness
of the tools already at our disposal to
prevent risk segmentation and to make
further strides in ensuring that
beneficiaries have access to an array of
plan options that represent real choice.
We have been conservative in our use of
the low enrollment non-renewal
authority as demonstrated by our
adoption of enrollment thresholds that
ensure that only the plans that attract
negligible interest from the market are
non-renewed, so few additional non-
renewals are likely to occur under this
authority in the coming years. Also, we
measure meaningful differences on a
relative basis, generally using a 95
percentile threshold to arrive at the
annual limits. As plan sponsors reduce
the additional value offered in their
benefit packages, the 95 percentile
threshold will be expected to converge
toward the value of basic plans.
Consequently, we will need to explore
alternative methodologies to ensure
meaningful differences remain among a
plan sponsor’s PDP offerings.
Nevertheless, the comments have
given us reason to conduct further
analysis of this issue and continue our
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00030 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29873
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
close observation of the developments
in the Part D market. Therefore, we are
not finalizing this proposal. It may be,
as the commenters suggest, that as the
coverage gap closes, the problems of risk
segmentation and large numbers of plan
options may solve themselves. Should
that not turn out to be the case, we may
revisit the issues of plan number limits
and changes to basic and enhanced bid
structures, keeping in mind the
comments we received in response to
this proposal. In the event that we make
this or a similar proposal again, we
would only do it as part of a new
rulemaking process, during which we
would solicit public comment once
more before deciding whether to
publish final regulations.
After consideration of the public
comments we received, we are not
finalizing our proposal to limit PDP
sponsors to offering no more than two
bids per PDP region.
14. Applicable Cost-Sharing for
Transition Supplies. Transition Process
Under Part D § 423.120(b)(3)
We established transition
requirements under § 423.120(b)(3) for
Part D sponsors to address the needs of
new Part D plan enrollees who are
transitioning from other prescription
drug coverage (Part D or otherwise), and
whose current drug therapies may not
be included on their Part D plan’s
formulary (including Part D drugs that
are on a plan’s formulary but require
prior authorization or step therapy
under the plan’s utilization management
requirements). While § 423.120(b)(3)(iii)
specifies that PDP plans must provide a
temporary fill when an enrollee requests
a fill of a non-formulary drug during the
transition time period (including Part D
drugs that are on a plan’s formulary but
require prior authorization or step
therapy under a plan’s utilization
management rules), it does not currently
specify the cost sharing that should
apply to such fills. Current guidance (at
§ 30.4.9 of Chapter 6 of the Medicare
Drug Benefit Manual, found at http://
www.cms.gov/Medicare/Prescription-
Drug-Coverage/
PrescriptionDrugCovContra/Downloads/
Chapter6.pdf) states that a Part D
sponsor may charge cost sharing for a
temporary supply of drugs provided
under its transition process. Further,
cost sharing for transition supplies for
low-income subsidy (LIS) eligible
beneficiaries cannot exceed the
statutory maximum copayment
amounts. However, for non-LIS
enrollees, we stated that a sponsor must
charge cost sharing based on one of its
approved drug cost sharing tiers (if the
sponsor has a tiered benefit design), and
this cost sharing must be consistent
with cost sharing that the sponsor
would charge for non-formulary drugs
approved under a coverage exception.
This guidance created a great deal of
confusion on the part of sponsors and
beneficiaries because it can result in
beneficiaries paying different cost
sharing for formulary drugs subject to
utilization management edits (such as
prior authorization or step therapy)
during transition than specified in their
tiered benefit design. It is possible that
beneficiaries may pay more during
transition than for his or her drug’s
normal designated formulary tier.
Conversely, it is also possible that the
beneficiary may pay more once the
utilization management edit had been
satisfied than he or she did under the
transition fill.
We believe that more consistent
treatment of formulary and non-
formulary drugs, respectively, will
simplify the benefit and reduce sponsor
and beneficiary confusion.
Consequently, we proposed to add a
paragraph at § 423.120(b)(3)(vi)
clarifying that when providing a
transition supply, the cost sharing is
determined as follows: A Part D sponsor
must charge cost sharing for a temporary
supply of drugs provided under its
transition process such that the
following conditions are met:
• For low-income subsidy (LIS)
enrollees, a sponsor must not charge
higher cost sharing for transition
supplies than the statutory maximum
copayment amounts.
• For non-LIS enrollees, a sponsor
must charge—
++ The same cost sharing for non-
formulary Part D drugs provided during
the transition that would apply for non-
formulary drugs approved through a
formulary exception in accordance with
§ 423.578(b); and
++ The same cost sharing for
formulary drugs subject to utilization
management edits provided during the
transition that would apply once the
utilization management criteria are met.
Comment: We received numerous
comments that this clarification in
regulation will simplify the rules for
transition policy and reduce beneficiary
confusion.
Response: We agree with these
commenters that this provision will
simplify the rules for transition cost
sharing and reduce beneficiary
confusion. We believe this requirement
will help ensure more consistent
treatment of transition cost sharing for
formulary and non-formulary drugs
across all Part D plans and removes any
ambiguity that Part D sponsors may
have had with respect to transition cost
sharing for formulary drugs that would
otherwise be subject to utilization
management edits.
Comment: One commenter wrote that
this requirement will further complicate
an already complex policy surrounding
transition fills.
Response: We disagree with this
commenter. This provision removes the
ambiguity surrounding the allowable
cost sharing when utilization
management edits are overridden during
transition for formulary drugs, and
ensures that beneficiaries will pay the
same cost sharing for such formulary
drugs during transition and after
transition if the utilization management
criteria are met. There has been a great
deal of confusion from both sponsors
and beneficiaries with respect to the
proper cost sharing that should apply in
these situations during transition and
both we and many commenters believe
this provision provides the necessary
clarification.
In light of the overwhelmingly
positive comments on this proposal, we
are finalizing this provision without
modification.
15. Interpreting the Non-Interference
Provision (§ 423.10)
Since the MMA created the Part D
benefit in 2003, we have never formally
interpreted section 1860D–11(i) of the
Act, which is known as the
noninterference provision. In practice
we have generally invoked the spirit of
this provision in declining to intervene
in negotiations or disputes involving
payment-related contractual terms
between participants in the drug
distribution channel. However, it is
increasingly clear from the many
questions that continue to arise when
working with stakeholders on matters
ranging from lawsuits to policy
clearance to complaint resolution that
the agency and all Part D stakeholders
would benefit from a clear, formal
interpretation of these limits on our
authority. Some stakeholders appear to
believe the prohibition on interference
in negotiations extends far beyond the
boundaries that we consider relevant,
while others insist our authority extends
into arbitrating matters that seem to us
to clearly fall within the intended
prohibition. Therefore, we proposed an
interpretation through rulemaking in
order to clarify and codify the extent of
these limits on our authority.
The noninterference provision at
section 1860D–11(i) of the Act provides
that, ‘‘In order to promote competition
under this part and in carrying out this
part, the Secretary: (1) May not interfere
with the negotiations between drug
manufacturers and pharmacies and PDP
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00031 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29874
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
sponsors; and (2) may not require a
particular formulary or institute a price
structure for the reimbursement of
covered Part D drugs.’’ In beginning
with the words ‘‘In order to promote
competition under this part and in
carrying out this part. . .’’ we believe
that the Congress intended that the
activities addressed in the rest of the
provision should take place through
private market competition. We
interpret this to mean two separate but
related goals. The first goal is that the
Secretary through CMS should promote
private market competition in the
selection of Part D drugs for Part D
sponsor formularies. The second goal is
that CMS should not create any policies
that would be expected to interfere with
competitive market negotiations leading
to the selection of drug products to be
covered under Part D formularies.
Therefore, in light of these two goals we
believe there is both a duty to act—to
promote competition in the private
market for Part D drugs—and a duty to
refrain from acting—to avoid
intervention in private market
negotiations that take place in the
context of that competitive market.
Consequently, as an initial matter, in
light of our interpretation of the general
purpose of section 1860D–11(i) of the
Act, we proposed a general rule at
§ 423.10(a) that CMS promotes fair
private market competition in the
market for Part D drugs.
There is also a duty to avoid
intervention in private market
negotiations that take place in the
context of that competitive market. We
believe the intent of 1860D–11(i) is to
ensure that we do not create any
policies or become a participant in any
discussions that could be expected to
interfere with negotiations leading to
the selection of drug products to be
covered under Part D formularies. By
this we mean selection by Part D
sponsors (or other intermediary
contracting organizations) of specific
manufacturers’ products for inclusion
on formularies, formulary tier
placement, and negotiations of
acquisition costs, rebates, and any other
price concessions. We believe this
interpretation is consistent with a
textual reading of 1860D–11(i) and with
how private market transactions
determine which prescription drug
products are covered under Part D
plans. We outlined aspects of the
complex process of private market
competition for prescription drugs
described in detail elsewhere (such as in
the 2007 CBO report entitled
‘‘Prescription Drug Pricing in the Private
Sector’’ at: http://www.cbo.gov/
publication/18275) to support our
reading of the distinctly different types
of negotiations between the three parties
in ‘‘between drug manufacturers and
pharmacies and PDP sponsors’’. These
distinct sets of negotiations in the
private market between manufacturers
and pharmacies on the one hand, and
between manufacturers and plan
sponsors on the other hand, support our
textual reading of section 1860D–
11(i)(1) of the Act to prohibit CMS
involvement in negotiations between
manufacturers and pharmacies, and
between manufacturers and plan
sponsors. There are also separate price
negotiations between plan sponsors (or
their intermediary contracting
organizations) and pharmacies around
the negotiated prices required for
network participation. However, since
the statute establishes numerous
requirements that CMS must regulate
concerning access to network
pharmacies and negotiated prices, we
believe that a CMS role in negotiations
between plan sponsors and pharmacies
is not prohibited under section 1860D–
11(i)(1) of the Act, but rather under
section1860D–11(i)(2), as discussed in
this section.
Section 1860D–11(i)(1) of the Act
states that we ‘‘may not interfere with
the negotiations between drug
manufacturers and pharmacies and PDP
sponsors’’. We believe that the term
‘‘interference’’ in this context should be
interpreted as prohibiting our
involvement in discussions between
manufacturers and their distribution
channel customers (such as wholesalers
and pharmacies) or the ultimate
purchasers of prescription drugs (such
as plan sponsors and PBMs) leading to
signed contracts. We also believe
section 1860D–11(i)(1) of the Act should
be interpreted as prohibiting our
involvement in arbitration of
agreements already executed between
any of these parties. Therefore, we
interpret the prohibition in section
1860D–11(i)(1) of the Act on
interference in negotiations to pertain to
discussions either between prescription
drug manufacturers and pharmacies, or
between prescription drug
manufacturers and Part D sponsors (or
their intermediary contracting
organizations, hereafter included by
association whenever we refer to Part D
sponsors). Our interpretation is based
on the sequential phrasing of the clause
‘‘negotiations between drug
manufacturers and pharmacies and PDP
sponsors.’’ Because in general these
negotiations are not among all three
parties at once, and because
manufacturers separately contract with
pharmacies for the purchase of
inventory and with sponsors for
formulary placement, we believe the
quoted phrase can be interpreted as
recognizing these distinct types of
negotiations. Therefore, in our proposed
rule we stated that under such a
reading, the prohibition on interference
in negotiations, as described in section
1860D–11(i)(1) of the Act, would not
pertain to negotiations between Part D
sponsors and pharmacies. In hindsight,
given the strong reaction of most
commenters a better way to have
articulated CMS’ long-standing position
would have been to focus on what
‘‘interfere’’ means and to interpret it to
mean a sort of hindering or influence
beyond the implementation and
enforcement of statutory requirements.
This is the case because there are
numerous statutory provisions that
require us to directly intervene in the
contractual relationship between Part D
sponsors and network pharmacies, and
these provisions clearly signal that the
Congress expected CMS involvement in
at least some of these negotiations. The
Congress has provided many contractual
requirements for CMS to enforce
between sponsors and pharmacies; just
the drug-cost-related of these include:
interpretation of what ‘‘access to
negotiated prices’’ means, any-willing-
pharmacy standard terms and
conditions, prohibition on any
requirement to accept insurance risk,
prompt payment, and payment standard
update requirements. Consequently, we
believe that Part D sponsors and
pharmacies do not have sole discretion
to interpret these specific matters. We
would be obligated to intervene in
disputes over whether proposed or
finalized contractual arrangements
violated our rules in any area where our
oversight is directed under the statute.
So we believe it is clear that such
involvement could not be what the
Congress intended to prohibit.
Therefore, we proposed at § 423.10(b)
that CMS may not be a party to
discussions between prescription drug
manufacturers and pharmacies, or
between drug manufacturers and Part D
sponsors, and may not arbitrate the
meaning of or compliance with the
terms and conditions of agreements
reached between these parties, except as
necessary to enforce CMS requirements
applicable to those agreements. Thus,
we could only be involved in such
discussions in order to explain CMS
requirements and to ensure compliance
with Part D rules and regulations. We
also add that nothing in this prohibition
limits our authority to require
documentation of and access to all such
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00032 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29875
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
agreements, or to require the inclusion
of terms and conditions in agreements
when necessary to implement
requirements under the Act.
The first part of the section 1860D–
11(i)(2) of the Act states that CMS ‘‘may
not require a particular formulary’’. The
noninterference clause must be read in
context of the other provisions that give
CMS authority with respect to
formularies, so we proposed to interpret
the term ‘‘particular formulary’’ to mean
the selection of specific manufacturer
licensed drug products to be on
formulary, or on any particular tier of a
formulary, assuming the product meets
the definition of a Part D drug. We
believe the first part of section 1860D–
11(i)(2) of the Act would prohibit us
from developing formulary guidelines
that prefer one manufacturer’s product
over another’s in Part D formularies,
leading to more limited formularies
such as provided by the Department of
Defense and the Veteran’s
Administration. The most efficient
formularies will make formulary
selections and then exclude all or most
competing multi-source and
therapeutically equivalent brand
products in order to concentrate volume
and maximize rebates. Given the size of
the Part D market, if CMS were able to
similarly limit access to Part D
formularies to certain products, this
would bestow significant competitive
advantage on the manufacturers of
selected products and significant
competitive disadvantage on
manufacturers of competing products.
Such limits would be expected to
fundamentally alter supply and demand
in the marketplace. This prohibited sort
of formulary drug product selection
would be distinguished from CMS
formulary requirements that may
require particular types of drug entities
to be on all formularies, or on preferred
tiers, in order to provide non-
discriminatory access to drugs necessary
to treat conditions in all Medicare
beneficiaries, or to address drug classes
of clinical concern. Therefore, we
proposed a provision prohibiting
establishment of formulary drug product
selection at § 423.10(c) that would
specify that CMS does not determine the
specific drug products to be included on
Part D sponsor formularies or any tier
placement of such products, except as
required to comply with
§§ 423.120(b)(1)(v) or 423.272(b)(2).
The second part of section 1860D–
11(i)(2) of the Act states that CMS ‘‘may
not institute a price structure for the
reimbursement of covered Part D
drugs’’. Again, the noninterference
clause must be read in context of the
other provisions that give CMS
responsibilities in a number of areas
that pertain to pricing, so we stated our
view that the phrase ‘‘price structure’’
refers to establishing either absolute or
relative indices of price for Part D drugs.
Specifically, we believe the intent of
this provision is to prohibit two types of
intervention by CMS. The first
prohibited activity is that CMS may not
require Part D drug acquisition costs or
sales prices to be a function of (be
defined relative to) any particular
published or unpublished pricing
standard, either existing or future. Thus,
we could not require that Part D prices
be based on, or be any particular
mathematical function (such as a
percentage or multiple) of established
pricing standards such as Average
Wholesale Price, Wholesale Average
Cost, Average Manufacturer Price,
Average Sales Price, Federal Supply
Schedule, 340b pricing, etc. The second
prohibited activity is that CMS cannot
require price concessions (on any
standard or basis) to be at any specific
(absolute) dollar amount or equal to a
level specified in other legislative
requirements for other federal programs.
Thus, we could not, for example, set
minimum or maximum dollar prices for
a drug product or require that Part D
prices be offered at acquisition cost, or
at the ‘best price’ applicable under the
Medicaid program. However, since the
statute requires us to regulate many
aspects of how drug costs are made
available and displayed to beneficiaries
and treated in Part D bidding and
payment processes, it is clear that we
have an important role to play in
establishing rules for consistent
treatment of drug costs in the program.
Consequently, we may establish
definitions of what constitutes a pricing
standard, a price concession, a cost, etc.
We may also establish rules concerning
how drug costs are treated under Part D,
including, but not limited to, how such
amounts are disclosed in the
marketplace, projected in Part D bids,
made available to beneficiaries at point
of sale, reported in Explanation of
Benefits (EOBs), submitted to CMS, and
treated in CMS payments to Part D
sponsors. Therefore, we proposed a
provision prohibiting establishment of
drug price reimbursement
methodologies at § 423.10(d) that
specifies that CMS does not establish
drug product pricing standards or the
dollar level of price concessions at any
stage in the drug distribution channel
for Part D drugs. Nothing in our
proposed regulation would have limited
our authority to require full disclosure
or uniform treatment and reporting of
drug costs and prices.
We received numerous comments on
this proposed interpretation, both
supportive and strongly critical.
Different commenters asserted different
‘‘plain readings’’ of the statute. The
wide variation in interpretations of the
statutory prohibition evidenced in these
comments, in our view, confirms our
belief that this provision is not
consistently understood by all
stakeholders. Although the
interpretation we proposed to codify is
the same interpretation we have been
operating under in managing the Part D
program since before the beginning of
the Part D program, many commenters
perceived our proposal as a change in
interpretation. And as noted previously,
in hindsight we could have better
articulated our policy rationale than by
stating that the prohibition in section
1860D–1(i)(1) did not apply to
negotiations between sponsors and
pharmacies. These widely differing
reactions to our proposal to codify our
current interpretation lead us to
understand that additional work needs
to be done to better explain our policy,
as well as to address the concerns and
arguments advanced by numerous
commenters. Consequently, we will not
finalize the proposed regulatory
provision at § 423.10 in this final rule,
and do not intend to codify this
provision without issuing an additional
future notice of proposed rulemaking.
Comment: Some commenters
supported our interpretation and
regulatory proposal; others supported
the interpretation but did not believe
there was any need to codify our
interpretation in regulation. One
commenter supported our intent to
clarify and specify the limits of our
authority, but was very concerned about
the proposed exceptions to the
limitations on our authority and
requested greater specificity around the
particular CMS requirements that would
invoke the exceptions.
Response: We appreciate the
supportive comments, and can
understand the desire for greater
specificity in some areas.
Comment: Several commenters stated
that our interpretation violated the plain
reading of the statute, and then offered
differing interpretations of the plan
meaning of the statute. In particular,
many commenters asserted that the
phrase ‘‘between drug manufacturers
and pharmacies and PDP sponsors’’
essentially had the plain meaning of
prohibiting any and all negotiations
between any two of the parties. Other
commenters agreed with our
interpretation and that it represented
the plain meaning.
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00033 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29876
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
Response: These differing
interpretations of the statute confirm
our belief that the statutory language is
not universally understood in the same
way by all parties and would ultimately
benefit from formal interpretation and
codification in regulation.
Comment: Numerous commenters
understood us to be proposing that we
could now interfere in negotiations
between Part D sponsors and
pharmacies that we had previously
avoided.
Response: We intended to explain
how we could reconcile the distinct sets
of negotiations in the private market
between manufacturers and pharmacies,
between manufacturers and plan
sponsors, and between plan sponsors
and pharmacies with both the non-
interference provision and within the
context of the rest of the statute. Since
the statute establishes numerous
requirements that CMS must implement
concerning access to network
pharmacies and negotiated prices, we
sought to make that distinction in the
proposed rule by proposing that a CMS
role in negotiations between plan
sponsors and pharmacies is not
prohibited under section 1860D–11(i)(1)
of the Act, but rather under section
1860D–11(i)(2) of the Act. The strong
reaction of many commenters to this
interpretation has persuaded us that a
better way to have articulated this
distinction would have been to focus on
what ‘‘interfere’’ means and to interpret
it to mean a sort of hindering or
influence beyond the implementation
and enforcement of statutory
requirements. The Congress has
provided many contractual
requirements for CMS to enforce
between sponsors and pharmacies, and
we would be obligated to intervene in
disputes over whether proposed or
finalized contractual arrangements
violated our rules in any area where our
oversight is directed under the statute.
In other words, we sought to explain
that we could not involve ourselves in
negotiations between plan sponsors and
pharmacies except as necessary to fulfill
our requirements established under the
statute. From the many comments we
received on this issue, we conclude that
our explanation on this point in the
proposed rule conveyed the wrong
impression.
Comment: Numerous commenters
characterized our proposal as a change
in policy. These commenters frequently
cited examples of our previous
invocation of the prohibition on
interference in private market
negotiations as evidence of this alleged
change. For instance, commenters cited
a CMS response to a 2008 OIG report in
which CMS did not concur with several
OIG recommendations on the basis that
to do so would violate the non-
interference clause. This report,
‘‘Review of Medicare Part D Contracting
for Contract Year 2006’’ (A–06–07–
00082) is available at: http://
oig.hhs.gov/reports-and-publications/
archives/oas/cms_archive.asp.
Response: The interpretation put forth
in our proposed rule was intended to
represent the interpretation that the Part
D program has been operating under
since before the beginning of the Part D
program. We believe the examples cited
by commenters can all be traced back to
specific situations and topics that are
consistent with our proposal. For
instance, in the case of the 2008 OIG
report, the specific recommendations
with which CMS did not concur on the
basis of interference were
recommendations that violated that
provision in exactly the way we
proposed to prohibit in our proposed
rule. Specifically, we disagreed with
requiring Part D sponsors to disclose to
pharmacies the data source, basis, and
methodology used to develop
reimbursement rates, or to reveal to
pharmacies criteria for receiving higher
reimbursement rates available to certain
categories of pharmacies, and with CMS
determining whether reimbursement
rates for extended days’ supplies are
adequate. In other words, we disagreed
with CMS becoming a party to
discussions between Part D sponsors
and pharmacies on price structures or
the arbiter of the adequacy of
reimbursement methodologies. Thus, in
our view, our responses to the OIG
report were entirely consistent with our
proposed regulation. (We note that
section III.A.17 of this final rule
addresses changes to the prescription
drug pricing standard requirements
established under MIPPA, but still does
not require disclosure of data source,
basis, and methodology used to develop
reimbursement rates.) We believe that
the perception of a change in
interpretation arises from both the lack
of a common understanding of the
statutory prohibition, and from the
absence of any discussion of how our
previous statements on the record on
this topic do or do not conform to our
proposals. The numerous examples
provided by commenters will be very
helpful in developing such an
explanation in any future rulemaking on
this policy.
16. Pharmacy Price Concessions in
Negotiated Prices (§ 423.100)
We have learned that some Part D
sponsors have been reporting costs and
price concessions to CMS in different
ways. This reporting differential matters
because this variation in the treatment
of costs and price concessions affects
beneficiary cost sharing, CMS payments
to plans, federal reinsurance and low-
income cost-sharing (LICS) subsidies,
and manufacturer coverage gap discount
payments. Differential treatment of costs
would also be expected to affect plan
bids. If the projected net costs a sponsor
is liable for in its bid are understated
because the sponsor has been reporting
certain types of price concessions as
direct or indirect remuneration (DIR)
rather than as price concessions that
affect the negotiated price, it follows
that the sponsor may be able to offer a
lower bid than its competitors and may
achieve a competitive advantage
stemming not from greater efficiency,
but rather from a technical difference in
how costs are reported to CMS. When
this happens, such differential reporting
could result in bids that are no longer
comparable, and in premiums that are
no longer valid indicators of relative
plan efficiency. Therefore, we proposed
changes to rectify this concern.
Negotiated prices are the payment
amounts pharmacies receive from plans
for covered Part D drugs dispensed to
plan enrollees. CMS payments to plans
are based on the reporting of negotiated
prices (through PDE reporting) that are
actually paid and are then offset by any
other price concessions (submitted in
aggregate through the separate annual
DIR reporting process). CMS establishes
rules for cost and price concession
reporting through both PDE and DIR
guidance and other payment
reconciliation rules, and has regulated
the definition of negotiated price and
how it is to be treated in Part D benefit
administration and in payment
reconciliation. Since 2010, the
regulatory definition at § 423.100 has
been: ‘‘Negotiated prices means prices
for covered Part D drugs that: (1) The
Part D sponsor (or other intermediary
contracting organization) and the
network dispensing pharmacy or other
network dispensing provider have
negotiated as the amount such network
entity will receive, in total, for a
particular drug; (2) Are reduced by
those discounts, direct or indirect
subsidies, rebates, other price
concessions, and DIR that the Part D
sponsor has elected to pass through to
Part D enrollees at the point of sale; and
(3) Include any dispensing fees.’’
We intended clause 2 to primarily
refer to price concessions from parties
other than pharmacies, since these
would be price concessions that were
not based on the sale of the drug by the
pharmacy and calculated when the
claim adjudicated and, in fact, could not
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00034 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29877
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
be calculated until a later date. In
particular, we expected these other non-
claim-based price concessions to be in
the form of rebates offered by
prescription drug manufacturers. Since
prescription drugs are dispensed by
pharmacies and purchased through
transactions between Part D sponsors (or
their intermediary contracting
organizations) and pharmacies,
manufacturers are never in a position to
apply price concessions to negotiated
prices at point of sale. We now
understand that clause 2 is ambiguous
and permits sponsors and their
intermediaries to elect to take some
price concessions from pharmacies in
forms other than the negotiated price
and report them outside the PDE. When
this occurs, the increased negotiated
prices generally shift costs to the
beneficiary, the government and
taxpayer, and when applicable to
certain brand name drugs, to
prescription drug manufacturers. (The
mechanism of this sort of cost shift was
discussed at length in the analogous
context of lock-in pricing in our 2008
proposed rule entitled ‘‘Medicare
Program; Revisions to the Medicare
Advantage and Prescription Drug
Benefit Programs’’ which was published
on May 16, 2008 in the Federal
Register, 73 FR 28563 through 28566.)
In addition, when price concessions
from pharmacies are reflected in forms
other than the negotiated price, the
degree of price concession that the
pharmacy has agreed to is no longer
reflected in the negotiated prices
available at point of sale or reflected on
the Medicare Prescription Drug Plan
Finder (Plan Finder) tool. Thus, the true
price of drugs at individual pharmacies
is no longer transparent to the market.
Consequently, consumers cannot
efficiently minimize both their costs
(cost sharing) and costs to the taxpayers
by seeking and finding the lowest-cost
drug/pharmacy combination. Moreover,
as the coverage gap closes, there are
fewer and fewer beneficiaries who are
exposed to the full cost of drug
products, either at the point of sale or
as reflected in Plan Finder estimates.
When this occurs, the basis of
competition shifts from prices to cost
sharing, and the pricing signals
available to the market can be distorted
when lower cost sharing is not aligned
with lower prices. Thus, we believe the
exclusion of pharmacy price
concessions from the negotiated price
thwarts the very price competition that
the Congress intended with respect to
how private plans would compete with
other plans on both premiums and
negotiated prices.
We are aware that certain pharmacy
price concessions are being excluded
from the determination of the negotiated
price because they are being
characterized as ‘‘network access fees’’,
‘‘administrative fees,’’ ‘‘technical fees’’
or ‘‘service fees’’ that are frequently
imposed through PBM-issued manuals
rather than explicit contractual terms.
Pharmacies and pharmacy organizations
report that they do not receive anything
of value for those fees other than the
ability to participate in the Part D
network. The itemized types of services
for which their payments are offset
reportedly include things such as
transaction fees for submission of
claims, help desk support, information
technology and telecommunication
systems connectivity, electronic funds
transfers, and other expenses associated
with credentialing, maintaining, and
auditing pharmacy networks. These fees
take the form of deductions from
payments to pharmacies for drugs
dispensed, but in our view clearly
represent charges that offset sponsor/
PBM operating costs. We believe that if
the sponsor or its intermediary
contracting organization wishes to be
compensated for these services and have
those costs treated as administrative
costs, such costs should be accounted
for in the administrative costs of the
Part D bid. If instead these costs are
deducted from payments made to
pharmacies for purchases of Part D
drugs, such costs are price concessions
and must be treated as such in Part D
cost reporting. This is the case
regardless of whether the deductions are
calculated on a per-claim basis or not.
In our view, the decision on how such
network management costs are funded
between the PBM and the sponsor is not
governed by our rules, but our rules do
require that price concessions be fully
disclosed and net against drug costs in
reconciliation. We have also heard from
pharmacies that some sponsors apply
dispensing fees to claims when they are
adjudicated at point of sale, but require
that these fees later be rebated back to
the sponsor and deducted from payment
remittances. Such practices again
misstate the negotiated price. Our
proposal would require that dispensing
fees could only be applied at point of
sale if they are received and retained by
the pharmacy in the negotiated price.
Some stakeholders have
recommended that certain incentive
payments to pharmacies, such as
generic dispensing incentive fees,
should not be included in negotiated
prices. If these payments are included,
they explain, the negotiated prices
appear higher at the more efficient
pharmacy as the result of the additional
incentive payment. This higher price
then proportionally increases costs
borne by beneficiaries, the government,
and manufacturers. These incentives
really represent amounts that the
sponsor is willing to bear in order to
encourage the most efficient drug
choices, which will drive down total
costs overall, and thus the sponsor is
willing to bear a disproportionate share
of such expense. We agree with this
argument and we believe that this sort
of arrangement would not conflict with
our proposed requirement that all price
concessions be reflected in the
negotiated price since such additional
payments are the opposite of price
concessions. Instead such incentive fees
represent contingent price increases that
cannot be predicted in advance.
Therefore, they cannot be programmed
to be applied at point of sale or reflected
in the price posted on Plan Finder. We
believe it would be appropriate to treat
this particular sort of price increase
differently than price decreases because
including such amounts in the
negotiated price (incentive fee
component) at point of sale could
disguise the relative competitiveness of
the underlying pharmacy prices.
Incentive fees also primarily benefit the
plan sponsor who benefits from the
lower costs associated with the
incentivized behavior, rather than the
beneficiary. Therefore, in this case, we
agree that it would be more appropriate
for such incentive payments to be
excluded from the negotiated price, and
reported later in reconciliation as
negative DIR. When reported as negative
DIR, these amounts disproportionately
affect (increase) the amounts the
sponsor is liable for in risk sharing,
which is appropriate given the intent of
the incentives to promote least-cost drug
product selection at point of sale. Least-
cost drug product selection will directly
reduce the sponsor’s allowable risk
corridor costs, so any incentive paid to
encourage this behavior would be
expected to be more than offset by the
ingredient costs savings achieved
through avoidance of higher-cost drug
selection. This is so because, as we
learned from numerous commenters to
the 2014 draft Call Letter, the incentive
payments are generally in the range of
a dollar or two and the difference
between preferred and non-preferred
drug products is generally much greater.
Therefore, we proposed to revise the
definition of negotiated prices at
§ 423.100 to require that all price
concessions from pharmacies are
reflected in these prices. Specifically we
proposed to redefine negotiated prices
to mean prices for covered Part D drugs
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00035 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29878
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
that: (1) The Part D sponsor (or other
intermediary contracting organization)
and the network dispensing pharmacy
or other network dispensing provider
have negotiated as the amount such
network entity will receive, in total, for
a particular drug; and (2) are inclusive
of all price concessions and any other
fees charged to network pharmacies;
and (3) include any dispensing fees; but
(4) exclude additional contingent
amounts, such as incentive fees, only if
these amounts increase prices and
cannot be predicted in advance; and (5)
may not be rebated back to the Part D
sponsor (or other intermediary
contracting organization) in whole or in
part.
We received the following comments
on our proposed revisions and our
responses follow.
Comment: We received a significant
number of comments in support of this
provision based on the improved
transparency of pharmacy price
concessions. One commenter stated the
belief that PDPs and their contracted
PBMs are circumventing the Medicare
Modernization Act by hiding pharmacy
charge backs as overall administrative
surcharges. These commenters stated
that amounts charged to pharmacies in
the form of ‘‘administrative fees,’’
‘‘network access fees’’ or rebates of
dispensing fees appeared to be vehicles
for price concessions. Another
commenter believed that the proposed
provision would alleviate the
complexity of tracking actual drug
reimbursement and help ensure that
reimbursement structures are not
actually increasing Medicare costs.
Several commenters stated that
inclusion of accurate costs in the Plan
Finder tool would be of benefit to
consumers, and added that drug prices
must be accurate and transparent to
help seniors compare plan costs.
We also received some comments in
opposition to the proposed provision.
These commenters stated that some
price concessions that benefit the Part D
program do not lend themselves to
inclusion in negotiated prices. A few
commenters stated that savings from
lower point-of-sale prices would be
reflected in higher enrollee premiums
and increased premium subsidies. Other
commenters stated that payments
received from pharmacies to PBMs were
for services provided and should not be
considered price concessions. One
commenter stated that just because
pharmacies pay for and benefit from
services from PBMs does not necessarily
make the fees price concessions. A few
commenters opposed the provision on
the grounds that it would place new
limitations on the terms sponsors will
be able to negotiate with network
pharmacies and stated that CMS is
limiting the tools available to sponsors
to offer varied incentive-based
agreements such as providing additional
compensation for increased dispensing
of generic medicines or superior
customer service. Other commenters
thought that Part D sponsors and PBMs
should be able to retain the flexibility to
determine which concessions to pass
through to beneficiaries through drug
prices or lower premiums. To bolster
this argument one commenter quoted
from our 2009 rule in which we stated
that the statute says prices will ‘‘take
into account’’ price concessions not
include them all, and that a ‘‘plain
reading of this demonstrates the
Congress’ intent to be permissive of Part
D sponsors to choose how much of their
negotiated price concessions to pass
through to Part D beneficiaries at the
point of sale’’.
One of the commenters who opposed
the provision suggested that, as an
alternative, CMS use its existing
authority to require plans to disclose
both in the bid pricing tool (BPT) and
through DIR, specific line-item reporting
of performance-based DIR received from
network pharmacies. Several
commenters urged CMS to use its
existing DIR reporting authority to
capture price concessions attributable to
risk-based performance measures,
which often require retrospective
performance review and therefore
cannot be captured in negotiated prices.
The commenters argued that the DIR
process must be used to allow sponsors
to maintain innovative payment
arrangements that yield efficient and
quality pharmacy networks. One of
these commenters voiced support for ‘‘a
competitive and level playing field for
all sponsors’’ and urged CMS to create
clear and comprehensive regulatory
guidance with respect to pharmacy
price concessions.
Response: We appreciate the detailed
comments we received in response to
our proposal. We continue to believe it
is critical that negotiated prices reported
on PDEs have a consistent meaning
across the Part D program in order to
preserve a level playing field in bidding
and cost reporting. As we stated in the
proposed rule, we intended clause 2 of
the existing definition of negotiated
price to primarily refer to price
concessions from parties other than
pharmacies, since these would be price
concessions that were not based on the
sale of the drug by the pharmacy and
calculated when the claim adjudicated
and, in fact, could not be calculated
until a later date. Our proposal to
require all pharmacy price concessions
be included in the negotiated price
would ensure that negotiated prices
have a consistent meaning, provide for
increased transparency in cost reporting
to CMS, and allow for meaningful price
comparisons between Part D sponsors.
While we recognize that some
pharmacy price concessions are
contingent upon risk or incentive based
arrangements, we provided an
illustration of how such price
concessions could adjust future
negotiated prices, rather than adjusting
the current quarter’s prices downward
through DIR reporting. Consequently,
we did not believe that our proposal
would limit Part D sponsors’ ability to
enter into such contracting relationships
with their network pharmacies. We did
not propose placing additional
restrictions around such arrangements,
only that their resulting costs must be
transparent to all concerned.
Nevertheless, we are persuaded by the
comments that there may be some price
concessions from pharmacies that are
based upon contingencies that cannot be
known at the point-of-sale and that
these price concessions should be
distinguished from all other pharmacy
price concessions and continue to be
reported as direct or indirect
remuneration. This would be also be
consistent with the commenter who
pointed out the statutory language that
negotiated prices will ‘‘take into
account’’ price concessions. While we
had proposed including all price
concessions from pharmacies in the
negotiated price to provide maximum
price transparency, we believe that there
is room for further discussion with
industry to determine whether there are
specific types of arrangements that do
not lend themselves to accurate
inclusion in the negotiated prices. As
long as all types of price concessions are
consistently ‘‘taken into account’’ in the
same way by each sponsor in preparing
bids and reporting costs, bids and point-
of-sale negotiated prices can remain
comparable. Therefore, in response to
comments we are revising our proposed
definition of negotiated price to allow a
narrow exception to the requirement
that all pharmacy price concession be
included in the negotiated price for
those contingent pharmacy price
concessions that cannot reasonably be
determined at the point-of-sale. We
intend to identify in our DIR reporting
guidance which types of price
concessions from pharmacies would
meet the standard for this exception,
and we intend to consult with industry
in developing our guidance in this area.
Any contingent pharmacy price
concessions or incentive payments that
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00036 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29879
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
can be determined at the point-of-sale
must be included in negotiated prices.
We agree with the commenter who
pointed out that not all fees that
pharmacies pay to PBMs are price
concessions. But as discussed in the
NPRM, when such fees take the form of
deductions from payments to
pharmacies for Part D drugs dispensed,
such costs are price concessions and
must be treated as such in Part D cost
reporting. This is the case regardless of
whether the deductions are calculated
on a per-claim basis or not. Standard
treatment of all price concessions will
bring improved transparency to
pharmacy payments. We disagree that
this change is inconsistent with the
MMA because the MMA established
Medicare Part D as a voluntary, private-
market-based program what would rely
on private plans to provide coverage
and to bear some of the financial risk for
drug costs. These private plans would
determine premiums through a bid
process and would compete with other
plans based on premiums and
negotiated prices. While Part D sponsors
may lose some flexibility in deciding
how much of the price concessions
should be applied to beneficiaries at the
point of sale or through reduced
premium, consistency in how specific
types of price concessions are ‘‘taken
into account’’ in negotiated prices is
necessary in order to preserve reliance
on market competition between plans,
which is a cornerstone of the Medicare
Part D program.
Comment: A few commenters
questioned CMS’ authority to
implement the proposed change and
some asserted that the non-interference
provision prohibits CMS from defining
negotiated prices.
Response: We disagree with these
comments. We have the authority to
interpret the provisions of section
1860D–2(d)(1)(B) and believe our
interpretation is appropriate. We also
have a history of regulating on cost and
price concession reporting. We
established detailed guidance for
accurate and consistent cost and price
concession reporting through both PDE
and DIR guidance and other payment
reconciliation rules, and have twice
before regulated the definition of
negotiated price and how it is to be
treated in Part D benefit administration
and in payment reconciliation. In the
original Part D rule, negotiated prices
were mainly defined as ‘‘prices for
covered Part D drugs that were available
to beneficiaries at the point of sale at
network pharmacies’’. This definition
permitted sponsors or their
intermediaries to include PBM spread in
the price. Therefore, on January 12,
2009 we published in the Federal
Register the final rule with comment
entitled, ’’ Medicare Advantage and
Prescription Drug Benefit Programs:
Negotiated Pricing and Remaining
Revisions’’ (74 FR 4131), to clarify that
negotiated prices must be the amounts
actually received by the pharmacy for
the drug. We are now once again
revising the definition.
Comment: Several commenters
addressed the effective date of the
proposed rule. Commenters advocated
for a prospective implementation only,
or expressed the hope that the rule
could be delayed until 2016. They
stated that time was needed to allow
collaboration with the industry, enable
CMS to capture the changes in detailed
guidance, and give Part D sponsors time
to revise their pharmacy network
contracts.
Response: In response to these
comments we are postponing
implementation of this provision until
the 2016 contract year and will use this
time to work with the industry to
develop guidance on when the
exception previously described applies.
After considering comments received,
we are finalizing the provision as
proposed with modification to require
that negotiated prices be inclusive of all
price concessions from network
pharmacies except contingent price
concessions that cannot reasonably be
determined at the point-of-sale. We also
modified the language in paragraph (4)
by clarifying that additional contingent
amounts, such as incentive fees, that
increase prices are always excluded
from the negotiated price by removing
the word ‘‘may,’’ and we also replaced
‘‘cannot be predicted in advance’’ with
‘‘cannot reasonably be determined at the
point-of-sale’’ to parallel paragraph (2).
Finally, we have modified the effective
date of this provision to 2016 to avoid
disruption of the existing regulation
which will be applicable for the rest of
2014 and 2015.
17. Preferred Cost Sharing (§§ 423.100
and 423.120)
In our original rule implementing the
Part D Program, we codified an
interpretation of section 1860D–
4(b)(1)(B) of the Act at § 423.120(a)(9)
that permitted Part D sponsors to offer
lower cost sharing at a subset of network
pharmacies, dubbed ‘‘preferred
pharmacies,’’ than at other in-network
pharmacies. This lower cost sharing was
subject to certain conditions that
seemed straightforward to us at the
time, but which have proven to need
clarification. We have recently
discussed this concern in the
Announcement of Calendar Year (CY)
2014 Medicare Advantage Capitation
Rates and Medicare Advantage and Part
D Payment Policies and Final Call Letter
(2014 Call Letter) on pages 175 and 176
[at http://www.cms.gov/Medicare/
Health-Plans/
MedicareAdvtgSpecRateStats/
Downloads/Announcement2014.pdf].
Section 1860D–4(b)(1)(B) of the Act
contemplates the possibility of sponsors
offering lower cost sharing at some
network pharmacies than is offered in
conjunction with the any willing
pharmacy terms and conditions
mandated in the immediately preceding
paragraph (A). However, a plan’s ability
to reduce cost sharing is contingent
upon one condition: ‘‘In no case shall
such a reduction result in an increase in
payments made by the Secretary under
section 1860D–15 of the Act to a plan.’’
In our original proposed rule entitled
‘‘Medicare Program; Medicare
Prescription Drug Benefit; Proposed
Rule,’’ published on August 3, 2004 in
the Federal Register, 69 FR 46658
through 46659, we did not offer an
interpretation of this language but
implied that any assessment of whether
the condition was met would be a
matter of actuarial equivalence analysis.
We proposed to codify the requirements
in regulation with the following two
conditions: ‘‘. . . the plan must still
meet the requirements under
§ 423.104(e)(2) and (5); and [a]ny cost
sharing reduction must not increase
CMS payments under § 423.329.’’ In the
final regulation entitled ‘‘Medicare
Program; Medicare Prescription Drug
Benefit; Final Rule’’, published on
January 28, 2005 in the Federal
Register, 70 FR 4247 through 4255, we
reiterated the language from the
aforementioned proposed rule (69 FR
46658). ‘‘However, we note that while
these within-network distinctions are
allowed, the statute also requires that
such tiered cost-sharing arrangements in
no way increase our payments to Part D
sponsors. Therefore, tiered cost-sharing
arrangements based on within-network
distinctions could be included in Part D
plans’ benefits subject to the same
actuarial tests that apply to formulary-
based tiered cost-sharing structures.
Thus, a reduction in cost sharing for
preferred pharmacies in a Part D plan
network could be offered through higher
cost sharing for non-preferred
pharmacies (or as alternative
prescription drug coverage).’’ (70 FR
4254, January 28, 2005.) This statement
was immediately followed by an
expression of our intent to ensure that
such network benefit designs were non-
discriminatory: ‘‘We recognize the
possibility that Part D plans could
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00037 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29880
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
effectively limit access in portions of
their service areas by using the
flexibility provided in § 423.120(a)(9) of
our final rule to create a within-network
subset of preferred pharmacies. In other
words, in designing its network, a Part
D plan could establish a differential
between cost-sharing at preferred versus
non-preferred pharmacies—while still
meeting the access standards in
§ 423.120(a)(1) of our final rule—that is
so significant as to discourage enrollees
in certain areas (rural areas or inner
cities, for example) from enrolling in
that Part D plan. We emphasize that
such a network design has the potential
to substantially discourage enrollment
by certain Part D enrollees, and that we
have the authority under section
1860D–11(e)(2)(D) of the Act to disallow
benefit designs that are discriminatory.’’
However, what we failed to
sufficiently explain in 2005 was that if
cost sharing cannot rise beyond a
certain level, then in return for lower
cost sharing, preferred networks must
reduce drug costs paid by the plan in
order to prevent an increase in CMS
payments to the plan. In part this
omission may have been because we
presumed that Part D sponsors would
motivate enrollees to go to a subset of
pharmacies through lower cost sharing
only if those pharmacies offered
significantly lower negotiated prices,
and thus would provide a competitive
advantage for the sponsor in lowering
costs. As the concerns expressed in the
2014 Call Letter indicate, this does not
seem to have been the case for some
sponsors. However, if drug costs
(negotiated prices) are not lower in
return for lower cost sharing, and the
lower cost sharing cannot be completely
offset by higher cost sharing on other
beneficiaries due to our cost-sharing-
outlier limits, then the amount that
must be subsidized by the government
and the taxpayer will increase.
Therefore, we proposed to clarify that
preferred cost sharing should signal
consistently lower costs. When lower
cost sharing correctly signals the best
prices on drugs, then choosing
pharmacies on the basis of that lower
cost sharing lowers not only beneficiary
out-of-pocket costs, but also Part D plan
and other government subsidy costs.
Lower plan and government subsidies
translate into lower CMS payments to
plans, consistent with the statutory
requirements at section 1860D–
4(b)(1)(B) of the Act. Therefore, we
proposed to revise § 423.120(a)(9) to
state: ‘‘Preferred cost-sharing in network
pharmacies. A Part D sponsor offering a
Part D plan that provides coverage other
than defined standard coverage may
reduce copayments or coinsurance for
covered Part D drugs obtained through
a subset of network pharmacies, as long
as such preferred cost sharing is offered
in accordance with the requirements of
§ 423.120(a)(8) and for Part D drugs with
consistently lower negotiated prices
than the same drugs when obtained in
the rest of the pharmacy network.’’ We
proposed that by ‘consistently lower’ we
mean that sponsors must offer
beneficiaries and the Part D program
better (lower) negotiated prices on all
drugs in return for the lower cost
sharing. In practice we believe this
would mean that whatever pricing
standard is used to reimburse drugs
purchased from network pharmacies in
general, a lower pricing standard must
be applied to drugs offered at the
preferred level of cost sharing. We
welcomed comments on alternative
approaches to ensuring that the offering
of preferred cost sharing does not
increase our payments. We proposed
that any alternative methodology must
be based solely on the level of
negotiated prices and thus consistent
with our proposal to amend that
definition (section III.A.15. of this final
rule). As discussed in that section, we
proposed to revise the definition to
specify that all price concessions from
pharmacies must be reflected in the
negotiated price in order to promote
transparent price competition, as well as
to eliminate differential cost reporting
and cost shifting that interfere with a
fair and transparent competitive bidding
process. We requested that any
alternative methodology suggestions be
accompanied by specific proposals for
how we could objectively validate
compliance through data we already
collect.
In addition, we solicited comments on
whether we should also establish
standards on how much lower drug
costs should be in return for preferred
cost sharing. We are aware that there is
a wide range of savings projections
associated with the use of limited
networks. For instance, a January 2013
study prepared for the Pharmaceutical
Care Management Association (PCMA)
provides various estimates ranging from
5 percent to 18 percent [at http://
www.pcmanet.org/images/stories/
uploads/2013/visante-
pcma%20pharmacy%20networks
%20study%201-24-13%20final.pdf]. We
solicited comment on whether Medicare
should require a minimum level of
savings, such as 10 percent or 15
percent, over the costs available at retail
cost-sharing rates. We believe that
substantial discounts in this range
would be necessary to balance the
extremely low preferred cost sharing
rates offered by many sponsors in 2013.
We also solicited comments on how
broadly preferred cost sharing should be
applied to drugs on a sponsor’s
formulary. For instance, is it reasonable
to offer cost sharing as low as $0 for
only the least expensive generics on
formulary? Or should preferred cost
sharing have to apply to a minimum
percentage of formulary products to be
a meaningful benefit instead? Or should
preferred cost sharing have to apply to
all drugs available at pharmacies
offering preferred cost sharing? This
would require that the prices of all
drugs at those pharmacies could be no
higher than the prices at the other
network pharmacies. Such a policy
would prevent sponsors from offering
lower prices on drugs with preferred
cost sharing while offering higher prices
on other drugs not subject to preferred
cost sharing. Our concern is that
without such rules, it is possible that
the beneficiary is motivated to change
pharmacies in order to pay very low
copays on some drugs, but the program
may end up paying higher costs on
other drugs the beneficiary purchases at
the same pharmacy out of convenience.
We also proposed a clarification in
terminology to better describe the
application of the policy to a sponsor’s
approved Part D pharmacy network.
Specifically, we proposed to delete the
definitions of ‘‘preferred pharmacy’’ and
‘‘non-preferred pharmacy’’ from
§ 423.100 and to add a new definition of
preferred cost sharing. ‘‘Preferred cost
sharing’’ would mean lower cost sharing
for certain covered Part D drugs at
certain network pharmacies offered in
accordance with the requirements of
§ 423.120(a)(9). We would then require
that Part D sponsors would revise any
marketing materials to reflect the
revised nomenclature, and eliminate
any references to preferred or non-
preferred network pharmacies. We
solicited comment on whether any
further clarifications of terminology are
needed for this policy proposal.
We received the following comments
and our responses follow:
Comment: Many commenters strongly
supported our proposal to require
consistently lower negotiated prices at
pharmacies offering preferred cost
sharing. These commenters found it
troubling that some Part D plans’
negotiated prices were not lower for
some drugs at pharmacies offering
preferred cost sharing and stated that
the alignment of preferred cost sharing
with lower negotiated prices is
necessary to ensure that arrangements
with pharmacies to offer preferred cost
sharing do not cost the government
more and provide savings for
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00038 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29881
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
beneficiaries. The commenters assert
that the current framework is not
transparent and allows PBMs to
maximize profits by moving as much
volume as possible to their mail order
pharmacies with little, if any, savings
for the beneficiary, and even the
possibility that the beneficiary could
pay more than they would at a
pharmacy without preferred cost
sharing.
However, other commenters strongly
opposed our proposal to require
consistently lower negotiated prices at
pharmacies offering preferred cost
sharing. While no commenters dispute
that benefit designs that provide
preferred cost sharing at some network
pharmacies must not increase payment
to Part D plans, many dispute our
proposal to make this determination
based entirely upon negotiated prices.
They assert that the reference in the
statute to ‘‘an increase in payments’’
does not refer solely to negotiated prices
but must also take into consideration
the direct subsidy, reinsurance
subsidies, end of year reconciliation,
and beneficiary premiums. Several
commenters said that we do not have
the authority to implement this proposal
because it violates the section 1860D–
11(i) statutory non-interference
provision that prohibits CMS from
instituting a price structure for the
reimbursement of Part D drugs. One
commenter said that while they share
our objectives for preferred cost sharing
arrangements to lower costs for the Part
D program and beneficiaries, they
believe these arrangements can be
beneficial if the price concessions are
reflected in prices at the pharmacies
and/or used to lower premiums.
Commenters also stated that requiring
lower negotiated prices for every drug
will restrict the flexibility that Part D
sponsors need to negotiate discounts
with pharmacies, which will lead to
increased prices and beneficiary
disruption. Moreover, commenters
argue that savings from preferred cost
sharing cannot be determined at the
individual drug level because that does
not account for different drug mixes at
different pharmacies that could better be
determined by actuarially sound
aggregate methods of comparison. One
commenter recommended that we
implement a ‘‘fixed basket of drugs’’
approach similar to our Out-of-Pocket
(OOPC) tools used for determining
meaningful differences between basic
and enhanced plans. A number of
commenters also contend that such a
consistently-lower-price requirement is
unworkable because their contracts
frequently have a ‘‘lesser of’’ provision
to ensure they only pay the pharmacies’
usual & customary prices when such
prices are lower than the negotiated rate
and they would have no way to ensure
that pharmacy usual & customary prices
are never lower at pharmacies that do
not offer preferred cost sharing. Finally,
most commenters opposed CMS
establishing standards on how much
lower drug costs should be in return for
preferred cost sharing.
Response: We appreciate the
significant support we received for the
proposal and continue to believe that
the proposal would provide a
transparent mechanism for ensuring
compliance with the statutory
requirement that prohibits benefit
designs with preferred cost sharing at
certain network pharmacies from
increasing payments to plans. While we
agree that basing increased payments to
plans entirely on negotiated prices is
not the only possible interpretation of
the statutory requirement, we believe it
is a reasonable interpretation that would
allow us to uniformly apply the
statutory requirement while also
providing price transparency to
beneficiaries and maximizing price
competition.
Nevertheless, we premised this
proposal on our related proposal to
change the definition of ‘‘Part D
Negotiated Price’’ to include all
pharmacy price concessions. If we are
going to use negotiated prices as the sole
basis for determining increased
payments to plans for purposes of
section1860D–4(b)(1)(B) of the Act, then
all pharmacy price concessions must be
in the negotiated price because the price
would need to have the same meaning
at every network pharmacy.
Consequently, because we are finalizing
a different definition of negotiated price
than originally proposed, one that will
allow for the exclusion of some
pharmacy price concessions from the
negotiated price, we will not be
finalizing our proposal to require
consistently lower negotiated prices at
pharmacies offering preferred cost
sharing. Clearly if some price
concessions are not reflected in the
negotiated price, a higher negotiated
price may not result in increased
payments to plans. We also are not
finalizing an alternative requirement at
this time, in light of the comments that
suggested different approaches because
we intend to consider them further as
we determine how best to ensure, in a
transparent manner, that preferred cost
sharing does not increase payments to
plans. While we are not finalizing the
proposal, we disagree with the
commenter who stated that CMS does
not have the authority to implement
such a requirement because it is
consistent with our obligation to
implement and enforce many statutory
requirements under the Part D program
that directly or indirectly affect
negotiations between pharmacies and
Part D sponsors, in particular section
1860D–4(b)(1)(B) of the Act, and
including several other closely related
statutory provisions contained in
section 1860D–4(b)(1) of the Act. For
example, we have previously
established retail and non-retail
pharmacy network adequacy
requirements under this authority to
ensure convenient pharmacy access as
required under section 1860D–4(b)(1)(C)
of the Act.
Comment: Some commenters asserted
that our April 2013 study (‘‘Negotiated
Pricing between Preferred and Non-
Preferred Pharmacy Networks’’, posted
at: http://www.cms.gov/Medicare/
Prescription-Drug-Coverage/
PrescriptionDrugCovGenIn/.
Downloads/PharmacyNetwork.pdf)
that we cited as showing some
negotiated prices for drugs were higher
at pharmacies offering preferred cost
sharing than the rest of the network was
flawed. Therefore, they contend that our
rationale for the proposal was flawed.
They point out that this study only
looked at prescription drug event (PDE)
data and did not take into consideration
any direct or indirect remuneration.
They claim that even if you accept the
results of this study as stated, it shows
only that drug prices were ‘‘slightly
higher’’ and only in ‘‘a few’’ preferred
networks in ‘‘some plans’’. In addition,
commenters raised methodological
concerns because the CMS study was
not normalized for different drug mix
and utilization between plans, which
they said will bias the results and lead
to incorrect conclusions that will
contribute to higher costs for
beneficiaries and the Part D program.
Response: We appreciate the detailed
comments regarding the validity of our
study and the conclusions that we drew.
However, we disagree with the assertion
that our study was flawed and believe
some commenters misinterpreted our
findings. Specifically, despite the
comments, we did not conclude that our
findings showed that some pharmacies
with preferred cost sharing were more
expensive than some other pharmacies
that were not offering preferred cost
sharing. We acknowledge that this study
did not take into consideration price
concessions reported as DIR or
differences in drug mix, and therefore
agree that one cannot make that
conclusion given the current definition
of negotiated price and variability
among plans on what is included in the
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00039 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29882
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
price. Nevertheless, we believe the
findings of some higher negotiated
prices at some pharmacies offering
preferred cost sharing demonstrates that
we cannot assume point-of-sale
negotiated prices are always lower at
pharmacies offering preferred cost
sharing and, therefore cannot assume
that benefit designs with some
pharmacies offering preferred cost
sharing never increase payments to
plans. Instead, we believe our study
highlighted this vulnerability and the
need for us to propose a transparent and
consistent method for ensuring these
benefit designs do not increase
payments to plans.
Comment: Some commenters strongly
supported our proposal to remove the
definitions of preferred and non-
preferred pharmacies and replace them
with a definition of preferred cost
sharing. These commenters agreed that
the term ‘‘preferred pharmacy’’ is
confusing for beneficiaries who
sometimes interpret this to mean non-
preferred pharmacies are out-of-
network. Other commenters opposed
the proposal because they believe the
change in terminology will be confusing
for beneficiaries. They note that under
the current framework plans may
already refer to non-preferred
pharmacies as ‘‘other network
pharmacies’’ and, therefore, there is no
need for this change. Moreover, some
commenters opposed removing the term
‘‘preferred pharmacy’’ because they
believe it refers not only to lower cost
sharing but also quality of services.
Another commenter who was
supportive of the proposed change also
raised concerns about beneficiary
confusion from the change in
terminology and urged CMS to consider
education and outreach efforts to help
beneficiary understand the new
terminology and add related language to
Medicare & You.
Response: We appreciate the
comments we received on this proposal.
We agree with supporters that this
change will help avoid confusion
regarding pharmacy network status and
more accurately reflect what is meant by
preferred. While any change has the
potential to initially create some
confusion, we disagree that
substantively this change will be more
confusing to beneficiaries going
forward. In addition, we are perplexed
by the comments that said their
identification of preferred pharmacies
also takes into consideration the quality
of pharmacy services because that was
never part of the regulatory definition.
Nevertheless, we are not finalizing this
proposal because it is so closely tied to
the other preferred cost sharing proposal
to revise § 423.120(a)(9) that is not being
finalized as a result of changes to the
definition of negotiated price in this
final rule (as described in section
III.A.25 of this final rule).
After considering of the public
comments received, we are not
finalizing the proposed changes to
§§ 423.120(a)(9) and 423.100. We will
undertake notice and comment
rulemaking if we are going to make
changes to these provisions in the
future.
18. Prescription Drug Pricing Standards
and Maximum Allowable Cost
(§ 423.505(b)(21)
We proposed a change to the
regulations governing the disclosure and
updating of prescription drug pricing
standards used by Part D sponsors to
reimburse network pharmacies to make
clear that drug pricing based on
maximum allowable cost is subject to
these regulations. Section 173 of MIPPA
amended sections 1860D–12(b) and
1857(f)(3) of the Act to add a provision
requiring the regular updating of
prescription drug pricing standards.
Specifically, for plan years beginning on
or after January 1, 2009, CMS’s contracts
with Part D sponsors must include a
provision requiring sponsors to update
any standard they use to reimburse
network pharmacies based on the cost of
the drug to accurately reflect the market
price of acquiring the drug. These
updates must occur not less frequently
than once every 7 days, beginning with
an initial update on January 1 of each
year.
We codified this requirement in
§ 423.505(b)(21). We also amended
§ 423.505(i)(3) with respect to contracts
or written arrangements between Part D
sponsors and pharmacies or other
providers, first tier, downstream and
related entities. Specifically,
§ 423.505(i)(3)(viii)(A) requires that
sponsors’ pharmacy contracts include a
provision establishing regular updates
of any prescription drug pricing
standard used by the Part D sponsor,
consistent with § 423.505(b)(21), and
§ 423.505(i)(3)(viii)(B) requires that a
Part D sponsor’s pharmacy contract
indicate the source used by the Part D
sponsor for making any such pricing
updates. We finalized these regulations
in a final rule entitled, ‘‘Medicare
Program; Medicare Advantage Program
and Prescription Drug Benefit
Programs’’ at 76 FR 54600 (September 1,
2011) (‘‘September 2011 final rule’’).
We stated in the preamble to the
September 2011 final rule that a
‘‘prescription drug pricing standard’’ is
an accepted methodology based on
published drug pricing. In the preamble
to the proposed rule, we explained that
this was because we were unaware at
the time that there is at least one
standard based, at least in part, on costs
of the drugs that is not based strictly on
published drug pricing, which is
maximum allowable cost prices. Now
that we have become aware of these
types of pricing standards, we wish to
amend our regulatory requirement. We
believe that the updating requirement
should apply to pricing standards based
on the cost of a drug, even when the
standard is not based on published drug
pricing, an approach consistent with the
intent of the statute. The text of section
173 of MIPPA indicates the provision’s
purpose—Part D sponsors must update
their prescription drug pricing
standards regularly ‘‘to accurately
reflect the market price of acquiring the
drug.’’ We believe that this statement of
purpose indicates that the Congress
intended to provide pharmacies with a
means of ensuring that they have
current data on the amount of
reimbursement that they can expect,
including in cases when the
reimbursement is based upon maximum
allowable cost prices.
When the source of a prescription
drug pricing standard is published
publicly, such as with AWP or WAC,
pharmacies can determine their
reimbursement for all drugs at any given
time and can monitor these sources to
ensure they are being reimbursed
correctly. However, when a prescription
drug pricing standard is not published
publicly, network pharmacies are
unable to promptly determine whether
their reimbursement is consistent with
their contractual arrangements. This, in
turn, presents risks to the Medicare Part
D program in a number of ways. For
example, disclosure of the source used
to determine drug prices is necessary for
pharmacies to ensure accurate payment
of their claims, which is necessary for
accuracy in the costs submitted to CMS
by Part D sponsors on PDEs without
unnecessary later adjustments that are
disruptive to the operation of the Part D
program.
In addition, when network
pharmacies are unable to determine
whether their reimbursement is
consistent with their contractual
arrangements, the accuracy of the prices
displayed in the Medicare Prescription
Drug Plan Finder (‘‘MPDPF’’) is
questionable. While these prices only
provide an estimate of Part D drugs
costs at particular pharmacies,
beneficiaries do use the MPDPF to make
drug purchasing choices. If a pharmacy
does not know what it will be paid for
drugs on any given day, it cannot test
the MPDPF and validate the prices.
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00040 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29883
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
Thus, there is no assurance that the
posted prices are accurate, and
pharmacies are deprived of the
opportunity to compete based on more
accurate prices, and beneficiaries may
make choices based on erroneous
estimated drug costs. This is contrary to
the public policy goal of facilitating
competition in the health care system
and supporting consumers to be
informed purchasers of health care.
Also, when we compare posted prices to
prices submitted on PDEs to evaluate
the estimates provided in the MPDPF,
there can be no assurance that those
values correspond to the payments
pharmacies actually receive.
For this and other reasons detailed in
the preamble to the proposed regulation,
as well as in response to comments
received on the proposed regulation, we
are defining ‘‘prescription drug pricing
standard’’ in regulation. Specifically, in
§ 423.501 a ‘‘prescription drug pricing
standard’’ is now defined as ‘‘any
methodology or formula for varying the
pricing of a drug or drugs during the
term of a pharmacy reimbursement
contract that is based on the cost of a
drug, which includes, but is not limited
to, drug pricing references and amounts
that are based upon average wholesale
price, wholesale acquisition cost,
average manufacturer price, average
sales price, maximum allowable cost, or
other cost, whether publicly available or
not.’’ In addition, we are finalizing the
following technical changes to make the
regulations on prescription drug pricing
standards easier to reference: (1) To
combine the current requirements
contained in § 423.505(b)(21) (i) and (ii)
into (i) and eliminate the reference to
the effective contract year 2009 as no
longer necessary. These requirements
generally state that Part D sponsors
agree to update any prescription drug
pricing standard (as would be defined in
§ 423.501) on January 1 of each contract
year and not less frequently than once
every 7 days thereafter. Also, we are
moving the current requirement to
indicate the source used for making any
such updates to (b)(21)(ii) from
§ 423.505(i)(3)(viii)(B), so that it is
clearer by its placement in the
regulation that this requirement is on
Part D sponsors.
For new paragraph
§ 423.505(b)(21)(iii), we are finalizing a
new requirement and not a technical
change, that Part D sponsors agree in
their contracts with CMS to disclose all
individual drug prices to be updated to
the applicable pharmacies in advance of
their use for reimbursement of claims, if
the source for any prescription drug
pricing standard is not publicly
available. This means, in conjunction
with the proposed definition of a
‘‘prescription drug pricing standard’’
discussed previously, that Part D
sponsors have to convey to network
pharmacies the actual maximum
allowable cost prices to be changed in
advance. We are requiring that the
actual maximum allowable cost prices
be disclosed in advance because, if the
pharmacies are not able to use the
updates as a reference against which
they can check their reimbursements,
there would be no point to the statutory
requirement.
As a final technical change, we are
eliminating language in
§ 423.505(i)(3)(viii)(A) about
establishing regular updates of any
prescription drug pricing standard used
by the Part D sponsor, which is
duplicative to language in
423.505(b)(21). As a result of the
changes described previously, there
would be no paragraphs (A) and (B) of
§ 423.505(i)(3)(viii) (which we note will
be redesignated as § 423.505(i)(3)(vii)
due to other changes in this final rule),
and this provision simply requires that,
if applicable, each and every contract
governing Part D sponsors and first tier,
downstream, and related entities, must
contain provisions addressing the
prescription drug pricing standard
requirements of § 423.505(b)(21). We
believe these changes will make the
regulation text easier to reference and
understand.
Comment: We received a very
significant number of supportive
comments for our proposal. These
commenters asserted that maximum
allowable cost prices are a source of
deep and ongoing concern for
pharmacies. Specifically, these
commenters assert that PBMs update
maximum allowable cost prices of drugs
for which the drug costs are declining
in a timely manner, but do not do so
when the drug costs are increasing.
These commenters asserted in particular
that there were significant spikes in the
acquisition costs for certain generic
drugs in Fall 2013, but that PBMs did
not update their maximum allowable
cost prices accordingly. These
commenters also offered specific
examples of maximum allowable cost
prices of drugs that they asserted
resulted in reimbursement that was
below pharmacy acquisition costs for
the drugs, yet the drugs were not
available on the market at lower prices.
These commenters stated that
pharmacies were forced not to stock
certain drugs due to inadequate
reimbursement based on maximum
allowable cost prices of drugs,
sometimes creating access issues for
patients. These commenters further
stated that the pharmacies are even in
danger of going out of business
altogether due to the low maximum
allowable cost prices for drugs, and that
if pharmacies are forced to close their
doors for this reason, there would be
even greater health care access issues in
many communities.
The supportive comments stated that
greater transparency in maximum
allowable cost prices of drugs would not
only give pharmacies the ability to shop
for more cost-effective versions of
generic drugs, but would improve
pharmacies’ ability to evaluate Medicare
Part D plan contract proposals, plan
their business staffing levels and
potential capital investments, and
monitor claims reimbursements and
appeal when it appears that there has
been a reimbursement error.
Conversely, some other commenters
opposed our proposal. One commenter
asserted that our proposal was based
upon anecdotal complaints from
pharmacies. This commenter stated that
PBMs make their most utilized
maximum allowable cost list available
upon request to any pharmacy that asks
for it, and that pharmacies almost never
make such a request.
Response: We thank the commenters
for their supportive comments of our
proposal. Given the voluminous number
of supportive comments we received,
we disagree with the commenter that
stated that our proposal was based upon
anecdotal pharmacy complaints.
However, we were surprised to learn
that pharmacies do not routinely request
PBMs’ most utilized maximum
allowable cost lists, and wonder if
pharmacies do not realize that they are
available upon request. We agree with
the supportive commenters that greater
drug price transparency will further
increase competition in the drug market
which can lead to even lower drug
prices. Therefore, we encourage
pharmacies to make requests for the
most utilized maximum allowable cost
lists from the PBMs with which they do
business, and thank the commenter for
this suggestion.
Comment: Many commenters support
our proposal out of concern that the
uncertainties surrounding current
maximum allowable cost prices for
drugs fall more heavily on smaller rural
and community pharmacies and may
limit beneficiary access. Additionally,
these commenters expressed support for
greater drug price transparency for
Medicare beneficiaries.
Response: We thank the commenters
for their support of our proposal.
Comment: Some commenters opposed
our proposal, asserting that it would
increase costs by requiring a specific
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00041 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29884
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
time period (which many commenters
interpreted to be 7 days advance notice)
for advance notice, as generic drug costs
generally decrease over time. It also
appeared that some commenters
asserted that requiring any advance
notice of maximum allowable cost
prices would increase costs, including
one who made a general assertion that
it would permit pharmacies and drug
manufacturers to ‘‘game the system’’ by
modifying the timing of their various
transactions in a manner that capitalizes
on the pricing changes. Other
commenters stated that the proposal
would interfere with a mechanism that
incentivizes pharmacies to purchase the
least expensive generic drug available.
Finally, some commenters opposed the
requirement, asserting that requiring
price updates at least every 7 days is
redundant of the frequent updates that
are inherent in a maximum allowable
cost pricing mechanism and only adds
administrative cost.
Conversely, many commenters
requested that PBMs be required to give
at least 7 days prior notice before a
maximum allowable cost price change.
One commenter opposed the proposal,
but recommended as an alternative that
maximum allowable cost prices be
updated every 7 business days, and not
necessarily beginning on January 1 of
each year. Another commenter opposed
the proposal, but recommended as an
alternative that the no-less-than-7 day
update requirement for maximum
allowable cost prices be extended to no
less than every 14 days.
Response: This requirement does not
specify any particular time period for
advance notice of maximum allowable
cost prices to network pharmacies. The
requirement is that maximum allowable
cost prices of drugs must be updated at
least every 7 days and disclosed in
advance of their use, if the source for
any prescription drug pricing standard
is not publicly available. Also, if generic
prices generally decrease over time,
updating maximum allowable cost
prices for drugs at least every 7 days
generally should have a downward
pressure on overall drug costs.
Therefore, we do not agree with the
commenters that the requirement will
necessarily increase costs. Also,
maximum allowable cost prices
currently must be disclosed at point-of-
sale (POS) in order for a drug claim to
process, so we do not believe that mere
advance notice changes the drug claims
processing system so significantly as to
permit gaming of the system,
particularly since the commenter that
raised this concern provided no detail to
back up its assertions about how any
such gaming would occur. Furthermore,
we do not understand the argument for
the status quo—that disclosing
maximum allowable cost prices only at
POS better incentivizes pharmacies to
purchase the least expensive generic
drugs compared to requiring some
advance notice of those prices to the
pharmacies. We think pharmacies will
still be incentivized to acquire a drug at
the lowest cost possible regardless of
whether disclosed maximum allowable
cost prices are declining or increasing.
We further were not persuaded by the
argument that the requirement is
redundant, as it seems to suggest that
the Part D sponsors/PBMs will
frequently update maximum allowable
cost prices anyway and disclose them at
POS, but requiring them to be updated
at least every 7 days and disclosed in
advance adds significant administrative
costs. In fact, we think just the
opposite—that negligible administrative
costs will be incurred by Part D
sponsors due to this requirement, since
they are using and updating maximum
allowable cost prices for reimbursement
of drug claims already and must make
minimal changes to that current system
to comply with this requirement. In
other words, so long as Part D sponsors
are updating maximum allowable cost
prices as frequently as commenters
asserted that the prices change and
using them for reimbursement, then the
new updating and disclosure
requirement changes nothing for that
sponsor, other than that the sponsor
must now disclose the maximum
allowable cost prices to its network
pharmacies in advance of their use
(rather than just at point-of-sale) in a
way that enables the pharmacy to
connect a claim to the correct drug price
at the appropriate point in time in order
to validate the price. However, we
acknowledge that to the extent the
assertions of some commenters are
true—that PBMs update maximum
allowable cost prices only when drug
prices are declining, but not when they
are increasing—then we would agree
that this requirement may also result in
more updating for PBMs.
In addition, we note that the
requirement to update prescription drug
pricing standards every 7 days
beginning on January 1 of each year is
a statutory one. We do not have the
authority to implement different update
timing requirements, nor to disregard
the January 1 start date every year.
Comment: Some commenters stated
that our proposal was operationally
infeasible, as there are different
maximum allowable cost lists for
different pharmacies, types of
pharmacies, types of programs
(commercial, Medicare D, TRICARE,
etc.) and over 100,000 drugs are subject
to maximum allowable cost prices,
(sometimes daily). Some other
commenters stated that sending network
pharmacies a stream of continuous
maximum allowable cost pricing
updates would be a nuisance and
distraction and not helpful to network
pharmacies. One commenter did not
object to our proposal, as long the
requirement can be met in a manner that
is efficient, such as on a look-up basis
through a secure internet site that
network pharmacies can access at any
time to obtain the most current
maximum allowable cost pricing for a
particular drug.
