
Global pharma looks to India:
Prospects for growth
Pharmaceuticals and Life Sciences

Table of contents
Introduction 03
Background 04
A fast growing economy
An expanding pharmaceutical market
Government-provided healthcare improving, but private healthcare dominates
Domestic market overview 09
Background
Consolidation underway, despite challenges
Contract manufacturing
Vaccines
Over the counter market holds signicant potential
Reaching the untapped rural market
Growing Research & Development 15
Overview
Clinical trials
Biotech and biosimilars on track for growth
Other growth areas
Bioinformatics
Stem cell research
Medical devices
Global Pharma’s evolving business models and options in India
Background
Export-oriented business (Contract Research and Manufacturing Services)
Licensing
Franchising
Joint ventures
Wholly-owned subsidiaries
Practical concerns 27
Infrastructure
Tax environment
Counterfeiting
Intellectual property
Conclusion 30
Related reading: Pharma 2020
References 32
Acronyms 38
31
20
23
Global pharma looks to India: Prospects for growth 3
Introduction
The pharmaceutical industry’s main
markets are under serious pressure.
North America, Europe and Japan jointly
account for 82% of audited and
unaudited drug sales; total sales
reached US$773 billion in 2008,
according to IMS Health. Annual growth
in the European Union (EU) has slowed
to 5.8%, and sales are increasing at an
even more sluggish rate in Japan (2.1%)
and North America (1.4%).
1
Impending
policy changes, promoting the use of
generics in these key markets are
expected to further dent the top- and
bottom-line of global pharma majors.
The industry is bracing itself for some
fundamental changes in the
marketplace and is looking at newer
ways to drive growth.
Further, higher R&D costs, a relatively
dry pipeline for new drugs, increasing
pressure from payers and providers for
reduced healthcare costs and a host of
other factors are putting pressure on the
global pharmaceutical companies.
Pharma companies are looking for new
ways to boost drug discovery potential,
reduce time to market and squeeze
costs along the whole value chain.
How can industry leaders best face
these challenges? Analysis by
PricewaterhouseCoopers (PwC) shows
that several regions offer considerable
promise, either as places with untapped
demand for effective drugs or as
suitable areas for conducting research
and development (R&D) and/or clinical
trials. In this paper we shall examine the
opportunities available in India.
India’s population is growing rapidly, as
is its economy – creating a large middle
class with the resources to afford
Western medicines. Further, India’s
epidemiological prole is changing, so
demand is likely to increase for drugs
for cardio-vascular problems, disorders
of the central nervous system and other
chronic diseases. Together these factors
mean that India represents a promising
potential market for global
pharmaceutical manufacturers.
More than that, India has a growing
pharmaceutical industry of its own. It is
likely to become a competitor of global
pharma in some key areas, and a
potential partner in others. India has
considerable manufacturing expertise;
Indian companies are among the world
leaders in the production of generics
and vaccines. As both of these areas
become more important, Indian
producers are likely to take a large role
on the world stage – and potentially
partner with global pharma companies
to market their wares outside of India.
Indian companies have also started
entering into the realm of R&D; some of
the leading local producers have now
started conducting original research.
India has the world’s second biggest
pool of English speakers and a strong
system of higher education, so it should
be well-positioned to serve as a source
for research talent. A new patent regime
provides better protection of intellectual
property rights, although some issues
remain. Clinical trials can also be
conducted here much more cost-
effectively than in many developed
nations, and some local companies are
beginning to develop the required
expertise. All of these factors add up to
a strong case for partnering with Indian
companies around R&D, including
clinical testing.
Further, healthcare has become one of
the key priorities of the Indian
Government and it has launched new
policies and programmes to boost
local access and affordability to
quality healthcare.
Global players in the pharma industry
cannot afford to ignore India. The
country, many predict, will be the most
populous in the world by 2050. India will
make its mark as a growing market,
potential competitor or partner in
manufacturing and R&D, and as a
location for clinical trials.
4 PricewaterhouseCoopers
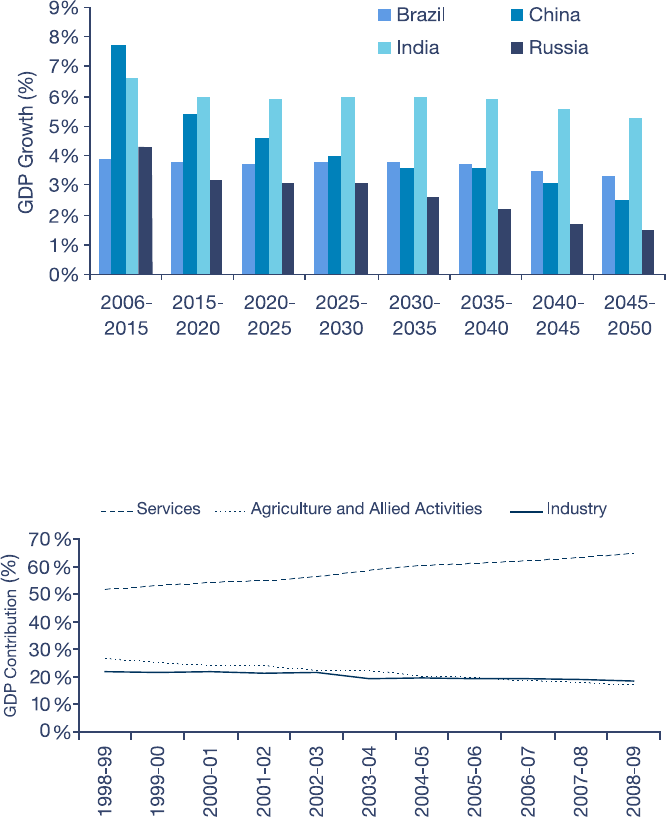
A fast growing economy
The Indian economy is worth about
US$1,243 billion and rapidly getting
bigger.
2
Real GDP growth reached 9%
in the year to March 2008.
3
The rate of
increase has since slowed down due to
the global nancial crisis; in the year to
March 2009, growth eased to 6.7%.
4
Even so, most forecasters believe that
India will continue to show robust
growth over the long-term; a survey of
professional forecasters performed for
the Reserve Bank of India (RBI)
anticipates growth improving to 6% in
the year ending March 2010,
5
and
expects robust growth of 7.8% p.a for
the next ten years.
6
Previous forecasts
such as those of Goldman Sachs
suggest that India will be the only
emerging economy to maintain such an
outstanding pace over the longer term,
i.e. to 2050 (see Figure 1).
7
Two factors underlie this favourable
outlook: India’s demographic prole and
a robust services sector. India’s
population is currently just over 1.1
billion and projected to rise to 1.6 billion
by 2050 – a 45.5% increase that will see
it outstrip China as the world’s most
populous state.
8
India has also utilised
its strengths in IT to become a major
offshore business services provider, in
marked contrast with most of Asia,
which has relied on manufacturing for
its recent growth. As a result, services
now account for 64.5% of India’s GDP
(see Figure 2).
9
While a strong services
sector heralds well for continued
economic prosperity, it also suggests
why India looks to be important for
research and development as well as
drug manufacture; the country’s
experience delivering on outsourcing
opportunities in other knowledge-critical
areas such as IT should serve it well in
its bid to offer such services in pharma,
biotech and related areas.
Figure 1: India is forecast to grow by at least 5% a year for the next 41 years
Source: BRICs and Beyond, Goldman Sachs, November 2007.
Figure 2: India is shifting from agriculture to services
Source: Reserve Bank of India Annual Reports.
Background

Global pharma looks to India: Prospects for growth 5
An expanding pharmaceuticals
market
India’s pharmaceuticals industry looks
set for a solid long-term growth. It
already ranks fourteenth in the global
league table, with sales of almost
US$19 billion in March 2009.
10
However,
PwC estimates that it will rise to
approximately US$50 billion by 2020 –
a 163% in the space of eleven years.
11
Indeed, in our report, Pharma 2020:
The vision, we anticipate that India will
be one of the industry’s top 10 markets
by 2020.
This growth will be driven by the
expanding economy and increasing per
capita GDP. In 2008, India’s middle
class constituted 13% of the
population, according to the National
Council of Applied Economic
Research.
12
While this remains a fairly
small proportion of the total population,
it represents a substantial increase from
a mere 3% in 1995.
13
If the economy
continues to grow faster than those of
the developed world and the literacy
rate keeps rising, around a third of the
population (34%) is expected to join the
middle class in the near future.
14
While
these consumers still earn substantially
less than their US or European
counterparts, they are rapidly acquiring
the buying power necessary to afford
modern healthcare, particularly if
purchasing power parity is considered.
One source estimates that at least 60
million Indians – a market as big as the
UK – can already afford to buy Western
medicines.
15
Aggressive pricing
strategies will be necessary, however, to
make in-roads into India’s price-
sensitive market.
India’s federal Government currently
mandates price controls on essential
drugs, however, these are under review.
Price controls are carried out on certain
The bottom line:
Increased buying
power and
epidemiological
changes should
spur dramatic
growth in sales
volumes, but India
remains a price-
sensitive market.
drugs by virtue of the Drugs Price
Control Order (DPCO), supervised by
the National Pharmaceutical Pricing
Authority (NPPA). The 347 price-
controlled drugs included in 1979 were
reduced to 143 in 1987.
16
At present, 74
bulk drugs are covered under the
DPCO.
17
The Government’s draft
pharmaceutical policy in 2006 sought to
expand the scope of essential drugs
and evoked a sharp reaction from the
industry. They argued that it would
adversely affect R&D activities in India,
as companies would stay away from
investing in new drugs. To date, no
further action on the proposed policy
changes have been taken and it
currently looks unlikely that the DPCO
will be expanded.
The Indian Government’s Department of
Pharmaceuticals has also initiated
operations for a peoples’ medicines
shop, called ‘Jan Aushadhi,’ in various
locations. These shops sell generic
medicines at much cheaper rates
than the price of corresponding
branded medicines.
18
Some multinational pharma companies
are already taking measures to reach a
larger patient population by reducing
drug prices and increasing affordability.
One example: Merck & Co. has
launched differential pricing through
Januvia, its anti-diabetic drug, which is
priced at approximately US$1 per dose
in India – a fth of its price in the US.
19
Indian companies like Biocon have also
followed a similar pricing strategy.
Biocon has launched its monoclonal
antibody BIOMAb EGFR at one-fourth
of its price in the global markets.
20
It’s also likely that India will require
different types of drugs in the future.
Like almost every other emerging
economy, India is experiencing
epidemiological changes. Thanks to
6 PricewaterhouseCoopers
greater afuence and better hygiene,
the population is ageing; by 2028, an
estimated 199 million Indians will be 60
or older, up from about 91 million in
2008.
21
Besides that, it has the largest
pool of diabetic patients, for example,
with more than 41 million people
suffering from the disease (see sidebar
on India’s insulin dependence).
22
The
pattern of demand for medicines is
shifting accordingly. In 2001, anti-
infective and gastrointestinal drugs and
vitamins accounted for 50% of the
domestic market. By 2012, they are
expected to account for just 36%.
Conversely, drugs for cardio-vascular
problems, disorders of the central
nervous system and other chronic
diseases will account for 64% of
total sales, up from 50% in 2001
(see Figure 3).
These factors help to explain why India
is expected to be among the top
markets for many pharmaceutical
India’s insulin dependence
The number of Indians with
diabetes is projected to reach
73.5 million in 2025. The direct
and indirect costs of treating
such patients are currently about
US$420 per person per year. If
these costs remained the same
as they are now, India’s total
bill for diabetes would be about
US$30 billion by 2025. But as
its economic wealth grows and
standards of care improve,
treatment costs are likely to rise.
The US spends an average
US$10,844 per year on each
patient with diabetes. If India’s
per capita expenditure rose to
just one-tenth of this level, the
total cost of treating all patients
with diabetes would be US$79.7
billion by 2025. The value of
prophylaxis in India alone would
thus be substantial; preventing
10% of the population from
developing diabetes would save
nearly US$8 billion a year.
Source: PricewaterhouseCoopers,
Pharma 2020: The vision
companies. It currently represents about
8% of the global drugs market by
volume and only around 1% by value,
23
but the Indian consumer’s rapidly
increasing purchasing power and the
country’s changing epidemiological
prole could jointly improve its price/
volume mix.
In order to get drugs to consumers at
the right price, though, improvements to
local supply chains will need to take
place. One source estimates that
logistics comprise 45-55% of the costs
in the Indian pharmaceutical supply
chain from factory to shelf.
24
India has
historically had a pharma supply chain
with a number of stops between the
initial production and nal consumer.
The arrival of Goods and Services Tax
(GST) may prove to be a strong
incentive for greater streamlining, as
such middle men could potentially add
substantially to the nal cost of
medications in a price-sensitive market.
0%
20%
40%
60%
80%
100%
2001-02
Source: ORGIMS Data, Crisil Research, Pharmaceuticals: Review Indian formulation market (2008)
Figure 3: India’s therapeutic needs are changing
2006-07 2011-12
Anti-infectives
Vitamins/Minerals
Others
Respiratory
Gastrointestinal
Neuro/CNS
Antidiabetic
Cardiovascular
Pain/Analgesics,
Gynaeclogical &
Dermatology
15%
3%
8%
5%
24%
14%
12%
10%
9%
13%
4%
10%
5%
18%
9%
11%
9%
20%
13%
5%
10%
6%
17%
8%
11%
9%
21%