One commenter requested that we
require maximum allowable cost prices
to be disclosed via a certain consistent
format layout and delivery method and
include industry standard drug
identifiers, such as Generic Pricing
Indicators (GPI), and that the data
format allow for efficient data analysis
such as MS Excel, or a text document
that could be converted to Excel.
Response: We were not persuaded by
the commenters that stated our proposal
was operationally infeasible. It does not
make sense to us that Part D sponsors/
PBMs can manage the complexity in
pharmacy reimbursement described in
the comments, but cannot manage to
modify that existing system in order to
disclose the prices in advance of their
use to network pharmacies, and update
them at least every 7 days. Rather, we
were persuaded by the commenter that
described one option for meeting the
requirement—through a secure internet
site that allowed network pharmacies to
look up their drug prices. This option
would be compliant with the
prescription drug pricing standard
requirement, so long as the site or other
delivery method to convey maximum
allowable cost prices enables
pharmacies to connect a claim to the
correct drug price at the appropriate
point in time in order to validate the
price. We decline to require a certain
format layout and delivery method for
disclosure of maximum allowable cost
prices, but note these matters can be
addressed by the parties in their
negotiations.
Comment: Some commenters asserted
that requiring the disclosure of
maximum allowable cost methodology
would increase Part D program costs by
revealing competitive information.
Many other commenters requested that
we require PBMs to disclose the specific
NDCs used to compute maximum
allowable cost prices on drugs.
Response: Our proposal did not
require Part D sponsors/PBMs to
disclose their maximum allowable cost
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00042 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29885
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
methodology, nor the proprietary data
source or basis used to develop
reimbursement rates. We note that
423.505(b)(21)(ii) will require a Part D
sponsor to indicate the source for
making updates to a prescription drug
pricing standard. In the case of publicly
available standards, the sponsor would
identify the standard. In the case of
maximum allowable cost pricing that is
not publicly available, the sponsor
would indicate that the standard is
maximum allowable cost pricing to
meet this particular requirement. We
also decline to require Part D sponsors
to disclose the specific NDCs used to
compute maximum allowable cost
prices. However, we note that these
matters can be addressed in contractual
negotiations.
Comment: Some commenters asserted
that maximum allowable cost prices are
not a prescription drug pricing standard,
and that CMS is exceeding its statutory
authority in making it one. One
commenter asserted that the Congress’
intent in enacting section 173 of MIPPA
was to ensure that pricing standards are
timely adjusted when market prices
fluctuate and not to ensure that
pharmacies have current data on
reimbursement amounts. This
commenter also stated that when a
payment methodology uses non-public
costs for setting prices, payment
amounts may have no direct
relationship to fluctuations in
acquisition costs. Many commenters
specifically supported the language
‘‘includes, but is not limited to’’ in the
proposed definition of prescription drug
pricing standard, stating that without
this language, PBMs will shift to a
different drug claim reimbursement
mechanism over time and assert that the
new mechanism is not subject to the
prescription drug pricing standard
regulation. Another commenter
helpfully pointed out that our proposed
definition of ‘‘prescription drug pricing
standard’’ mistakenly referred to
‘‘wholesale average cost’’ instead of
‘‘wholesale acquisition cost.’’
Response: We thank the commenters
for their supportive comments and note
that we are finalizing the definition of
‘‘prescription drug pricing standard’’ as
proposed, with the exception of
changing ‘‘wholesale average cost’’ to
‘‘wholesale acquisition cost.’’ We
disagree with the commenters that
maximum allowable cost prices are not
a prescription drug pricing standard,
and we disagree that we are exceeding
our authority in specifying in regulation
that maximum allowable cost prices,
like other prescription drug standards,
must be updated in accordance with the
statutory requirements. In our view, it is
clear that Congress believed that if a
standard is based on the cost of a drug
(whether directly or indirectly), it must
be updated to accurately reflect the
market price of acquiring the drug.
Since the statutory language of section
173 of MIPPA does not exclude
maximum allowable cost prices from
the term ‘‘prescription drug pricing
standard,’’ and maximum allowable cost
prices are based on the cost of the drug
and thus fluctuate and are updated, we
believe it is reasonable to interpret the
term, ‘‘prescription drug pricing
standard,’’ to include maximum
allowable cost prices. As such, they
must be treated as any other
prescription drug pricing standard
under the statutory and regulatory
requirements. In the case of published
prescription drug pricing standards, the
standards themselves provide
pharmacies with current data on
reimbursement amounts. In the case of
non-published ones, disclosing the
prices themselves in advance of their
use provides this data. We agree with
the commenter who asserted that
MIPPA section 173 is intended to
ensure that prices are adjusted timely,
but we disagree that it necessarily
follows that the Congress did not intend
to ensure that pharmacies had access to
current data on reimbursement
amounts. We believe that the
requirement for timely updating of
reimbursement standards must include
sufficient transparency so that
pharmacies can determine that the
updating requirement is being fulfilled.
The disclosure requirements we are
finalizing here are consistent with the
updating requirement, and are
appropriate to ensure sufficient
transparency.
Comment: Many commenters stated
that having current data on the amount
of reimbursement pharmacies can
expect in turn impacts costs that plan
sponsors submit to CMS, as well as
prices displayed on Medicare
Prescription Drug Plan Finder (MPDFP).
Other commenters asserted that the
MPDFP is updated every 2 weeks with
pricing that is effectively a month old,
and that the validity of estimated prices
on the MPDPF does not depend on the
ability of pharmacies to verify the prices
shown, and that this responsibility is on
Part D sponsors. One commenter stated
that our requirement would necessitate
more frequent updating of the MPDPF.
Response: We thank the commenters
for their supportive comments of our
similar assertions in the preamble to the
proposed rule. Our proposal does not
affect the current process for Part D
sponsors to submit drug price for the
MPDPF. Our point about the MPDPF in
the preamble to the proposed rule was
that this requirement will enable
pharmacies to validate maximum
allowable cost prices in the MPDPF.
While we agree with the commenters
that the MPDPF is not a real-time
information system, but rather reflects
drug prices at a point in time, we note
that these prices should be the correct
prices for that point in time. Currently,
however, pharmacies have no ready way
to validate the prices in the MPDPF that
are based on maximum allowable cost
prices if they choose to do so. Once
maximum allowable cost prices are
disclosed to pharmacies in a way that
enables pharmacies to connect a claim
to the correct drug price at the
appropriate point in time, they will be
able to validate prices in the MPDPF
and alert sponsors, or CMS, to any
issues.
Comment: A few commenters
requested a delay in the effective date
for implementation of this requirement
until January 1, 2016. This delay would
provide for more preparation time.
Response: We were persuaded by
comments to delay the effective date of
this proposal until 2016 to give Part D
sponsors time to consider the format
layout and delivery method for
conveying maximum allowable cost
prices to network pharmacies in a
manner that allows the pharmacies to
connect a claim to the correct drug price
at the appropriate point in time in an
efficient way.
Comment: Many commenters
requested that we include a definition
for which drugs can be included on a
maximum allowable cost list, and
requirements for an appeals process for
challenging maximum allowable cost
prices and for standards related to
pharmacy audits. One commenter stated
that it sends 200 requests per month to
PBMs to increase their maximum
allowable cost reimbursement rates to
be closer to pharmacy acquisition costs
and that very few are ever responded to,
and fewer still are ever adjusted.
Response: These comments are out of
scope of our proposal.
In light of all the comments received,
we are finalizing this proposal without
change, except for correcting the error in
the definition for prescription drug
pricing standard previously noted and
delaying the effective date until January
1, 2016.
19. Any Willing Pharmacy Standard
Terms & Conditions (§ 423.120(a)(8))
Section 1860D–4(b)(1)(A) of the Act
requires Part D plans to permit any
pharmacy meeting the plan’s Terms and
Conditions (T&C) to participate in the
plan’s network. We used this authority
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00043 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29886
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
to establish requirements under
§ 423.120(a)(8) and 423.505(b)(18) that
plan sponsors have reasonable and
relevant T&C for network participation
in their standard contract, and allow
any pharmacy meeting the T&C to
participate as a network pharmacy for
that plan. Section 1860D–4(b)(1)(B) of
the Act permits sponsors to reduce cost
sharing ‘‘below the level otherwise
required,’’ notwithstanding paragraph
(A). Thus, the statute permits a
‘‘preferred’’ cost sharing level to be
offered at some network pharmacies.
Since the beginning of the program, we
have required sponsors to offer standard
T&Cs to any willing pharmacy in order
to achieve broad network access, but
have permitted sponsors to offer
different T&Cs in return for preferred
cost sharing to a smaller subset of its
network. We have previously stated that
we believed our interpretation of these
two seemingly conflicting statutory
provisions struck an appropriate
balance between the need for broad
pharmacy access and the need for Part
D plans to have appropriate contracting
tools to lower costs. In this section we
proposed that in place of sponsors
having one contract with standard terms
for any willing pharmacy and a second
preferred cost sharing contract for a
limited subset of pharmacies, that
sponsors instead have standard T&C for
network participation that list all
combinations of cost sharing and
negotiated prices possible for retail
settings under the plan, allowing any
willing pharmacy the opportunity to
offer preferred cost sharing if the
pharmacy can offer the requisite level of
negotiated prices.
When discussing cost sharing,
distinctions are made in this section
between plans offering a preferred cost
sharing level and plans that do not. For
the purposes of this section, the cost
sharing levels offered at retail
pharmacies not contracted to offer
preferred cost sharing are referred to as
standard cost sharing levels. Cost
sharing levels offered at retail
pharmacies at the preferred T&C are
referred to as preferred cost sharing
levels.
We have heard from many
pharmacies, many of them small
independent community pharmacies,
that plans do not offer any willing
pharmacy the opportunity to offer
preferred cost sharing. Instead, some
pharmacies are being offered only the
plan’s standard T&C, at the highest level
of beneficiary cost sharing. We received
more than 200 comments in response to
our discussion of this topic in the
Announcement of Calendar Year (CY)
2014 Medicare Advantage Capitation
Rates and Medicare Advantage and PDP
Payment Policies and Final Call Letter
(2014 Call Letter) pp. 175 and 176 at
http://www.cms.gov/Medicare/Health-
Plans/MedicareAdvtgSpecRateStats/
Downloads/Announcement2014.pdf.
Most of these comments were from
pharmacies concerned about barriers to
entry for participation in preferred
networks, and many of these argued that
such limited networks violate the
statutory intent of the network access
provisions at section 1860D–4(b)(1) of
the Act. In particular, these commenters
disagreed that such barriers were
consistent with the any willing
pharmacy requirement as stated in
1860D–4(b)(1)(A) of the Act.
Consequently, we reviewed our
original regulatory interpretation of
these provisions, not only in light of
these complaints, but also in light of our
experience in the Part D program. We
believe that an alternative reading of
sections 1860D–4(b)(1)(A) and (B) of the
Act to reduce barriers is not only
permissible, but also it would have the
following key policy benefits, which we
describe as follows:
• Increased access for beneficiaries to
preferred level cost sharing with any
willing pharmacy able to agree to the
T&C that include preferred cost sharing.
• Improved opportunity for
competition among pharmacies
contracting with the sponsor to charge
no more than the ceiling price stated in
the contract for preferred cost sharing,
reducing costs charged to the program.
• Improved clarity for beneficiaries
surrounding cost sharing levels
available at retail and mail order
pharmacies.
We have heard the assertion that
limited networks achieve greater savings
than broader networks, and that
moreover, allowing more participants
into a limited network than those hand-
picked by the sponsor will necessarily
lead to increased prices. However, we
have been running a natural experiment
of sorts relative to this assertion in the
Part D program. If limited networks per
se led to significantly lower costs, we
would see consistently significant
savings in those network segments
relative to the rest of the sponsors’
networks. However, an April 2013
analysis by CMS, ‘‘Negotiated Pricing
Between Preferred and Non-Preferred
Pharmacy Networks’’, reviewed actual
program experience and indicated that
this is not the case across the board (see
http://www.cms.gov/Medicare/
Prescription-Drug-Coverage/
PrescriptionDrugCovGenIn/Downloads/
PharmacyNetwork.pdf). As the 2012
claims show, there is wide variation in
discounting across sponsors. Consistent
savings are not seen uniformly. In some
cases, pharmacies extending high
discounts are ones that have been
excluded from limited networks offering
preferred cost sharing, while some
pharmacies within the limited networks
offer effectively no discounts compared
to the rest of the network.
We have also heard the argument that
the pharmacies in currently limited
networks are offering deeper discounts
solely in return for increased market
share and that they will withdraw such
offers if the limited network is opened
up to other pharmacies that can meet
those T&C. We are skeptical that such
participants in the highly competitive
retail market will abandon their market
share by returning to the broader
network T&C. As some network
pharmacies offering standard cost
sharing have been able to extend
discounts in pricing even deeper than
what is seen in some pharmacies
offering preferred cost sharing, it is not
obvious that negotiated prices would
necessarily increase in the aggregate in
the event that a limited number of
pharmacies consider changing from
preferred to standard cost sharing. We
have also been informally told by one
sponsor with preferred cost sharing in a
limited network that its preferred cost-
sharing T&C already are offered to any
willing pharmacy. For these reasons, we
do not believe that our proposal would
result in increased prices.
We also believe that there is a limit to
the number of cost sharing levels offered
under a benefit plan that can be well
understood by beneficiaries. When
establishing its network, a Part D
sponsor does not offer identical T&C for
network participation to every
pharmacy. Certain terms will
necessarily differ among contracts with
the different types of pharmacies
needed to provide all Part D drugs, if for
no other reason than to address the
different access and service standards
established by CMS. These various
types include at a minimum: Retail,
mail-order, long-term care institutional,
limited-distribution-drug specialty, and
home infusion therapy pharmacies.
Terms will also differ with respect to
negotiated prices and the level of cost
sharing that a pharmacy’s claims will be
subject to. For instance, long-term care
institutional, specialty, and infusion
pharmacies are generally offered at the
standard level of cost sharing (for the
applicable formulary tier) for a month’s
supply of a covered drug. Retail and
mail-order pharmacies, in contrast,
currently may contract with plans to be
offered at more than one cost sharing
level.
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00044 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29887
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
Cost sharing at retail and mail-order
pharmacies currently vary on three
dimensions: Whether the cost sharing is
standard or preferred, on the quantity
dispensed (or ‘‘days’ supply’’), and on
dispensing location.
We proposed that a more simplified
benefit design, incorporating these three
variables and accommodating a more
clearly defined set of cost sharing levels,
would promote better understanding of
Part D plan benefits, both in terms of
beneficiary cost sharing and prices
charged to the program, as well as
streamlined contracting options. We
also proposed to expressly state the total
number of possible cost-sharing levels,
to clarify expectations and to preempt
the introduction of additional or
unauthorized cost-sharing levels in the
future.
For prescriptions not subject to Long
Term Care, specialty pharmacy, or home
infusion pricing, the interaction of the
following four provisions of section
1860D–4(b)(1) of the Act point to three
authorized levels of cost sharing:
Standard, preferred, and extended days’
supplies for retail and mail order
pharmacies. We proposed to minimize
the number of variations on these three
levels to the following options and to
ensure that standard T&C for network
participation offer every level available
for each respective pharmacy type. First,
we proposed to limit long term care,
specialty, and infusion pharmacy cost
sharing to the standard monthly rate, as
is industry practice today. Second, we
proposed to limit retail pharmacies to
the three authorized levels; either the
standard or preferred monthly rate (for
supplies up to 34 days), and one
extended days’ supply cost sharing rate
not exceeding three times the monthly
retail rate (either three times the
standard monthly retail rate or three
times the preferred monthly retail rate,
depending upon the T&C of the
pharmacy’s contract). Third, we
proposed to limit the levels of cost
sharing at mail-order pharmacies to one
monthly rate and one extended day mail
order cost sharing rate (for any supplies
greater than 34 days) for reasons
discussed previously. We additionally
solicited comments on the frequency of
mail order being used to fill
prescriptions lasting one month or less.
We note that these proposals would not
alter our requirements around the
dispensing of any days’ supplies less
than 30 days, which is still subject to
the ‘‘daily cost sharing’’ provision at
§ 423.153(b)(4).
In summary, we proposed to use the
authority in section 1860D–4(b)(1)(C)(i)
of the Act to establish rules defining
convenient access within a Part D
pharmacy network, combined with the
authority in section 1860D–4(b)(1)(A) of
the Act to revise the any willing
pharmacy requirements, to ensure that
any pharmacy that can meet the
applicable T&C for offering standard or
preferred cost sharing can join the
network on those terms. We believe the
network access provisions in section
1860D–4(b)(1) of the Act support
expanding § 423.120(a)(8) to all levels of
cost sharing offered under a sponsor’s
benefit plans. We believe that doing so
supports the Congressional intent to
have plans compete on negotiated prices
by making this price competition more
open and accessible to pharmacies.
Specifically, we proposed to revise
§ 423.120(a)(8) to require that, in
establishing its contracted pharmacy
network, a Part D sponsor offering
qualified prescription drug coverage
must comply with all of the following
requirements:
• Must offer and publicly post
standard terms and conditions for
network participation for each type of
pharmacy in the network subject to the
following:
++ May not require a pharmacy to
accept insurance risk as a condition of
participation in the PDP sponsor’s
contracted pharmacy network.
++ Must offer payment terms for
every level of cost sharing offered under
the sponsor’s plans consistent with CMS
limitations on the number and type of
cost sharing levels, and for every type of
similarly situated pharmacy.
• Must contract with any willing
pharmacy able to meet one set of the
terms and conditions offered by that
plan for that type of pharmacy.
We also proposed to make conforming
changes to the contracting provisions at
§ 423.505(b)(18) to require Part D
sponsors to agree to have standard T&C
for network participation that meet the
requirements described in
§ 423.120(a)(8), with reasonable and
relevant T&C of participation for each
type of pharmacy in its network. We
believe these proposed requirements
would better ensure that each Part D
plan: (1) Provides convenient access to
Part D drugs in all Part D settings and
to the extent practical, at all cost sharing
levels; and (2) offers cost sharing levels
that encourage beneficiaries to make
choices that minimize costs not only for
themselves, but also to the Medicare
Part D program as a whole. We solicited
comments on these proposals to expand
the any willing pharmacy T&C and to
streamline the levels of cost sharing
offered under those standard T&C. We
believe these proposals would increase
beneficiary understanding of and access
to cost sharing that is better aligned
with the lowest negotiated prices,
improve market competition, and
increase downward pressure on total
program costs. We received more than
4,000 comments on these proposals and
our response follows:
Comment: This proposal received
significant support from commenters
citing an interest in expanding access to
preferred cost sharing and creating a
more level playing field for small and
independent pharmacies. Many
reported that the lower cost sharing at
a limited number of pharmacies offering
preferred cost sharing leads many
beneficiaries to drive sometimes great
distances to access these savings, even
when they have a stated preference to
stay with a local pharmacy, or one
where they have a long-term history
with the pharmacist. Many other
commenters reported that some current
marketing practices are mistakenly
interpreted as suggesting that only
pharmacies offering preferred cost
sharing can be used by enrollees of that
plan, also leading many beneficiaries to
leave their preferred choice of where to
access pharmacy services.
Response: We appreciate the strong
support we received for this proposal.
We agree with many of the commenters
who wrote that beneficiaries should be
able to choose where they obtain their
pharmacy services, and we are very
concerned to hear that the current
incentives (and potentially current
marketing of pharmacies offering
preferred cost sharing) lead many
beneficiaries to believe that only those
pharmacies offering preferred cost
sharing can be used. We are also
concerned by the many comments
reporting that beneficiaries are now
driving 30–60 miles to the nearest
pharmacy offering preferred cost
sharing, or are feeling forced into using
mail-order services, despite a preference
to stay with a local pharmacy. We share
the concerns of commenters who
suggest that current contracting
practices by sponsors, only extending
preferred cost sharing T&C with select
pharmacies, are being interpreted by
Medicare beneficiaries as a violation of
the Any Willing Pharmacy provision in
statute. While the Any Willing
Pharmacy provision applies only to
participation in a plan’s pharmacy
network, not the subset of pharmacies
offering preferred cost sharing, many
commenters reported that access to
preferred cost sharing does not align
with beneficiaries’ expectation for
choice of pharmacy service provider.
That is, if a plan offers preferred cost
sharing, beneficiaries assume they will
be able to access that cost sharing at
their own ‘‘preferred’’ pharmacy.
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00045 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29888
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
Comment: Some commenters asserted
that requiring plan sponsors to allow
any willing pharmacy to accept publicly
disclosed terms and conditions to offer
preferred cost sharing to plan enrollees,
in exchange for requisite drug price
discounts, would limit sponsors’ ability
to negotiate significant discounts from a
more limited number of pharmacies.
Some of these commenters stated that
they did not believe CMS had the
authority to make this change. A few
commenters suggested that CMS use its
current authority to respond to plan
offerings that we determine to be
discriminatory in the availability and
access they provide to preferred cost
sharing, and to reject plans failing to
offer fair access. Many of the opponents
of this proposal objected to publicly
posting contract T&C, as potentially
undermining price competition. These
commenters suggested that this change
would ultimately result in higher drug
costs, as a higher number of pharmacies
offering preferred cost sharing would
lead to a decrease in the volume of
enrollees electing to use any one of
these pharmacies, and as a result
pharmacies would not be as willing to
negotiate deeply discounted drug prices
without the promise of a high volume of
enrollees. Some commenters submitted
economic analyses in support of their
claims. Some, but not all opponents
questioned CMS’ assumption that
pharmacies currently offering preferred
cost sharing would not elect to
discontinue offering preferred cost
sharing if such terms and conditions
were available to any willing pharmacy.
Response: We continue to believe that
reduced preferred cost sharing offered to
plan enrollees should be aligned with
reduced drug prices charged to the
program, aligning the cost sharing price
signals with high value plans offering
reduced drug pricing. We believe that
opening up these limited networks to
any pharmacy willing to charge no more
than the contract’s ceiling price to
qualify for offering the lower preferred
cost sharing may be necessary to restore
price competition in these networks. We
disagree with the comments suggesting
that this provision violates the non-
interference provision. Expanding
access to preferred cost sharing aligns
with the authority to establish rules
defining convenient access within a Part
D network, combined with the authority
to interpret the any willing pharmacy
requirements. We believe the network
access provisions in section 1860D–
4(b)(1)(A) of the Act support expanding
§ 423.120(a)(8) to establish access
standards for all levels of cost sharing
offered under a sponsor’s benefit plans,
and that this expansion aligns with
Congressional intent to have open
competition between plans based on
negotiated price.
Numerous comments from opponents
of the provision cited published
analyses that predate Part D on the
elimination of selective contracting
practices at the state level and higher
drug expenditures noted after this
change. However, we are concerned that
traditional analyses that study drug
expenditures after an expansion of a
previously limited network may not be
directly relevant to the Part D market.
While we recognize the general parallels
between the studies submitted for
consideration and the any willing
pharmacy proposal, any attempt to
generalize these studies to the Part D
benefit would need to incorporate
multiple other variables, especially
given the revenue streams other than
point-of-sale pricing that may distort
other economic incentives. The studies
submitted offer only limited explanation
of what trends in utilization, pricing,
and care management surrounded the
state-level changes, and without that
context we do not consider these
analyses persuasive. Further supporting
our concerns, one commenter provided
alternative economic analysis that
supported our assumption that within
the Part D market expanding access to
any willing pharmacy may not affect
drug prices.
While we continue to believe that
there are benefits in increasing
transparency and in permitting
pharmacies willing to charge reduced
prices in exchange for offering preferred
cost sharing, in light of these comments
we believe it is necessary to further
analyze the potential impacts on the
Part D market. Considering the
conflicting comments and analyses
submitted, and the potential
consequences of implementing any
changes based on incorrect
assumptions, we believe it is important
to wait and to spend additional time
considering the evidence for potential
financial impacts within the Part D
benefit. We will be closely studying
preferred cost sharing practices,
including the associated point-of-sale
drug pricing, going forward. In response
to the comments suggesting that CMS
use its current authority to respond to
plan offerings that we determine to be
discriminatory in its proposed
availability and access to preferred cost
sharing, we will further explore our
authority in this area. In addition, we
plan to closely monitor beneficiaries’
access to preferred cost sharing, as well
as drug pricing by pharmacies offering
preferred cost sharing, to determine
whether future rulemaking in this area
is necessary.
In summary, pending further study,
we are not finalizing the any willing
pharmacy contracting proposed
provision changes to § 423.120(a)(8) or
423.505(b)(18), nor the proposed
changes to limit the authorized levels of
cost sharing. We will engage in further
notice and comment rulemaking on this
issue as warranted in the future.
20. Enrollment Requirements for
Prescribers of Part D Covered Drugs
(§ 423.120(c)(5) and (6))
To improve our ability to oversee the
Medicare Part D program, we proposed
to implement section 6405(c) of the
Affordable Care Act effective January 1,
2015. This section provides the
Secretary with authority to require that
prescriptions for covered Part D drugs
be prescribed by a physician or eligible
professional (as defined at section
1848(k)(3)(B) of the Act (42 U.S.C.
1395w–4(k)(3)(B)) who is enrolled in the
Medicare program pursuant to section
1866(j) of the Act (42 U.S.C. 1395cc(j)).
We generally proposed in revised
§ 423.120(c)(5) and new paragraph (6)
that a prescriber of Part D drugs must
have (1) an approved enrollment record
in the Medicare program, or (2) a valid
opt-out affidavit on file with a Part A/
Part B Medicare Administrative
Contractor (A/B MAC) in order for a
prescription to be eligible for coverage
under the Part D program. More
specifically, we proposed the following:
• Under § 423.120(c)(5)(ii)(A) and (B),
a Part D sponsor must deny or must
require its PBM to deny a pharmacy
claim for a Part D drug if: (1) An active
and valid physician or eligible
professional National Provider Identifier
(NPI) is not contained on the claim; or
(2) the physician or eligible professional
(i) is not enrolled in the Medicare
program in an approved status, and (ii)
does not have a valid opt-out affidavit
on file with an A/B MAC.
• Under § 423.120(c)(5)(ii)(C) and
(c)(6)(ii), to receive payment for a drug,
a beneficiary’s request for
reimbursement from a Part D sponsor
must be for a Part D drug that was
dispensed in accordance with a
prescription written by a physician or
eligible professional who: (1) Is
identified by his or her legal name in
the request; and (2) is either enrolled in
Medicare in an approved status or has
a valid opt-out affidavit on file with an
A/B MAC.
• Under § 423.120(c)(6)(i), in order for
a Part D sponsor to submit to CMS a
prescription drug event (PDE) record,
the PDE must pertain to a claim for a
Part D drug that was dispensed in
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00046 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29889
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
accordance with a prescription written
by a physician or eligible professional
who is either (1) enrolled in Medicare
in an approved status, or (2) has a valid
opt-out affidavit on file with an A/B
MAC.
• Under § 423.120(c)(6)(iii), a Part D
sponsor must deny or must require its
PBM to deny a pharmacy claim for a
drug (or a request for reimbursement
from a Medicare beneficiary for a drug)
if the claim does not meet the
requirements of § 423.120(c)(6)(i) or (ii),
respectively.
The overriding purpose of these
provisions is to help ensure that Part D
drugs are prescribed only by physicians
and eligible professionals who are
qualified to do so under state law and
under the requirements of the Medicare
program.
Our proposed enrollment deadline of
January 1, 2015 was intended to give
physicians and eligible professionals at
least 6 months after the publication of
a final rule to complete the Medicare
enrollment process. We solicited
comments regarding the propriety of
this effective date.
The Medicare enrollment process
requires that an A/B MAC screen and
validate each enrollment application
submitted by a physician or eligible
professional prior to the decision to
approve or deny enrollment in the
Medicare program. The enrollment
application collects identifying
information about the applicant and his
or her credentials, such as licensure
status. We have been concerned about
instances where unqualified individuals
are prescribing Part D drugs. In fact, in
a June 2013 report the OIG found that
the Part D program inappropriately paid
for drugs ordered by individuals who
clearly did not appear to have the
authority to prescribe. (See ‘‘Medicare
Inappropriately Paid for Drugs Ordered
by Individuals Without Prescribing
Authority’’ (OEI–02–09–00608).) There
have also been reports that the
prescriptions of physicians with
suspended licenses have been covered
by the Part D program. This should not
happen, and we believe we can better
address these and similar vulnerabilities
by verifying the credentials of
prescribers through either the Medicare
enrollment process or their submission
of a valid opt-out affidavit.
With respect to the latter, we note that
under section 1802(b) of the Act and the
implementing regulations at § 405.400 et
seq., certain physicians and eligible
professionals can opt-out of the
Medicare program and enter into private
contracts with Medicare beneficiaries.
By entering into such contracts, these
individuals do not bill the Medicare
program for non-emergency services
they furnish to beneficiaries.
Under our proposal, in short, the
prescriptions of a physician or eligible
professional who is not enrolled in
Medicare and does not have a valid opt-
out affidavit on file with an A/B MAC
would not be covered under the Part D
program. As explained in the proposed
rule, CMS would furnish or make
available to Part D sponsors a list of
physicians and eligible professionals
who have an approved Medicare
enrollment record or who have a valid
opt-out affidavit on file with an A/B
MAC.
We also solicited comments on the
following issues:
• Whether all pharmacies should be
required to enroll in Medicare in order
to dispense covered Part D drugs.
(Alternatively, we sought comment on
whether requiring Medicare enrollment
for network pharmacies is a ‘‘best
practice’’ in pharmacy contracting by
plan sponsors, and should be an integral
part of sponsors’ required fraud, waste
and abuse programs.)
• Whether doctors of dental surgery
or dental medicine, including family
dentists, should be required to enroll in
Medicare in order to prescribe covered
Part D drugs. (Note that we did not
propose to exclude dentists from our
requirements. Sections 423.120(c)(5)
and (6) were intended to apply to
dentists.)
We received a significant number of
comments regarding these proposed
provisions. Summaries of the comments
as well as our responses follow:
Comment: A number of commenters
opposed our proposed changes to
§ 423.120(c)(5) and the addition of
§ 423.120(c)(6). Several commenters
were concerned that these requirements
would disrupt Medicare beneficiaries’
current relationships with their
physicians or otherwise prevent patients
from seeing certain physicians, hence
denying them care. One commenter
stated that it appears that state licensure
alone is no longer sufficient for an
individual to prescribe drugs, and that
§ 423.120(c)(5) and (6) would
inappropriately limit one’s ability to
prescribe when he or she is otherwise
permitted to do so under state law. The
requirement to enroll is particularly
disconcerting, the commenter added,
considering that the prescribing
individual (as opposed to the pharmacy)
is not even receiving reimbursement
from Medicare for the prescribed drug.
Another commenter stated that
medication should be based on a
patient’s needs, rather than on whether
a physician is in the Medicare system.
Several commenters also requested
further clarification regarding the intent
of our proposed revisions.
Response: The central purpose of our
changes to § 423.120(c), as alluded to
previously, is to ensure that we can
verify that the prescriber is
appropriately licensed and certified, is
not excluded or debarred from
Medicare, and is otherwise qualified
under Medicare regulations to prescribe
Part D drugs. Again, we have been
concerned that unqualified individuals
are prescribing such drugs, and the
previously-referenced OIG report bears
this out. The enrollment process will
help ensure that Medicare beneficiaries
and the Trust Funds are protected,
which is why we intend to proceed with
our proposal. We note further that these
changes are fully consistent with our
requirement in § 424.507 that
physicians and eligible professionals
who order or certify certain services and
items are either enrolled in Medicare or
have a valid opt-out affidavit on file
with an A/B MAC.
Comment: Several commenters
contended that Medicare should not
require physicians who do not
participate in or take Medicare to enroll
in the program.
Response: Our changes to § 423.120(c)
permit a physician or eligible
professional who has a valid opt-out
affidavit on file with an A/B MAC to
prescribe Part D drugs.
Comment: Many commenters, some of
whom supported our proposed changes,
expressed concern about the proposed
January 1, 2015 date. Several of them
requested that the implementation of
§ 423.120(c)(5) and (6) be delayed until
2016 or even 2017 to give CMS,
prescribers, and plan sponsors adequate
time to prepare and to address all
operational and system challenges.
Other commenters suggested that CMS
utilize a phased-in approach, similar to
that which was used for CMS’
implementation of § 424.507. These
commenters asserted that this would
help ensure that patient care is not
interrupted, that all information
regarding prescribers’ enrollment
statuses is correct, that appropriate
system testing is done, that CMS
engages in regular communication with
all affected stakeholders, and that CMS
can more accurately report the number
of physicians and eligible professionals
who will be affected by our proposal.
Additional commenters recommended
that any revised implementation date be
on January 1 so as to coincide with the
beginning of the new plan year.
Response: We agree with these
commenters regarding the need to allow
adequate time to prepare. Therefore, we
are revising § 423.120(c)(5) and (6) to
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00047 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29890
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
establish an effective date of June 1,
2015. We understand the commenters’
desire for a January 1 date, but we do
not believe a delay until January 1, 2016
is feasible given our aforementioned
program integrity concerns. A June 1,
2015 date, we believe, strikes an
appropriate balance between the need to
have sufficient time to prepare and the
need to ensure that only qualified
individuals are prescribing Part D drugs.
We wish to assure plan sponsors,
prescriber and supplier organizations,
and beneficiary advocacy groups that
we will regularly communicate with
them in the months leading up to the
June 1, 2015 effective date to address
whatever concerns they have and to
keep them abreast of CMS’ preparations
for implementation.
Plan sponsors, prescribers,
beneficiaries, and other affected parties
should note that existing policies that
will be superseded by our changes
remain intact (and should continue to
be adhered to) through May 31, 2015.
In order to: (1) Help ensure that
stakeholders can effectively determine
which provisions apply to them before
and after June 1, 2015, (2) simplify and
consolidate our proposed changes to
§ 423.120(c), and (3) eliminate potential
duplication between the provisions we
proposed in (c)(5)(ii) and in (c)(6), we
are making several technical revisions.
The existing version of paragraph (c)(5)
will remain intact with the exception of
the addition of the ‘‘Before June 1, 2015,
the following are applicable’’ language
at the very beginning of the paragraph.
We are not finalizing our proposed
changes to paragraph (c)(5)(ii), but are
instead merging them with our addition
of paragraph (a)(6). Hence, our final
version of new paragraph (c)(6) will
read as follows:
‘‘(6) Beginning June 1, 2015, the
following are applicable—
(i) A Part D sponsor must deny, or
must require its pharmaceutical benefit
manager (PBM) to deny, a pharmacy
claim for a Part D drug if an active and
valid physician or eligible professional
(as defined in section 1848(k)(3)(B)(i) or
(ii) of the Act) National Provider
Identifier (NPI) is not contained on the
claim.
(ii) A Part D sponsor must deny, or
must require its PBM to deny, a
pharmacy claim for a Part D drug if the
physician or eligible professional (when
permitted to write prescriptions by
applicable State law)—
(A) Is not enrolled in the Medicare
program in an approved status; and
(B) Does not have a valid opt-out
affidavit on file with an A/B Medicare
Administrative Contractor (MAC).
(iii) A Part D sponsor must deny, or
must require its PBM to deny, a request
for reimbursement from a Medicare
beneficiary for a drug if the request is
not for a Part D drug that was dispensed
in accordance with a prescription
written by a physician or, when
permitted by applicable State law, other
eligible professional (as defined in
section 1848(k)(3)(B)(i) or (ii) of the Act)
who—
(A) Is identified by his or her legal
name in the request; and
(B)(1) Is enrolled in Medicare in an
approved status; or
(2) Has a valid opt-out affidavit on file
with an A/B MAC.
(iv) In order for a Part D sponsor to
submit to CMS a prescription drug event
(PDE) record, the PDE must contain an
active and valid individual prescriber
NPI and must pertain to a claim for a
Part D drug that was dispensed in
accordance with a prescription written
by a physician or, when permitted by
applicable State law, an eligible
professional (as defined in section
1848(k)(3)(B)(i) or (ii) of the Act) who:
(A) Is enrolled in Medicare in an
approved status, or
(B) Has a valid opt-out affidavit on file
with an A/B MAC.
We note that in our final version of
§ 423.120(c)(6)(iv), we have included
the language ‘‘must contain an active
and valid individual prescriber NPI.’’
This is not a new mandate, for a PDE
must currently have the required NPI
under § 423.120(c)(5)(i). We are simply
clarifying that this requirement
continues on and after June 1, 2015.
Again, these are merely technical
revisions. They do not involve any
changes to our proposed policies.
Comment: A commenter stated that
proposed § 423.120(c)(5) and (6) reflect
CMS’ continued efforts to protect the
Medicare program from inappropriate
payments for prescription drugs.
Response: We appreciate the
commenter’s support.
Comment: A commenter requested
that CMS furnish sub-regulatory
guidance concerning the following
issues related to § 423.120(c)(5) and (6):
(1) the pharmacy’s capability at point of
service (POS) to verify that the
prescriber’s NPI and Medicare
enrollment are valid; (2) whether plan
sponsors will be expected to deny at the
point of service if the beneficiary’s
prescriber has not completed either the
enrollment process or an opt-out
affidavit; (3) how CMS will disseminate
relevant information to plan sponsors
on a timely basis to enable sponsors to
set up point of service edits and prevent
negative beneficiary impacts; (4)
whether CMS will require sponsors to
allow pharmacies to override these
denials, similar to other Prescriber ID
edits; (5) which party (assuming CMS
requires sponsors to pay claims at point
of service and investigate post-claim
payment) will be financially responsible
when it is subsequently confirmed that
the prescriber is not enrolled or has not
validly opted-out; and (6) how CMS and
sponsors will ensure that beneficiaries’
access to needed Medicare-covered
drugs are not delayed or denied due to
this new process. Other commenters
requested clarification regarding
whether § 423.120(c)(5) and (6) establish
any new responsibilities for plan
sponsors or pharmacies.
Response: We anticipate
disseminating, as deemed necessary,
sub-regulatory or other guidance to
address the topics raised by the
commenter and any new requirements
for plan sponsors and pharmacies.
Furthermore, and as already stated, we
will regularly communicate with plan
sponsors, prescriber and supplier
associations, and beneficiary
organizations prior to the June 1, 2015
effective date to address their concerns.
Comment: A commenter expressed
concern that there would be a flood of
CMS–855 enrollment application forms
or opt-out affidavit submissions by
physicians and practitioners. The
commenter asserted that this could
cause application processing delays
and, consequently, the denial of claims
for drugs prescribed by practitioners
whose applications could not be
processed to completion before the
implementation date. Another
commenter requested information
regarding the process and timeline for
Medicare enrollment. Another
commenter suggested that CMS could
give a grace period to accept PDEs for
physicians and eligible professionals
who have applied for enrollment but are
still awaiting the outcome of their
application submission. Yet another
commenter stated that the large number
of revalidation applications being
submitted could delay the processing of
prescribers’ CMS–855 applications.
Response: We believe that our
extension of the effective date to June 1,
2015 will give physicians and eligible
professionals plenty of time to submit
their enrollment applications or opt-out
affidavits to their A/B MACs and to
have the latter process these materials to
completion before § 423.120(c)(6) is
implemented. Therefore, we do not
believe that the grace period suggested
by the third commenter is or will be
necessary. As we stated in the proposed
rule, we believe that the number of
prescribers who are neither Medicare-
enrolled nor have validly opted-out is
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00048 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29891
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
very low in any event, given that many
physicians and eligible professionals
furnish or order Part B services.
Nevertheless, we will monitor this
situation as June 1, 2015 approaches,
and will communicate with plan
sponsors, prescriber and supplier
organizations, and beneficiary advocacy
groups about progress in physician and
eligible professional enrollment in
Medicare pursuant to the requirements
of § 423.120(c)(6).
Information on the general provider
enrollment process and the timeframes
for application processing can be found
on CMS’ Web site at http://
www.cms.gov/Medicare/Provider-
Enrollment-and-Certification/
MedicareProviderSupEnroll/index.html.
Comment: Several commenters
questioned the accuracy of the
verification process, specifically as it
relates to PECOS. The commenters
stated that PECOS may not capture all
enrolled individuals and that the
information in the system may either be
inaccurate or inconsistent with the data
in NPPES. Another commenter
requested that CMS permit enrollment
via PECOS or a contractor’s legacy
system.
Response: We are continuously
enhancing PECOS and are confident
that all enrolled and opted-out
prescribers will be accurately reflected
in the system. In addition, all current
enrollments have been transitioned to
the PECOS system and all new
enrollments are directly entered into
PECOS.
Comment: A number of commenters
requested information about how plan
sponsors and pharmacies will be able to
determine that a prescription was
written by a prescriber who is enrolled
or has opted-out. One commenter
recommended that CMS clarify whether
the NPI would be used as the primary
identifier of whether a particular
physician or practitioner is enrolled.
Other commenters requested further
clarification regarding: (1) How our
proposal will be operationalized; (2)
whether the proposed list will include
all enrolled and opt-out prescribers and
will be sufficiently complete; (3)
whether or how often CMS will update
the list; (4) how plan sponsors will have
access to the file; (5) when CMS will
define the standard format; (6) whether
there will be start and end-dates in the
file; (7) whether there will be an
indicator for physicians who are in a
pended status; (8) the extent to which
NPPES will be used in prescriber
validation; (9) whether plan sponsors
will still be required to review the OIG/
System Access Management ((SAM);
formerly GSA) databases; (10) how
deceased prescribers and taxonomy data
will be handled; and (11) how plan
sponsors and pharmacists will identify
revoked or limited supplier statuses.
Response: As already indicated, we
will make available to plan sponsors
and pharmacies a complete list of
prescribers who are either enrolled in
Medicare or who have opted-out. The
list will be regularly updated. The NPI
will be one of several identifiers that
can or will be used. We will, as deemed
necessary, elaborate further on the
verification process, the specific
contents of the aforementioned list, the
specific frequency with which the list
will be updated, and various operational
aspects of our requirements via sub-
regulatory or other guidance.
Comment: One commenter
encouraged CMS to include a review of
the prescriber’s taxonomy code to
confirm prescribing authority as part of
the Medicare enrollment process for
physicians and other eligible
professionals.
Response: We appreciate this
suggestion and will take it under
advisement as we continue our efforts to
enhance the provider enrollment
process.
Comment: A commenter requested
clarification concerning whether an
individual who enrolls in Medicare
solely to prescribe Part D drugs will be
required to revalidate his or her
enrollment every 5 years per § 424.515.
Another commenter sought clarification
regarding whether enrollment pursuant
to § 423.120(c)(5) and (6) would subject
the enrollee to all of the enrollment
requirements outlined in §§ 424.500
through 424.570 (such as revalidation,
deactivation, retention of medical
documentation).
Response: We reserve the right to
apply applicable requirements in
§§ 424.500 through 424.570 to
individuals enrolled in Medicare solely
to prescribe Part D drugs. This would
include the requirement in § 424.515 to
revalidate one’s enrollment every 5
years.
Comment: A commenter requested
that CMS conduct a formal analysis to
determine the percentage of prescribers
with an active enrollment status by
comparing the prescriber NPIs
submitted on the PDEs to the Medicare
enrollment records. The commenter was
concerned that if the unenrolled
prescribers disproportionately reflect
certain supplier types or geographic
areas, this could cause disruptions. The
commenter also stated that CMS should
develop a process for allowing
prescribers who are authorized under
state law to prescribe but are not eligible
to be enrolled in Medicare to still
prescribe Part D drugs that would be
covered.
Response: Prior to the June 1, 2015
date, we will, as deemed necessary,
share information with plan sponsors
regarding the numbers and percentages
of prescribers who are enrolled in
Medicare. As for the final comment, the
prescriber must either opt-out of the
Medicare program or otherwise comply
with all Medicare enrollment
requirements. We cannot enroll a
prescriber who is ineligible to enroll in
Medicare regardless of the individual’s
status under state law, for we are bound
by our established enrollment
procedures. Consequently, we cannot
establish the exception process
envisioned by the commenter.
Comment: To limit POS denials that
could affect beneficiary access and
compromise patient care, a commenter
made several recommendations
regarding § 423.120(c)(5) and (6). First,
the prescriber enrollment files provided
by CMS should be the single and
authoritative source of prescriber
enrollment for all federal health care
programs. This would eliminate
duplication of effort, streamline the
enrollment process for prescribers,
ensure the consistent application of
CMS requirements, and eliminate the
need to review NPPES, the DHHS OIG
List, and the SAM. Second, a CMS and
industry task force should be developed
to establish data integrity criteria,
identify the minimum necessary data
elements, establish file dissemination
frequency to support real-time
validations, and ensure that appropriate
information is communicated to the
pharmacy and patient. Third, a process
should be developed to address changes
in a prescriber’s enrollment status (and
to notify beneficiaries of such changes)
after the most recent files have been
disseminated and before the next update
will be available. Fourth, there should
be changes to the PDE to support and
accept multiple Submission
Clarification Codes, as well as a process
for CMS to convey more accurate
information to the A/B MACs to update
their files. Fifth, a CMS call center
should be established to support
prescriber and beneficiary inquiries on
the prescriber’s enrollment status. Sixth,
there should be a CMS prescriber
outreach and education effort to
emphasize the importance of enrollment
and to address various prescriber
questions.
Response: We appreciate the
commenter’s suggestions and address
them as follows.
Regarding the first recommendation,
the aforementioned list will be the
authoritative list of prescribers who are
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00049 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29892
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
enrolled in Medicare or have opted-out.
However, it will not contain information
regarding said individuals’ enrollment
in other federal health care programs.
We do not believe such an all-
encompassing list is feasible at the
present time due to the differing
requirements and standards of these
various programs.
We will continue to work with the
health care industry to ensure that the
files CMS disseminates contain the
information necessary for plan sponsors,
pharmacies, and prescribers to enforce
and comply with all CMS requirements.
This will include appropriate updates to
reflect changes in a prescriber’s status,
as alluded to in the commenter’s third
suggestion.
We will consider making changes to
the PDE as deemed necessary to
facilitate the appropriate
implementation of and adherence to
§ 423.120(c)(6). We will also, as deemed
necessary, furnish guidance regarding:
(1) appropriate information for
prescribers and beneficiaries concerning
the enrollment status of prescribers; (2)
the importance of enrollment; and (3)
vehicles for addressing prescriber
inquiries.
Comment: A commenter
recommended that in order to stop fraud
on a prepayment basis and to ensure
that Medicare beneficiaries are
protected from physicians and eligible
professionals who prescribe controlled
substances without a valid DEA
registration number, CMS should revise
§ 423.120(c)(6) to require Part D plan
sponsors to make payments to a
pharmacy or Medicare beneficiary when
a Part D controlled substance is
prescribed by a physician or eligible
professional who has a valid and active
DEA registration number.
Response: We do not believe this
revision is necessary, for we will be able
to revoke an individual’s ability to
prescribe such drugs under
§ 424.535(a)(13) (as explained in more
detail later in this section). We believe
that § 423.120(c)(6) as currently crafted
(aside from the effective date) will
achieve our goal of ensuring that only
qualified physicians and eligible
professionals can prescribe Part D drugs.
We further note that having a DEA
certificate does not necessarily mean
that a prescriber is in compliance with
all Medicare requirements.
Comment: A commenter requested
clarification regarding whether, if a
claim is rejected at the POS, a plan will
be required to provide beneficiaries
with a list of prescribers that are
enrolled in the Medicare program.
Response: No. This will not be
required.
Comment: A commenter stated that
§ 423.120(c)(5) and (6) do not take into
account the thousands of patients
currently serviced by interns and
residents who are yet to be licensed but
are authorized by state governments to
examine, treat and prescribe for their
patients provided they function under
the supervision of an attending
physician. The commenter sought
clarification concerning whether these
as yet unenrolled individuals would be
able to order prescriptions. Another
commenter requested that CMS furnish
guidance: (1) On how situations in
which a resident’s enrollment status has
changed should be handled; and (2) for
teaching hospitals regarding the
importance of ensuring that residents
comply with Medicare enrollment rules
(including updating enrollment data as
needed).
Response: Section 423.120(c)(6) does
not prohibit interns and residents from
prescribing Part D drugs to the extent
that these individuals are otherwise
qualified to prescribe such drugs under
applicable law and regulations and to
either enroll in Medicare or validly opt-
out of the program.
We will, as deemed necessary, issue
guidance concerning the importance of
complying with Medicare enrollment
rules.
Comment: A number of commenters
requested additional clarification
regarding the number of physicians who
are not enrolled in Medicare.
Response: Although a sizable majority
of physicians nationwide are enrolled in
Medicare, we do not have a precise
number.
Comment: Several commenters
recommended that CMS make available
for Medicare Advantage plans and other
stakeholders access to national, real-
time data—preferably in a single file—
to use in identifying excluded, non-
enrolled, and opt-out suppliers. One
commenter added that any opt-out file
should have the physician’s NPI and
specialty, as well as the expiration date
of his or her opt-out agreement.
Response: The file alluded to earlier
that will be distributed to plan sponsors
will be updated regularly. Specific
information regarding the frequency of
the updates and the contents of the file
will, as deemed necessary, be
disseminated via sub-regulatory or other
guidance.
Comment: A commenter questioned
whether there is evidence of higher
levels of fraud, waste, or abuse by
suppliers who are not enrolled in the
Medicare program versus those who are
enrolled, and whether increasing the
number of enrolled suppliers per
§§ 423.120(c)(5) and (6) will provide an
avenue for unscrupulous but unenrolled
prescribers to defraud Medicare.
Response: The enrollment process, as
explained earlier, is designed to ensure
that we can verify that a supplier meets
all CMS requirements, such as
licensure. Without this process,
unqualified and fraudulent suppliers
would be able to enter Medicare and bill
the program, resulting in billions of
dollars being improperly paid to such
individuals and organizations. We
maintain that CMS’ enrollment process
reduces the amount of potential fraud,
waste and abuse. Furthermore, we do
not see how § 423.120(c)(6) will provide
an avenue for unscrupulous persons to
defraud Medicare. To the contrary, it
will protect the Medicare program by
ensuring that only qualified and
legitimate individuals can prescribe Part
D drugs.
Comment: A commenter disagreed
with CMS’ proposal to allow a
physician who has opted-out of the
Medicare program to prescribe Part D
drugs to Medicare beneficiaries. The
commenter stated that CMS does not
have the legal authority to revoke the
prescribing privileges of a physician or
eligible professional who has been
convicted of health care fraud but is in
an opt-out status or is practicing via
private contract.
Response: Section 1802(b) of the Act
is clear that certain physicians and
practitioners may opt-out of the
Medicare program and enter into private
contracts with Medicare beneficiaries.
We believe that to require such
individuals to enroll in Medicare would
be inconsistent with this statutory
provision.
Comment: A commenter suggested
that CMS purge all opt-out affidavits if
they are more than 2 years old and
establish a systematic process to purge
all opt-out affidavits on a regular basis.
Response: We appreciate this
suggestion and will take it under
advisement as we continue our efforts to
enhance the integrity of the Medicare
program.
Comment: Several commenters stated
that numerous other federal
requirements (for example, DEA
certificate) and state regulations (for
example, state medical licensing boards)
already exist to ensure that medications
are only prescribed by qualified
individuals. Rather than implement
another bureaucratic hurdle, the
commenters contended that these other
federal and state regulations should be
tightened as needed. One commenter
stated that because there are multiple
safeguards currently in place through
the OIG, there is no need for
§ 423.120(c)(5) and (6). Other
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00050 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29893
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
commenters stated that § 423.120(c)(5)
and (6) are unnecessary because (i) Part
D sponsors are already required to
review NPPES to verify a prescriber’s
NPI and other data; (ii) states already
license and regulate prescribers; and
(iii) pharmacists are responsible for
determining that prescriptions are
written by licensed individuals.
Response: We disagree with these
commenters. Data lists that are
prepared, administered and updated by
agencies outside of CMS frequently do
not capture the information we need to
confirm that a supplier meets Medicare
requirements. The CMS enrollment
process is the most practical, thorough,
and effective means of securing and
verifying all necessary information on
physicians and eligible professionals.
Comment: Several commenters
expressed support for our proposed
provisions but sought assurances that
plans would not be penalized for filling
prescriptions if, at the time the drug was
dispensed, the plan did not know of the
prescriber’s termination. Another
commenter did not believe there should
be retroactive enrollment terminations;
this would eliminate recoupment of
payment from pharmacies or Part D
sponsors for prescribers who were
shown as enrolled by the most current
information available at the time the
prescription was filled. Another
commenter requested clarification as to
whether there would be performance-
score safeguards established for plans
that appropriately deny drugs based on
the information available to them
through the MACs or other parties
responsible for maintaining said list.
Another commenter expressed concern
about the impact that these
requirements would have on plan
performance measures due to an
increased number of complaints from
beneficiaries relating to prescriptions
that could not be filled, or with respect
to which payment would be denied.
Response: It is important to note that
our requirements are directed
specifically at individuals who
prescribe Part D drugs. Individuals who
prescribe are required to enroll in
Medicare (or validly opt-out of
Medicare) in order to do so. As such,
plan sponsors would be required to pay
only for those prescriptions written by
physicians or eligible professionals
who, according to CMS, are enrolled in
Medicare in an approved status or who
have validly opted-out of Medicare. We
will, as deemed necessary, further
address these issues via sub-regulatory
or other guidance.
Comment: Several commenters
believed that the administrative burden
of these provisions would outweigh any
potential benefits in deterring fraud,
waste and abuse; this would be
especially true for plan sponsors that
would have to verify a particular
prescriber’s enrollment or opt-out
status. The commenters requested that
CMS more closely study the potential
administrative impact of these
provisions.
Response: We have studied the
impact of these provisions and believe
that the benefits to Medicare
beneficiaries, the Medicare Trust Funds,
and the program as a whole of
confirming that physicians and eligible
professionals are qualified to prescribe
Part D drugs far outweigh the burden to
prescribers of completing the
enrollment process or submitting an
opt-out affidavit. Besides, as mentioned
in the proposed rule, a large majority of
physicians and eligible professionals
who prescribe Part D drugs are already
enrolled in Medicare; hence, our
provisions will have no impact on these
individuals. Furthermore, those who are
impacted will have ample time to
complete the enrollment or opt-out
process due to the extension of the
compliance date to June 1, 2015.
Comment: A commenter suggested
that CMS issue warnings to prescribers
for a 6 to 12-month period prior to
rejecting claims that fail to meet the
necessary criteria.
Response: We appreciate this
suggestion. We are exploring various
means of alerting prescribers who are
neither enrolled in Medicare nor have
submitted a valid opt-out affidavit of the
need to comply with the requirements
of § 423.120(c)(6).
Comment: A commenter suggested
that CMS consider using technology that
already exists within the pharmacy
industry for validating prescriber data,
for this would (when compared to the
batch processes): (1) Improve patient
access to care as the most timely data is
made available at the time of
prescription drug dispensing; (2)
decrease costs associated with audits
and recovery of funds resulting from
out-of-date data; and (3) increase
consistency of data among the multiple
MACs and pharmacies. Another
commenter stated that CMS should
avoid using a PDF file similar to that
which exists for the current ordering/
certifying edits and instead create a
database containing this information.
Response: We are contemplating
various formats in which the
previously-discussed list might be
disseminated to plan sponsors.
Comment: A commenter requested
clarification as to: (1) Whether CMS is
proposing a new provider enrollment
process for Part D in addition to the
current enrollment process for obtaining
Medicare billing privileges; and (2) how
a Part D revocation would impact Part
B billing by the same practitioner.
Response: The provider enrollment
process under § 423.120(c)(6) will be the
same as that which is used for
physicians and eligible professionals
enrolling in Medicare in order to
comply with § 424.507. A revocation
under § 424.535(a) would eliminate the
individual’s ability to prescribe covered
Part D drugs because he or she would
no longer be enrolled in Medicare;
hence, the requirements of
§ 423.120(c)(6) would no longer be met.
Comment: Several commenters
requested that CMS exclude dentists
from proposed § 423.120(c)(5) and (6)’s
application because the provisions
would place an unnecessary burden on
dentists and their Medicare-eligible
patients, and would not address CMS’
desire to stop fraud and abuse. One
commenter added that it is unaware of
high-billing levels associated with
prescriptions written by dentists for
Medicare-eligible patients, yet the
administrative burden on dentists
would be significant. Another
commenter expressed concern that the
proposal could negatively impact plan
members, in that members who receive
prescriptions written by dentists not
enrolled in the program would be
financially responsible for such
prescriptions because they would no
longer be covered. Another commenter
noted that Medicare beneficiaries
enrolled in dual eligible SNPs may
receive comprehensive dental benefits,
including certain invasive procedures.
Dentists may prescribe antibiotics in
these circumstances, and these drugs
should be covered under Medicare Part
D. However, since dentists are not
typically enrolled in Medicare, our
proposal could interfere with this
coverage. Other commenters
recommended that CMS exclude from
§ 423.120(c)(5) and (6)’s purview those
suppliers who do not normally see
Medicare beneficiaries or receive
Medicare payment (including
psychiatrists and Veterans’
Administration (VA) doctors) and
enable them to (after a grace period)
register with Medicare in a limited
capacity to enable them to write
prescriptions for Medicare beneficiaries.
Response: While we recognize the
concerns of these commenters, we do
not believe dentists, psychiatrists, VA
physicians, or any other physicians or
eligible professionals should be granted
special exemptions from § 423.120(c)(6).
The issue of primary concern to us is
not the typical volume of drugs these
individuals prescribe but the need to
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00051 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29894
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
ensure and confirm that Medicare
payments are only made for Part D
drugs that are prescribed by qualified
physicians and eligible professionals.
This is precisely the concern that the
OIG expressed in its previously-
referenced report. Moreover, we believe
that our extension of the effective date
to June 1, 2015 will afford these
individuals more than adequate time to
complete the enrollment or opt-out
process, hence easing the burden on
them.
Comment: One commenter: (1)
Favored requiring dentists to enroll in
Medicare (or have a valid opt-out
affidavit on file) in order to prescribe
Part D drugs; and (2) believed that a
January 1, 2015 effective date was
reasonable.
Response: We agree with this
commenter’s first comment and intend
to apply § 423.120(c)(6) to dentists.
While we appreciate the commenter’s
second comment, we believe that a June
1, 2015 effective date is more
appropriate.
Comment: A commenter requested
clarification concerning how these
provisions would be enforced in cases
of out-of-network benefits, which permit
plan enrollees to receive healthcare
items and services (including
prescription medicines) across the
country. Another commenter stated that
if CMS allows point of service overrides,
the Prescription Drug Events (PDEs)
should be accepted and final, with no
requirement for plans/sponsors to
provide a retroactive look back. Other
commenters suggested that CMS should:
(1) Require plans to hold beneficiaries
harmless from the consequences of non-
coverage for a non-compliant supplier
for at least one fill of the prescription;
(2) require plans to reach out to the
beneficiary and the supplier to explain
the issue, allowing sufficient time for
the beneficiary to see another supplier
or for the supplier to correct his or her
enrollment status; and (3) reach out to
policy makers in the states that permit
foreign prescriptions, to determine what
kind of alternate supplier credential
checking might be available to ensure
that beneficiaries who spend portions of
the year in other countries can access
their medications without interruption
or the unneeded expense of additional
physician visits.
Response: We will, as deemed
necessary, address these matters via
sub-regulatory guidance or future
rulemaking.
Comment: A commenter requested
clarification as to whether
§ 423.120(c)(6) applies even if a
physician or eligible professional is
state-licensed but is neither Medicare-
enrolled nor has opted-out.
Response: Yes, it applies.
Comment: A commenter requested
information as to the following: (1)
Whether plan sponsors would remain
responsible for ensuring that a
prescriber is properly enrolled in
Medicare; (2) whether prescriber
validation should occur at the point-of-
sale and whether plan sponsors are not
permitted to ‘‘flow down’’ the
responsibility for this verification
process to their network pharmacies;
and (3) whether CMS could prohibit
Part D plans from reversing pharmacy
claims with prescriber verification
errors found in audits if the prescriber
enrollment verification found by that
plan was later found to be inaccurate.
Response: We will, as deemed
necessary, address these matters via
sub-regulatory guidance or future
rulemaking.
Comment: A commenter stated that
because the vast majority of prescribing
physicians and other practitioners are
already enrolled as Medicare suppliers,
§§ 423.120(c)(5) and (6) should not
impose a great burden on prescribers.
However, the commenter encouraged
CMS to make any requirements for
beneficiary requests for reimbursement
from Part D sponsors as clear and
concise as possible for beneficiaries.
Prescribers should be able to quickly
generate forms for patients who want to
submit them to their plan sponsors
directly.
Response: We agree with the
commenter’s first statement, and will
attempt to ensure that beneficiaries
understand the requirements for
requesting reimbursement.
Comment: A commenter urged CMS
to require plans to cover the costs
associated with the charge-back if there
is an error in the claim related to
Medicare enrollment, and that the cost
for verification and correction of any
claims be borne by the plan through
their administrative costs.
Response: We are not prepared in this
final rule to issue a definitive statement
regarding costs associated with charge-
backs. Any such statement will, as
deemed necessary, be addressed via
sub-regulatory or other guidance.
Comment: A commenter urged CMS
to explore options to reduce member
disruptions and to allow plans to
manage prescribers not meeting these
requirements. Such options could
include: (1) Allowing a period of ‘‘soft
edits’’ to effectively track and manage
potential future disruptions; (2)
applying our requirements only to new
fills; or (3) allowing prescriptions to be
grandfathered up to a year after the
effective date.
Response: We believe that our
extension of the effective date to June 1,
2015, as well as CMS’ outreach efforts,
will greatly reduce the potential for
coverage disruptions. However, we will
monitor the progress of the
implementation of § 423.120(c)(6) to
ensure that such disruptions do not
occur.
Comment: A commenter stated that
the proposed rule did not address how
Part D beneficiaries in the U.S.
territories would be impacted by
proposed § 423.120(c)(5) and (6).
Response: We anticipate conducting
outreach, as needed, for beneficiaries in
U.S. territories regarding how they may
be affected by these provisions.
Comment: A commenter expressed
concern that CMS had proposed to no
longer allow Part D coverage for foreign
prescriptions.
Response: We did not propose to deny
coverage for foreign prescriptions. We
simply proposed to require that all
prescribers of Part D drugs be enrolled
in Medicare or in a valid opt-out status.
We may, as deemed necessary, further
address this issue via sub-regulatory
guidance.
Comment: Several commenters
requested clarification concerning
whether plan sponsors would be able to
accept a pharmacy claim for an
automatically-generated refill
prescription if the prescriber is not
enrolled in Medicare. The commenters
also recommended that § 423.120(c)(5)
and (6) only be applied to new
prescriptions.
Response: The pharmacy claims
described by the commenters will not be
covered if the prescriber is not enrolled
in Medicare and does not have a valid
opt-out affidavit on file with an A/B
MAC, regardless of whether the
prescription is new or a refill.
Comment: A number of commenters
opposed the notion of requiring
pharmacies to enroll in Medicare in
order to distribute Part D drugs. They
expressed concern about the burden and
cost involved for pharmacies, and the
potential disruption to the Part D
program that would result if thousands
of pharmacies were required to enroll.
One commenter stated that Part D
sponsors or their PBMs have direct
contractual relationships with
pharmacies and perform their own
credentialing and verifications before
allowing pharmacies into their
networks; sponsors have the necessary
experience and expertise to identify and
remove unlicensed, fraudulent or
otherwise unqualified pharmacies from
their networks.
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00052 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29895
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
Response: Because we concur with
these contentions, we do not intend to
apply § 423.120(c)(6) to pharmacies at
this time.
Comment: A commenter requested
clarification concerning whether the
pharmacy requirement for enrollment
refers to Part B DMEPOS supplier
enrollment for drugs.
Response: Our earlier reference to
pharmacy enrollment pertains to Part D
drugs. However, as stated previously,
we are not applying § 423.120(c)(6) to
pharmacies at this time.
Comment: One commenter supported
the notion of requiring pharmacy
enrollment.
Response: We appreciate this
comment. However, as already stated,
we do not intend to apply
§ 423.120(c)(6) to pharmacies at this
time.
Given this, we are finalizing our
proposed provisions in § 423.120(c)
with several exceptions. First, the
January 1, 2015 effective date is changed
to June 1, 2015. Second, the existing
version of paragraph (c)(5) will remain
intact with the exception of the addition
of the ‘‘Before June 1, 2015, the
following are applicable’’ language at
the very beginning of the paragraph.
Third, we are not finalizing our
proposed changes to paragraph (c)(5)(ii),
but are instead merging them with our
addition of paragraph (a)(6). Our final
version of new paragraph (c)(6) will
thus read as follows:
‘‘(6) Beginning June 1, 2015, the
following are applicable—
(i) A Part D sponsor must deny, or
must require its pharmaceutical benefit
manager (PBM) to deny, a pharmacy
claim for a Part D drug if an active and
valid physician or eligible professional
(as defined in section 1848(k)(3)(B)(i) or
(ii) of the Act) National Provider
Identifier (NPI) is not contained on the
claim.
(ii) A Part D sponsor must deny, or
must require its PBM to deny, a
pharmacy claim for a Part D drug if the
physician or eligible professional (when
permitted to write prescriptions by
applicable State law)—
(A) Is not enrolled in the Medicare
program in an approved status; and
(B) Does not have a valid opt-out
affidavit on file with an A/B Medicare
Administrative Contractor (MAC).
(iii) A Part D sponsor must deny, or
must require its PBM to deny, a request
for reimbursement from a Medicare
beneficiary for a drug if the request is
not for a Part D drug that was dispensed
in accordance with a prescription
written by a physician or, when
permitted by applicable State law, other
eligible professional (as defined in
section 1848(k)(3)(B)(i) or (ii) of the Act)
who—
(A) Is identified by his or her legal
name in the request; and
(B)(1) Is enrolled in Medicare in an
approved status; or
(2) Has a valid opt-out affidavit on file
with an A/B MAC.
(iv) In order for a Part D sponsor to
submit to CMS a prescription drug event
(PDE) record, the PDE must contain an
active and valid individual prescriber
NPI and must pertain to a claim for a
Part D drug that was dispensed in
accordance with a prescription written
by a physician or, when permitted by
applicable State law, an eligible
professional (as defined in section
1848(k)(3)(B)(i) or (ii) of the Act) who:
(A) Is enrolled in Medicare in an
approved status, or
(B) Has a valid opt-out affidavit on file
with an A/B MAC.
These revisions to our proposed
paragraph (c)(6) do not involve any
changes from our proposed policy. They
are merely technical changes designed
to better fit the existing regulatory text.
21. Improper Prescribing Practices
(§§ 424.530 and 424.535)
a. Background and Program Integrity
Concerns
We stated in the preamble to the
proposed rule that notwithstanding our
proposed provisions in § 423.120(c),
additional program safeguard
enhancements were necessary to protect
the Medicare Trust Funds from fraud,
waste and abuse, and to ensure that Part
D drugs are prescribed only by qualified
suppliers. Along with the
aforementioned OIG report (‘‘Medicare
Inappropriately Paid for Drugs Ordered
by Individuals Without Prescribing
Authority’’ (OEI–02–09–00608)), we
cited another OIG report titled,
‘‘Prescribers with Questionable Patterns
in Medicare Part D’’ (OEI–02–09–
00603). This report highlighted a
number of instances in which
physicians and eligible professionals
prescribed inordinate amounts of drugs
to Part D beneficiaries in 2009. For
example—
• Medicare paid a total of $9.7
million—151 times more than the
average—for one California physician’s
prescriptions; most of this physician’s
prescriptions were filled by two
independent pharmacies, both of which
the OIG had identified as having
questionable billing;
• One hundred and eight general-care
physicians each ordered an average of
71 or more prescriptions per
beneficiary, more than 5 times general-
care physicians’ national average of 13;
• An Ohio physician ordered more
than 400 drugs each for 13 of his 665
beneficiaries; and
• A Texas physician ordered more
than 400 prescriptions each for 16
beneficiaries and prescribed 700 or
more drugs for 3 of these beneficiaries.
The OIG also noted examples of
physicians prescribing a high
percentage of Schedule II and III drugs
in 2009. In one case, 78 percent of the
prescriptions a Florida physician
ordered were for Schedule II drugs even
though the OIG found that 4 percent of
the prescriptions ordered by prescribers
nationwide were for Schedule II drugs.
For one beneficiary, the physician
prescribed a 605-day supply of
morphine sulfate, a 524-day supply of
oxycodone HCl, a 460-day supply of
fentanyl, and a 347-day supply of
hydromophone HCl.
The OIG has recommended that CMS
exercise greater oversight of the Part D
program, not only to curb the specific
practices outlined previously but also to
stem the overall risk of fraud and abuse
that the program presents. The OIG has
expressed particular concern over the
potential for beneficiaries to become
addicted to or otherwise be seriously
harmed by certain drugs if they are
inappropriately prescribed in
dangerously excessive amounts. We
share this concern, particularly as we
continue to receive reports of improper
prescribing practices. The difficulty, as
we explained in the proposed rule, is
that CMS does not possess the legal
authority to take administrative action
against the prescriber. This means, in
many cases, that the individual can
continue prescribing drugs that will be
covered under Part D and, if he or she
is enrolled in Medicare, remain so
enrolled to furnish medical services. We
believe this is inconsistent with: (1) The
OIG’s recommendations in its various
Part D reports; and (2) our goals of
protecting and promoting the health and
safety of Medicare beneficiaries and of
safeguarding the Medicare Trust Funds.
To this end, and as we explain in this
section, we proposed several changes to
Part 424, subpart P.
b. Drug Enforcement Administration
(DEA) Certification of Registration
The DEA implements and enforces
Titles II and III of the Comprehensive
Drug Abuse Prevention and Control Act
of 1970, and the Controlled Substances
Import and Export Act, as amended, and
collectively referred to as the Controlled
Substances Act (CSA) (21 U.S.C. 801–
971); the implementing regulations for
these statutes are in 21 CFR Parts 1300
through 1321. The CSA makes
possession of authority under state law
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00053 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29896
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
to dispense controlled substances a
requirement for both obtaining and
maintaining a DEA Certificate of
Registration.
We view a DEA Certificate of
Registration to prescribe controlled
substances as similar to a state’s
requirement that a physician or eligible
professional be licensed or certified by
the state to furnish health care services.
Indeed, we are concerned that a
physician or eligible professional’s
improper prescribing practices may be
duplicated in the Medicare program. To
address these issues, we proposed the
following:
• Adding a new § 424.530(a)(11)
granting CMS the authority to deny a
physician or eligible professional’s
Medicare enrollment application if: (1)
His or her DEA Certificate is currently
suspended or revoked; or (2) the
applicable licensing or administrative
body for any state in which the
physician or eligible professional
practices has suspended or revoked the
physician or eligible professional’s
ability to prescribe drugs, and such
suspension or revocation is in effect on
the date he or she submits his or her
enrollment application to the Medicare
contractor.
• Adding a new § 424.535(a)(13)
granting CMS the authority to revoke a
physician or eligible professional’s
Medicare enrollment if: (1) His or her
DEA Certificate is suspended or
revoked; or (2) the applicable licensing
or administrative body for any state in
which the physician or eligible
professional practices suspends or
revokes his or her ability to prescribe
drugs. Again, this approach is consistent
with our requirement that suppliers
maintain compliance with all applicable
licensure and certification requirements.
(We also solicited comments on
whether our proposed additions of
§§ 424.530(a)(11) and 424.535(a)(13)
should be expanded to include
pharmacy activities.)
We believe that the loss of the ability
to prescribe drugs via a suspension or
revocation of a DEA Certificate or by
state action is a clear indicator that a
physician or eligible professional may
be misusing or abusing his or her
authority to prescribe such substances.
We also believe that our proposed
provisions were consistent with the
OIG’s recommendations and, equally
important, are necessary to protect
Medicare beneficiaries and the Trust
Funds.
We received a number of comments
related to our proposal. Summaries of
the comments and our responses are as
follows:
Comment: Several commenters
expressed support for §§ 424.530(a)(11)
and 424.535(a)(13), stating that these
provisions would help reduce abusive
prescribing.
Response: We appreciate the support
of these commenters.
Comment: Various commenters
recommended that CMS: (1) Verify a
DEA registration number submitted on
the CMS–855I or the CMS–855O with
the DEA prior to enrolling a physician
or eligible professional into Medicare;
(2) require physicians and eligible
professionals to report a change
(voluntary termination, revocation,
suspension) in their DEA registration
number within 30 days of the change;
(3) modify the CMS–855I and CMS–
855O to require that physicians and
eligible professionals report a DEA
registration number suspension or
revocation within 30 days; (4) require
that a physician or eligible professional
have a DEA number for each state in
which the physician or eligible
professional is prescribing controlled
substances; (5) require its Part D
sponsors to establish the necessary edits
to deny a prescription for a controlled
substance when the physician or
eligible professional does not maintain
a validly issued and active DEA
registration number in the state where
the prescription was written; (6) refer to
the DEA the name and NPI of any
physician or eligible professional who is
enrolled in Medicare in multiple states
and who is only using a single DEA
registration number to prescribe
controlled substances to Medicare
beneficiaries; and (7) establish a data
matching agreement with the DEA to
verify the DEA registration numbers
assigned by the DEA for all physicians
and eligible professionals enrolled in
Medicare. Another commenter
suggested that CMS establish a 3-year
reenrollment bar under § 424.535(c) for
any physician or eligible practitioner
who is revoked pursuant to
§ 424.535(a)(13), or at least identify in
the final rule what the reenrollment bar
length will be. The commenter also
recommended that the reenrollment bar
apply to Medicare Advantage
Organizations, not simply the Part B
Medicare program and Part D drugs.
Response: We appreciate these
suggestions and will take them into
consideration as part of our ongoing
efforts to strengthen payment safeguards
in the Medicare program.
Comment: Several commenters
recommended that CMS allow
physicians and eligible professionals to
self-report a DEA license revocation or
suspension (or a state licensing body
revocation or suspension associated
with prescribing drugs) within 30 days
of the revocation, suspension, or
voluntary surrender of their DEA
registration.
Response: We do not believe that a
physician or eligible professional
should be permitted to evade
§ 424.535(a)(13) and the subsequent
reenrollment bar merely by reporting
the DEA certificate suspension or
revocation to CMS. The issues of
concern to us are the certificate
revocation or suspension itself and the
consequent need to protect Medicare
beneficiaries and the Trust Funds, and
not so much the physician or eligible
professional’s voluntary revelation of
the revocation or suspension.
Comment: A commenter requested
that CMS furnish two lists to Part D
sponsors: (1) A list of physicians and
eligible professionals who have a DEA
registration number that CMS has
confirmed with the DEA; and (2) a list
of physicians and eligible professionals
who do not have a valid and active DEA
registration number. The data on these
lists, the commenter suggested, could be
broken down by state.
Response: We appreciate this
suggestion and will take it under
advisement as we continue our efforts to
strengthen the integrity of the Part D
program.
Comment: Several commenters
requested clarification concerning
whether CMS intends to implement
§ 424.535(a)(13) retrospectively and
revoke the Medicare billing privileges of
physicians and eligible professionals
who have had their DEA number
suspended or revoked. One commenter
opposed a retroactive application of our
proposal.
Response: We retain the discretion to
revoke the billing privileges of an
enrolled physician or eligible
professional whose DEA certificate is
suspended or revoked at the time
§ 424.535(a)(13) becomes effective.
Comment: A commenter requested
CMS’ rationale for permitting an
individual to enroll in Medicare after
the DEA has: (1) Denied him or her a
DEA certificate of registration; or (2)
suspended or revoked a DEA
registration number and the suspension
or revocation is still in force.
Response: In the commenter’s second
scenario, we would be able to deny the
individual’s enrollment under
§ 424.530(a)(11). As for the first
scenario, our focus in preparing our
proposed rule was on individuals who
had active DEA certificate suspensions
or revocations. We nonetheless
appreciate the commenter’s apparent
suggestion and may consider addressing
it in future rulemaking.
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00054 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29897
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
Comment: Several commenters
recommended that § 424.535(a)(13) not
be applied in cases where a physician’s
DEA number was suspended due to
substance abuse issues and the
physician is in counseling.
Response: We do not believe that a
blanket exemption from
§ 424.535(a)(13)’s potential application
for such individuals is warranted or
justified. However, we note that
§ 424.535(a)(13), like most other
revocation reasons in § 424.535, is
discretionary, meaning that CMS is not
required to exercise its revocation
authority. Although we have the
discretion to invoke § 424.535(a)(13)
regardless of the grounds for the DEA
certificate revocation or suspension, we
would also be able to take into account
the circumstances surrounding the
suspension or revocation prior to
making a final determination.
Comment: Several commenters
requested clarification as to whether: (1)
Proposed § 424.535(a)(13) applies to
non-controlled substances; and (2)
whether a voluntary surrender of a DEA
certificate (for instance, a semi-retired
physician wishes to prescribe only non-
controlled substances) would invoke
§ 424.535(a)(13). The commenters
believed that non-controlled substances
should be excluded from
§ 424.535(a)(13)’s purview if the
prescriber otherwise maintains the legal
authority to prescribe such drugs, is in
good standing with a state professional
licensing board, and has not engaged in
abusive prescribing. At a minimum, one
commenter suggested, CMS should refer
a potential case to the state for review
prior to making a decision.
Response: We explained in the
proposed rule that a DEA certificate of
registration is not required to dispense
non-controlled substances. Thus, if
one’s DEA certificate is suspended or
revoked, he or she would still be able
to prescribe non-controlled substances
absent some other restrictive action
taken by the DEA or the state (although
his or her billing privileges could still
be revoked under § 424.535(a)(13)). Yet
we note that § 424.535(a)(13) can be
invoked if the applicable licensing or
administrative body for any state in
which the individual practices suspends
or revokes his or her ability to prescribe
drugs. Therefore, if the state rescinds
the person’s ability to prescribe any
drugs, the individual (should
§ 424.535(a)(13) be invoked) would be
prohibited from prescribing Part D
controlled and non-controlled drugs.
The voluntary surrender of a DEA
certificate would not constitute grounds
for revocation under § 424.535(a)(13).
The provision as written is limited to
certificate revocations and suspensions.
However, we may consider addressing
this issue via future rulemaking.
Comment: Several commenters
recommended that CMS: (1) Explain
how it will obtain information from the
DEA regarding registration numbers that
are valid, approved, revoked,
suspended, voluntarily surrendered,
etc.; (2) make available to Part D
sponsors the information necessary to
deny a Part D claim for controlled
substances when a physician or eligible
professional does not have a valid and
active DEA registration number in the
state in which the prescription is
written; and (3) explain whether this
data will be in the file that is to be used
for the enforcement of §§ 423.120(c)(5)
and (6).
Response: We will, as deemed
necessary, address these issues via sub-
regulatory or other guidance.
Comment: Several commenters
requested clarification concerning
whether a physician would be able to
reenroll in Medicare after the
suspension or revocation of his or her
DEA registration is lifted.
Response: If we revoke a physician’s
billing privileges under § 424.535(a)(13),
the physician would be able to submit
a CMS–855 application for enrollment
upon the expiration of his or her
reenrollment bar.
Comment: A commenter
recommended that CMS clarify whether
physicians and eligible professionals
have 30 days to report a DEA
registration number revocation per
§ 424.516(d).
Response: The individual would be
required to report this information to
CMS under § 424.516(d) to the extent
the CMS–855 mandates that such
information be disclosed on the
application.
Comment: A commenter suggested
that CMS revise and update item B1 in
section 3 of the CMS–855I and the
CMS–855O, which states ‘‘Any
revocation or suspension of a license to
provide health care by any state
licensing authority; this includes the
surrender of such a license while a
formal disciplinary proceeding was
pending before a state licensing
authority,’’ to read as follows: ‘‘Any
revocation or suspension of a license to
provide health care by any state
licensing authority or Drug Enforcement
Administration Registration number.
This includes the surrender of such a
license while a formal disciplinary
proceeding was pending before a state
licensing authority.’’ The commenter
also sought clarification regarding
whether CMS will indeed treat a DEA
registration number denial or revocation
as a final adverse legal action.
Response: We appreciate this
suggestion and will take it under
advisement as we continue our efforts to
strengthen the integrity of the Part D
and Part B programs.
At this stage, CMS does not have the
legal authority to treat a DEA certificate
revocation or suspension as a final
adverse action because the current
definition of the latter term in § 424.502
does not specifically include DEA
actions. However, we may address this
issue through future rulemaking.
Comment: A commenter supported
our proposal to require Part D
physicians and eligible professionals
who prescribe controlled substances to
obtain and maintain a valid DEA
certificate of registration as a condition
of enrollment. Yet the commenter
recommended that the provision apply
only to those individuals who prescribe
controlled substances; this would avoid
impacting the ability of practitioners
providing services solely in local public
health departments to prescribe non-
controlled medications.
Response: As stated previously, if
one’s DEA certificate is suspended or
revoked, he or she would still be able
to prescribe non-controlled substances
absent some other restrictive action
taken by the DEA or a state (although
his or her billing privileges could still
be revoked under § 424.535(a)(13)).
However, if the state in which the
individual practices suspends or
revokes his or her ability to prescribe
any drugs, the individual (should
§ 424.535(a)(13) be invoked) would be
prohibited from prescribing Part D
controlled and non-controlled drugs.
Comment: Several commenters
opposed our proposed addition of
§ 424.535(a)(13), stating that a
suspended DEA certificate or state
license does not necessarily reflect one’s
inability to treat Medicare patients
safely and at a high standard. This is
particularly true, one commenter
contended, considering that many DEA
certificate or licensure revocations,
suspensions, or restrictions are due to
the physician or practitioner’s medical
illness, usually drug abuse and
dependence. Such individuals generally
complete treatment programs
successfully and should be given a
second chance. At a minimum, the
commenter maintained, CMS should
take into account such situations in
determining whether to invoke
§ 424.535(a)(13).
Response: As explained earlier,
§ 424.535(a)(13) is a discretionary
authority, and CMS can use its
discretion to take into account the
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00055 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29898
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
individual’s particular circumstances in
determining whether a revocation is
warranted. But we caution that we are
not required to do so, and there may be
instances in which we decide that the
certificate revocation or suspension
alone, on its face, is sufficient to justify
invoking § 424.535(a)(13).
For the reasons stated in this section,
we are finalizing our proposed additions
of §§ 424.530(a)(11) and 424.535(a)(13).
c. Patterns or Practices of Prescribing
We also proposed to add a new
§ 424.535(a)(14) that would permit CMS
to revoke a physician or eligible
professional’s Medicare enrollment if
CMS determines that he or she has a
pattern or practice of prescribing Part D
drugs that—
• Is abusive and represents a threat to
the health and safety of Medicare
beneficiaries; or
• Fails to meet Medicare
requirements.
We chose not to define ‘‘abusive’’ and
‘‘threat to the health and safety of
Medicare beneficiaries’’ in the proposed
rule, primarily because the myriad of
questionable situations that could
warrant the possible application of
§ 424.535(a)(14) requires that CMS have
the flexibility to address each case on its
own merits. We believed that the
sounder approach was to propose a list
of criteria that we would use in
determining whether a prescriber is
engaging in prescribing practices
sufficient to warrant a revocation.
In determining instances of a pattern
or practice of prescribing that is abusive
and a threat to the health and safety of
Medicare beneficiaries, we proposed to
consider several factors, including—
• Whether there are diagnoses to
support the indications for which the
drugs were prescribed;
• Whether there are instances where
the necessary evaluation of the patient
for whom the drug was prescribed could
not have occurred (for example, the
patient was deceased or out of state at
the time of the alleged office visit);
• Whether the physician or eligible
professional has prescribed controlled
substances in excessive dosages that are
linked to patient overdoses;
• The number and type(s) of
disciplinary actions taken against the
physician or eligible professional by the
licensing body or medical board for the
state or states in which he or she
practices, and the reason(s) for the
action(s);
• Whether the physician or eligible
professional has any history of ‘‘final
adverse actions’’ (as that term is defined
in § 424.502);
• The number and type(s) of
malpractice suits that have been filed
against the physician or eligible
professional related to prescribing that
have resulted in a final judgment against
the physician or eligible professional or
in which the physician or eligible
professional has paid a settlement to the
plaintiff(s) (to the extent this can be
determined);
• Whether any State Medicaid
program or any other public or private
health insurance program has restricted,
suspended, revoked, or terminated the
physician or eligible professional’s
ability to prescribe medications, and the
reason(s) for any such restriction,
suspension, revocation, or termination;
and
• Any other relevant information
provided to CMS.
In determining whether a physician or
eligible professional has a pattern or
practice of prescribing that fails to meet
Medicare requirements, we proposed to
consider the following factors, including
whether the physician or eligible
professional—
• Has a pattern or practice of
prescribing without valid prescribing
authority;
• Has a pattern or practice of
prescribing for controlled substances
outside the scope of the prescriber’s
DEA Certificate of Registration;
• Has a pattern or practice of
prescribing drugs for indications that
were not medically accepted—that is,
for indications neither approved by the
Food and Drug Administration (FDA)
nor medically accepted under 1860D–
2(e)(4) of the Act—and whether there is
evidence that the physician or eligible
professional acted in reckless disregard
for the health and safety of the patient.
Many patterns and practices of
prescribing, though perhaps
questionable on their face, do not upon
investigation involve abusive or
fraudulent behavior nor involve
substandard medical care. As such, we
proposed to base any revocation under
proposed § 424.535(a)(14) on situations
that fall outside the norm of appropriate
prescribing, and only after carefully
considering the relevant factors. A
thorough, detailed investigation by CMS
of the physician or eligible
professional’s prescribing practices
would be a prerequisite for the use of
§ 424.535(a)(14). Honest physicians and
eligible professionals who engage in
reasonable prescribing activities would
not be impacted by our proposal. We
noted further that CMS, rather than the
Part D plans or the A/B MACs, would
make all determinations under our
proposed provisions, though
information contained in referrals from
Part D Plan sponsors may be used as
part of CMS’ analysis to make
revocation decisions.
We received a high volume of
comments regarding proposed
§ 424.535(a)(14). Comment summaries
and our responses are as follows.
Comment: A number of commenters
opposed our proposed addition of
§ 424.535(a)(14). They generally stated
that this revocation reason would
negatively impact Medicare
beneficiaries by restricting access to
important medications and disrupting
current care plans, hence creating a
chilling effect on the practice of
medicine. They asserted that the
proposed provision could dissuade
physicians from appropriate
prescribing. What may be considered
excessive prescribing for the general
population, they added, could be
clinically appropriate given a patient’s
individual circumstances, particularly
in pain management; many ‘‘off-label’’
uses are clinically appropriate and
represent the standard of care,
especially with cancer patients. Several
commenters also stated that the process
of finding the right medication for a
particular individual may involve trial
and error over the course of months, if
not years; decisions about specific
medications to prescribe must be based
on clinical observations, knowledge of
past history, awareness of side effects,
and a process of collaboration between
doctor and patient. One commenter
stated that policies that markedly limit
the use of substances to treat chronic
pain could increase the suicide rate.
Response: We appreciate these
comments and fully recognize the
commenters’ concerns. We certainly
understand that each patient is
different, as is: (1) His or her specific
medical condition; (2) the setting in
which he or she is being treated; and (3)
the types and doses of medications that
may legitimately be required. As
alluded to in the proposed rule and as
we more emphatically state here, we
only intend to invoke § 424.535(a)(14) in
very limited and exceptional
circumstances. For this reason, we do
not believe that § 424.535(a)(14) will
have a chilling effect on physician or
practitioner prescribing activities or will
restrict beneficiaries’ access to
medications. Indeed, it will become
clear to honest and legitimate
prescribers (once § 424.535(a)(14)
becomes effective and is implemented)
that our focus is restricted to cases of
improper prescribing that are so
egregious that the physician or
practitioner’s removal from the
Medicare program is needed to protect
Medicare beneficiaries.
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00056 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29899
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
Comment: Many commenters
contended that state medical licensing
boards are the appropriate bodies to
review prescribing practices; one such
commenter stated that prescription
restrictions under Part D should only be
imposed if the state board finds a
pattern of negligence in prescribing
practices. Other commenters
recommended that CMS, in lieu of
utilizing § 424.535(a)(14), refer cases of
improper prescribing to the applicable
state board for its review and
disposition, with one commenter adding
that CMS could then decide whether to
take action based on the state’s findings.
This commenter stated that such
investigatory actions should be left to
the state; having both CMS and the state
undertake separate investigations would
be duplicative and redundant, perhaps
slowing down both investigations in the
process.
Response: We recognize the leading
position of state medical boards in
monitoring the practice of medicine.
However, such bodies operate
independently of CMS. They play no
role in overseeing the Medicare
program, a responsibility that rests
exclusively with CMS. As such, we
must be able to rapidly take steps on our
own volition (without having to wait for
possible action by state licensing boards
or other bodies) to protect Medicare
beneficiaries and the Trust Funds from
abusive behavior.
Comment: Several commenters
asserted that CMS lacks the statutory
authority for § 424.535(a)(14).
Response: We disagree. As we stated
in the proposed rule, sections 1102 and
1871 of the Act give the Secretary the
authority to establish requirements for
the efficient administration of the
Medicare program. We believe that
§ 424.535(a)(14) is necessary to help
ensure the integrity and efficiency of the
Medicare program.
Comment: Several commenters
opposed the use of
§ 424.535(a)(14)(i)(F), which addresses
prescription-related malpractice suits,
as a criterion. One commenter
contended that CMS’ assertion that the
existence of such a lawsuit is somehow
equivalent to liability is incorrect. The
commenter, as well as others, stated that
many liability insurers settle cases with
little or no merit. Another commenter
stated that it would be difficult for CMS
to verify the existence of such suits and
settlements, while another commenter
contended that certain physician
specialties at high risk for malpractice
suits could be unfairly targeted under
§ 424.535(a)(14).
Response: We did not assert in the
proposed rule (and do not in this final
rule) that such a lawsuit automatically
equates to liability. We realize that
certain cases are settled with no
admission or even existence of liability.
Nonetheless, it would be inappropriate
and even irresponsible for CMS to
completely disregard situations where a
physician or practitioner has, for
example, been sued several times for
prescription-related malpractice and has
either settled one of the cases or has had
at least one final judgment against him
or her.
We stress that § 424.535(a)(14)(i)(F)
will represent only one of several factors
in our § 424.535(a)(14) determinations,
and it will not in and of itself be
dispositive.
With respect to the next-to-last
comment, we included the language ‘‘to
the extent this can be determined’’ at
the end of proposed
§ 424.535(a)(14)(i)(F) based on our
recognition that it may occasionally be
difficult to ascertain the specific
outcome of such suits.
Regarding the last comment, and as
already stated: (1) We only intend to
invoke § 424.535(a)(14) in very limited
and exceptional circumstances; (2) we
will account for the patient’s particular
situation and setting in determining
whether a § 424.535(a)(14) revocation is
warranted; and (3) § 424.535(a)(14)(i)(F)
is only one of a number of factors we
will consider.
Comment: Several commenters stated
that CMS’ proposal is duplicative of
current safety mechanisms, ignores the
long history of states regulating the
licensure process, adds yet another layer
of regulatory burden and administrative
costs to the program, and gives the
federal government an excessive amount
of latitude without furnishing clear
objectives. They added that CMS has
stepped outside its statutory authority
and into regulating the practice of
medicine, and has also usurped the
authority of state boards to regulate the
practice of medicine. They requested
that CMS work with the medical
community through pre-rulemaking
activities, such as listening sessions,
town halls, and the issuance of requests
for information (RFI), to better develop
any future proposals to address the
agency’s concerns. Another commenter
stated that CMS should focus on
preventing individuals who do not have
the authority to prescribe (such as
massage therapists) from prescribing
Part D drugs rather than on applying
§ 424.535(a)(14).
Response: Section 424.535(a)(14) is
not an attempt by CMS to regulate the
practice of medicine or to usurp state
medical boards’ roles in doing so. States
remain free to take action against
physicians and practitioners as they
deem fit. Again, though, Medicare is a
distinct program that is under the
purview of CMS, not the states. We
must have the ability to remove abusive
prescribers from the Medicare program
without having to obtain or wait for
approval from state licensing boards or
other bodies that do not have oversight
of Medicare.
As mentioned earlier, we have the
authority under sections 1102 and 1871
of the Act to establish requirements for
the efficient administration of the
Medicare program. We believe this
includes ensuring that the Part D
program is properly administered, and
that Medicare beneficiaries and the
Trust Funds are protected. We believe
that § 424.535(a)(14) will be an
important part of these objectives.
We appreciate the recommendation
that we work with the medical
community in developing future
proposals and will take it under
advisement.
As for the final comment, our
addition of (c)(6) is aimed at stemming
the problem of unqualified prescribers.
Yet we disagree with the implication
that this issue should be our sole focus.
Other matters, such as egregious and
dangerous prescribing practices by
physicians and eligible professionals,
must be addressed as well.
Comment: Several commenters
expressed concern about the potential
application of § 424.535(a)(14) to
hospice and palliative physicians. They
stated that medications furnished in a
hospice or palliative setting often
require doses and indications that are
generally not seen in conventional care.
Such doses, they contend, are often
necessary to relieve pain and furnish
comfort to terminally ill patients, noting
also that dosages might vary depending
on what stage of the dying process the
patient is in; terminally ill patients, they
state, require different pain management
strategies and often higher doses of
opioids than those who are not
terminally ill. The possible application
of § 424.535(a)(14) to hospice and
palliative physicians, they asserted,
could prevent these physicians from
prescribing needed medications to
dying patients due to concerns about
prescribing outside the usual norms.
They requested an exception to
§ 424.535(a)(14) when the patient is
specifically receiving hospice or
palliative services. Another commenter
suggested exempting from
§ 424.535(a)(14) those physicians who
are ABMS-board certified in hospice
and palliative medicine, or medical
directors certified by the Hospice
Medical Director Certification Board.
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00057 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29900
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
Response: We decline to establish a
specific exception for hospice or
palliative physicians or services, for this
would eliminate our ability to take
action against truly egregious and
dangerous prescribing practices that
may occur in such settings. However, as
stated earlier, we fully understand that
each patient is different, as is his or her
specific condition and needs. We will
operate under this overriding principle
when considering whether
§ 424.535(a)(14) should be invoked in a
particular instance.
Comment: A number of commenters
contended that several of the criteria
identified by CMS are beyond the
expertise of CMS regulators.
Response: We disagree. We have
physicians and other medical personnel
on staff who we anticipate may be
consulted, as needed, in potential
§ 424.535(a)(14) cases.
Comment: A commenter stated that
because of the limited number of
certified hospice and palliative
physicians, most hospice and palliative
patients will be cared for by their
primary care physician or mid-level
practitioner. The commenter
recommended that CMS add an appeals
process with peer-review to ensure that
good clinicians are not penalized
unduly. Other commenters expressed
concern that the proposed rule made no
mention of appeal rights, while one
commenter requested how physicians
can defend themselves against a
§ 424.535(a)(14) revocation.
Response: A physician or eligible
professional whose Medicare billing and
prescribing privileges are revoked under
§ 424.535(a)(14) may appeal the
revocation per 42 CFR part 498. Also, as
already mentioned, we anticipate that
physicians and other medical personnel
of CMS may be consulted, as needed, in
potential § 424.535(a)(14) cases.
Comment: One commenter stated that
CMS should clarify the term ‘‘necessary
evaluation’’ as it is used in
§ 424.535(a)(14)(i)(B); the commenter
explained that a hospice or palliative
physician must often rely on the
evaluations of the nurses and is not
always able to physically see a
homebound patient. The commenter
was concerned that he or she would not
be able to adjust dosages without seeing
the patient. Another commenter stated
that in applying this criterion, CMS
should focus more on the prescriber’s
status than on beneficiaries who may be
evaluated outside of their normal
residence.
Response: We are not in a position to
further clarify or define the term
‘‘necessary evaluation’’ in this rule, for
we must retain the flexibility to address
the variety of factual scenarios that
could potentially implicate
§ 424.535(a)(14). However, we recognize
the commenter’s concern, and as stated
earlier we will account for the patient’s
particular needs and circumstances.
We intend to review all aspects of the
prescriber’s and the patient’s statuses
and physical locations when examining
this criterion.
Comment: A commenter
recommended that in lieu of adopting
its proposed new revocation policy,
CMS should use its existing regulatory
authority under § 405.371 to suspend
Part D prescribing privileges when there
is a credible allegation of fraud. If CMS
believes it lacks the legal authority to
implement a payment suspension that
precludes a physician or eligible
professional from prescribing, ordering,
or certifying services for a Medicare
beneficiary when a credible allegation of
fraud exists, CMS should consider
proposing a new policy that expands on
the existing provisions in § 405.371 and
allow the public to comment on this
policy. Another commenter requested
that CMS explain how a revocation
under § 424.535(a)(14) is different from
an OIG exclusion based on a conviction
of fraud. Another commenter contended
that CMS, through § 424.535(a)(14),
would essentially be making fraud
determinations that CMS lacks the
statutory authority to undertake.
Response: We disagree with the first
commenter’s recommendation and the
third commenter’s statement because
abusive or inappropriate prescribing
does not necessarily involve fraudulent
behavior, although it could well involve
improper payments. We further believe
that revocation is a more appropriate
remedy for abusive prescribing than a
payment suspension. In the latter
situation, the prescriber would remain
enrolled in Medicare despite his or her
improper prescribing; we believe this
goes against the overall objective of
§ 424.535(a)(14), which is to protect
Medicare beneficiaries and the Trust
Funds from abusive behavior.
Comment: A commenter suggested
that CMS should, prior to finalizing
§ 424.535(a)(14), solicit comments on a
process of notification and opportunity
to correct prior to implementing a
revocation under § 424.535(a)(14). Other
commenters likewise stated that before
revoking a supplier under
§ 424.535(a)(14), CMS should notify the
supplier of the potential revocation and
enable the supplier to respond.
Response: We disagree. Providing a
physician with an opportunity to take
corrective action would not be
appropriate under these circumstances,
given that CMS would have based its
revocation action on a prescriber
engaging in a pattern or practice of
abusive prescribing over some period of
time. One of our goals with
§ 424.535(a)(14) is to place prescribers
on notice that abusive prescribing
practices can result in the individual’s
losing his or her Medicare billing
privileges. To permit an abusive
prescriber to avoid revocation by simply
modifying his or her behavior
temporarily would undermine this
objective and, more importantly, would
not undo the harm that may have been
done to Medicare beneficiaries because
of the prescriber’s practices.
Comment: Several commenters
suggested that CMS be required to
consult with and receive written
approval from the OIG and/or the
Department of Justice prior to any
invocation of § 424.535(a)(14).
Response: We do not agree. As
mentioned earlier, CMS administers the
Medicare program. We must be able to
expeditiously remove abusive
prescribers from the Medicare program
without having to secure prior approval
from law enforcement. Indeed, failure to
take such quick action would be
inconsistent with the spirit of the two
aforementioned OIG reports that urged
CMS to exercise greater oversight of the
Part D program.
Comment: A commenter opposed the
criterion in § 424.535(a)(14)(i)(B) that
reads, ‘‘Whether there are instances
where the necessary evaluation of the
patient for whom the drug was
prescribed could not have occurred (for
example, the patient was deceased or
out of state at the time of the alleged
office visit).’’ The commenter stated that
this factor does not address whether the
physician or eligible professional is out
of the country when the new
prescription for a Part D drug was given
to a beneficiary. Another commenter
stated that the criterion does not: (1)
Outline cases where a physician or
eligible professional is allowed under
state law to prescribe Part D drugs over
the phone to a Medicare beneficiary
who is on vacation and may need a Part
D prescription; and (2) differentiate
between a prescription for a new Part D
drug a day after the death of a Medicare
beneficiary and a refill of an existing
Part D medication by the spouse or
child after the death of the Medicare
patient. This commenter requested that
CMS rescind this criterion unless it
furnishes more information, such as
how it will be used as a factor in making
a revocation determination. Another
commenter requested the removal of
this criterion if it will be based solely on
PDE data.
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00058 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29901
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
Response: The example cited in
§ 424.535(a)(14)(i)(B) is not the only one
to which the criterion could apply. The
term ‘‘for example’’ indicates that
multiple factual scenarios are
envisioned. Such is the case with
§ 424.535(a)(14)(i)(B). We will consider
the specific facts of each situation in
determining whether the resolution of
this factor weighs in favor of a
revocation under § 424.535(a)(14). In
addition, we will consider information
besides PDE data when evaluating this
criterion.
Comment: A commenter opposed the
criterion outlined in
§ 424.535(a)(14)(i)(D) regarding the
number and type(s) of disciplinary
actions. The commenter contended that
CMS did not indicate whether it would
use a particular state licensing board
decision (for example, a reprimand or
fine) as its basis for taking an action
under § 424.535(a)(14). The commenter
stated that CMS should rescind this
portion of its proposal unless it: (1)
Provides more information regarding the
state medical board actions that would
be used as a factor in making a decision
under § 424.535(a)(14); and (2) affords
the public an opportunity to comment
on CMS’ implementation approach.
Response: We are not in a position to
outline every conceivable disciplinary
action that a state medical board could
impose. Such actions vary widely by
state and by magnitude, which is why
§ 424.535(a)(14)(D) accounts for the
specific type of disciplinary action
involved.
Comment: Several commenters stated
that the term ‘‘abusive’’ should be
stricken from the rule because it is too
broad and subjective. Others requested
that CMS at least provide more
clarification and guidance: (1) As to the
meaning of the terms ‘‘abusive,’’
‘‘excessive dosage,’’ ‘‘improper
prescribing practices,’’ and ‘‘threat to
patient health and safety’’; and (2)
regarding the steps that would be taken
if the agency determines that a
prescriber’s Medicare enrollment should
be revoked; one commenter stated that
CMS Publication 100–18, Chapter 9,
contains a definition of ‘‘abusive’’
whereas our proposed rule did not.
Another commenter recommended that
this guidance incorporate evidence-
based guidelines and research along
with the patient’s history.
Response: We did not define these
terms in the proposed rule and decline
to do so in this final rule because of the
need to retain our flexibility in
addressing a variety of factual scenarios.
Any revocation under § 424.535(a)(14)
would be processed in the same manner
as all other revocations, with the
exception that with these revocations,
the applicable Part D plan sponsor(s)
would also be notified of CMS’
revocation action so that the sponsor
can terminate the individual’s
prescribing privileges.
Comment: A commenter stated that
while CMS noted in its proposed rule
that it would conduct a complete and
thorough investigation prior to any
revocation, there are no safeguards to
ensure a full investigation. The
commenter added that CMS did not
identify who would conduct these
investigations. Other commenters
requested information as to the process
for determining whether abusive
prescribing or a threat to patient health
and safety exists. Another commenter
stressed the need for a clearly defined
protocol that would be followed before
any revocation decision is made.
Response: We stated in the proposed
rule and reiterate here that in every case
we will carefully consider all of the
relevant factors before invoking
§ 424.535(a)(14); this will include a
review of all of the evidence before us,
including the patient’s particular needs,
circumstances, and setting. CMS and
contractor staff will conduct the
investigations, with CMS personnel
performing the evaluation of the factors
and making the final determination.
More detailed information regarding the
review process will, as deemed
necessary, be disseminated via sub-
regulatory or other guidance.
Comment: Several commenters
requested clarification regarding
whether CMS intends to implement
§ 424.535(a)(14) retrospectively. They
supported a strictly prospective
application.
Response: We reserve the right to
revoke the billing privileges of a
physician or eligible professional
enrolled as of the effective date of this
rule who has engaged or is engaging in
abusive prescribing as described in
§ 424.535(a)(14). However, the effective
date of the revocation would not be
earlier than the effective date of this
final rule.
Comment: A commenter questioned
whether CMS will routinely scour its
data for suppliers with suspicious
prescribing patterns and, if so, what
CMS will then do.
Response: Consistent with our current
practices, we will be alert for such
prescribing patterns. Once a pattern is
detected, we will conduct a review and
investigation using our existing
procedures. If, based on this review, we
believe that a situation involving
abusive prescribing may exist, we will
determine whether action under
§ 424.535(a)(14) is warranted.
Comment: A commenter stated that
proposed § 424.535(a)(14) is
unnecessary because the OIG has the
ability to exclude from Medicare (under
42 U.S.C. 1320a–7(b)(6)(B)) any
individual who has furnished items or
services to patients substantially in
excess of the patients’ needs or of a
quality that does not meet
professionally recognized standards of
care.
Response: While we recognize that
the OIG has its exclusion authority,
CMS is the agency directly responsible
for administering the Medicare program
and for protecting Medicare
beneficiaries and the Trust Funds.
Consequently, CMS should be able to
use its own authority to pursue
administrative actions to address our
concerns regarding abusive prescribing.
We also reiterate that the OIG has
recommended that CMS exercise greater
oversight over the integrity of the Part
D program and has noted its concern
about abusive prescribing. Therefore, we
believe it is proper for CMS (and
consistent with the OIG’s
recommendations) to implement
§ 424.535(a)(14).
Comment: Several commenters
expressed support for our proposed
addition of § 424.535(a)(14). One
commenter stated that this will allow
for more effective monitoring of
improper prescribing behaviors. The
commenter noted that inappropriate
prescribing can result in overutilization
of medications that increase program
costs without providing any health
benefit and can harm beneficiaries.
Another commenter stated that
§ 424.535(a)(14) will enable CMS to
exercise greater control over the Part D
program.
Response: We appreciate the support
of these commenters.
Comment: Several commenters
recommended that CMS explain in the
final rule whether CMS or Medicare
contractors will use clinical staff
(physicians and pharmacists) in
determining whether Part D prescription
drug abuse has occurred and whether a
revocation under § 424.535(a)(14) is
warranted.
Response: As stated earlier, we may
use clinical staff, as needed, in making
§ 424.535(a)(14) determinations.
Comment: One commenter
recommended that CMS, in lieu of
finalizing § 424.535(a)(14), revoke the
Medicare billing and/or prescribing
privileges of individuals under
§ 424.535(a)(10) when the medical
documentation does not support the
Part D prescription written by the
physician or eligible professional. The
commenter believed that this approach
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00059 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29902
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
would be easier and more cost-effective
to implement and would avoid the need
for CMS to make clinical judgments.
Response: Section 424.535(a)(10) does
not apply to Part D prescriptions.
Consequently, § 424.535(a)(10) cannot
be used in lieu of § 424.535(a)(14).
Comment: A commenter requested
clarification concerning whether CMS
or its Medicare contractors will conduct
medical document reviews to determine
whether an abusive prescribing pattern
exists.
Response: Medical document reviews
are one of several actions we may
undertake in determining whether an
invocation of § 424.535(a)(14) is
warranted.
Comment: With respect to the
criterion regarding diagnoses to support
indications for which the drugs were
prescribed, a commenter: (1) Questioned
how CMS will cross-reference Part D
prescriptions with appropriate
diagnoses; and (2) stated that CMS
should include all scientifically-
supported indications, whether on the
FDA labeling or not.
Response: We will, as deemed
necessary, furnish sub-regulatory or
other guidance to address the
commenter’s first issue. We agree with
the commenter’s second comment, and
intend to include all scientifically-
supported indications irrespective of
whether they are on the FDA labeling.
Comment: One commenter expressed
concern about the potential impact of
§ 424.535(a)(14) on pharmacies and
their patients. The commenter stated
that beneficiaries may see an
interruption in the continuity of their
health care if their physician is no
longer qualified to be a Medicare
supplier; the commenter believed there
should be options available to ensure
that health care is not interrupted.
Response: As explained earlier, we
only intend to invoke § 424.535(a)(14) in
exceptional circumstances.
Consequently, we do not believe that
patient access in general will be
impacted.
Comment: In referring to the criterion
in § 424.535(a)(14) regarding private
insurers, a commenter stated that CMS
does not have the statutory authority to
make enrollment and revocation
decisions based upon the actions of
commercial health insurers. The
commenter urged CMS to explain its
legal justification for invoking
§ 424.535(a)(14) on this ground. The
commenter also suggested that CMS
explain how it will obtain information
regarding private insurer actions taken
against physicians and practitioners and
whether these insurers will be required
to furnish such data to CMS.
Response: We disagree with the
commenter’s assertion that we do not
have the authority to consider the
actions of private insurers in
determining whether a § 424.535(a)(14)
is appropriate. Again, we have the
authority under sections 1102 and 1871
of the Act to establish requirements for
the efficient administration of the
Medicare program. If private insurers
have taken actions against a particular
physician or practitioner for
questionable prescribing activities, we
believe it would be appropriate for us to
consider this information in light of our
obligation to oversee the Part D program
in a responsible manner. We will
attempt to work with private insurers to
facilitate the appropriate exchange of
information.
Comment: A commenter opposed the
following criterion: ‘‘Whether the
physician or eligible professional has
prescribed controlled substances in
excessive dosages that are linked to
patient overdoses.’’ The commenter
contended that CMS: (1) Did not
provide the source or sources that it will
use to obtain this information, and (2)
already has a similar reason for
revocation in § 424.535(a)(3) regarding
felony convictions. The commenter
stated that if CMS adopts this criterion,
CMS should add the following language
to the end thereof: ‘‘that result in a
felony conviction of criminal neglect or
misconduct.’’ The commenter also
recommended that CMS: (1) Cite the
sources it will use to obtain information
on patient overdoses; and (2) defer to
the state medical boards regarding
whether a physician or eligible
professional is posing an immediate risk
to Medicare Part D beneficiaries
(assuming CMS intends to consider state
actions in its § 424.535(a)(3)
determinations).
Response: We are unclear as to the
specific information to which the
commenter is referring; namely,
whether the commenter is alluding to
published clinical data (for example,
professional journals) or to information
regarding a particular patient’s
overdose. If it is the latter, as we
suspect, we intend to use both publicly
available and internal data to determine
whether cases of excessive prescribing
exist. To the extent this data is obtained
from state medical boards, CMS (for
reasons alluded to earlier) does not
believe a prior determination by the
state of an immediate risk to Part D
beneficiaries is necessary.
Comment: A commenter suggested
that in lieu of revoking an individual
under § 424.535(a)(14), CMS should
place the physician under a payment
suspension and deactivate his or her
Medicare billing privileges. Another
commenter recommended that CMS
consider a sliding scale to include
lower-level consequences (such as
suspensions) for less severe
occurrences.
Response: We disagree. With a
payment suspension, the physician
would remain enrolled in Medicare and
be able to prescribe Medicare Part D
drugs and provide Medicare Part B
services (although he or she would have
Medicare payments withheld for a
period of time). Moreover, there would
be no legal basis under § 424.540 to
deactivate the supplier’s prescribing
privileges, which is why revocation is
the most appropriate remedy to address
these situations.
Comment: A commenter stated that
the sole use of PDE data to identify
prescriber trends is insufficient to
determine abusive practices, for such
data cannot distinguish between: (1)
Legitimate high dose and frequency of
prescriptions; and (2) illegitimate
prescribing.
Response: We agree and intend to use
various data sources to detect such
practices.
Comment: Several commenters stated
that prescribers may avoid long-term
care practice for fear of being revoked
from Medicare.
Response: We disagree. We
mentioned earlier that we only intend to
invoke § 424.535(a)(14) in exceptional
circumstances involving truly abusive
behavior. Accordingly, we do not
believe that § 424.535(a)(14) will deter
prescribers from practicing in long-term
care settings.
Comment: A commenter suggested
that CMS provide additional data to
plans in order to improve a plan’s
ability to identify inappropriate patterns
and to apply claims processing edits
correctly/timely.
Response: We agree and are
considering various means of doing so.
Comment: One commenter stated that
a revocation under § 424.535(a)(14)
alone will not suspend or revoke the
practitioner’s right to prescribe drugs
under state law, meaning that patients
other than Medicare beneficiaries would
still be at risk. This is especially true,
the commenter added, considering that
§ 424.535(a)(14) does not require such
CMS revocations to be reported to the
state.
Response: It is possible that a
prescriber revoked under
§ 424.535(a)(14) may still be able to
retain his or her state license. However,
we are currently working with the states
to facilitate a closer exchange of
information regarding Medicare actions
taken against physicians and
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00060 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29903
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
practitioners, which may facilitate
concomitant action taken by states.
Comment: Several commenters
requested clarification as to whether a:
(1) Part D plan sponsor would be
penalized if it fills a prescription from
a terminated supplier when information
about the termination is not available
(for example, in cases of retroactive
termination or an error with CMS
records); and (2) whether a pharmacy
would be penalized for filling a
prescription order that has been
approved through the claims
adjudication process. The commenter
opposed the application of such
penalties.
Response: We believe these comments
are outside the scope of this final rule.
Comment: A commenter
recommended that CMS remove the
following as a factor for revocation:
‘‘Has a pattern or practice of prescribing
for controlled substances outside the
scope of the DEA Certificate of
Registration.’’ The commenter instead
suggested that CMS work with the DEA.
Response: We disagree with the
commenter. As previously mentioned,
we are responsible for administering the
Medicare program and must be able to
take quick action against abusive
prescribers without the prior approval
of another agency.
Comment: A commenter stated that,
on its face, each criterion appears
reasonable. However, the commenter
expressed concern that the rule does not
provide guidance on the application and
weight given to each factor, which could
allow for subjective, contradictory, and
discriminate revocation decisions
(especially with respect to the last
criterion that permits CMS to consider
‘‘any other relevant information
provided to CMS.’’).
Response: We understand the
commenter’s concern and note that we
will, as deemed necessary, be issuing
sub-regulatory guidance that explains in
more depth the operational details of
the § 424.535(a)(14) determination
process. In addition, CMS, rather than
its contractors, will make all final
determinations. This will ensure greater
overall uniformity, as well as a more
consistent application of the various
factors.
Comment: While supporting much of
our proposed addition of
§ 424.535(a)(14), a commenter expressed
concerns regarding several criteria.
First, the commenter (referring to
proposed § 424.535(a)(14)(i)(A)) stated
that there are many reasons why a
physician might prescribe a particular
drug without a formal diagnosis (for
instance, the physician may be unable
to conduct a full evaluation due to
distance, cultural preference, etc.).
Second, the commenter recommended
that a statute of limitations be imposed
regarding the individual’s final adverse
action history.
Response: We recognize that there
may be instances where a formal
diagnosis does not or cannot occur. In
applying § 424.535(a)(14)(i)(A), we will
consider the reason such a diagnosis did
not take place. Regarding the
commenter’s second concern, we do not
favor a statute of limitations for the final
adverse action criterion; CMS must be
able to retain its flexibility in this
regard. Nonetheless, we will take into
account when the adverse action
occurred when analyzing whether it
supports a finding of abusive
prescribing.
Comment: A commenter
recommended that CMS forgo adopting
§ 424.535(a)(14) and instead work with
the Congress to suspend Coverage and
Payment for Questionable Part D
Prescriptions, as described in the FY
2015 Department of Health and Human
Services performance budget.
Response: We continue to work with
the Congress in our efforts to enhance
Part D program integrity, and we believe
that § 424.535(a)(14) is an important
step in this direction.
Comment: With respect to the
criterion dealing with state disciplinary
actions, a commenter suggested that
CMS monitor prescriber licensure
statuses and status changes in lieu of
state disciplinary actions. The
commenter stated that many states do
not publish state board disciplinary
actions in a standardized format that
can be easily used to ascertain a
prescriber’s practicing privileges.
Response: We recognize that state
disciplinary data may not always be
available. To the extent that it is,
though, we do not believe it should be
completely disregarded, even if the
action did not result in a licensure
suspension or revocation.
Comment: A commenter requested
that CMS ensure that innovative abuse-
deterrent technologies are employed as
a tool in working to curb prescription
drug abuse in Medicare. Another
commenter recommended that CMS
utilize health information technology
systems to collect and organize data for
measuring performance, supporting
clinical decisions, and evaluating
quality improvement processes; drug
utilization procedures and prescription
drug monitoring programs (PDMPs), for
instance, could be important tools for
improving public health and clinical
practice.
Response: We appreciate these
suggestions and note that we are
considering various technological and
system-based means of enhancing our
oversight of the Part D program.
Comment: A commenter offered
several suggestions. First, CMS should
furnish examples in the final rule as to
the process of identifying and
quantifying a pattern or practice as well
as the actual revocation process of
Medicare enrollment. Second, CMS
should offer additional educational
opportunities for suppliers regarding
Medicare prescribing practices, which
would place physicians and eligible
professionals on notice that they must
meet Medicare requirements and must
prescribe properly.
Response: We agree with the
commenter’s second recommendation
and, as stated earlier, plan to conduct
outreach regarding prescribing
practices. As for the first suggestion, we
are not in a position in this final rule to
furnish specific examples of when we
would conclude that abusive
prescribing exists and a § 424.535(a)(14)
revocation is warranted; again, we must
retain our flexibility to address a variety
of factual scenarios.
The revocation process will be the
same as that which currently exists for
all other revocation reasons under
§ 424.535(a), the lone exception being
that Part D plan sponsors will be
notified of a revocation action under
§ 424.535(a)(14).
Comment: One commenter
recommended that CMS revise the
regulation to permit denial of an
enrollment application due to
prescribing practices that are either
abusive and/or represent a threat to the
health and safety of Medicare
beneficiaries.
Response: We appreciate this
suggestion and may consider it as part
of a potential future rulemaking effort.
Comment: One commenter
recommended that CMS obtain and
consider a recommendation from the
plan sponsor’s medical director as to
whether the prescribing pattern falls
outside the standard of care and
represents a therapeutic use for which
safety and efficacy is not otherwise
supported by available scientific
evidence.
Response: We appreciate this
suggestion, but note that CMS staff
includes medical personnel who, as
stated earlier, may be consulted as
needed in potential § 424.535(a)(14)
cases.
Comment: A commenter requested
that CMS eliminate the criterion dealing
with patterns and practices of
prescribing without authority and
instead utilize processes already in
place to validate prescriptive authority
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00061 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29904
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
at the point of sale. The commenter also
recommended that CMS work with
industry stakeholders to develop a
streamlined process for capturing data
that will be used in CMS’
§ 424.535(a)(14) determinations.
Response: We disagree with the first
comment. The issue is not the technical
or logistical means of validating
prescriptive authority but whether the
unauthorized prescribing of drugs is
indicative of abusive prescribing. As for
the second comment, we are somewhat
unclear as to the commenter’s specific
request; nevertheless, we will consult
with plan sponsors and pharmacy
interest groups as needed to ensure that
our new provisions are effectively
implemented.
Comment: A commenter expressed
support for CMS’ decision not to define
‘‘abusive’’ and ‘‘threat to the health and
safety of Medicare beneficiaries’’ and to
allow CMS the flexibility to address
each case on its own merits. However,
it urged CMS to review the list of
criteria on a periodic basis and consider
additions and modifications to reflect
advances in clinical best practices and
the evolution of abusive prescribing
patterns or practices.
Response: We appreciate the
commenter’s support and intend to
regularly review (and, if needed, update
via further rulemaking) the criteria in
§ 424.535(a)(14) to account for changes
in the medical field.
Comment: A commenter suggested
that in making § 424.535(a)(14)
determinations, CMS should: (1) Take
into account historical information in
the National Practitioner Data Bank
(such as past DEA registration
suspensions); and (2) consider the
relative severity of any state licensure
sanctions.
Response: We appreciate this
comment. As stated earlier, a physician
or eligible professional’s final adverse
action history (both past and present)
will be a criterion for us to consider; the
severity of any such actions or sanctions
will be taken into account as well.
Comment: A commenter stated that
proposed § 424.535(a)(14) may run
counter to Medicare regulations that
protect patient rights, creating the
possibility that systematic limitations
on prescribing practices may constitute
a violation of patients’ rights to pain
assessments, palliative care, and the
provision of hospice care.
Response: We disagree with the
commenter. We do not believe that
§ 424.535(a)(14) will hinder the ability
of Medicare beneficiaries to receive
appropriate medications, particularly
considering that: (1) § 424.535(a)(14)
will only be applied in egregious
instances; and (2) the patient’s
particular needs, circumstances, and
setting will be taken into account.
Comment: A commenter stated that
there needs to be additional information
in the final rule as to how this
information would be provided to PBMs
and how PBMs should administer it; for
example, guidance is needed on how to
manage suppliers that are licensed in
multiple states but have an action
against them in one state but not the
other(s).
Response: We will, as deemed
necessary, disseminate sub-regulatory or
other guidance that addresses the issues
the commenter has raised.
Comment: Several commenters
believed that § 424.535(a)(14) could be
strengthened even further by permitting
revocation of enrollment based on
prescribing practices that are abusive
and/or represent a threat to the health
and safety of Medicare beneficiaries, as
opposed to requiring that both of these
criteria be met. The commenters stated
that some prescribing practices might be
fraudulent and abusive but not
necessarily representative of a threat to
the health and safety of Medicare
beneficiaries.
Response: We agree, and will revise
§ 424.535(a)(14) accordingly.
Specifically, the language in
§ 424.535(a)(14)(i) that reads, ‘‘The
pattern or practice is abusive and
represents a threat to the health and
safety of Medicare beneficiaries,’’ will
be changed to ‘‘The pattern or practice
is abusive or represents a threat to the
health and safety of Medicare
beneficiaries or both.’’ This will give us
further flexibility in addressing cases of
abusive prescribing, which in turn will
enable us to better protect Medicare
beneficiaries and the Trust Funds.
Given these comments and our
responses, we are finalizing our
addition of § 424.535(a)(14) with the
exception noted in the previous
paragraph.
We also received a number of
comments that, in general, either
applied to all of our proposed
provisions or were not precisely related
to any specific proposal. Our summary
of the comments and are responses are
as follows.
Comment: Several commenters
opposed the use of the CMS–855O on
various grounds. First, the CMS–855O
does not collect practice location or
medical storage information, which the
commenter believes is a significant
vulnerability and is inconsistent with
CMS’ existing regulations. Second, use
of the CMS–855O is inappropriate
because proposed and final regulations
(in which the notice-and-comment
process is used) regarding its use and
implementation were not published in
the Federal Register. Third, the
commenter contended that CMS does
not have the statutory or legal basis to
use an enrollment application other
than for the express purpose of enrolling
a provider or supplier; as such, CMS
exceeded its legal authority to
implement the CMS–855O for the sole
purpose of ordering and certifying
services and items in the Medicare
program. Fourth, the commenter
contended that CMS lacks the statutory
and regulatory basis to establish a
registration process for Medicare. Fifth,
the CMS–855O is duplicative of the
CMS–855I form, the latter of which was
subject to notice-and-comment; also,
requiring a physician who is enrolled
(via the CMS–855I) solely to order or
certify services or items to then
complete the CMS–855I if he or she
wishes to bill Medicare increases the
paperwork burden.
Response: These comments are
outside the scope of this final rule.
Comment: A commenter
recommended that to improve
transparency, CMS should post the
name, NPI, and reason for each
Medicare enrollment revocation or
payment suspension and the duration of
the revocation or payment suspension
on the Medicare Provider/Supplier
Enrollment Web site. The commenter
believed that this information would be
useful for Medicare Advantage
Organizations, Part D sponsors, and the
public.
Response: We appreciate this
suggestion, and will take it under
advisement as we continue our efforts to
strengthen the integrity of the Part D
program.
Comment: Several commenters
recommended that CMS: (1) Explain
why it does not believe the inclusion of
the practice location on the CMS–855O
is essential to identifying the physician
or non-physician practitioner; (2)
require that all physicians and non-
physician practitioners report their
practice locations; (3) mandate that only
physicians with a defined specialty be
permitted to prescribe Part D drugs; (4)
remove from the list of physicians and
eligible professionals with an approved
enrollment record any physician or non-
physician practitioner with an
undefined or unlisted physician or non-
physician specialty code; (5) explain
why it did not solicit comments on the
use of an electronic signature in the
Internet-based versions of the CMS–
855O and the CMS–855I; (6) provide the
authority to implement and use the
CMS–855O beginning in July 2011 and
explain why it did not choose to solicit
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00062 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29905
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
public comments on changes to
regulatory provisions found in
§§ 424.502 and 424.505 for almost 3
years after adopting and using the CMS–
855O; (7) explain why it is using the
CMS–855O rather than the CMS–855I
since the CMS–855O, in the
commenter’s view, essentially
duplicates the CMS–855I; (8) modify
and use the CMS–855I (rather than
continue using the CMS–855O) because
CMS cannot verify the practice location
of a physician who registers using the
CMS–855O; (9) explain why CMS has
not proposed to revise § 424.500 to
accommodate the registration of
physicians and non-physician
practitioners for the sole purpose of
ordering/certifying services and items in
the Medicare program; (10) disenroll all
physicians and practitioners enrolled
via the CMS–855O and require them to
enroll via the CMS–855I; and (11)
provide the number of individuals
enrolled or registered into the Medicare
program using the CMS–855O since July
2011.
Response: These comments are
outside the scope of this final rule.
Comment: A commenter suggested
that the final rule take action against
physicians who report via the Internet
that they are board certified when in
fact they are not.
Response: We appreciate this
suggestion. While we are unable to
include such a provision in this final
rule because we did not propose it, we
will take it under advisement as we
continue our efforts to strengthen the
integrity of the Part D program.
Comment: A commenter
recommended that CMS require
physicians and eligible professionals to
have an active Medicare enrollment to
order Part B drugs.
Response: This comment is outside
the scope of this final rule.
Comment: A commenter
recommended that CMS not allow
physicians and eligible professionals
who have opted-out of Medicare to
order or certify services and items when
they have been suspended or revoked by
a state licensing body.
Response: This comment is outside
the scope of this final rule.
Comment: A commenter
recommended that CMS clarify whether
a Part D Medicare beneficiary will need
to provide his or her physician’s name
and NPI to his or her plan sponsor if he
or she submits a Part D claim for
payment.
Response: We will, as deemed
necessary, address this issue via sub-
regulatory or other guidance.
Comment: A commenter
recommended that CMS exclude
physicians and eligible professionals
who have opted out of the Medicare
program from prescribing Part D
covered drugs because CMS does not
have the legal authority in either the
Social Security Act or existing
regulations to revoke the prescribing
privileges of a physician or eligible
professional who has opted-out of the
Medicare program.
Response: As we stated previously,
section 1802(b) of the Act is clear that
certain physicians and practitioners
may opt-out of the Medicare program
and enter into private contracts with
Medicare beneficiaries. We believe that
to require such individuals to enroll in
Medicare would be inconsistent with
this statutory provision.
Comment: A commenter
recommended that CMS explain why
the April 14, 2012 and September 11,
2011 Federal Register Notices soliciting
comments on the CMS–855O state that
physicians and practitioners submitting
this form are registering rather than
enrolling in Medicare, while the April
2013 proposed rule states that they are
enrolling in Medicare; the commenter
stated that existing regulations do not
provide for a registration process.
Response: This comment is outside
the scope of this final rule.
Comment: A commenter stated that
diagnosis codes should be placed on
prescriptions to assess their
appropriateness.
Response: This comment is outside
the scope of this final rule.
Comment: A commenter: (1)
Requested clarification regarding
whether a revocation under our
proposed provisions would affect a
physician or eligible professional’s
Medicaid enrollment; (2) requested
clarification concerning how a State
Medicaid agency would differentiate
between one’s enrollment via the CMS–
855I and an enrollment via the CMS–
855O; and (3) suggested that CMS
provide a complete list of individuals
who can only order, certify or prescribe
in the Medicare program.
Response: With respect to the first
comment, any Medicare revocation
results in the termination of the
provider or supplier’s Medicaid
enrollment pursuant to § 455.416(c).
The second comment is outside the
scope of this final rule. As for the third
comment, and as alluded to in both the
proposed rule and in this final rule, we
plan to make available to Part D
sponsors a list of physicians and eligible
professionals who have an approved
enrollment record or a valid opt-out
affidavit on file with an A/B MAC. We
do not intend at this time to modify this
list (nor to create a separate list) to
identify those individuals who are
enrolled solely to order, certify, or
prescribe in the Medicare program.
However, we may consider this as part
of a future enhancement.
Comment: A commenter suggested
that CMS require private insurers in Part
D to report suspected fraud, waste and
abuse to Medicare’s fraud contractor.
Another commenter stated that CMS
should: (1) Encourage Part D sponsors to
voluntarily report suspected instances
of physician and eligible professional
misconduct or abusive prescribing, and
(2) institute measures to ensure a two-
way working dialogue between the
sponsors and the MEDIC.
Response: We are working to ensure
that Part D plans consistently and
regularly refer suspected fraud, waste,
and abuse to the MEDIC and that there
is appropriate communication between
them.
Comment: A commenter requested
that steps be taken to ensure that a
beneficiary is notified when his or her
physician’s billing privileges have been
revoked, and that an exception be made
for emergency or urgent care situations.
Similarly, another commenter requested
clarification as to whether claims will
be processed for emergency and urgent
care services furnished by opt-out
physicians and, if so, how processors
will identify claims in that scenario.
One commenter requested that CMS
furnish guidance regarding: (1) How
Part D prescribers can complete an opt-
out affidavit; (2) how opt-out prescribers
will be identified in the file; (3) which
(if any) edits will apply to opt-out
prescribers; (4) how various enrollment
statuses (for example, an enrollment
application or opt-out affidavit is
pending) should be handled; (5) how
terminations should be handled and
whether changes in enrollment
(including suspensions and revocations)
will be communicated to plan sponsors
at least 30 business days in advance; (6)
whether the enrollment/opt-out file will
be made available to prescriber data
vendors; (7) whether an alert process
will be established for reinstated or new
enrollments that occur between file
deliveries; (8) whether override
processes will be developed; (9)
whether procedures for notifying
beneficiaries of a change in an
individual’s ability to prescribe Part D
drugs will be established; (10) whether
a special call center for Part D
prescribing issues related to enrollment
will be created; and (11) how felony
convictions, exclusions, debarments,
and State Medicaid program prescriber
sanctions should be treated for purposes
of claim denials and coding (for
instance, whether they should be treated
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00063 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29906
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
as Medicare revocations or OIG
exclusions for purposes of claim denials
and coding).
Response: We will, as deemed
necessary, address the aforementioned
issues via sub-regulatory or other
guidance.
Comment: A commenter
recommended that CMS reestablish the
systematic deactivation of Medicare
billing and prescribing privileges if the
physician or non-physician practitioner
has not billed the Medicare program in
more than a year to ensure consistency
with an OIG recommendation to this
effect. The commenter also
recommended that CMS provide the
number of physicians and eligible
professionals who have completed the
CMS–855I and who have not billed the
Medicare program in more than a year
as of March 7, 2014, the ending
comment date for this proposed rule.
Response: These comments are
outside the scope of this final rule.
Comment: A commenter stated that a
revocation would be too serious a
penalty for a DEA registration
suspension or revocation or for
improper prescribing.
Response: While we recognize
commenter’s concern regarding the
severity of a revocation action, this
action will in some cases be necessary
to protect Medicare beneficiaries and
the Trust Funds.
Comment: A commenter sought
clarification regarding whether CMS
would notify the prescriber at least 30
days in advance of a forthcoming
revocation.
Response: As already stated, the
operational procedures for revoking
suppliers under §§ 424.535(a)(13) and
(14) will be the same as those which
currently exist for other revocations
under § 424.535(a), the sole exception
being that Part D plans will be notified
of the revocation.
Comment: A commenter suggested
that CMS centralize all exclusion, opt-
out, and other lists in one location (to
the maximum extent possible),
preferably via a format that Part D plans
can download and convert into a file
format compatible with data analytics
programs. This would enable plans to
act more quickly against excluded
suppliers. Another commenter urged
that CMS update such lists
expeditiously so that plan sponsors can
take action as needed.
Response: We appreciate the first
commenter’s suggestion and may
consider this as part of a future
enhancement. We agree with the second
commenter’s recommendation and
stress that CMS attempts to update its
existing lists (and will attempt to update
the aforementioned enrolled/opted-out
prescriber list) as quickly as possible.
Comment: A commenter requested
clarification concerning whether: (1)
Plan sponsors are only supposed to
deny coverage in the state in which a
physician’s license is revoked or
whether denials should be for all states;
and (2) whether CMS will continue to
permit a physician with a CMS waiver
to continue practicing in rural areas
when he or she is the only physician
available, even though he or she is
revoked.
Response: We believe these comments
are outside the scope of this final rule.
Comment: A commenter requested
clarification regarding whether a
physician who is prohibited from
prescribing controlled substances for the
treatment of non-cancer related chronic
pain or obesity would have their
Medicare billing privileges and/or
prescribing privileges revoked by the
Medicare program.
Response: As stated in
§ 424.535(a)(13), if the applicable
licensing body for any state in which
the physician practices suspends or
revokes his or her ability to prescribe
controlled drugs, we have the discretion
to revoke his or her Medicare billing
and prescribing privileges. Should we
exercise this discretion, the physician
would be unable to prescribe covered
Part D drugs because he or she would
no longer be enrolled in Medicare.
Comment: A commenter
recommended that CMS explain the
process it uses to identify medical
licenses that are surrendered while a
formal disciplinary proceeding was
pending before a state licensing
authority.
Response: This comment is outside
the scope of this final rule.
Comment: A commenter encouraged
CMS to consider participation in the
Council for Affordable Quality
Healthcare (CAQH) universal
credentialing application process used
by many private sector healthcare
systems. Having one ‘‘portal’’ for
physicians to become credentialed for
both Medicare and private sector health
plans, the commenter believed, would
reduce the administrative burden for
physicians.
Response: We appreciate this
comment but believe it is outside the
scope of this final rule.
Comment: One commenter, while
expressing support for §§ 424.530(a)(11)
and 424.535(a)(13) and (14), requested
that CMS delay its implementation until
CMS (1) has fully field-tested the
Medicare enrollment and reporting
program, and (2) demonstrates that the
program will operate at a high level of
accuracy, with frequent updates, and
with consistently reliable linkages to
and from other federal and state
databases. Another commenter
recommended that the criterion in
§ 424.535(a)(14) regarding diagnosis
codes be delayed until the effects of the
ICD–10 transition are reviewed.
Response: While we appreciate the
first commenter’s support, we do not
believe that the implementation of these
provisions (including the criteria in
(a)(14)) should be delayed. As explained
earlier, CMS must take steps to ensure
the integrity of the Part D program and
to protect both Part D beneficiaries and
the Trust Funds.
We are neither finalizing nor
proposing any regulatory changes as a
result of these miscellaneous comments.
22. Broadening the Release of Part D
Data (§ 423.505)
We proposed to revise our regulations
governing the release of Part D data to
expand the release of unencrypted
prescriber, pharmacy, and plan
identifiers contained in prescription
drug event (PDE) records, as well as to
make other changes to our policies
regarding release of Part D PDE data. For
background, in the May 28, 2008
Federal Register (76 FR 30664), we
published a final rule entitled,
‘‘Medicare Program; Medicare Part D
Claims Data,’’ (hereinafter referred to as
the ‘‘Part D data final rule’’) to
implement regulations that govern the
collection of PDE data under the
authority of section 1860D–12(b)(3)(D)
of the Act and the disclosure of this data
in accordance with section 1106 of the
Act. The provisions governing the
collection and disclosure of PDE data
are codified at § 423.505(b)(8), (f)(3), (1)
and (m). The Part D data final rule
governed the collection and disclosure
of the original 37 elements of PDE data,
but was updated to apply to any
additional elements that were added to
the PDE record. This update was in a
final rule issued in April 2010 (75 FR
19678) entitled, ‘‘Medicare Program;
Policy and Technical Changes to the
Medicare Advantage and the Medicare
Prescription Drug Benefit Programs’’
(hereinafter referred to as the ‘‘April
2010 final rule’’).
In the preamble to the Part D data
final rule (73 FR 30671), we stated, ‘‘we
[ ] believe that it is in the interest of
public health to share the information
collected under [the authority of 1860D–
12(b)(3)(D)] with entities outside of
CMS.’’ We explained that the release of
PDE data assists CMS in evaluating the
Medicare Part D program and assessing
related policies. We further stated such
release was in the interest of public
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00064 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29907
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
health and would improve the clinical
care of beneficiaries.
In addition to setting forth the
significant public policy reasons for
disclosure of PDE data, we made clear
in the preambles of both the Part D data
final rule and the April 2010 rule that
our primary concerns in releasing PDE
data are protecting the confidentiality of
beneficiary identifiable information and
commercially sensitive data of Part D
sponsors. Therefore, as described in the
Part D data final rule and the April 2010
rule, the release of PDE data is subject
to certain protections, described here
generally, such as encryption of
beneficiary information and aggregation
of commercially sensitive data of Part D
sponsors. In addition, whenever PDE
data is released, we only release the
minimum data necessary for a given
purpose, as determined in the sole
discretion of CMS after review of the
requestor’s detailed request for data. If
releasing data to an external entity for
research purposes, CMS indicated in the
Part D data final rule that the requestor
must be a legitimate researcher,
meaning the requestor has the requisite
experience and is working for, or on
behalf of, a reputable institution. (In the
preamble to the Part D data final rule
(73 FR 30674 citing 45 CFR 164.501), we
used the definition of ‘‘research’’
contained in the HIPAA Privacy Rule,
which defines the term as ‘‘a systematic
investigation, including research
development, testing, and evaluation,
designed to develop or contribute to
generalizable knowledge.’’) In the Part D
data final rule (73 FR 30674), we also
indicated that, consistent with our
current policies for Part A and B data,
identifiable Part D data would not be
disclosed for commercial purposes.
In the preamble to the proposed rule,
we stated that we believe the current
limitations on the release of certain data
elements hinder the use of PDE data in
a health care environment that is
substantially transforming due to the
Affordable Care Act, and that these
limitations therefore also inhibit
accompanying insights into prescription
drug benefit plans that could result from
broader release of the data. We further
stated that our experience has led us to
conclude that broader release of PDE
data to external entities can increase the
positive contributions researchers make
to the evaluation and function of the
Part D program and improve the
efficiency of the program and the
clinical care of its beneficiaries, which
is in the interest of public health. For
these reasons, we stated that increased
access to prescriber, pharmacy, and plan
identifiers by all categories of requestors
is of utmost importance and will
facilitate research by entities outside
CMS that involves identifiable plans,
prescribers, and pharmacies.
Furthermore, we stated that we could
relax the current policies on the release
of this PDE data, while still protecting
beneficiary confidentiality and
commercially sensitive data of Part D
sponsors.
Specifically, we proposed to permit
the release of unencrypted prescriber,
pharmacy, and plan identifiers
(including internal plan/pharmacy
identification numbers on the claim that
represent reference numbers assigned by
the plan at the time a drug is dispensed)
contained in PDE records to all current
categories of requestors (including other
HHS entities and the Congressional
oversight agencies, non-HHS executive
branch agencies and states, and external
entities). We noted that because the
minimum necessary policy will still
apply to all such releases, our proposal
was more a formality with respect to
HHS entities/Congressional oversight
agencies and non-HHS executive branch
agencies/states, since this data is
available in unencrypted format to these
same entities under the current Part D
data regulations ‘‘if needed.’’ For this
reason, in the proposed rule, we focused
on the release of unencrypted
prescriber, pharmacy, and plan
identifiers to external entities as
discussed later in this section.
We acknowledged in the preamble to
the proposed rule that there still may be
concerns about releasing unencrypted
prescriber, plan, and pharmacy
identifiers to outside entities based on
comments that were received in
response to our original proposed Part D
data regulations, and that were
discussed in the Part D data final rule
(73 FR 30675). In particular, we
addressed concerns that this
information could be used by
pharmaceutical companies and others
who may want to influence physicians’
prescribing patterns and interfere with
their professional judgment. As we
stated in the proposed rule, it is our
view today, however, that the vast
majority of physicians have prescribed
and do prescribe what they believe are
the appropriate medications for their
patients, and they should have no
concerns with transparency in their
prescribing patterns. Moreover, we
stated that there are other measures in
place to prevent inappropriate influence
by external entities on prescribers, such
as section 6002 of Affordable Care Act
and the federal Anti-Kickback Law
(section 1128B(b) of the Act). We also
pointed out that when data are
completely transparent, it is easier for
the attempts of some to use the data for
purposes of inappropriate manipulation
to be countered by others who have
access to the same data. We also noted
that it appears prescriber data are
already available commercially from
pharmacy data aggregators. For these
reasons, we stated that we believe that
our earlier concerns expressed in the
Part D data final rule about the release
of unencrypted prescriber identifiers in
PDE data to external entities are no
longer warranted.
In the proposed rule, in conjunction
with our proposal to broaden the release
of unencrypted prescriber identifiers,
we also highlighted our response to a
comment discussed in the Part D data
final rule, which argued that providing
access to linked physician identifiable
claims in order to pool them with
employer data would allow analysis to
reduce cost of care delivery and
improve the quality of care (73 FR
30676). We noted that in response to the
comment, we did not disagree with the
commenter, but referenced a variety of
pay for performance and value-based
health care initiatives being undertaken
by CMS at the time in an effort to
encourage health care providers to
furnish high quality health care and to
provide cost and quality information to
consumers. We also noted that in our
response to the comment, we had stated
that we intended to use PDE data in
those activities, but we declined to
adopt a policy that would include
making unencrypted prescriber
identifiers available for release to
external entities (except when needed to
link to another data set). In this
proposed rule, however, we
acknowledged that, in light of the goals
of the Affordable Care Act to improve
the quality of health care, including
through better access to information, we
now agree with the commenter
regarding the importance of providing
access to prescriber-identifiable claims
in order to allow researchers to pool
them with employer data and conduct
broader research.
We noted in the proposed rule that
our current policy on release of
ingredient cost and dispensing fee data
would not change under our proposal,
meaning the minimum necessary data
regarding ingredient costs and
dispensing fees would continue to be
available for release in disaggregated
form only to other HHS entities and
congressional oversight agencies. Non-
HHS executive branch agencies and
external entities could still only obtain
the minimum necessary ingredient cost
and dispensing fee data, and only in
aggregated form.
With respect to our proposal to
broaden the release of unencrypted plan
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00065 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29908
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
identifiers, we also explained in the
proposed rule that an analysis of Part D
plans, their network pharmacies, and
average drug costs, can already be
accomplished through data posted on
CMS’ Web site and/or purchased in
public use files. Additionally, the
Medicare Prescription Drug Plan Finder
(‘‘MPDPF’’) allows users to view and
compare all available prescription drug
plan choices, including plan and
pharmacy specific estimates of the costs
of individual drugs. These data can be
manipulated by researchers to reveal
information about specific plans and
pharmacies that contribute to the
evaluation and functioning of the Part D
program and can be used to improve the
public health. Therefore, in light of the
fact that plan data is already publicly
available and the public policy rationale
for increasing access to PDE data by all
categories of requestors, we stated that
plan identifiers should be available in
an unencrypted format.
For the same reasons that we
proposed to make prescriber and plan
identifiers available for release in an
unencrypted format, we explained in
the proposed rule that we no longer see
a reason that pharmacy identifiers
should not be available for release in an
unencrypted format. Accordingly, we
also proposed to release unencrypted
pharmacy identifiers to all categories of
requestors.
We addressed one final aspect of our
policies governing the release of Part D
data in the proposed rule. As discussed
previously, in the preamble to the Part
D data final rule (73 FR 30664), we
explained that consistent with CMS’s
existing policies with respect to Parts A
and B data, CMS would not release PDE
data for commercial purposes (but
external researchers may be funded by
commercial firms if the researchers are
free to publish their results regardless of
the findings). However, for the same
reasons that we proposed to make
changes to our rules governing the
release of PDE data, we also solicited
comment on the current restriction on
the release of PDE data for commercial
purposes. We noted that we were not
making a specific proposal in this
regard, but rather, that we wished to
receive comments on this issue for
consideration.
In addition to the proposed changes
with respect to prescriber, pharmacy,
and plan identifiers described
previously, and our request for
comment on the restriction on the
release of Part D PDE data for
commercial purposes, we proposed a
few other changes to our regulations
governing the submission, use, and
release of PDE data, including some
changes intended to clarify our existing
policies with respect to several issues
related to PDE data. First, we proposed
to add supporting program integrity
purposes, including coordination with
states, as an additional purpose deemed
necessary and appropriate by the
Secretary for which a Part D sponsor
must agree to submit all data elements
included in all its drug claims under
section 1860D–12(b)(3)(D) of the Act.