Global pharma looks to India: Prospects for growth 7
Further, the consolidation of the pharma
industry and emergence of pharma retail
chains are likely to lead to more
concentration in the supply chain. The
increasing requirements posed by some
formulations like biologics, which
require advanced expertise such as the
ability to maintain the cold chain and
avoid shocks during the distribution
process, will also play a role. Inventory
reduction and the reduction of order
cycle time will be key objectives for
companies looking to optimise their
supply chains in order to offer their
drugs at affordable prices.
25
Government-provided
healthcare improving, but
private healthcare dominates
The Indian Government is currently
in the throes of a much needed
programme to reform the health care
system. After years of under-funding,
most public health facilities provide only
basic care. Moreover, three quarters of
medical facilities are located in urban
areas, leaving the majority of rural
The bottom line:
India’s healthcare
system is
struggling to
meet the needs
of its vast
population, but
government
programmes and
reforms in the
health insurance
industry should
improve the
situation.
workers without access to hospitals or
pharmacies (see Table 1).
26
Many of
the poor rely exclusively on alternative
forms of treatment such as Ayurvedic
medicine
27
, Unani
28
and Acupuncture.
The Indian Government has made the
provision of healthcare as one of its
Doctors
60 per 100,000 people
29
Nurses
80 per 100,000 people
30
Pharmacies 367,000 (urban),
183,000 (rural)
31
Hospitals 30,000 (67% public,
23% private)
32
Hospital
beds
1.7 million
(one per 1,000 people)
33
Health
centers
171,687 (including
145,272 sub-centres
with basic facilities)
34
Sources: World Health Organisation (2008)
Modern Pharmaceuticals (December 2008);
Health System in India: Opportunities and
Challenges for Improvement (July 2005)
Expresspharmaonline.com (2007); and
World Health Organisation (2007).
Table 1: India healthcare facilities

who do have some insurance, the
main provider is the Government-run
General Insurance Company (GIC),
along with its four subsidiaries, but
private insurance is on the rise. The
health insurance market in India has
undergone liberalisation in recent years.
Further, the Insurance Regulatory
and Development Authority (IRDA)
eliminated tariffs on general insurance
as of January 1, 2007, and sales have
been going up accordingly. In 2007-08,
almost US$1.2 billion worth of medical
insurance policies were sold in India –
up from US$160 million in 2001-02.
42
But widespread use of health insurance
could take many years, not least
because the insurance companies lack
the data they require to assess health
risks accurately and the only products
they sell work on an indemnity basis
– that is, they reimburse the patient
after he or she has paid the healthcare
provider’s bill, making such policies
less attractive.
2010-11 also allocated US$ 2,920
million under the National Rural Health
Mission (NRHM), an increase of 15%
over the previous year.
40
However, critics suggest that the
authorities are doing too little too
late, and those who can afford it have
turned to the private sector instead. In
2008, fee-charging private companies
accounted for 80% of India’s US$48.6
billion expenditure on healthcare,
while central and local Government
accounted for only around 20%. Private
rms are now thought to provide about
80% of all outpatient care and as much
as 55% of all in-patient care.
41
Some costs for care may be covered
by the insurance industry in the future,
although the current lack of general
coverage remains a challenge. In 2007,
only 11% of the population had any
form of health insurance coverage.
For the small percentage of Indians
key priorities. It launched a new policy
to build more hospitals, boost local
access to healthcare and improve
the quality of medical training, and
promised to increase public expenditure
on healthcare to 2-3% of GDP, up from
a current low of 1%.
35
The 2008-09
Union Budget highlighted a ve year
tax holiday for setting up hospitals
anywhere in India, especially in tier-2
and tier-3 towns.
36
The Government
further allocated US$51 million for a
new health insurance scheme to provide
a health cover of US$745 for every
worker (including his/her family) in the
unorganised sector falling below
poverty line (BPL),
37
which was
increased to US$76 million in 2009-10
budget.
38
The recent budget (2010-11)
extended the coverage to another 20%
of the Indian population covered by the
NREGA (National Rural employment
Guarantee Act) programme, who have
worked for more than 15 days during
the preceding nancial year.
39
Budget
8 PricewaterhouseCoopers

Global pharma looks to India: Prospects for growth 9
The bottom line:
2008 saw M&A
in the pharma
sector in India
more than
double against
the previous
year, despite
the challenges
posed by the
global recession.
Background
I
ndia’s domestic pharmaceutical
industry was worth around US$11 billion
in March 2009 and PwC estimates it
will rise to approximately US$30 billion
by 2020.
43
The domestic market is very
fragmented; more than 10,000 rms
collectively control about 70% of the
market.
44
Many of the local players
are generics producers specialising
in anti-infectives. In 1972, the federal
Government passed a law allowing local
producers to manufacture drugs that
were still under patent, as long as they
used different processes.
45
The lack
of a patent system that conformed to
international standards helped spawn
a domestic industry that excelled in
reverse engineering novel drugs and
launching copycat versions at home and
in other emerging markets. Wholesale
marketing of generic versions of drugs
patented since 1995 and still under
patent has not been permitted since
2005 (see Intellectual Property Rights
on page 29), so market strategies are
changing and some generics
producers are looking further aeld
for new markets.
India’s manufacturing clout has made it
a massive threat to established generics
rms – India now produces more
than 20% of the world’s generics.
46
Moreover, around US$70 billion worth
of drugs are expected to go off patent
in the US over the next three years,
and India is well-positioned to take
a substantial share of the resulting
new generics markets.
47
Indian
companies today account for 35% of
the Abbreviated New Drug Application
(ANDA) approvals granted by the US
Food and Drug Administration (FDA)
until February 2009.
48
India’s generic
houses are now entering into strategic
alliances with global pharma companies
to strengthen their generic portfolio
and jointly market these drugs globally,
Domestic market overview

for example Pzer has entered into
alliances with Aurobindo and Claris to
market their drugs in offshore markets.
Similarly, GlaxoSmithKline (GSK) has
acquired exclusive rights for Dr. Reddy’s
Laboratories’ (DRL) pipeline of over 100
generics for sale in emerging markets.
In addition to partnering with global
pharma, some Indian companies are
also setting up their own marketing
subsidiaries abroad.
India’s pharmaceutical exports totalled
around US$8 billion in 2009 and PwC
estimates they will rise to approximately
US$20 billion by 2020.
49
Over the
past several years companies such
as DRL, Cipla and Lupin have grown
internationally in their own right as well.
Other Indian pharma companies like
Glenmark Pharma, Orchid and Aurobindo
also have wholly owned subsidiaries in
different parts of the globe.
DRL has grown from a small rm into
an international business with annual
sales of more than US$1.4 billion,
about 84% of them outside India.
50
The
company’s acquisition of Germany’s
Betapharm positioned it as one of the
largest generics companies in the world;
it is currently one of the largest suppliers
of drugs to the US. It is also one of the
largest active pharmaceutical ingredient
(API) manufacturers globally.
Cipla is another company with
revenues of over US$1.1 billion, 56%
of which come from outside India.
51
It is one of the largest manufacturers
of antiretroviral drugs in the World.
52
In 2007, an Avesta-Cipla joint
venture acquired Siegfried Biologics,
a Switzerland based company, to
manufacture US FDA and European
Medicines Agency (EMEA) compliant
biopharmaceuticals for the global
markets.
53
Meanwhile, Lupin is the
biggest producer of Lisinopril, an API
used in the treatment of hypertension.
54
Lupin’s acquisition of Multicare
Pharmaceuticals of Philippines has
propelled it into position as a top
generics player in the Phillipines.
55
The deal represented Lupin’s sixth
acquisition since 2008.
Consolidation underway, despite
challenges
The Indian pharma industry as a whole is
moving on a consolidation path. The year
2008 saw 57 mergers and acquisitions,
56
a 128% increase over the previous year.
57
Total investment in pharmaceutical,
healthcare and biotechnology sectors
was second among industry sectors in
terms of deal value at US$5.57 billion,
marginally below the Telecommunication
sector which had total transactions worth
US$5.78 billion in 2008. In the same year,
India’s largest pharma company, Ranbaxy
Laboratories, was acquired by Japan’s
Daiichi Sankyo. This was a landmark
deal in the Indian pharma history, where
Ranbaxy’s promoters relinquished
their entire stake to the acquirers. The
transaction paved the way for other
promoters to consider whether they are
better served growing their businesses
independently or by realigning with other
partners who may be able to help them
to take their businesses to the next level
of growth.
In 2008, the world went through a credit
crunch, followed by a prolonged global
economic downturn in the last quarter of
2008 and throughout 2009, both of which
have also had a negative impact on the
Indian pharma industry. The impact of
the downturn, coupled with volatility in
the Rupee, depleted the nancial
position of several Indian pharma
companies, especially those which had
substantial foreign borrowings on their
balance sheets.
Sustaining acquisition heavy structures
became increasingly difcult in 2008.
10 PricewaterhouseCoopers