The regulation at § 423.505(f)(3)
currently contains a non-exclusive list
of purposes deemed necessary and
appropriate. Thus, we indicated that we
believe the use of these data for
supporting program integrity purposes
has always been included, even though
not explicitly listed. However, given the
importance of our ability to release PDE
data for program integrity purposes,
including for coordination with states
on program integrity, we proposed to
add this purpose explicitly to the non-
exclusive list in § 423.505(f)(3).
Second, we proposed to clarify that
non-final action data (for example,
information on claims subject to
subsequent adjustment) are available to
entities outside of CMS. We explained
that non-final action data are captured
through the data element, ‘‘Original
versus Adjusted PDE (Adjustment/
Deletion code).’’ We further explained
that this is a PDE field which
distinguishes original from adjusted or
deleted PDE records, which allows
sponsors to make adjustments to the
original PDE record to ensure accurate
payment. The information included in
these revised PDE records is thus not
point-of-sale data. With the increasing
focus on coordination of care, we noted
that requests for access to non-final
action PDE data have understandably
also increased, and that non-final action
data are also routinely requested for
evaluation and research projects. We
noted that the Part D data final rule (73
FR 30683) included an appendix that
explained in more specific detail the
restrictions relative to the available PDE
elements for the different categories of
requestors. Specifically, we noted that
this appendix stated (73 FR 30685) that
the data element ‘‘Original versus
Adjusted PDE (Adjustment/Deletion
code)’’ was available to other (that is,
non-CMS) HHS entities and the
congressional oversight agencies, while
for non-HHS executive branch agencies,
states, and external entities, it stated
that ‘‘Final Action claims would be
provided, so this element should not be
needed.’’ Thus, we noted that this
appendix did not explicitly address the
question of whether non-final action
data would be available for release to
non-HHS executive branch agencies,
states, and external entities, because
such data were not expected to be
needed by these requestors. However,
since it is clear that these entities do
need access to non-final data, we
proposed to clarify that non-final action
data are also available for release to non-
HHS executive branch agencies, states,
and external entities under the Part D
data final rule.
Due to our proposals to make changes
to our policies governing the release of
PDE data described previously, we
proposed to make corresponding
changes to the current applicable
regulatory text. In addition, we also
proposed to eliminate the appendix that
accompanied the Part D data final rule
(73 FR 30683) that explained in more
specific detail which PDE elements
would be available to different
categories of requestors, and any
restrictions that applied. We stated that
we believed this appendix is no longer
necessary, as our proposed changes in
policy would eliminate most of the
distinctions with respect to the PDE
data available for release to the different
categories of requesters, with the
exception of Total Drug Costs, which
will continue to be available in
disaggregated form only to other (that is,
non-CMS) HHS entities and the
congressional oversight agencies, and
we proposed to revise the regulation at
§ 423.505(m)(1)(iii)(B) to account for
this distinction. We also proposed to
revise this provision to clarify that we
will continue to exclude sales tax from
the aggregation, if necessary for the
project. Finally, we proposed changes to
the regulatory text to incorporate notes
from the current Appendix that are not
addressed by the existing reference to
CMS data sharing procedures in
§ 423.505(m)(1)(ii).
We received the following comments
on our proposed revisions to the
regulations governing the release of Part
D data:
Comment: We received a number of
comments regarding these proposed
revisions, many of which strongly
supported our proposed revisions to the
Part D data regulations. Several
commenters commended CMS’ ongoing
work to improve the efficiency of the
Medicare program and the clinical care
of its beneficiaries, which these
commenters asserted will be better
facilitated through increased data
transparency that facilitates additional
research. These commenters stated that
releasing unencrypted physician, plan,
and pharmacy identifiers in Part D PDE
data under the parameters we proposed
will allow researchers to answer a
broader range of questions about the
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00066 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29909
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
program. These commenters further
stated that greater access to Part D PDE
data will help ensure that this data is
used to maximum effect in the creation
of knowledge and understanding about
the program and around clinical care
received by beneficiaries. Commenters
additionally noted that the increased
availability of PDE data will enable
researchers to conduct in-depth
comparisons of medications provided
through different outlets, which could
enable CMS to take proactive measures
to achieve cost savings. One commenter
also stated that the public has a
significant interest in provider, plan,
and pharmacy professional conduct, as
these entities are government licensed
and regulated, and Medicare payments
are publicly funded. Finally, these
commenters noted that our existing
‘‘minimum necessary’’, ‘‘legitimate
researcher’’, and ‘‘aggregation’’ policies
are sufficient to provide some common
sense parameters for release of
unencrypted identifiers.
Potential areas of research suggested
by the commenters were linking
information on Part D plan features
(such as premiums, cost-sharing, and
formularies) to health outcomes and the
quality of health care provided to
Medicare beneficiaries. Other
commenters asserted that broader access
to prescriber, plan, and pharmacy
identifiers in PDE data will facilitate
research in particular for conditions for
which there are very few viable
treatments, no available cure, and much
more work to be done with respect to
researching and developing safe and
effective medicines. These commenters
welcomed the availability of additional
information to spur further knowledge,
investigation, and progress on how to
best treat—and ensure appropriate
coverage for treating—complex health
conditions.
Response: We thank these
commenters for their comments in
support of our proposed changes.
Comment: Other commenters opposed
our proposed revisions to the policies
governing the release of Part D data.
These commenters asserted that the
existing framework for release of Part D
PDE data fully accommodates the needs
of government entities and legitimate
researchers, and strikes the appropriate
balance between these needs and the
legitimate concerns of health care
providers and Part D plan sponsors
regarding the widespread dissemination
of sensitive data, including data that
specifically identifies them. One
commenter stated that CMS had not
articulated a reason for the need to
identify specific plans, pharmacies, and
prescribers, and that necessary research
can be accomplished with encrypted
identifiers. One commenter requested a
clarification on the meaning of ‘‘new
health care environment.’’ Some
commenters asserted that prescriber,
plan, and pharmacy identifiers in PDE
data are commercially sensitive
information, and that release of these
identifiers would undermine
competition and may lead to higher
costs in the Part D program and less
choice. A few of these commenters
asserted similarly that prescription drug
benefit plans could potentially reverse
engineer competitively sensitive data
regarding other plans, which could have
an anti-competitive effect on the Part D
bidding process.
Response: We think the preamble to
the proposed rule provided a clear
description of the ways in which the
Affordable Care Act is transforming the
health care system in this country—by
spearheading the drive toward an
information- and value-based system,
and the compelling reasons for the
release of unencrypted prescriber, plan,
and pharmacy identifiers in Part D PDE
data to allow for additional research to
achieve this goal. Specifically, it is in
the interest of public health to share this
information with entities outside of
CMS, as the work of these entities will
assist CMS in evaluating the Medicare
Part D program and assessing related
policies to improve the clinical care of
beneficiaries. We also note that when
more data is released about the
Medicare Part D program, the potential
research topics expand as well. For
instance, commenters supportive of the
proposed expansion in the release of
Part D data offered examples of
potential areas of new research, such as
linking information on Part D plan
features (such as premiums, cost-
sharing, and formularies) to health
outcomes and the quality of health care
provided to Medicare beneficiaries.
Such research is not possible with
encrypted plan identifiers, because the
researchers would not know the specific
features of the unidentified plans.
In addition, we are not persuaded that
these identifiers are commercially
sensitive data. As we stated in the
preamble to the proposed rule (79 FR
1990), an analysis of Part D plans, their
network pharmacies, and average drug
costs, can already be accomplished
through data posted on CMS’ Web site
and/or purchased in public use files.
Additionally, the MPDPF allows users
to view and compare all available
prescription drug plan choices,
including plan and pharmacy specific
estimates of the costs of individual
drugs. Moreover, we noted that it
appears that prescriber data are already
available commercially from pharmacy
data aggregators. These data can
currently be manipulated by researchers
to reveal information about specific
plans, pharmacies and prescribers. For
these reasons, we have concluded that
prescriber, plan and pharmacy
identifiers are not commercially
sensitive information, and that it is
appropriate to share this information in
an unencrypted format when it is
needed for a particular study or project.
Comment: Some commenters
supported our assertion in the proposed
rule that release of unencrypted
identifiers in Medicare Part D PDE data
subject to our current data release
policies, including our minimum
necessary and legitimate research
policies, will not result in data
recipients using the data
inappropriately, such as to influence
physicians’ prescribing patterns or
interfere with physicians’ professional
judgment. These commenters stated that
physicians are trained to use their best
medical judgment in making
prescription decisions for their patients.
Other commenters disagreed,
asserting that the release of unencrypted
identifiers has the potential, for
instance, to influence prescribing
patterns and physician judgment, or
otherwise to be used to draw incorrect
or inaccurate conclusions that could be
damaging to the reputations of
professionals and health care
organizations. These commenters
asserted that inappropriate influence
may adversely affect the quality of care
for beneficiaries. One commenter stated
that the Affordable Care Act’s additional
reporting requirements with respect to
physician prescribing do not address
this type of influence, and that CMS has
assumed that release of this data will
not adversely affect beneficiaries, rather
than carefully considering the impact of
release. Another commenter stated that
data and statistics are valuable in
observing trends among patient
populations, but that they are a blunt
instrument when applied to individuals.
One commenter opposed the
indiscriminate release of data to any
requesting external entity, including to
data aggregators that have little
knowledge of the Medicare Part D
program. A few commenters encouraged
CMS to present the data in a way that
considers the quality of the services
provided, including an explanation of
the data limitations, and allows for the
opportunity to correct information, for
instance, to include patient non-
compliance in the case of release of
prescriber identifiers. Finally, these
commenters stated that disputing
inaccurate findings takes significant
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00067 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29910
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
time, effort, and expense, and even then,
it is often impossible to fully mitigate
the harm caused.
Response: While we are sensitive to
the concerns regarding undue influence
raised by the commenters, for the
reasons discussed in the proposed rule,
we agree with those commenters that
did not believe releasing these data
would result in improper influence on
physician prescribing patterns or
otherwise interfere with the exercise of
professional judgment. In addition, we
believe CMS’ current release policies
will also limit inappropriate use of the
data. In order for a researcher to gain
access to CMS data, the researcher must
submit a research protocol and receive
approval of the protocol from CMS. In
addition, all requestors are required to
sign a Data Use Agreement with the
agency that limits the use of the data to
only the approved purposes. The agency
carefully considers all data requests to
ensure that the use of the data will not
exploit or negatively impact Medicare
beneficiaries. Furthermore, we do not
believe the professional research
community would support the
dissemination of faulty analyses and
would be quick to offer criticisms of
poor research, should this happen
despite our careful evaluation of all data
requests. We also disagree that data and
statistics are only valuable in observing
trends among patient populations. As
we lead the effort to provide high
quality care and better health at lower
costs, data analysis at various levels of
specificity is crucial. For example,
analyses at the provider or supplier
level, when properly adjusted to
account for differences in patient
populations, could provide insight into
differences in the way a given condition
is treated and help develop best
practices. In addition, unencrypted
prescriber identifiers have valuable uses
beyond reporting on individual
physician prescribing patterns. For
example, unencrypted identifiers in Part
D data can be linked to other sources of
data, such as claims data from other
payers, electronic health records, and
clinical data such as lab results, in order
to facilitate broader and more complex
research projects.
Additionally, we were not persuaded
that CMS should release data in a way
that considers the quality of the services
provided, includes an explanation of the
data limitations, or allows for the
opportunity to correct information. This
is precisely what professional
researchers do, and as we previously
noted, we think the professional
research community would be quick to
offer criticisms of poor research, should
a project fail to address these issues
appropriately. Moreover, if CMS were to
analyze data before its release for
research, this practice would undermine
the independent nature of the analyses
performed by outside researchers.
Comment: Some commenters
specifically supported the release of
non-final action PDE records, asserting
such data would permit researchers to
explore data for a better understanding
of the Medicare Part D program. The
comments included a specific example
of how non-final action data can assist
researchers in exploring prescription
adherence and abandonment by tracking
and accounting for adjusted or deleted
prescriptions. In contrast, other
commenters specifically opposed the
release of non-final data, asserting that
this information can easily be
misinterpreted and may cause false
conclusions that impact providers. One
commenter opposed our proposed
clarification regarding release of non-
final action data stating that CMS had
failed to articulate a reason for releasing
non-final action data other than that it
had received requests for it.
Response: We disagree with the
commenter that asserted that CMS failed
to articulate a reason for releasing non-
final action data. We think we did
articulate a reason for releasing non-
final action data. It is the same as the
overarching reason to release Part D PDE
data, which we discussed at length in
the proposed rule. Specifically, it is in
the interest of public health to share this
information with entities outside of
CMS, as research conducted by these
entities may assist CMS in evaluating
the Medicare Part D program and
assessing related policies to improve the
clinical care of beneficiaries. In
addition, as we stated in the preamble
to the proposed rule, the release of non-
final action data is necessary due to the
increased focus on coordination of care
in the Medicare program and indeed in
the health care system as a whole.
Comment: One commenter opposed
the proposal to allow release of
unencrypted prescriber and pharmacy
identifiers stating that the current
release policies already allow external
entities to link unencrypted identifier
elements to another data set.
Response: We disagree. Under our
current regulations, the identifiers must
be re-encrypted after the link has been
made. Under the regulations as
finalized, once linked, research at
higher levels of specificity can be
conducted that is not possible with
encrypted identifiers. For instance, a
researcher conducting a study on
medication adherence will have many
more factors to consider and explore
when the prescribers, plans, and
pharmacies involved in the research are
identifiable. Is adherence related to plan
features? Physician location and/or
specialty? Pharmacy organization filling
the prescription? All three? The
research possibilities will expand, as the
additional connections that can be
explored by researchers expand. Drilling
down to higher and higher levels of
specificity to understand and
potentially solve a problem is the very
nature of 21st century data-driven
research, and we believe it is essential
that the Part D data release policies keep
current.
Comment: One commenter opposed
our proposal to release unencrypted
plan identifiers, asserting that plan
information is gathered only for Part D
administration purposes.
Response: We disagree. Section
423.505(f)(3) of the regulations states
that a plan sponsor agrees to submit to
the Secretary all data elements for
purposes deemed necessary and
appropriate by the Secretary, including,
but not limited to conducting
evaluations of the overall Medicare
program. As we noted previously,
commenters offered examples of
potential areas of research that will be
enabled through access to unencrypted
plan identifiers, such as linking
information on Part D plan features
(including premiums, cost-sharing, and
formularies) to health outcomes and the
quality of health care provided to
Medicare beneficiaries.
Comment: One commenter stated that
CMS should more specifically define
‘‘legitimate researcher’’ to ensure that
Part D data is not released for
competitive or commercial purposes
contrary to CMS’ current policy.
Response: Under current CMS data
sharing policies, the agency evaluates
all research requests to ensure that the
researcher has the expertise to conduct
the proposed study. In addition, we
must approve the research protocol
before any data is shared with a
researcher. We believe that this review
process contains appropriate safeguards
to prevent inappropriate use of the data
and, as such, we do not believe it is
necessary to define a ‘‘legitimate
researcher.’’ Furthermore, we believe a
variety of different types of individuals
could submit a valid research request.
Comment: Some commenters
supported our proposal to broaden the
release of Part D data, so long as
beneficiary privacy is protected. One
commenter suggested that a bio-
statistician conduct an expert review of
the data sets to be released in the
context of the permitted use(s) to ensure
beneficiary privacy in the context of the
permitted uses of the data. Another
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00068 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29911
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
commenter stated that our proposed
expansion of the available data will
compromise beneficiary privacy and
requested that an approval process
similar to an IRB be established to
evaluate requests for such information
to weigh the risks and benefits of
disclosure. Another commenter stated
that CMS should ensure that its efforts
to protect beneficiary confidentiality do
not create such onerous data request
processes that qualified researchers are
discouraged from attempting to access
Part D data. Another commenter stated
that CMS should establish and impose
appropriate penalties for any breach of
privacy related to beneficiary
identifiable information.
Response: All users accessing
beneficiary identifiable data are
required to sign CMS’ Data Use
Agreement (DUA), which addresses
privacy and security for the data CMS
discloses. In addition, the DUA
currently does, and will continue to
have, enforcement mechanisms,
including criminal penalties. CMS
would make use of these provisions in
the event of any breach or violation of
the terms of the DUA. The DUA also
contains provisions regarding access to
and storage of CMS data to ensure that
beneficiary identifiable information is
stored in a secure system. We believe
these restrictions are necessary in order
to ensure that data is only requested in
compliance with the requirements of the
regulations and CMS data sharing
procedures, and that data shared by
CMS is appropriately protected and is
not reused or redisclosed without the
necessary approval. Given that
researchers have successfully been
accessing to CMS data under the terms
of this DUA for years, we do not believe
these requirements are too burdensome.
With regard to the suggestion that CMS
have a bio-statistician review the data
sets to be released to ensure beneficiary
privacy, we do not believe this is
necessary given the beneficiary privacy
protections in the DUA. However, to the
extent that CMS releases any de-
identified, summarized data sets based
on the Part D data, the agency carefully
reviews the proposed release to ensure
that it does not put beneficiary privacy
at risk. Finally, we disagree that the
expansion of the available data will
compromise beneficiary privacy or that
additional procedures are necessary in
order to safeguard beneficiary privacy.
CMS has an established process to
evaluate requests for data to ensure that
there are appropriate safeguards in place
to protect beneficiary privacy. We
believe this process contains the
necessary checks to ensure that the risks
of the disclosure are minimal.
Comment: One commenter stated that
CMS should be as transparent as
possible under its data use agreements,
asserting that the public, as well as the
parties involved, must be able to readily
determine the manner in which data are
released, the purpose for the release of
the data and the parties to whom the
data are released.
Response: We are strongly committed
to transparency. In particular, we have
established processes to ensure that
beneficiaries can request information
about to whom their protected health
information or personally identifiable
information has been disclosed, as well
as the purpose for the release of the
data. Beneficiaries interested in
requesting access to this information
should contact the CMS Freedom of
Information Act (FOIA) Office (http://
www.cms.gov/center/freedom-of-
information-act-center.html).
Comment: One commenter stated that
CMS should consider whether a
proliferation of analyses of outdated
Part D data will truly benefit the Part D
program, when CMS has the ability to
commission studies and data analysis
that would more knowledgeably take
into account a comprehensive
understanding of the continually
changing dynamics of the Part D
prescription drug market.
Response: We use Part D data to
conduct a variety of studies and
analyses. However, this work does not
even begin to cover the scope of
possible analyses that could be
performed using Part D data. We believe
that by limiting Part D data analysis to
that supported by CMS, the agency
would be inhibiting important research
and innovation that has the potential to
result in higher quality care at lower
costs in the Medicare Part D program,
and indeed for all Americans.
After review of the comments we
received, we are finalizing our proposed
changes to the regulations governing the
release of Part D data. Specifically, we
are finalizing the following revisions to
the applicable regulatory text:
• Section 423.505(f)(3) is revised to
add supporting program integrity
purposes, including coordination with
states, as an additional purpose.
• Section 423.505(m)(1)(iii) is revised
to remove references to encrypting
certain identifiers since prescriber, plan,
and pharmacy identifiers are no longer
be subject to encryption when released.
• Section 423.505(m)(1)(iii)(A) is
revised to clarify that, subject to the
restrictions contained in paragraph
(m)(1), all elements on the claim are
available not only to HHS, but also to
other executive branch agencies and
states, since there is no longer any
distinction between the two categories.
• Section 423.505(m)(1)(iii)(B) is
revised to incorporate a note from the
appendix that is being eliminated,
which states: ‘‘Upon request, CMS
excludes sales tax from the aggregation
at the individual level, if necessary for
the project’’ at the end of the provision.
• Section 423.505(m)(1)(iii)(C) is
deleted as no longer necessary since
unencrypted plan identifiers, including
the internal plan/pharmacy
identification numbers, are available for
release.
• Section 423.505(m)(1)(iii)(D) is re-
lettered as (C) and references to
encryption of pharmacy and prescriber
identifiers are deleted, since these
identifiers are available for release in
unencrypted format. Additional
language regarding beneficiary
identifiers is added to reflect the current
policy on release of this identifier. In
addition, we are including the
statement, ‘‘Public disclosure of
research results will not include
beneficiary identifying information,’’ at
§ 423.505(m)(1)(iii)(C)(2), which also
reflects current policy as described in
the appendix that is being eliminated.
• Section 423.505(m)(3) re-lettered as
(m)(3)(i) and (m)(3)(ii) is added to
incorporate a note from the appendix
that is being eliminated about the status
of the Congressional Research Service as
an external entity when it is not acting
on behalf of a Congressional committee
in accordance with 2 U.S.C. 166(d)(1).
With respect to our policy not to
release Part D data for commercial
purposes, we did not make a specific
proposal but solicited comments for
general consideration. We received
comments on both sides of this topic,
and thank all the commenters. The
following is a summary of the
comments:
Comment: Commenters that desire a
change in the policy applauded CMS for
soliciting comment on this topic. These
commenters stated that in order to
improve and modernize the U.S. health
care system, greater alignment of
stakeholder incentives is required, and
that CMS is keenly aware of this pivotal
requirement for success. These
commenters stated that the challenge of
quantifying greater efficiency and
evidence of improvement as part of
overall health reform requires more
access to the unique data sets in federal
data, and that the current restriction on
the use of these data for commercial
purposes will grow increasingly
challenging in the future as Medicare
employs more value-based payment
incentives, and as Medicare data are
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00069 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29912
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
included in broader multi-payer sets,
such as those being established by the
Patient-Centered Outcomes Research
Institute. These commenters further
stated that because the quality and
efficiency of all physician groups,
health plans, hospital systems, and
other providers and suppliers can be
enhanced using data, any notion that
commercial interest is limited and
discrete is outdated. They stated that
PDE data is a valuable asset to all types
of commercial health care entities in
limiting the incidence of fraud,
obtaining practice pattern feedback,
managing health care delivery to deliver
value, developing best practice
standards, and conducting comparative
effectiveness research.
The commenters also stated that
eliminating or reducing this current
restriction on the release of data for
commercial purposes is consistent with
CMS’ desire to foster transparency and
competition in the Part D program
during a period of sweeping change to
the health care system. These
commenters asserted, for example, that
suppliers need to incorporate accurate
data into their product pricing and
discounting strategies to align their
approaches with the system-wide drive
toward value-based decision-making
and high quality care.
Commenters additionally stated that
the more access there is to Medicare
data, the more dramatically the
bandwidth for research will be
increased, leading to increased quality
of care, system efficiency, and consumer
satisfaction in the Medicare programs
and health care system in general. These
commenters noted that there is deep
scientific and analytic expertise within
organizations that are currently
excluded from accessing CMS data.
These commenters asserted that the
standard for data release by CMS should
be whether the research proposed is of
high quality and whether it has the
potential to improve program
administration or the health of the
covered population, rather than
financial benefit and profit status of the
organization proposing the research.
These commenters further noted that
CMS has in place strong research merit
criteria, rules, and obligations for data
use and individual privacy protections,
and that these processes and this
oversight are sufficient to determine
whether a requestor should have access
to PDE or other identifiable data,
regardless of the researcher’s affiliation.
Some commenters stated that broader
release of Part D data would not only
further public health research and
analysis of the Part D program, but also
would serve to further educate
consumer organizations, patient
advocates, and ultimately beneficiaries
about the program generally, as well as
coverage and prescribing patterns under
various plans.
Some commenters stated that they
would support changing the policy on
non-release of Part D data for
commercial purposes, so long as CMS
ensured that release of the data would
be conditioned on its use for
improvement of one or more aspects of
the Part D program, and CMS carefully
screened potential recipients of the data
for demonstrated expertise in using
research data to improve health
programs, as well as for any potential
conflicts of interest or other concerns.
Commenters that believe that the
policy of non-release for commercial
purposes should remain unchanged
stated that health care entities have
legitimate concerns regarding the
widespread dissemination of sensitive
data, such as data that specifically
identifies them. These commenters also
stated that strong program oversight and
public health and public policy
imperatives do not exist to
counterbalance these concerns.
One commenter stated that CMS lacks
the authority to release Part D data for
commercial purposes, because the
authority cited by CMS limits releases to
those required for program purposes
and for improving public health. The
same commenter asserted that the right
to make data available for purely
commercial reasons is a right inherent
in the ownership of the data, and that
CMS has never previously asserted an
ownership over, or right to control the
use of, data not obtained through access
to a CMS system. This commenter stated
that by granting itself this right to
release Part D PDE data for purely
commercial purposes, CMS would be
exercising a right inherent in ownership
of the data.
In light of all the comments received
on both sides of this particular topic, we
continue to believe that the best
approach is for our policy regarding the
release of Part D data for commercial
purposes to remain consistent with the
policies for the release of data from
Medicare Parts A and B. As we
discussed, in the Part D data final rule
(73 FR 30672), the procedures that we
use to make Part D data available are
built upon the practice that was already
in place with respect to the release of
Part A and B data. Furthermore, absent
specific reasons for treating the data
differently, we believe it is appropriate
to have consistent policies for the
release of data across Medicare Parts A,
B, and D. Therefore, although we are not
changing our policy against releasing
Part D data for commercial purposes at
this time, we note that in the event the
policy regarding the release of Parts A
and B data for commercial purposes
were to change, we would also revise
our Part D data sharing policies to be
consistent with that change.
23. Establish Authority To Directly
Request Information From First Tier,
Downstream, and Related Entities
(§§ 422.504(i)(2)(i), and 423.505(i)(2)(i))
Under section 1857(d)(2) and 1860D–
12(b)(3)(c) of the Act, existing
regulations at §§ 422.504(i) and
423.505(i) establish various conditions
that entities contracting as a first tier,
downstream, or related entity (FDR) to
an MA organization or Part D sponsor
must agree to in order to participate in
the MA or Part D program. One such
condition at §§ 422.504(i)(2)(i) and
423.505(i)(2)(i) is that HHS, the
Comptroller General, or their designees
have the right to audit, evaluate, and
inspect any books, contracts, computer
or other electronic systems, including
medical records and documentation of
the first tier, downstream, and related
(FDR) entities related to CMS’ contract
with the Part C and D sponsor.
CMS (or its designee(s)) conduct
routine audits of Part D sponsors and
MA organizations, as well as conduct
audits to investigate allegations of
noncompliance with Part C and/or Part
D rules and requirements. While
§§ 422.504(d) and 423.505(d) address
Part D and MA organizations’ own
maintenance of records and the rights of
CMS to inspect those records,
§§ 422.504(i)(2)(i) and 423.505(i)(2)(i)
also require plan sponsors to require
their FDRs to agree to a CMS right to
inspection. Plan sponsors regularly
contract with FDRs to perform critical
Part C and D operating functions. For
example, many (if not most) Part D
sponsors delegate critical Part D
functions to their PBMs. As a result,
many of the records that we or our
designees would need to review and
evaluate when we audit a Part D
sponsor or MA organization reside with
its FDRs.
Our existing regulation at § 423.505
(i)(3)(iv) states that the contracts
between the Part D sponsor and its FDRs
must indicate whether records held by
the FDR pertaining to the Part D
contract will be provided to the sponsor
to provide to CMS (upon request), or
will be provided directly to CMS or its
designees by the FDR (the Part C
regulation is silent on this matter). As
such, we have not previously required
Part C or Part D FDRs to provide
information directly to CMS.
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00070 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29913
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
Two separate reports by the OIG
(OEI–03–08–00420, dated October 2009
and OEI 03–11–00310, dated January
2013), have highlighted barriers
experienced by the Medicare Drug
Integrity Contractor (MEDIC), the entity
contracted by CMS to be responsible for
detecting and preventing fraud, waste,
and abuse in the Medicare Parts C and
D programs nationwide, in obtaining
requested information in an expeditious
manner. The 2009 OIG report discussed
that CMS’ and its designees’ (in this
case, the MEDIC) lack of authority to
directly obtain information from
pharmacies, PBMs, and physicians has
hindered the MEDIC’s ability to
investigate potential fraud and abuse
and the OIG recommended that CMS
change its regulations to establish its
authority to obtain necessary
information directly from FDRs. The
OIG’s 2013 report reiterated the
recommendation that CMS have a more
direct route to obtain records held by
FDRs so that CMS would be able to
obtain necessary records in a timely
fashion. While the 2013 report pointed
out that sponsors and their FDRs
generally cooperate in providing the
information requested by the MEDIC, it
often takes months for it to reach the
MEDIC because the MA organization or
Part D sponsor acts as a gatekeeper.
In the past, we chose not to be
prescriptive regarding whether a first
tier, downstream, or related entity must
make its books and records available to
us directly or through the Part C or D
sponsor. As a consequence of what we
have learned through the OIG
investigations and the seriousness with
which we approach our fraud, waste,
and abuse oversight obligations, we
proposed to specify at
§§ 422.504(i)(2)(ii) and 423.505(i)(2)(ii)
that HHS, the Comptroller General, or
their designees have the right to audit,
evaluate, collect, and inspect any
records by obtaining them directly from
any first tier, downstream, or related
entity.
We further proposed to revise the
regulation at §§ 422.504(i)(2)(i) and
423.505(i)(2)(i) to make clear that CMS
and its designees may ‘‘collect’’ records,
in addition to our existing authority to
‘‘audit, evaluate, and inspect’’
information. The addition of ‘‘collect’’
removes any doubt that, in addition to
our other options for obtaining records,
we have the authority to request
information to be reviewed in some
location other than onsite at a sponsor’s
or FDR’s facility. Furthermore, this
proposed provision is intended to
clarify only that CMS may contact FDRs
directly and request that they provide
Part C or D-related information directly
to CMS. The question as to whether
CMS has the authority to enter the
premises of FDRs is to be determined by
interpreting other applicable statutory
and regulatory authority.
Finally, we also proposed to delete
the existing provision at
§ 423.505(i)(3)(iv) which gives Part D
sponsors the choice as to how
information sought from their FDRs will
be provided to CMS. Section 423.505
would be renumbered so that
paragraphs (v) through (viii) would
become paragraphs (iv) through (vii).
We received the following comments
and our responses follow:
Comment: Several MA organizations,
Part D sponsors, and industry
associations (about a dozen
organizations all together) raised their
opposition to this proposal. The main
argument made by these parties is that
CMS lacks the legal authority to directly
access information from FDRs since our
contractual relationship in these
situations is with the MA organizations
and PDP sponsors, not the FDRs
themselves. One physician’s group
raised the concern that this provision
would increase the likelihood of audits.
Response: We acknowledge that we
do not have the authority to directly
regulate FDRs. However, we believe our
proposal allows us to achieve our goal
of securing the right of access to FDR
records because it relies on our ability
to require the sponsors to incorporate
such a right of access as part of the
contractual relationship between the
sponsors and their FDRs. That is, the
proposal requires MA organizations and
PDP sponsors to require their FDRs to
agree to respond to direct requests for
information made by CMS. As a
practical matter, the mechanism for
sponsors to require their FDRs to agree
to such a provision is through the
contractual relationship between the
parties. We do not believe that this
proposal in any way changes the
likelihood of an audit, but merely affects
the flow of information which we would
be otherwise requesting.
Comment: The MA organizations and
PDP sponsors that commented also
maintained that since they are
ultimately accountable to CMS for the
work performed under their Medicare
contracts, they have the right to
participate in and manage the
information provided to their
contracting partner (CMS) on their
behalf by their FDRs. The MA
organizations and PDP sponsors further
asserted that allowing the MEDIC or
other government-related entities to
circumvent the plan sponsors would
lead to the collection of potentially
erroneous information since the plan
sponsor would not have the opportunity
to properly vet the information flowing
from the FDR to CMS. As a result, CMS’
and the MEDIC’s information collection
from FDRs may become less efficient
than it is under the current regulatory
regime. Several commenters argued that
the proposal would in fact increase
burden, in contrast to CMS’ suggestion
in the proposed rule that the proposal
would reduce burden.
Response: We maintain that having
direct access to information from FDRs
is an essential tool in combating fraud,
waste, and abuse which we should be
authorized to use. That said, we
appreciate concerns raised about MA
organizations and PDP sponsors’
interests in managing information
flowing to CMS, and the concern that
such information could at times be
flawed or erroneous without the quality
review performed by the MA
organization or PDP sponsor.
Consequently, we wish to clarify that
CMS and the MEDICs will default to the
current practice of requesting
information held by FDRs via an initial
request to the MA organization or PDP
sponsor. However, we will use the
‘‘direct access’’ route in circumstances
where either (a) the results of data
analytics, complaints, and/or
investigations indicate a suspicion of
fraud, waste, or abuse in the Medicare
Part C or D programs or (b) in the case
of an urgent law enforcement matter.
We will publish sub-regulatory
guidance on CMS’ standards for
determining when direct requests of
FDRs would be appropriate. We believe
that this approach promotes CMS’ anti-
fraud efforts by increasing fraud
investigators’ access to critical Part C
and D program information and will
likely increase the speed with which
investigators may get access to critical
FDR information, but at the same time
allows for continued MA organization
and PDP sponsor control and review of
information in appropriate
circumstances. We also wish to provide
assurance that CMS’ contractor, the
MEDIC, would not be permitted to
independently determine under what
circumstances it would be appropriate
to bypass the MA organization or PDP
sponsor in favor of requesting
information directly from the FDR; CMS
would be directly involved in all such
determinations. This approach also
minimizes any loss of quality or
potential for errors in the requested
information as well as the placement of
any additional burden on sponsors or
FDRs.
Comment: Several commenters
requested that if CMS finalizes the
provision, that we revise the regulatory
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00071 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29914
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
language to state that CMS would notify
the MA organization or PDP sponsor
upon a direct request to one of its FDRs.
Response: While we had previously
stated in this final rule discussion that
MA organizations and PDP sponsors
would be notified when there is a direct
request for information made of an FDR,
we agree that it is reasonable for us to
specify this commitment in regulation.
As such, we have added at
§§ 422.504(i)(2)(iii) and 423.505(i)(2)(iii)
language stating that except in
exceptional circumstances, CMS will
provide notification to the MA
organization or PDP sponsor that a
direct request for information has been
made to one of its FDRs. The
exceptional circumstance exception is
included to allow for the possibility that
the MA organization or PDP sponsor
could be one of the parties to the fraud
investigation, in which case it may not
be appropriate to provide such
notification.
Therefore, we are finalizing this
provision with the modification that
CMS will provide notification to the MA
organization or PDP sponsor that a
direct request for information has been
made to one of its FDRs, except in
exceptional circumstances.
24. Eligibility of Enrollment for
Incarcerated Individuals (§§ 417.1,
417.460, 422.74, 423.44)
Entitlement and enrollment in the
Medicare program (Part A and Part B) is
contingent on entitlement to Social
Security retirement and disability
benefits as outlined in sections 226 and
226A of the Act, and enrollment in the
Medicare program for individuals not
receiving retirement or disability
benefits is outlined in sections 1818 and
1818A of the Act. These sections do not
preclude entitlement to or enrollment in
the Medicare program for individuals
who are incarcerated in prisons or other
penal facilities. However, section
1862(a)(3) of the Act excludes Medicare
payment for services which are paid
directly or indirectly by another
government entity, including federal,
state and local prisons, and penal
facilities. Given that Medicare
entitlement flows from entitlement to
Social Security retirement and disability
benefits, we established regulations at
§ 411.4(b) and implemented section
1862(a)(3) of the Act through a payment
exclusion process in the FFS program,
outlined in section 50 of Chapter 16 of
the Medicare Benefit Policy Manual and
section 10.4 of the Medicare Claims
Payment Manual.
The Medicare payment exclusion
process includes the receipt of
incarceration status for individuals via
regular data transfers from the SSA to
CMS. Once we receive the data, the
incarceration status is noted on the
individual’s record and is retained in
the FFS claims processing systems.
Upon receipt of submitted FFS claims,
CMS denies payment of both Part A and
Part B claims for individuals with
records on which incarceration is
denoted, subject to the narrow
exception provided in § 411.4(b). The
denial of claims continues until the
individual is no longer incarcerated and
that information is reported by SSA to
CMS. Individuals who are entitled to
premium-free Part A will maintain their
entitlement and will remain enrolled in
Part B as long as premiums are paid.
Similarly, individuals who are enrolled
in premium Part A and/or Part B
maintain their enrollment as long as
premiums are paid. Sections
1851(a)(3)(B), 1860D–1(a)(3)(A), and
1876(a)(1)(A) of the Act outline the
eligibility requirements to enroll in MA,
Part D, and Medicare Health
Maintenance Organization/Competitive
Medical Plans (cost plans). In all
options, individuals must have active
Medicare coverage. Specifically, to
enroll in MA, an individual must be
entitled to Part A and enrolled in Part
B; to enroll in a PDP, an individual must
be eligible for Part D by either being
entitled to Part A and/or enrolled in Part
B; to enroll in a Medicare cost plan, an
individual must be enrolled in Part B
but Part A is not required.
In addition, sections 1851(b)(1)(A),
1860D–1(b)(1)(B)(i), and 1876(d) of the
Act provide that Medicare beneficiaries
are eligible to enroll in an MA plan,
PDP, or cost plan only if they reside in
the geographic area served by the plan,
known as the plan’s ‘‘service area.’’ As
noted earlier, an individual who is
incarcerated still meets the eligibility
requirements for Part A and Part B and
is eligible generally to enroll in an MA
plan, PDP, or cost plan. However,
residence in a plan’s service area is also
a condition for eligibility to enroll in an
MA plan, PDP or cost plan. See
§§ 422.50(a)(3)(i) for MA plans,
423.30(a)(1)(ii) for PDPs, and 417.422(b)
for cost plans. If a member no longer
resides in the service area, plans must
disenroll that individual per rules at
§§ 422.74(a)(2)(i) and 422.74 (d)(4) for
MA plans, 423.44(b)(2)(i) for PDPs, and
417.460(b)(2)(i) for cost plans.
a. Changes in Definition of Service Area
for Cost Plans (§ 417.1)
In order to implement the exclusion
from Medicare coverage for incarcerated
individuals under section 1862(a)(3) of
the Act in the case of MA plans and
PDPs, we explicitly excluded facilities
in which individuals are incarcerated
from an MA plan’s service area by
including this exclusion in the
definition of ‘‘service area’’ when those
regulatory definitions were adopted (54
FR 41734 and 72 FR 47410).
Specifically, ‘‘service area,’’ under
§§ 422.2 for MA plans and 423.4 for
PDPs, is defined so that facilities in
which individuals are incarcerated are
considered outside of the service area.
We did not include a similar service
area exclusion in the case of cost plans.
To the extent that cost plans do not
incur costs for incarcerated enrollees
because their health care costs are
covered by the facility, there would be
no costs claimed on the cost report, and
therefore, no Medicare payment.
Nonetheless, to ensure that no cost
payments are made, we proposed to
revise the definition of service area in
§ 417.1 to specifically note that facilities
in which individuals are incarcerated
are not a part of the service area. This
adjustment will ensure parity among the
various Medicare plan coverage options
and be the basis for ensuring that
services are not paid by the Medicare
Trust Funds for those who are not
eligible for them.
b. Involuntary Disenrollment for
Incarcerated Individuals Enrolled in
MA, PDP and Cost Plans (§§ 417.460,
422.74, and 423.44)
Sections 1860D–1(b)(1)(B)(i),
1851(b)(1)(A), and 1876(a)(1)(A) of the
Act provide that individuals whose
permanent residence is outside the
plan’s service area are ineligible to
enroll in or to remain enrolled in the
MA, Part D, or cost plan. Based on the
definition of service area established in
§§ 422.2 and 423.4, this applied to
individuals who were incarcerated as
well. As such, individuals who became
incarcerated while enrolled were
ineligible to remain enrolled because
they did not meet the eligibility
criterion of residing in the MA plan or
PDP’s service area. As noted previously,
the regulations for cost plans currently
do not exclude incarcerated individuals
from enrolling or remaining enrolled in
these plans.
At the time of the implementation of
Part D, the data regarding incarceration
were not as robust as they are at the
present time. To compensate, we
provided instructions in sub-regulatory
guidance that required MA plans and
PDPs to investigate a notification from
CMS of an individual’s incarcerated
status. If a plan could not confirm an
enrollee’s status, the plan would then
apply the more-general policy for
investigation of a possible out-of-area
status, which would allow an
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00072 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29915
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
incarcerated individual to remain
enrolled in the plan for up to 6 or 12
months for MA plans or PDPs,
respectively. Cost plans, on the other
hand, are not currently subject to
similar instructions. Therefore
individuals are currently not
disenrolled from cost plans solely
because they are determined to be
incarcerated.
Given that the data CMS receives from
SSA today regarding the incarceration
status of Medicare beneficiaries are
reliable enough for the purpose of
involuntary disenrollment from MA,
Part D, and cost plans, we proposed in
the preamble of the January 2014 notice
of proposed rulemaking to amend
§§ 417.460(b)(2)(i), 417.460(f)(1)(i),
422.74(d)(4)(i), 422.74(d)(4)(v) and add
423.44(d)(5)(iii) and 423.44(d)(5)(iv) to
establish that MA organizations, PDPs,
and cost plan organizations must
disenroll individuals incarcerated for 30
days or more upon notification of such
status from CMS. Our proposal
indicated that CMS, as a part of this
change, would review the incarceration
data provided by SSA and, where
possible, involuntarily disenroll
individuals who are incarcerated based
on the data provided by SSA and notify
the plan in which the individual is
enrolled of this involuntary
disenrollment. For all such
disenrollments under our proposal, the
effective date of disenrollment would be
the first of the month after the
incarceration start date, as reported by
SSA. Such disenrolled individuals
would maintain Medicare Part A and
Part B coverage through FFS, provided
they continue to pay premiums, as
applicable, and payment of FFS claims
would be based upon existing
regulations outlined at 42 CFR 411.4(b).
In connection with this change, we also
proposed to deny enrollment requests
for individuals if data received by CMS
indicates an active incarceration status
of at least 30 days. Based on the data
received from SSA, if incarceration is
denoted, we will deny that enrollment
and notify the plan of the denial. This
would replace the current process
requiring plans to accept the enrollment
and immediately begin the process to
verify that the individual was out of the
plan’s service area. We indicated our
intent to provide operational
instructions in subregulatory guidance.
We received the following comments
on our proposal:
Comment: We received general
support for our proposals. Specifically,
commenters appreciated the
clarification that individuals released
from incarceration are eligible for a
special election period (SEP) to enroll in
an MA or Part D plan.
Response: We appreciate the support
expressed by the commenters. We note
that the SEP related to release from
incarceration (that is, change in
residence) is not new or tied to this
proposal. Details about this SEP can be
found in section 30.4.1 of Chapter 2 of
the Medicare Managed Care Manual and
section 30.3.1 of Chapter 3 of the
Medicare Prescription Drug Benefit
Manual.
Comment: A few commenters had
suggestions for how we should
implement this proposal. Specifically,
they suggested that we issue updated
guidance and develop new model
notices. They also suggested that the
best vehicle for providing updates to
incarcerated status on members would
be through the MARx system or daily
transaction reply reports (DTRRs).
Response: We agree that manual and
operational guidance will be necessary
in order for MA, Part D and cost plans
to implement this provision
appropriately. We will evaluate whether
new or revised model notices are
needed and we will share these with
plans as soon as possible. We also agree
that transmission of data through MARx
and DTRRs would make the most sense
in terms of sharing incarcerated status
with plans.
Comment: A commenter requested
that CMS notify MA organizations and
Part D sponsors of involuntary
disenrollments on the day of
incarceration. This commenter also
suggested that we consider permitting
MA and Part D plans to disenroll
members as of the incarceration start
date (as opposed to the first day of the
month following the incarceration start
date) to be in line with rules governing
Qualified Health Plans (QHPs).
Response: Notification to plans and
sponsors on the day that incarceration
begins is not possible, since CMS
receives the data from SSA once a
month, and only after the correctional
facility provides it to SSA. We would
also note that plan enrollment and the
corresponding payment to plans by
CMS occurs in full calendar month
increments. Even if we were able to
provide plans with real time
incarceration data, an involuntary
disenrollment date other than the last
day of the month is not possible.
We understand that QHPs may have
different disenrollment effective dates
because they can disenroll on days other
than the first of the month. However, as
previously stated, MA, Part D and cost
plan effective dates begin and end on a
monthly basis (that is, the first day of
the month). Therefore, we cannot use
the date of incarceration as the
disenrollment effective date.
Comment: A commenter requested
that we clarify if there will be an option
for plans to disenroll a member if they
receive information from the State
Medicaid agency that an individual is
incarcerated.
Response: If a plan receives
information from an entity other than
CMS or receives from CMS, via existing
MARx processes, an indication of
possible out of area status due to
incarceration, there is already a process
outlined in sub-regulatory guidance for
plans to determine whether an
individual is residing outside of the
service area, which is what
incarceration is considered. For cases in
which CMS does not receive data
confirming the incarceration of the
individual, the MA organization or Part
D sponsor must establish that the
individual is no longer residing in the
plan’s service area due to incarceration
as outlined in Section 50.2.1 in Chapter
2 of the Medicare Managed Care Manual
and Chapter 3 of the Medicare
Prescription Drug Benefit Manual.
In addition, as outlined in Section
50.2.1 in Chapter 17, Subchapter D of
the Medicare Managed Care Manual,
cost plans must disenroll individuals
who permanently move out of the
service area based upon written
statement from the beneficiary or other
reasonable evidence that establishes the
individual no longer resides in the
plan’s service area. With the change in
definition of service area for cost plans
as reflected in the proposed change at
§ 417.1, cost plans must establish that
the individual is no longer residing in
the plan’s service area if they receive
information regarding incarceration
from CMS or another entity.
Comment: Two commenters suggested
creating a Part B SEP to ease the
transition for beneficiaries after they are
released from incarceration to ensure
access to Medicare Part B benefits as
they re-enter the community.
Oftentimes, the commenters cited, these
beneficiaries lose their Medicare Part B
coverage because they are unable to pay
their premiums during their
incarceration and are not eligible for a
Part B SEP upon their release. As a
result, if these individuals sign up for
Part B at a later date, there is the
likelihood that they will have to pay a
late enrollment penalty.
Response: This comment is outside
the scope of this rulemaking. However,
we would like to note that SEPs for Part
B and premium Part A are outlined in
statute and CMS does not have the
authority to establish additional SEPs.
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00073 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29916
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
1
Ali Shirvani-Mahdavi, Ph.D. & Melissa Haeffner,
Ph.D., Rewarding Wellness: The Science Behind
Effective Wellness Incentive Programs (2014).
After consideration of the public
comments received, we are taking the
following action on our proposals:
• The definition of service area for
cost plans at § 417.1 is finalized without
modification.
• To articulate that the geographic
area is the HMO or CMP’s service area
as defined in § 417.1, we are finalizing
the language at § 417.460(b)(2)(i) with
the minor modification of adding the
word ‘‘service.’’
• To articulate that the basis of the
disenrollment for incarceration is due to
the individual not residing in the plan’s
service area, the regulation text at
§§ 417.460(f)(1)(i), 422.74(d)(4)(i)(A),
and 422.74(d)(4)(v)) is finalized with
modification.
• Due to an inadvertent omission, the
proposed regulatory text changes to
§ 423.44(d)(5)(iii) and (iv) were not
published in the proposed rule. Because
our preamble was clear that our
proposed changes were applicable to
Part D, and the comments received
demonstrated that readers understood
our intent, we are adding and finalizing
regulatory text changes at
§ 423.44(d)(5)(iii) and (iv).
• A proposed change to the definition
of ‘‘service area’’ was inadvertently
published in the January 2014 proposed
rule at § 422.2. That revised definition is
not being finalized.
Finally, we recognize that in our
discussion of the proposed rule we
described our intent that ineligibility
for—as well as involuntary
disenrollment from—MA, Part D, and
cost plans would be based on a period
of incarceration of 30 days or more. As
we will note in implementing guidance
for these final rules, we will determine
eligibility based on confirmed
incarceration data from SSA, not a 30-
day timeframe.
25. Rewards and Incentives Program
Regulations for Part C Enrollees
(§ 422.134)
Every year, CMS receives inquiries
from MA organizations that wish to
expand the scope of the rewards and
incentives that currently may be offered
to beneficiaries enrolled in their MA
plans. In some cases, MA organizations
wish to extend rewards and incentives
already offered to their commercial
members to their Medicare enrollees.
There is some evidence to suggest that
health-driven reward and incentive
programs for currently enrolled
members of health plans may lead to
meaningful and sustained improvement
to their health behaviors and health
outcomes.
1
CMS would like to enable MA
organizations to offer health-driven
rewards and incentives programs that
may be applied to more health-related
services and activities than are allowed
under our current guidance. We
proposed to amend our regulations to
establish parameters for rewards and
incentives programs offered to enrollees
of MA plans. Because we are concerned
about the possibility that such programs
would be targeted only to healthier
enrollees, and discourage sicker
enrollees from participating in such
incentives and in remaining enrolled in
the plan, we also proposed to include
specific requirements regarding rewards
and incentives so as to ensure that such
programs do not discriminate against
beneficiaries on the basis of health
status or disability, or other
impermissible bases for discrimination.
Section 1856(b)(1) of the Act provides
authority for the establishment of MA
standards by regulation that are
consistent with and carry out Part C,
and section 1857(e)(1) of the Act
provides authority to impose contract
requirements that CMS finds ‘‘necessary
and appropriate’’ and that are not
inconsistent with Part C. Section
1852(b)(1)(a) of the Act states that MA
organizations may not discriminate
against beneficiaries on the basis of
health status and that CMS may not
approve an MA plan if that offering is
susceptible to discrimination based on
an individual’s health status.
Furthermore, section 1857(g)(1)(D) of
the Act provides authority for taking
intermediate sanction action against an
MA organization which ‘‘engages in any
practice that would reasonably be
expected to have the effect of denying
or discouraging enrollment by eligible
individuals’’ as a result of their health
status or history. We proposed to rely
upon the aforementioned rulemaking
and substantive authority to establish
requirements for rewards and incentives
programs offered by MA organizations
to Medicare beneficiaries enrolled in
their MA plans.
Specifically, we proposed adding a
new provision at § 422.134 that would
authorize MA organizations to offer
reward and incentive programs to their
current Medicare enrollees to encourage
their participation in activities that
focus on promoting improved health,
preventing injuries and illness, and
promoting efficient use of health care
resources. We proposed requiring that
reward-eligible activities be designed so
that all enrollees are able to earn
rewards without discrimination based
on race, gender, chronic disease,
institutionalization, frailty, health
status, and other impairments. This
proposed requirement would not
preclude MA organizations from
offering rewards and incentives
programs that target a specific disease,
chronic condition or preventive service.
Rather, the goal of having a non-
discrimination requirement is to prevent
particularly vulnerable populations
from being disproportionately
underserved. MA organizations may not
use this provision to ‘‘cherry pick’’
healthier enrollees. Therefore, any
rewards and incentives program
implemented by an MA organization
under this proposal must accommodate
otherwise qualified beneficiaries who
receive services in an institutional
setting or who need a modified
approach to enable effective
participation.
To meet the proposed CMS
requirements, a reward or incentive
would have to be earned by completing
the entire health-related service or
activity and may not be offered for
completion of less than all required
components of the eligible service or
activity. An MA organization would
define what qualifies as an ‘‘entire
service or activity’’ within its program
design. This proposed requirement is
tied to interpreting the value of the
service provided as it relates to the
value of the reward. Under this
proposal, rewards and incentives would
be subject to a monetary cap in an
amount CMS determines could
reasonably be expected to affect enrollee
behavior while not exceeding the value
of the health-related service or activity
itself. As part of our proposal, we
indicated the intent to provide guidance
on this qualitative standard on a regular
basis.
In addition, our proposed regulation
would require MA organizations that
offer rewards and incentives programs
to provide information about the
effectiveness of such programs to CMS
upon request. If CMS determines that
the rewards and incentives programs are
not in compliance with our regulatory
standard, we proposed that we may
require that the MA organization modify
the basic parameters of the program.
We received the following comments
and our responses are as follows:
Comment: We received several
comments in support of this proposal,
approving of our effort to allow MA
organizations to make rewards and
incentives programs more widely
available to enrollees. Several
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00074 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29917
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
commenters noted that facilitating
beneficiary engagement in health
behaviors and practices will help to
achieve better health outcomes.
Response: We thank the commenters
for their support.
Comment: Several organizations
expressed concern over the requirement
that rewards and incentives programs be
non-discriminatory and available to all
enrollees. They requested clarification
that such programs may target specific
chronic conditions, diseases and other
health care needs.
Response: In response to comments,
we have strengthened the regulation to
ensure that rewards and incentives
programs will not be discriminatory. As
revised, the non-discrimination
requirement of the provision is based on
the substantive requirement of section
1852(b)(1)(A) of the Act (which states
that MA organizations may not
discriminate against beneficiaries on the
basis of health status) and expands upon
it by identifying other impermissible
bases for discrimination, including race,
national origin, and gender. The
regulation is meant to prevent rewards
and incentives programs from being
used to unfairly benefit healthier
enrollees while excluding or
disadvantaging enrollees who are less
healthy or have a disability. MA
organizations may establish rewards and
incentives for specific chronic
conditions, diseases, or other health
care needs so long as the rewards and
incentives program is not
discriminatory.
Comment: We received several
comments stating that the requirement
that a beneficiary must complete a
whole service or activity is too narrow
to permit effective program designs and
requesting that CMS provide greater
flexibility in this area.
Response: We proposed to require
that rewards and incentives be offered
in connection with an entire service or
activity so that CMS and MA
organizations can interpret the value of
a reward or incentive in relation to the
service or activity for which it is being
given. MA organizations may reasonably
define the scope of the ‘‘entire service
or activity’’ in their program design. For
example, a MA organization may decide
to offer rewards and incentives for
participation in a smoking cessation
program. The MA organization may
decide to give smaller rewards for each
class attended or give one larger reward
for completing a set number of classes,
as long as the value of the reward
reflects the value of the service and
adheres to the monetary cap designated
by CMS. We are revising
§ 422.134(c)(1)(i) to eliminate the phrase
‘‘completion of’’ to make it possible for
portions of a service or activity to be
defined as the ‘‘entire service or
activity.’’ We emphasize that the value
limitation applies to each ‘‘entire
service or activity’’ such that the value
of the reward or incentive offered may
not be greater than the value of the
service or activity itself.
Comment: Several commenters
cautioned against rewards and incentive
programs because they have the
potential to disproportionately penalize
low-income, minority beneficiaries, and
beneficiaries with disabilities.
Response: We understand the
commenters’ concerns and consequently
emphasize here (and elsewhere in this
preamble) that all rewards and
incentives programs must be non-
discriminatory and may not
disproportionately penalize any groups,
specifically the vulnerable.
Additionally, as discussed in a previous
response, we have revised the regulation
text to strengthen the non-
discrimination language.
Comment: Several commenters
suggested that CMS solicit data from
rewards and incentives programs on a
regular basis rather than ‘‘on request.’’
Commenters are particularly interested
in outcomes data. In addition, one
commenter asked about CMS’
requirements for the format of that
information.
Response: We have noted these
comments and will consider adopting a
rewards and incentives program
reporting cycle in the future.
Comment: Several commenters do not
support rewards and incentive program
designs that include increased
beneficiary cost-sharing as a penalty for
not participating in such a program.
Response: The provision as finalized
only allows programs that will provide
rewards and incentives to beneficiaries.
It does not allow MA organizations to
penalize beneficiaries for non-
participation by any means, including
through increased cost-sharing. We also
note that § 422.134(c)(2)(i) prohibits
rewards and incentives from being
offered in the form of cash or monetary
rebates; we would consider a discount
on cost-sharing to be such a prohibited
reward and incentive. Furthermore,
CMS regulations requiring uniformity of
benefits (42 CFR 422.100(d)(2)) preclude
MA plans from charging enrollees of a
plan different premiums or cost-sharing
for the same service.
Comment: A few commenters asked
how this new provision will impact the
rewards and incentives guidance that is
currently located in the Medicare
Marketing Guidelines.
Response: This provision will
supersede any previously issued
rewards and incentives program
guidance. Upon finalization of this rule,
we will update our subregulatory
guidance accordingly.
Comment: A few commenters asked
CMS to provide more specific
information regarding rewards and
incentives programs. They asked for
guidance on calculating the value of the
activities for which the plan would like
to offer rewards and incentives, whether
the rewards and incentives may be used
to decrease cost-sharing or premiums
and whether there is a limit on how
often an MA organization may offer a
reward or incentive.
Response: The provision provides an
MA organization with great flexibility in
designing its own rewards and
incentives program. At this time, we
will rely on the MA Organizations to
reasonably value the activities/services
for which they offer rewards and
incentives. In this final rule, we neither
identify limits for how often rewards
and incentives may be offered nor do we
set a maximum monetary value for the
rewards and incentives. However, if we
determine such guidance is needed to
apply the standard in § 422.134(c)(1)(iii)
that the reward or incentive be expected
to impact enrollee behavior without
exceeding the value of the health-related
service or activity itself, we will provide
it through subregulatory guidance.
Rewards and incentives may never be
used to decrease cost-sharing or plan
premiums. In addition to the
prohibition at § 422.134(c)(2)(i), CMS
regulations requiring uniformity of
benefits (42 CFR 422.100(d)(2)) preclude
MA plans from charging enrollees of a
plan different premiums or cost-sharing
for the same service. Thus, a MA plan
may not offer lower cost-sharing or
premiums for plan benefits, as a reward
or incentive.
Comment: Two commenters asked
that we expand this provision to include
Part D plans.
Response: We have noted the
comment. At this time, the rewards and
incentives program provision only
applies to Part C.
Comment: One commenter requested
that SNPs be allowed greater flexibility
in rewards and incentives program
design.
Response: The current provision and
the parameters set forth are applied to
all types of MA plans, including SNPs.
At this time, we do not intend to
provide SNP-specific rewards and
incentives program rules or guidance.
Comment: Several commenters asked
how rewards and incentives will be
accounted for in plan bids and one
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00075 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29918
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
commenter suggested that the costs
should be identified as an
administrative cost for care management
services in the bid.
Response: A rewards and incentives
program would be included in the bid
as a non-benefit expense and would not
be entered in the PBP. Per CMS OACT
Bidding Guidance, (available at:
https://www.cms.gov/Medicare/Health-
Plans/MedicareAdvtgSpecRateStats/
Bid-Forms-and-Instructions.html),
‘‘non-benefit expenses are all of the bid-
level administrative and other non-
medical costs incurred in the operation
of the MA plan.’’ We also wish to clarify
that the costs of a rewards and
incentives program would not
necessarily be related only to care
management services and that plans
must comply with applicable bidding
requirements.
Comment: Several commenters
requested that CMS clarify whether
rewards and incentives programs would
be offered as a benefit or otherwise.
Response: Our policy has been, and
continues to be, that rewards and
incentives programs are not benefits.
Comment: One commenter requested
that CMS, in considering additional
parameters for reward and incentive
programs, consider shared decision-
making and tiered networks. The same
commenter also stated that our proposal
removes a great deal of flexibility for
plans to develop these programs and
constrains employer group plans from
providing these programs to the entire
employer group.
Response: We are not clear what is
meant by the request that in our
consideration of additional parameters
we consider shared decision-making
and tiered networks. We note that
shared decision-making and tiering of
medical benefits are strategies that MA
organizations may use to influence
enrolled beneficiaries’ health care
decisions. Rewards and incentives are
another tool CMS is making available to
MA organizations to encourage
enrollees to engage in activities/services
that are intended to improve health and/
or decrease enrollee risk for illness. MA
organizations have the flexibility to use
these tools together or as separate
programs designed to improve enrollees’
health.
We are not aware of what flexibilities
plans may be using currently in
providing rewards and incentive
programs to enrollees that the
commenter believes CMS proposed to
remove. We specifically solicited
information on this topic from MA
organizations in both the proposed rule
and in the CY 2014 Call Letter and have
received no information that would lead
us to believe that our proposed rewards
and incentives program would limit,
rather than expand, current plan
flexibilities. The current guidance on
rewards and incentive programs that
may be offered to plan enrollees,
included in the Medicare Marketing
Guidelines, allows a very limited use of
rewards and incentives to promote
enrollee use of Medicare-covered
preventive services. Therefore, we do
not see how our proposed rewards and
incentives program framework could
remove plans’ flexibilities rather than
expand them.
After consideration of the public
comments received, we are finalizing
the proposed Rewards and Incentives
Program Regulations for Part C Enrollees
rule with modifications to subparagraph
(b)(1) to include ‘‘national origin,
including limited English proficiency,’’
and ‘‘disability.’’ In subparagraph (b)(1)
we are also changing the text from
‘‘institutionalization’’ to ‘‘whether a
person resides or receives services in an
institutional setting’’ and from ‘‘other
impairments’’ to ‘‘other prohibited
basis.’’ These changes clarify the scope
of the categories of beneficiaries
included in the context of prohibited
discrimination and address comments
expressing concern about the possible
disproportionate impact of rewards and
incentives programs.
Additionally, we are modifying
paragraph (c)(1)(i) to eliminate the
phrase ‘‘completion of’’ from the
regulation text to make it possible for
smaller increments of service or activity
to be defined as the ‘‘entire service or
activity.’’ However, we emphasize that
the value of any reward must reflect the
value of the service and adhere to any
monetary cap that has been determined
by CMS under § 422.134(c)(1)(iii).
Finally, we note that we have made a
technical change to delete the phrase
‘‘all of the following’’ from the
introductory language at paragraph (c).
B. Improving Payment Accuracy
1. Implementing Overpayment
Provisions of Section 1128J(d) of the
Social Security Act (§ 422.326 and
423.360)
This section of the final rule
implements Section 6402 of the
Affordable Care Act, which established
new section 1128J(d) of the Social
Security Act (‘‘the Act’’) entitled
Reporting and Returning of
Overpayments. Section 1128J(d)(4)(B) of
the Act defines the term overpayment as
any funds that a person receives or
retains under title XVIII or XIX to which
the person, after applicable
reconciliation, is not entitled under
such title. The definition of person at
section 1128J(d)(4)(C) includes a
Medicare Advantage organization (as
defined in section 1859(a)(1) of the Act)
and a Part D sponsor (as defined in
section 1860D–41(a)(13) of the Act). The
definition does not include a
beneficiary.
Section 1128J(d)(1) of the Act requires
a person who has received an
overpayment to report and return the
overpayment to the Secretary, the state,
an intermediary, a carrier, or a
contractor, as appropriate, at the correct
address, and to notify the Secretary,
state, intermediary, carrier or contractor
to whom the overpayment was returned
in writing of the reason for the
overpayment. Section 1128J(d)(2) of the
Act requires that an overpayment be
reported and returned by the later of (1)
the date which is 60 days after the date
on which the overpayment was
identified; or (2) the date any
corresponding cost report is due, if
applicable. Section 1128J(d)(3) of the
Act specifies that any overpayment
retained by a person after the deadline
for reporting and returning an
overpayment is an obligation (as defined
in 31 U.S.C. 3729(b)(3)) for purposes of
31 U.S.C. 3729.
Finally, section 1128J(d)(4)(A) of the
Act defines ‘‘knowing’’ and
‘‘knowingly’’ as those terms are defined
in 31 U.S.C. 3729(b). Specifically, the
terms ‘‘knowing’’ and ‘‘knowingly’’
‘‘mean that a person with respect to
information: (1) Has actual knowledge
of the information; (2) acts in deliberate
ignorance of the truth or falsity of the
information; or (3) acts in reckless
disregard of the truth or falsity of the
information.’’ There need not be ‘‘proof
of specific intent to defraud.’’
To implement section 1128J(d) of the
Act for the Part C Medicare Advantage
program and the Part D Prescription
Drug program, we proposed two new
sections, §§ 422.326 and 423.360,
respectively, both titled, ‘‘Reporting and
Returning of Overpayments.’’ These
sections proposed rules for MA
organizations and Part D sponsors to
report and return an identified
overpayment to the Medicare program.
We use the term Part D sponsor, as
defined at § 423.4, to refer to the entities
that offer prescription drug plans (PDPs)
under part 423 and thus are subject to
section 1128J(d) of the Act.
We also proposed conforming
amendments to §§ 422.1, 422.300, and
423.1 that add a reference to section
1128J(d) of the Act to the existing list of
statutory authorities for the regulations
governing the MA organizations and
Part D sponsors. We also proposed to
amend §§ 422.504(l) and 423.505(k) to
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00076 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29919
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
incorporate a reference to the proposed
§§ 422.326 and 423.360, respectively, in
order to extend the existing data
certification requirement to data that
MA organizations and Part D sponsors
submit to CMS as part of fulfilling their
obligation to return an overpayment
under section 1128J(d) of the Act.
Section 422.504(l) refers to certification
of data ‘‘as a condition for receiving a
monthly payment’’ and § 423.505(k)
refers to certification of data for
enrollees ‘‘for whom the organization is
requesting payment.’’ Thus, we
proposed to add a requirement that
applies after CMS has completed
prospective monthly payments for a
year, and organizations are no longer
‘‘requesting payment’’ because
applicable reconciliation has occurred.
Applicable reconciliation, we stated, is
the point when organizations submit
their final data for the previous payment
year. Accordingly, if an MA
organization or Part D sponsor has
identified an overpayment, there clearly
is a different state of ‘‘best knowledge,
information, and belief’’ than the state
of knowledge, information, and belief
that existed prior to applicable
reconciliation. Thus, we proposed to
require that the CEO, CFO, or COO must
certify (based on best knowledge,
information, and belief) that information
the MA organization or Part D sponsor
submits to CMS for purposes of
reporting and returning of overpayments
under §§ 422.326 and 423.360 is
accurate, complete, and truthful.
We reminded all stakeholders that
even in the absence of a final regulation
on these statutory provisions, MA
organizations and Part D sponsors are
subject to the statutory requirements
found in section 1128J(d) of the Act and
could face potential False Claims Act
liability, Civil Monetary Penalties (CMP)
Law liability, and exclusion from
Federal health care programs for failure
to report and return an overpayment.
Additionally, MA organizations and
Part D sponsors continue to be obliged
to comply with our current procedures
for handling inaccurate payments.
In response to the January 10, 2014
proposed rule, we received
approximately 30 pieces of
correspondence from organizations and
individuals. In this section of the final
rule, we describe our proposals, respond
to the public comments, and state our
final policies.
We did not receive any comments on
our proposed amendments to
§§ 422.504(l) and 423.505(k) to
incorporate a reference to the proposed
§§ 422.326 and 423.360, respectively, in
order to extend the existing data
certification requirement to data that
MA organizations and Part D sponsors
submit to CMS as part of fulfilling their
obligation to return an overpayment
under section 1128J(d) of the Act. We
did not receive any comments on our
conforming amendments to §§ 422.1,
423.300, and 423.1. Therefore, we are
finalizing these amendments as
proposed.
a. Terminology (§§ 422.326(a) and
423.360(a))
We proposed definitions of 3 terms.
First, we proposed to adopt the statutory
definition of overpayment, where an
overpayment exists when—after
‘‘applicable reconciliation’’—an MA
organization or Part D sponsor is not
entitled to funds it has received and/or
retained. In order to clarify the statutory
definition of overpayment, we proposed
definitions of 2 key terms at
§§ 422.326(a) and 423.360(a): ‘‘Funds’’
and ‘‘applicable reconciliation.’’
We proposed to define ‘‘funds’’ as
payments an MA organization or Part D
sponsor has received that are based on
data that these organizations submitted
to CMS for payment purposes. We also
noted that MA organizations and Part D
sponsors have responsibility for the
accuracy, completeness, and
truthfulness of data they submit under
existing §§ 422.504(l) and 423.505(k).
For Part C, the data submitted by the
MA organization to CMS includes
§§ 422.308(f) (enrollment data) and
422.310 (risk adjustment data). For Part
D, data submitted by the Part D sponsor
to CMS includes data submitted under
§§ 423.329(b)(3), 423.336(c)(1), 423.343,
and data provided for purposes of
supporting allowable costs as defined in
§ 423.308 of this part which includes
data submitted to CMS regarding direct
or indirect remuneration (DIR).
There are additional payment-related
data CMS uses to calculate Part C and
Part D payments that are submitted
directly to CMS by other entities, such
as the Social Security Administration
(SSA), which is the authoritative source
for data they submit to CMS. We believe
that MAOs and Part D sponsors cannot
be held accountable for the accuracy of
data controlled and submitted to CMS
by other entities.
For example, the SSA is the
authoritative source for date of death.
An MA organization or Part D sponsor
generally does not submit a date of
death directly to CMS’ systems; it comes
from the SSA data feed. When the SSA
submits to CMS corrected data regarding
a beneficiary’s date of death, CMS’
systems recalculate the payments made
to the plan for that beneficiary and
recoup the incorrect payment in a
routine retroactive payment adjustment
process.
We stated that when CMS recoups an
incorrect payment from an MA
organization or Part D sponsor based on
data corrections submitted by
authoritative sources such as the SSA,
CMS would not consider this
recoupment to be the return of an
overpayment by an MA organization or
Part D sponsor under proposed
§§ 422.326 and 423.360. Therefore, the
proposed meaning of ‘‘funds’’ refers to
a payment amount that an MA
organization or Part D sponsor received
from CMS that is based on data that the
MA organization or Part D sponsor
controls and submits to CMS.
We stated that the term ‘‘applicable
reconciliation’’ refers to an event or
events after which an overpayment can
exist under section 1128J(d) of the Act,
and we proposed definitions of the term
applicable reconciliation that are
specific to Part C and Part D.
For Part C, we proposed that
applicable reconciliation occurs on the
date that CMS announces as the final
deadline for risk adjustment data
submission. For each payment year, we
apply three sets of risk scores to adjust
payments: Initial and midyear risk
scores during the payment year (both
sets are based on incomplete diagnosis
data from the data collection year); and
final risk scores after the payment year
using data MA organizations submit on
or before the final deadline for risk
adjustment data (which reflects
complete data for the data collection
year). We also stated that the final risk
adjustment data submission deadline
would function as the Part C applicable
reconciliation date.
For Part D sponsors, we proposed that
applicable reconciliation is the later of
either: The annual deadline for
submitting prescription drug event
(PDE) data for the annual Part D
payment reconciliations referred to in
§ 423.343 (c) and (d) or the annual
deadline for submitting DIR data. The
annual deadline for submitting PDE data
is the last federal business day prior to
June 30th of the year following the
benefit year being reconciled. The
annual deadline for submitting DIR data
is announced annually through
subregulatory guidance and generally
occurs around the last business day in
June the year following the benefit year
being reconciled. We selected these
events to define the Part D applicable
reconciliation because these data are
used for the purposes of determining
final Part D payment reconciliation. We
noted that MA organizations would still
have to submit all final risk adjustment
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00077 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29920
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
diagnoses for Part D by the final risk
adjustment data submission deadline.
In summary, we proposed an
approach to defining applicable
reconciliation that establishes dates that
differ for Part C and Part D. We asked
for comment on this approach.
We noted that payment errors
identified as a result of any corrections
to risk adjustment data submitted by
MA organizations (and other
organizations required to submit risk
adjustment data to CMS) on or before
the annual final risk adjustment data
submission deadline are handled as part
of the current annual process of risk
adjustment payment reconciliation.
Because these payment errors are prior
to the date defined in this final rule as
‘‘applicable reconciliation’’, we stated
that we do not consider these errors to
be overpayments for the purpose of
§§ 422.326 and 423.360. That is, any
deletions of risk adjustment data in the
file submitted on or before the final risk
adjustment data submission deadline for
a payment year, would result in
payment errors that are addressed with
processes that have been in place prior
to our codification of section 1128J(d) of
the Act in proposed §§ 422.326 and
423.360.
Likewise, for Part D, any payment
errors identified as a result of any
corrections to PDE or DIR data
submitted on or before the later of the
annual deadline for submitted PDE and
DIR data are handled as part of the
current Part D reconciliation process,
and we do not consider these errors to
be overpayments for the purpose of
§ 423.360.