Global pharma looks to India: Prospects for growth 11
Some Indian companies which made
signicant acquisitions were now nding
it difcult to integrate their foreign
acquisitions with the Indian operations
due to severe pricing pressures.
Legislative reforms imposed in
acquisitions’ home markets also had an
impact. Further, some companies
booked losses on foreign currency
convertible bonds (FCCBs), negatively
impacting overall protability.
Nonetheless, investor condence has
remained fairly stable and deals continue
despite challenges. The average deal size
in 2008 was around US$15.34 million,
20% higher than US$12.82 million in
2007. The pharma sector had 57 deals,
of which 17 deals were domestic. There
were a total of 22 pharma private equity
(PE) deals worth US$337.41 million.
Private equity players and investment
funds played an active role in the deal
market. Some of the investments were
those of Citi Venture and Everest Capital
of about US$23.6 million in Nectar
Lifesciences.
58
Similarly, Kotak Private
Equity Group, an arm of Kotak Mahindra
Bank, invested about US$10 million
in Intas Biopharmaceuticals.
59
Gujarat
Biotech Venture Fund invested US$12.7
million in Century Pharmaceuticals and
SME Growth Fund invested US$7 million
in Centaur Group.
60
Further, in 2009 another landmark deal
was announced, with sano-aventis
acquiring controlling stakes in the
leading Indian vaccine manufacturer
Shanta Biotech.
Elsewhere we discuss some of the
strategies that Indian companies
employed to stay aoat during the crisis,
including greater focus on leveraging
their strengths in newer structures like
Contract Research & Manufacturing
Services (CRAMS), biotech & clinical
trials, and increasing penetration in
rural markets.
Contract manufacturing
Contract manufacturing is a strong
segment of the domestic market. Indian
rms have several advantages over their
Western rivals. The expertise gained in
manufacturing generics through reverse-
engineering has helped some companies
streamline the process for getting
manufacturing up and running. Costs are
very competitive; indeed, they are only
two-fths of those involved in setting
up and running a new manufacturing
facility in the West.
61
They can operate
on signicantly lower margins, given
their low development and labour costs.
Currently their key area of strength in
The bottom line:
2008 saw M&A
in the pharma
sector in India
more than
double against
the previous
year, despite
the challenges
posed by the
global recession.
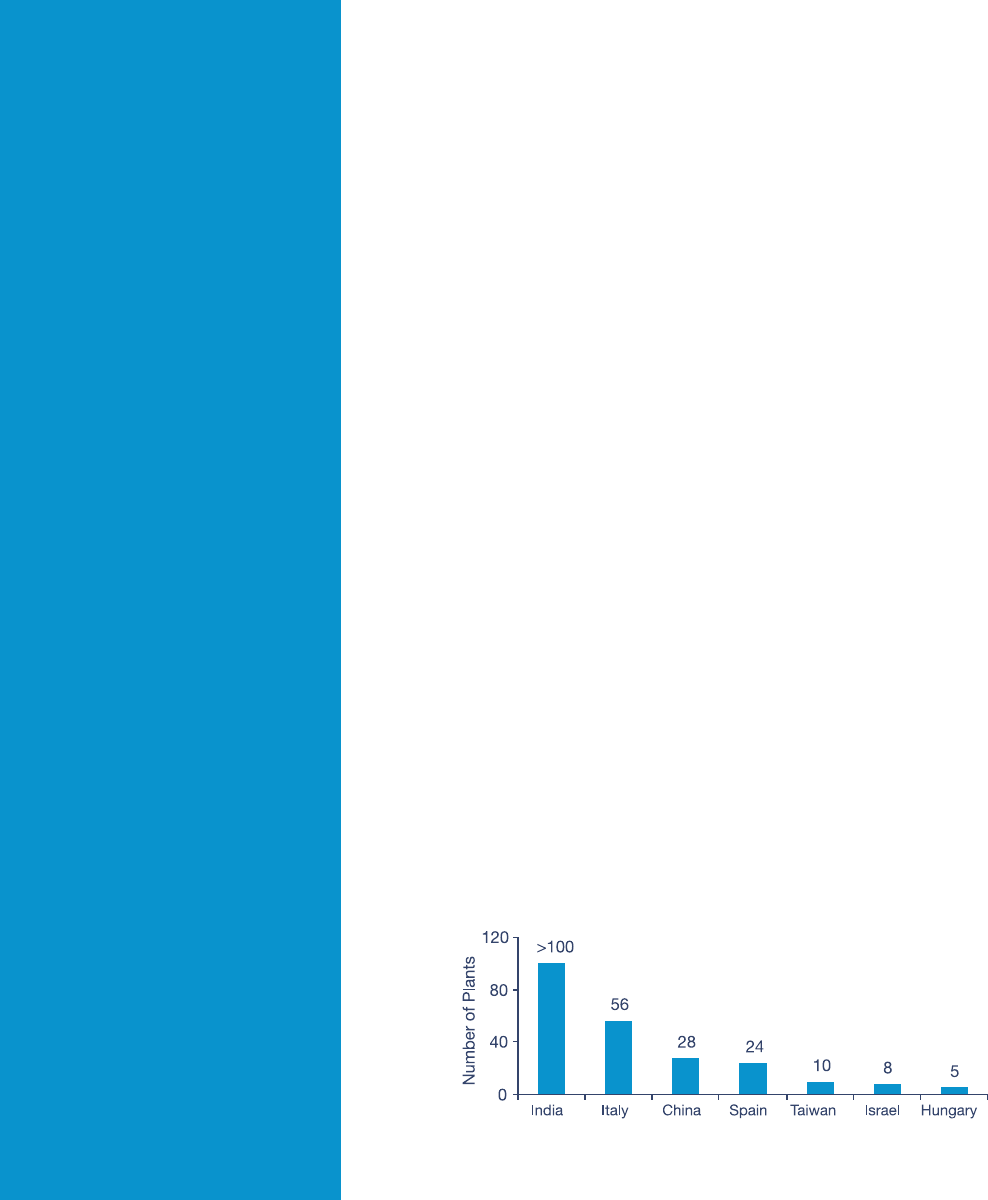
12 PricewaterhouseCoopers
outsourcing is the manufacture of APIs.
Some Indian pharma companies could
probably benet signicantly by moving
towards specialty APIs in the future.
The Indian contract manufacturing
segment was worth around US$605
million in 2008 and is expected to reach
around US$916 million in 2010.
62
The
US FDA has already approved over
100 manufacturing sites – more than
in any country except the US (see
Figure 4).
63
Among six ofces that the
US FDA has overseas, two are located
in India, in Delhi and Mumbai.
64
All
domestic producers are also obliged to
comply with India’s Good Manufacturing
Practices, under Schedule M of the
Drugs and Cosmetics Act, 1940.
Indian manufacturers are currently facing
some scrutiny around quality issues. In
2009, the US FDA took action against a
few Indian companies after conducting a
series of inspections and issuing warning
letters against these drug makers.
While such sanctions clearly pose
signicant challenges, some analysts
see an opportunity as well. Indian
companies are aggressively improving
their manufacturing standards in
response, and are therefore likely to
be better positioned to take advantage
of the upsurge in generics production
The bottom line:
Indian pharma
companies have
solid expertise
in contract
manufacturing
and recent
scrutiny around
quality issues is
driving signicant
improvement in
manufacturing
standards.
Figure 4: India has more US FDA-approved manufacturing plants than any country
except the US
Source: Crisil Research, Bulk drug exports to scale up in the regulated markets (December 2008) for India;
ICICI Securities, Indian Pharma Sector: Sector Update (December 2008) for Italy, China, Spain, Taiwan, Israel
and Hungary.
expected as patents expire over the next
ve years.
65
Some Indian manufacturers are also now
incorporating Lean Manufacturing and
Six Sigma principles to help them boost
operational efciency and further improve
quality, while facilitating compliance.
66
Vaccines
Vaccines are another prominent area
of growth. India is one of the largest
vaccine producers in the world, with
many new vaccines set to be launched
in the next ve years. The vaccines
segment was around US$780 million in
March 2008, growing at a compounded
annual growth rate (CAGR) of 15%.
67
India currently exports vaccines to
about 150 countries. It also meets
around 40-70% of the World Health
Organisation (WHO) demand for the
DPT (diphtheria, pertussis or whooping
cough, and tetanus) and the BCG
(bacille calmette-guérin) vaccine against
tuberculosis, and almost 90% of its
demand for the measles vaccine.
68
The Serum Institute of India, founded
in 1966, is a leading player which
produces and supplies low-cost, life-
saving vaccines for children and adults.
The Institute is also the world’s largest
producer of measles and DPT vaccines.

Global pharma looks to India: Prospects for growth 13
It has been commissioned by the WHO
to develop vaccines against the latest
strain of H1N1. An estimated two out
of every three immunised children in
the world have received a vaccine
manufactured by the Serum Institute.
69
As the risk of global pandemics
grows, so do potential markets for
new vaccines.
OTC market holds signicant
potential
Globally, over-the-counter (OTC) drug
sales have been increasing in recent
years. This trend is driven in part by
aggressive efforts of global pharma
companies to leverage the brand equity
that major products have attained
during the patent period. Other major
winners in the OTC category include
products where patients continue to buy
particular remedies following an initial
doctor’s prescription.
OTC drugs may have even stronger
potential in India. An increasing number
of Indians are already dipping into their
own pockets to buy OTC drugs. The
OTC market was worth about US$1.8
billion in 2009 and is expected to grow
at 18% a year to reach about US$3
billion in 2012.
70
The Government is
now considering plans to expand the
list of drugs which can be sold outside
pharmacies, since many common
household remedies are more difcult
to obtain in India than in other
developing countries. An expansion of
the list would substantially increase
the potential market opportunity in
this segment.
Although the term ‘OTC’ has no legal
recognition, all the drugs that are not
included in the list of ‘prescription
only drugs’ are considered as non-
The bottom line:
OTC sales
are on the
increase, offering
opportunities
to achieve high
volumes and
enhance pharma
brands in India.
prescription drugs (or OTC drugs). OTC
proprietary drugs are also regulated by
the Drugs and Cosmetics Act and the
Drugs and Cosmetics Rules. However,
as they do not require a drug license
they can be sold by non-chemists, so
sales channels are more extensive. As
discussed, much of India’s population
relies on self-medication, and the
purchasing power of the middle class
is growing. These trends should drive
growth in cough and cold formulations,
gastrointestinals, analgesics, and
dermatologicals. Only a few OTC
active ingredients, e.g. acetylsalicylic
acid and ephedrine and its salts, fall
under the current DPCO price control.
Counterfeits of popular OTC drugs are
however a major issue.
Indian consumers are also placing more
emphasis on prevention and wellness,
which should contribute to continued
increases in sales of OTC vitamins
and minerals. The market is already
growing strongly. Protable OTC drugs
for some of India’s largest pharma
companies include articial sweeteners,
emergency contraceptive pills and
nutritional supplements.
The popularity of Ayurvedic therapies
should also contribute to the sales of
related OTC formulations. Some of
the leading OTC brands in India are
registered as ‘Ayurvedic Medicines’
because of their plant-based natural
active ingredients. There are no price
controls on ‘Ayurvedic Medicines’.
Some global pharma companies are
already launching OTC products in India
or buying OTC products. Novartis India
launched Calcium Sandoz as an OTC
supplement in 2000 and has now come
out with Otrivin nasal drops in a spray
form.
71
Pzer has launched Listerine,
Benadryl, Caladryl and Benylin in India,

14 PricewaterhouseCoopers
which were later sold to Johnson and
Johnson.
72
In the future, India may
also serve as a manufacturing location
for OTC products destined for other
markets. In August 2009, US-based
OTC manufacturer Perrigo announced
the purchase of 85% of Indian contract
manufacturer Vedants. The company
plans to shift some of its current
production from facilities in Israel and
Germany to India by 2011.
India’s regulatory framework permits
advertising for OTC products, and
consumers can buy them without a
doctor’s prescription. However, a wider
distribution network will also boost the
growth of such products. Currently
about half of OTC sales come from
chemists, while grocery stores and
general stores account for over a third
of the sales.
73
Pharma companies are
also targeting post ofces to sell OTC
drugs in rural India. This move could
substantially increase the access of
OTC drugs, especially in areas where
there are no pharmacies.
Reaching the untapped rural
market
Although urbanisation continues,
around 70% of India’s population still
resides in rural areas. As already noted,
the population residing in villages has
signicantly reduced access to quality
treatment and medicines. Many pharma
companies are thinking beyond larger
cities and targeting rural sectors.
While urban markets are currently
more lucrative and will continue to
represent a focus for the industry,
the untapped potential of Indian rural
markets is now seen as the next volume
driver. Rising income levels leading
to more affordability, improving health
infrastructure, and increasing incidence
of lifestyle diseases along with the use
of health insurance are fuelling the
growth in rural areas.
Indian companies are devising a
number of strategies to increase rural
penetration. For instance, Lupin has
a strong brand franchise in the anti-
infective, pain management, and
gastrointestinal segments – these three
areas account for 40% of domestic
formulations sales. The company
has a dedicated rural eld force of
more than 300 people and is rapidly
expanding it. Piramal Healthcare has
also announced a new initiative to target
the mass market, focused on general
practitioners, to cater to rural markets.
Piramal plans to employ a eld-force of
approximately 800 people.
Companies looking to access rural
markets face many hurdles, including
lack of communication, language
barriers, high penetration of spurious
drugs, lack of adequate infrastructure,
such as marketing and distribution
channels for niche therapeutic
segments in particular, poor storage
facilities, and insufcient sales
personnel deployment. Global pharma
companies eyeing rural markets will
need to forge alliances and partnerships
to overcome these obstacles.
The bottom line:
While urban
markets will
remain the focus
in the near-term,
getting treatment
out to the 70%
of the population
residing outside
of these areas
represents the
next volume
driver.
Global pharma looks to India: Prospects for growth 15
Overview
PwC estimates that India’s 10 largest
drug rms spent US$480 million on R&D
in 2008. The bulk of this investment
went towards developing new
formulations, however R&D in the Indian
pharmaceuticals industry is changing.
The new patent regime means
companies need to be more innovative,
rather than relying solely on reverse-
engineering existing formulations. The
reliance on anti-infectives is also likely
to lessen. As already noted, as the
illnesses of afuence and age increase,
the demand for many other types of
pharmaceuticals will rise, and Indian
pharma companies need to begin
transforming their portfolios accordingly.
India has widely acknowledged
chemistry skills. Several leading
domestic producers have begun to
conduct original research into new
chemical entities and novel drug
delivery systems. Amongst others,
Ranbaxy has commenced phase-III
clinical trials for its new anti-malarial
combination drug. Other companies are
looking to shift to clinical areas with a
growth opportunity, such as diabetes
(see sidebar on India’s insulin
dependence on page 6). Piramal Life
Sciences has initiated phase-I trials of
a new experimental drug for diabetes-
metabolic syndrome in Canada. DRL is
conducting phase–III trials for its Type II
diabetes drug. Other areas of innovation
are also being explored; Biocon has 7
and Wockhardt has 10 new chemical
entities in their R&D pipelines.
However India offers limited capabilities
in preclinical and complex Biology
research. Preclinical capabilities in
India are limited to clinical trials in
rodents and dogs, with almost none
for primates. The capabilities mostly
reside with Indian pharmaceutical
companies, developed through in-
house R&D programmes – Government
involvement in this area is minimal.
Some Government institutes do offer
basic biology services, but the level of
innovation generated by such facilities
is fairly modest. Multinationals will
need to partly/completely own or
share technology with available Indian
Contract Research Organisations
(CROs) in order to achieve innovative
results. The Indian contract research
segment was estimated at around
US$485 million in 2008 and is expected
to reach around US$1 billion in 2010.
74
Despite Indian pharma companies’
growing expertise in later stages of
the R&D process, many of the drug
candidates initially formulated in India
are likely to be further developed by
Western drug makers, because few
Indian companies can afford the high
costs and failure rates associated
with pushing a drug right through
the pipeline. Several Indian rms
have already entered into research
partnerships with multinationals; DRL
and Torrent have joined forces with
Novartis, for example, while Ranbaxy
has formed alliances with GSK and
Schwarz Pharmaceuticals. Glenmark
has formed an alliance with Napo
Pharmaceuticals and Piramal Healthcare
has formed an alliance with Eli Lilly. By
selling developing and licensing rights
for the US, Japan and Western Europe,
but retaining rights within emerging
markets, some Indian pharmaceutical
companies are able to gain immediate
revenues, while retaining future access
to India’s growing domestic market.
A number of Indian pharma companies
have spun off their R&D divisions
into separate units in order to scale
up resources and to attract focused
investments. DRL started the trend
in R&D spin-offs in 2005. Piramal Life
Sciences, Piramal Healthcare’s R&D
division, was recently demerged from
the latter. Sun Pharma Advanced
Research and Ranbaxy Life Science
Research have also been demerged
from their parent companies Sun
Pharma and Ranbaxy respectively.
Some spin offs have faced difculties
stemming from uncertain resources
and declining PE interest in research.
Several companies are now seeking a
collaborative approach towards drug
discovery, in order to mitigate the
risk associated with failure of a
drug molecule.
India’s R&D base is still small, but it has
several advantages that should serve
it well in the future. Some 70 million
people speak English
75
– more than in
any other country except the US – and
it has an excellent tertiary education
system; every year, it turns out about
115,000 scientists with Master’s
degrees, and 12,000 with PhDs.
76
Many
of these scientists have traditionally
Growing Research
& Development
16 PricewaterhouseCoopers
gone abroad, but companies like
Ranbaxy are now actively trying to
lure them back with the prospect of
opportunities for original research.
Salaries are also very much lower than
they are in North America or Western
Europe. Wage costs within the Indian
pharmaceutical industry are about one-
third of those in developed countries.
77
To achieve its potential and convert
these opportunities into global
success stories, the Indian pharma
industry requires the support and
collaboration of all stakeholders,
including the Government, academia
and nancial investors. Collaboration
will be essential; but to date only a few
Indian pharmaceutical companies have
partnered with academic institutes
to carry out basic research.
78
Such
cooperations can help accelerate
the research process in some areas.
Partnering with academia can also
help develop the sophisticated skills
needed for high-level research.
Pharma players who can leverage the
research capability of academic and
Government institutes, through mutually
benecial collaborative models, will gain
signicant competitive advantage.
Amongst emerging economies, India
has the unique advantage of its recent
successes in the global software and
IT services market. In this respect,
India offers one of the very few
examples of an emerging economy
that has managed to attract Foreign
Direct Investment (FDI) in the area
of high-tech software development,
while successfully inserting itself as
a competitive presence in the very
heart of Silicon Valley. Biotech, another
knowledge-based sector, is now
experiencing a similar boom. Drawing
on the success of IT enterprise parks,
the Government also inaugurated the
rst phase of its rst biotech-IT park
– Bangalore Helix in June 2007. The
project is part of efforts to position
India as a global hub for bioinformatics
and biotech.
Clinical Trials
India’s developing research skills are
matched by its growing involvement in
clinical testing. The country historically
lacked the expertise to perform clinical
trials because most companies only
tested different processes for producing
copycat versions of Western products
and the rules were quite lenient. Several
drug makers have also been caught
behaving unethically or even illegally.
The Supreme Court and Drug Controller
General of India (DCGI) have criticised a
few India pharma companies for testing
new drugs without getting patients’
consent or for violating protocol.
However, during the past few years
a number of big contract research
organisations have set up businesses
in India, including Quintiles, Omnicare,
PharmaNet and Pharm-Olam. Most
of the multinationals, Novo Nordisk,
sano-aventis, Novartis and GSK
among them, have likewise started
running clinical trials here – and some,
such as Pzer and Eli Lilly, have been
conducting tests locally for a while.
In January 2005, the federal
Government amended Schedule Y
of the Drugs and Cosmetics Act to
make the rules on clinical trials more
consistent with international practice.
79
The Health Ministry is planning to add
a new Schedule Y-1 to the Drugs and
Cosmetic Rules 1945 to further improve
the situation.
80
Early stage testing of
molecules discovered outside India is
still restricted, but multinationals can
now conduct trials where, previously,
they could only conduct trials in any
particular phase after completing the
same phase of testing elsewhere.
81
At present, though, the industry still
lacks a strong regulatory framework.
Good Laboratory Practices (GLP)
certication remains a voluntary

Global pharma looks to India: Prospects for growth 17
The bottom line:
Insufcient
regulatory
oversight is
currently a barrier,
however India’s
many advantages
- overall costs
are only 50%
of comparable
US-based
programmes
- should spur
dramatic growth
in clinical testing
in the next 2-5
years.
process, although most Indian pharma
companies dealing with international
clients or exporting to foreign regulated
markets look to attain such certication.
The National Good Laboratory Practice
Compliance Monitoring Authority was
established under the Department
of Science and Technology in April
2002. While this was undoubtedly a
step in the right direction, there are
still only about 33 GLP inspectors
82
and about 12 GLP certied labs in the
country.
83
In addition, the ruling on
whether a trial design violates ethical
principles is left to individual local ethics
committees. There is no central register
of Ethical Committee decisions. Better
infrastructure for regulation, ethics
review and monitoring is required.
84
Registration of new clinical trials is now
mandatory on the Indian council of
medical research's (ICMR) web based
clinical trials registry. The government
plans to make inspection of clinical trial
sites an ongoing activity by increasing
the number of inspectors, training them
for site inspection and developing
a checklist for audits. Further, the
government is also working on a
proposal to register CRO’s in India.
85
This type of more rigorous regulatory
oversight, together with increasing
interest from foreign rms, should help
to boost the Indian clinical trials market.
Expectations are already high; some
observers expect the market could
reach US$2 billion annually by 2012, up
from just US$300 million in 2008.
86
The strong anticipated growth reects
some of the attractions India holds
for this market. According to a study
by Rabo India Finance, a subsidiary
of the Netherlands based Rabo Bank,
the huge patient population offers vast
genetic diversity, making the country
“an ideal site for clinical trials.” Further,
many people are “treatment-naïve” and
relatively easy to access. The United
Nations reports that around 30% of the
population lives in urban areas;
87
and
over 67 million people live in India’s six
biggest cities alone (see Table 2).
Population
(Data in ‘000)
City 2005 2010
Bangalore 6,465 7,229
Kolkata (Calcutta)
14,282
15,577
Chennai (Madras) 6,918 7,559
Delhi 15,053 17,015
Hyderabad 6,117 6,761
Mumbai (Bombay) 18,202 20,072
Table 2: Urban India
Source: United Nations, World Urbanization Prospects
(2007)
18 PricewaterhouseCoopers
The ratio of doctors to patients – at
60 per 100,000 people – is also
relatively high, although the quality
of medical training is not as good as
it is in some other emerging nations.
The country’s 289 medical colleges
are over subscribed and the emphasis
is on quantity rather than quality.
88
These problems are compounded by
lack of experience. India has only 500
to 1,000 investigators in the country
as compared to 50,000 in the United
States, suggesting that most companies
would need to make a major investment
in training during study start-ups.
89
Some Indian pharma companies are
already developing a reputation for
a nimble, rapid approach to clinical
testing that looks to streamline the
clinical trial process and bring new
drugs to market faster. For example,
Glenmark now routinely looks to
incorporate “proof of mechanism” into
every phase–I study.
90
Most pharma
companies save this step for phase–II.
But the most obvious benet of
conducting clinical trials in India is the
potential for cost savings. Clinical trials
account for over 40% of the costs of
developing a new drug.
91
In terms of
cost efciency, India offers substantial
advantages – the cost of conducting
a trial here is lower by 50% than in the
United States.
92
The federal Government
is alive to the strength of this argument.
Drugs and materials imported for clinical
trials are exempt from customs duties.
Clinical trials also remain exempt from
service tax.
Biotech and biosimilars on track
for growth
India is home to a small biotechnology
industry, based largely in Karnataka,
with other clusters of activity in West
Bengal, Maharashtra, Andhra Pradesh,
Hyderabad, Kerala and Ahmedabad. In
2008-09, the sector generated sales of
US$2.64 billion
93
representing a CAGR
of 26%, but both the federal and state
Governments have been actively
promoting biotech research initiatives
and are targeting revenues of US$5
billion by 2010 -11.
94
The leading
domestic players include Serum
Institute of India, which focuses on
immuno-biologicals and vaccines;
Biocon, which concentrates on
recombinant DNA technologies,
bioprocesses, fermentation-based
small molecules and enzymes; and
Panacea Biotec, which specialises in
novel drug delivery techniques and
pharmacogenomics (see Table 3).
Several initiatives have been launched
by the Government to give impetus
to the thriving biotech industry. The
Biotechnology Industry Partnership
Programme (BIPP) has been launched
by the Department of Biotechnology
(DBT) to support high-end
biotechnology research programmes
capable of generating globally
recognised intellectual property. It
specically focuses on transformational
research and development. The
DBT has also drafted the National
Biotechnology Regulatory Act in order
to set up the National Biotechnology
Regulatory Authority (NBRA). The NBRA
is expected to be an autonomous body
formed specically to regulate the
biotechnology segment and reduce
regulatory overlap.
96
Further funding support from the
Government will be critical in ensuring
continued growth in the biotech
industry. The Government can play a
vital role in funding incubation and early
stage ventures.
A growing biotech industry should
help India to gain a share of the global
opportunity currently emerging around
biosimilars. The biosimilars market is
Revenues
(US$ million)
2008–09
Serum Institute of India 242.12
Biocon 198.29
Panacea Biotec 129.79
Rasi Seeds 81.63
Nuziveedu Seeds 79.11
Novo Nordisk 71.72
Siro Clinpharm 60.86
Novozymes South Asia 54.34
Shantha Biotech 53.68
Jubilant 52.60
Source: Biospectrum – ABLE, 2009
95
Table 3: India’s top 10 biotech rms
Company

Global pharma looks to India: Prospects for growth 19
The bottom line:
India’s developing
biotech industry
and cost
advantages
should drive
signicant
growth in local
development of
biosimilars for the
global market.
glaritus. DRL has already launched
lgrastim and rituximab in emerging
markets and has a pipeline of 10
biogenerics in various stages.
104
The challenge for the development of
biosimilars arises from the fact that
biologics are more complex than small
molecules and chemically synthesised
drugs; therefore their replica are – in
contrast to ‘traditional’ small-molecule
generics – ‘similar’ but not identical to
the original drug. Consequently, the
registration of biosimilars requires more
data than is required for generics, and
manufacturers have to demonstrate
efcacy and safety in pre-clinical
and clinical studies. This makes the
registration of biosimilars a costly and
time-consuming process, and lessens
the chances of a successful launch.
Developing biosimilars is costlier than
developing chemical based generics,
requires a greater capital investment
and operating costs of manufacturing
are higher. These factors mean that
developing biosimilars represents a
higher risk area of R&D.
Pharma companies need to balance
the risks and rewards when considering
whether to enter the biosimilars
market. The decision to enter the
market should only be made based on
a clearly dened long-term biosimilar
strategy, including development and
manufacturing capabilities, marketing,
pricing and regulatory expertise. India’s
cost advantages in many of these areas
likely to grow by around US$2 billion
by 2014, to reach a total of US$19.4
billion, following key patent expiration
for epoetin alpha, lgrastim, interferon
beta 1a, interferon alpha, human growth
hormone (hGH), and insulin-glargine.
97
This represents a CARG of 89.1% from
2009 to 2014. All told, around US$25
billion worth of biologics are expected
to go off patent by 2016.
98
These
patent expirations open the route for
biosimilars, the equivalent of generics
for biologics.
Indian biotech companies are slowly
building capabilities in development
and manufacturing of biosimilars. Intas
Biopharmaceuticals is now developing
a biosimilar of a protein used to treat
the side effect of cancer therapy,
for example.
99
Biocon has initiated
registration of its human recombinant
insulin with the European regulatory
agency, EMEA and intends to launch it
by 2011.
100
Reliance Life Sciences has
launched three biosimilars—ReliPoietin
(Erythropoietin), ReliGrast (GCSF),
and ReliFeron (Interferon Alpha 2b) in
the domestic market in 2008 and is
currently conducting clinical studies
for erythropoetin and granulocyte
colony stimulating factor (GCSF) in
Europe.
101
Wockhardt has launched
its recombinant erythropoietin, Wepox
and insulin, Wosulin in the domestic
market
102
and is conducting clinical
trials in the US for Wosulin.
103
It has built
capacities in erythropoetin, hepatitis
vaccine, recombinant insulin and insulin