Finally, we stated our expectation that
MA organizations and Part D sponsors
must be continuously diligent regarding
the accuracy and completeness of
payment-related data they submit to
CMS for a payment year, whether
during or after that payment year, and
whether before or after applicable
reconciliation dates. This expectation is
based on existing requirements at
§§ 422.310, 422.504(l), 423.329(b)(3)(ii),
and 423.505(k), and proposed
amendments that clarify and strengthen
these requirements.
We did not receive any comments on
the proposed definitions of the terms
‘‘funds’’ or ‘‘overpayment.’’ (See the
next section for comments and
responses on the provision regarding
‘‘identified overpayment’’.) We received
the following comment on the term
‘‘applicable reconciliation’’, and our
response follows.
Comment: Some commenters
supported CMS’ proposal to have
separate applicable reconciliation dates
for the Part C and Part D programs,
noting that this approach is simpler and
more practical than the alternative CMS
described (where there would be 2
applicable reconciliation dates for the
Part D program—one for risk adjustment
and another for PDE and DIR data).
Response: We appreciate the support.
We will finalize our proposal that the
Part C applicable reconciliation date
will be the same as the final risk
adjustment data submission deadline,
and the Part D applicable reconciliation
date will be the later of: The annual
deadline for submitting prescription
drug event (PDE) data for the annual
Part D payment reconciliation referred
to in § 423.343(c) and (d) or the annual
deadline for submitting DIR data.
We would like to note that the final
risk adjustment data submission
deadline will still apply to diagnosis
data for both Part C and Part D risk
scores for beneficiaries in MA–PD plans.
After consideration of the public
comments received, we are finalizing
the provisions at §§ 422.326(a) and
423.360(a) as proposed.
b. General Rules for Overpayments
(§ 422.326(b) Through (c); § 423.360(b)
through (c))
We proposed at §§ 422.326(b) and
423.360(b) that if an MA organization or
Part D sponsor has identified that it has
received an overpayment, the MA
organization or Part D sponsor must
report and return that overpayment in
the form and manner set forth in the
section. In paragraphs §§ 422.326(c) and
423.360(c), we proposed that the MA
organization or Part D sponsor has
identified an overpayment if it has
actual knowledge of the existence of the
overpayment or acts in reckless
disregard or deliberate ignorance of the
existence of the overpayment. We noted
that the terms ‘‘reckless disregard’’ and
‘‘deliberate ignorance’’ are part of the
definitions of ‘‘knowing’’ and
‘‘knowingly’’ in section 1128J of the Act,
which provides that the terms
‘‘knowing’’ and ‘‘knowingly’’ have the
meaning given those terms in the False
Claims Act (31 U.S.C. 3729(b)(3)). We
stated that without such a proposal to
include ‘‘reckless disregard’’ and
‘‘deliberate ignorance’’, some MA
organizations and Part D sponsors might
avoid performing activities to determine
whether an overpayment exists. We also
provided that if an MA organization or
Part D sponsor has received information
that an overpayment may exist, the
organization must exercise reasonable
diligence to determine the accuracy of
this information, that is, to determine if
there is an identified overpayment.
Finally, in paragraphs §§ 422.326(d)
and 423.360(d), we proposed the
requirements for reporting and returning
an identified overpayment. An MA
organization or Part D sponsor must
report and return any identified
overpayment it received no later than 60
days after the date on which it
identified it received an overpayment.
The statute provides an alternative
deadline: The date any corresponding
cost report is due, if applicable. We
proposed that this alternative deadline
is not applicable to the Parts C or D
programs because, in general, MA
organizations and Part D sponsors are
paid based on their bids, and not based
on their actual incurred costs.
The MA organization or Part D
sponsor must notify CMS, using a
notification process determined by
CMS, of the amount and reason for the
overpayment. Also within this 60-day
time period, the organization must
return identified overpayments to CMS
in a manner specified by CMS,
including the amount and reason for the
overpayment. We proposed to codify at
paragraph (3) the statutory requirement
that any overpayment retained by an
MA organization or Part D sponsor after
the 60-day deadline for reporting and
returning is an obligation under 31
U.S.C. 3729(b)(3).
We also emphasized that an MA
organization and Part D sponsor are
deemed to have returned the
overpayment when they have taken the
actions that we will specify, in
forthcoming operational guidance, to
submit the corrected data that is the
source of the overpayment. We will
recover the returned overpayment
through routine processing according to
the systems schedule established in the
annual operations budget. That is,
payments are recovered through the
established payment adjustment
process, not on the 60-day schedule that
applies to each MA organization or Part
D sponsor that has identified an
overpayment. Rerunning reconciliation
each time an entity identifies an
overpayment that triggers its 60-day
clock is simply not feasible for CMS.
Finally, we proposed that there will
be circumstances when we may ask the
MA organization or Part D sponsor to
provide an auditable estimate of the
overpayment amount, reason for
overpayment, and make a payment to
CMS. This may occur, for example,
when an overpayment is identified after
the final Part D reopening for a contract
year has occurred but prior to the end
of the look-back period or if an MA
organization or Part D sponsor had a
thoroughly-documented catastrophic
loss of stored data. Information about
the nature of such a request would be
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00078 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29921
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
detailed in forthcoming operational
guidance.
We received the following comments
on general rules for overpayments and
our responses follow.
Comment: Several commenters
requested that CMS clarify when the 60-
day period begins. Specifically, does the
period begin once the MA organization
or Part D sponsor has identified that
there is an overpayment or once the
organization has determined the exact
amount of the overpayment? A
commenter expressed concern that the
proposed rule does not appear to
acknowledge that the amount of an
overpayment must be quantified before
it is ‘‘identified.’’ Another commenter
requested that CMS address the
situation where an MA organization or
Part D sponsor becomes aware of an
issue or error that may have resulted in
one or more overpayments, but could
not determine, with reasonable
certainty, the amount of the
overpayment(s) within a 60-day period.
Response: It is important to
understand the distinctions among
identifying, reporting, and returning an
overpayment in this rulemaking for the
purposes of the MA and Part D
programs. Once an organization has
identified that it has received an
overpayment, the 60-day period for
reporting and returning the
overpayment begins. Because of the
nature of the Part C and Part D
programs, we did not propose that
‘‘identified’’ includes completion of the
act of quantification of an overpayment
amount. Rather, we proposed that
identification of an overpayment means
knowing that the MA organization or
Part D sponsor has submitted erroneous
data to CMS that caused CMS to
overpay the organization.
An organization can identify or assess
that there is a problem with data
submitted to CMS, and determine that it
is incorrect data, prior to actually
calculating what the payment impact is
of that erroneous data. For the MA and
Part D programs, the relevant factor is
identifying that the data is incorrect and
will result in an overpayment. For
example, a risk adjustment diagnosis
that has been submitted for payment but
is found to be invalid because it does
not have supporting medical record
documentation would result in an
overpayment. Under this provision, the
day after the date on which the
organization has confirmed an
identified overpayment—because the
organization knows that the diagnosis is
not supported by documentation—is the
first day of the 60-day period for
reporting and returning the
overpayment. As another example, an
MA organization may find that data
used to calculate Healthcare
Effectiveness Data and Information Set
(HEDIS) measures that the organization
submitted to CMS are found to be
invalid; when the organization has
confirmed that it has identified invalid
data leading to an overpayment, this is
the first day of the 60-day period for
reporting and returning the
overpayment.
Then, during the 2-month period for
reporting and returning the
overpayment, the organization must
determine what data should be
submitted to CMS to correct the
identified overpayment, and then must
engage in the reporting and returning
process that we will describe in
forthcoming guidance. This reporting
and returning process will involve: (1)
Notifying CMS that an overpayment
exists, including notification of the
reason and estimated amount for that
overpayment; and (2) submitting the
corrected data to CMS.
In other words, we believe that the
MA organization and Part D sponsor
will discover through appropriate
payment evaluation procedures when a
60-day period would begin under the
requirements of this provision, because
‘‘day one’’ of the 60-day period is the
day after the date on which organization
has determined that it has identified the
existence of an overpayment. Once the
organization ‘‘starts the clock,’’ it has 60
days to submit to CMS the corrected
data that is the basis of the
overpayment. It is the act of submitting
the corrected data to CMS, along with a
reason and an amount of the
overpayment (which may be an
estimate), that constitutes fulfillment of
the requirement to report and return the
overpayment.
As we stated in the January 10, 2014
proposed rule preamble (79 FR 1997),
‘‘It also is important to note that the MA
organization and Part D sponsor are
deemed to have returned the
overpayment when they have taken the
actions that we will specify, in
forthcoming operational guidance, to
submit the corrected data that is the
source of the overpayment’’. We will
recover the returned overpayment
through routine CMS payment
processes. That is, payments will
continue to be recovered through the
established payment adjustment
processes and schedules. As a result the
payment recovery may not occur within
the 60-day window triggered by
identifying an overpayment. Rerunning
payment reconciliations and conducting
payment recovery within CMS payment
systems each time an entity identifies an
overpayment that triggers its 60-day
clock is simply not feasible for CMS.
We will release operational guidance
on the process an organization will use
for informing CMS that it has identified
a Part C and/or Part D overpayment.
This guidance will also address how an
organization will be required to provide
a reason for and the amount of the
overpayment (which may be estimated).
We seek to reduce burden and
implement an efficient process for
administering the reporting and return
of overpayments, so we are considering
making use of existing procedures for
organizations to communicate payment
data issues to CMS. For example, MA
organizations and Part D sponsors have
used the Remedy system for a number
of years to inform CMS of payment
issues and provide relevant information
on that issue.
In the forthcoming operational
guidance, we will address the question
of how to report the overpayment
amount, including estimation of the
overpayment amount and updates under
certain scenarios.
Comment: A commenter contended
that, applying the principles adopted by
CMS in the RADV audit context, an
overpayment cannot exist for a
particular MA contract unless CMS’
payments as a whole to the MA
organization pursuant to the contract are
inaccurate in light of an appropriate FFS
Adjuster applied to the entire contract.
Potential overpayments can be
determined, therefore, based only on
processes such as CMS’ RADV audits,
which are designed to measure whether
contract-level payments to an MA
organization are accurate when
compared to an appropriate FFS
Adjuster. The commenter further
contended that to the extent an MA
organization develops processes
intended to measure payment accuracy
at the contract-level, the MA
organization would be required to report
and repay inaccuracies calculated after
applying CMS’s FFS Adjuster, and
consistent with prior CMS guidance,
this is the sole instance in which an
‘‘overpayment’’ can be determined for
purposes of proposed § 422.326.
Response: We disagree with the
commenter. Our RADV methodology
does not change our existing contractual
requirement that MA organizations must
certify (based on best knowledge,
information, and belief) the accuracy,
completeness, and truthfulness of the
risk adjustment data they submit to
CMS. Further, this decision does not
change the long-standing risk
adjustment data requirement that a
diagnosis submitted to CMS by an MA
organization for payment purposes must
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00079 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29922
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
be supported by medical record
documentation.
However, we are clarifying the link
between the § 422.326 overpayment
provisions and RADV audits under
§ 422.311 by adding a condition to the
requirement at § 422.326(d), as follows:
an MA organization must report and
return any overpayment it received no
later than 60 days after the date on
which it identified it received an
overpayment. We are adding to
paragraph (d) the provision ‘‘unless
otherwise directed by CMS for the
purpose of § 422.311.’’ Thus, when an
MA organization has a contract selected
for a RADV audit, during the audit the
MA organization will not be allowed to
report and return an overpayment under
§ 422.326 that is due to errors in the
data used to risk-adjust payments for the
audited contract for the payment year
that is the subject of the RADV audit.
We will notify the MA organization
about the timeline for reporting and
returning any overpayments for a
contract under a RADV audit. This new
provision protects the integrity of the
RADV audit process, including the
sampling frame of beneficiaries in a
selected MA plan, whose diagnoses will
be audited.
Comment: A commenter stated that
there will be many circumstances and
situations where entities receiving an
overpayment will not have the ability to
repay funds within the 60-day period
without undue hardship.
Response: MA organizations and Part
D sponsors have an obligation to pay an
overpayment owed under Section
1128J(d). As noted previously, our
recovery of overpayments will occur
through routine payment processing
cycles and schedules. In most
circumstances, MA organizations and
Part D sponsors will be submitting
corrected data, which will be re-run by
CMS and then CMS will recover the
overpayment.
Comment: A commenter noted that 60
days is not a sufficient timeframe, as
identifying and quantifying
overpayments can be a very involved
process. Another commenter stated that
most overpayments are identified
through analyses and studies, such as
internal RADV studies; the commenter
requested that the 60-day time period
begin at the conclusion of the internal
study, so that overpayments can be
referred to CMS after all issues have
been identified and confirmed.
Response: We provide that the 60-day
period is the time period for reporting
and returning an identified
overpayment, after the organization has
conducted the activities needed to
identify that it has received an
overpayment. As explained previously,
for the purposes of the MA and Part D
programs, the MA organization or Part
D sponsor must report and return the
identified overpayment, which is due to
incorrect data it has submitted to CMS,
no later than 60 days after the date on
which the organization identified it
received the overpayment. Subsequently
and within the 60-day period the MA
organization or Part D sponsor is
required to report and return the
overpayment. Reporting the
overpayment involves notifying CMS of
the reason for and the amount of the
overpayment. Returning the
overpayment is deemed to have
occurred through the act of correcting
the erroneous data submitted to CMS,
for example, by deleting incorrect PDEs
or risk adjustment data. Note that if an
organization identifies one set of
erroneous data that has caused an
overpayment, the organization must
begin the 60-day clock on that date, and
if subsequent overpayments are
identified, the organization must begin
subsequent 60-day reporting and
returning periods.
Comment: A commenter questioned
whether CMS will be identifying criteria
for organizations to use to determine an
overpayment.
Response: We have specified in this
final rule the specific types of ‘‘funds’’
that are subject to the provisions under
this section through the definition of
‘‘funds’’. Funds are payments an
organization has received that are based
on data that the organization submitted
to CMS for payment purposes. We will
not provide additional criteria or a
checklist.
Comment: A commenter stated that
logically, an MA organization or Part D
sponsor cannot return an overpayment
until it has calculated the exact amount
that it must return. It might take a
considerable amount of time for the MA
organization or Part D sponsor to audit
its records to determine the amount,
whether there is an issue in previous
years, and whether extrapolation, or
case by case analysis, is appropriate.
The commenter was concerned that
while a plan sponsor might be able to
report to CMS that it has identified an
issue within 60 days, a plan sponsor
may not have enough information after
identification to be able to report the
exact amount. Therefore, the commenter
requested that CMS clarify that the 60
days begins once the organization has
identified the exact amount of the
overpayment. The commenter
suggested, as an alternative, that if the
MA organization or Part D sponsor has
notified CMS that it believes there is an
overpayment, but it will take more than
60 days to determine the exact amount,
CMS consider allowing a ‘‘tolling’’ of
the 60 days so that the organization may
determine the amount it must return to
CMS. Under this ‘‘tolling’’ process, the
organization would be required to notify
CMS within 60 days of identifying that
an overpayment likely exists, but would
be provided additional time by CMS to
determine the exact amount.
Response: We have not used the
phrase ‘‘exact amount’’ in this rule-
making. For the MA and Part D
programs, we define overpayment in the
regulation as ‘‘funds’’ the organization
has received to which it is not entitled,
and then defines ‘‘funds’’ as any
payment based on data submitted by an
MA or Part D organization. Because of
the nature of the Part C and Part D
programs, the key focus in
implementing these statutory provisions
for the MA and Part D programs is thus
correcting the incorrect data that the
organization submitted to CMS that
resulted in an overpayment. We will
then run reconciliation on its routine
operational schedule to recover
overpayment amounts based on the
corrected data. The purpose of the 60
days is to provide the MA or Part D
organization with sufficient time to
correct the incorrect data submitted to
CMS using established data correction
processes. MA organizations and Part D
sponsors are deemed to have returned
the overpayment when they have taken
the actions to submit the corrected data
that is the source of the identified
overpayment. Within the 60 days the
MA organization and Part D sponsor
must also report the overpayment
amount (or estimated amount). If an
estimated overpayment amount is
reported, it may be higher or lower than
the actual overpayment amount
recovered because additional payment
data submitted into the CMS payment
system from other sources may be
incorporated into the payment
calculations.
Comment: A commenter stated that it
is unclear what may occur post-
reconciliation if both parties have been
overpaid. For example, if CMS owes the
Part D sponsor $10 million due to
activity post-reconciliation and a $2
million overpayment is discovered, the
commenter questioned whether we will
still require that the $2 million be
refunded within 60 days or whether the
sponsor will be allowed to offset
amounts owed by CMS. The commenter
recommended that if an overpayment
would be reduced or fully covered by a
reopening, that CMS allow sponsors to
request a reopening and offset the
reopening amount due from the
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00080 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29923
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
overpayment pending completion of the
reopening.
Response: For both the Part C and Part
D programs, the provisions regarding
reporting and returning identified
overpayments become effective the day
after the date of applicable
reconciliation. As we have stated, MA
organizations and Part D sponsors are
deemed to have returned the
overpayment when they have submitted
corrected data that is the source of the
overpayment. We will recover the
overpayment amount through routine
processing. For Part D, that means that
if an overpayment is discovered after
the initial reconciliation but prior to the
reopening described at § 423.346, a Part
D sponsor may request a reopening and
submit the corrected data to fulfill its
obligation to return the overpayment.
The overpayment will be reconciled
through the routine reopening process.
Comment: A commenter stated that
the onus on plans for the calculation of
an overpayment amount creates a risk
that CMS may be overpaid/underpaid in
the monies returned.
Response: As explained in proposed
rule (79 FR 1997), we will recover
overpayments through the correction of
erroneous data and established payment
adjustment processes. Therefore, we
believe that the risk the commenter
mentions does not exist because CMS’
systems will calculate the exact amount
to be recovered.
Comment: A few commenters
objected to the fact that the proposed
rule does not address situations in
which a sponsor has overpaid CMS, and
requested that this regulation also set
forth rules by which CMS handles an
organization’s overpayments to CMS.
Response: This final rule is intended
to implement section 1128J(d) of the
Act, which pertains only to
overpayments the government made to
contracting MA organizations and Part
D sponsors.
Comment: A commenter requested
that MA organizations and Part D
sponsors be able to submit auditable
estimates of an overpayment in lieu of
determining which data is in error and
submitting corrected data, given the fact
that the administrative costs of
determining a specific set of data deletes
is significant relative to the size of the
issue. The commenter recommended
that CMS permit plans to proactively
suggest the use of such tools to resolve
potential overpayments.
Response: The use of auditable
estimates is intended only for a limited
set of circumstances. This may occur,
for example, when the Part D reopening
occurs prior to the end of the look-back
period or if an MA organization or Part
D sponsor had a thoroughly-
documented catastrophic loss of stored
data. Information about the nature of
such a request would be detailed in
forthcoming operational guidance.
Therefore, we will not allow, on a
routine basis, submission of auditable
estimates in lieu of submission of
corrected data. By recovering
overpayments based on the corrected
payment data, we will be more likely to
ensure that the most accurate
overpayment amounts are returned to
the Medicare Trust Fund.
Comment: A commenter expressed
concern that this final rule could
impose a boundless duty to troll
medical records in search of unknown
vulnerabilities, and requested that CMS
make clear that Part C and Part D plans
are not obliged to proactively search for
an overpayment without reason to
believe that a specific overpayment
exists.
Response: The focus of this final rule
is on ensuring that MA and Part D
organizations return an overpayment
when it is identified. For many years
organizations have been obliged to
submit accurate, complete, and truthful
payment-related data, as described at
§§ 422.504(l) and 423.505(k). Further,
CMS has required for many years that
diagnoses that MA organizations submit
for payment be supported by medical
record documentation. Thus, we have
always expected that MA organization
or Part D sponsor implement, during the
routine course of business, appropriate
payment evaluation procedures in order
to meet the requirement of certifying the
data they submit to CMS for purposes of
payment. Therefore, we do not believe
that §§ 422.326 and 423.360 represent
such a new requirement.
Comment: A commenter requested
that CMS confirm that the data
submission requirement under this
section is based on enrollment data and
risk adjustment scores, and thus does
not apply to direct overpayments from
providers.
Response: Once an overpayment is
identified, the MA or Part D
organization is responsible for
correcting the data that caused the
overpayment. This is data that is
routinely submitted to CMS for payment
purposes, such as, risk adjustment data.
Comment: A commenter requested
that CMS clarify if changes in a
beneficiary’s low income subsidy (LIS)
status could result in an overpayment
under this provision.
Response: As we stated in the
proposed rule, we believe that MA
organizations and Part D sponsors
cannot be held accountable for the
accuracy of the data controlled and
submitted to CMS by other entities. (We
emphasize here that the term ‘‘other
entities’’ used to discuss these
overpayment provisions does not
include the following parties referenced
in §§ 422.504(i) and 423.505(i): first tier,
downstream, and related entities,
contractors, or subcontractors to the MA
organization or Part D sponsor.) It is the
Social Security Administration and the
states that notify CMS of individuals
whom they have determined to be
eligible for the Part D LIS. We in turn
provide the subsidy information,
including effective date and level of
subsidy, to the Part D plan in which the
beneficiary enrolls. Although, we will
not consider an overpayment to have
occurred strictly due to changes in a
beneficiary’s LIS status, Part D sponsors
are required to adjust prescription drug
event (PDE) data to accurately reflect the
beneficiary’s LIS status.
Comment: A commenter supported
our proposal for when overpayments
have been identified.
Response: We appreciate the
commenter’s support for our proposal.
Comment: A few commenters
requested that CMS provide more clarity
or an example of what is meant by ‘‘acts
in reckless disregard or deliberate
ignorance.’’
Response: We are revising our
definition of an identified overpayment
to state that an MA organization or Part
D sponsor has identified an
overpayment when it has determined, or
should have determined through the
exercise of reasonable diligence, that the
MA organization or Part D sponsor has
received an overpayment.
As to the circumstances that give rise
to a duty to exercise reasonable
diligence, we are not able to anticipate
all factual scenarios in this rulemaking.
MA organizations and Part D sponsors
are responsible for ensuring that
payment data they submit to CMS are
accurate, truthful, and complete (based
on best knowledge, information, and
belief), and are expected to have
effective and appropriate payment
evaluation procedures and effective
compliance programs as a way to avoid
receiving or retaining overpayments.
Thus, at a minimum, reasonable
diligence would include proactive
compliance activities conducted in good
faith by qualified individuals to monitor
for the receipt of overpayments.
However, conducting proactive
compliance activities does not mean
that the person has satisfied the
reasonable diligence standard in all
circumstances. In certain circumstances,
for example, reasonable diligence might
require an investigation conducted in
good faith and in a timely manner by
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00081 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29924
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
qualified individuals in response to
credible information of a potential
overpayment.
We note that in discussing the
standard term ‘‘reasonable diligence’’ in
the preamble, we are interpreting the
obligation to ‘‘report and return the
overpayment’’ which is contained in
section 1128J(d) of the Social Security
Act. We are not seeking to interpret the
terms ‘‘knowing’’ and ‘‘knowingly’’,
which are defined in the Civil False
Claims Act and have been interpreted
by a body of False Claims Act case law.
Comment: Some commenters thought
that we had an overly broad
interpretation of the statute and that
there was no statutory basis for CMS to
interpret the term ‘‘identified’’ in
section 6402 of the Affordable Care Act
to include ‘‘reckless disregard or
deliberate ignorance of the existence of
the overpayment.’’ A commenter stated
that the term ‘‘knowing’’ is not actually
used in the overpayment standard set
forth in section 6402(d) of the
Affordable Care Act, so the mere
existence of an errant reference to the
False Claims Act definition of
‘‘knowing’’ does not give CMS sufficient
basis to apply the expansive False
Claims Act knowledge standard to the
definition of ‘‘identified’’ under section
6402. This commenter noted that in an
earlier version of the Affordable Care
Act, H.R. 3962, used the False Claims
Act knowledge standard in the section
on reporting and returning of
overpayments. The commenter also
stated that the final version of the
Affordable Care Act enacted by the
Congress used the term ‘‘identified,’’
and not the word ‘‘knowledge.’’ This
commenter believed that the Congress’s
explicit rejection of the False Claims Act
knowledge standard, and use of the term
‘‘identified’’ in the final legislative
language weighs against incorporating
the False Claims Act knowledge
standard into the regulatory provision.
Response: We disagree with the
commenters’ arguments. While we
acknowledge that the terms ‘‘knowing’’
and ‘‘knowingly’’ are defined but not
otherwise used in section 1128J(d), we
believe that the Congress intended for
section 1128J(d) to apply broadly. If the
requirement to report and return
overpayments applied only to situations
where the MA organization or Part D
sponsor has actual knowledge of the
existence of an overpayment, then these
entities could easily avoid returning
improperly received payments and the
purpose of the section would be
defeated. Thus, we decline to read a
narrow actual knowledge limitation into
the law as suggested by commenters.
Comment: Several commenters
recommended that CMS remove the
language relating to ‘‘reasonable
diligence’’ from the proposed
regulation. These commenters believed
that an identified overpayment should
be limited to actual knowledge of an
overpayment.
Response: For the reasons discussed
previously, we decline to read a narrow
actual knowledge limitation into the law
as suggested by commenters.
Comment: A few commenters were
concerned that by adding a reasonable
diligence requirement, CMS appears to
be suggesting that a much lower level of
sponsor behavior—a failure to act
reasonably—could trigger potential
False Claims Act liability. One
commenter stated that the phrase
‘‘reasonable diligence’’ is not a
recognized or defined standard and is
overly vague as to the obligations of
plans to follow through on information
received regarding a potential
overpayment. The commenters have
serious concerns about the implication
of such a standard.
Response: We understand the
commenters’ concerns. However, we do
not believe that it is inappropriate to
expect that MA organizations and Part
D sponsors act reasonably. We note that
it is the statute that establishes liability
under the False Claims Act for failure to
report and return identified
overpayments, pursuant to section
1128J(d)(3).
c. Look-Back Period for Reporting and
Returning Overpayments
We proposed at §§ 422.326(e) and
423.360(e) to codify a look-back period
for MA organizations and Part D
sponsors. MA organizations and Part D
sponsors would be required to report
and return any overpayment that they
identify within the 6 most recent
completed payment years. The statute of
limitations related to the False Claims
Act is 6 years from the date of the
violation or 3 years from the date the
relevant government official learns of
the situation, but in no case more than
10 years from the date of the violation.
CMS proposed 6 years as the look-back
period because we believe this best
balances government’s interest in
having overpayments returned with
entities’ interest in finality. Six years
also is consistent with the CMP
provisions, and maintenance of records
requirements under the contracts. We
also proposed that overpayments
resulting from fraud would not be
subject to this limitation of a look-back
period.
We received the following comments
on the look-back period, and our
responses follow.
Comment: We received a few
comments recommending that we
shorten the 6-year look-back period. A
commenter noted that permitting greater
finality in overpayment reporting and
recovery will decrease administrative
costs and free up resources to focus on
benefits. This commenter also stated
that an organization would have to
retain a significant amount of
documentation to fully support and
justify payments, more than what they
would retain under CMS’s 10-year
record retention requirement. Several
commenters recommended that the
look-back period be 3 years to align with
the RAC look-back period. A commenter
noted that the 3-year period would also
be consistent with the federal
government’s treatment of government
contractors that are subject to the
Federal Acquisition Regulation. A
couple of commenters recommended
that we implement a 4-year look-back
period to align with the 4-year period
that Medicare Administrative
Contractors can reopen Medicare fee-
for-service payment determinations.
Response: We disagree with the
commenters’ recommendations to
shorten the look-back period. We note
that section 1128J(d) of the Act has no
time limit to the obligation to report and
return overpayments received by a
provider or supplier. However, as we
stated in the preamble to our proposed
rule and again in this preamble to our
final rule, we proposed 6 years as the
look-back period because we believe
this best balances government’s interest
in having overpayments returned with
entities’ interest in finality. Six years is
consistent with the CMP provisions, and
maintenance of records requirements
under the contracts. It is also consistent
with the False Claims Act in that the
statute of limitations related to the False
Claims Act is 6 years from the date of
the violation or 3 years from the date the
relevant government official learns of
the situation, but in no case more than
10 years from the date of the violation.
We believe that our final rule does not
create additional recordkeeping burden
or cost. Under § 422.504(d) and
§ 423.505(d), MA organizations and Part
D sponsors are required to maintain for
10 years books, records, documents, and
other evidence of accounting procedures
and practices related to costs, financial
statements, cash flow, etc.
Comment: A commenter requested
that CMS clarify the parameters of the
6-year look-back provision.
Response: As we stated in the
preamble to the proposed rule and this
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00082 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29925
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
final rule and again in §§ 422.326(e) and
423.360(e), MA organizations and Part D
sponsors are required to report and
return any overpayment that they
identify within the 6 most recent
completed payment years. That would
mean, for example, after the initial
reconciliation that takes place for Part D
payments (that is, the determination on
the final amount of direct subsidy
described in § 423.329(a)(1), final
reinsurance payments described in
§ 423.329(c), the final amount of the low
income subsidy described in
§ 423.329(d), or final risk corridor
payments as described in § 423.336) for
contract year 2015 (which will take
place at the end of 2016), Part D
sponsors are obligated to report and
return overpayments under § 423.360
for contract years 2010 through 2015.
Comment: A few commenters
recommended that CMS impose the
same limitation on the look-back period
for all overpayments, even those relating
to fraud. A commenter noted that under
the statutory scheme set forth in section
6402 of the Affordable Care Act, the
existence of an overpayment does not
depend on, or otherwise reflect, the
existence of fraud. Commenters also
requested clarification from CMS
whether MA organizations and Part D
sponsors that become aware of an
overpayment prior to the look-back
period have an obligation to investigate
and determine whether that
overpayment resulted from fraud. These
commenters were concerned that MA
organizations and Part D sponsors
would have to investigate potential
overpayments indefinitely, no matter
how far in the past they may have
occurred, because these organizations
would have to determine whether there
was any fraud in connection with the
potential overpayment in order to
determine whether a reporting
obligation exists.
Response: Upon further review, we
agree with the commenters’ suggestion
that CMS impose the same limitation on
the look-back period for all
overpayments. Six years is consistent
with the more commonly applicable
FCA statute of limitations as well as the
statute of limitations under section
1128A of the Act. Therefore, we have
elected to establish a 6-year look-back
period regardless of the nature of the
overpayment, and we have amended the
regulation text at §§ 422.326(e) and
423.360(e) accordingly. We note that the
government may have other avenues for
pursuing the return of overpayments
due to false and fraudulent claims
outside of these provisions.
Finally, we note that an MA
organization’s and Part D sponsor’s
obligation to investigate and identify
false and fraudulent claims is outside
the scope of this final rule.
After consideration of the public
comments received on the overpayment
provisions, we are finalizing as
proposed the following provisions:
§§ 422.1, 422.300, 422.504(l), 423.1, and
423.505(k). We are finalizing the
provisions at § 422.326, with the
following modifications. First, we add at
the end of paragraph (d) the phrase
‘‘unless otherwise directed by CMS for
the purpose of § 422.311.’’ Second, we
strike the following sentence in the
proposed paragraph on the six-year
look-back period: ‘‘Overpayments
resulting from fraud are not subject to
this limitation of the lookback period.’’
To increase clarity we also revise
paragraph (c) regarding identified
overpayments. We also are making a
technical correction by redesignating
proposed paragraph (d)(3) on
enforcement as paragraph (e), and
redesignating proposed paragraph (e) on
the six-year look-back period as
paragraph (f), and revising new
paragraph (e) on enforcement to say
‘‘Any overpayment retained by an MA
organization is an obligation under 31
U.S.C. 3729(b)(3) if not reported and
returned in accordance with paragraph
(d) above.’’
Finally, we are finalizing the
provisions at § 423.360 with the
following modifications. We strike the
following sentence in the proposed
paragraph on the six-year look-back
period: ‘‘Overpayments resulting from
fraud are not subject to this limitation
of the lookback period.’’ To increase
clarity we also revise paragraph (c)
regarding identified overpayments. We
also are making a technical correction
by redesignating proposed paragraph
(d)(3) on enforcement as paragraph (e),
and redesignating proposed paragraph
(e) on the six-year look-back period as
paragraph (f), and revising new
paragraph (e) on enforcement to say
‘‘Any overpayment retained by a Part D
sponsor is an obligation under 31 U.S.C.
3729(b)(3) if not reported and returned
in accordance with paragraph (d).’’
2. Risk Adjustment Data Requirements
(§ 422.310)
We proposed several amendments to
§ 422.310 to strengthen existing
regulations related to the accuracy of
risk adjustment data. We proposed to
renumber existing paragraph
§ 422.310(e) as paragraph (e)(2) and add
new paragraph (e)(1), which would
require that any medical record reviews
conducted by an MA organization must
be designed to determine the accuracy
of diagnoses submitted under
§§ 422.308(c)(1) and 422.310(g)(2).
Under our proposal, medical record
reviews conducted by an MA
organization could not be designed only
to identify diagnoses that would trigger
additional payments by CMS to the MA
organization; medical record review
methodologies would have to be
designed to identify errors in diagnoses
submitted to CMS as risk adjustment
data, regardless of whether the data
errors would result in positive or
negative payment adjustments.
We also proposed to amend
§ 422.310(g) regarding deadlines for
submission of risk adjustment data; our
proposal was to restructure and revise
subparagraph (g)(2) and add
subparagraph (g)(3). Our current
procedures generally permit submission
of risk adjustment data after the final
risk adjustment submission deadline
only to correct overpayments. Thus, we
proposed, at § 422.310(g)(2)(ii) to
explicitly permit late submissions only
to correct overpayments but not to
submit diagnoses for additional
payment so that the regulation text
would be consistent with our
procedures.
Finally, we proposed to make two
additional changes in paragraph (g).
First, we proposed the deletion of the
January 31 deadline in paragraph (2)
and replacing it with the statement that
CMS will announce the deadline by
which final risk adjustment data must
be submitted to CMS or its contractor.
We noted that the risk adjustment data
submission deadline would also
function as the Part C applicable
reconciliation date for purposes of
proposed § 422.326 on overpayment
rules, also discussed in this final rule.
Second, we proposed adding paragraph
(3) to § 422.310(g). Proposed paragraph
(3) cites § 422.326 as the source of rules
for submission of corrected risk
adjustment data after the final risk
adjustment data submission deadline,
that is, after applicable reconciliation as
defined at § 422.326(a).
In response to the January 10, 2014
proposed rule, we received
approximately 25 pieces of
correspondence from organizations and
individuals regarding these proposals.
We received the following public
comments and our responses follow.
Comment: Many commenters
expressed concern about the vagueness
and overly broad statement of CMS’
proposal to amend § 422.310(e) to
require that medical record reviews
conducted by MA organizations be
designed to determine the accuracy of
diagnoses they submitted to CMS. Some
commenters thought this implied a
requirement to verify every diagnosis
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00083 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29926
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
submitted by every provider, while
others thought this implied a restriction
on the ability of plans to identify what
medical records to review. Other
commenters believed the proposed
amendment limited plans’ ability to
review medical records for operational
purposes other than risk-adjusted
payment, such as focusing on only a
portion of a medical record for a subset
of beneficiaries in order to enhance
HEDIS scores, conduct contract
compliance reviews, and validate claims
processing and billing.
Finally, a few commenters argued that
CMS should offset the payment impact
of diagnoses an MA organization
submitted to CMS that were later found
through medical record reviews to not
be supported by medical record
documentation by adjusting the amount
of CMS’ overpayment to the MA
organization for the level of error in
equivalent diagnoses in FFS claims
data. Specifically, the commenters
argued that CMS should give MA
organizations a credit for erroneous
diagnoses they submitted from their
providers’ claims up to the rate
identified by CMS as the applicable FFS
Adjustor in the RADV program. The
commenters also argued that there is no
reason to require that both MA and FFS
diagnosis data be scrutinized for error
rates when determining retroactive
payment adjustments, while not
engaging in a similar adjustment process
when paying plans prospectively.
Response: We thank the commenters
for their input. We are not finalizing the
proposed amendment to § 410.322(e).
However, we emphasize that our
decision to not finalize this regulatory
proposal does not change CMS’ existing
contractual requirement that MA
organizations must certify (based on
best knowledge, information, and belief)
the accuracy, completeness, and
truthfulness of the risk adjustment data
they submit to CMS. Further, this
decision does not change the long-
standing risk adjustment data
requirement that a diagnosis submitted
to CMS by an MA organization for
payment purposes must be supported by
medical record documentation.
Comment: A few commenters
supported CMS’ proposal to remove the
current date of January 31 as the annual
final risk adjustment data submission
deadline and replace it with the
provision that CMS will announce the
deadline annually, with the proviso that
CMS’ timing of this annual deadline
always allows sufficient opportunity for
organizations to make final data
submissions. Several other commenters
stated their concern about this proposed
change in deadline, including a concern
that CMS might announce a deadline
earlier than January 31 in some years.
These commenters requested that CMS
clarify that the annual deadline would
never be before January 31, and a few
commenters suggested that the
regulation state that the deadline is
January 31 but may be extended.
Finally, a few commenters requested
that CMS not change the January 31 date
to a floating date, in order to allow
operational stability.
Response: We are not finalizing this
proposal at this time.
Comment: Many commenters
disagreed with CMS’ proposal under
§ 410.322(g)(2) that, after the final risk
adjustment data submission deadline,
CMS would permit submission of data
to correct overpayments but not permit
late submission of diagnosis data that
would result in additional payment,
asserting that this asymmetrical
approach does not promote CMS’ stated
goal of improving payment accuracy.
The commenters maintained that an MA
organization should be allowed to
submit additional diagnoses after the
final risk adjustment data submission
deadline to correct not only an
overpayment to the MA organization,
but also an underpayment. A
commenter recommended that, after the
final deadline, MA organizations should
be able to submit paired deletions-
additions of diagnoses as long as the
result is not an increased payment to the
organization but a smaller reduction in
payment than would otherwise occur if
only the deletion were submitted; for
example, an MA organization may want
to delete the diagnosis code for diabetes
with acute complications and replace it
with the code for diabetes without
complications so that it loses only some
of the payment. Finally, a commenter
requested that CMS allow exceptions to
the general rule that no new diagnoses
may be submitted after the final risk
adjustment data submission deadline for
special circumstances such as system
failures, file formatting issues, and other
technical problems.
Response: For a given payment year
(which is a calendar year), CMS applies
diagnoses from the previous year (the
data collection year) to calculate
beneficiary risk scores used to risk-
adjust payments to MA organizations in
the payment year. MA organizations
must finalize any corrections and new
submissions of diagnosis data for a data
collection year by January 31 of the year
after the payment year. That is, we
allow 13 months after the end of the
diagnosis year for MA organizations to
identify errors in data they have
submitted (that is, deleting diagnoses
from CMS’ systems) and to identify and
submit additional diagnoses that were
not submitted during the diagnosis year.
We believe that is a very reasonable
period of time to finalize risk
adjustment data for a diagnosis year.
These risk adjustment processes have
been in place for many years, and we
believe it is the responsibility of MA
organizations to have internal audit
processes in place allowing them to
finalize their risk adjustment data for a
payment year by the conclusion of this
13-month period. Therefore, we are
finalizing, as proposed, the provision
codified at § 422.310(g)(2)(ii) that, after
the final deadline, an MA organization
may submit risk adjustment data to
correct overpayments but not to add
payments.
Comment: A commenter supported
CMS’ proposal to limit post-deadline
modifications to deletions of incorrect
diagnoses but requested that CMS offer
one additional opportunity to eliminate
unsupported diagnosis codes in advance
of a RADV audit.
Response: When we are preparing to
initiate a RADV audit cycle, all MA
organizations are notified that they
should eliminate unsupported
diagnoses from CMS’ systems by a date
specified in the notice. Subsequently,
we inform the contracts that have been
selected for RADV.
In summary, after consideration of the
public comments received, we are not
finalizing the proposed amendment to
§ 410.322(e). Also, we are not finalizing
at this time our proposal at
§ 422.310(g)(2)(ii) to remove the current
date of January 31 as the annual final
risk adjustment data submission
deadline and replace it with the
provision that CMS will announce the
deadline annually. We are finalizing as
proposed the restructuring of
§§ 422.310(g)(2) and the 422.310(g)(2)(ii)
provision to prohibit submission of
diagnoses for additional payment after
the final risk adjustment data
submission deadline. We did not
receive any comments on subparagraph
(g)(3) and are finalizing it as proposed.
3. RADV Appeals
a. Background
We published final Risk Adjustment
Data Validation (RADV) appeals
regulations in the April 15 2010 Federal
Register (75 FR 19677). These rules
were proposed and finalized under our
authority to establish Medicare
Advantage (MA) program standards at
section 1856(b)(1) of the Act and are
found at § 422.311 et seq. Since
finalizing these rules in 2010, we
conducted additional RADV audits and
determined that some of the appeals
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00084 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29927
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
provisions finalized in the 2010 RADV
Appeals final rule should be modified to
strengthen and streamline the RADV
appeals process and to prevent
confusion. Therefore, we proposed
revisions to the RADV appeals
regulations on January 10, 2014. These
proposed RADV provisions will apply
to any RADV determinations issued on
or after the effective date of this
regulation.
We proposed changing certain RADV
definitions at § 422.2. Specifically, we
proposed removing the definition Initial
Validation Contractor (IVC); removing
the definition of RADV payment error
calculation appeal process; and
removing the definition of ‘‘One Best
Medical Record for the purposes of
Medicare Advantage Risk Adjustment
Data Validation (RADV)’’. In addition,
we proposed adding one new definition
by specifically defining the RADV
appeals process. We also proposed
revising the definitions of ‘‘Risk
Adjustment Data Validation (RADV)’’
and ‘‘attestation process’’ within the
RADV appeals context. Furthermore, we
proposed amending RADV definitions at
§ 422.2 to specify that the Secretary,
along with CMS, could conduct RADV
audits.
At § 422.311, we proposed to update
select RADV appeals terminology. We
proposed amending the RADV
regulations by adopting one common
term to refer to RADV audit reports:
‘‘RADV Audit Report’’. As mentioned
earlier, we proposed removing from the
RADV regulations the term—‘‘Initial
Validation Contractor, or IVC,’’ since
RADV medical record review process no
longer utilizes ‘‘initial’’ and
‘‘secondary’’ validation contractors to
conduct medical record review under
RADV. Instead, we now utilize medical
record reviewers to code medical
records who may be employed by the
same or different medical record review
contractors.
At § 422.311(c)(1), we proposed to
simplify the RADV appeals process by
combining the two existing RADV
appeal procedures—one for medical
record review and one for payment error
calculation—into one set of
requirements and one process
comprised of three administrative steps:
Reconsideration, hearing officer review,
and CMS Administrator-level review.
Combining these existing RADV
medical record review determination
and payment error calculation appeals
policies and processes improves the
overall appeals process by simplifying
the overall RADV appeals process and
reducing burden on all parties involved
in the RADV appeals process. We also
believed that doing so improves overall
RADV appeals procedures by providing
clarity that leads to greater efficiencies
in adjudicating RADV appeals. Within
this overall framework, we also
proposed defining issues that would be
eligible for RADV appeal at
§ 422.311(c)(2) and issues that would
not be eligible for RADV appeals under
this combined-appeal process at
§ 422.311(c)(3). We further proposed
defining the manner and timing of a
request for RADV appeal at
§ 422.311(c)(2)(iii), a reconsideration
process at § 422.311(c)(6), a hearing
process at § 422.311(c)(7)(iv), and an
Administrator-level review at
§ 422.311(c)(8).
At § 422.311(a), we proposed that the
Secretary, along with CMS, be permitted
to conduct RADV audits beginning with
the effective date of this regulation.
Because of the absence of a clearly-
defined burden of proof standard for
RADV medical record review
determination appeals, at § 422.311(c)(4)
we proposed adoption of a burden of
proof standard for all RADV
determinations—be they payment error
calculation or RADV medical record
review determinations—whereby the
burden would be on MA organizations
to prove, based on a preponderance of
the evidence, that CMS’s
determination(s) was (were) erroneous.
At § 422.311(b)(2) we proposed
changing the compliance date for
meeting RADV audit requirements for
the validation of risk adjustment data to
the due date when MA organizations
selected for RADV audit must submit
medical records to the Secretary—and
not only CMS.
We received comments from health
plans, managed care industry trade
associations, providers, provider trade
associations and other interested
parties. These comments have resulted
in changes to the previously described
proposals, as discussed later in this
section. Some of the comments we
received did not apply to the proposed
RADV appeals processes. However,
because some of these comments apply
to underlying RADV audit process, we
are responding to certain comments
because they appear to be relevant to the
RADV appeals process. Other comments
were clearly outside the scope of our
proposed rule, so we have not included
responses to those comments.
b. RADV Definitions
We proposed to amend the RADV
definitions at § 422.2 as follows:
• Removing the following definitions:
++ ‘‘Initial Validation Contractor
(IVC)’’ means the first level of medical
record review under the RADV audit
process.
++ ‘‘RADV payment error calculation
appeal process’’ means an
administrative process that enables MA
organizations that have undergone
RADV audit to appeal the CMS
calculation of an MA organization’s
RADV payment error.
++ ‘‘The one best medical record for
the purposes of Medicare Advantage
Risk Adjustment Validation (RADV)’’
means the clinical documentation for a
single encounter for care (that is, a
physician office visit, an inpatient
hospital stay, or an outpatient hospital
visit) that occurred for one patient
during the data collection period. The
single encounter for care must be based
on a face-to-face encounter with a
provider deemed acceptable for risk
adjustment and documentation of this
encounter must be reflected in the
medical record.
• Adding the following definition:
++ ‘‘RADV appeal process’’ means an
administrative process that enables MA
organizations that have undergone
RADV audit to appeal the Secretary’s
medical record review determinations
and the Secretary’s calculation of an MA
organization’s RADV payment error.
• Revising the following definitions:
++ Risk adjustment data validation
(RADV) audit means a payment audit of
a Medicare Advantage (MA)
organization administered by CMS or
the Secretary that ensures the integrity
and accuracy of risk adjustment
payment data.
++ ‘‘Attestation process’’ means a
CMS-developed RADV process that
enables MA organizations undergoing
RADV audit to submit CMS-generated
attestations for eligible medical records
with missing or illegible signatures or
credentials. The purpose of the CMS-
generated attestations is to cure
signature and credential issues for
eligible medical records. CMS-generated
attestations do not provide an
opportunity for a provider or supplier to
replace a medical record or for a
provider or supplier to attest that a
beneficiary has the medical condition.
We received no comments specifically
recommending modifications to the
proposed definitions as stated, though
we did receive comments regarding the
policy behind some of these definition
changes. The policy comments will be
addressed later in this rule, though we
are finalizing the specific definitions
without modification.
c. Publication of RADV Methodology
In the October 22, 2009 proposed rule,
and as reinforced in the April 15, 2010
final rule, we indicated that we would,
‘‘publish its RADV methodology in
some type of public document—most
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00085 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29928
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
likely, a Medicare Manual, so that the
public can review and provide comment
as it deems necessary’’. We also
indicated that we would provide an
annual notice of RADV audit
methodology. Our last RADV-related
notice of methodology was published in
February 2012. We will continue to
publish a notice of the methodology
employed, but will do so only if there
is a change in the RADV methodology
that would require publication. We note
that these notices of RADV audit
methodology updated information
provided on RADV audit methodology
provided in the October 22, 2009
proposed rule and April 15, 2010 final
rule.
In addition, we provided in the
October 22, 2009 proposed rule
preamble that we would provide an
expanded explanation of methodology
and payment error calculation factors as
a part of each audit report of findings
that we send to MA organizations that
undergo RADV audit. Such explanation
and factors have been and will continue
to be part of the RADV audit report(s)
that CMS provides health plans that
have undergone RADV audits.
d. Proposal To Update RADV Appeals
Terminology (§ 422.311)
Current RADV regulations utilize the
following terms for the CMS-issued
RADV audit report: Audit report post
medical record review; RADV audit
report; IVC-level RADV audit report;
and RADV audit report of finding. This
use of multiple terms to refer to what is
the same audit report (the RADV audit
report that CMS issues following
conclusion of the medical record review
portion of the audit) is potentially
confusing. Therefore, we proposed
amending the RADV regulations
throughout to adopt one common term
to refer to RADV audit reports: ‘‘RADV
Audit Report’’. By standardizing
terminology throughout the RADV
regulations, the proposed amendment
provides clarity which may lead to
increased efficiency.
As mentioned earlier in the
description of RADV-related definitions
that have changed, we have revised
certain RADV-related definitions to
accommodate changes to both the RADV
audit process and the RADV appeals
process. One definition that we have
removed from the RADV regulations is
Initial Validation Contractor, or IVC.
The RADV medical record review
process no longer utilizes ‘‘initial’’ and
‘‘secondary’’ validation contractors to
conduct medical record review under
RADV. Instead we now utilize medical
record reviewers to code medical
records undergoing RADV review.
These reviewers may be employed by
the same or different medical record
review contractors. Therefore, the term
‘‘IVC’’ is no longer relevant to the RADV
audit process. As a result, we proposed
to remove this term from the RADV
regulations at the following citations:
§ 422.311(c)(2)(i)(B) through (D);
§ 422.311(c)(2)(ii)(B),
§ 422.311(c)(2)(iii)(A), § 422.111(c)(2)(v),
(vi), § 422.311(c)(3)(ii)(A), and
§ 422.311(c)(3)(iii)(A) and (B). We
invited comment on this proposal.
Comment: A commenter requested
that CMS verify that the medical record
review error determination standard,
which presently requires multiple
review determinations by independent
coders to confirm a CMS–HCC coding
error, remains in effect and is not
altered by this proposed rule.
Response: While we did not propose
RADV coding changes, we believe the
question merits a response. We believe
that our proposal to remove the
definition of ‘‘Initial Validation
Contractor’’ (IVC) may have led some to
believe that we were abandoning RADV
audit processes that require multiple
levels of independent medical record
review (coding) by independent
reviewers before we will confirm a
CMS–HCC coding error. This standard
has not changed, notwithstanding the
removal of the term IVC from the RADV
appeals rules. We continue to utilize
medical record reviewers to code
medical records undergoing RADV
review, though these reviewers may
now be employed by the same or
different medical record review
contractors. The principle of
independent review and multiple
confirmations of an identified CMS–
HCC remain in effect.
e. Proposal To Simplify the RADV
Appeals Process
Currently, there are two types of
RADV-related appeals processes
described in our regulations at
§ 422.311: Medical record review-
determination appeals and RADV
payment error calculation appeals.
RADV medical record review-
determination appeal requirements and
procedures are discussed at
§ 422.311(b)(3) and § 422.311(c)(2).
Medical record review determination
appeal is a two-stage administrative
appeal process—the first step is a
hearing by a hearing officer, followed by
a CMS Administrator—level review.
This appeal procedure provides MA
organizations with an opportunity to
appeal RADV medical record review
determinations that are made by coders
reviewing the medical record
documentation submitted by MA
organizations undergoing RADV audit.
The second type of RADV appeal,
payment error calculation appeal, is
discussed at § 422.311(c)(3). Payment
error calculation appeal is a three-
pronged appeal process:
reconsideration, followed by a hearing
officer review, followed by CMS
Administrator—level review. This
appeal process was specifically
designed to afford MA organizations the
opportunity to appeal CMS’s contract-
level RADV payment error calculation.
We proposed that the administrative
appeals language described at
§ 422.311(b)(3) and § 422.311(c)(2) for
RADV medical record review
determination appeals and
§ 422.311(c)(3) for RADV payment error
calculation appeals be replaced with
new regulatory language proposed
§ 422.311(c)(1), that combines the two
existing RADV appeal policies and
procedures into one set of requirements
and one process. We proposed to
combine the two RADV appeals
processes into one combined RADV
appeals process that is comprised of
three administrative steps:
Reconsideration, hearing officer review,
and CMS Administrator-level review. A
three-step administrative appeals
process comprising reconsideration,
hearing officer review, and
Administrator-levels of review is a
common administrative appeals model
used elsewhere within the Medicare
managed care program, such as in
appealing contract award
determinations and intermediate
sanctions. The combined RADV appeal
process that we proposed at new
§ 422.311(c)(1), also has the benefit of
simplifying what is today a complex
two-track appeal process into one
process. While both CMS and the MA
industry will benefit from simplifying
this process, MA organizations also
obtain an additional level of review
under the combined approach since MA
organizations will be afforded a
reconsideration appeal step for medical
record review determinations that is
today—not part of the existing RADV
appeal process. Shortening the existing
two-track appeal process should also
reduce the resources and level of effort
needed from both MA organizations and
CMS in participating in a RADV appeal
proceeding. Under this proposal, MA
organizations can simply request to
appeal their RADV audit findings one
time and specify whether they want to
appeal either their medical record
review determination(s), payment error
calculation, or both. The specific details
regarding this proposed process follow.
We proposed these changes based upon
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00086 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29929
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
our experience with RADV appeals and
because we hope to reduce the burden
associated with undertaking RADV
appeals on both MA organizations and
CMS. The details of the proposed policy
and procedure follow.
(1) Issues Eligible for RADV Appeal
Current regulations at §§ 422.311(c)(2)
et seq., and 422.311(c)(3) et seq., specify
RADV-related medical record review
and payment error calculation
documents and issues eligible for the
medical record review determination
and payment error calculation appeal
processes. We proposed to amend the
policies and procedures around issues
eligible for RADV appeals at
§ 422.311(c)(2) and § 422.311(c)(3) by
combining proposed policies and
procedures for the existing two-pronged
appeal approach into one set of policies
and procedures for RADV appeals at the
new § part 422.311(c)(2)(iv). At
§ 422.311(c)(2)(i), we proposed that as a
general rule, MA organizations may
appeal RADV medical record review
determinations and RADV payment
error calculation, though in order to be
eligible to pursue these appeals, we
specify at proposed § 422.311(c)(2)(i)(A)
and (B) that MA organizations must
adhere to established RADV audit
procedures and requirements and
adhere to RADV appeals procedures and
requirements. At § 422.311(c)(2)(ii) we
proposed that failure to follow RADV
audit procedures and requirements and
RADV appeals procedures and
requirements will render the MA
organization’s request for RADV appeal
invalid. Furthermore, at proposed
§ 422.311(c)(2)(iii) we stipulate that the
MA organization’s written request for
medical record review determination
appeal must specify the audited HCC(s)
that have been identified pursuant to
RADV audit as being in error, and
further specify that MA organizations
must provide a justification in support
of the audited HCC(s) that the MA
organization elects to appeal. At
§ 422.311(c)(2)(i)(iv) we proposed that
for each audited HCC, MA organizations
may appeal one medical record that has
undergone RADV medical record review
and that if an attestation was submitted
to cure a signature or credential issue,
that attestation may likewise be
included in the HCC appeal. For
example, if an MA organization
submitted a medical record that did not
contain a signature and/or credential—
and the MA organization submitted an
attestation to cure the error that CMS
subsequently failed to accept—the MA
organization could choose to appeal
CMS’s determination to not accept the
submitted attestation. We reiterate that
the purpose of CMS-generated
attestations is to cure signature and
credential errors associated with an
eligible submitted medical record and
not to provide an opportunity for a
provider or supplier to attest that a
beneficiary has a certain medical
condition. Evidence for the existence of
the medical condition is found in a
medical record.
We proposed to modify our language
at § 422.311(c)(2)(i)(v) to clarify existing
RADV appeals provisions which
stipulate that MA organizations must
adhere to the ‘‘one best medical record’’
policy. Under changes to the RADV
audit methodology announced by CMS
in February 2012, we now allow MA
organizations to submit more than one
medical record (that is, more than the
‘‘one best medical record’’) during the
RADV audit process to validate an
audited CMS–HCC. However, for
purposes of appealing a CMS medical
record review determination, we will
not permit organizations to appeal
multiple medical records but will
instead—require that MA organizations
identify a record from amongst those
records submitted, and to submit that
record for appeal. For each audited
HCC, MA organizations may appeal
only one medical record that has
undergone RADV review. This policy
was published in the February 2012
White Paper and is not included in this
final rule.
At § 422.311(c)(2)(vi) we proposed
that a written request for RADV
payment error calculation appeal must
clearly specify the MA organization’s
own RADV payment error calculation
and must also specify where the
payment error calculation was
erroneous.
(2) Issues Not Eligible for RADV
Appeals
At § 422.311(c)(3) we proposed
documents and issues that are ineligible
for RADV appeals. Consistent with the
overall approach of combining into one
RADV appeals process what was
heretofore two separate RADV appeals
processes—by way of this new proposed
section, we propose to amend existing
regulations at § 422.311(c)(3). At new
§ 422.311(c)(3), we proposed that MA
organizations’ request for appeal may
not include HCCs, medical records or
other documents beyond the audited
HCC, selected medical record and any
accompanying attestation that the MA
organization chooses to appeal. We
specify at § 422.311(c)(3)(ii) that the MA
organizations may not appeal CMS’s
medical record review determination
methodology or CMS’s payment error
calculation methodology. This is a
clarification to existing RADV
regulations at § 422.311(c)(3)(D) which
specifies that MA organizations may not
appeal CMS’s payment error calculation
methodology. At § 422.311(c)(3)(iii) we
specify that MA organizations may not
appeal RADV medical record review-
related errors when appealing RADV
error-calculation issues since medical
record review determination issues
must be resolved before we can
calculate RADV payment errors. And at
§ 422.311(c)(3)(iv) we specify that RADV
errors that result from an MA
organization’s failure to submit a
medical record are not eligible for
appeal.
(3) Manner and Timing of a Request for
RADV Appeal
We proposed to replace existing
RADV regulations at § 422.311(c)(2)(iii)
et seq., and § 422.311(c)(3)(iii) et seq.,
regarding the manner and timing of a
request for RADV appeals. Again, at
§ 422.311(c)(5), we proposed to combine
the formerly two separate sets of
requirements and procedures into one
RADV appeals process addressing the
request for RADV appeal. At
§ 422.311(c)(5)(i) we proposed that at
the time the Secretary issues her RADV
audit report, the Secretary notifies
audited MA organizations that they may
appeal RADV HCC errors that are
eligible for medical record review
determination appeal and may appeal
the Secretary’s RADV payment error
calculation. At § 422.311(c)(5)(ii) we
specify that MA organizations have 30
days from the date of CMS’s issuance of
the RADV audit report to file a written
request with CMS for RADV appeal.
This request for RADV appeal must
specify whether the MA organization
requests medical record review
determination appeal, whether the MA
organization requests RADV payment
error calculation appeal, or whether the
MA organization requests both medical
record review determination appeal and
RADV payment error calculation
appeal—and in each instance—the
issues with which the MA organization
disagrees, and the reasons for the
disagreements. See proposed regulations
at § 422.311(c)(6).
In proposed § 422.311(c)(5)(ii), we
specify that while MA organizations
may now elect to appeal either medical
record review determination, payment
error calculation, or both—they must
notify CMS which issues they will
appeal at the same time. This new
provision replaces existing RADV
appeals requirements regarding
notification at § 422.311(c)(2)(iii) and
§ 422.311(c)(3)(iii)(C).
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00087 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29930
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
For MA organizations that elect both
medical record review determination
appeal and RADV payment error
calculation appeal, we specify at
§ 422.311(c)(5)(iii)(A) and (B) that the
Secretary will adjudicate the request for
RADV payment error calculation
following conclusion of reconsideration
of the MA organization’s request for
medical record review determination
appeal. This is necessary because RADV
payment error calculations are based
upon the outcomes of medical record
review determinations. For example, for
an MA organization that appeals both
medical record review determinations
and payment error calculations, the
reconsideration official would first
adjudicate and rule on the medical
record review determinations and then
proceed to recalculate the RADV
payment error.
(4) Reconsideration Stage
Under current RADV appeals
procedures, only the RADV payment
error calculation appeal process
contains a reconsideration step. We
proposed to amend existing regulations
at § 422.311(c)(3)(iii)(C) and
§ 422.311(c)(3)(v), (vi), and (vii) by
proposing a new reconsideration stage
for RADV appeals at § 422.311(c)(6) et
seq. Reconsideration is the first stage of
the new RADV appeals process and will
apply to both medical record review
determinations and error calculation
issues being appealed. Therefore, MA
organizations that elect to appeal RADV
audit findings de facto begin the appeal
process with the reconsideration step.
At proposed § 422.311(c)(6)(i) we
specify that a MA organization’s written
request for medical record review
determination reconsideration must
specify the audited HCC identified as
being in error that the MA organization
wishes to appeal; and to provide a
justification in support of the audited
HCC chosen for appeal. At proposed
§ 422.311(c)(6)(ii) we specify that the
MA organizations’ written request for
payment error calculation
reconsideration must include the MA
organization’s own RADV payment error
calculation that clearly indicates where
the RADV payment error calculation
was erroneous. The request for payment
error calculation reconsideration may
also include additional documentary
evidence pertaining to the calculation of
the error that the MA organization
wishes the reconsideration official to
consider.
At proposed § 422.311(c)(6)(iii) we
describe the conduct of the
reconsideration process that is being
proposed. We specify that for medical
record review determination
reconsideration, a medical record
review professional who was not
involved in the initial medical record
review determination of the disputed
HCC reviews the medical record and
accompanying dispute justification; and
reconsiders the initial audited HCC
medical record review determination.
For payment error calculation
reconsideration, we ensure that a third
party not involved in the initial RADV
payment error calculation reviews the
RADV payment error calculation,
reviews the MA organization’s own
RADV payment error calculation, and
recalculates the payment error in
accordance with CMS’s RADV payment
error calculation procedures.
At proposed § 422.311(c)(6)(iv), we
specify that the reconsideration official
issues a written reconsideration
decision to the MA organization, and
that the reconsideration official’s
decision is final unless the MA
organization disagrees with the
reconsideration official’s decision. If the
MA organization disagrees with the
reconsideration official’s decision, it
may request a hearing.
(5) Hearing Stage
Existing regulations at
§ 422.311(c)(2)(iv) through (ix) and
§ 422.311(C)(4) et seq., specify the
procedures under which CMS conducts
hearings under the RADV appeals
process for medical record review and
payment error calculation. We proposed
to replace these provisions with new
hearing requirements and procedures at
§ 422.311(c)(7)(iv).
At § 422.311(c)(7)(i), we proposed that
at the time the RADV appeals
reconsideration official issues his/her
reconsideration determination to the
MA organization, the reconsideration
official notifies the MA organization of
any RADV audited HCC errors and or
payment error calculations that are
eligible for RADV hearing. At
§ 422.311(c)(7)(ii), we specify that a MA
organization that requests a hearing
officer review must do so in writing in
accordance with procedures established
by CMS. At § 422.311(c)(7)(iii), we
specify that a written request for a
hearing must be filed with the Hearing
Officer within 30 days of the date the
MA organization receives the
reconsideration officer’s written
reconsideration decision. If the MA
organization appeals the medical record
review reconsideration determination,
the written request for RADV hearing
must include a copy of the written
decision of the reconsideration official;
must specify the audited HCCs that the
reconsideration official confirmed as
being in error; and must specify a
justification as to why the MA
organization disputes the
reconsideration official’s determination.
If the MA organization appeals the
RADV payment error calculation, the
written request for RADV hearing must
include a copy of the written decision
of the reconsideration official and must
include the MA organization’s own
RADV payment error calculation that
clearly specifies where the CMS’s
payment error calculation was
erroneous.
At § 422.311(c)(7)(iv), we proposed
that a CMS hearing officer conduct the
RADV hearing. At § 422.311(c)(7)(v), we
specify terms and conditions under
which a hearing officer may be
disqualified. A hearing officer may not
conduct a hearing in a case in which he
or she is prejudiced or partial to any
party or has any interest in the matter
pending for decision. A party to the
hearing who objects to the assigned
hearing officer must notify that officer
in writing at the earliest opportunity.
The hearing officer must consider the
objections, and may, at his or her
discretion, either proceed with the
hearing or withdraw. If the hearing
officer withdraws, another hearing
officer will conduct the hearing. If the
hearing officer does not withdraw, the
objecting party may, after the hearing,
present objections and request that the
officer’s decision be revised or a new
hearing be held before another hearing
officer. The objections must be
submitted in writing to CMS.
At § 422.311(c)(7)(vi), we proposed
that the hearing officer reviews the
medical record and any accompanying
attestation that the MA organization
selected for review, the reconsideration
official’s payment error calculation (if
appealed), the reconsideration official’s
written determination, and the written
justification submitted by the MA
organization and CMS in response to the
reconsideration official’s determination.
At § 422.311(c)(7)(vii), we proposed
RADV appeal hearing procedures. We
proposed that the hearing officer has
full power to make rules and establish
procedures, consistent with the law,
regulations, and rulings. These powers
include the authority to dismiss the
appeal with prejudice and take any
other action which the hearing officer
considers appropriate, including for
failure to comply with RADV audit and
appeals rules and procedures. We
proposed that the hearing be altogether
on the record unless the hearing officer,
at his or her full discretion, approves a
parties request for a live or telephonic
hearing regarding some or all of the
medical records in dispute, or if the
hearing office schedules a live or
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00088 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29931
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
telephonic hearing on its own motion.
The hearing officer’s review will be
solely limited to the record. The record
is comprised of the RADV reviewed
medical record and any accompanying
attestation that the MA organization
selected for review, the reconsideration
official’s payment error calculation (if
appealed), the reconsideration official’s
written determination, the written
justification submitted by the MA
organization in response to the
reconsideration official’s determination,
and written briefs from the MA
organization explaining why they
believe the reconsideration official’s
determination was incorrect. In
addition, the record will be comprised
of a brief from CMS that responds to the
MA organization’s brief.
In terms of specifying the conduct of
the hearing, we proposed at
§ 422.311(c)(7)(vii)(B) that the hearing
officer neither receives testimony nor
accepts any new evidence that is not
part of the record. At § 422.311(c)(7)(vii)
we proposed that the hearing officer be
given the authority to decide whether to
uphold or overturn the reconsideration
official’s decision, and pursuant to this
decision—to send a written
determination to CMS and the MA
organization, explaining the basis for
the decision.
At § 422.311(c)(7)(ix), we proposed
that in accordance with the hearing
officer’s decision, a third party not
involved in the initial RADV payment
error calculation recalculate the MA
organization’s RADV payment error and
issue a new RADV audit report to the
appellant MA organization and CMS.
For MA organizations appealing the
RADV payment error calculation only,
we proposed that a third party not
involved in the initial RADV payment
error calculation recalculate the MA
organization’s RADV payment error and
issue a new RADV audit report to the
appellant MA organization and CMS.
At§ 422.311(c)(7)(x) we proposed that
the hearing officer’s decision be final
unless the decision is reversed or
modified by the CMS Administrator.
(6) CMS Administrator Review Stage
Existing regulations at
§ 422.311(c)(2)(x) et seq., and
§ 422.311(C)(4)(vi) et seq., specify the
CMS Administrator-level review
procedures that CMS adheres to under
the current RADV appeals process for
medical record review determinations
and payment error calculation. We
proposed to replace these regulations
with new RADV appeal-related CMS
Administrator review requirements and
procedures at § 422.311(c)(8).
At § 422.311(c)(8)(i) and (ii), we
proposed that a request for CMS
Administrator review must be made in
writing within 30 days of receipt of the
hearing officer’s decision; and must be
filed with the CMS Administrator by
CMS or an MA organization. At
§ 422.311(c)(8)(iii), we proposed that
after receiving a request for review, the
CMS Administrator has the discretion to
elect to review the hearing officer’s
decision or to decline to review the
hearing officer’s decision. At
§ 422.311(c)(8)(iv) we proposed that if
the CMS Administrator elects to review
the hearing decision—the Administrator
acknowledges the decision to review the
hearing decision in writing and notifies
CMS and the MA organization of their
right to submit comments within 15
days of the date of the notification. At
§ 422.311(c)(8)(iv)(B), we proposed that
the CMS Administrator be limited to the
review of the record and that the record
be comprised of the hearing record, and
written arguments from the MA
organization and/or CMS explaining
why either or both parties believe the
hearing officer’s determination was
correct or incorrect.
Regarding Administrator-level review
procedures at § 422.311(c)(8)(vi), we
proposed that the Administrator reviews
the record and determines whether the
hearing officer’s determination should
be upheld, reversed, or modified. At
§ 422.311(c)(8)(v), we proposed that the
Administrator render his or her final
decision in writing to the parties within
60 days of acknowledging his or her
decision to review the hearing officer’s
decision. At § 422.311(c)(8)(vi), we
proposed that the decision of the
hearing officer become final if the
Administrator declines to review the
hearing officer’s decision or does not
make a decision within 60 days.
Combining these existing RADV
medical record review determination
and payment error calculation appeals
policies and processes improves the
overall appeals process by strengthening
the depth and integrity of these
procedures. We also believe that doing
so improves overall RADV appeals
procedures by providing clarity that
leads to greater efficiencies in
adjudicating RADV appeals. We
welcomed comments on these
proposals.
We received the following comments
and our response follows:
Comment: Several commenters agreed
that combining the RADV medical
record review determination and
payment error calculation appeals
policies and processes into one
combined appeals process strengthens
the overall appeals process and should
reduce administrative burden. A
commenter disagreed with this
assessment. Another commenter
indicated that appealing both a medical
record review determination and the
payment error calculation concurrently
within a 30-day timeframe would be
problematic.
Response: We continue to believe the
combining two RADV appeals processes
into one combined appeals process will
improve efficiency and reduce
administrative burden. Previously, MA
organizations wishing to appeal both
medical record review determinations
and a RADV payment error calculation
would have been required to participate
in two hearings and two Administrator-
level reviews. Under our proposal, these
same organizations need only
participate in one hearing and one
Administrator review. Regarding the
notion that appealing both a medical
record review determination and the
payment error calculation concurrently
within a 30-day timeframe would be
problematic, we believe the commenter
misunderstood how the proposed
process is intended to work. The
proposed provision at
§ 422.311(c)(5)(ii)(c) states that for MA
organizations that appeal both medical
record review determination appeal and
RADV payment error calculation
appeal—the Secretary adjudicates the
request for RADV payment error
calculation appeal following conclusion
of reconsideration of the MA
organization’s request for medical
record review determination appeal and
not concurrently as the commenter
asserted. However, to provide additional
clarity to the provision, we have
amended § 422.311(c)(5)(iii)(B) to state
that MA organization’s request for
appeal of their RADV payment error
calculation will not be adjudicated until
appeals of RADV medical record review
determinations filed by the MA
organization have been completed and
the decisions are final for that stage of
appeal. We trust this clarifies this
provision and CMS therefore finalizes
this proposal.
Comment: A commenter objected to
the proposed provision at
§ 422.311(c)(2)(ii) that failure to follow
RADV audit procedures and
requirements and RADV appeals
procedures and requirements will
render the MA organization’s request for
RADV appeal invalid. This commenter
stated procedural issues should not
render an appeal invalid unless they
undermine the integrity of the audit
results or are otherwise significantly
prejudicial.
Response: We disagree. RADV is an
inherently complex administrative
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00089 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29932
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
process and the appeals procedures we
have proposed are likewise detailed and
comprehensive. Failure by MA
organizations to follow RADV audit
procedures could compromise the
integrity of the administrative record
that will serve as the foundational
document that will be considered
during any appeals process. Moreover, if
we were to make subjective case-by-case
determinations regarding what defines
‘‘undermining the integrity of the audit
process,’’ then we would compromise
our ability to establish objective review
standards upon which to base appeals
determinations. Therefore, we are
finalizing this provision as proposed.
Comment: Several commenters
requested that CMS allow MA
organizations to appeal determinations
made on ‘‘additional’’ CMS–HCCs
abstracted during the medical record
review process. Some commenters
asserted that these additional CMS–
HCCs are underpayments for which they
are entitled payment. These commenters
also asserted that MA organizations that
do not receive credit for what they
believe to be additional CMS–HCCs
present in a submitted medical record
should be entitled to appeal the fact that
they did not receive credit.
Response: We disagree. We note that
an additional CMS–HCC is a CMS–HCC
that CMS uncovers during the review of
the MA organization’s submitted
medical record(s) for which it had not
received payment. We acknowledge that
in certain circumstances when CMS
uncovers these additional CMS–HCCs,
the MA organization can in fact receive
credit for these newly-discovered
diagnoses codes to offset the
overpayment findings resulting from the
medical record review of the audited
CMS–HCC. The RADV process
addresses additional CMS–HCCs, or
‘‘additionals,’’ as they are termed,
through the application of rules for
crediting a sampled enrollee with
additional CMS–HCCs that are
identified incidentally, during medical
record review. We emphasize that these
‘‘additional’’ diagnoses were not
submitted for payment by MA
organizations during the data collection
period for enrollees selected in the
sample, and yet in certain instances we
provide audited MA organizations
credit through our RADV medical
record review process. At its core,
RADV is an audit process that is
intended to validate the CMS–HCCs that
were submitted voluntarily by MA
organizations in order to determine
whether the risk adjustment portion of
payment were properly made. We
would note that the data collection
period for any given payment year
provides a substantial amount of time
for MA organizations to submit and/or
correct enrollee diagnoses data to reflect
an enrollee’s health status. The RADV
audit process is not intended to serve as
a de facto mechanism for extending the
data collection deadlines under which
MA organizations operate. For these
reasons, MA organizations will not be
permitted to appeal additional CMS–
HCC determinations found under the
RADV audit for which MA
organizations did not receive credit.
Comment: At § 422.311(c)(2)(i), CMS
proposed that for each audited CMS–
HCC, MA organizations may appeal one
medical record that has undergone
RADV medical record review and that if
an attestation was submitted to cure a
signature or credential issue, that
attestation may likewise be included in
the CMS–HCC appeal. In response to
this proposal, a commenter requested
that CMS allow MA organizations to use
an attestation to replace a medical
record. Another commenter
recommended that CMS change the
attestation process embedded in the
existing RADV audit procedures so that
when CMS notifies an MA organization
that an audited CMS–HCC was not
validated due to lack of signature or
credential, CMS would likewise allow,
after medical record review, submission
of an attestation to cure the identified
RADV error.
Response: The purpose of the CMS-
generated attestations is to provide MA
organizations with an opportunity to
cure signature and credential CMS–HCC
validation errors for eligible medical
records. CMS-generated attestations are
not intended to provide an opportunity
for a MA organizations, provider or
supplier to replace a medical record; or
for a provider or supplier to attest that
a beneficiary has the medical condition
reflected in the CMS–HCC at issue. Risk
adjustment rules require that allowable
diagnoses be verified in a medical
record, not attestation. Regarding the
recommendation that CMS notifies an
MA organization that an audited CMS–
HCC was not validated due to lack of
signature or credential pursuant to
RADV medical record review, we
believe MA organizations bear the
responsibility for identifying records
that do not contain signature or
credentials, and should do so at the
same time they submit medical records
to CMS for RADV medical record
review.
Comment: At § 422.311(c)(2)(iv), CMS
proposed a provision which stipulates
that notwithstanding these changes, for
purposes of appealing a CMS medical
record review determination, we will
not permit MA organizations to appeal
multiple medical records but will
instead require MA organizations to
identify one medical record from
amongst the records submitted, and
submit that record for appeal. For each
audited CMS–HCC, MA organizations
may appeal only one medical record
that has undergone RADV review.
Several commenters objected to CMS’s
proposal that RADV appeals be limited
to one medical record selected by the
MA organization. These commenters
believe CMS should not require MA
organizations to select one medical
record to appeal, but should rather
permit MA organizations to appeal
multiple medical records as part of the
proposed RADV appeal process.
Response: We believe these comments
are in part responding to information
that we provided in February 2012
regarding changes in RADV
methodology. At that time, we
announced that CMS would allow MA
organizations to submit more than one
medical record for CMS–HCC validation
during the RADV medical record review
stage of the RADV audit process. While
we now permit MA organizations to
submit more than one medical record
during the RADV audit process to
validate an audited CMS–HCC, only one
medical record is required and
ultimately utilized by CMS to validate
an audited CMS–HCC or conversely, to
make a determination that the audited
CMS–HCC is not present in the
submitted medical record. Since one
medical record is sufficient to validate
an audited CMS–HCC, we believe it is
reasonable to limit MA organizations to
selecting one medical record for
purposes of RADV appeal. Guidelines
set forth in the International
Classification of Diseases, Ninth Clinical
Revision (ICD–9) specify that the
information necessary to abstract a code
be contained in entirety in
documentation for one encounter (either
inpatient or outpatient). Multiple
records cannot be combined to obtain
sufficient documentation for a
diagnosis. Furthermore, risk adjustment
rules specify that only one diagnosis
submission throughout the entire data
collection period initiates a risk score
adjustment. Given this, multiple
medical record support is not required
to confirm the diagnosis. Therefore, the
appeal record should be carefully
selected to ensure payment is validated.
Therefore, we do not accept this
recommendation.
Comment: Several commenters
objected to CMS’s proposal at
§ 422.311(c)(3) to not permit MA
organizations to appeal either medical
record review determination or payment
error calculation methodology. A
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00090 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29933
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
commenter stated that MA organizations
should be allowed to identify and
explain their objections to audit and
appeals procedures and requirements
without losing their ability to pursue the
administrative appeals process.
Response: At § 422.311(c)(3)(ii), we
proposed that MA organizations would
not be permitted to appeal CMS’s
medical record review determination
methodology or CMS’s payment error
calculation methodology. We proposed
this requirement for the same reason
that we finalized the RADV appeals
requirement in 2010 that MA
organizations could not appeal the
RADV payment error calculation
methodology. The payment error
calculation methodology would be
known to audited MA organizations
before their RADV audit began. MA
organizations that questioned or did not
otherwise understand the methodology
would have an opportunity to seek
clarification from CMS regarding the
methodology at that time.
In December 2010, in response to
questions from the MA industry
regarding our RADV payment error
calculation methodology, we published
a white paper describing our RADV
payment error calculation
methodologies, and invited public
comment. In response to comments
received in February 2012, we
published a RADV-related notice of
methodology specifying the RADV
payment error calculation methodology
that the agency would utilize on a
moving-forward basis.
This same principle applies to the
way we conduct medical record review
within the RADV audit context. We
have long adhered to the International
Classification of Diseases, Ninth
Revision, Clinical Modification, or ICD–
9–CM, system to classify and assign
codes to health conditions abstracted
from medical records that MA
organizations submit to validate audited
CMS–HCCs. ICD–9–CM standards are
widely available to the public and will
be available to MA organizations before
RADV audits are initiated. We
anticipate continuing to adhere to these
standards until such time as new coding
standards (for example, ICD–10) are
universally adhered to in the United
States. We continue to believe that it is
essential that CMS adhere to a
universally accepted coding
classification system that is widely
available in the public domain when
conducting RADV audits.
We disagree that MA organizations
lose their ability to pursue the
administrative appeals process
described at § 422.311 when they
identify and explain objections to audit
and appeals procedures. MA
organizations can fully execute their
rights to RADV administrative appeals
as described at § 422.311 by following
applicable regulations. Those rules
clearly specify issues that are eligible for
RADV appeal at § 422.311(c)(2) and
§ 422.311(c)(3); and issues that are
ineligible for RADV appeal at
§ 422.311(c)(3). To the extent an MA
organization appeals RADV issues that
are eligible for RADV appeal that
request for appeal will go forward. To
the extent an MA organization appeals
issues that are ineligible for RADV
appeal; we will not act upon that
request for RADV appeal. The act of
identifying and explaining objections to
audit and appeals procedures will not in
and of itself nullify an MA
organization’s request to appeal issues
that are eligible for RADV appeal.
Comment: At § 422.311(c)(5)(ii) we
proposed that MA organizations have 30
days from the date of CMS’s issuance of
the RADV audit report to file a written
request with CMS for RADV appeal. At
§ 422.311(c)(7)(iii), we proposed that a
written request for a hearing must be
filed with the Hearing Officer within 30
days of the date the MA organization
receives the reconsideration officer’s
written reconsideration decision. At
§ 422.311(c)(8)(i) and (ii), we proposed
that a request for CMS Administrator
review must be made in writing within
30 days of receipt of the hearing officer’s
decision. Several commenters requested
that CMS consider providing MA
organizations additional time at each of
these steps within the RADV appeals
process to elect to pursue further
appeals activity. In most instances,
these commenters requested CMS
provide a 60-day response time instead
of the proposed 30-day response time.
Response: We agree with the
commenters’ recommendations and will
change the proposed response times
from 30 days to 60 days at
§ 422.311(c)(5)(ii), § 422.311(c)(7)(iii),
and § 422.311(c)(8)(i) and (ii).
Comment: A commenter
recommended that CMS correct a cross-
reference error between preamble
language and regulation text. The error
pertains to language at
§ 422.311(c)(6)(iv)(C) which references
the hearing process in accordance with
paragraph (c)(8). As stated in the
preamble, we believe this should be a
reference to (c)(7), which sets forth the
rules for requesting a hearing. Paragraph
(c)(8) relates to review by the CMS
administrator.
Response: We agree with commenter’s
recommended edit and have changed
the regulation text to specify paragraph
(c)(7) and not (c)(8).
Comment: A commenter
recommended that CMS clarify the
provision at § 422.311(c)(7)(vii)(B)(2)(i)
that states that either party be allowed
to request a live or telephonic hearing,
still subject to the hearing officer’s
discretion. The commenter
recommended that both CMS and the
MA organization be allowed to request
a live or telephonic hearing, still subject
to the hearing officer’s discretion.
Response: Proposed
§ 422.311(7)(vii)(B)(2)(i) specifies that
‘‘the parties may request a live or
telephonic hearing . . .’’ The term ‘‘the
parties’’ in this instance means CMS or
the MA organization, and not either
CMS or the hearing officer. Therefore,
either organization that is a party of the
hearing process may request a live or
telephonic hearing. This clarification
notwithstanding, the CMS
Administrator nevertheless maintains
the independent discretion to elect to
review the hearing officer’s decision or
to decline to review the hearing officer’s
decision. See § 422.311(7)(vii)(B)(2)(iii).
Comment: In proposed
§ 422.311(c)(8)(i) and (ii), CMS requires
that a request for CMS Administrator—
level review be filed with the CMS
Administrator by either CMS or an MA
organization. A commenter
recommended that CMS accept adverse
decisions by its hearing officers and not
be permitted to appeal them to the CMS
Administrator. Another requested
clarification whether an MA
organization would be given a
‘‘meaningful opportunity’’ to appeal a
hearing officer’s decision that is
favorable to CMS (and not to the MA
organization) if CMS’ inaction allows
the hearing officer’s decision to become
finalized.
Response: We disagree that only MA
organizations should be provided an
opportunity to appeal a hearing officer’s
adverse determination to the
Administrator. Doing so would provide
MA organizations with a level of due
process not available to CMS, thus
weighing the appeals process in favor of
MA organizations. Consequently, we
believe that both parties should be able
to appeal a hearing officer’s unfavorable
decision to the Administrator level of
review. Regarding the question of
whether an MA organization would be
given a meaningful opportunity to
appeal a hearing officer’s decision that
is favorable to CMS if CMS’ inaction
allows the hearing officer’s decision to
become finalized, we reiterate that a
decision that is favorable to CMS would
inherently be unfavorable to the MA
organization rendering them eligible to
request review by the CMS
Administrator so long as other appeals-
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00091 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29934
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
pre-requisites (for example, following of
applicable rules, etc.) have been met. At
proposed § 422.311(c)(8)(ii) we specify
that an MA organization that has
received a hearing officer’s decision
may request review by the CMS
Administrator.
Comment: Several commenters
objected to CMS’s proposed RADV
appeals-related documentation
standards. A commenter requested that
CMS reconsider its position that the
medical record that they designate for
RADV appeal be selected from one of
the medical records that they originally
submitted for medical record review
under RADV audit. Another commenter
requested that CMS reconsider its
position that errors resulting from an
outright failure by an MA organization
to submit a medical record are not
eligible for RADV appeal by the MA
organization.
Response: Both of these
recommendations suggest that CMS
should extend, not have or otherwise
not adhere to a medical record
submission deadline when conducting
RADV audits. It is our position that
establishing realistic medical record
submission deadlines is essential for
conducting RADV audits timely.
Conducting any type of audit activity
absent the establishment of realistic
documentation submission standards
increases the burden and costs
associated with completing the audit
tasks on all parties involved. In fact, in
response to industry concerns that we
were not providing sufficient time for
MA organizations to obtain and submit
the medical records necessary to
validate CMS–HCCs, we earlier
extended the RADV audit medical
record submission window from 3
months to 5 months. We believe 5
months is sufficient time for MA
organizations to locate and submit
medical records necessary to validate an
audited CMS–HCC. Therefore, we
reaffirm that the medical record that an
MA organization selects to support its
appeal of an adverse CMS–HCC
determination must come from records
that the MA organizations submitted to
CMS for audit.
Comment: Several commenters
objected to what they contend is a
burden that RADV audits impose upon
the physicians and physician practices
that must produce medical records
necessary to conduct audits. A provider-
based trade association requests that
MA organizations requesting medical
records for a RADV audit be required to
provide documentation on the scope of
the audit from CMS, as providers
believe there have been abuses in terms
of the amount of requests and data
demands which exceed the actual
requirements. By requiring MA
organizations to provide documentation
of the CMS RADV audit request and the
specific medical records required, this
commenter contended that CMS will
ensure it receives all necessary
documentation, while also ensuring MA
organizations are not using the RADV
audit to unduly burden providers. We
note that outside of the proposed rule,
CMS has also received letters arguing
that the burden associated with RADV
audits is not limited to the CMS’ audits
but also extends to internal audit
activity undertaken by MA
organizations that mimic the RADV
audits that we undertake for Medicare
payment validation. These commenters
raised concerns that MA organizations
were misrepresenting their internal
audit activity as official CMS RADV
audits.
Response: In an effort to minimize the
burden associated with this activity, we
have developed best practices that we
encourage MA organizations to employ
in their efforts to gather medical records
from providers and hospitals. To the
extent MA organizations employ these
practices; it is our belief that the impact
of RADV audits on providers can be
minimized. We also understand the
increasing need for providers to be able
to distinguish when they are being
asked for medical records in association
with an MA organization’s own audit or
in accordance with an official Medicare
program RADV audit which is subject to
statutory requirements. Therefore, we
issue letters on our letterhead that MA
organizations must use when requesting
medical records from providers when
the request is specifically related to an
official CMS RADV audit. Providers
may rely upon these letters as an
indicator that a given medical record
request is for CMS’ RADV audit process,
and providers may request this
authorizing letter before responding to
requests by an MA organization.
f. Proposal To Expand Scope of RADV
Audits
Federal regulations at § 422.311(a)
specify that RADV audits are conducted
by CMS. We proposed to amend this
regulation at § 422.311(a) by specifying
that the Secretary of the Department of
Health and Human Services, along with
CMS, may conduct RADV audits
beginning with the effective date of this
regulation. We also proposed to amend
RADV definitions at § 422.2 to specify
that The Secretary of the Department of
Health and Human Services, along with
CMS, may conduct RADV audits. We
welcomed comment on this proposal.
We received the following comments
and our response follows:
Comment: Many commenters objected
to proposed § 422.311(a) which specifies
that the Secretary, along with CMS,
could conduct RADV audits beginning
with the date when CMS’ proposed
RADV appeals rule change became
effective. Some of these commenters
also objected to CMS’s proposal to
amend RADV definitions at § 422.2 to
specify that the Secretary, along with
CMS, could conduct RADV audits.
Another commenter requested
clarification for the rationale and
mechanics of allowing HHS to conduct
RADV audits, citing concerns about
maintaining consistency in the audit
process.
Response: We conduct RADV audits
to help ensure the integrity of the
Medicare program though activities
aimed at determining whether certain
payments should have been made by
Medicare. The Secretary (including the
Office of Inspector General (OIG)—
pursuant to OIG’s authority under the
Inspector General Act of 1978, 5 U.S.C.
App.) clearly has the authority to
conduct RADV audit activity. Our
proposing this provision and the related
change in definition simply clarifies
what is already an existing statutory
authority. In response to the
commenters requested clarification on
the mechanics of how the Secretary
would conduct RADV audits, we would
note that the Secretary or OIG, will
provide instructions regarding its RADV
audit at the time the Secretary or OIG
notifies selected organizations of
pending RADV audit activity.
g. Proposal To Clarify the RADV
Medical Record Review Determination
Appeal Burden of Proof Standard
Our regulations at § 422.311(c)(3)(iv)
specify that for RADV payment error
calculation appeals, MA organizations
bear the burden of proving that CMS
failed to follow its stated RADV
payment error calculation methodology.
However, RADV regulations do not
specify a burden of proof standard for
the RADV medical record review
determination appeal process. The
absence of a clearly-defined burden of
proof standard for RADV medical record
review determination appeals creates an
appeal environment where MA
organizations, CMS and RADV appellate
officials are free to interpret and apply
different burden of proof standards
when arguing or reviewing appeals
cases. We proposed to amend the rule
with new § 422.311(c)(4) which
specifies that the burden of proof for all
RADV determinations—be they
payment error calculation or RADV
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00092 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29935
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
medical record review determinations—
is on MA organizations to prove, based
on a preponderance of the evidence,
that CMS’s determination was
erroneous.
This approach would stand in
contrast to a burden of proof standard in
which the MA organization were to
prove that a valid diagnoses exists on
the record, and that therefore, the
audited HCC has been validated. This
proposed amendment to the rule
provides the medical record review
determination process a clear burden of
proof standard which more aligns with
the existing RADV payment error
calculation appeals burden of proof
standard. Doing so also improves the
overall RADV appeals procedures by
providing clarity that leads to greater
efficiencies in adjudicating RADV
appeals. We invited comment on this
proposal.
We received the following comments
and our response follows:
Comment: Several commenters
objected to proposed § 422.311(c)(4)
which specifies that the burden of proof
for all RADV determinations—be they
payment error calculation or medical
record review determinations—resides
with the MA organizations, based on a
preponderance of the evidence
standard, that CMS’s RADV audit
determination(s) was erroneous. These
commenters recommended revising the
regulation to place the burden of
supporting an affirmative finding that a
payment error has been made, on CMS.
A commenter also requested that CMS
more clearly define how a
‘‘preponderance of the evidence’’
burden of proof standard would be
applied.
Response: In developing this
proposal, we reviewed other types of
burdens of persuasion, such as the
burden to establish by ‘‘clear and
convincing’’ evidence that a fact exists
or does not exist. First, we based our
decision to propose a preponderance of
the evidence standard on CMS
precedence in other appeals processes.
Second, we determined that it may not
seem fair to the MA organizations to set
a high expectation for persuasion,
especially for those MA organizations
which have not gone through a RADV
appeals process before. We determined
that it would not set as high a standard
as ‘‘clear and convincing’’ or ‘‘beyond a
reasonable doubt’’ for these cases at this
time. Proof that evidence as a whole is
of a degree which is more probable than
not is sufficient to overturn a CMS
determination.
h. Proposal To Change RADV Audit
Compliance Date
Currently, the compliance date for
RADV audits is the due date when MA
organizations selected for RADV audit
must submit medical records to CMS or
its contractors. We proposed to change
the compliance date for meeting RADV
audit requirements for the validation of
risk adjustment data to the due date
when MA organizations selected for
RADV audit must submit medical
records to the Secretary—and not only
to CMS. See proposed regulation
language at § 422.311(b)(2).
We received no comments on this
proposal and therefore are finalizing
this provision without modification.
B. Improving Payment Accuracy
4. Recovery Audit Contractor (RAC)
Determination Appeals (Proposed Part
422 Subpart Z and Part 423 Subpart Z)
a. Background
Section 306 of the Medicare
Prescription Drug, Improvement and
Modernization Act of 2003 (MMA)
required the Secretary to conduct a
demonstration to determine whether
recovery auditors could be used
effectively to identify improper
payments paid under Medicare Part A
and Part B claims. We conducted the
demonstration from March 2005 to
March 2008 in six states. The Recovery
Audit demonstration established
recovery auditors as a successful tool in
the identification and prevention of
improper Medicare payments.
In December 2006, the Tax Relief and
Health Care Act of 2006 (TRHCA) (Pub.
L. 109–432) was enacted. Section 302(a)
of the TRHCA created a permanent
Medicare Recovery Audit Contractor
(RAC) program and added a new
paragraph (h) to section 1893 of the Act
that required us to establish a national
recovery audit program for Medicare
Part A and Part B. The national
Medicare Fee-For-Service (FFS)
Recovery Audit program was
established on January 1, 2010.
Section 6411(b) of the Affordable Care
Act amended section 1893(h)(1) of the
Act by requiring the establishment of
recovery audit programs for Medicare
Parts C and D, in addition to the RAC
program already in place for Medicare A
and B.
On December 27, 2010, we published
a notice in the Federal Register (75 FR
81278) requesting comments on how to
best implement the RAC program for
Parts C and D. Analysis of the comments
received assisted us with
implementation of the Part C and D
RACs.
In January 2011, we entered into a
recovery audit contract for Part D. The
Part D RAC began recouping identified
overpayments in 2012. On December 7,
2012, we published a Request for
Quotation (RFQ) via the General
Services Administration’s (GSA) eBuy
seeking quotations on the
implementation of a Medicare Part C
RAC. We anticipate the award of a Part
C RAC contract in FY 2014.
Given that we began recouping
overpayments determined by the Part D
RAC in 2012, and we anticipate
recouping overpayments in Part C after
awarding a Part C RAC contract in FY
2014, it is appropriate to provide a
codified administrative appeals process
to allow for plans to challenge the
overpayment findings generated by the
RACs just as we provide for challenges
to overpayment determinations
elsewhere in the Medicare program. In
crafting our proposed appeals process
for Parts C and D RAC determinations,
we reviewed existing appeals processes
in other areas, including Parts A and B
RAC determinations, Part C RADV
Audits, Part D payments, etc.
b. Proposed RAC Appeals Process
After reviewing the agency’s existing
appeal processes, we determined that
the general mechanisms set forth in
§ 422.311 and § 423.350 offered the most
appropriate models for the Part C and D
RAC appeals process.
The Part D RAC currently reviews
PDE data to identify overpayments and
underpayments that are paid back to the
plans. When overpayments are
identified, Part D plans are notified and
funds are recovered. If a plan disagrees
with the calculated overpayment
amounts or whether the overpayments
are proper, the plan may appeal the Part
D RAC’s determination directly to the
CMS Center for Program Integrity.
A multilevel independent appeals
process is an important component of
the Part C and Part D RAC program as
it allows plans to appeal determinations
they contend are made in error. The
administrative appeals mechanisms in
this final rule would apply to all Part C
and Part D RAC determinations. As we
implement the Part C RAC, we would
determine if additional changes to the
proposed appeals process are necessary.
Based on the foregoing, we proposed
to add a new subpart Z in Parts 422 and
423, respectively that would include the
proposed provisions discussed in this
section. In accordance with CMS
direction and criteria, the Part C or Part
D RAC would conduct an issue specific
audit of CMS’ payment(s) to plans. An
independent validation of all Part C and
Part D RAC-identified improper
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00093 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29936
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
payments would be conducted. If both
the Part C or Part D RAC and the
independent validation determine that
an improper payment was made, the
Part C or Part D RAC would send a
notice of improper payment to the plan.
If the Part C or Part D RAC determines
an overpayment was made to the plan,
it would send a demand letter
requesting repayment. The demand
letter would: (1) Explain the reason for
the overpayment determination; (2)
explain our recoupment process; and (3)
contain instructions on how the plan
may appeal the Part C or Part D RAC’s
finding. There would be no minimum
monetary threshold for an appeal at any
level.
The following 3-level process sets
forth our proposed administrative
appeals process for overpayment
determinations by the Part C and Part D
RACs. Please note that the appeals
process set forth applies to both
§ 422.2600 and § 423.2600. Because the
sections largely mirror one another,
discussions in this preamble would
apply to both programs, unless
otherwise noted. (1) Reconsiderations
(§ 422.2605 and § 423.2605)
At § 422.2605 and § 423.2605, we
proposed that if the plan believes the
part C or Part D RAC did not apply
CMS’ stated payment methodology
correctly, a plan may appeal the
determination to an independent
reviewer. CMS’ payment methodology
itself, however, is not subject to appeal.
That is, while miscalculations and
factual or data errors may be appealed,
the plan may not appeal the substantive
basis for the overpayment
determination. This is consistent with
the approach to Part D reconciliation
appeals at § 423.350(a)(1), which states
that the Part D plan may appeal ‘‘if CMS
did not apply its stated payment
methodology correctly.’’ The Part D
reconciliation appeals process does not
permit the underlying payment
methodology to be appealed.
Examples of appealable issues would
include, but are not limited to: (1) A
Part C or Part D RAC determination that
a plan provider/pharmacy was excluded
from Medicare when the service was
furnished; (2) a Part C or Part D RAC
determination that a payment was a
duplicate payment; or (3) whether the
Part C or Part D RAC miscalculated an
overpayment.
In paragraph (a), we proposed that the
plan’s request for reconsideration must
be filed with the independent reviewer
within 60 calendar days from the date
of the demand letter. In paragraph (b)(1),
we proposed that the request for
reconsideration must be in writing and
must provide evidence or reasons or
both to substantiate the request. In
paragraph (b)(2), we proposed that the
plan must include with its request all
supporting documentation, evidence,
and substantiation it wants the
independent reviewer to consider. This
material must be submitted in the
format requested by CMS.
Documentation, evidence, or
substantiation submitted after the filing
of the reconsideration request would not
be considered.
In paragraph (c), we proposed that
CMS may file a rebuttal to the plan’s
reconsideration request. The rebuttal
must be submitted to the independent
reviewer within 30 calendar days of the
independent reviewer’s notification to
CMS that it has received the plan’s
reconsideration request. We would
notify and send its rebuttal to the plan
at the same time it is submitted to the
independent reviewer. In paragraph (d),
we proposed that the independent
reviewer would conduct the
reconsideration. Specifically, the
independent reviewer would review the
notification of improper payment, the
evidence, and findings upon which it
was based, and any evidence that the
plan or CMS submitted in accordance
with regulations. In paragraph (e), we
proposed that the independent reviewer
would inform CMS and the plan of its
decision in writing. In paragraph (f), we
proposed that a reconsideration
decision would be final and binding
unless the plan requests a hearing in
accordance with § 422.2605 and
§ 423.2605. Finally, in paragraph (g), we
proposed that a plan that is dissatisfied
with the independent reviewer’s
reconsideration decision would be
entitled to a review by a hearing official
as provided in § 422.2610 and
§ 423.2610.
(2) Hearing Official Determinations
(§ 422.2610 and § 423.2610)
In proposed § 422.2610 and
§ 423.2610, we outline the process for
requesting review of the record by a
CMS hearing official. In paragraph (a),
we proposed that a request for review
must be filed with CMS within 15 days
from the date of the independent
reviewer’s issuance of a determination.
The request must be in writing and must
provide a basis for the request. In
paragraph (b), we proposed that the plan
must submit with its request all
supporting documentation, evidence,
and substantiation that it wants to be
considered. Documentation, evidence,
or substantiation submitted after the
filing of the request would not be
considered.
In paragraph (c), we proposed that a
CMS-designated hearing official would
conduct the review. A hearing would
not be conducted, either live or via
telephone, unless the hearing official, in
his or her sole discretion, chooses such
a mechanism. In all cases, the hearing
official’s review would be limited to
information that: (1) The Part C or Part
D RAC used in making its
determinations; (2) the independent
reviewer used in making its
determinations; (3) the plan submits
with its hearing request; and (4) CMS
submits per paragraph (d). Neither the
plan nor CMS would be allowed to
submit new evidence.
In paragraph (d), we proposed that
CMS may file a rebuttal to the plan’s
hearing request. The rebuttal must be
submitted within 30 calendar days of
the plan’s submission of its hearing
request. CMS would send its rebuttal to
the plan at the same time it is submitted
to the hearing official. In paragraph (e),
we proposed that the CMS hearing
official would decide the case within 60
days and send a written decision to the
plan and CMS, explaining the basis for
the decision. In paragraph (f), we
proposed that the hearing official’s
decision would be final and binding,
unless the decision was reversed or
modified by the CMS Administrator in
accordance with § 422.2615 and
§ 423.2615.
(3) Administrator Review (§ 422.2615
and § 423.2615)
In proposed § 422.2615 and
§ 423.2615, we discuss the
Administrator review process. In
paragraph (a), we proposed that if a plan
is dissatisfied with the hearing official’s
decision, the plan may request that the
CMS Administrator review the decision.
The request must be filed with the CMS
Administrator within 15 calendar days
of the date of the hearing official’s
decision. The request must provide
evidence or reasons or both to
substantiate the request. In paragraph
(b), we proposed that the plan must
submit with its request all supporting
documentation, evidence, and
substantiation that it wants to be
considered. Neither the plan nor CMS
would be allowed to submit new
evidence. Documentation, evidence or
substantiation submitted after the filing
of the request would not be considered.
In paragraph (c), we proposed that
after receiving a request for review, the
Administrator would have the
discretion to review the hearing
official’s decision in accordance with
paragraph (e) or to decline to review
said decision.
In paragraph (d), we proposed that the
Administrator would notify the plan of
whether he or she intends to review the
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00094 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29937
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
hearing official’s decision. If the
Administrator declines to review the
hearing official’s decision, the hearing
official’s decision is final and binding.
If the Administrator agrees to review the
hearing official’s decision, CMS may file
a rebuttal statement within 30 days of
the Administrator’s notice to the plan
that the request for review has been
accepted. CMS would send its rebuttal
statement to the plan at the same time
it is submitted to the Administrator. In
paragraph (e), we proposed that if the
Administrator agrees to review the
hearing official’s decision, the
Administrator would determine, based
upon this decision, the hearing official
record, and any arguments submitted by
the plan or CMS in accordance with this
section, whether the determination
should be upheld, reversed, or
modified. The Administrator would
furnish a written decision to the plan
and to CMS. The Administrator’s
decision would be final and binding.
We received the following comments
and our responses follow:
Comment: Several commenters
expressed concern over the proposed
15-day timeframe for plan sponsors to
request review by a Hearing Official and
also the proposed 15-day timeframe to
request review by the Administrator.
Commenters believe that a 15-day
timeframe for requesting additional
review may result in unnecessary
appeals and that 30 days is a more
appropriate timeframe for plan sponsors
to evaluate if additional appeals for
review are appropriate. Commenters
pointed out that a 30-day timeframe is
typical among other similar CMS
appeals processes.
Response: We agree with commenters
that a 15-day timeframe for requesting
additional review by a Hearing Official
or the Administrator may not provide
enough time for plan sponsors to make
an appropriate decision regarding
additional appeals for review and we
are finalizing this rule with a 30-day
timeframe for such requests. This
timeframe will also make the Parts C
and D RAC Appeals process more
structurally similar to existing appeals
processes such as the RADV Appeals
process.
Comment: A commenter requested
that CMS clarify the distinction between
‘‘payment methodology’’ and ‘‘findings
of the applied methodology’’ given that
CMS proposed that ‘‘payment
methodology’’ is not subject to appeal.
This commenter believes that this
distinction is critical to providing
meaningful appeal rights to plan
sponsors. The commenter provided an
example such as when the RAC
determines that a payment received by
the Part D sponsor should have been
treated as Direct and Indirect
Remuneration (DIR).
Response: We agree with the
commenter that this distinction is
critical to providing a meaningful
appeals process to plan sponsors. In the
proposed rule, we indicated that
miscalculations and factual or data
errors may be appealed as ‘‘findings of
the applied methodology’’. If a plan
sponsor believes that a Part D RAC
incorrectly classified a payment as DIR,
for example, this would be a question of
fact regarding the findings of the
applied methodology that the plan
sponsor is entitled to appeal.
Comment: A commenter questioned
why new evidence could not be
submitted at subsequent levels of appeal
after the first level reconsideration and
requested that CMS allow new evidence
to be submitted at each level of appeal.
Response: We disagree with the
commenter. We do not believe it is
common for evidence relevant to a RAC
determination to be unavailable to a
plan sponsor 60 days after a Notice of
Improper Payment is received by the
plan sponsor. This is the relevant
timeframe for requesting a
reconsideration and submitting relevant
evidence and documentation to the
independent reviewer. Also, we do not
believe it is generally appropriate for
plan sponsors to withhold relevant
evidence from the independent reviewer
at the Reconsideration stage of appeal
and we want to safeguard the program
from this type of activity. We have
modeled our proposed process after
existing CMS appeals processes that do
not allow the submission of new
evidence at higher levels of appeal, such
as the CMS RADV appeals process. We
also note that in addition to the plan
sponsor not being permitted to submit
new evidence at subsequent levels of
appeal, we are also precluded from
submitting new evidence at subsequent
levels of appeal.
Comment: A commenter questioned
why CMS did not define ‘‘designated
independent reviewer’’ and suggested
that in order to ensure that the first level
appeals reviewer is both qualified and
independent of the RAC, the regulation
should specify the necessary
qualifications for this position. The
commenter further suggested that the
regulation contain a specific conflict of
interest provision that would disallow
any financial or other relationship
between the RAC and the independent
reviewer.
Response: We agree with the
commenter that the integrity of the
proposed appeals process is imperative
and that the designated independent
reviewer be both qualified and
independent of the RAC. We decline to
specify the necessary qualifications for
this position in the regulation and we
decline to add a specific conflict of
interest provision in the regulation. We
believe that the independence of the
reviewer will be self-evident as the
reviewer will not be affiliated with the
RAC and we have no incentive to select
independent reviewers who are lacking
the qualifications to fulfill this task.
Comment: A commenter requested
that CMS make clear that the Part D
sponsor is not required to make any
payment with respect to a RAC finding
until the sponsor has exhausted the
administrative appeals process. The
commenter also requested that CMS
clarify that any final and binding
decision by the Administrator does not
preclude judicial review.
Response: We agree that final Part D
payment adjustments based on RAC
findings will not be made until all
administrative appeal rights are
exhausted. This is our current practice
under the existing appeals process and
will continue to be the practice under
the formal three-level appeals process
being implemented in this final rule. We
also agree with the commenter that any
final and binding decision by the
Administrator under this rule does not
preclude judicial review.
After review of the public comments
received on these proposals, we are
finalizing our proposals with one
modification. In §§ 422.2610(a) and
422.2615(a) and § 423.2610(a) and
§ 423.2615(a), we are revising the
timeframe for MA organizations and
Part D plan sponsors, respectively, to
request review by a Hearing Official or
the Administrator from 15 days to 30
days.
C. Implementing Other Technical
Changes
1. Definition of a Part D Drug (§ 423.100)
Section 1860D–2(e) of the Act defines
a covered Part D drug as a drug that may
be dispensed only upon a prescription
and that is described in paragraph (A)(i),
(A)(ii), or (A)(iii) of section 1927(k)(2) of
the Act; or a biological product
described in clauses (i) through (iii) of
paragraph (B) of such section, or insulin
described in paragraph (C) of such
section and medical supplies associated
with the injection of insulin (as defined
in regulations of the Secretary), and
such term includes a vaccine licensed
under section 351 of the Public Health
Service Act (and, for vaccinations
administered on or after January 1,
2008, its administration), and any use of
a covered Part D drug for a medically
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00095 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29938
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
accepted indication (as defined in
paragraph (4)). We codified this
definition in § 423.100.
a. Combination Products
The FDA approves and regulates
many products that include drug-drug
and drug-device combinations.
However, for the purposes of the Part D
program, only combination products
approved and regulated by the FDA as
drugs, vaccines, or biologics are
potentially eligible for Part D coverage,
in line with the Part D drug definition.
We proposed to address this issue in
regulation to codify and clarify policy
we previously addressed through
guidance.
We proposed to add paragraph (vii)
under the definition of a Part D drug to
further clarify that only those
combination products approved and
regulated in their combination form by
the FDA as a drug, vaccine, insulin, or
biologic, as described in paragraph (i),
(ii), (iii), or (v) of the Part D drug
definition, may be eligible for Part D
coverage. Our proposal would make it
clear that the definition of a Part D drug
excludes products where a combination
of items are bundled or packaged
together for convenience (such as one
box packaging together multiple
products, each in separate bottles),
where the bundle has not been
evaluated and approved by the FDA.
This proposal would not affect products
where multiple active ingredients
(including at least one Part D eligible
prescription-only ingredient) are
incorporated into a single pill or single
injection, as such products would have
had to go through FDA approval in this
combined form, meeting the Part D
requirement. Combination products that
are FDA approved would then be
treated like other Part D drugs, eligible
for coverage only when being used for
a medically accepted indication and not
otherwise excluded from Part D
coverage (for example, because it is
covered as prescribed and dispensed or
administered under Medicare Part B).
This proposed policy is intended to
clarify that a combination product
containing at least one constituent
ingredient that would, if dispensed
separately, meet the definition of a Part
D drug is eligible for Part D coverage
only if it has received FDA approval in
its combined form. Combination
products not FDA approved as drugs
under the Federal Food, Drug, and
Cosmetics Act would not satisfy section
1927(k)(2)(A)(i) of the Act, defining
covered outpatient drugs as those
approved for safety and effectiveness as
a prescription drug. Combination
vaccines not licensed as a vaccine under
section 351 of the Public Health Service
Act similarly would not satisfy the
definition of a Part D drug as defined in
section 1860D–2(e)(1) of the Act.
Our proposal would not require that
all constituent ingredients of a
combination product be FDA-approved
prescription drugs. An example would
be an FDA-approved prescription drug
that combines a Part D drug with a non-
Part D covered vitamin. Conversely, a
product combining a Part D drug with
a medical food, dietary supplement, or
another Part D drug, where the
combined product has not received FDA
approval as a prescription drug, vaccine,
or biologic would not be eligible for Part
D coverage.
Comment: A commenter noted it
supported the proposed policy
regarding combination products.
Response: We appreciate the support
for our proposal.
Comment: A commenter requested
that CMS provide clarification on what
constitutes a vitamin versus what
constitutes a dietary supplement.
Response: In the preamble, we
provided an example of a Part D drug
combined with a vitamin that would be
eligible for coverage, if FDA approved in
the combined form. We also provided
an example of a Part D drug combined
with a dietary supplement that would
not be eligible for coverage because the
FDA had not approved that
combination. We did not mean to imply
that only approved combinations
involving vitamins would be eligible,
nor did we mean to distinguish between
vitamins as opposed to dietary
supplements in that paragraph. Our
intent was to distinguish their eligibility
for coverage by the fact that one of these
combined products was approved as a
combination drug product by the FDA
and the other was not.
After consideration of the public
comments we received, we are
finalizing this provision with a
technical modification to improve the
clarity of the provision.
b. Barbiturates and Benzodiazepines
We also proposed to amend the
definition of a Part D drug to address
certain exclusions by revising paragraph
(2)(ii). When the Part D benefit started
in 2006, all uses of barbiturates and
benzodiazepines were excluded from
coverage by statute. In 2008, section 175
of the MIPPA amended section 1860D–
2(e)(2)(A) of the Act to include coverage
for barbiturates when used in the
treatment of epilepsy, cancer, or a
chronic mental health disorder and for
benzodiazepines when used for any
medically accepted indication, effective
January 1, 2013. In 2010, section 2502
of the Affordable Care Act amended
section 1927(d) of the Act, to remove
barbiturates and benzodiazepines from
the list of drugs subject to exclusion
from coverage, effective for services
provided on or after January 1, 2014.
Thus, this subsequent statutory change
effectively includes barbiturates as a
Part D drug for all medically accepted
indications. The proposed revision to
§ 423.100 would conform our definition
of Part D drug to the new statutory
requirement by removing from
paragraph (2)(ii) the clause ‘‘barbiturates
when used to treat epilepsy, cancer, or
a chronic mental health disorder; and
benzodiazepines.’’
We did not receive any comments
regarding this proposal.
We note that an error appeared in the
corresponding regulations text of the
January 10, 2014 proposed rule. In the
regulations text (79 FR 2062), we made
a typographical error in an amendatory
instruction and inadvertently did not
remove the previously noted clause
from the definition of ‘‘Part D drug’’ at
§ 423.100(2)(ii). Therefore, we are
making the required corrections in the
regulations text of this final rule.
c. Medical Foods
We proposed to add paragraph (2)(iii)
to the list of exclusions from the
definition of Part D drug to specify that
medical foods, as defined in 21 U.S.C.
360ee, are not Part D drugs. Medical
foods are not described in paragraphs
A(i), A(ii) or A(iii) of section 1927(k)(2)
of the Act, and therefore, do not meet
the statutory definition of a covered Part
D drug, nor do they fall under other
categories eligible for Part D coverage
listed in the Part D drug definition, such
as biologics, vaccines, and insulin.
Moreover, as described previously in
the section on combination products, a
product with relevant components
including some or all ingredients
meeting the definition of a Part D drug
would not be eligible for Part D coverage
unless the combined product has also
been approved by the FDA as a drug,
vaccine, or biologic.
The proposed clarifications involving
coverage for approved combination
products and non-coverage of medical
foods would not affect current policies
surrounding Part D coverage of
parenteral nutrition. (See the Part D
manual guidance, Chapter 30.7
regarding the payment for parenteral
and enteral nutrition items and
services.) Extemporaneously
compounded prescription drug products
(addressed separately in Chapter 6 of
the Part D manual and in § 423.120) also
would not be affected by the proposed
changes. Part D coverage for
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00096 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29939
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
extemporaneously compounded
prescriptions is available for the
ingredients that independently meet the
definition of a Part D drug when the
product needed is one requested by the
provider to meet a specific medical
need, where there is no commercially
available alternative. The convenience
packaging of unapproved combination
products for broad distribution does not
meet the criteria set out specifically for
extemporaneously compounded
prescriptions.
Comment: A commenter that strongly
disagreed with the proposal stated that
there are medically indicated nutritional
supplements such as food thickeners,
caloric supplements, and probiotics
which should be covered if prescribed
by a physician.
Response: The definition of a
‘‘covered Part D drug’’ found in section
1860D–2(e)(1) of the Act does not allow
us to cover food thickeners, caloric
supplements, and probiotics even if
prescribed by a prescription. These
items do not meet any of the
requirements of that section.
After consideration of the public
comment we received, we are finalizing
this provision without modification.
2. Special Part D Access Rules During
Disasters or Emergencies (§ 423.126)
Section 1860D–4(b) of the Act
requires us to ensure beneficiaries have
access to covered Part D drugs. When a
disaster strikes or is imminent,
beneficiaries may find they have trouble
accessing drugs through normal
channels or must move to safer
locations far away from their regular
pharmacies. In order to ensure that
beneficiaries do not run out of their
medications during or as a result of a
disaster or emergency, we issued
guidance on December 18, 2009,
identifying when, in the course of a
disaster, Part D sponsors would be
expected to relax ‘‘refill-too-soon’’ (RTS)
edits. We proposed to codify a revised
version of that policy. Proposed
§ 423.126(a)(1)(i) would require Part D
sponsors to relax RTS edits in the event
of any imminent or occurring disaster or
emergency that would hinder an
enrollee’s access to covered Part D
drugs. By this we mean that there is an
anticipated or actual disaster or
emergency, as evidenced by a
declaration of a disaster or emergency
issued by an appropriate federal, state or
local official, and it is reasonable to
conclude that such disaster or
emergency or preparation therefore
would make it difficult for beneficiaries
to obtain refills of their medications
because the disaster or emergency or
anticipation thereof has affected, or will
affect, their ability to have timely access
to their usual pharmacies. For example,
if federal, state or local authorities issue
mandatory evacuation orders to
populations or segments of the
population in a geographic area, it
would be reasonable to conclude that
the evacuation would hinder an LTC
resident’s ability to get a refill after he
or she is evacuated from the facility. In
such an instance, then, Part D sponsors
with enrollees in the affected area
would be required to relax RTS edits so
that the LTC pharmacies could provide
beneficiaries with refills to take with
them to the location to which they are
being evacuated.
Our proposed requirement would
apply to one refill for each drug the
beneficiary is taking for refills sought
within 30 days of the date the plan
sponsor began relaxing RTS edits. We
believe this timeframe would be
sufficient to ensure that beneficiaries
who are unable to obtain refills during
the emergency or disaster will be able to
do so as soon as they can safely access
a network pharmacy. We solicited
comment as to whether 30 days after the
date of the triggering declaration
provides an appropriate amount of time
to ensure that beneficiaries do not run
out of their medications. In particular,
we would be interested in learning
about any situations in which a
beneficiary affected by an actual or
impending disaster or emergency would
be likely to go to a pharmacy more than
30 days after the triggering declaration
such that the resumption of RTS edits
after 30 days would be problematic. We
also solicited comment as to how it
would be feasible for Part D sponsors to
identify pharmacies or beneficiaries
located in affected areas for which they
would be required to relax edits and,
how long it might then take to program
the necessary changes.
Although we believed our proposal
provides a general framework for when
RTS edits must be relaxed, we solicited
comment on whether we should impose
more particular requirements in cases
where a disaster or emergency could
result in a voluntary or mandatory
evacuation of an LTC facility. We are
also concerned that if a disaster strikes
the area in which an LTC facility is
located but not the area in which its
servicing LTC pharmacy is located, the
appropriate edits may not be relaxed.
Accordingly, we solicited comment as
to whether it would be more feasible to
establish beneficiary specific edits
limited to residents of LTC facilities in
affected areas given that evacuation
decision-making is rarely a
straightforward, linear process (for
example, not just based on the
declaration of a disaster or emergency),
but rather, often involves myriad
facility-specific factors. In particular, we
solicited comment on the practicality of
requiring Part D sponsors to relax RTS
edits for residents of a particular LTC
facility after that facility decides on its
own initiative to evacuate through use
of National Council on Prescription
Drug Programs (NCPDP) Submission
Clarification Code (SCC) code 13, which
conveys that there is an emergency. We
solicited comment as to whether use of
this code number, 13, is specific enough
to signal that sponsors need to loosen
RTS edits and whether it would be
practical for LTC facilities to request
that their LTC pharmacies enter the SCC
code 13. Lastly, we stated we would be
interested in any other ideas on how to
structure workable edits or institute
manual procedures to best target only
enrollees who live in LTC facilities
located in areas affected by a disaster.
We also stated that we would be
interested in hearing from any
commenters who would recommend
any other triggering events that would
require Part D sponsors to relax RTS
edits. In particular, we solicited
comment as to whether it would be
feasible to require sponsors to relax
edits after the issuance by the National
Weather Service (NWS) of a Hurricane
or Tropical Storm watch or warning.
The NWS typically issues watches 36
hours in advance of adverse weather
conditions possibly hitting an area,
while the NWS issues watches 48 hours
(2 days) in advance of those conditions
possibly hitting an area. All watches/
warnings are posted on the NWS Web
site immediately after their issuance.
We solicited comment as to whether
watch/warnings would require RTS
overrides in the whole state, or just
areas under the watch or warning. We
also stated that we were interested in
comments regarding the time generally
needed to move residents of LTC
facilities with their medication supplies
to safety.
Lastly, we believe that sponsors are in
the best position to determine how to
relax the specific RTS edits when
required under our proposal. However,
we also wish to ensure that all sponsors
relax RTS edits in a consistent manner
in order that enrollees have the same
critical access to drugs when disasters
and emergencies are imminent or have
occurred—regardless of the specific
plan in which they are enrolled.
Accordingly, we solicited comments on
the types of situations that might arise
and the extent to which sponsors should
be allowed to exercise some discretion
in complying with this proposed
requirement.
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00097 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29940
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
And, as has been the case under our
current guidance, Part D sponsors may
consider extending the implementation
of the RTS edits but are not required to
do so. However, if sponsors choose to
reinstate the RTS edits, they need to
work closely with enrollees who
indicate that they are still displaced or
otherwise impacted by the disaster or
emergency.
Comment: Several commenters
supported CMS’s proposal. A
commenter commended CMS’s efforts to
ensuring access to critical and other
drugs during times of crises.
Response: We thank the commenters
for the support.
Comment: Several commenters were
concerned that the policy was not clear
enough to ensure that Part D sponsors
would apply it consistently. A
commenter suggested that requiring
sponsors to ‘‘reasonably conclude’’
whether a beneficiary would have
difficulty obtaining refills would result
in an inconsistent relaxation of edits
and suggested instead that CMS provide
clear direction by exercising its section
1135 waiver authority. Another
commenter requested that CMS issue
HPMS alerts to advise Part D sponsors
on when to relax edits every time a
trigger event occurred not only because
of the subjective nature of the sponsor
assessment but because it also depended
on whether a sponsor knew that about
a declaration. A commenter requested
that we make it clear sponsors would
only be obligated to relax edits when
operationally possible.
Response: We appreciate the
suggestions and will consider them in
the future.
Comment: A commenter suggested
that CMS allow beneficiaries enrolled in
mail order pharmacy programs to use
local retail or hospital pharmacies
during emergencies when disasters or
other emergencies interfere with their
receipt of drugs through the mail.
Response: While we appreciate the
concerns, we did not propose any
changes with respect to mail order
during disasters and emergencies and
we are not adopting this
recommendation at this time.
Comment: Several commenters
responded to our request for comment
by agreeing that it was appropriate to
limit the window for relaxed edits to 30
days after the date of declaration.
Another commenter suggested that the
30 day period should start running
when the emergency actually occurred
because declarations often do not take
place until later—in which case the
proposed timeframe might actually
exceed 30 days.
Response: We appreciate the
comments, and will use them to inform
possible future rulemaking.
Comment: Several commenters
believed use of the NCPDP submission
code appropriate, while another
concluded it would not work because it
was beneficiary specific and not specific
to LTC facilities. A commenter stated it
was difficult to operationalize RTS
overrides by areas and that it was
typically done by state. Comments on
the feasibility of relying on NWS
watches and warnings ranged from
several commenters who thought it
inappropriate to ever relax edits on
account of such warnings because they
might never occur, to a commenter who
thought it appropriate to limit such
application solely to hurricane and
tropical storm warnings, to another
commenter who thought both types of
warnings appropriate triggers and
suggested that CMS also rely on
advisories from NWS. However, not all
commenters discussed warnings and
watches in the context of LTC facilities
and, in fact, a commenter questioned
whether our proposal even applied to
non-LTC situations.
Response: We appreciate the
comments and will use them to inform
possible future rulemaking.
Comment: A few commenters
requested that we broaden our policy by
allowing sponsors to relax edits more
often than proposed. A commenter
suggested we allow sponsors make
determinations regarding whether to
relax edits ‘‘well before’’ declarations
were issued rather than wait for their
issuance, and other commenters
identified specific situations that they
felt should prompt such a determination
such as local challenges and severe
weather (such as tornadoes) and
accompanying difficulties (such as
power outages extending for multiple
days). Another commenter requested
that CMS automatically grant special
access rules when a state of emergency
is declared in a state or region thereof
rather than leave the discretion to apply
those rules to sponsors.
In contrast, several commenters
requested that we revise the regulation
so that sponsors would be able to relax
edits less often than proposed.
Observing that many anticipated snow
storms that did not actually take place
last winter, a commenter requested that
CMS not allow Part D sponsors to relax
edits for government declarations that
merely announced the possibility, rather
than the occurrence, of disasters or
emergencies. Another commenter
suggested that we limit application of
the policy to declarations only from
federal and state authorities because it
was difficult for Part D sponsors with
large service areas to track declarations
by local authorities.
Another commenter recommended
that we retain the current guidance.
Response: As a result of the comments
we received on this issue, we are not
finalizing this proposal. We have
concluded that we need to carefully
consider our options and consequently
have decided to leave in place current
guidance. There was simply not a
consensus regarding any aspect of the
proposed regulation to sufficiently
inform a decision to finalize. For
instance, a number of commenters
expressed opposing views: Some
requested that we broaden our policy by
allowing sponsors to relax edits more
often than proposed, while others
suggested that we curtail the
circumstances under which sponsors
would be permitted to relax edits. Some
contractors liked the discretionary
aspects of the proposal and the existing
guidance while others sought bright line
indicators—although sometimes just to
trigger the times when discretion might
be applied. Several commenters appear
to have misunderstood our proposal.
We believe it is important to ensure
that beneficiaries receive drugs in the
event of disasters or anticipated
disasters that might hinder their access
to such drugs for a period of time. But
we are concerned that if sponsors do not
uniformly relax edits under similar
circumstances, beneficiaries in different
plans will be treated disparately. We
hope to prevent situations in which, for
instance, two beneficiaries living in the
same area are affected by the same
disaster, but one beneficiary is able refill
a prescription that otherwise would
been subject by RTS edits, while the
other, who is enrolled in a different
plan, is not. The variety of comments
and responses suggests that resolving
these issues may require more focused
inquiry. In the meantime, the current
guidance will remain in place (found in
Prescription Drug Benefit Manual,
Chapter 5, Benefits and Beneficiary
Protections, Section 50.12). We again
thank all the commenters including
those that took the time to respond to
our specific solicitations. We will keep
their suggestions in mind as we
carefully consider our options for the
future, including whether to address our
regulatory proposals in future
rulemaking.
3. Termination of a Contract Under Parts
C and D (§§ 422.510 and 423.509)
a. Cross-Reference Change (§ 423.509(d))
Section 1857(h)(1)(B) and 1860D–
12(b)(3)(F) of the Act describes the
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00098 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29941
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
2
We note that although the preamble accurately
reflected this proposal, the regulation text for
§ 423.756(a)(2), (79 FR 2070), erroneously did not
reflect the proposed grammatical correction.
3
In the preamble to our proposal, we mistakenly
referred to the language as being deleted by using
‘‘and’’ instead of ‘‘through’’.
procedures for termination for both Part
C and Part D plan sponsors respectively.
We codified organizations’ appeal rights
under subpart N of parts 422 and 423.
Under the Part C § 422.510(d), a
reference to the appeal rights ‘‘in
accordance with subpart N’’ is made.
However, in the corresponding section
for Part D Plan sponsors at § 423.509(d),
the reference to the appeal rights reads
‘‘in accordance with § 423.642.’’ The
Part C and Part D references should be
the same.
We proposed to align the Part C and
Part D appeal rights language under
§§ 422.510(d) and 423.509(d) by
replacing the inconsistent language at
§ 423.509(d) to now read ‘‘in accordance
with subpart N of this part.’’
b. Terminology Changes (§§ 422.510 and
423.509)
Sections 1857(c) and 1860D–
12(b)(3)(B) of the Act authorize CMS to
terminate contracts with MA
organizations and Part D plan sponsors
respectively. In the current termination
regulations at §§ 422.510 and 423.509,
there is inconsistent use of the terms
‘‘days’’ and ‘‘calendar days’’. Therefore,
we proposed to replace the word ‘‘days’’
with ‘‘calendar days’’ in both §§ 422.510
and 423.509.
c. Technical Change To Align Paragraph
Headings (§ 422.510(b)(2))
Sections 1857(c)(2) and 1860D–
12(b)(3)(B) of the Act provide CMS with
the authority to terminate contracts, for
Part C and Part D sponsors respectively.
The Part C paragraph heading at
§ 422.510(b)(2) incorrectly reads
‘‘Expedited termination of contract by
CMS.’’ Therefore, we proposed to revise
the paragraph heading of § 422.510(b)(2)
to read ‘‘Immediate termination of
contract by CMS’’. This change will also
make it consistent with the
corresponding heading for Part D, in
§ 423.509(b)(2).
d. Terminology Change
(§ 423.509(b)(2)(C)(ii))
Sections 1857(c)(2) and 1860D–
12(d)(3)(B) of the Act provide CMS with
the authority to terminate contracts, for
Part C and Part D sponsors respectively.
In § 423.509(b)(2)(C)(ii) the regulation
incorrectly references ‘‘MA
organization.’’ This section concerns
Part D, so the correct reference is ‘‘Part
D Plan Sponsor’’. Therefore, we
proposed to change § 423.509(b)(2)(C)(ii)
to appropriately reference Part D plan
sponsor; not MA organization, as it
currently states.
We received no comments on these
proposals and are therefore finalizing
these provisions without modification.
4. Technical Changes Regarding
Intermediate Sanctions and Civil Money
Penalties
Sections 1857(g) and 1860D–
12(b)(3)(E) of the Act provide us with
the authority to impose intermediate
sanctions (sanctions) and CMPs on Part
C and Part D sponsors, respectively.
a. Technical Changes to Intermediate
Sanctions Notice Receipt Provisions
(§§ 422.756(a)(2) and 423.756(a)(2))
Under §§ 422.756(a)(2) and
423.756(a)(2) the current language states
that written requests for rebuttal by the
MA organization or Part D plan sponsor
must be received within ‘‘10 calendar
days from the receipt of notice’’. The
language in other sections of this
subpart refers to receipt of a notice as
‘‘days after receipt of this notice.’’ All
sections should be consistent.
Therefore, we proposed to modify the
language at §§ 422.756(a)(2) and
423.756(a)(2) to state ‘‘10 calendar days
after receipt of the notice’’. In addition,
we proposed to correct grammatical
errors in current §§ 422.756(a)(2) and
423.756(a)(2) by revising the language in
both §§ 422.756(a)(2) and 423.756(a)(2)
2
to add the word ‘‘the’’ before notice; as
proposed, the second sentence in each
paragraph (a)(2) would read ‘‘CMS
considers receipt of the notice as the
day after the notice is sent by fax, email,
or submitted for overnight mail.’’
b. Cross-Reference Changes
(§ 422.756(b)(4) and § 423.756(b)(4))
Under § 422.756(b)(4) and
§ 423.756(b)(4), we reference the
procedures MA organizations and Part D
plan sponsors must follow for
requesting a hearing to appeal the
imposition of intermediate sanctions
and civil money penalties. MA
organizations and Part D sponsors must
adhere to hearing procedures
promulgated within subpart N of the
regulations, not just §§ 422.660 through
422.684 and §§ 423.650 through
423.662, respectively, as currently cited
in §§ 422.756(b)(4) and 423.756(b)(4).
Therefore, we proposed to modify the
language at §§ 422.756(b)(4) and
423.756(b)(4) so that it would read that
MA organizations and Part D sponsors
‘‘must follow the right to a hearing
procedures as specified in subpart N’’.
c. Technical Changes (§§ 422.756(d) and
423.756(d))
In §§ 422.756(d) and 423.756(d) we
provide alternatives to sanctions,
including non-renewal or termination of
the organizations contract. However, the
paragraph heading of both §§ 422.756(d)
and 423.756(d) only refers to
terminations by CMS. Therefore, we
proposed to revise the paragraph
heading to ‘‘Non-renewal or termination
by CMS’’ in both sections to reflect the
content specified within the provision.
Within §§ 422.756(d) and 423.756(d),
we state that we may decline to
authorize the renewal of an
organization’s contract in accordance
with § 422.506(b)(2) and (b)(3) for MA
organizations and in accordance with
§ 423.507(b)(2) and (b)(3) for Part D plan
sponsors. However, all of paragraph (b)
in §§ 422.506 and 423.507 applies to
§§ 422.756(d) and 423.756(d),
respectively. Therefore, we proposed to
change both provisions §§ 422.756(d)
and 423.756(d) to read ‘‘§ 422.506(b)’’
and ‘‘§ 423.507(b)’’, respectively.
3
Within §§ 422.756(d) and423.756(d),
we refer to the ‘‘sanctions described in
paragraph (c)’’ but in each section,
paragraph (c) refers to the effective date
and duration of sanctions, rather than
sanctions which are actually described
in §§ 422.750 and 423.750, respectively.
Therefore, we proposed to change the
current language at § 422.756(d) to read
‘‘In addition to or as an alternative to
the sanctions described in § 422.750
. . .’’ and change the language at
§ 423.756(d) to read ‘‘In addition to or
as an alternative to the sanctions
described in § 423.750.’’ to correct this
mistake.
d. Technical Changes To Align the Civil
Money Penalty Provision With the
Authorizing Statute (§§ 422.760(a)(3)
and 423.760(a)(3))
The provisions at §§ 422.760(a)(3) and
423.760(a)(3) state, ‘‘the harm which
resulted or could have resulted from
conduct of an MA organization’’ and
‘‘the harm which resulted or could have
resulted from conduct of a Part D plan
sponsor’’, respectively. However, this
language is not consistent with the
authorizing statutory provisions, nor is
it consistent with other provisions in
corresponding sections.
Therefore, we proposed to align the
language with that used in paragraphs
(b)(1) and (2) from that same section in
both §§ 422.760(a)(3) and 423.760(a)(3).
The language would be revised to state
‘‘The adverse effect to enrollees which
resulted or could have resulted . . .’’ in
both §§ 422.760(a)(3) and 423.760(a)(3)
to track the statutory language.
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00099 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29942
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
e. Technical Changes To Align the Civil
Money Penalty Hearing Notice Receipt
Provisions (§§ 422.1020(a)(2),
423.1020(a)(2), 422.1016(b)(1), and
423.1016(b)(1))
Sections 1857(g)(4) and 1860D–
12(b)(3)(E) of the Act provides us with
the authority to impose civil money
penalties on MA organizations and Part
D plan sponsors, respectively. Under
§§ 422.1020(a)(2) and 423.1020(a)(2), we
discuss our procedures for requesting an
appeal of a CMP. The current language
in both sections state written requests
for appeal ‘‘must be filed within 60
calendar days from the receipt of notice
of initial determination.’’ However, this
language does not align with the appeal
language in subpart N for requesting a
hearing.
Therefore, we proposed to change the
language at §§ 422.1020(a)(2) and
§ 423.1020(a)(2) to align it with the
language within subpart N for appeals.
Specifically, we proposed to change the
language in both §§ 422.1020(a)(2) and
423.1020(a)(2) to read ‘‘after receipt’’
instead of ‘‘from the receipt’’, so it reads
‘‘within 60 calendar days after receipt of
the notice of initial determination’’.
In addition, under §§ 422.1016 and
423.1016, we furnish our procedures for
filing briefs with the Administrative
Law Judge or Departmental Appeals
Board, and opportunity for rebuttal. The
provisions at §§ 422.1016(b)(1) and
423.1016(b)(1) state, ‘‘the other party
will have 20 days from the date of
mailing or personal service to submit
any rebuttal statement or additional
evidence’’. However, this language is
not consistent with provisions in other
corresponding sections. Therefore, we
proposed to revise the language at
§§ 422.1016(b)(1) and 423.1016(b)(1) to
state ‘‘The other party will have 20 days
from the date of mailing or in person
filing . . .’’ to maintain consistency.
We received no comments on these
proposals and therefore are finalizing
these provisions without modification.
IV. Collection of Information
Requirements
Under the Paperwork Reduction Act
of 1995, we are required to provide 30-
day notice in the Federal Register and
solicit public comment before a
collection of information requirement is
submitted to the Office of Management
and Budget (OMB) for review and
approval. In order to fairly evaluate
whether an information collection
should be approved by OMB, section
3506(c)(2)(A) of the Paperwork
Reduction Act of 1995 requires that we
solicit comment on the following issues:
• The need for the information
collection and its usefulness in carrying
out the proper functions of our agency.
• The accuracy of our estimate of the
information collection burden.
• The quality, utility, and clarity of
the information to be collected.
• Recommendations to minimize the
information collection burden on the
affected public, including automated
collection techniques.
We solicited public comment on each
of these issues for the following sections
of this document that contain
information collection requirements
(ICRs).
A. ICRs Related to Improper Prescribing
Practices and Patterns
Our additions of §§ 424.530(a)(11),
424.535(a)(13), and 424.535(a)(14) will
likely result in an increase in denials,
revocations, and associated appeals.
However, we are unable to estimate the
number of denials, revocations, and
appeals. We do not have data available
that can be used to make such
projections, as each situation would
have to be carefully reviewed and
addressed on a case-by-case basis.
Therefore, we cannot estimate the
potential concomitant increase in the
ICR burden, though, as we stated in the
proposed rule, we believe any such
increase will be minimal.
We received no comments on the
potential ICR burden of
§§ 424.530(a)(11), 424.535(a)(13), and
424.535(a)(14).
B. ICRs Related to Applicants or Their
Contracted First Tier, Downstream, or
Related Entities To Have Experience in
the Part D Program Providing Key Part
D Functions (§ 423.504(b)(8)(i) Through
(iii))
Proposed § 423.504(b)(8)(i) through
(iii) would require that Part D
organizations seeking a new Medicare
contract must have arrangements in
place such that either the applicant or
a contracted entity that will be
performing certain key Part D functions
has at least 1 full benefit year of
experience providing the function or
providing the function for another Part
D plan sponsor. The burden associated
with this requirement is the time and
effort put forth by Part D applicants to
answer questions about such experience
as part of the Part D application process.
For entities that hold an existing Part D
contract, or whose parent or another
subsidiary of that parent has already
held a Part D sponsor contract for at
least a year, it is estimated that it will
take each Part D applicant for a new
contract 2 minutes to provide 1 or 2 new
sentences in the organizational history
section of the application, and 1 minute
to respond to yes-no questions about
experience with the 3 functions for
which experience is required, for a total
of 3 minutes per applicant. For entities
new to Part D, it is estimated that it will
take each Part D applicant for a new
contract 2 minutes to provide 1 or 2 new
sentences in the organizational history
section of the application, 1 minute to
respond to yes-no questions about
experience with the 3 functions for
which experience is required, and 1
additional minute to provide at least 1
contract number of an existing or recent
Part D sponsor under which the entity
to provide the key function obtained its
experience, for a total of 4 minutes.
Based on the number of Part D
applications we receive each year, we
would anticipate no more than 60 Part
D applications for a new contract, of
which no more than 15 would be
entities new to Part D. Thus, the burden
for the 45 existing entities at 3 minutes
each, plus the burden for the 15 new
entities at 4 minutes each, brings the
total burden hours to approximately
3.25 hours. If approved, the new
application questions would be
addressed under currently approved
OMB control number (OCN) 0938–0936.
We received no comments on this
proposal and therefore are finalizing
this provision without modification.
C. ICRs Related to Eligibility of
Enrollment for Incarcerated Individuals
(§ § 417.460, 422.734, and 423.44)
We proposed to amend
§§ 417.460(b)(2)(i), 417.460(f)(1)(i),
422.74(d)(4)(i)(A), 422.74(d)(4)(v), and
423.44(d)(5) to clarify the eligibility
requirement for residing in the plan’s
service area related to incarceration for
the purposes of enrolling into and
remaining enrolled in MA, Part D, and
Medicare cost plans. To implement
these regulations, we would relay data
to plans regarding an individual’s
incarceration through the MARx system
so that the plans would be aware of the
individual’s eligibility when requesting
enrollment and notify the plans of loss
of eligibility for current members. This
data is already available to us. Thus no
new data would be collected, and there
is no new information collection or
burden on organizations.
We received no comments on the ICRs
for this proposal and therefore are
finalizing the ICR assessment without
modification.
D. ICRs Related to Rewards and
Incentives Program Regulations for Part
C Enrollees (§ 422.134)
This requirement does not impose any
new information collection
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00100 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2
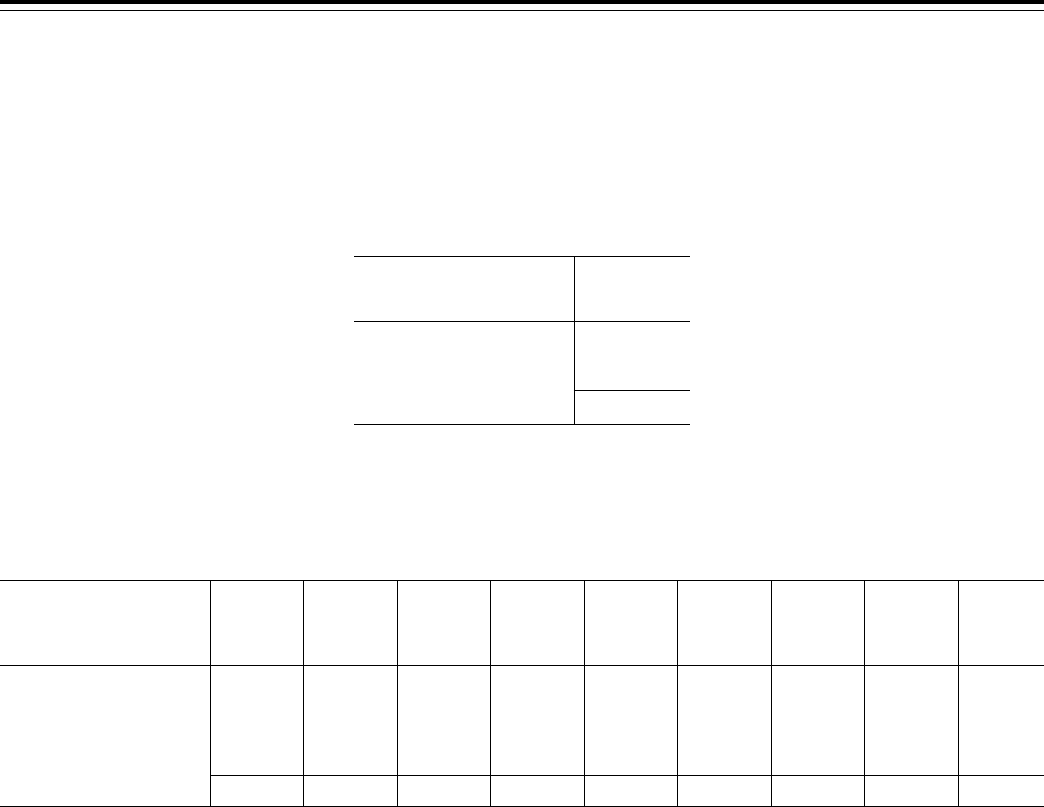
29943
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
requirements. This is an existing
recordkeeping requirement in which
MA organizations must retain
information pertaining to any rewards
and incentives programs in accordance
with our regulations at 42 CFR 422.118.
We believe the burden associated with
this requirement is exempt from the
PRA under 5 CFR 1320.3(b)(2) as we
believe this is a usual and customary
business practice. Furthermore, any
requests to furnish the information in a
form and manner we designate are
unique, that is, non-standardized and
specific to each individual MA
organization.
We received no comments on the ICR
assessment for this proposal and
therefore are finalizing this assessment
without modification.
E. ICR Related To Recovery Audit
Contractor Determinations (Part 422,
Subpart Z and Part 423, Subpart Z)
The information collection burden
associated with our proposed
requirements consists of the submission
of requests for: (1) Reconsiderations; (2)
CMS hearing official determinations;
and (3) CMS Administrator reviews.
Based on existing Part D appeals data,
we estimate that plans will file the
following numbers of requests on an
annual basis:
T
ABLE
4—E
STIMATED
N
UMBER OF
P
ART
C & D RAC A
PPEAL
R
EQUESTS
Type of request
Number of
requests per
year
Reconsideration .................... 104
CMS Hearing Official ............ 10
Administrator Review ............ 2
Total ............................... 116
The reasons for the decrease in
requests at higher appeal levels are that:
(1) The plan may succeed in its appeal
and thus have no need to appeal to the
next level; and (2) the plan may simply
wish to forgo further appeals. We stress
that the figures in Table 4 are mere
projections, though, again, they are
based on the number of Part D appeals
that have been submitted to date.
We estimate that it would take a plan
5 hours to prepare and file an appeal
request. In terms of cost, it has been our
experience that most appeals have been
prepared by high-level officials of the
plan. According to the most recent wage
data provided by the Bureau of Labor
Statistics (BLS) for May 2012, the mean
hourly wage for the category of ‘‘General
and Operations Managers’’—which we
believe, considering the variety of
officials who have submitted appeals, is
the most appropriate category—is
$55.22. With fringe benefits and
overhead, the per hour rate is $83.35.
Multiplying this figure by 580 hours (or
116 submissions × 5 hours) results in a
projected annual cost burden of
$48,343, as outlined in Table 5.
T
ABLE
5—E
STIMATED
A
NNUAL
R
EPORTING
/R
ECORDKEEPING
B
URDEN
Regulation section(s)
OMB control
No.
Respond-
ents
Responses
Burden per
response
(hours)
Total annual
burden
(hours)
Hourly labor
cost of
reporting
($)
Total labor
cost of
reporting
($)
Total
capital/
mainte-
nance costs
($)
Total cost
($)
§ 422.2605 ................................. N/A 52 52 5 260 83.35 83.35 0 21,671.00
§ 422.2610 ................................. N/A 5 5 5 25 83.35 83.35 0 2083.75
§ 422.2615 ................................. N/A 1 1 5 5 83.35 83.35 0 416.75
§ 423.2605 ................................. N/A 52 52 5 260 83.35 83.35 0 21,671.00
§ 423.2610 ................................. N/A 5 5 5 25 83.35 83.35 0 2083.75
§ 423.2615 ................................. N/A 1 1 5 5 83.35 83.35 0 416.75
Total ................................... N/A 116 116 N/A 580 .................... .................... 0 48,343
We received no comments on the ICR
assessment for this proposal and
therefore are finalizing this assessment
without modification.