20 PricewaterhouseCoopers
could help it gain a stronghold globally
in this growing market.
Bioinformatics in India
The modern process for drug discovery
and testing now generates very large
quantities of data through computer
modeling and simulations, genetic
sequencing, and other data-intensive
processes. Further, as we noted in Pharma
2020: The vision, pharma companies are
under increasing pressure to document
the efcacy of their products; tracking
patient outcomes represents a further
source of large quantities of data. In order
to facilitate the storage, management,
retrieval and analysis of this large pool
of data, a new subsector of the IT sector
has emerged – bioinformatics. Tools have
been developed which can help lower
cost, improve efciency, and streamline
the process of documenting a drug’s
efcacy throughout development until
launch and beyond.
India’s strength in the IT sector and its
growing pharmaceutical sector are driving
growth of this emerging area. Revenues
for the Indian bioinformatics industry
were around US$48 million as of March
2009. It is an export driven segment with
earnings of around US$37 million from
overseas. Domestic revenues contribute
around US$11 million.
105
Some companies
provide only specialised bioinformatics
services; in other cases, local life sciences
companies are integrating bioinformatics
services into a complete portfolio of
research capabilities.
India is now actively targeting the
bioinformatics market, with the
construction of its rst biotech-IT park
in Bangalore, at a total cost of about
US$87 million.
106
The rst phase of the
park has been completed and a tender for
the development for phase–II is expected
soon from the local state Government.
Several Indian companies, including the
Bangalore based Strand Genomics and
Ocimum Biosolutions, have already made
forays into the bioinformatics industry.
Recently, Ocimum was granted a patent
for its method and system to manage
and query gene expression data based
on quality.
107
The Institute of Bioinformatics has also
developed a comprehensive database
of all known human proteins and their
characteristics, and the Centre for
DNA Fingerprinting and Diagnostics in
Hyderabad along with Sun Microsystems
has operationalised a Centre of
Excellence focusing primarily on medical
bioinformatics.
108
Some global pharma
companies are already drawing on the
emerging resources. Tata Consultancy
Services has signed a deal with GSK
to set up a support centre in Mumbai
for the company’s global drug
development programme. Biocon has
taken its tie-up with Bistol-Myers Squibb
The bottom line:
India’s existing
knowledge
capital in IT
provides a natural
base for the
development of
bioinformatics
research and
operations.
Other growth areas

Global pharma looks to India: Prospects for growth 21
The bottom line:
India has made
considerable
progress in stem
cell research
and is well-
positioned to
leverage growing
capabilities in this
area.
the National Centre for Cell Sciences in
Pune and the National Brain Research
Centre near Delhi, are investigating the
use of stem cells to regenerate nerve,
heart and adult muscle cells, and repair
damaged bone tissue. The L.V. Prasad
Eye Institute has also treated blindness
using stem cells derived from the eye.
While the Indian Government is strongly
promoting biotech generally, concrete
Government funding for stem cell
research in India still lags far behind
that provided in other countries such
as the US. There are also no laws
per se governing stem cell research,
although there are specic guidelines
which classify stem cell use into three
categories: permissive, restricted,
and prohibited. The Indian Council of
Medical Research is currently drawing
up plans for a national stem cell
initiative to promote clinical applications
of stem cell research in ophthalmology,
cardiology and spinal cord repair,
and build links between scientists
and doctors. India’s ex-president Dr.
A.P.J. Abdul Kalam had also identied
stem cell research as one of the
areas on which the country should
focus its efforts.
Given India’s growing presence in
biotech, drug discovery, and clinical
testing, the country may be well
further by setting up a dedicated
research facility, through its subsidiary
Syngene International.
Stem cell research
Stem cells are seen by many as a
powerful tool for improving the research
and development process in the pharma
industry. Stem cells are being used to
develop some types of direct therapeutic
applications; they are also becoming
increasingly important as a tool to test
potential drug toxicity.
India has already made considerable
progress in this area. India’s entry into
stem cell research has progressed
from a few institutions to currently
over 40 institutions and hospitals
involved in stem cell research.
109
In
2008, Stempeutics, a leading stem
cell company, launched its second
stem cell laboratory on the Manipal
University campus for advanced stem
cell research in human embryonic stem
cells.
110
Further activities followed in
2009 – one example is a joint venture
formed by StemCyte in India with Apollo
Hospitals and Cadila Pharmaceuticals
to provide stem cell therapies.
111
Several
major research institutes, such as the
National Centre for Biological Sciences
in Bangalore, the Centre for Cellular
and Molecular Biology in Hyderabad,

22 PricewaterhouseCoopers
about 50-60%, implantable devices
which are around 20-30%, and simple
plastic disposables which are
around 20%.
The sector became regulated in 2005
under the Drugs and Cosmetics Act.
The Ministry of Health and Family
Welfare declared 10 products to be
classied and listed as drugs under the
Act. The list was expanded in March
2009 to include 19 more products.
Under the Act, import registration
requires product approval from another
country’s regulatory organisation such
as the US FDA or the EU medical
devices directive.
114
The manufacture
of any new type of a medical device is
not covered under the Act and requires
approval from an expert committee put
together for the purpose.
In contrast to other biotech-related
areas such as stem cell research and
bioinformatics, the medical devices
sector lacks the necessary regulatory
and R&D support. Institutional support
is also required for testing and validating
facilities, as well as human resource
The bottom line:
Medical devices
represent a
signicant
potential market,
however the
sector currently
lacks the
regulatory and
R&D support
necessary to
achieve its
full potential
and faces stiff
competition
from Europe and
China.
positioned to take a leading role in
leveraging the potential of stem cell
technology throughout the pharma
value chain.
Medical devices
Many pharmaceutical companies
such as Bayer Healthcare, Johnson
and Johnson Medical India (JJMI),
Roche, and Piramal Healthcare are also
looking to medical devices as a path
to growth. The Indian medical devices
and supplies market is at a nascent
stage and was estimated at US$2.75
billion in 2008. This is about 1.25% of
the global medical devices and supplies
market of around US$220 billion in
2008. By 2012, India’s medical devices
market is expected to nearly double to
around US$5 billion.
112
Improving health
infrastructure such as an increasing
number of hospitals, clinics and
clinical laboratories and telemedicine
services are expected to drive demand.
The production of low value medical
supplies and disposables is dominated
by domestic manufacturers, whereas
the high end medical equipment is
generally imported.
113
The sector
consists of the large medical-dental-
surgical equipment segment which is

Global pharma looks to India: Prospects for growth 23
Global Pharma’s evolving
business models and
options in India
development. In the future the industry
is expected to face stricter regulation
and competition from Europe as well
as China.
Background
The global pharmaceutical industry is
changing. In a report by PwC Pharma
2020: Challenging business models,
we describe how the pharmaceutical
business model is witnessing a
paradigm shift from a fully integrated
company structure towards a future
where companies use a wide range
of outsourcing, partnership initiatives
and other contractual and relationship
arrangements to create networks of
collaboration and discovery. Eli Lilly,
for example, is currently transforming
itself from a traditional fully integrated
pharmaceutical company into a fully
integrated pharmaceutical network,
in order to leverage on a wider range
of resources beyond its physical
The bottom line:
Global pharma
players can take
advantage of a
variety of options
to maximise
their investment
in India. As
many pharma
companies
turn to more
collaborative
business models,
Indian companies
are likely to play
an increasingly
important
partnering role.
boundaries. It aims to get better
access to innovation, reduce its costs,
manage its risks effectively and
improve productivity.
This evolution in pharma business
models has enormous repercussions
for the Indian pharmaceutical sector,
and related sectors like biotechnology.
Indian companies now have an
unprecedented opportunity to
partner with global players across
a wide range of activities, from
contract manufacturing and licensing
arrangements, to franchising and
joint venture opportunities. The range
of option spans a wide spectrum
of levels of ownership and control,
from straightforward outsourcing
of manufacturing to licensing
arrangements to more involved joint
ventures and partially or wholly-owned
subsidiaries (see Figure 5). The amount
of investment risk varies accordingly.
Figure 5 : Evolving business models
Out-licensing -
e.g.
Ethypharm
-
Solvay;
Glenmark
-
Forest,
Glenmark
-
Teijin
In-licensing
-
e.g.
Elder- Enzymotec,
Elder
-
Daiwa;
Lupin -ItalFarmaco
From India
-
e.g. Dishman,
Glenmark,Orchid
and Aurobindo
Into India
-
e.g. Pfizer,
GlaxoSmithKline
and Novartis
E.g.
Fortis Healthcare,
Medicine Shoppe
E.g.
Novavax
-
Cadila;
Novotech
-
ETI Klinical
E.g. Cipla
,
DRL,
Dishman
and GVK
Evolving
Business
Models
Partially or
wholly owned
subsidiaries
E xport
Oriented
B us ines s
-
CR AMS
L icens ing
Franchising
Joint
Ventures
24 PricewaterhouseCoopers
Big Pharma is already well aware of
India’s importance. Many of them
have been sourcing products from
Indian manufacturers for some years,
but have now started setting up their
own production facilities. Sandoz,
the generics arm of Novartis, has two
manufacturing plants and a research
centre for developing formulations
and processes, based in Thane,
near Mumbai. Pzer also operates a
manufacturing base in Thane. GSK
has facilities based in Mumbai and
Nashik; Apotex has a research centre
and manufacturing plant in Bangalore;
and Teva has an R&D centre in
Greater Noida, having already bought
a manufacturing operation in Uttar
Pradesh in 2003. Mid-tier global pharma
companies are present as well – Watson
Pharma, Lonza, Eisai Pharmaceuticals,
Ethypharm and Astellas all have
manufacturing or research facilities in
India. While their presence is certainly
on the increase, only two foreign
multinationals rank in the top 10 Indian
companies, measured by sales – and
even they only have 6.4% of the market
between them (see Table 4).
Export-oriented business:
CRAMS
Outsourcing has been the traditional
method of doing business with Indian
companies. Historically, the focus for
the pharmaceutical industry has been
on lower value add manufacturing
Country sales,
12 months
to Q4 2008,
US($millions)
Country sales,
12 months
to Q4 2007,
US($millions)
Growth, 12 months
to Q4 2008/2007,
Fixed rate US$(%)
Cipla 510 468 9.0%
Aurobindo 477 416 14.5%
Sun Pharmaceuticals 449 340 31.9%
Piramal Healthcare 428 354 20.9%
GSK 399 395 1.2%
Ranbaxy (Daiichi) 368 385 -4.5%
Cadila Healthcare 357 324 10.4%
Lupin 286 261 9.2%
Dr. Reddy’s
Laboratories
232 218 6.3%
Glenmark 134 146 -8.5%
Source: Annual Reports (2009) & Company Reports
115
Table 4: Only two foreign multinationals rank among the top 10 pharmaceutical
companies in India
activities such as APIs and generics,
and India continues to play an important
role in these segments.
In recent years, India’s pharma
companies have also begun to move
up the value chain. Foreign companies
are now increasingly tapping India’s
growing research skills in addition to
its manufacturing skills. Players such
as Dishman and GVK-Biosciences
undertake contract research for western
companies. Low costs, availability of
skilled talent and a large patient pool
continue to be growth drivers for the
CRAM segment in India. Ensuring that
products and research comply with
all relevant regulatory frameworks
continues to be a challenge when
outsourcing to Indian pharmaceutical
players, although the situation
is improving.
Licensing
Multinationals are also striking licensing
agreements to get a share of the
Indian pie. For example, Elder
Pharmaceuticals has entered into
an exclusive in-licensing deal
with Israel’s Enzymotec to sell the
latter’s cholesterol-reducing dietary
supplement, CardiaBeat, in India.
116
Major pharma
company
Global pharma looks to India: Prospects for growth 25
Elder has also entered into another deal
with Daiwa Pharmaceutical of Japan
to introduce Daiwa’s nutraceuticals
into the Indian markets.
117
Lupin has
in-licensed Lupenox, a cardiovascular
drug from ItalFarmaco, an Italian
pharmaceutical company.
118
In recent years, a wide array of out-
licensing arrangements have also
emerged. Ethypharm out-licensed
and entered into a supply agreement
for Mesalazine with Solvay Pharma.
119
Glenmark has out-licensing deals with
Forest, Teijin, Eli Lilly and Merck &
Co.
120
Claris sealed a deal with Pzer
to license out 15 injectable generic
medicines for pain, infections and
other conditions.
121
Most developmental costs are borne by
the licensor in licensing arrangements,
resulting in the licensee paying a high
unit cost and having little control over
manufacture. However, licensing can be
effectively used to establish a common
platform in order to gain rapid in-market
acceptance and create a complete
therapy range through arrangements
such as cross-licensing.
Franchising
India’s retailing industry also offers huge
opportunities for foreign companies to
either set up their own retail franchisee
or enter into collaboration with existing
players. Medicine Shoppe India,
the master franchisee of US-based
Medicine Shoppe International has
already forayed the market and plans
to expand 1,000 stores by 2010.
122
Fortis Healthcare plans to open a chain
of 1,000 stores by 2012, of which the
US$200 million has been committed.
123
Franchising arrangements can
leverage on purchasing power from the
franchisor buying in large quantities and
passing down savings to franchisees.
Continued business support from
the franchisor such as technology,
products, training and marketing is
an added advantage. However, there
are restrictions on how the business
must be managed in order to retain
consistency among franchises. All
franchisees are obligated to conform
accurately to the initial business model.
Joint Ventures
Joint ventures (JVs) are becoming
a more prevalent option for
companies looking to capitalise on
the opportunities presented in India.
Foreign companies are increasingly
looking at local partners to work with
in order to increase their presence
in India. Domestic partners bring
together extensive local expertise due
to their familiarity with the business
environment, knowledge support and
the networked capabilities of other
local pharmaceutical companies. These
advantages, along with low production
costs, skilled labor and faster drug
development can be productively
utilised by western pharmaceutical
companies coming into India. As noted,
India is home to more then 100 US FDA
approved plants, so foreign companies
looking for local partners can access a
substantial manufacturing base.
R&D joint ventures are also growing
in popularity. Some Indian companies
are collaborating with overseas players
to enhance their vaccine development
capabilities, for example. Panacea
Biotech has a joint venture with Chiron
for development and marketing of
vaccines.
124
Similarly, Novavax and
Cadila Pharmaceuticals have a joint
venture for the development and
manufacture of vaccines and other
biopharmaceutical products in India.
125
Other joint ventures focus on biotech
or new biosimilars technologies.
Novavax and Cadila Pharmaceuticals
signed an agreement in March 2009
to form a joint venture, CPL Biologicals.
CPL will develop and manufacture
vaccines, biological therapeutics
and diagnostics in India using
technology contributed from Novavax
and Cadila Pharmaceuticals.
126
Clinical testing also offers opportunities.
In 2009 Novotech, an Australia based
clinical research company, entered into
a strategic venture with ETI Klinical
to service the growing demand for
clinical research and clinical and data

management services in India.
127
Joint ventures offer many ways for
partners to pool their Intellectual
Property (IP) and to share risks and
rewards equally. These types of
arrangements can be particularly
attractive to biotechnology or national
pharmaceutical companies who wish to
retain some control over development
and to sell the resulting product in
some markets, but who lack the ability
to undertake global development and
commercialisation. However, the prot
and/or sales split may be determined as
much by the companies’ relative market
strengths as by the value of their
initial IP.
Partially or Wholly owned
subsidiaries
Some multinational companies have
also increased their stake in their Indian
subsidiaries to take advantage of the
India opportunity. Pzer has been able
to increase its stake in its Indian arm,
Pzer India, from 41.2% to around
72%.
128
Similarly, Novartis AG has
hiked its stake to 76.42% in its Indian
subsidiary Novartis India from 50.9%.
129
Other companies are using local
subsidiaries to set up their own sales
and marketing organisations, either
organically or through acquisitions. GSK
has headquartered its wholly-owned
subsidiary SB Asia in India. Novartis has
two wholly owned companies in India
– Novartis Consumer Health Private
Limited and Sandoz India Private
Limited. Pharmacia India Private
Limited remained as a wholly owned
subsidiary and was not consolidated
with Pzer India during Pzer’s
acquisition of Pharmacia.
130
Unlike in some other sectors,
fully owned subsidiaries in the
pharmaceutical industry offer little risk
in terms of sharing critical data and
competitive advantage, as most are
subject to strong control by the parent
company. Pharmaceutical companies
willing to have wholly owned operations
in India can gain value from being
present across the value chain, from
drug discovery to clinical trials through
to manufacturing. Other benets may
include tax advantages.
Requirements of a JV
Foreign companies forming a JV
in India can invest directly via
the automatic route i.e. no prior
approval of the Government is
required. Foreign direct investment
up to 100% is permitted in
pharmaceutical sector under the
automatic route. In cases where
the foreign investor has an existing
venture or tie-up in India in the
same eld as on 12 January
2005, prior Foreign Investment
Promotion Board (FIPB) approval is
required. The FIPB generally grants
its approval on the basis of a no
objection certicate (NOC) from the
existing JV partner.
26 PricewaterhouseCoopers