V. Regulatory Impact Analysis
A. Statement of Need
The purpose of this final rule is to
make revisions to the MA program (Part
C) and Prescription Drug Benefit
Program (Part D), implement provisions
specified in the Affordable Care Act,
and make other changes to the
regulations based on our continued
experience in the administration of the
Part C and Part D programs. This final
rule makes changes that are necessary
to: Clarify various program participation
requirements and make other
clarifications and technical changes.
B. Overall Impact
We have examined the impacts of this
rule as required by Executive Order
12866 on Regulatory Planning and
Review (September 30, 1993), Executive
Order 13563 on Improving Regulation
and Regulatory Review (January 18,
2011), the Regulatory Flexibility Act
(RFA) (September 19, 1980, Pub. L. 96–
354), section 1102(b) of the Social
Security Act, section 202 of the
Unfunded Mandates Reform Act of 1995
(March 22, 1995, Pub. L. 104–4),
Executive Order 13132 on Federalism
(August 4, 1999), and the Congressional
Review Act (5 U.S.C. 804(2)).
Executive Orders 12866 and 13563
direct agencies to assess all costs and
benefits of available regulatory
alternatives and, if regulation is
necessary, to select regulatory
approaches that maximize net benefits
(including potential economic,
environmental, public health and safety
effects, distributive impacts, and
equity). Executive Order 13563
emphasizes the importance of
quantifying both costs and benefits, of
reducing costs, of harmonizing rules,
and of promoting flexibility. A
regulatory impact analysis (RIA) must
be prepared for major rules with
economically significant effects ($100
million or more in any 1 year). This
final rule has been designated an
‘‘economically significant’’ rule under
section 3(f)(1) of Executive Order 12866.
Accordingly, we have prepared a
regulatory impact analysis that details
the anticipated effects (costs, savings,
and expected benefits), and alternatives
considered. Finally, in accordance with
the provision of the Executive Order
12866, this final rule was reviewed by
the Office of Management and Budget.
The RFA requires agencies to analyze
options for regulatory relief of small
entities, if a rule has a significant impact
on a substantial number of small
entities. For purposes of the RFA, small
entities include small businesses,
nonprofit organizations, and small
governmental jurisdictions. The great
majority of hospitals and most other
health care providers and suppliers are
small entities, either by being nonprofit
organizations or by meeting the SBA
definition of a small business (having
revenues of less than $7.0 million to
$35.5 million in any 1 year). Individuals
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00101 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29944
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
and states are not included in the
definition of a small entity. This final
rule primarily affects the federal
government, Medicare Advantage plans,
and Part D Sponsors.
Part D sponsors and MA plans,
entities that will be affected by the
provisions of this rule, are not generally
considered small business entities. We
determined that there were very few MA
plans and Part D sponsors that fell
below the size thresholds for ‘‘small’’
businesses established by the Small
Business Administration (SBA).
Currently, the SBA size threshold is
$35.5 million in total annual receipts for
health insurers (North American
Industry Classification System, or
NAICS, Code 524114) and we have
confirmed that most Part D sponsors
have Part D receipts above the $35.5
million threshold.
While a very small rural plan could
fall below the threshold, we do not
believe that there are more than a
handful of such plans. A fraction of MA
organizations and sponsors are
considered small businesses because of
their non-profit status. HHS uses as its
measure of significant economic impact
on a substantial number of small
entities, a change in revenue of more
than 3 to 5 percent. Consequently, we
do not believe that this threshold will be
reached by the requirements in this final
rule because this final rule will have
minimal impact on small entities.
Therefore, an analysis for the RFA will
not be prepared because the Secretary
has determined that this final rule will
not have a significant impact on a
substantial number of small entities.
In addition, section 1102(b) of the Act
requires us to prepare an analysis if a
rule may have a significant impact on
the operations of a substantial number
of small rural hospitals. This analysis
must conform to the provisions of
section 604 of the RFA. For purposes of
section 1102(b) of the Act, we define a
small rural hospital as a hospital that is
located outside of a metropolitan
statistical area and has fewer than 100
beds. We are not preparing an analysis
for section 1102(b) of the Act because
the Secretary has determined that this
final rule will not have a significant
impact on the operations of a substantial
number of small rural hospitals.
Section 202 of the Unfunded
Mandates Reform Act of 1995 (UMRA)
also requires that agencies assess
anticipated costs and benefits before
issuing any rule whose mandates
require spending in any 1 year by state,
local, or tribal governments, in the
aggregate, or by the private sector of
$100 million in 1995 dollars, updated
annually for inflation. In 2014, that
threshold is approximately $141
million. This final rule is not expected
to reach this spending threshold.
Executive Order 13132 establishes
certain requirements that an agency
must meet when it promulgates a final
rule that imposes substantial direct
requirement costs on state and local
governments, preempts state law, or
otherwise has federalism implications.
Based on CMS Office of the Actuary
estimates, we do not believe that this
final rule imposes substantial direct
requirement costs on state and local
governments, preempts state law, or
otherwise has federalism implications.
Table 10 details the final rule’s
impacts by entity, including the federal
government and MA organizations and
Part D sponsors. We note that the
estimated savings do not represent net
social benefits because they consist of
transfers of value from drug
manufacturers, pharmacies, and
incarcerated individuals to the federal
government, MA organizations, Part D
sponsors and beneficiaries who
continue in the programs.
C. Anticipated Effects
1. Effects of Closing Cost Contract Plans
to New Enrollment
We proposed to ensure that
organizations do not move enrollees
from one of their cost or MA plan types
to another based on financial or some
other interest, and to revise
§ 422.503(b)(5) so that an entity seeking
to contract as an MA organization must
‘‘not accept new enrollees under a
section 1876 reasonable cost contract in
any area in which it seeks to offer an
MA plan if the MA organization and
reasonable cost contract are offered by
the same parent organization.’’ We
believe this provision will have minimal
or no financial impact as only a handful
of parent organizations currently offer
MA and cost plans in the same service
area. In addition, as the regulation
requires that affected cost plans close to
new enrollment, not that they terminate
operations, we believe that there will be
little or no impact to beneficiaries. We
are finalizing the provision as proposed,
with the revisions specified in our
response to public comments earlier in
this document.
2. Effects of Authority To Impose
Intermediate Sanctions and Civil Money
Penalties
We proposed to make two changes to
existing authority for the imposition of
intermediate sanctions and civil money
penalties (CMPs). First, under the
Affordable Care Act, new authority was
provided to the Secretary, which now
permits CMS to impose intermediate
sanctions for additional contract
violations in the areas of marketing and
enrollment. This new authority further
permits CMS to impose intermediate
sanctions on contracting organizations’
that employ or contract with
organizations, agents, and suppliers
who commit any of the contract
violations contained in §§ 422.752 and
423.752.
Second, we are clarifying our
authority to impose CMPs for the
aforementioned contract violations.
Current regulations designate the OIG as
the sole government agency with the
authority to impose CMPs for the
contract violations contained in
§§ 422.752 and/or 423.752. We are
modifying the language of these
provisions to clarify that CMS or the
OIG may impose CMPs for these
contract violations except the provision
that relates to the misrepresentation of
falsification of information furnished to
CMS, an individual or entity.
We believe these provisions will not
result in additional burden to sponsors
nor will they have a financial impact on
sponsors.
3. Effects of Contract Termination
Notification Requirements and Contract
Termination Basis
In current regulations, we are required
to provide 90-day notice to
organizations whose contracts are being
terminated by CMS. The authorizing
statute at section 1857(h)(1)(B) and
1860D–12(b)(3)(F) of the Act states that
the Secretary must provide reasonable
notice and opportunity for hearing
(including the right to appeal the initial
determination) before terminating a
contract (except under certain
circumstances). We proposed to modify
the notice timeframe from 90 days to 45
days. We believe these provisions will
not result in additional burden to
sponsors nor will it have a financial
impact on sponsors.
4. Effects of Reducing the Burden of the
Compliance Program Training
Requirements
We proposed to lessen the burden
placed on contracting organizations and
their first tier, downstream and related
entities (FDRs). Current regulations
specify that contracting organizations
are required to provide general
compliance program training for their
FDRs upon initial contracting and
annually thereafter. To lessen this
burden, we will require all contracting
organizations to accept a certificate of
completion of the CMS Standardized
General Compliance Program Training
and Education Module as evidence of
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00102 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29945
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
satisfaction of this program requirement.
Under this program change, contracting
organizations will not be permitted (or
required) to develop or implement
organization specific training for FDRs.
We anticipate that this will greatly
reduce the burden on various sectors of
the industry including, but not limited
to, insurance providers, hospitals,
suppliers, pharmacists and physicians.
We anticipate that this change will
actually provide savings for sponsors
and the FDRs since FDRs will only have
to take one training as opposed to the
possible numerous trainings they may
take under current requirements.
Additionally, sponsors will save
because they will not be required to
provide training materials to each FDR
with which they contract.
We believe these provisions will not
result in additional burden to sponsors
nor will they have a financial impact on
sponsors.
5. Effects of Procedures for Imposing
Intermediate Sanctions and Civil Money
Penalties Under Part C and D
We proposed to make changes to our
authority for imposing intermediate
sanctions and for determining when
such sanctions will be lifted. Sections
1857(g) and 1860D–12(b)(3)(E) of the
Act provide the Secretary the ability to
impose intermediate sanctions on MA
organizations and PDP sponsors.
Intermediate sanctions consist of
suspension of enrollment, suspension of
marketing and suspension of payment.
Current regulations governing
intermediate sanctions are contained in
subparts O of part 422 and Part 423.
Sections 422.756 and 423.756 provide
specific procedures for imposing
intermediate sanctions and include
provisions, which address: The duration
of the sanction; and the standard that
we apply when determining if a
sanction should be lifted. As specified
in the Act and regulations, when
intermediate sanctions are imposed on
contracting organizations, the sanctions
remain in place until the Secretary/CMS
is satisfied that the basis for the sanction
determination has been corrected and is
not likely to recur.
In the October 2009 proposed rule (74
FR 54634), we proposed a change that
included a rule that allows us to require
a plan under a marketing and/or
enrollment sanction to market or accept
enrollments or both for a limited period
of time. As we explained in that
proposed rule, the purpose of the test
period is to assist us in making a
determination as to whether the
deficiencies that are the bases for the
intermediate sanctions have been
corrected and are not likely to recur.
The test period provides us with the
opportunity to observe a sanctioned
plans ability to enroll or market to
Medicare beneficiaries prior to lifting
the sanction.
We proposed to extend the
applicability of such a test period to
include all intermediate sanctions and
to clarify that while we may require a
sponsor to receive enrollments during
this test period, the sponsor will not
receive any LIS annual or auto
facilitated reassignments.
We believe these provisions will not
result in additional burden to sponsors
nor will they have a financial impact on
sponsors.
6. Effects on Timely Access to Mail
Order Services
We proposed to establish a fulfillment
requirement for mail order
prescriptions. We believed it was
necessary and appropriate to establish
mail order fulfillment requirements
defining maximum turnaround times
from when the pharmacy receives the
prescription order to when it is shipped.
This would underscore the importance
of consistent and reliable access to
medications, protecting beneficiaries
from inconsistent or unreliable practices
that may otherwise jeopardize timely
access to prescriptions.
Comments persuaded us that we had
not considered all relevant implications
of this proposal and we decided not to
finalize this provision. This in turn
means that there will be no financial
impact.
7. Effects of the Modification of the
Agent/Broker Compensation
Requirements
The current independent agent
compensation structure (as originally
published as CMS–4138–IFC2 in
November 2008) is comprised of a 6-
year cycle which ended December 31,
2013. MA organizations and Part D
sponsors provide an initial
compensation payment to independent
agents for new enrollees or unlike plan
changes (Year 1), and pay a renewal rate
(equal to 50 percent of the initial year
compensation) for Years 2 through 6.
We proposed revising this existing
compensation structure. MA
organizations and Part D sponsors will
have the discretion to decide, on an
annual basis, whether to pay initial and/
or renewal compensation payments to
their independent agents. For new or
unlike plan change enrollments, MA
organizations and Part D sponsors could
make an initial payment that is no
greater than the fair market value (FMV)
amount for such services, set annually
by CMS in guidance interpreting these
regulations. For renewals in Year 2 and
subsequent years, the MA organization
or Part D sponsor could pay up to 35
percent of the FMV amount for that
year. We are finalizing the provision
with an up to 50 percent payment for
renewals, instead of the proposed 35
percent. We also proposed that plans
not recover compensation when the
disenrollment is not a result of the
agent’s behavior. We are not
implementing the changes with respect
to the recovery of compensation, but
will finalize language to keep the
existing situation, which requires full
recoupment if a member disenrolls
within the first 3 months of enrollment
except in limited circumstances. In
addition to the agent and broker
compensation structures, we are setting
limits on referral fees for agents and
brokers.
We do not believe that any of these
revisions will have a significant increase
in burden or financial impact. Our
existing compensation rules require that
MA organizations and Part D sponsors
pay on a calendar year basis, not a
rolling year basis. Our regulations are
restating existing requirements, to
ensure consistency. While some MA
organizations and Part D sponsors may
have to make significant systems
changes to ensure compliance, these
changes are not based on this final rule
but are required to meet existing
requirements. MA organizations and
Part D sponsors will likely have to make
some systems modifications, such as
paying between January 1 and December
31 of each year. However, we do not
believe these will be of significant
impact. Although some changes will be
necessary, we believe the small cost and
burden of the changes will outweigh the
cost and burden of the existing multi-
tier approach by simplifying the
compensation structure for independent
agent brokers.
8. Effects of Drug Categories or Classes
of Clinical Concern
We are not finalizing the proposed
criteria or their application to the
categories and classes of clinical
concern.
9. Effects of Medication Therapy
Management Program (MTMP) under
Part D
Current regulations require that Part D
sponsors must have established a
Medication Therapy Management
Program that targets beneficiaries who:
(1) Have multiple chronic diseases with
three chronic diseases being the
maximum number a Part D plan sponsor
may require for targeted enrollment; (2)
are taking multiple Part D drugs, with
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00103 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29946
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
eight Part D drugs being the maximum
number of drugs a Part D plan sponsor
may require for targeted enrollment; and
(3) are likely to incur costs for covered
Part D drugs in an amount greater than
or equal to $3000, as increased by an
annual percentage. We specified in
guidance that while Part D sponsors are
permitted to target beneficiaries with
select chronic diseases, they must
include at least five of nine core chronic
diseases in their criteria. These
provisions have generated wide
variability in MTM programs. Moreover,
despite opt-out enrollment, completion
rates for comprehensive medication
reviews (CMR) remain very low.
We proposed to broaden the MTM
criteria to require that Part D sponsors
target beneficiaries who have two or
more chronic diseases and are taking
two or more covered Part D drugs. We
proposed to set the annual cost
threshold at an amount commensurate
with the annual amount of Part D costs
incurred by individuals that meet the
first two criteria regarding multiple
chronic conditions and use of multiple
covered Part D drugs. Applying this
methodology, we would have set the
cost threshold at $620 which is the
approximate cost of filling two generic
prescriptions. We proposed to revise
this number periodically to reflect more
up-to-date information regarding the
drug spending of beneficiaries that have
two or more chronic conditions and use
two covered Part D drugs. We estimated
that 2.5 million beneficiaries are
currently eligible for MTM services, 13
percent opt-out of the MTM program,
and 10 percent of participating
beneficiaries will receive an annual
CMR. We also estimated that an average
CMR requires 35 minutes to complete
and the average hourly compensation
(including fringe benefits, overhead,
general, and administrative expenses
and fee) of the MTM provider is $120
(labor cost per CMR is $70), and that it
costs $0.91 to print and mail a CMR
summary in CMS’ standardized format.
Therefore, the estimated total annual
cost of providing CMRs in all settings is
$15,422,925 ($70.91/CMR x 217,500
CMRs). Previously, prior to the
availability of more precise opt-out and
CMR rates, we estimated that the total
burden associated with conducting
CMRs and delivering the CMR written
summary in CMS’ standardized format
was 1,192,429 hours with a cost of
$143,363,555, including delivery of
1,896,500 CMRs in all settings under the
current eligibility criteria, and
implementation and mailing costs for
the CMR summary in standardized
format (see OMB Control No. 0938–
1154). We do not currently have data or
estimates to determine the costs
associated with quarterly targeted
medication reviews and follow-up
interventions, if necessary.
In the proposed rule, we estimated
that 18 million beneficiaries would be
eligible for MTM services based on the
proposed criteria. Using the same opt-
out, CMR, and expense rates as before,
the estimated total annual cost of
providing CMRs in all settings would be
$111,045,060 ($70.91/CMR × 1,566,000
CMRs). This was below previous
estimates.
We were unable to definitively score
the proposed changes to the eligibility
criteria because the portion of the
administrative costs attributable to
MTM is not a specific line item that can
be easily extracted from the bid.
Although the increase in the number of
CMRs was estimated to cost $111
million, we cited evidence in the
proposed rule that showed that MTM
services may generate overall medical
savings.
We are not finalizing these proposals.
Therefore, the increased burden
estimates associated with increasing
eligibility from 2.5 million beneficiaries
to 18 million beneficiaries are removed.
10. Effects of Requirement for
Applicants or Their Contracted First
Tier, Downstream, or Related Entities to
Have Experience in the Part D Program
Providing Key Part D Functions
Based on CMS’ authority at section
1860D–12(b)(3)(D) of the Act to adopt
additional contract terms, not
inconsistent with the Part C and D
statutes, that are necessary and
appropriate to administer the Part D
program, we proposed at
§ 423.504(b)(8)(i) through (iii) that Part
D organizations seeking a new Medicare
contract must have arrangements in
place such that either the applicant, or
a contracted entity that will be
performing certain key Part D functions,
has at least one full benefit year of
experience providing key Part D
functions. This proposal ensures that
applicants take advantage of the
abundant Part D industry expertise and
experience that exists today in the
development of their Part D program
operations, rather than relying on
technical assistance from CMS and
having their inexperience place
beneficiaries’ access to prescription
drugs at risk. We believe this provision
will have a very minor savings impact
on the federal budget, based on savings
of time and effort (staff time and
contracted auditor time and resources)
that the government would spend on
overseeing the disproportionate level of
problems experienced by organizations
operating Part D plans without prior
Part D experience. For each
inexperienced organization allowed into
the program in the absence of this
proposal, we would anticipate a savings
of 1,000 staff hours at an average rate of
$50 per hour, for a total of $50,000 in
employee time, plus an additional
savings of $200,000 in contractor dollars
to conduct an emergency audit, for a
total of $250,000. In the absence of this
proposal, we would anticipate no more
than two such inexperienced entities
beginning Part D operations per year, for
a total annual savings of $500,000.
The burden associated with this
proposal on industry will be minimal,
with a total estimated number of labor
hours of 3.25 to submit information
during the Part D application process.
Using the same average hourly salary as
previously mentioned, the total cost to
Part D applicants will be $162.50. We
do not believe there are any non-
administrative costs to industry
associated with this proposal, as Part D
applicants are already required to have
arrangements in place to perform the
key Part D functions discussed in our
proposal.
The main anticipated effect from this
proposal is ensuring that only entities
with some experience with Part D in
critically important functional areas are
permitted to offer new Part D contracts,
thus strengthening the Part D program
by enhancing the qualification criteria.
We considered the alternate proposal of
requiring the prior Part D experience to
be tied to specific quality outcomes. We
rejected the alternative because we
believed it added unnecessary
complexity and burden to the process,
and we believe a simple experience
requirement is currently sufficient.
11. Effects of Requirement for
Applicants for Stand Alone Part D Plan
Sponsor Contracts To Be Actively
Engaged in the Business of the
Administration of Health Insurance
Benefits
Based on CMS’ authority at section
1860D–12(b)(3)(D) of the Act to adopt
additional contract terms, not
inconsistent with the Part C and D
statutes, that are necessary and
appropriate to administer the Part D
program, we proposed at
§ 423.504(b)(9)(i) through (ii) that
organizations seeking to offer a stand-
alone prescription drug plans (PDP) for
the first time must have either: (i)
Actively offered health insurance or
health benefits coverage for 2
continuous years immediately prior to
submitting an application, or (ii)
actively managed prescription drug
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00104 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29947
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
benefits for a company offering health
insurance or health benefits coverage for
5 continuous years immediately prior to
submitting an application. This
proposal will ensure that applicants
have substantial experience in
administering health insurance benefits
prior to becoming a Part D sponsor. We
believe this provision will have a very
minor savings impact on the federal
budget, based on savings of time and
effort (staff time and contracted auditor
time and resources) that the government
would spend on overseeing the
disproportionate level of problems
experienced by organizations operating
stand-alone PDPs without prior health
insurance administration experience.
For each inexperienced organization not
allowed into the program in the absence
of this proposal, we would anticipate a
savings of 1,000 staff hours at an average
rate of $50 per hour, for a total of
$50,000 in employee time, plus an
additional savings of $200,000 in
contractor dollars to conduct an
emergency audit, for a total of $250,000.
In the absence of this proposal, we
would anticipate no more than two such
inexperienced entities beginning Part D
operations per year, for a total annual
savings of $500,000.
The burden associated with this
proposal on industry will be minimal,
with a total estimated number of labor
hours of 3.25 to submit information
during the Part D application process.
Using the same average hourly salary as
previously mentioned, the total cost to
Part D applicants will be $162.50. We
do not believe there are any non-
administrative costs to industry
associated with this proposal, as Part D
applicants are already required to be
licensed in at least one state prior to
offering Part D benefits.
The main anticipated effect from this
proposal is ensuring that only entities
with some experience administering
health insurance benefits will be
permitted to offer new stand-alone
PDPs, thus strengthening the Part D
program by enhancing the qualification
criteria. CMS considered the alternate
proposal of requiring the prior health
insurance benefit administration
experience to be tied to specific quality
outcomes. We rejected this alternative
because we believed it added
unnecessary complexity and burden to
the process, and we believe a simple
experience requirement is currently
sufficient.
12. Effects of Limit Parent Organizations
To One Prescription Drug Plan (PDP)
Sponsor Contract per PDP Region
This provision has no quantifiable
impact because the savings that might
be achieved likely will be offset by the
burden necessary with the consolidation
activities and legal work necessary to
implement these changes.
13. Effects of Limit Stand-Alone
Prescription Drug Plan Sponsors To
Offering No More Than Two Plans per
PDP Region
As this proposal is not being
finalized, there will be no financial
impact.
14. Effects of Applicable Cost-Sharing
for Transition Supplies: Transition
Process Under Part D
We proposed to add at
§ 423.120(b)(3)(vi) a paragraph clarifying
that a Part D sponsor must charge cost
sharing as follows: (a) For low-income
subsidy (LIS) enrollees, a sponsor must
not charge higher cost sharing for
transition supplies than the statutory
maximum copayment amounts; (b) for
non-LIS enrollees, a sponsor must
charge: (1) The same cost sharing for
non-formulary Part D drugs provided
during the transition that would apply
for non-formulary drugs approved under
a coverage exception; and (2) the same
cost sharing for formulary drugs subject
to utilization management edits
provided (for example, prior
authorization and step therapy) during
the transition that would apply once the
utilization management criteria are met.
Because increases or decreases in cost
sharing during transition supplies under
the various circumstances are likely to
offset one another, we anticipate that
there will be no cost impact on plans.
15. Effects of Interpreting the Non-
Interference Provision
We proposed to formally interpret
section 1860D–11(i) of the Act, referred
to as the non-interference provision.
This provision prohibits CMS from
interfering with the negotiations
between drug manufacturers and
pharmacies and Part D sponsors, and
requiring a particular formulary or
instituting a price structure for the
reimbursement of covered part D drugs.
We have not formally interpreted the
statutory provision, which has resulted
in different stakeholders having
different views about its scope.
Consequently, we believe that a clear
interpretation of the statutory provision
will remove ambiguity. As we are not
finalizing this proposal, there is no
change in regulatory impact.
16. Effects of Pharmacy Price
Concessions in Negotiated Prices
We proposed to revise the definition
of negotiated prices at § 423.100 to
specify that all pharmacy price
concessions must be included in the
negotiated price. This will preclude the
differential reporting that is taking place
today in the realm of reporting drug
costs and price concessions from
network pharmacies. The rule will
change current policy that permits
sponsors to elect which price
concessions from pharmacies to report
outside the PDE. This practice currently
allows price concessions to be applied
disproportionately to costs that plans
are liable for, and thus may shift more
low-income cost-sharing subsidy and
reinsurance costs to the government, as
well as to manufacturers in the
calculation of coverage gap discount
payments. A sponsor that engages in
this practice can reduce its bid and
achieve a competitive advantage relative
to a sponsor that applies all price
concessions to the negotiated price—a
competitive advantage stemming not
from greater efficiency, but from a
technical difference in how costs are
reported to CMS. Meanwhile, the higher
the negotiated price, the higher
beneficiary coinsurance will be, the
faster the beneficiary is moved through
the benefit, and the higher government
subsidies for low-income cost sharing
(LICS) and reinsurance subsidies will
be. Our proposal will impose consistent
treatment of drug price reporting.
Our proposal to require all price
concessions to be reflected in the
negotiated price received by the
pharmacy would not necessarily change
the level of price concessions received
from network pharmacies, but will
impose a single consistent price
concession reporting process on all Part
D sponsors. Therefore, it is not clear that
any contractual arrangements between a
subset of sponsors and network
pharmacies will require renegotiation,
since only the form of the price
concession, rather than its level, will be
affected by this proposal.
In addition, when price concessions
from pharmacies are in forms other than
the negotiated price, the degree of price
concession that the pharmacy has
agreed is no longer reflected in the
negotiated prices available at point of
sale or reflected on the Medicare
Prescription Drug Plan Finder (Plan
Finder) tool. Thus, the true price of
drugs at individual pharmacies is no
longer transparent to the market.
Consequently, consumers cannot
efficiently minimize both their costs
(cost sharing) and costs to the taxpayers
by seeking and finding the lowest-cost
drug/pharmacy combination. This
proposal will ensure that the actual
level of price competition is transparent
to the Part D market.
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00105 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29948
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
Under current policy, a sponsor may
be able to offer a lower bid than its
competitors and may achieve a
competitive advantage stemming not
from greater efficiency, but from a
technical difference in how costs are
reported to CMS. When this happens,
such differential reporting may result in
bids that are no longer comparable, and
in premiums that are no longer valid
indicators of relative plan efficiency.
The changes we proposed will lend to
Part D bids being more accurately
comparable and premiums more
accurately reflecting relative plan
efficiencies. The lowest premiums will
more accurately direct beneficiaries to
the plans that have the lowest costs to
the program overall.
We do not collect sufficient detail in
price concession data reported to CMS
to quantify the impact of this change to
standardize price concession reporting.
We believe that only certain sponsors
are engaging in the differential reporting
practices today, and these sponsors face
close competition from larger
competitors that do not appear to be
employing the same strategies.
Consequently, if the sponsors
employing these tactics increase their
bids to maintain margin, they could
likely risk losing market share.
Therefore, we would expect these
sponsors to carefully consider the risk of
losing market share before raising their
bids in response to our regulatory
proposals, particularly those that are
committed to the LIS market.
We are finalizing the provision with
modification to require that negotiated
prices be inclusive of all price
concessions from network pharmacies
except those contingent price
concessions that cannot reasonably be
determined at the point of sale. We
expect that the effect of regulation to
require consistent and transparent
pricing will not only provide higher-
quality information to the Part D market,
but also promote increased price
competition among network
pharmacies. This expectation is
consistent with economic theory that
holds that increased price transparency
will increase price competition. We
believe pharmacies will support
including the full price concession in
the point-of-sale price, and fully
transparent price competition will align
beneficiary and taxpayer interests in
minimizing costs. Our rule will not
change the level of price concessions
and therefore costs under the program
as a whole, but will apply consistency
to how these are reported to CMS and
treated in bidding and payment
processes. Therefore, we anticipate that
there will be no cost impact on plans.
17. Effects of Preferred Cost Sharing
We proposed to require that sponsors
may offer reduced copayments or
coinsurance for covered Part D drugs
obtained through a subset of network
pharmacies, as long as such preferred
cost sharing is in return for consistently
lower negotiated prices relative to the
same drugs when obtained in the rest of
the pharmacy network. Therefore, we
intended to clarify that preferred cost
sharing should consistently be aligned
with and accurately signal lower costs.
We proposed that by ‘‘consistently
lower’’ we meant that sponsors must
offer better prices on all drugs in return
for the lower cost sharing. In practice
we believe this would mean that
whatever pricing standard is used to
reimburse drugs purchased from
network pharmacies in general, a lower
pricing standard must be applied to
drugs offered at the preferred level of
cost sharing. Our analysis shows that
most sponsors offering preferred cost
sharing are currently achieving these
levels of savings, and therefore our
proposed policy would only require a
change in price concession levels or
reporting for a limited number of
sponsors. Our proposal would apply a
consistent expectation across all
sponsors to compete on the same basis
on negotiated prices, including in
related-party pharmacy operations.
After considering the public
comments, we are not finalizing the
proposal to revise § 423.120(a)(9) to
require consistently lower negotiated
prices for Part D drugs obtained through
pharmacies offering preferred cost
sharing than the same Part D drugs
when obtained in the rest of the
pharmacy.
This proposal will not be finalized
and we will not engage in further
rulemaking without re-proposing in a
future rule, eliminating any estimated
costs for implementation at this time.
18. Effects of Maximum Allowable Cost
Pricing Standard
We proposed a change to the
regulations at §§ 423.501, 423.505(b)(21)
and 423.505(i)(3)(vii) governing the
disclosure and updating of prescription
drug pricing standards used by Part D
sponsors to reimburse network
pharmacies to make clear that drug
pricing based on maximum allowable
cost (MAC) is subject to these
regulations. In the final rule at 76 FR
54600 (September 1, 2011), we did not
estimate a regulatory impact for Part D
sponsors to comply with the
prescription drug pricing standard
requirements, and we do not believe
these changes would result in any
regulatory impact. Read together, the
new provisions in §§ 423.501,
423.505(b)(21), and 423.505(i)(3)(viii)
require sponsors, when applicable, to
include provisions in network
pharmacy contracts, to address the
disclosure of MAC prices themselves to
be updated to the applicable pharmacies
in advance of their use for
reimbursement of claims, because the
source of the MAC prices is not publicly
available. Addressing prices that will be
paid to a subcontractor is an activity
undertaken in the normal course of
business. Also, whether to use MAC
prices is voluntary for Part D sponsors.
Finally, sponsors must have procedures,
systems, and technology currently in
place to use these prices for
reimbursement of pharmacy claims in
the normal course of business. These
systems will have to be adapted to also
disclose the prices to pharmacies in
advance of their use, which we believe
will involve negligible effort for Part D
sponsors’ existing employees and/or
subcontractors. Therefore, we estimate
the impact of these provisions to be
negligible.
19. Effects of Any Willing Pharmacy
Standard Terms & Conditions
Proposed changes to § 423.120(a)(8)
would require Part D sponsors to offer
the contract terms and conditions (T&C)
for every level of cost sharing offered
under a Part D plan (preferred, standard
retail, mail order, etc.) to any willing
pharmacy. We expected the burden for
Part D sponsors to amend contracts,
where necessary, to offer every level of
cost sharing would be negligible.
Sponsors already must meet any willing
pharmacy requirements for retail and
mail order cost sharing. In 2013, nearly
half of non-employer group Part D
sponsors were designing and marketing
plans with T&C for preferred cost
sharing levels. For these sponsors, the
only change associated with this
proposal would have been to ensure that
now T&C for all levels of cost sharing,
including preferred, are being offered (if
they are not already) to all interested
pharmacies. For the other half of Part D
sponsors not currently offering preferred
cost sharing options, this proposal did
not require them to start.
Part D sponsors already negotiate
contracts regularly with pharmacies in
order to meet network access
requirements. We estimated that for
sponsors who currently offer benefit
packages with a preferred cost sharing
level (approximately 500 plans), an
estimated new burden of 5,000 legal
hours (500 plans x 10 hours) for revising
contract language and 2,000 hours (500
plans x 4 hours) for additional contract
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00106 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29949
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
support staff time negotiating with and
assisting pharmacies contracting at the
preferred cost sharing level for the first
time. The estimated cost associated with
this change is the estimated number of
hours multiplied by available average
hourly rates ($62.93 per hour for a
lawyer, $32.22 per hour for a financial
specialist [May 2012 wage data from
Bureau of Labor Statistics Occupational
Employment Statistics]), plus 48 percent
for fringe benefits and overhead, which
equals a first year cost of $561,053.20.
Once a sponsor had revised contracts to
meet the proposed requirement, no
extraordinary additional expenses were
anticipated for subsequent years. For a
plan not currently offering preferred
cost sharing levels, it was expected that
preferred cost sharing terms and
conditions would be offered to any
willing pharmacy if they ever decide to
offer them.
Any new burden on pharmacies was
similarly expected to be negligible, as
they are already reviewing and
implementing terms from contracts,
often annually. Pharmacies were not
being directed to choose one set of T&C
over another, but rather would have
gained the option to review and
implement terms for preferred cost
sharing, if they so choose to accept the
applicable negotiated pricing terms.
Beneficiaries were expected to benefit
from an increased number of
pharmacies offering preferred cost
sharing levels.
We received the following comments
and our response follows:
Comment: Some commenters believed
that there would be additional costs not
reflected in the impact analysis,
resulting from the proposed change to
pharmacy contracts. One commenter
believed that the estimates provided for
revising contract language and
especially negotiating new contracts
with pharmacies were too low, and a
few commenters stated that it would
take more than 6 months to implement
these changes.
Response: We appreciate the
comments. However, this proposal will
not be finalized and we will not engage
in further rulemaking without re-
proposing in a future rule, eliminating
any estimated costs for implementation
at this time.
20. Effects of Enrollment Requirements
for Prescribers of Part D Covered Drugs
We proposed that prescribers must
either be enrolled in Medicare or have
validly opted-out in order for their
prescriptions to be covered under the
Part D program. This will entail Part D
sponsors or their designated PBMs
checking the prescriber’s individual NPI
to determine whether the prescriber is
enrolled or in a valid opt-out status in
Medicare before paying a claim from a
network pharmacy or a request for
reimbursement from a beneficiary.
When we promulgated the NPI PDE
requirement in a final regulation
published on April 12, 2012 (77 FR
22072), we estimated the impact for
PBMs and plan organizations to contract
for or build prescriber ID validation
services. Thus, while § 423.120(c)(6)
entails a new requirement for Part D
sponsors, we do not believe it will have
any new or additional impact because
Part D sponsors must already have
prescriber validation capabilities to
meet the NPI PDE requirement.
We presume that if a beneficiary’s
prescriber is not enrolled or does not
enroll in Medicare, the beneficiary will
find a new prescriber who is enrolled,
rather than go without needed
medications. Solely from this
perspective, we do not project any
savings from this provision. We believe
there will be savings, though, from the
fact that certain unqualified individuals
will no longer be able to prescribe Part
D drugs, for they will be unable to meet
Medicare requirements. However, we
are unable to estimate a particular
savings figure because we do not know
how many such individuals there will
be.
21. Effects of Improper Prescribing
Practices and Patterns
Our additions of §§ 424.530(a)(11) and
424.535(a)(13) will likely result in
additional application denials,
revocations, and associated appeals. The
DEA Web site found at http://
www.deadiversion.usdoj.gov/crim_
admin_actions/index.html contains a
list of physicians, eligible professionals,
and pharmacies that have had their DEA
Certificate of Registration suspended or
revoked since 2000. However, we do not
have data available to assist us in
calculating the possible costs to
physicians and eligible professionals in
lost potential billings or the possible
costs or savings to the government
arising from these two provisions.
Section 424.535(a)(14) will result in
an increase in the total number of
revocations and associated appeals. Yet
we are unable to project the number of
providers and suppliers that will be
revoked under § 424.535(a)(14) because
we do not have data available that can
be used to make such an estimate. Thus,
we cannot project: (1) The potential
costs to providers and suppliers in lost
billings, or (2) the possible costs or
savings to the government arising from
this provision.
We received the following comments
regarding the impact of proposed
§§ 424.530(a)(11), 424.535(a)(13), and
424.535(a)(14).
Comment: A commenter disagreed
with CMS’ determination that
§§ 424.530(a)(11), 424.535(a)(13), and
424.535(a)(14) do not have federalism
implications, contending that these
provisions usurp the role of state
licensing boards. The commenter
recommended that CMS explain: (1) the
federalism impacts of these provisions;
and (2) the steps that it took to consult
with state and local officials early in the
process of developing the proposed rule.
Response: We maintain that these
three provisions have no federalism
implications, for CMS is not usurping
the authority of states to take action
against a physician or practitioner with
respect to his or her licensure status.
Moreover, as stated earlier, CMS (not
state licensing boards) is the agency
responsible for administering the
Medicare program. Therefore, we must
have the ability to independently take
steps to protect Medicare beneficiaries
and the Trust Funds.
Comment: One commenter stated that
CMS did not furnish reasonable
alternatives to the establishment of
§ 424.535(a)(14).
Response: In light of the very serious
problem of abusive prescribing, as
outlined by the OIG, CMS did not
believe there were any reasonable
alternatives to its proposal. Prompt
action was necessary to protect
Medicare beneficiaries and the Trust
Funds.
No modifications are being made to
§§ 424.530(a)(11), 424.535(a)(13), and
424.535(a)(14) as a result of these
comments.
22. Effects of Broadening the Release of
Part D Data
We proposed to revise our regulations
governing the release of Part D data to
expand the release of unencrypted
prescriber, plan, and pharmacy
identifiers contained in prescription
drug event (PDE) records to external
entities, as well as to make other
changes to our policies regarding the
use and release of PDE data, as currently
codified at § 423.505 (f)(3), (l) and (m).
These proposals would not impose any
new costs on any stakeholders.
Medicare Part D plan sponsors are
already required to, and do, submit the
information that may be used or
released in accordance with these
proposals. Therefore, although we are
finalizing the revisions to the Part D
data regulations as proposed, we are not
including any assessment of this final
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00107 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29950
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
policy for the regulatory impact
statement.
23. Effects of Establish Authority To
Directly Request Information From First
Tier, Downstream, and Related Entities
Pursuant to sections 1857(d)(2) and
1860D 12(b)(3)(c) of the Act, we are now
proposing to specify at
§§ 422.504(i)(2)(ii) and 423.505(i)(2)(ii)
that HHS, the Comptroller General, or
their designees have the right to audit,
evaluate, collect, and inspect any
records directly from any first tier,
downstream, or related entity. This
regulatory change would not grant CMS
(or the MEDIC, the contractor that
conducts fraud investigations on our
behalf) any oversight authority beyond
what we already possess.
In enabling CMS or its designee(s) to
directly request information from a first
tier, downstream, or related entity, we
would provide a more efficient avenue
to obtain necessary information. This
proposal would change the current
policy, which requires going through
the plan sponsor in order to collect
information. Our proposal would save
money and time for CMS as well as the
plan sponsor.
We anticipate that adoption of this
proposal would result in cost savings for
plan sponsors. Under the current
regulatory structure, assuming that the
MEDIC (the CMS contractor that
typically would put forth such requests)
puts forth 1000 requests per year to Part
C and D sponsors, each request requires
the plan sponsor to spend 5 hours
developing and making the request for
information from its first tier,
downstream, or related entity, and
communicating the results of that
request back to CMS. At a rate of $55
per hour, plan sponsors may save a total
of $275,000 in employee costs in the
aggregate. Additionally, we believe this
provision will have a very minor
savings impact on the federal budget.
This calculation is based on the savings
in time and effort the MEDIC will
experience (2 hours per information
request) resulting from the ability to
request information directly from first
tier, downstream, and related entities.
The 2 hours reflects the time the MEDIC
currently spends resolving ambiguities
in the request or in the information
provided in response that are created by
the presence of an intermediary (that is,
the plan sponsor) between the requestor
(MEDIC) and the custodian of the
information (that is; first tier,
downstream, or related entity).
In addition to cost savings, this
regulatory change will reduce the
administrative burden on plan sponsors.
The plan sponsor will no longer have to
act as the gatekeeper between the
MEDIC and its first tier, downstream, or
related entity.
We do not anticipate any additional
burden relating to the requirement that
we alert the plan sponsor that we are
contacting its first tier, downstream or
related entity since CMS will be merely
copying the plan sponsor on the request.
24. Effects of Eligibility of Enrollment
for Incarcerated Individuals
We proposed to amend
§§ 417.460(b)(2)(i), 417.460(f)(1)(i),
422.2, 422.74(d)(4)(i)(A), 422.74(d)(4)(v),
423.4, and 423.44(d)(5) to clarify the
eligibility requirement for residing in
the plan’s service area related to
incarceration for the purposes of
enrolling into and remaining enrolled in
MA, Part D, and Medicare cost plans.
We expect the impact of this change to
be primarily that of savings to the MA
and Part D programs. In CY 2012, there
were close to 50 million Medicare
beneficiaries. Approximately 34.4
million of those beneficiaries were
enrolled in MA plans, PDPs, or cost
plans which accounts for 68.8 percent of
the total Medicare population. In the
same year, an average of 21,329
Medicare beneficiaries enrolled in MA
or Part D plans were identified by SSA
as being incarcerated.
We issued guidance to MA plans and
PDPs to investigate each individual’s
incarcerated status and disenroll the
individual for no longer residing in the
plan’s service area if the plan confirmed
incarcerated status. If the MA plan or
PDP could not confirm the incarcerated
status, those plans were to continue to
investigate each instance of
incarceration for up to 6 or 12 months
and disenroll the individuals at the end
of that time following
§§ 422.74(b)(4)(ii)/423.44(b)(5)(ii) if they
could not verify the incarcerated status
sooner. As a result, plans received
capitated payments when individuals
were ineligible to receive payment of
Medicare benefits. Section 1876 Cost
contracts had no such instructions to
disenroll individuals who are
incarcerated. By directing MA plans,
PDPs, and cost plans to disenroll
incarcerated individuals at the time of
notification from CMS, we intend to
prevent improper payment for these
individuals to MA plans, PDPs, and cost
plans for periods when they were
ineligible to receive such services.
Based on the data for capitation
payments for MA and PDPs, as well as
the prepayments provided to cost plans,
we estimate that the disenrollment of
incarcerated individuals would result in
a decrease in improper payments made
by CMS and would result in a cost
savings of $73 million in 2015.
We estimate, based on the numbers
mentioned previously, that this change
could save the MA program
approximately $27 million in 2015,
increasing to $103 million in 2024, and
could save the Part D program (includes
the Part D portion of MA PD plans)
approximately $46 million in 2015,
increasing to $153 million in 2024. As
cost plans are paid based on the
reasonable costs of delivering Medicare
covered services to their enrollees,
instead of the fixed capitation amounts
paid to MA and PDPs, we believe the
impact to cost plans associated with this
provision to be negligible.
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00108 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29951
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
T
ABLE
6—P
ROJECTED
N
UMBER OF
I
NDIVIDUALS
D
ISENROLLED
D
UE
T
O
I
NCARCERATION AND
E
STIMATED
S
AVINGS TO THE
M
EDICARE
A
DVANTAGE
P
ROGRAM BY
P
ROVISION FOR
C
ALENDAR
Y
EARS
2015 T
HROUGH
2024
2015 2016 2017 2018 2019 2020 2021 2022 2023 2024
Totals
(CYs 2015–
2024)
Projected number of incarcerated
beneficiaries enrolled in MA plans 6,280 7,750 9,221 10,691 12,162 13,234 13,969 14,704 15,440 16,175 ........................
Projected federal impact due to in-
carcerated individuals disenrolled
6 months sooner ........................... ¥$27 million ¥$35 million ¥$43 million ¥$52 million ¥$62 million ¥$70 million ¥$78 million ¥$86 million ¥$94 million ¥$103 million ¥$650 million
Note: Estimates reflect scoring by the CMS, Office of the Actuary, and 2012 incarceration data provided by the SSA.
T
ABLE
7—P
ROJECTED
N
UMBER OF
I
NDIVIDUALS
D
ISENROLLED
D
UE
T
O
I
NCARCERATION AND
E
STIMATED
S
AVINGS TO THE
M
EDICARE
P
ART
D P
ROGRAM BY
P
ROVISION FOR
C
ALENDAR
Y
EARS
2015 T
HROUGH
2024
2015 2016 2017 2018 2019 2020 2021 2022 2023 2024
Totals
(CYs 2015–
2024)
Projected number of incarcer-
ated beneficiaries enrolled in
MA plans ............................... 52,605 60,076 67,547 75,018 82,489 87,937 91,672 95,408 99,144 102,879 ........................
Projected federal impact due to
incarcerated individuals
disenrolled 6 months sooner ¥$46 million ¥$55 million ¥$65 million ¥$77 million ¥$90 million ¥$102 million ¥$113 million ¥$125 million ¥$138 million ¥$153 million ¥$965 million
Note: Estimates reflect scoring by the CMS, Office of the Actuary, and 2012 incarceration data provided by the SSA.
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00109 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29952
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
We received the following comment:
Comment: One commenter requested
additional information on the
assumptions used to calculate the
savings related to our proposal to
disenroll incarcerated individuals, such
as the percentage of membership of
incarcerated beneficiaries.
Response: The following chart
provides the assumptions used to
calculate the savings previously
outlined:
T
ABLE
8—A
SSUMPTIONS FOR
E
LIGIBILITY OF
E
NROLLMENT FOR
I
NCARCERATED
I
NDIVIDUALS
2015 2024
Expected MAPD Enrollment due to be disenrolled (A) ........................................................................................... 6,280 16,175
Average Part C per Capita Costs ($) (B)* ............................................................................................................... 10,024 14,845
Average Length of Stay (years) (C) ........................................................................................................................ 0.5 0.5
Gross Savings ($millions) (A x B x C) .................................................................................................................... 31.5 120.1
Savings from Part A Trust Fund ($millions) (D) ...................................................................................................... 14.7 53.1
Savings from Part B Trust Fund ($millions) (E) ...................................................................................................... 16.8 67.0
Savings Net of Member Premium ($millions) (D + 0.75 x E) ................................................................................. 27.3 103.3
* Note: Part C per Capita Costs are derived from the 2014 mid-session review.
After consideration of the public
comment received, we are finalizing the
policy without modification.
25. Rewards and Incentives Program
Regulations for Part C Enrollees
(§ 422.134)
This provision permits plans to
provide limited rewards and incentives
to enrollees who participate in activities
that focus on promoting improved
health, preventing injuries and illness,
and promoting efficient use of health
care resources. While there would be a
cost associated with providing rewards
and incentives there may be savings as
a result of healthier behavior. Because
plans are not required to provide
rewards and incentives and CMS does
not have a means of calculating the
costs and benefits of rewards/incentives
at this time, we are not providing an
impact analysis for this provision.
26. Effects of Improving Payment
Accuracy: Reporting Overpayments,
RADV Appeals, and LIS Cost Sharing
This section proposes only technical
changes for overpayment reporting,
RADV appeals, and CMS’ treatment of
diagnoses for additional payment after
the final risk adjustment data
submission deadline. These technical
changes will not result in costs to MA
organizations and Part D sponsors, nor
do we expect the impact of these
technical changes to result in savings.
27. Effects of Part C and Part D RAC
Determination Appeals
In section III.B.4. of this final rule, to
establish an administrative appeals
process for overpayment determinations
by the Part C and Part D RACs. The cost
associated with these provisions
involves the preparation and
submission of appeal requests by plans.
We estimate this cost to be $48,343 as
summarized in the following Table 9.
T
ABLE
9—S
UMMARY OF
RAC D
ETERMINATION
A
PPEALS
Provision description
Costs
(in $millions)
Benefits
Submission of MA plans’ first level Request for Reconsider-
ation.
0.02167 Administrative appeal rights and accuracy in recovery de-
mands.
Submission of Part D plans’ first level Request for Reconsid-
eration.
0.02167 Administrative appeal rights and accuracy in recovery de-
mands.
Submission of MA plans’ second level Request for Review .... 0.00208 Administrative appeal rights and accuracy in recovery de-
mands.
Submission of Part D plans’ second level Request for Review 0.00208 Administrative appeal rights and accuracy in recovery de-
mands.
Submission of MA plans’ third level Request for Review by
the CMS Administrator.
0.0004 Administrative appeal rights and accuracy in recovery de-
mands.
Submission of Part D plans’ third level Request for Review by
the CMS Administrator.
0.0004 Administrative appeal rights and accuracy in recovery de-
mands.
28. Effects of the Technical Changes to
the Definition of Part D Drug
There is no impact associated with
this provision as it is a technical change
to regulation language.
29. Effects of Special Part D Access
Rules During Disasters
In § 423.126(a), we proposed to codify
requirements similar to existing
guidance that pertains to relaxing
‘‘refill-too-soon’’ (RTS) edits to permit
one refill in the event of any imminent
or occurring disaster or emergency that
would hinder an enrollee’s access to
covered Part D drugs.
The proposed changes would not
have resulted in any additional costs.
For one, we currently expect through
guidance that sponsors will relax edits
after the issuance of certain federal
declarations. We also do not anticipate
that providing a general framework for
when sponsors must relax RTS edits
would necessitate an increase in
resources because it is currently not
uncommon for Part D sponsors to relax
edits for particular individuals under
certain circumstances.
The provisions would have required
Part D sponsors to relax ‘‘refill-too-
soon’’ (RTS) edits when, as evidenced
by a declaration of a disaster or
emergency or its imminence by an
appropriate federal, state, or local
official, it is reasonable to conclude that
an occurring or imminent disaster or
emergency would make it difficult for
beneficiaries to obtain refills of their
medications. Relaxing RTS edits in
these circumstances would benefit
beneficiaries by better ensuring that
they do not run out of their medications
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00110 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2
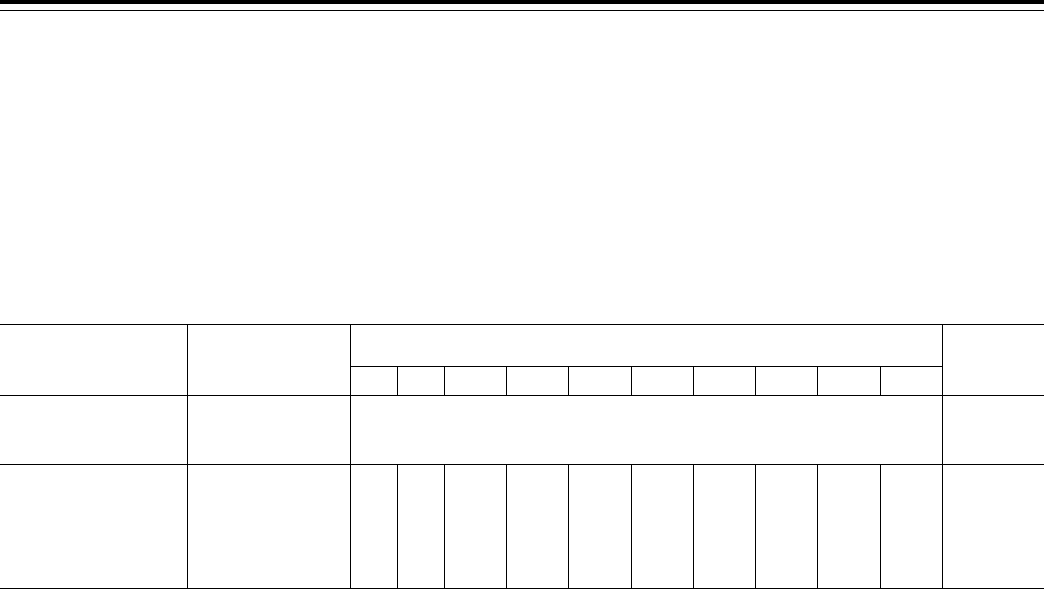
29953
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
when a disaster is imminent or after it
strikes.
As this proposal is not being
finalized, there will be no financial
impact.
30. Effects of Termination of a Contract
Under Parts C and D
The changes to §§ 422.510 and
423.509 are minor technical and
clarifying revisions and include making
language consistent, aligning titles and
correcting references. These technical
and clarifying changes will not result in
additional burden to MA organizations
or Part D sponsors nor will they have a
financial impact on such entities.
31. Effects of Technical Changes
Regarding Intermediate Sanctions and
Civil Money Penalties
The changes to §§ 422.756 and
423.756 are minor technical and
clarifying revisions and include making
language consistent, aligning titles and
correcting references. These technical
and clarifying changes will not result in
additional burden to MA organizations
or Part D sponsors nor will they have a
financial impact on such entities.
T
ABLE
10—E
STIMATED
1
A
GGREGATE
S
AVINGS TO THE
H
EALTH
C
ARE
S
ECTOR BY
P
ROVISION FOR
C
ALENDAR
Y
EARS
2015 T
HROUGH
2024
Provision Regulation section(s)
Calendar year
($ in millions)
Total
($ in millions)
CYs 2015–
2019 2015 2016 2017 2018 2019 2020 2021 2022 2023 2024
Federal
Government
(Medicare)
Impacts
A.24. Eligibility of Enroll-
ment for Incarcerated In-
dividuals
2
.
§ 417.460(b)(2)(i),
§ 417.460(f)(1)(i),
§ 422.2,
§ 422.74(d)(4)(i)(A),
§ 422.74(d)(4)(v),
§ 423.4, and
§ 423.44(d)(5).
¥73 ¥90 ¥108 ¥129 ¥152 ¥172 ¥191 ¥211 ¥232 ¥256 ¥1615
Total ($ in millions) ...... ................................. ¥73 ¥90 ¥108 ¥129 ¥152 ¥172 ¥191 ¥211 ¥232 ¥256 ¥1615
Notes:
1
Estimates of savings reflect scoring by the CMS, Office of the Actuary. Also, only provisions that are being finalized with savings or cost exceeding $1,000,000
are listed. Other provisions either have no expected savings or cost, have a savings or cost that is difficult to score, have a cost that is expected to be counterbal-
anced by savings, have a savings or cost under $1,000,000, or, were not finalized.
2
Supporting 2012 incarceration data provided by the SSA.
D. Expected Benefits
1. Drug Categories or Classes of Clinical
Concerns (§ 423.102(b)(2)(v))
Proposed codification of the
categories or classes of clinical concern
provisions would assist PBMs in
applying the Part D plans and managing
the Part D sponsor’s benefit packages
more efficiently.
However, we are not codifying the
propose criteria or applying them to the
drug categories and classes of clinical
concern. Thus, this does not apply.
2. Medication Therapy Management
Program Under Part D
We anticipated that many more
beneficiaries would have access to MTM
services and believed that the proposed
changes would have simplified the
MTM criteria and minimized
beneficiary confusion when choosing or
transitioning between plans. Moreover,
we believed the proposed changes
would have reduced disparity and
allowed more beneficiaries with drug
therapy problems to receive MTM
services.
However, we are not finalizing these
proposals, so these expected benefits are
no longer applicable.
E. Alternatives Considered
1. Modifying the Agent/Broker
Compensation Requirements
In the preamble of this final rule, we
outlined a few alternative compensation
schedules. Ultimately we determined
that the best approach was a two-tiered
payment schedule, incorporating an
initial payment and a continuous
renewal payment.
2. Any Willing Pharmacy Standard
Terms & Conditions
We considered the alternative of
maintaining the current process where
Part D plans can limit pharmacy access
to preferred cost-sharing contracts. We
have observed this in practice to be
limiting market competition, creating a
barrier to entry, and further, not
producing the savings to the program
that were initially anticipated.
We are not finalizing this proposal.
3. Pharmacy Price Concessions in
Negotiated Prices
We did not identify any alternatives
that both maintained consistent
reporting among sponsors leading to
comparable bids, and maximized price
competition.
4. Special Part D Access Rules During
Disasters or Emergencies
We did not consider alternatives to
requiring Part D sponsors to lift ‘‘refill
too soon’’ (RTS) edits in the event of any
imminent or occurring disaster or
emergency that would hinder an
enrollee’s access to covered Part D
drugs. It is important for the well-being
and health of beneficiaries that they be
able to obtain their medications after
disasters strike. Furthermore, given the
complexities of moving large numbers
of people with different health
conditions to safer locations, we also
believed we had no alternative but to
require Part D sponsors to relax RTS
edits when a disaster is imminent and
access to services might be jeopardized
rather than waiting for it to strike.
We are not finalizing this proposal.
5. Drug Categories or Classes of Clinical
Concerns
The critical policy decision was how
broadly or narrowly to establish criteria
and exceptions to those criteria
pursuant to Affordable Care Act
provisions. Broad criteria might easily
encompass many classes of drugs and
significantly increase costs to the Part D
program by eliminating the need for
manufacturers to aggressively rebate
their products for formulary placement.
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00111 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29954
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
Only narrow criteria would limit the
number of categories or classes of
clinical concern receiving additional
protections under the Affordable Care
Act. Similarly, broad exceptions further
limit the products within those
categories or classes of clinical concern
that would receive additional protection
under the Affordable Care Act.
However, we are not codifying the
propose criteria or applying them to the
drug categories and classes of clinical
concern. Thus, this does not apply.
6. Medication Therapy Management
Program (MTM) Under Part D
In the proposed rule, we considered
leaving the maximum number of
multiple chronic diseases a plan may
require for targeted enrollment at three,
but believed this threshold significantly
limited the number of beneficiaries who
qualified for MTM services and was
inconsistent with literature concerning
the relative risk of the combination of
multiple disease states and the need for
access to MTM interventions. Similarly,
we considered other numbers of Part D
drugs less than eight, but again believed
these thresholds decreased access to
MTM services, contributed to
beneficiary confusion, and led to racial
disparities in access to MTM services.
We also considered other cost
thresholds less than $3,000, for
example, $900 or $1,200, which roughly
coincide with cost thresholds achieved
by taking 3 or 4 generic drugs, and we
solicited stakeholder comment on where
the threshold might alternatively be set.
7. Requirement for Applicants or Their
Contracted First Tier, Downstream, or
Related Entities To Have Experience in
the Part D Program Providing Key Part
D Functions
Based on our authority at section
1860D–12(b)(3)(D) of the Act to adopt
additional contract terms that are
necessary and appropriate to administer
the Part D program, we proposed
changes at § 423.504(b)(8)(i) through (iii)
that Part D organizations seeking a new
Medicare contract must have
arrangements in place such that either
the applicant or a contracted entity that
will be performing certain key Part D
functions has at least 1 full benefit year
of experience providing the function for
another Part D plan sponsor. This
proposal ensures that applicants take
advantage of the abundant Part D
industry expertise and experience that
exists today in the development of their
Part D program operations, rather than
relying on technical assistance from
CMS and having their inexperience
place beneficiaries’ access to
prescription drugs at risk. We believe
this provision will have a very minor
savings impact on the federal budget,
based on savings of time and effort (staff
time and contracted auditor time and
resources) that the government would
spend on overseeing the
disproportionate level of problems
experienced by organizations operating
Part D plans without prior Part D
experience. For each inexperienced
organization allowed into the program
in the absence of this proposal, we
would anticipate a savings of 1,000 staff
hours at an average rate of $50 per hour,
for a total of $50,000 in employee time,
plus an additional savings of $200,000
in contractor dollars to conduct an
emergency audit, for a total of $250,000.
In the absence of this proposal, we
would anticipate no more than two such
inexperienced entities beginning Part D
operations per year, for a total annual
savings of $500,000.
The burden associated with this
proposal on industry would be minimal,
with a total estimated number of labor
hours of 3.25 to submit information
during the Part D application process.
Using the same average hourly salary as
previously mentioned, the total cost to
Part D applicants would be $162.50. We
do not believe there are any non-
administrative costs to industry
associated with this proposal, as Part D
applicants are already required to have
arrangements in place to perform the
key Part D functions discussed in our
proposal.
The main anticipated effect from this
proposal is ensuring that only entities
with some experience with Part D in
critically important functional areas are
permitted to offer new Part D contracts,
thus strengthening the Part D program
by enhancing the qualification criteria.
We considered the alternate proposal of
requiring the prior Part D experience to
be tied to specific quality outcomes. We
rejected the alternative because we
believed it added unnecessary
complexity and burden to the process,
and we believe a simple experience
requirement is currently sufficient.
F. Accounting Statement and Table
As required by OMB Circular A–4
(available at http://
www.whitehouse.gov/omb/circulars/
a0004/a-4/pdf), in Table 11 we have
prepared an accounting statement
showing the transfers associated with
the provisions of this final rule for CYs
2015 through 2019.
T
ABLE
11—A
CCOUNTING
S
TATEMENT
: C
LASSIFICATIONS OF
E
STIMATED
T
RANSFERS
F
ROM
C
ALENDAR
Y
EARS
2015
TO
2024
[$ in millions]
Category
Transfers
Discount rate
Period covered
7% 3%
Annualized Monetized Transfers (Federal) ............................................................... ¥$150.27 ¥156.27 CYs 2015–2024
Whom to Whom? ....................................................................................................... Federal Government to MA Organizations and Part D
Sponsors
Note: Monetized figures in 2014 Dollars.
G. Conclusion
We estimate the savings to the federal
government from implementing these
provisions will be $73 million in CY
2015. The savings will increase
annually. In CY 2024, the federal
government savings from implementing
these provisions will be $256 million.
For the entire estimated period, CYs
2015 through 2024, we estimate the total
federal government (Medicare) impact
to result in savings of approximately
$1.615 billion. We note that these
savings do not represent net social
benefits because they consist of transfers
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00112 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29955
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
of value from drug manufacturers,
pharmacies, and incarcerated
individuals to the federal government,
MA organizations, Part D sponsors and
beneficiaries who continue in the
programs
List of Subjects
42 CFR Part 417
Administrative practice and
procedure, Grant programs—health,
Health care, Health insurance, Health
maintenance organizations (HMO), Loan
programs—health, Medicare, and
Reporting and recordkeeping
requirements.
42 CFR Part 422
Administrative practice and
procedure, Health facilities, Health
maintenance organizations (HMO),
Medicare, Penalties, Privacy, and
Reporting and recordkeeping
requirements.
42 CFR Part 423
Administrative practice and
procedure, Emergency medical services,
Health facilities, Health maintenance
organizations (HMO), Health
professionals, Medicare, Penalties,
Privacy, Reporting and recordkeeping
requirements.
42 CFR Part 424
Administrative practice and
procedure, Emergency medical services,
Health facilities, Health maintenance
organizations (HMO), Health
professionals, Medicare, Penalties,
Privacy, Reporting and recordkeeping
requirements.
For the reasons set forth in the
preamble, the Centers for Medicare &
Medicaid Services amends 42 CFR
Chapter IV as follows:
PART 417—HEALTH MAINTENANCE
ORGANIZATION, COMPETITIVE
MEDICAL PLANS, AND HEALTH CARE
PREPAYMENT PLANS
■ 1. The authority citation for part 417
continues to read as follows:
Authority: Secs. 1102 and 1871 of the
Social Security Act (42 U.S.C. 1302 and
1395hh), secs. 1301, 1306, and 1310 of the
Public Health Service Act (42 U.S.C. 300e,
300e–5, and 300e–9), and 31 U.S.C. 9701.
■ 2. Section 417.1 is amended by
revising the definition of ‘‘service area’’
to read as follows:
§ 417.1 Definitions.
* * * * *
Service area means a geographic area,
defined through zip codes, census
tracts, or other geographic
measurements, that is the area, as
determined by CMS, within which the
HMO furnishes basic and supplemental
health services and makes them
available and accessible to all its
enrollees in accordance with
§ 417.106(b). Facilities in which
individuals are incarcerated are not
included in the geographic service area
of an HMO or CMP plan.
* * * * *
■ 3. Section 417.460 is amended by
revising paragraph (b)(2)(i) and adding
paragraphs (f)(1)(i)(A) through (C) to
read as follows:
§ 417.460 Disenrollment of beneficiaries
by an HMO or CMP.
* * * * *
(b) * * *
(2) * * *
(i) Moves out of the HMO’s or CMP’s
geographic service area or is
incarcerated.
* * * * *
(f) * * *
(1) * * *
(i) * * *
(A) Incarceration. The HMO or CMP
must disenroll an individual if the HMO
or CMP establishes, on the basis of
evidence acceptable to CMS, that the
individual is incarcerated and does not
reside in the geographic service area of
the HMO or CMP per § 417.1.
(B) Notification by CMS of
incarceration. When CMS notifies an
HMO or CMP of disenrollment due to
the individual being incarcerated and
not residing in the geographic service
area of the HMO or CMP, as per § 417.1,
the disenrollment is effective the first of
the month following the start of
incarceration, unless otherwise
specified by CMS.
(C) Exception. The exception in
paragraph (f)(2) of this section does not
apply to individuals who are
incarcerated.
* * * * *
PART 422—MEDICARE ADVANTAGE
PROGRAM
■ 4. The authority citation for part 422
continues to read as follows:
Authority: Secs. 1102 and 1871 of the
Social Security Act (42 U.S.C. 1302 and
1395hh).
■ 5. Section 422.2 is amended by—
■ A. Revising the definition of
‘‘Attestation process’’.
■ B. Removing the definition of ‘‘Initial
Validation Contractor (IVC)’’.
■ C. Adding the definition of ‘‘RADV
appeal process’’.
■ D. Removing the definition of ‘‘RADV
payment error calculation appeal
process’’.
■ E. Revising the definition of ‘‘Risk
adjustment data validation (RADV)
audit’’.
■ F. Removing the definition of ‘‘The
one best medical record for the purposes
of Medicare Advantage Risk Adjustment
Validation (RADV)’’.
The revisions and additions read as
follows:
§ 422.2 Definitions.
* * * * *
Attestation process means a CMS-
developed RADV audit-related process
that is part of the medical record review
process that enables MA organizations
undergoing RADV audit to submit CMS-
generated attestations for eligible
medical records with missing or
illegible signatures or credentials. The
purpose of the CMS-generated
attestations is to cure signature and
credential issues. CMS-generated
attestations do not provide an
opportunity for a provider or supplier to
replace a medical record or for a
provider or supplier to attest that a
beneficiary has the medical condition
* * * * *
RADV appeal process means an
administrative process that enables MA
organizations that have undergone
RADV audit to appeal the Secretary’s
medical record review determinations
and the Secretary’s calculation of an MA
organization’s RADV payment error.
* * * * *
Risk adjustment data validation
(RADV) audit means a payment audit of
a MA organization administered by the
Secretary that ensures the integrity and
accuracy of risk adjustment payment
data.
* * * * *
■ 6. Section 422.74 is amended by
revising paragraph (d)(4)(i)(A) and
adding paragraph (d)(4)(v) to read as
follows:
§ 422.74 Disenrollment by the MA
organization.
* * * * *
(d) * * *
(4) * * *
(i) * * *
(A) Out of the MA plan’s service area
or is incarcerated as specified in
paragraph (d)(4)(v) of this section.
* * * * *
(v) Incarceration. (A) The MA
organization must disenroll an
individual if the MA organization
establishes, on the basis of evidence
acceptable to CMS, that the individual
is incarcerated and does not reside in
the service area of the MA plan as
specified at § 422.2 or when notified of
the incarceration by CMS as specified in
paragraph (d)(4)(v)(B) of this section.
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00113 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29956
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
(B) Notification by CMS of
incarceration. When CMS notifies the
MA organization of the disenrollment
due to the individual being incarcerated
and not residing in the service area of
the MA plan as per § 422.2,
disenrollment is effective the first of the
month following the start of
incarceration, unless otherwise
specified by CMS.
* * * * *
■ 7. Section 422.134 is added to subpart
C to read as follows:
§ 422.134 Reward and incentive programs.
(a) General rule. The MA organization
may create one or more programs
consistent with the standards of this
section that provide rewards and
incentives to enrollees in connection
with participation in activities that
focus on promoting improved health,
preventing injuries and illness, and
promoting efficient use of health care
resources.
(b) Non-discrimination. Reward and
incentive programs—
(1) Must not discriminate against
enrollees based on race, national origin,
including limited English proficiency,
gender, disability, chronic disease,
whether a person resides or receives
services in an institutional setting,
frailty, health status or other prohibited
basis;
(2) Must be designed so that all
enrollees are able to earn rewards; and
(3) Are subject to sanctions at
§ 422.752(a)(4).
(c) Requirements. (1) A rewards and
incentives program must —
(i) Be offered in connection with the
entire service or activity;
(ii) Be offered to all eligible members
without discrimination;
(iii) Have a monetary cap as
determined by CMS of a value that may
be expected to impact enrollee behavior
but not exceed the value of the health
related service or activity itself; and
(iv) Otherwise comply with all
relevant fraud and abuse laws,
including, when applicable, the anti-
kickback statute and civil money
penalty prohibiting inducements to
beneficiaries.
(2) Reward and incentive items may
not—
(i) Be offered in the form of cash or
other monetary rebates; or
(ii) Be used to target potential
enrollees.
(3) The MA organization must make
information available to CMS upon
request about the form and manner of
any rewards and incentives programs it
offers and any evaluations of the
effectiveness of such programs.
§ 422.300 [Amended]
■ 8. Section 422.300 is amended by
removing the phrase ‘‘and 1858 of the
Act.’’ and adding in its place the phrase
‘‘1858, and 1128J(d) of the Act.’’
■ 9. Section 422.310 is amended by
revising paragraph (g)(2) and adding
paragraph (g)(3) to read as follows:
§ 422.310 Risk adjustment data.
* * * * *
(g) * * *
(2) After the payment year is
completed, CMS recalculates the risk
factors for affected individuals to
determine if adjustments to payments
are necessary.
(i) Prior to calculation of final risk
factors for a payment year, CMS allows
a reconciliation process to account for
risk adjustment data submitted after the
March deadline until the final risk
adjustment data submission deadline in
the year following the payment year.
(ii) After the final risk adjustment data
submission deadline, which is January
31 of the year following the payment
year, an MA organization can submit
data to correct overpayments but cannot
submit diagnoses for additional
payment.
(3) Submission of corrected risk
adjustment data in accordance with
overpayments after the final risk
adjustment data submission deadline, as
described in paragraph (g)(2) of this
section, must be made as provided in
§ 422.326.
■ 10. Section 422.311 is amended as
follows:
■ A. In paragraph (a), by removing the
phrase ‘‘CMS annually’’ and adding in
its place the phrase ‘‘the Secretary
annually’’.
■ B. In paragraph (b)(2), by removing the
phrase ‘‘to CMS or its contractors’’ and
adding in its place the phrase ‘‘to the
Secretary’’.
■ C. By removing paragraph (b)(3).
■ D. By revising paragraph (c).
The revision reads as follows:
§ 422.311 RADV audit dispute and appeal
processes.
* * * * *
(c) RADV audit appeals. (1) Appeal
rights. MA organizations that do not
agree with their RADV audit results may
appeal.
(2) Issues eligible for RADV appeals.
(i) General rules. MA organizations may
appeal RADV medical record review
determinations and the Secretary’s
RADV payment error calculation. In
order to be eligible for RADV appeal,
MA organizations must adhere to the
following:
(A) Established RADV audit
procedures and requirements.
(B) RADV appeals procedures and
requirements.
(ii) Failure to follow RADV rules.
Failure to follow the Secretary’s RADV
audit procedures and requirements and
the Secretary’s RADV appeals
procedures and requirements will
render the MA organization’s request for
appeal invalid.
(iii) RADV appeal rules. The MA
organization’s written request for
medical record review determination
appeal must specify the following:
(A) The audited HCC(s) that the
Secretary identified as being in error.
(B) A justification in support of the
audited HCC selected for appeal.
(iv) Number of medical records
eligible for appeal. For each audited
HCC, MA organizations may appeal one
medical record that has undergone
RADV review. If an attestation was
submitted to cure a signature or
credential-related error, the attestation
may be included in the HCC appeal.
(v) Selection of medical record for
appeal. The MA organization must
select the medical record that undergoes
appeal.
(vi) Written request for RADV
payment error calculation appeal. The
written request for RADV payment error
calculation appeal must clearly specify
the following:
(A) The MA organization’s own RADV
payment error calculation.