Global pharma looks to India: Prospects for growth 27
Infrastructure
Insufcient energy infrastructure and
inadequate transport infrastructure
has historically posed challenges for
companies operating in India. The
situation is denitely improving, as
the Government focuses attention
on infrastructure needs. The Indian
infrastructure sector continues to be
viewed as an investment opportunity,
despite the global slowdown.
In early 2009, the Indian Government
was reported to be mulling over a plan
to use part of its foreign exchange
reserves to fund certain forms of
infrastructure spending. It is also
keen to encourage public-private
partnerships (PPPs) in infrastructure
development projects. The Union
Ministry of Health and Family Welfare,
along with the pharmaceutical industry
and airport developers GVK and GMR,
plan to set up dedicated cargo zones to
handle the import and export of pharma
products.
131
Such initiatives could spur
substantial improvements in India’s
infrastructure over the medium-term.
Tax environment
India is expected to implement a new
Direct Tax Code, pending approval,
by April 2011, which should simplify
the existing tax structure. The new
tax code proposes a reduction in the
corporate tax rate from the current 30%
to 25% and an unlimited carry forward
of business losses. A dual system
GST has also been proposed for April
2010. The implementation of the new
GST may face delays. The new system
Practical concerns
would impose taxes at both federal and
state levels and differentiate between
goods and services. The new dual
GST is designed to aggregate different
indirect taxes currently levied, in order
to simplify and integrate the current
system of indirect taxation.
India already offers a variety of tax
concessions to the pharmaceutical
sector, including tax holidays for
industrial operations established in
free trade zones or under-developed
areas; deduction of prots earned from
exports; liberal depreciation allowances;
deduction of capital R&D expenditure;
and relief on all contributions
to approved domestic research
institutions. For pharma manufacturing
units, there is an additional weighted
deduction of 200% for expenditures
relating to in-house R&D.
132
Further,
recently, a new provision has been
added to provide 125% weighted
deduction for expenditure incurred
towards outsourcing of R&D activities.
At present, foreign direct investment
in manufacture of drugs and
pharmaceuticals including those
involving use of recombinant DNA
technology is freely permitted up to
100% under the automatic route,
i.e., without obtaining any prior
regulatory approval.
Of particular interest for pharma
companies may be the special
economic zones (SEZs). In order to
incentivise the country’s export sector,
the Government has formulated the
SEZ policy, which offers cost and tax
benets. On the corporate tax front,
units set up in SEZs enjoy 100%
The bottom line:
India offers
some attractive
tax benets
for pharma
companies and
reductions in
customs duties
should also
help global
manufacturers
compete in the
price-sensitive
Indian market.
28 PricewaterhouseCoopers
income tax exemption on export prots
in the rst ve years of operation, 50%
exemption for the next ve years, and
50% exemption on the reinvested
export prots in the following ve years.
Companies located in SEZ also benet
from various Indirect Tax benets such
as exemption from payment of Customs
Duty; Excise Duty; Central Sales Tax
and refund and exemption of
Service Tax.
Currently, SEZs must adhere to a
positive net foreign exchange
obligation policy (i.e. where the total
value of exports should be more than
the total value of imports) under the
Import Export policy, in order to retain
SEZ status. A proposal has been
made to exempt pharma SEZs from
this requirement.
133
In an effort to attract companies to
SEZs, some of these are located in
modern industrial areas. The Jawaharlal
Nehru Pharma City, India’s rst and
largest pharma industrial estate,
includes a SEZ. The facility is located
near Visakhapatnam, in close proximity
to many chemical manufacturing hubs,
and offers common infrastructure
for resident pharma companies.
There are three other pharma SEZs
located in Andhra Pradesh, and four
in Maharashtra, as well as one on the
outskirts of Dehra Dun in Uttarakhand,
so global pharma companies have a
range of options.
At this stage, it may also be pertinent
to note that the draft Direct Tax Code
Bill published by the Government
presently does not provide for SEZ-
related incentive schemes. However,
recent press releases suggest that
the Finance Minister has identied
proposed incentive provisions as one of
the areas for detailed examination prior
to nalisation of the Direct Tax Code.
134
Overall, India offers a favourable
environment as far as taxation policies
for pharmaceuticals are concerned. In
addition to the attractive tax benets
for companies pursuing innovative
R&D in India, the recent budget 2010
-11 has provided certain benets to
pharmaceutical industry. In this budget,
a uniform, concessional basic duty
of 5%, countervailing duty (CVD) of
4% with full exemption from special
additional duty has been prescribed on
all medical equipment, while the parts
and accessories for manufacture of
these equipment has been prescribed
only the basic custom duty of 5%
and exempted from CVD and special
additional duty. Specied inputs of
orthopaedic implants and medical
equipment and devices such asassistive
devices, rehabilitation aids, etc. are fully
exempted from import duty.
135
The 2009
-10 budget reduced the customs duty
on import of inuenza vaccine and nine
specic life saving drugs and bulk drugs
used for the manufacture of such drugs
to 5%. This will better enable foreign
drug-makers to sell products at a lower
price point and better compete in India’s
highly price sensitive market.
Counterfeiting
Counterfeit drugs have been a serious
issue in India. The Organisation of
Pharmaceutical Producers of India
(OPPI) has spearheaded various
initiatives to combat the problem. It has
conducted several seminars and worked
closely with the Ministry of Health to
develop policies for controlling the
production and sale of ‘spurious’ drugs.
It has also published a series of anti-
counterfeiting guidelines for the industry
as a whole. Surprisingly, a recent
nationwide survey conducted by the
health ministry, published in December
2009 nds a much lower incidence
of spurious drugs in the country than
Global pharma looks to India: Prospects for growth 29
previous industry estimates. It found the
prevalence of spurious drugs at 0.046%
of all medicines sold to customers, in
contrast to results of an earlier survey
funded by the WHO and undertaken
by the International Pharmaceutical
Federation, which concluded that 3.1%
of drugs in India were counterfeit.
136
While such ndings are a positive
sign, companies should remain alert to
possible counterfeiting issues.
Intellectual Property Rights
The federal Government introduced
product patents for all industrial sectors
under the Patents (Amendment) Act,
2005 – in line with the commitment
India made when it signed up to the
Trade-Related Aspects of Intellectual
Property Rights (TRIPS) Accord in 1995.
This regulation aims to balance the
interests of domestic and multinational
drug makers. It represents a major
improvement on the previous rules, but
some issues remain. Firstly, it does not
apply to drugs patented before 1995.
137
Copies of drugs patented between
1995 and the introduction of the law will
probably not be withdrawn.
The Ordinance also allows third parties
to oppose an application for a patent,
which will prolong the period required
to issue a grant. It permits compulsory
licensing in some circumstances other
than national emergencies and public
health crises – provisions that could be
abused for commercial gain.
Further, patent rights for ‘mail box’
applications led will only accrue
from the date the patent is granted.
138
Lengthy delays are common, as the
Indian Patent Ofce lacks sufcient
resources to process applications very
rapidly. While a ‘mail box’ application
is pending, generic manufacturers can
freely produce the same drug
without fear of incurring any liability
for damages.
Post 2005 India has made several
amendments to better protect
intellectual property rights and enable
global pharma companies to bring
their patented products to India,
while protecting the interest of home
grown companies. The Satwant Reddy
committee’s report on data protection
has recommended pro-patent
amendments and data exclusivity for a
period of ve years.
139
The enforcement
regime is also changing, but the legal
system is currently too overburdened
for these improvements to be
immediately effective.
In December 2008, the Delhi High
Court’s landmark judgment in
favor of Bristol-Myers Squibb, the
patent holder for the leukemia drug
Dasatinib, restrained Hetero Drugs
from manufacturing and marketing
generic versions of the drug. In the past,
marketing approvals were sometimes
granted by the DCGI independently
of the patent status of the drug in
question. The judgment establishes
a link between patent and marketing
approvals granted by the IPR ofce
and the DCGI.
140
In June 2009, Novartis’ cancer drug
Glivec was not awarded a patent
for lack of improved efcacy under
section 3(d) of the Indian Patent Act
and its high price as ruled by the
Intellectual Property Appellate Board
(IPAB).
141
The former justication has
since come under scrutiny. In August
2009, the Government accepted the
recommendations of the Mashelkar
committee supporting patenting of
incremental innovation. The Mashelkar
report also pointed that efforts were
required to provide drugs at affordable
prices to the people of India and to
prevent the granting of frivolous
patents and evergreening.
30 PricewaterhouseCoopers
Conclusion
The Indian market is impossible to
ignore, given its economic prospects.
Foreign companies view India as a
potential signicant contributor of
future sales and are ramping up their
investments in the country accordingly.
India’s domestic market looks promising
for global pharma looking to launch
new products. The country’s growing
capabilities in contract manufacturing,
R&D and clinical trials also make it a
preferred outsourcing partner for global
pharma at every stage of the value
chain. So what strategy should foreign
pharmaceutical companies eager
to enter the country or expand their
existing operations adopt?
One approach is to call on India’s
increasing expertise in biotechnology,
bioinformatics and clinical testing.
Several overseas companies have
outsourced research and clinical trials
to Indian contractors, while others
have entered into collaborative R&D
arrangements to supplement their R&D
productivity. Many foreign companies
have also already initiated research on
neglected diseases. We believe that
many more will do so, as the patent
regime is strengthening. This will enable
them to capitalise on the cost savings to
be gained from shifting some research
activities to India, without jeopardising
their most valuable intellectual property.
Another approach is to tap into the
growing domestic market. Foreign
companies with a product portfolio
spanning across different therapeutics
segments can look at bringing newer
products in India by entering into
collaborative networks across the value
chain, from sourcing and manufacturing
to marketing and distribution. These
companies will have to understand
how to get their product to market and
develop a realistic pricing strategy,
particularly as India is still far away
from a widespread shift to an insured
payer model.
India’s pharma market is highly
fragmented and remains extremely
price sensitive. Affordable healthcare
continues to pose a challenge, although
there are a number of healthcare
initiatives by the Government underway
to improve the situation for India’s vast
population. Indian courts and regulatory
authorities are very sensitive to pricing
issues in making decisions around
intellectual property. Pharma companies
coming into India may need to consider
a differential pricing. They will need
to evaluate access to medicines, a
volume-based pricing strategy and
take into account gradually increasing
per capita incomes to come up with
acceptable price levels for their drugs.
Global pharma companies will then
need to decide how to manufacture
their products, and identify and develop
strong local partners.
One way to build a presence in India
may be through an increased presence
in the OTC market. Promoting a range
of OTC products could serve as means
of building brand awareness and as a
source of new revenues. Indigenous
producers dominate the generics
business, and about 97% of all drugs
sold in India are already off patent. The
OTC market is, by contrast, relatively
undeveloped. Indian consumers already
pay privately for the lion’s share of their
healthcare, and the Government is too
hampered by budgetary constraints
to reverse this pattern. In future, then,
it seems likely that access to OTC
medicines will be improved and the
market will continue to expand.
The pharmaceutical business model
is witnessing a paradigm shift, moving
from a fully integrated company
structure towards a future where
companies use a wide range of
outsourcing, partnership initiatives
and other contractual and relationship
arrangements to create networks of
collaboration and discovery. Investing
in India will be a vital component of
this networked future. Companies
that will be most successful in doing
business in India will be those that are
most adept at managing and mixing a
range of contractual relationships and
partnership strategies.
Some practical issues will need to be
addressed, regardless of the business
model selected. Infrastructure decits
continue to exist, although some are
being addressed. Intellectual property
protection has improved substantially
but some holes remain. And while
the regulatory environment in India
has improved substantially in recent
years, the industry still faces a number
of question marks. Finalisation of
Government policies around drug
price control, access to OTC drugs,
tax policy, intellectual property
protection and infrastructure spending
is still pending.
Nonetheless, India’s appeal is growing
rapidly in a number of respects. It
has long been a formidable player in
pharmaceutical manufacturing, but
its socio-economic strengths provide
even greater grounds for optimism. If
the economy outpaces that of every
other emerging country for the next
half century, as many commentators
expect, large portions of the population
will be able to afford modern medicines.
India’s increasing scientic expertise
will also equip it to play a signicant
role in researching and developing
those drugs. It has a large pool of
highly educated, English speaking
scientists who can undertake research
and conduct trials more cheaply and in
some cases faster than their Western
peers. These are major advantages in
a world where drug development
costs are soaring and getting to
market fast is vital.
Global pharma looks to India: Prospects for growth 31
PricewaterhouseCoopers’
Pharma 2020 series
Pharma 2020: The vision
First in the series, the report highlights
a number of issues that will have a
major bearing on the industry over the
next 11 years. The publication outlines
the changes we believe will best help
pharmaceutical companies realise the
potential the future holds to enhance
the value they provide to shareholders
and society alike.
Pharma 2020: Virtual R&D
Second in the series, the report explores
opportunities to improve the R&D
process. This paper proposes that new
technologies will enable the adoption
of virtual R&D and by operating in
a more connected world, industry,
in collaboration with researchers,
Governments, healthcare payers and
providers, can address the changing
needs of society more effectively.
Pharma 2020: Marketing the future
Third in the series, the report
discusses the key forces reshaping the
pharmaceutical marketplace, including
the growing power of healthcare payers,
providers and patients, and the changes
required to create a marketing and sales
model that is t for the 21st century.
Pharma 2020: Challenging
business models
Fourth in the series, the report explains
why Pharma’s fully integrated business
models may not be the best option
for the pharma industry in 2020 and
why more creative collaboration
models may be more attractive. The
paper also evaluates the advantages
and disadvantages of the alternative
business models and how each
stands up against the challenges
facing the industry.
Pharma 2020: Taxing times ahead
Fifth in the series, the report discusses
the implications changes to the
business model and political and
economic trends may have on how the
pharma industry is taxed. The report
focuses on the challenges ahead,
but also shows how companies can
adapt their tax planning to support the
provision of outcomes-based healthcare
and remain competitive.
The entire Pharma 2020 series is
available for download at
www.pwc.com/pharma2020
Related reading
32 PricewaterhouseCoopers
1. IMS Health, “Total Unaudited and Audited Global Pharmaceutical Market by Region” (March 2009).
2. IMF, World Economic Outlook Database (October 2009).
3. Government of India: Union Budget and Economic Survey (2008-09).
4. National Accounts Division - Press Release and Statements, Ministry of Statistics and Programme Implementation, Government of India (29 May,
2009).
5. Survey of Professional Forecasters conducted by the Reserve Bank of India, Results of the Ninth Round Q2:2009-10 Press Release (November
16, 2009).
6. Survey of Professional Forecasters conducted by the Reserve Bank of India, op. cit.
7. Goldman Sachs, BRICS Report (October 2003).
8. US Census Bureau, International Data Base.
9. Reserve Bank of India, Macroeconomic and Monetary Developments, Second Quarter Review (2009-10).
10. Department of Pharmaceuticals, Third Round Up of Developments in the Pharmaceutical Sector (July, 2009).
11. PwC Analysis, assuming the market grows at the rate of 9.2% p.a.
12. “Will a resentful middle class dene electoral outcomes?”, National Council of Applied Economic Research (October 17, 2008), accessed May 30,