(B) Where the Secretary’s RADV
payment error calculation was
erroneous.
(3) Issues ineligible for RADV appeals.
(i) MA organizations’ request for appeal
may not include HCCs, medical records
or other documents beyond the audited
HCC, RADV-reviewed medical record,
and any accompanying attestation that
the MA organization chooses for appeal.
(ii) MA organizations may not appeal
the Secretary’s medical record review
determination methodology or RADV
payment error calculation methodology.
(iii) As part of the RADV payment
error calculation appeal— MA
organizations may not appeal RADV
medical record review-related errors.
(iv) MA organizations may not appeal
RADV errors that result from an MA
organization’s failure to submit a
medical record.
(4) Burden of proof. The MA
organization bears the burden of proof
by a preponderance of the evidence in
demonstrating that the Secretary’s
medical record review determination(s)
or payment error calculation was
incorrect.
(5) Manner and timing of a request for
RADV appeal. (i) At the time the
Secretary issues its RADV audit report,
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00114 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29957
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
the Secretary notifies audited MA
organizations of the following:
(A) That they may appeal RADV HCC
errors that are eligible for medical
record review determination appeal.
(B) That they may appeal the
Secretary’s RADV payment error
calculation.
(ii) MA organizations have 60 days
from date of issuance of the RADV audit
report to file a written request with CMS
for RADV appeal. This request for
RADV appeal must specify one of the
following:
(A) Whether the MA organization
requests medical record review
determination appeal, the issues with
which the MA organization disagrees,
and the reasons for the disagreements.
(B) Whether the MA organization
requests RADV payment error
calculation appeal, the issues with
which the MA organization disagrees,
and the reasons for the disagreements.
(C) Whether the MA organization
requests both medical record review
determination appeal and RADV
payment error calculation appeal, the
issues with which the MA organization
disagrees, and the reasons for the
disagreements.
(iii) For MA organizations that appeal
both medical record review
determination appeal and RADV
payment error calculation appeal:
(A) The Secretary adjudicates the
request for RADV payment error
calculation following conclusion of
reconsideration of the MA
organization’s request for medical
record review determination appeal.
(B) An MA organization’s request for
appeal of its RADV payment error
calculation will not be adjudicated until
appeals of RADV medical record review
determinations filed by the MA
organization have been completed and
the decisions are final for that stage of
appeal.
(6) Reconsideration stage. (i) Written
request for medical record review
reconsideration. A MA organization’s
written request for medical record
review determination reconsideration
must specify the following:
(A) The audited HCC that the
Secretary identified as being in error
that the MA organization wishes to
appeal.
(B) A justification in support of the
audited HCC chosen for appeal.
(ii) Written request for payment error
calculation. The MA organization’s
written request for payment error
calculation reconsideration—
(A) Must include the MA
organization’s own RADV payment error
calculation that clearly specifies where
the Secretary’s RADV payment error
calculation was erroneous; and
(B) May include additional
documentary evidence pertaining to the
calculation of the payment error that the
MA organization wishes the
reconsideration official to consider.
(iii) Conduct of the reconsideration.
(A) For medical record review
determination reconsideration, a
medical record review professional who
was not involved in the initial medical
record review determination of the
disputed audited HCCs does the
following:
(1) Reviews the medical record and
accompanying dispute justification.
(2) Reconsiders the initial audited
medical record review determination.
(B) For payment error calculation
reconsideration, CMS ensures that a
third party not involved in the initial
RADV payment error calculation does
the following:
(1) Reviews the Secretary’s RADV
payment error calculation.
(2) Reviews the MA organization’s
RADV payment error calculation;
(3) Recalculates the payment error in
accordance with CMS’s RADV payment
error calculation procedures.
(iv) Effect of the reconsideration
official’s decision. (A) The
reconsideration official issues a written
reconsideration decision to the MA
organization.
(B) The reconsideration official’s
decision is final unless the MA
organization disagrees with the
reconsideration official’s decision.
(C) If the MA organization disagrees
with the reconsideration official’s
decision, they may request a hearing in
accordance with paragraph (c)(7) of this
section.
(7) Hearing stage. (i) Errors eligible for
hearing. At the time the reconsideration
official issues his or her reconsideration
determination to the MA organization,
the reconsideration official notifies the
MA organization of any RADV HCC
errors or payment error-calculations that
are eligible for RADV hearing.
(ii) General hearing rules. A MA
organization that requests a RADV
hearing must do so in writing in
accordance with procedures established
by CMS.
(iii) Written request for hearing. The
written request for a hearing must be
filed with the Hearing Officer within 60
days of the date the MA organization
receives the reconsideration officer’s
written reconsideration decision.
(A) If the MA organization appeals
medical record review reconsideration
determination, the written request for
RADV hearing must—
(1) Include a copy of the written
decision of the reconsideration official;
(2) Specify the audited HCCs that the
reconsideration official confirmed as
being in error; and
(3) Specify a justification why the MA
organization disputes the
reconsideration official’s determination.
(B) If the MA organization appeals the
RADV payment error calculation
reconsideration determination, the
written request for RADV hearing must
include the following:
(1) A copy of the written decision of
the reconsideration official.
(2) The MA organization’s own RADV
payment error calculation that clearly
specifies where the Secretary’s payment
error calculation was erroneous.
(iv) Designation of hearing officer. A
hearing officer will conduct the RADV
hearing.
(v) Disqualification of the hearing
officer. (A) A hearing officer may not
conduct a hearing in a case in which he
or she is prejudiced or partial to any
party or has any interest in the matter
pending for decision.
(B) A party to the hearing who objects
to the designated hearing officer must
notify that officer in writing at the
earliest opportunity.
(C) The hearing officer must consider
the objections, and may, at his or her
discretion, either proceed with the
hearing or withdraw.
(D) If the hearing officer withdraws,
another hearing officer conducts the
hearing.
(E) If the hearing officer does not
withdraw, the objecting party may, after
the hearing, present objections and
request that the officer’s decision be
revised or a new hearing be held before
another hearing officer. The objections
must be submitted in writing to the
Secretary.
(vi) Hearing Officer review. The
hearing officer reviews the following:
(A) For a medical record review
determination appeal, the hearing
officer reviews all of the following:
(1) The RADV-reviewed medical
record and any accompanying
attestation that the MA organization
selected for review.
(2) The reconsideration official’s
written determination.
(3) The written brief submitted by the
MA organization or the Secretary in
response to the reconsideration official’s
determination.
(B) For a payment error calculation
appeal, the hearing officer reviews all of
the following:
(1) The reconsideration official’s
written determination.
(2) Briefs addressing the
reconsideration decision.
(vii) Hearing procedures. (A)
Authority of the Hearing Officer. The
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00115 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29958
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
hearing officer has full power to make
rules and establish procedures,
consistent with the law, regulations, and
the Secretary rulings. These powers
include the authority to dismiss the
appeal with prejudice and take any
other action which the hearing officer
considers appropriate, including for
failure to comply with such rules and
procedures.
(B) The hearing is on the record. (1)
Except as specified in paragraph
(c)(viii)(B)(2) of this section, the hearing
officer is limited to the review of the
record.
(2)(i) Subject to the hearing officer’s
full discretion, the parties may request
a live or telephonic hearing regarding
some or all of the disputed medical
records.
(ii) The hearing officer may, on his or
her own-motion, schedule a live or
telephonic hearing.
(3) The record is comprised of the
following:
(i) Written decisions described at
paragraphs (c)(6)(iv) and (7)(vi) of this
section.
(ii) Written briefs from the MA
organization explaining why they
believe the reconsideration official’s
determination was incorrect.
(iii) The Secretary’s optional brief that
responds to the MA organization’s
brief—
(4) The hearing officer neither
receives testimony nor accepts any new
evidence that is not part of the record.
(5) Either the MA organization or the
Secretary may ask the hearing officer to
rule on a motion for summary judgment.
(viii) Hearing Officer decision. The
hearing officer decides whether to
uphold or overturn the reconsideration
official’s decision, and sends a written
determination to CMS and the MA
organization, explaining the basis for
the decision.
(ix) Computations based on hearing
decision. (A) Once the hearing officer’s
decision is considered final in
accordance with paragraph (c)(7)(x) of
this section, a third party not involved
in the initial RADV payment error
calculation recalculates the MA
organization’s RADV payment error and
issues a new RADV audit report to the
appellant MA organization and CMS.
(B) For MA organizations appealing
the RADV error calculation only, a third
party not involved in the initial RADV
payment error calculation recalculates
the MA organization’s RADV payment
error and issues a new RADV audit
report to the appellant MA organization
and CMS.
(x) Effect of the Hearing Officer’s
decision. The hearing officer’s decision
is final unless the decision is reversed
or modified by the CMS Administrator.
(8) CMS Administrator review stage.
(i) A request for CMS Administrator
review must be made in writing and
filed with the CMS Administrator.
(ii) CMS or a MA organization that
has received a hearing officer’s decision
and requests review by the CMS
Administrator must do so within 60
days of receipt of the hearing officer’s
decision.
(iii) After receiving a request for
review, the CMS Administrator has the
discretion to elect to review the hearing
officer’s decision or to decline to review
the hearing officer’s decision.
(iv) If the CMS Administrator elects to
review the hearing decision—
(A) The CMS Administrator
acknowledges the decision to review the
hearing decision in writing and notifies
CMS and the MA organization of their
right to submit comments within 15
days of the date of the notification; and
(B) The CMS Administrator is limited
to the review of the record. The record
is comprised of the following:
(1) The record is comprised of
documents described at paragraph
(c)(7)(vii)(B)(3) of this section.
(2) The hearing record.
(3) Written arguments from the MA
organization or CMS explaining why
either or both parties believe the hearing
officer’s determination was correct or
incorrect.
(C) The CMS Administrator reviews
the record and determines whether the
hearing officer’s determination should
be upheld, reversed, or modified.
(v) The CMS Administrator renders
his or her final decision in writing to the
parties within 60 days of acknowledging
his or her decision to review the hearing
officer’s decision.
(vi) The decision of the hearing officer
is final if the CMS Administrator—
(A) Declines to review the hearing
officer’s decision; or
(B) Does not make a decision within
60 days.
■ 11. Section 422.326 is added to
subpart G to read as follows:
§ 422.326 Reporting and returning of
overpayments.
(a) Terminology. For purposes of this
section—
Applicable reconciliation occurs on
the date of the annual final deadline for
risk adjustment data submission
described at § 422.310(g), which is
announced by CMS each year.
Funds means any payment that an
MA organization has received that is
based on data submitted by the MA
organization to CMS for payment
purposes, including § 422.308(f) and
§ 422.310.
Overpayment means any funds that
an MA organization has received or
retained under title XVIII of the Act to
which the MA organization, after
applicable reconciliation, is not entitled
under such title.
(b) General rule. If an MA
organization has identified that it has
received an overpayment, the MA
organization must report and return that
overpayment in the form and manner
set forth in this section.
(c) Identified overpayment. The MA
organization has identified an
overpayment when the MA organization
has determined, or should have
determined through the exercise of
reasonable diligence, that the MA
organization has received an
overpayment.
(d) Reporting and returning of an
overpayment. An MA organization must
report and return any overpayment it
received no later than 60 days after the
date on which it identified it received
an overpayment, unless otherwise
directed by CMS for purposes of
§ 422.311.
(1) Reporting. An MA organization
must notify CMS, of the amount and
reason for the overpayment, using a
notification process determined by
CMS.
(2) Returning. An MA organization
must return identified overpayments in
a manner specified by CMS.
(e) Enforcement. Any overpayment
retained by an MA organization is an
obligation under 31 U.S.C. 3729(b)(3) if
not reported and returned in accordance
with paragraph (d) of this section.
(f) Look-back period. An MA
organization must report and return any
overpayment identified for the 6 most
recent completed payment years.
■ 12. Section 422.503 is amended by –
■ A. Adding paragraph (b)(4)(vi)(C)(3).
■ B. Adding reserved paragraph
(b)(4)(vi)(G)(4).
■ C. Revising paragraph (b)(4)(vi)(G)(5).
The revisions and additions are as
follows:
§ 422.503 General provisions.
* * * * *
(b) * * *
(4) * * *
(vi) * * *
(C) * * *
(3) An MA organization must require
all of its first tier, downstream, and
related entities to take the CMS training
and accept the certificate of completion
of the CMS training as satisfaction of
this requirement. MA organizations are
prohibited from developing and
implementing their own training or
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00116 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29959
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
providing supplemental training
materials to fulfill this requirement.
* * * * *
(G) * * *
(5)(i) Not accept, or share, a corporate
parent organization owning a
controlling interest in an entity that
accepts new enrollees under a section
1876 reasonable cost contract in any
area in which it seeks to offer an MA
plan.
(ii) Not accept, as either the parent
organization owning a controlling
interest of, or subsidiary of, an entity
that accepts new enrollees under a
section 1876 reasonable cost contract in
any area in which it seeks to offer an
MA plan.
* * * * *
■ 13. Amend § 422.504 by:
■ A. Revising paragraph (i)(2)(i).
■ B. Redesignating paragraph (i)(2)(ii) as
(i)(2)(iv).
■ C. Adding new paragraph (i)(2)(ii) and
paragraphs (i)(2)(iii) and (1)(5).
The revisions and additions read as
follows:
§ 422.504 Contract provisions.
* * * * *
(i)* * *
(2)* * *
(i) HHS, the Comptroller General, or
their designees have the right to audit,
evaluate, collect, and inspect any books,
contracts, computer or other electronic
systems, including medical records and
documentation of the first tier,
downstream, and entities related to
CMS’ contract with the MA
organization.
(ii) HHS, the Comptroller General, or
their designees have the right to audit,
evaluate, collect, and inspect any
records under paragraph (i)(2)(i) of this
section directly from any first tier,
downstream, or related entity.
(iii) For records subject to review
under paragraph (i)(2)(ii) of this section,
except in exceptional circumstances,
CMS will provide notification to the MA
organization that a direct request for
information has been initiated.
* * * * *
(l) * * *
(5) Certification of accuracy of data
for overpayments. The CEO, CFO, or
COO must certify (based on best
knowledge, information, and belief) that
the information provided for purposes
of reporting and returning of
overpayments under § 422.326 is
accurate, complete, and truthful.
* * * * *
■ 14. Amend § 422.510 as follows;
■ A. By redesigating paragraphs (a)(4)
through (15) as paragraphs (a)(4)(i)
through (xii).
■ B. By adding new paragraph (a)(4)
introductory text.
■ C. In newly redesignated paragraphs
(a)(4)(ii), (v), (vi), and (viii) by removing
the term ‘‘fails’’ and adding in its place
the term ‘‘failed’’.
■ D. In newly redesignated paragraphs
(a)(4)(iii), (iv), (vii), (ix), and (x), by
removing the term ‘‘Fails’’ and adding
in its place the term ‘‘Failed’’.
■ E. By revising newly redesignated
paragraph (a)(4)(xii).
■ F. By revising paragraph (b)(1)(i)
through (iii) and the heading for
paragraph (b)(2).
■ G. In paragraph (b)(2)(i)(C), by
removing the cross-reference ‘‘(a)(4) of
this section’’ and adding in its place the
cross reference ‘‘(a)(4)(i) of this section’’.
■ H. In paragraph (c)(2)(iii), by
removing the cross reference ‘‘(a)(4) of
this section’’ and adding in its place the
cross reference ‘‘(a)(4)(i) of this section’’.
The additions and revisions read as
follows:
§ 422.510 Termination of contract by CMS.
* * * * *
(a) * * *
(4) CMS may make a determination
under paragraph (a)(1), (2), or (3) of this
section if the MA organization has had
one or more of the following occur:
* * * * *
(xii) Has failed to report MLR data in
a timely and accurate manner in
accordance with § 422.2460 or that any
MLR data required by this subpart is
found to be materially incorrect or
fraudulent.
* * * * *
(b) * * *
(1) * * *
(i) CMS notifies the MA organization
in writing at least 45 calendar days
before the intended date of the
termination.
(ii) The MA organization notifies its
Medicare enrollees of the termination by
mail at least 30 calendar days before the
effective date of the termination.
(iii) The MA organization notifies the
general public of the termination at least
30 calendar days before the effective
date of the termination by releasing a
press statement to news media serving
the affected community or county and
posting the press statement prominently
on the organization’s Web site.
(2) Immediate termination of contract
by CMS.***
* * * * *
■ 15. Amend § 422.752 by adding
paragraphs (a)(9) through (12) and
revising paragraphs (c)(1) and (c)(2)(ii)
to read as follows:
§ 422.752 Basis for imposing intermediate
sanctions and civil money penalties.
(a) * * *
(9) Except as provided under § 423.34
of this chapter, enrolls an individual in
any plan under this part without the
prior consent of the individual or the
designee of the individual.
(10) Transfers an individual enrolled
under this part from one plan to another
without the prior consent of the
individual or the designee of the
individual or solely for the purpose of
earning a commission.
(11) Fails to comply with marketing
restrictions described in subpart V or
applicable implementing guidance.
(12) Employs or contracts with any
individual, agent, provider, supplier or
entity who engages in the conduct
described in paragraphs (a)(1) through
(11) of this section.
* * * * *
(c) * * *
(1) CMS. In addition to, or in place of,
any intermediate sanctions, CMS may
impose civil money penalties in the
amounts specified in the following:
(i) Section 422.760(b) for any of the
determinations at § 422.510(a), except
§ 422.510(a)(4)(i).
(ii) Section 422.760(c) for any of the
determinations at § 422.752(a) except
§ 422.752(a)(5).
(2) * * *
(ii) Determinations made under
§ 422.510(a)(4)(i).
■ 16. Amend § 422.756 as follows:
■ A. In paragraph (a)(2), by removing
the phrase ‘‘days from receipt’’ and
adding in its place ‘‘days after receipt’’
and by removing the phrase ‘‘considers
receipt of notice’’ and adding in its
place ‘‘considers receipt of the notice’’.
■ B. In paragraph (b)(4), by removing the
cross-reference ’’§ 423.660 through
§ 423.684 of this part.’’ and adding in its
place ‘‘Subpart N of this part.’’.
■ C. In paragraph (c)(3)(ii) introductory
text, by removing the phrase ‘‘In
instances where marketing or
enrollment or both intermediate
sanctions have been imposed,’’ and
adding in its place the phrase ‘‘In
instances where intermediate sanctions
have been imposed,’’.
■ D. Adding paragraph (c)(3)(ii)(C).
■ E. Revising paragraph (d).
The addition and revision read as
follows:
§ 422.756 Procedures for imposing
intermediate sanctions and civil money
penalties.
(c) * * *
(3) * * *
(ii) * * *
(C) During the limited time period,
sanctioned sponsoring organizations
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00117 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29960
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
offering Part D plans under the
benchmark that would normally
participate in the annual and monthly
auto enrollment process for enrollees
receiving the low income subsidy will
not be allowed to receive or process
these types of enrollments.
* * * * *
(d) Non-renewal or termination by
CMS. In addition to or as an alternative
to the sanctions described in § 422.750,
CMS may—
(1) Decline to authorize the renewal of
an organization’s contract in accordance
with § 422.506(b); or
(2) Terminate the contract in
accordance with § 422.510.
* * * * *
■ 17. Amend § 422.760 by revising
paragraph (a)(3) and the heading of
paragraph (b) and adding paragraph (c)
to read as follows:
§ 422.760 Determinations regarding the
amount of civil money penalties and
assessment imposed by CMS.
(a) * * *
(3) The adverse effect to enrollees
which resulted or could have resulted
from the conduct of MA organization;
* * * * *
(b) Amount of penalty imposed by
CMS. ***
* * * * *
(c) Amount of penalty imposed by
CMS or OIG. CMS or the OIG may
impose civil money penalties in the
following amounts for a determination
made under § 422.752(a):
(1) Civil money penalties of not more
than $25,000 for each determination
made.
(2) With respect to a determination
made under § 422.752(a)(4) or (a)(5)(i),
not more than $100,000 for each such
determination, except with respect to a
determination made under
§ 422.752(a)(5), an assessment of not
more than the amount claimed by such
plan or MA organization based upon the
misrepresentation or falsified
information involved.
(3) Plus with respect to a
determination made under
§ 422.752(a)(2), double the excess
amount charged in violation of such
paragraph (and the excess amount
charged must be deducted from the
penalty and returned to the individual
concerned).
(4) Plus with respect to a
determination made under
§ 422.752(a)(4), $15,000 for each
individual not enrolled as a result of the
practice involved.
■ 18. Amend § 422.1016 by revising the
first sentence of paragraph (b)(1) to read
as follows:
§ 422.1016 Filing of briefs with the
Administrative Law Judge or Departmental
Appeals Board, and opportunity for
rebuttal.
* * * * *
(b) * * *
(1) The other party will have 20
calendar days from the date of mailing
or in person filing to submit any rebuttal
statement or additional evidence.* * *
* * * * *
■ 19. Amend § 422.1020 by revising
paragraph (a)(2) to read as follows:
§ 422.1020 Request for hearing.
(a) * * *
(2) The MA organization or its legal
representative or other authorized
official must file the request, in writing,
to the appropriate Departmental
Appeals Board office, with a copy to
CMS, within 60 calendar days after
receipt of the notice of initial
determination, to request a hearing
before an ALJ to appeal any
determination by CMS to impose a civil
money penalty.
* * * * *
■ 20. Amend § 422.2274 by:
■ A. Revising the introductory text.
■ B. Redesignating paragraphs (a)
through (f) as (b) through (g).
■ C. Adding new paragraph (a).
■ D. Revising newly redesignated
paragraph (b).
■ E. Adding paragraph (h).
The revisions and addition read as
follows:
§ 422.2274 Broker and agent requirements.
If an MA organization uses agents and
brokers to sell its Medicare plans, the
following requirements in this section
are applicable.
(a) Definitions. For purposes of this
section, the following definitions are
applicable:
Compensation (1) Includes monetary
or non-monetary remuneration of any
kind relating to the sale or renewal of a
policy including, but not limited to—
(i) Commissions;
(ii) Bonuses;
(iii) Gifts;
(iv) Prizes or Awards; or
(v) Referral or Finder fees.
(2) Does not include—
(i) Payment of fees to comply with
State appointment laws, training,
certification, and testing costs;
(ii) Reimbursement for mileage to, and
from, appointments with beneficiaries;
or
(iii) Reimbursement for actual costs
associated with beneficiary sales
appointments such as venue rent,
snacks, and materials.
Like plan type means one of the
following:
(1) PDP replaced with another PDP.
(2) MA or MA–PD replaced with
another MA or MA–PD.
(3) Cost plan replaced with another
cost plan.
Unlike plan type means one of the
following:
(1) PDP replaced with an MA–PD or
an MA–PD replaced with a PDP.
(2) PDP replaced with a cost plan or
a cost plan replaced with a PDP.
(3) MA–PD replaced with a cost plan
or a cost plan replaced with an MA–PD.
Plan year means the year beginning
January 1 and ending December 31.
Renewal year means all years
following the initial enrollment year in
a like plan type.
(b) Compensation rules. An MA
organization must compensate
independent brokers and agents, if
compensation is paid, only according to
the following rules in this section.
(1) Compensation amounts. (i) For an
initial year enrollment of a Medicare
beneficiary into an MA plan, the
compensation must be at or below the
fair market value of such services,
published annually as a cut-off amount
by CMS.
(ii) For renewal years, compensation
may be up to 50 percent of the current
fair market value cut-off amounts
published annually by CMS.
(iii) If the MA organization contracts
with a third party entity such as a Field
Marketing Organization or similar type
entity to sell its insurance products, or
perform services (for example, training,
customer service, or agent
recruitment)—
(A) The total amount paid by the MA
organization to the third party and its
agents for enrollment of a beneficiary
into a plan, if any, must be made in
accordance with paragraph (b)(1) of this
section; and
(B) The amount paid to the third party
for services other than selling insurance
products, if any, must be fair-market
value and must not exceed an amount
that is commensurate with the amounts
paid by the MA organization to a third
party for similar services during each of
the previous 2 years.
(2) Aggregate compensation. (i) An
entity must not provide aggregate
compensation to its agents or brokers
greater than the renewal compensation
payable by the replacing plan on
renewal policies if an existing policy is
replaced with a like plan at any time.
(ii) An agent or broker must not
receive aggregate compensation greater
than the renewal compensation payable
by the replacing plan on renewal
policies if an existing policy is replaced
with a like plan type at any time.
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00118 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29961
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
(iii) The initial compensation is paid
for replacements between unlike plan
types.
(3) Compensation payment and
payment recovery. (i) Compensation
may only be paid for the enrollee’s
months of enrollment during a plan
year.
(ii)(A) Subject to paragraph (b)(3)(iii)
of this section, compensation payments
may be made at one time for the entire
current plan year or in installments
throughout the year.
(B) Compensation may not be paid
until January 1 of the enrollment year
and, if paid at all, must be paid in full
by December 31 of the enrollment year.
(iii) When a beneficiary disenrolls
from an MA plan, compensation paid to
agents and brokers must be recovered
for those months of the plan year for
which the beneficiary is not enrolled.
For disenrollments occurring within the
first 3 months, the entire compensation
must be recovered unless CMS
determines that recoupment is not in
the best interests of the Medicare
program.
(4) Compensation structure. (i) The
MA organization must establish a
compensation structure for new and
replacement enrollments and renewals
effective in a given plan year.
Compensation structures must be in
place by the beginning of the plan
marketing period, October 1.
(ii) Compensation structures must be
available upon CMS request including
for audits, investigations, and to resolve
complaints.
* * * * *
(h) Finder’s (referral) fees. Finder’s
(referral) fees paid to all agents and
brokers—
(1) May not exceed an amount that
CMS determines could reasonably be
expected to provide financial incentive
for an agent or broker to recommend or
enroll a beneficiary into a plan that is
not the most appropriate to meet his or
her needs; and
(2) Must be included in the total
compensation not to exceed the fair
market value for that calendar year.
Subpart Y—[Reserved]
■ 21. Part 422 is amended by adding
reserved subpart Y.
■ 22. Part 422 is amended by adding
subpart Z to read as follows:
Subpart Z—Part C Recovery Audit
Contractor Appeals Process
Sec.
422.2600 Payment appeals.
422.2605 Request for reconsideration.
422.2610 Hearing official review.
422.2615 Review by the Administrator.
Subpart Z—Part C Recovery Audit
Contractor Appeals Process
§ 422.2600 Payment appeals.
If the Part C RAC did not apply its
stated payment methodology correctly,
an MA organization may appeal the
findings of the applied methodology.
The payment methodology itself is not
subject to appeal.
§ 422.2605 Request for reconsideration.
(a) Time for filing a request. The
request for reconsideration must be filed
with the designated independent
reviewer within 60 calendar days from
the date of the demand letter received
by the MA organization.
(b) Content of request. (1) The request
for reconsideration must be in writing
and specify the findings or issues with
which the MA organization disagrees.
(2) The MA organization must include
with its request all supporting
documentary evidence it wishes the
independent reviewer to consider.
(i) This material must be submitted in
the format requested by CMS.
(ii) Documentation, evidence, or
substantiation submitted after the filing
of the reconsideration request will not
be considered.
(c) CMS rebuttal. CMS may file a
rebuttal to the MA organization’s
reconsideration request.
(1) The rebuttal must be submitted
within 30 calendar days of the review
entity’s notification to CMS that it has
received the MA organization’s
reconsideration request.
(2) CMS sends its rebuttal to the MA
organization at the same time it is
submitted to the independent reviewer.
(d) Review entity. An independent
reviewer conducts the reconsideration.
The independent reviewer reviews the
demand for repayment, the evidence
and findings upon which it was based
and any supporting documentation that
the MA organization or CMS submitted
in accordance with this section.
(e) Notification of decision. The
independent reviewer informs the CMS
and the MA organization of its decision
in writing.
(f) Effect of decision. A
reconsideration decision is final and
binding unless the MA organization
requests a hearing official review in
accordance with § 422.2610.
(g) Right to hearing official review. An
MA organization that is dissatisfied
with the independent reviewer’s
reconsideration decision is entitled to a
hearing official review as provided in
§ 422.2610.
§ 422.2610 Hearing official review.
(a) Time for filing a request. A MA
organization must file with CMS a
request for a hearing official review
within 30 calendar days from the date
of the independent reviewer’s issuance
of a reconsideration determination.
(b) Content of the request. (1) The
request must be in writing and must
specify the findings or issues in the
reconsideration decision with which the
MA organization disagrees and the
reasons for the disagreements.
(2) The MA organization must submit
with its request all supporting
documentation, evidence, and
substantiation that it wants to be
considered.
(3) No new evidence may be
submitted.
(4) Documentation, evidence, or
substantiation submitted after the filing
of the request will not be considered.
(c) CMS rebuttal. CMS may file a
rebuttal to the MA organization’s
hearing official review request.
(1) The rebuttal must be submitted
within 30 calendar days of the MA
organization’s submission of its hearing
official review request.
(2) CMS sends its rebuttal to the MA
organization at the same time it is
submitted to the hearing official.
(d) Conducting a review. A CMS-
designated hearing official conducts the
hearing on the record.
(1) The hearing is not to be conducted
live or via telephone unless the hearing
official, in his or her sole discretion,
requests a live or telephonic hearing.
(2) In all cases, the hearing official’s
review is limited to information that
meets one or more of the following:
(i) The Part C RAC used in making its
determinations.
(ii) The independent reviewer used in
making its determinations.
(iii) The MA organization submits
with its hearing request.
(iv) CMS submits in accordance with
paragraph (c) of this section.
(3) Neither the MA organization nor
CMS may submit new evidence.
(e) Hearing official decision. The CMS
hearing official decides the case within
60 days and sends a written decision to
the MA organization and CMS,
explaining the basis for the decision.
(f) Effect of hearing official decision.
The hearing official’s decision is final
and binding, unless the decision is
reversed or modified by the CMS
Administrator in accordance with
§ 422.2615.
§ 422.2615 Review by the Administrator.
(a) Request for review by
Administrator. If an MA organization is
dissatisfied with the hearing official’s
decision, it may request that the CMS
Administrator review the decision.
(1) The request must be filed with the
CMS Administrator within 30 calendar
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00119 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29962
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
days of the date of the hearing official’s
decision.
(2) The request must provide evidence
or reasons to substantiate the request.
(b) Content of request. The MA
organization must submit with its
request all supporting documentation,
evidence, and substantiation that it
wants to be considered.
(1) Documentation, evidence, or
substantiation submitted after the filing
of the request will not be considered.
(2) Neither the MA organization, nor
CMS may submit new evidence.
(c) Discretionary review. After
receiving a request for review, the CMS
Administrator has the discretion to
review the hearing official’s decision in
accordance with paragraph (e) of this
section or to decline to review said
decision.
(d) Notification of decision whether to
review. The Administrator notifies the
MA organization within 45 days of
receiving the MA organization’s hearing
request of whether he or she intends to
review the hearing official’s decision.
(1) If the Administrator agrees to
review the hearing official’s decision,
CMS may file a rebuttal statement
within 30 days of the Administrator’s
notice to the plan that the request for
review has been accepted. CMS sends
its rebuttal statement to the plan at the
same time it is submitted to the
Administrator.
(2) If the CMS Administrator declines
to review the hearing official’s decision,
the hearing official’s decision is final
and binding.
(e) CMS Administrator’s review. If the
CMS Administrator agrees to review the
hearing official’s decision, he or she
determines, based upon this decision,
the hearing record, and any arguments
submitted by the MA organization or
CMS in accordance with this section,
whether the determination should be
upheld, reversed, or modified. The
Administrator furnishes a written
decision, which is final and binding, to
the MA organization and to CMS.
PART 423—MEDICARE PROGRAM;
MEDICARE PRESCRIPTION DRUG
PROGRAM
■ 23. The authority citation for part 423
continues to read as follows:
Authority: Secs. 1102, 1860D–1 through
1860D–42, and 1871 of the Social Security
Act (42 U.S.C. 1302, 1395w–101 through
1395w–152, and 1395hh).
■ 24. Amend § 423.1 by adding new
references in numerical order to
paragraph (a)(1) to read as follows:
§ 423.1 Basis and scope.
(a) * * *
(1) * * *
1128J(d). Reporting and Returning of
Overpayments.
* * * * *
1860D–14A. Medicare coverage gap
discount program.
* * * * *
1860D–43. Condition for coverage of
drugs under this part.
* * * * *
■ 25. Amend § 423.44 by adding
paragraphs (d)(5)(iii) and (iv) to read as
follows:
§ 423.44 Involuntary disenrollment from
Part D coverage.
* * * * *
(d) * * *
(5) * * *
(iii) Incarceration. The PDP must
disenroll an individual if the PDP
establishes, on the basis of evidence
acceptable to CMS, that the individual
is incarcerated and does not reside in
the service area of the PDP as specified
at § 423.4 or when notified of an
incarceration by CMS as specified in
paragraph (d)(5)(iv) of this section.
(iv) Notification by CMS of
incarceration. When CMS notifies the
PDP of the disenrollment due to the
individual being incarcerated and not
residing in the service area of the PDP
as per § 423.4, disenrollment is effective
the first of the month following the start
of incarceration, unless otherwise
specified by CMS.
* * * * *
■ 26. In § 423.100, amend the definition
of ‘‘Part D drug’’ as follows:
■ A. By adding paragraph (1)(vii).
■ B. By revising paragraph (2)
introductory text.
■ C. In paragraph (2)(i), by removing ‘‘;
and’’ and adding in its place ‘‘.’’.
■ D. In paragraph (2)(ii), by removing
the phrase ‘‘; barbiturates when used to
treat epilepsy, cancer, or a chronic
mental health disorder; and
benzodiazepines.’’ and adding in its
place ‘‘.’’
■ E. By adding paragraph (2)(iii).
The additions read as follows:
§ 423.100 Definitions
* * * * *
Part D drug ***
(1) * * *
(vii) A combination product approved
and regulated by the FDA as a drug,
vaccine, or biologic described in
paragraphs (1)(i), (ii), (iii), or (v) of this
definition.
(2) Does not include any of the
following:
* * * * *
(iii) Medical foods, defined as a food
that is formulated to be consumed or
administered enterally under the
supervision of a physician and which is
intended for the specific dietary
management of a disease or condition
for which distinctive nutritional
requirements, based on recognized
scientific principles, are established by
medical evaluation, and that are not
regulated as drugs under section 505 of
the Federal Food, Drug, and Cosmetic
Act.
* * * * *
■ 27. Section 423.100 is further
amended, effective January 1, 2016, by
revising the definition of ‘‘Negotiated
prices’’ to read as follows:
§ 423.100 Definitions
* * * * *
Negotiated prices means prices for
covered Part D drugs that meet all of the
following:
(1) The Part D sponsor (or other
intermediary contracting organization)
and the network dispensing pharmacy
or other network dispensing provider
have negotiated as the amount such
network entity will receive, in total, for
a particular drug.
(2) Are inclusive of all price
concessions from network pharmacies
except those contingent price
concessions that cannot reasonably be
determined at the point-of-sale; and
(3) Include any dispensing fees; but
(4) Excludes additional contingent
amounts, such as incentive fees, if these
amounts increase prices and cannot
reasonably be determined at the point-
of-sale.
(5) Must not be rebated back to the
Part D sponsor (or other intermediary
contracting organization) in full or in
part.
* * * * *
■ 28. Amend 423.120 by:
■ A. Revising paragraph (b)(2)(v).
■ B. Adding paragraph (b)(3)(vi).
■ C. Adding paragraph (c)(5)
introductory text.
■ D. Adding paragraph (c)(6).
The revisions and additions read as
follows:
§ 423.120 Access to covered Part D drugs.
* * * * *
(b) * * *
(2) * * *
(v) Until such time as there are
established, through notice and
comment rulemaking, criteria to
identify, as appropriate, categories and
classes of clinical concern, the
categories and classes of clinical
concern are as specified in section
1860D–4(b)(3)(G)(iv) of the Act.
* * * * *
(3) * * *
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00120 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29963
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
(vi) A Part D sponsor must charge cost
sharing for a temporary supply of drugs
provided under its transition process
such that the following conditions are
met:
(A) For low-income subsidy (LIS)
enrollees, a sponsor must not charge
higher cost sharing for transition
supplies than the statutory maximum
copayment amounts.
(B) For non-LIS enrollees, a sponsor
must charge—
(1) The same cost sharing for non-
formulary Part D drugs provided during
the transition that would apply for non-
formulary drugs approved through a
formulary exception in accordance with
§ 423.578(b); and
(2) The same cost sharing for
formulary drugs subject to utilization
management edits provided during the
transition that would apply once the
utilization management criteria are met.
* * * * *
(c) * * *
(5) Before June 1, 2015, the following
are applicable:
* * * * *
(6) Beginning June 1, 2015, the
following are applicable: —
(i) A Part D sponsor must deny, or
must require its pharmaceutical benefit
manager (PBM) to deny, a pharmacy
claim for a Part D drug if an active and
valid physician or eligible professional
(as defined in section 1848(k)(3)(B)(i) or
(ii) of the Act) National Provider
Identifier (NPI) is not contained on the
claim.
(ii) A Part D sponsor must deny, or
must require its PBM to deny, a
pharmacy claim for a Part D drug if the
physician or eligible professional (when
permitted to write prescriptions by
applicable State law)—
(A) Is not enrolled in the Medicare
program in an approved status; and
(B) Does not have a valid opt-out
affidavit on file with an A/B Medicare
Administrative Contractor (MAC).
(iii) A Part D sponsor must deny, or
must require its PBM to deny, a request
for reimbursement from a Medicare
beneficiary for a drug if the request is
not for a Part D drug that was dispensed
in accordance with a prescription
written by a physician or, when
permitted by applicable State law, other
eligible professional (as defined in
section 1848(k)(3)(B)(i) or (ii) of the Act)
who—
(A) Is identified by his or her legal
name in the request; and
(B)(1) Is enrolled in Medicare in an
approved status; or
(2) Has a valid opt-out affidavit on file
with an A/B MAC.
(iv) In order for a Part D sponsor to
submit to CMS a prescription drug event
record (PDE), the PDE must contain an
active and valid individual prescriber
NPI and must pertain to a claim for a
Part D drug that was dispensed in
accordance with a prescription written
by a physician or, when permitted by
applicable State law, an eligible
professional (as defined in section
1848(k)(3)(B)(i) or (ii) of the Act) who—
(A) Is enrolled in Medicare in an
approved status, or
(B) Has a valid opt-out affidavit on file
with an A/B MAC.
* * * * *
■ 29. Section 423.360 is added to
subpart G to read as follows:
§ 423.360 Reporting and returning of
overpayments.
(a) Definitions. For the purposes of
this section the following definitions are
applicable:
Applicable reconciliation means the
later of either the annual deadline for
submitting—
(i) PDE data for the annual Part D
payment reconciliations referred to in
§ 423.343(c) and (d); or
(ii) Direct and indirect remuneration
data.
Funds for purposes of this section,
means any payment that a Part D
sponsor has received that is based on
data submitted by the Part D sponsor to
CMS for payment purposes, including
data submitted under § 423.329(b)(3),
§ 423.336(c)(1), § 423.343, and data
provided for purposes of supporting
allowable costs as defined in § 423.308
which includes data submitted to CMS
regarding direct or indirect
remuneration.
Overpayment means funds that a Part
D sponsor has received or retained
under title XVIII of the Act to which the
Part D sponsor, after applicable
reconciliation, is not entitled under
such title.
(b) General rule. If a Part D sponsor
has identified that it has received an
overpayment, the Part D sponsor must
report and return that overpayment in
the form and manner set forth in this
section.
(c) Identified overpayment. The Part D
sponsor has identified an overpayment
when the Part D sponsor has
determined, or should have determined
through the exercise of reasonable
diligence, that the Part D sponsor has
received an overpayment.
(d) Reporting and returning of an
overpayment. A Part D sponsor must
report and return any overpayment it
received no later than 60 days after the
date on which it identified it received
an overpayment.
(1) Reporting. A Part D sponsor must
notify CMS of the amount and reason
for the overpayment, using the
notification process determined by
CMS.
(2) Returning. A Part D sponsor must
return identified overpayments in a
manner specified by CMS.
(e) Enforcement. Any overpayment
retained by a Part D sponsor is an
obligation under 31 U.S.C. 3729(b)(3) if
not reported and returned in accordance
with paragraph (d) of this section.
(f) Look-back period. A Part D sponsor
must report and return any overpayment
identified within the 6 most recent
completed payment years.
§ 423.464 [Amended]
■ 35. Amend § 423.464 as follows:
■ A. In paragraph (f)(2)(i) introductory
text, by removing the phrase ‘‘a Part D
plan must—’’ and adding in its place ‘‘a
Part D plan must do all of the
following:’’.
■ B. In paragraph (f)(2)(i)(A), by
removing ‘‘; and’’ and adding in its
place ‘‘.’’.
■ 30. Amend § 423.501, effective
January 1, 2016, by a adding a definition
for ‘‘prescription drug pricing standard’’
to read as follows:
§ 423.501 Definitions.
* * * * *
Prescription drug pricing standard
means any methodology or formula for
varying the pricing of a drug or drugs
during the term of a pharmacy
reimbursement contract that is based on
the cost of a drug, which includes, but
is not limited to, drug pricing references
and amounts based on any of the
following:
(1) Average wholesale price.
(2) Wholesale acquisition cost.
(3) Average manufacturer price.
(4) Average sales price.
(5) Maximum allowable cost.
(6) Other cost, whether publicly
available or not.
* * * * *
■ 31. Amend § 423.503 by adding
paragraphs (a)(3) to read as follows:
§ 423.503 Evaluation and determination
procedures for applications to be
determined qualified to act as a sponsor.
(a) * * *
(3) CMS does not approve an
application when it would result in the
applicant’s parent organization, directly
or through its subsidiaries, holding
more than one PDP sponsor contract in
the PDP Region for which the applicant
is seeking qualification as a PDP
sponsor. A parent organization is an
entity that exercises a controlling
interest in the applicant.
* * * * *
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00121 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29964
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
■ 32. Amend § 423.504 by adding
paragraphs (b)(4)(vi)(C)(4) and (b)(8) and
(9) to read as follows:
§ 423.504 General provisions.
* * * * *
(b) * * *
(4) * * *
(vi) * * *
(C) * * *
(4) A Part D plan sponsor must
require all of its first tier, downstream
and related entities to take the CMS
training and accept the certificate of
completion of the CMS training as
satisfaction of this requirement. Part D
plan sponsors are prohibited from
developing and implementing their own
training or providing supplemental
training materials to fulfill this
requirement.
* * * * *
(8) If neither the applicant, nor its
parent or another subsidiary of the same
parent, holds a Part D sponsor contract
that has been in effect for at least 1 year
at the time it submits an application, the
applicant must have arrangements in
place such that the applicant and its
contracted first tier, downstream, or
related entities, in combination, have at
least 1 full-benefit year of experience
within the 2 years preceding the
application submission performing at a
minimum all of the following functions
in support of the operation of another
Part D contract:
(i) Authorization, adjudication, and
processing of prescription drug claims
at the point of sale.
(ii) Administration and tracking of
enrollees’ drug benefits in real time,
including automated coordination of
benefits with other payers.
(iii) Operation of an enrollee appeals
and grievance process.
(9) For organizations applying to offer
stand-alone prescription drug plans, the
organization, its parent, or a subsidiary
of the organization or its parent, must
have either of the following:
(i) For 2 continuous years
immediately prior to submitting an
application, actively offered health
insurance or health benefits coverage,
including prescription drug coverage, as
a risk-bearing entity in at least one State.
(ii) For 5 continuous years
immediately prior to submitting an
application, actively managed
prescription drug benefits for an
organization that offers health insurance
or health benefits coverage, including at
a minimum, all of the services listed in
paragraph (b)(8) of this section.
* * * * *
■ 33. Amend § 423.505 as follows:
■ A. In paragraph (f)(3)(v), by removing
‘‘,’’ and adding in its place ‘‘.’’.
■ B. In paragraph (f)(3)(vi), by removing
‘‘; and’’ and adding in its place ‘‘.’’.
■ C. By adding paragraph (f)(3)(viii).
■ D. In paragraph (i)(2)(i), by removing
the phrase ‘‘audit, evaluate and inspect’’
and adding in its place ‘‘audit, evaluate,
collect, and inspect’’.
■ E. By redesignating paragraph (i)(2)(ii)
as paragraph (i)(2)(iv).
■ F. By adding new paragraphs
(i)(2)(ii)and (i)(2)(iii).
■ G. By removing paragraph (i)(3)(iv).
■ H. By redesignating (i)(3)(v) through
(viii) as (i)(3)(iv) through (vii).
■ I. By adding paragraph (k)(7).
■ J. By adding a paragraph (m) heading.
■ K. By revising paragraphs (m)(1)(iii).L.
By revising paragraph (m)(3).
The revisions and additions read as
follows:
§ 423.505 Contract provisions.
* * * * *
(f) * * *
(3) * * *
(viii) Supporting program integrity
purposes, including coordination with
the States.
* * * * *
(i) * * *
(2) * * *
(ii) HHS, the Comptroller General or
their designees have the right to audit,
evaluate, collect, and inspect any
records under paragraph (i)(2)(i) of this
section directly from any first tier,
downstream, or related entity.
(iii) For records subject to review
under paragraph (i)(2)(ii) of this section,
except in exceptional circumstances,
CMS will provide notification to the
Part D sponsor that a direct request for
information has been initiated.
* * * * *
(k) * * *
(7) Certification of accuracy of data
for overpayments. The CEO, CFO, or
COO must certify (based on best
knowledge, information, and belief) that
the information provided for purposes
of reporting and returning of
overpayments under § 423.360 is
accurate, complete, and truthful.
* * * * *
(m) Release of data.
(1) * * *
(iii) Subject, in certain cases, to
encryption of beneficiary identifiers and
aggregation of cost data to protect
beneficiary confidentiality and
commercially sensitive data of Part D
sponsors, in accordance with all of the
following principles:
(A) Subject to the restrictions in this
paragraph, all elements on the claim are
available to HHS, other executive
branch agencies, and the States.
(B) Cost data elements on the claim
generally are aggregated for releases to
other executive branch agencies, States,
and external entities. Upon request,
CMS excludes sales tax from the
aggregation at the individual level if
necessary for the project.
(C) Beneficiary identifier elements on
the claim generally are encrypted for
release, except in limited circumstances,
such as the following:
(1) If needed, in the case of release to
other HHS entities, Congressional
oversight agencies, non-HHS executive
agencies and the States.
(2) If needed to link to another
dataset, in the case of release to external
entities. Public disclosure of research
results will not include beneficiary
identifying information.
* * * * *
(3)(i) CMS must make available to
Congressional support agencies (the
Congressional Budget Office, the
Government Accountability Office, the
Medicare Payment Advisory
Commission, and the Congressional
Research Service when it is acting on
behalf of a Congressional committee in
accordance with 2 U.S.C. 166(d)(1)) all
information collected under paragraph
(f)(3) of this section for the purposes of
conducting congressional oversight,
monitoring, making recommendations,
and analysis of the Medicare program.
(ii) The Congressional Research
Service is considered an external entity
when it is not acting on behalf of a
Congressional committee in accordance
with 2 U.S.C. 166(d)(1) for the purposes
of paragraph (m)(1) of this section.
* * * * *
■ 34. Section 423.505 is further
amended, effective January 1, 2016, by
revising paragraphs (b)(21) and (i)(3)(vii)
to read as follows:
§ 423.505 Contract provisions.
* * * * *
(b) * * *
(21)(i) Update any prescription drug
pricing standard (as defined in
§ 423.501) based on the cost of the drug
used for reimbursement of network
pharmacies by the Part D sponsor on
January 1 of each contract year and not
less frequently than once every 7 days
thereafter;
(ii) Indicate the source used for
making any such updates; and
(iii) Disclose all individual drug
prices to be updated to the applicable
pharmacies in advance of their use for
reimbursement of claims, if the source
for any prescription drug pricing
standard is not publicly available.
* * * * *
(i) * * *
(3) * * *
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00122 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29965
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
(vii) If applicable, provisions
addressing the drug pricing standard
requirements of § 423.505(b)(21).
* * * * *
■ 35. Amend § 423.509 as follows:
■ A. By redesignating paragraphs (a)(4),
through (7), (a)(8) introductory text,
(a)(8)(i) and (ii), and (a)(9) through (14)
as paragraphs (a)(4)(i) through (iv),
(a)(4)(v) introductory text, (a)(4)(v)(A)
and (B), and (a)(4)(vi) through (xi),
respectively.
■ B. By adding paragraph (a)(4)
introductory text.
■ C. In newly redesignated paragraphs
(a)(4)(ii), (iv), (v) introductory text, (vi),
and (vii), by removing the term ‘‘fails’’
and adding in its place the term
‘‘failed’’.
■ D. In newly redesignated paragraphs
(a)(4)(iii), (viii), and (ix), by removing
the term ‘‘fails’’ and adding in its place
the term ‘‘failed’’.
■ E. By revising newly redesignated
paragraphs (a)(4)(x) and (xi).
■ G. By revising paragraphs (b)(1)(i)
through (iv) and (b)(2)(i)(C).
■ H. In paragraph (b)(2)(ii), by removing
the phrase ‘‘MA organization’’ and
adding in its place the phrase ‘‘Part D
plan sponsor’’.
■ I. In paragraph (c)(2)(iii), by removing
the cross-reference ‘‘(a)(4) of this
section’’ and adding in its place the
cross-reference ‘‘(a)(4)(i) of this section’’.
■ J. In paragraph (d), by removing the
cross-reference ‘‘§ 423.642’’ and adding
in its place the cross-reference ‘‘subpart
N of this part’’.
The additions and revisions read as
follows:
§ 423.509 Termination of a contract by
CMS.
(a) * * *
(4) CMS may make a determination
under paragraph (a)(1), (2) or (3) of this
section if the Part D Plan sponsor has
had one or more of the following occur:
* * * * *
(x) Achieves a Part D summary plan
rating of less than 3 stars for 3
consecutive contract years. Plan ratings
issued by CMS before September 1,
2012 are not included in the calculation
of the 3-year period.
(xi)(A) Has failed to report MLR data
in a timely and accurate manner in
accordance with § 423.2460; or
(B) That any MLR data required by
this subpart is found to be materially
incorrect or fraudulent.
* * * * *
(b) * * *
(1) * * *
(i) CMS notifies the Part D plan
sponsor in writing at least 45 calendar
days before the intended date of the
termination.
(ii) The Part D plan sponsor notifies
its Medicare enrollees of the termination
by mail at least 30 calendar days before
the effective date of the termination.
(iii) The Part D plan sponsor notifies
the general public of the termination at
least 30 calendar days before the
effective date of the termination by
releasing a press statement to news
media serving the affected community
or county and posting the press
statement prominently on the
organization’s Web site.
(iv) CMS notifies the general public of
the termination no later than 30
calendar days after notifying the plan of
CMS’s decision to terminate the Part D
plan sponsor’s contract by releasing a
press statement.
(2) * * *
(i) * * *
(C) The contract is being terminated
based on the grounds specified in
paragraphs (a)(4)(i) and (xi) of this
section.
* * * * *
§ 423.642 [Amended]
■ 36. Amend § 423.642(c)(1) by
removing the phrase ‘‘90 calendar days’’
and adding in its place ‘‘45 calendar
days’’.
■ 37. Amend § 423.752 as follows:
■ A. By adding paragraphs (a)(7)
through (10).
■ B. By revising paragraph (c)(1).
■ C. In paragraph (c)(2)(ii), by removing
the phrase ‘‘pursuant to 423.509(a)(4)’’
and adding in its place the phrase
‘‘pursuant to § 422.510(a)(4)(i) of this
chapter’’.
The additions and revision read as
follows:
§ 423.752 Basis for imposing intermediate
sanctions and civil money penalties.
* * * * *
(a) * * *
(7) Except as provided under § 423.34,
enrolls an individual in any plan under
this part without the prior consent of
the individual or the designee of the
individual.
(8) Transfers an individual enrolled
under this part from one plan to another
without the prior consent of the
individual or the designee of the
individual or solely for the purpose of
earning a commission.
(9) Fails to comply with marketing
restrictions described in subpart V or
applicable implementing guidance.
(10) Employs or contracts with any
individual, agent, provider, supplier or
entity who engages in the conduct
described in paragraphs (a)(1) through
(9) of this section.
* * * * *
(c) * * *
(1) CMS. In addition to, or in place of,
any intermediate sanctions, CMS may
impose civil money penalties in the
amounts specified in either of the
following:
(i) Section 423.760(b) for any of the
determinations at § 423.509(a), except
§ 423.509(a)(4)(i).
(ii) Section 423.760(c) for any of the
determinations in paragraph (a) of this
section except § 422.752(a)(5) of this
chapter.
* * * * *
■ 38. Amend § 423.756 as follows:
■ A. In paragraph (a)(2), by removing
the phrase ‘‘days from receipt’’ and
adding in its place ‘‘days after receipt’’
and by removing the phrase ‘‘considers
receipt of notice’’ and adding in its
place the phrase ’’considers receipt of
the notice’’.
■ B. In paragraph (b)(4), by removing the
cross-reference ‘‘§ 423.650 through
§ 423.662 of this part.’’ and adding in its
place ‘‘Subpart N of this part.’’.
■ C. In paragraph (c)(3)(ii) introductory
text, by removing the phrase ‘‘In
instances where marketing or
enrollment or both intermediate
sanctions have been imposed,’’ and
adding in its place the phrase ‘‘In
instances where intermediate sanctions
have been imposed,’’.
■ D. Adding paragraph (c)(3)(ii)(C).
■ E. Revising paragraph (d).
The addition and revision read as
follows:
§ 423.756 Procedures for imposing
intermediate sanctions and civil money
penalties.
* * * * *
(c) * * *
(3) * * *
(ii) * * *
(C) During the limited time period,
sanctioned Part D plan sponsors under
the benchmark that would normally
participate in the annual and monthly
auto enrollment process for enrollees
receiving the low income subsidy will
not be allowed to receive or process
these types of enrollments.
(d) Non-renewal or termination by
CMS. In addition to or as an alternative
to the sanctions described in § 423.750,
CMS may decline to authorize the
renewal of an organization’s contract in
accordance with § 423.507(b), or
terminate the contract in accordance
with § 423.509.
(1) Decline to authorize the renewal of
an organization’s contract in accordance
with § 423.507(b); or
(2) Terminate the contract in
accordance with § 423.509.
* * * * *
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00123 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29966
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
■ 39. Amend § 423.760 as follows:
■ A. In paragraph (a) introductory text,
by removing the phrase ‘‘under
423.752(c)(1), CMS will consider as
appropriate:’’ and adding in its place the
phrase ‘‘under § 423.752(c)(1), CMS
considers the following as
appropriate:’’.
■ B. In paragraphs (a)(1) and (2), by
removing ‘‘;’’ and adding in its place
‘‘.’’.
■ C. Revising paragraph (a)(3).
■ D. In paragraph (a)(4) by removing ‘‘;’’
and adding in its place ‘‘.’’.
■ E. In paragraph (a)(5), by removing ‘‘;
and’’ and adding in its place ‘‘.’’.
■ F. Adding paragraph (c).
The revision and addition read as
follows:
§ 423.760 Determinations regarding the
amount of civil money penalties and
assessment imposed by CMS.
(a) * * *
(3) The adverse effect to enrollees
which resulted or could have resulted
from the conduct of the Part D sponsor.
* * * * *
(c) Amount of penalty imposed by
CMS or OIG. CMS or the OIG may
impose civil money penalties in the
following amounts for a determination
made under § 423.752(a):
(1) Civil money penalties of not more
than $25,000 for each determination
made.
(2) With respect to a determination
made under § 423.752(a)(4) or (a)(5)(i),
not more than $100,000 for each such
determination except with respect to a
determination made under
§ 423.752(a)(5), an assessment of not
more than the amount claimed by such
plan or PDP sponsor based upon the
misrepresentation or falsified
information involved.
(3) Plus with respect to a
determination made under
§ 423.752(a)(2), double the excess
amount charged in violation of such
paragraph (and the excess amount
charged must be deducted from the
penalty and returned to the individual
concerned).
(4) Plus with respect to a
determination made under
§ 423.752(a)(4), $15,000 for each
individual not enrolled as a result of the
practice involved.
■ 40. Amend § 423.1016 by revising the
first sentence in paragraph (b)(1) to read
as follows:
§ 423.1016 Filing of briefs with the
Administrative Law Judge or Departmental
Appeals Board, and opportunity for
rebuttal.
* * * * *
(b) * * *
(1) The other party will have 20
calendar days from the date of mailing
or in person filing to submit any rebuttal
statement or additional evidence. * * *
* * * * *
■ 41. Amend § 423.1020 by revising
paragraph (a)(2) to read as follows:
§ 423.1020 Request for hearing.
(a) * * *
(2) The Part D sponsor or its legal
representative or other authorized
official must file the request, in writing,
to the appropriate Departmental
Appeals Board office, with a copy to
CMS, within 60 calendar days after
receipt of the notice of initial
determination, to request a hearing
before an ALJ to appeal any
determination by CMS to impose a civil
money penalty.
* * * * *
■ 42. Amend § 423.2274 by:
■ A. Revising the introductory text.
■ B. Redesignating paragraphs (a)
through (f) as (b) through (g).
■ C. Adding new paragraph (a).
■ D. Revising newly redesignated
paragraph (b).
■ E. Adding paragraph (h).
The revisions and additions read as
follows:
§ 423.2274 Broker and agent requirements.
If a Part D sponsor uses agents and
brokers to sell its Part D plans, the
following requirements in this section
are applicable.
(a) Definitions. For purposes of this
section, the following definitions are
applicable:
Compensation—(1) Includes
monetary or non-monetary
remuneration of any kind relating to the
sale or renewal of a policy including,
but not limited to—
(i) Commissions;
(ii) Bonuses;
(iii) Gifts;
(iv) Prizes or Awards; or
(v) Referral or Finder fees.
(2) Does not include—
(i) Payment of fees to comply with
State appointment laws, training,
certification, and testing costs;
(ii) Reimbursement for mileage to, and
from, appointments with beneficiaries;
or
(iii) Reimbursement for actual costs
associated with beneficiary sales
appointments such as venue rent,
snacks, and materials.
Like plan type means one of the
following:
(1) PDP replaced with another PDP.
(2) MA or MA–PD replaced with
another MA or MA–PD.
(3) Cost plan replaced with another
cost plan.
Unlike plan type means one of the
following:
(1) PDP replaced with an MA–PD or
an MA–PD replaced with a PDP.
(2) PDP replaced with a cost plan or
a cost plan replaced with a PDP.
(3) MA–PD replaced with a cost plan
or a cost plan replaced with an MA–PD.
Plan year means the year beginning
January 1 and ending December 31.
Renewal year means all years
following the initial enrollment year in
a like plan type.
(b) Compensation rules. A Part D
sponsor must compensate independent
brokers and agents, if compensation is
paid, only according to the following
rules in this section.
(1) Compensation amounts. (i) For an
initial year enrollment of a Medicare
beneficiary into a Part D plan, the
compensation must be at or below the
fair market value of such services,
published annually as a cut-off amount
by CMS.
(ii) For renewal years, compensation
may be up to 50 percent of the current
fair market value cut-off amounts
published annually by CMS.
(iii) If the Part D sponsor contracts
with a third party entity such as a Field
Marketing Organization or similar type
entity to sell its insurance products, or
perform services (for example, training,
customer service, or agent
recruitment)—
(A) The total amount paid by the Part
D sponsor to the third party and its
agents for enrollment of a beneficiary
into a plan, if any, must be made in
accordance with paragraph (b)(1) of this
section; and
(B) The amount paid to the third party
for services other than selling insurance
products, if any, must be fair-market
value and must not exceed an amount
that is commensurate with the amounts
paid by the Part D sponsor to a third
party for similar services during each of
the previous 2 years.
(2) Aggregate compensation. (i) An
entity must not provide aggregate
compensation to its agents or brokers
greater than the renewal compensation
payable by the replacing plan on
renewal policies if an existing policy is
replaced with a like plan at any time.
(ii) An agent or broker must not
receive aggregate compensation greater
than the renewal compensation payable
by the replacing plan on renewal
policies if an existing policy is replaced
with a like plan type at any time.
(iii) The initial compensation is paid
for replacements between unlike plan
types.
(3) Compensation payment and
payment recovery. (i) Compensation
may only be paid for the enrollee’s
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00124 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29967
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
months of enrollment during a plan
year.
(ii)(A) Subject to paragraph (b)(3)(iii)
of this section, compensation payments
may be made at one time for the entire
current plan year or in installments
throughout the year.
(B) Compensation may not be paid
until January 1 of the enrollment year
and, if paid at all, must be paid in full
by December 31 of the enrollment year.
(iii) When a beneficiary disenrolls
from an MA plan, compensation paid to
agents and brokers must be recovered
for those months of the plan year for
which the beneficiary is not enrolled.
For disenrollments occurring within the
first 3 months, the entire compensation
must be recovered unless CMS
determines that recoupment is not in
the best interests of the Medicare
program.
(4) Compensation structure. (i) A Part
D sponsor must establish a
compensation structure for new and
replacement enrollments and renewals
effective in a given plan year.
Compensation structures must be in
place by the beginning of the plan
marketing period, October 1.
(ii) Compensation structures must be
available upon CMS request including
for audits, investigations, and to resolve
complaints.
* * * * *
(h) Finder’s (referral) fees. Finder’s
(referral) fees paid to all agents and
brokers—
(1) May not exceed an amount that
CMS determines could reasonably be
expected to provide financial incentive
for an agent or broker to recommend or
enroll a beneficiary into a plan that is
not the most appropriate to meet his or
her needs; and
(2) Must be included in the total
compensation not to exceed the fair
market value for that calendar year.
Subpart Y—[Reserved]
■ 43. Part 423 is amended by adding
reserved subpart Y.
■ 44. Part 423 is amended by adding
subpart Z to read as follows:
Subpart Z—Recovery Audit Contractor Part
D Appeals Process
Sec.
423.2600 Payment appeals.
423.2605 Request for reconsideration.
423.2610 Hearing official review.
423.2615 Review by the Administrator.
Subpart Z—Recovery Audit Contractor
Part C Appeals Process
§ 423.2600 Payment appeals.
If the Part D RAC did not apply its
stated payment methodology correctly, a
Part D plan sponsor may appeal the
findings of the applied methodology.
The payment methodology itself is not
subject to appeal.
§ 423.2605 Request for reconsideration.
(a) Time for filing a request. The
request for reconsideration must be filed
with the designated independent
reviewer within 60 calendar days from
the date of the demand letter received
by the Part D plan sponsor.
(b) Content of request. (1) The request
for reconsideration must be in writing
and specify the findings or issues with
which the Part D plan sponsor
disagrees.
(2) The Part D plan sponsor must
include with its request all supporting
documentary evidence it wishes the
independent reviewer to consider.
(i) This material must be submitted in
the format requested by CMS.
(ii) Documentation, evidence, or
substantiation submitted after the filing
of the reconsideration request will not
be considered.
(c) CMS Rebuttal. CMS may file a
rebuttal to the Part D plan sponsor’s
reconsideration request.
(1) The rebuttal must be submitted
within 30 calendar days of the review
entity’s notification to CMS that it has
received the Part D plan sponsor’s
reconsideration request.
(2) CMS sends its rebuttal to the Part
D plan sponsor at the same time it is
submitted to the independent reviewer.
(d) Review entity. An independent
reviewer conducts the reconsideration.
The independent reviewer reviews the
demand for repayment, the evidence
and findings upon which it was based,
and any evidence that the Part D plan
sponsor or CMS submitted in
accordance with this section.
(e) Notification of decision. The
independent reviewer informs CMS and
the Part D plan sponsor of its decision
in writing.
(f) Effect of decision. A
reconsideration decision is final and
binding unless the Part D plan sponsor
requests a hearing official review in
accordance with § 423.2610.
(g) Right to hearing official review. A
Part D plan sponsor that is dissatisfied
with the independent reviewer’s
reconsideration decision is entitled to a
hearing official review as provided in
§ 423.2610.
§ 423.2610 Hearing official review.
(a) Time for filing a request. A Part D
plan sponsor must file with CMS a
request for a hearing official review
within 30 calendar days from the date
of the independent reviewer’s issuance
of a determination.
(b) Content of the request. (1) The
request must be in writing and must
provide evidence or reasons or both to
substantiate the request.
(2) The Part D plan sponsor must
submit with its request all supporting
documentation, evidence, and
substantiation that it wants to be
considered.
(3) No new evidence may be
submitted.
(4) Documentation, evidence, or
substantiation submitted after the filing
of the request will not be considered.
(c) CMS rebuttal. CMS may file a
rebuttal to the Part D plan sponsor’s
hearing official review request.
(1) The rebuttal must be submitted
within 30 calendar days of the Part D
plan sponsor’s submission of its hearing
official review request.
(2) CMS sends its rebuttal to the Part
D plan sponsor at the same time it is
submitted to the hearing official.
(d) Conducting a review. A CMS-
designated hearing official conducts the
hearing on the record.
(1) The hearing is not to be conducted
live or via telephone unless the hearing
official, in his or her sole discretion,
requests a live or telephonic hearing.
(2) In all cases, the hearing official’s
review is limited to information that
meets one or more of the following:
(i) The Part D RAC used in making its
determinations.
(ii) The independent reviewer used in
making its determinations.
(iii) The Part D plan sponsor submits
with its hearing request.
(iv) CMS submits in accordance with
paragraph (c) of this section.
(3) Neither the Part D plan sponsor
nor CMS may submit new evidence.
(e) Hearing official decision. The CMS
hearing official decides the case within
60 days and sends a written decision to
the Part D plan sponsor and CMS,
explaining the basis for the decision.
(f) Effect of hearing official decision.
The hearing official’s decision is final
and binding, unless the decision is
reversed or modified by the CMS
Administrator in accordance with
§ 423.2610.
§ 423.2615 Review by the Administrator.
(a) Request for review by
Administrator. If a Part D plan sponsor
is dissatisfied with the hearing official’s
decision, it may request that the CMS
Administrator review the decision.
(1) The request must be filed with the
CMS Administrator within 30 calendar
days of the date of the hearing official’s
decision.
(2) The request must provide evidence
or reasons to substantiate the request.
(b) Content of request. The Part D plan
sponsor must submit with its request all
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00125 Fmt 4701 Sfmt 4700 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2

29968
Federal Register / Vol. 79, No. 100 / Friday, May 23, 2014 / Rules and Regulations
supporting documentation, evidence,
and substantiation that it wants to be
considered.
(1) Documentation, evidence, or
substantiation submitted after the filing
of the request will not be considered.
(2) Neither the Part D plan sponsor
nor CMS may submit new evidence.
(c) Discretionary review. After
receiving a request for review, the CMS
Administrator has the discretion to
review the hearing official’s decision in
accordance with paragraph (e) of this
section or to decline to review said
decision.
(d) Notification of decision whether to
review. The CMS Administrator notifies
the Part D plan sponsor within 45 days
of receiving the Part D plan sponsor’s
hearing request of whether he or she
intends to review the hearing official’s
decision. If the Administrator agrees to
review the hearing official’s decision,
CMS may file a rebuttal statement
within 30 days of the Administrator’s
notice to the plan sponsor that the
request for review has been accepted.
CMS sends its rebuttal statement to the
plan sponsor at the same time it is
submitted to the Administrator. If the
CMS Administrator declines to review
the hearing official’s decision, the
hearing official’s decision is final and
binding.
(e) Administrator review. If the CMS
Administrator agrees to review the
hearing official’s decision, he or she
determines, based upon this decision,
the hearing record, and any arguments
submitted by the Part D plan sponsor or
CMS in accordance with this section,
whether the determination should be
upheld, reversed, or modified. The CMS
Administrator furnishes a written
decision, which is final and binding, to
the Part D plan sponsor and to CMS.
PART 424—CONDITIONS FOR
MEDICARE PAYMENT
■ 45. The authority citation for part 424
continues to read as follows:
Authority: Secs. 1102 and 1871 of the
Social Security Act (42 U.S.C. 1302 and
1395hh).
■ 46. Amend § 424.530 by adding
paragraph (a)(11) to read as follows:
§ 424.530 Denial of enrollment in the
Medicare program.
(a) * * *
(11) Prescribing authority. (i) A
physician or eligible professional’s Drug
Enforcement Administration (DEA)
Certificate of Registration to dispense a
controlled substance is currently
suspended or revoked; or
(ii) The applicable licensing or
administrative body for any State in
which a physician or eligible
professional practices has suspended or
revoked the physician or eligible
professional’s ability to prescribe drugs,
and such suspension or revocation is in
effect on the date the physician or
eligible professional submits his or her
enrollment application to the Medicare
contractor.
* * * * *
■ 44. Amend § 424.535 by revising the
section heading and adding paragraphs
(a)(13) and (14) to read as follows:
§ 424.535 Revocation of enrollment in the
Medicare program.
(a) * * *
(13) Prescribing authority. (i) The
physician or eligible professional’s Drug
Enforcement Administration (DEA)
Certificate of Registration is suspended
or revoked; or
(ii) The applicable licensing or
administrative body for any state in
which the physician or eligible
professional practices suspends or
revokes the physician or eligible
professional’s ability to prescribe drugs.
(14) Improper prescribing practices.
CMS determines that the physician or
eligible professional has a pattern or
practice of prescribing Part D drugs that
falls into one of the following categories:
(i) The pattern or practice is abusive
or represents a threat to the health and
safety of Medicare beneficiaries or both.
In making this determination, CMS
considers the following factors:
(A) Whether there are diagnoses to
support the indications for which the
drugs were prescribed.
(B) Whether there are instances when
the necessary evaluation of the patient
for whom the drug was prescribed could
not have occurred (for example, the
patient was deceased or out of state at
the time of the alleged office visit).
(C) Whether the physician or eligible
professional has prescribed controlled
substances in excessive dosages that are
linked to patient overdoses.
(D) The number and type(s) of
disciplinary actions taken against the
physician or eligible professional by the
licensing body or medical board for the
State or States in which he or she
practices, and the reason(s) for the
action(s).
(E) Whether the physician or eligible
professional has any history of ‘‘final
adverse actions’’ (as that term is defined
in § 424.502).
(F) The number and type(s) of
malpractice suits that have been filed
against the physician or eligible
professional related to prescribing that
have resulted in a final judgment against
the physician or eligible professional or
in which the physician or eligible
professional has paid a settlement to the
plaintiff(s) (to the extent this can be
determined).
(G) Whether any State Medicaid
program or any other public or private
health insurance program has restricted,
suspended, revoked, or terminated the
physician or eligible professional’s
ability to prescribe medications, and the
reason(s) for any such restriction,
suspension, revocation, or termination.
(H) Any other relevant information
provided to CMS.
(ii) The pattern or practice of
prescribing fails to meet Medicare
requirements. In making this
determination, CMS considers the
following factors:
(A) Whether the physician or eligible
professional has a pattern or practice of
prescribing without valid prescribing
authority.
(B) Whether the physician or eligible
professional has a pattern or practice of
prescribing for controlled substances
outside the scope of the prescriber’s
DEA registration.
(C) Whether the physician or eligible
professional has a pattern or practice of
prescribing drugs for indications that
were not medically accepted—that is,
for indications neither approved by the
FDA nor medically accepted under
section 1860D–2(e)(4) of the Act—and
whether there is evidence that the
physician or eligible professional acted
in reckless disregard for the health and
safety of the patient.
* * * * *
(Catalog of Federal Domestic Assistance
Program No. 93.773, Medicare—Hospital
Insurance; Program No. 93.774, Medicare—
Supplementary Medical Insurance Program)
Dated: April 30, 2014.
Marilyn Tavenner,
Administrator, Centers for Medicare &
Medicaid Services.
Dated: May 1, 2014.
Kathleen Sebelius,
Secretary, Department of Health and Human
Services.
[FR Doc. 2014–11734 Filed 5–19–14; 2:56 pm]
BILLING CODE 4120–01–P
VerDate Mar<15>2010 19:14 May 22, 2014 Jkt 232001 PO 00000 Frm 00126 Fmt 4701 Sfmt 9990 E:\FR\FM\23MYR2.SGM 23MYR2
TKELLEY on DSK3SPTVN1PROD with RULES2