2009http://www.ncaer.org/downloads/MediaClips/Press/livemint-willaresentfulmiddle.pdf
13. “Observed and Expected Size of Middle Class, India: Economic Issues and Debates” (January 15, 2006), accessed at http://www.ncaer.org/
popuppages/eventdetails/e3feb2006/SKBery.pdf
14. “Will a resentful middle class dene electoral outcomes?” op. cit.
15. Deutsche Bank, “India’s Pharmaceutical Industry on Course for Globalisation” (April 2008).
16. India OTC Prole 2008, Organisation of Pharmaceutical Producers of India (2008).
17. The First Schedule, List of Price Controlled Drugs, Drug Prices Control Order (1995), accessed July 21, 2009, http://nppaindia.nic.in/drug_
price95/txt9.html
18. “Jan Aushadhi - A solution to improve access to medicines”, Pharmabiz.com (June 03, 2009) accessed September 3, 2009, http://www.
pharmabiz.com/article /detnews.asp?articleid=50025§ionid=46
19. S. Harachand, “India-specic pricing bucks trend”, ContractPharma (August 2008).
20. Indian Biotechnology Innovation & Growth (2007), accessed September 10, 2009, http://www.pharmexcil.com/v1/docs/TechSes-III/22.Biocon-
sandeep-Biotechnology.pdf
21. U.S. Census Bureau Data (2008).
22. Viswanathan Mohan and Rajendra Pradeepa, “Redesigning the urban environment to promote physical activity in Southern India”, DiabetesVoice,
Volume 52, Issue 1 (March 2007): 33-35.
23. Department of Chemicals and Petrochemicals, “IndiaChem Report” (October 2008).
24. “Logistics issues in Indian pharmaceutical industry” (16 March 2008), Express Pharma, accessed September 18, 2009, http://www.
expresspharmaonline.com /20080330/loggedon04.shtml
25. “Logistics issues in Indian pharmaceutical industry”, op cit.
References
Global pharma looks to India: Prospects for growth 33
26. Speech by The President of India, Shrimati Pratibha Devisingh Patil (February 1, 2009), accessed July 8, 2009, http://presidentondia.nic.in/
sp010209.html
27. Ayurveda is a system of medicine that prevents and cures illnesses through natural means lifestyle interventions, Medicinenet.com, accessed July
6, 2009, http://www.medterms.com/script/main/art.asp?articlekey=10787
28. Unani treatment is based on its natural and remarkable diagnosis methods, mainly dependent on the Temperament of the patient, hereditary
condition and effects, different complaints, signs and symptoms of the body, external observation, examination of the pulse, etc., Department of
Ayurveda, Yoga & Naturopathy, Unani, Siddha and Homoeopathy (AYUSH), accessed July 6, 2009, http://www.indianmedicine.nic.in/unani.asp
29. World Health Organisation, “World Health Statistics, 2007” (2008).
30. World Health Organisation, op. cit.
31. Rajendra Gupta, “Role of pharmacies and pharmacists - Emerging horizons”, Modern Pharmaceuticals (December 2008).
32. Ramani KV & Mavalankar Dileep, “Health System in India: Opportunities and Challenges for Improvement” (July 2005), Indian Institute of
Management.
33. Viveka Roychowdhury, “India’s booming pharma market” (October 15, 2007), ExpressPharma, accessed July 6, 2009, http://www.
expresspharmaonline.com/ 20071015/cphlworldwide05.shtml
34. World Health Organisation, “Primary Health Care Resources in India” (2007).
35. National Rural Health Mission, (2005-2012), Mission Document, accessed March 4, 2010, http://mohfw.nic.in/NRHM/Documents/Mission_
Document.pdf
36. “General Budget 2008-09”, Press Information Bureau, accessed May 24, 2009, http://pib.nic. in/release/release.asp?relid=35816
37. “Budget India 2008: What is New for Health Sector?”, Indian Pediatrics, Volume 45 (May 17, 2008): 399-400, accessed May 27, 2009, http://
medind.nic.in /ibv/t08/i5/ibvt08i5p399.pdf
38. India Union Budget 2009-10, accessed July 6, 2009, http://indiabudget.nic.in/ub2009-10/download_index.htm
39. “Union Budget 2010: Private hospitals likely to benet”, Economic Times , (February 26, 2010), accessed March 3, 2010, http://economictimes.
indiatimes.com/News/ Union-Budget/Union-Budget-2010-Private-hospitals-likely-to-benet/ articleshow/5620329.cms
40. “More for public health, less for research” Business Standard (February 27, 2010).
41. Ramani KV & Mavalankar Dileep, op. cit.
42. "Out-of-pocket spending on health care high: Report", Business Line, (December 10, 2008).
43. Department of Pharmaceuticals, Third Round Up of Developments in the Pharmaceutical Sector (July, 2009) and PwC Analysis.
44. Usha Sharma, “Nurturing Industry Needs”, Express Pharma (June 2009).
45. Sudip Chaudhuri, “Product Patent Protection and Development of the Pharmaceutical Industry: India’s Experience and its Relevance for LDCs in
Africa” (June 7, 2008), Indian Institute of Management.
46. CSM Services, Pharmaceutical Industry (March 25 2008), accessed July 7, 2009, http://www.csmservicesindia .com/news_details.asp?id=36
47. “$70 bn worth drugs to go off-patent, beneting India: Report”, The Economic Times (August 2, 2009).
34 PricewaterhouseCoopers
48. Joe C Mathew, “USFDA door wide open for Indian pharma cos” Business Standard, (March 6, 2009).
49. Department of Pharmaceuticals, op. cit, and PwC Analysis.
50. Dr. Reddy’s Laboratories Annual Report (2008-09) and PwC Analysis
51. Cipla Annual Report (2008-09) and PwC Analysis.
52. “BioSpectrum Top 20: Cipla (5)” BioSpectrum Asia (August 26, 2008), accessed July 10, 2009 http://www.biospectrumasia.com/
content/260808IND6931.asp.
53. “Avesthagen-CIPLA JV acquires state of the art biological company in Germany” (October 05, 2007), Avesta Biotherapeutics
54. Lupin Pharmaceuticals website, accessed May 30, 2009, http://www.lupinpharmaceuticals.com/mandideep.htm
55. “Lupin acquires majority stake in Multicare Pharmaceuticals Philippines, Inc.” (March 26, 2009), Lupin Pharmaceuticals
56. IndusView, Volume 4, Issue 5 (April 10, 2009).
57. Arshiya Khan, “India targets the world”, Express Pharma (September 16, 2008).
58. “Everest Capital, CVCI Invest $24 Million In Nectar Life sciences”, Vccircle.com (August 25 2008).
59. “Kotak PE pumps in $50 m into biotech cos”, The Financial Express (April 12, 2008).
60. “Will PE drive pharma?”, Express Pharma (16 November 2008).
61. Deutsche Bank, op. cit.
62. ‘India CMO biz set for 23% CAGR’ (March 2008), BioSpectrum Asia,– access at http://www.drreddys.com/media/pdf/bio_spectrum_mar2008.pdf
63. Crisil Research, “Bulk drug exports to scale up in the regulated markets” (December 2008).
64. US Department of Health & Human Services, “HHS Opens U.S. Food and Drug Administration Ofces in India” (January 15, 2009), accessed July
3, 2009, http://www.hhs.gov/news/press/2009pres/01/20090115a.html
65. “FOCUS: FDA Heat Fails To Scorch Appeal Of Indian Pharma” Dow Jones Newswire, (July 28, 2009).
66. “Lean Practices in a Life Sciences Organization”, Contract Pharma (October 2007).
67. Srinivas Rao, “Vaccine majors drive business”, Biospectrum (July 08, 2009).
68. “WHO lifts vaccine embargo on India”, Livemint along with The Wall Street Journal (April 20, 2009).
69. Srinivas Rao, “Vaccine majors drive business”, op. cit
70. "Piramal buys Cipla’s i-Pill for Rs 95 crore", The Times of India (March 24, 2010) and PwC Analysis.
71. “Now, Novartis’ Otrivin spray will be available over the counter”, Business Line, (October 01, 2009).
72. “Pzer sells Benadryl, Listerine to Johnson & Johnson” (January 01, 2008), Rediff News accessed October 10, 2009, http://www.rediff.com/
money /2008/jan/01list.htm
73. OTC drugs turn money spinners”, The Economic Times (23 September, 2009).
74. Pharmaceutical Outsourcing Landscape in India – An Overview, Zinnov (April 2006).
Global pharma looks to India: Prospects for growth 35
75. Kewal Krishan, “Localization & Language Technology Standards”, National Informatics Centre (2006).
76. Deutsche Bank, op. cit.
77. Biswajit Dhar and K.M.Gopakumar, “Post-2005 TRIPS scenario in patent protection in the pharmaceutical sector: The case of the generic
pharmaceutical industry in India”, (November, 2006).
78. “Impact of Patents on India Pharma Industry’s Growth and Compaison”, Journal of Intellectal Property Rights (September 2009), accessed
September 23, 2009, http://nopr.niscair.res.in/bitstream/123456789/6061/1/JIPR%2014(5)%20432-436.pdf
79. “Schedule Y amendment to help India tap US$ 1 billion by 2010: Experts”, Pharmabiz (September 8, 2005).
80. Joseph Alexander, “Health ministry to add ScheduleY1 to D&C rules to streamline clinical trials sector”, Pharmabiz.com (April 14, 2009).
81. Ministry of Health and Family Welfare, Central Drugs Standard Control Organization, Amendment to Schedule Y of The Drugs and Cosmetics
Rules, 1945, accessed July 7, 2009, http://cdsco.nic.in/html/Schedule-Y%20(Amended%20Version-2005)%20original.htm
82. “GLP certication will help to remove technical barriers”, Express Pharma (16 September 2009).
83. “Lack of globally approved labs may hit drug exports to Europe, Livemint along with The Wall Street Journal (May 27, 2008).
84. Ethical concerns in clinical trials in India: an investigation (February 2009), Centre for Studies in Ethics and Rights, Mumbai, India.http://www.
fairdrugs.org/uploads/les/Ethical_concerns_in_clinical_trials_in_India_An_investigation.pdf
85. 45th Annual meeting of Drug Information Association & FICCI, "Evolving regulatory scenario in India" (San Diego 2009)
86. “Clinical Research India: the Need for Education & Training”, Journal for Clinical Studies (November 2008).
87. United Nations Population Fund, “State of World Population 2007” (2007).
88. Central Bureau of Health Intelligence, “National Health Prole 2008” (2008).
89. Omkar S. Kanwar, “Global Clinical Trial in India:Prospect and Challenges” , FICCI, (November 2005), accessed July 3, 2009, www.cci.com/
media-room/speeches-presentations/2005/nov/bio-welcoem.pdf
90. “New Drugs, India Style”, Forbes Asia (October 5, 2009).
91. Alan Davies and Janet Jones “Tracking CRO Growth”, Pharma Focus Asia Issue 9 (2008)
92. Raja M. Mitra, “India’s Emergence as a Global R&D Center”, ITPS, Swedish Institute for Growth Policy Studies (July 2007).
93. “BioSpectrum-ABLE Top 20 Companies”, Biospectrum (June 2009).
94. Federation of Indian Chambers of Commerce and Industry, “Overview of the Biotech Sector in India” (2005).
95. “BioSpectrum-ABLE Top 20 Companies” op. cit
96. Joseph Alexander, “Draft bill to set up National Biotechnology Regulatory Authority ready” Pharmabiz.com, (July 01, 2008) and N Suresh,
“Glimpses into the new biotech regulatory set-up”, Biospectrum India (May 07, 2008).
97. “Biosimilar market growth depends on the US: DataMonitor”, Biospectrum Asia (June 23, 2009).
98. “Biocon, Dr Reddy’s, Reliance Life, Intas to dominate $19.4 bn global biosimilars market by 2014” (September 9, 2009), accessed August 7, 2009,
http://www.dancewithshadows.com/pillscribe/biocon-dr-reddys-reliance-life-intas-to-dominate-19-4-bn-global-biosimilars-market-by-2014/
36 PricewaterhouseCoopers
99. “Apotex and Intas to Develop Biosimilar of Peglgrastim” , BioPharm (February 11, 2009), accessed July 7, 2009, http://biopharminternational.
ndpharma. com/biopharm/News/Apotex-and-Intas-to-Develop-Biosimilar-of-Peglgr/ArticleStandard/Article/detail/580879
100. “Biocon, Dr Reddy’s, Reliance Life, Intas to dominate $19.4 bn global biosimilars market by 2014”, op. cit.
101. Narayanan Suresh, “The door opens slightly for biosimilars in US”, BiospectrumAsia (May 5, 2009).
102. Four Corners of the World (2005), Wockhardt, accessed August 8, 2009, http://www.wockhardtin. com/pdf1/2005/4_Four_Corners_of_the_world.
pdf
103. US National Institutes of Health (October 14, 2008) accessed August 3, 2009, http://clinicaltrials.gov/ct2/show/NCT00772265
104. “Biocon, Dr Reddy’s, Reliance Life, Intas to dominate $19.4 bn global biosimilars market by 2014”, op. cit.
105. “Growth Slows”, Biospectrum (June 2009).
106. “Bangalore Helix to open on June 7”, op. cit.
107. Ocimum Biosolutions receives US patent for gene expression data quality metrics”, Pharmabiz.com (July 16, 2009).
108. “New Sun Center Of Excellence Inaugurated in India” (August 15, 2005), Sun Systemnews, accessed May 24, 2009, https://sun.systemnews.com/
articles/90/ 3/news/14923
109. Sushmi Dey, “Selling stem cells”, Express Pharma (December 15, 2007).
110. “Stempeutics Launches its Second State-of-the-art Lab”, ExpressHealthcare, (August 2008).
111. Sonal Shukla, “Banking on Stem Cells”, ExpressHealthcare (May 2009).
112. Medical Devices and Equipment in India (June 2009), Cygnus Research
113. “Medical Devices: Sector Analysis, Recommendation for NIPER-Ahmedabad” (June 2009), National Institute of Pharmaceutical Education and
Research.
114. “Guidelines For Import And Manufacture Of Medical Devices” (October 2005), Ministry of Commerce accessed September 11, 2009, http://
commerce.nic.in/wto sub/TBT%20Notications%20by%20INDIA/19-S.O.%201468%20(E).pdf
115. Cipla annual report (2008-09); Sun Pharmaceuticals annual report (2008-09); GSK annual report (2008); Cadila Healthcare annual report (2008-09);
Piramal Healthcare annual report (2008-09); Ranbaxy (Daiichi) annual report (2008); Dr. Reddy’s Laboratories annual report (2008-09); Aurobindo
Pharmaceuticals annual report (2008-09); Lupin annual report (2008-09), Lupin Investor Presentation (2008-09) and Lupin Corporate Presentation
(2009-09); Glenmark annual report (2008-09).
116. “Elder Pharma to sell Enzymotec drug in India”, Livemint along with The Wall Street Journal (July 9, 2007).
117. “Elder Pharmaceuticals Inks In-Licensing Pact With Daiwa Pharmaceutical Co Ltd, Japan” (October 10, 2008), EquityBulls.com accessed
September 6, 2009, http://www.equitybulls.com/admin/news2006/news_det.asp?id=42023
118. PB Jayakumar, “Pharma rms eye in-licensing opportunities”, Business Standard, (April 26, 2007).
119. “Ethypharm Out-licenses Mesalazine NDDS to Solvay”, Merinews (Jan 21, 2009), accessed September 8, 2009, http://www.merinews.com/
catFull.jsp? articleID=15708992
Global pharma looks to India: Prospects for growth 37
120. Rupali Mukherjee, “Desi pharma companies grow beyond generics”, The Times of India (November 26, 2007).
121. “Claris Life eyes 2-3 bln rupee IPO in 12-18 months”, Reuters India (June 18, 2009).
122. “Pharmacy chains Rx for change” (2007), India Brand Equity Federation
123. “Pharmacy chains Rx for change” (2007) op. cit
124. “India’s health biotech sector at a crossroads”, Nature Biotechnology 25, 403 - 417 (2007).
125. “Cadila Launches Joint Venture With Novavax In India”, Novavax (July 9,2009), accessed July 16, 2009, http://www.novavax.com/download/
releases/CadPR_FO%5B1%5D.pdf
126. “Cadila Pharmaceuticals Launches Joint Venture With Novavax in India”, Reuters India (July 9, 2009).
127. “Novotech Signs Joint Venture with Indian CRO ETI Klinical”, Applied Clinical Trials Online (May 6, 2009), accessed Sptember 7, 2009, http://
appliedclinicaltrials online.ndpharma.com/appliedclinicaltrials/article/articleDetail.jsp?id=596897
128. Nina Mehta, “Pzer increases stake in Indian arm to 72%” , The Economic Times (18 July 2009).
129. “Novartis raises stake in Indian arm to 76.42%”, Business Standard (June 25, 2009).
130. Rumi Dutta, “Pharma multinationals activating Indian arms”, Business Standard (February 01, 2005).
131. “Pharma cargo zones planned at airports”, Business Standard (March 25, 2009).
132. 2010-11 Budget Key Features, accessed March 1, 2010, http://indiabudget.nic.in/ub2010-11/download_index.htm
133. Arun S, “Task force prescribes policy change to help pharma SEZs” (May 07, 2009), Financial Express.
134. “Critical areas on DTC for detailed examination identied: FM” (October 09, 2009), Ministry of Finance.
135. 2010-11 Budget Key Features. op. cit.
136. Radhieka Pandeya, “supply of fake drugs grossly overstated”, The Live Mint Journal (November 26, 2009).
137. The Patents (Amendment) Act, 2005, accessed July 6, 2009, www.patentofce.nic.in/ipr/patent/patent_2005.pdf
138. The third proviso, Section 11 A Sub-section (7) of The Patents (Amendment) Act, 2005.
139. Department of Chemicals & Petrochemicals, Report on Steps to be taken by Government of India in the context of Data Protection Provisions of
Article 39.3 of TRIPS Agreement” (May 31, 2007).
140. Khomba Singh, “Delhi HC debarred Generic durgmakers from marketing”, The Economic Times (January 7, 2009), accessed July 21, 2009, http://
economictimes .indiatimes.com/News/Delhi_HC_debarred_Generic_durgmakers_from_marketing/rssarticleshow/3946637.cms
141. PB Jayakumar, “Novartis loses battle for cancer drug patent”, Business Standard (July 20, 2009); India Glivec Patent Case FAQ, Novartis,
accessed July 21, 2009, http://www.novartis.com/downloads /about-novartis/india-glivec-patent-case-faq.pdf
38 PricewaterhouseCoopers
Acronyms
ANDA - Abbreviated New Drug Application
API - Active Pharmaceutical Ingredient
BCG - Bacille Calmette-Guérin
BIPP - Biotechnology Industry Partnership Programme
BPL - Below Poverty Line
CAGR - Compounded Annual Growth Rate
CRAMS - Contract Research & Manufacturing Services
CROs - Contract Research Organisations
CVD - Countervailing Duty
DBT - Department of Biotechnology
DCGI - Drug Controller General of India
DPCO - Drug Price Control Order
DPT - Diphtheria, Pertussis or Whooping Cough,
and Tetanus
DRL - Dr. Reddy’s Laboratories
EMEA - European Medicines Agency
EU - European Union
FCCBs - Foreign Currency Convertible Bonds
FDA - Food and Drug Administration
FDI - Foreign Direct Investment
FIPB - Foreign Investment Promotion Board
GCSF - Granulocyte Colony Stimulating Factor
GIC - General Insurance Company
GLP - Good Laboratory Practices
GSK - GlaxoSmithKline
GST - Goods and Sales Tax
hGH - Human Growth Hormone
IP - Intellectual Property
IPAB - Intellectual Property Appellate Board
IRDA - Insurance Regulatory and
Development Authority
JJMI - Johnson and Johnson Medical India
JVs - Joint ventures
NPPA - National Pharmaceutical Pricing Authority
NREGA - National Rural Employment Guarantee Act
NRHM - National Rural Health Mission
OPPI - Organisation of Pharmaceutical Producers
of India
OTC - Over The Counter
PE - Private Equity
PPPs - Public-Private Partnerships
PwC - PricewaterhouseCoopers
R&D - Research and Development
RBI - Reserve Bank of India
SEZs - Special Economic Zones
SIA - Secretariat for Industrial Assistance
TRIPS - Trade Related Aspects of Intellectual
Property Rights
WHO - World Health Organisation

Territory industry contacts
Argentina
Diego Niebuhr
[54] 11 4850 4705
Australia
John Cannings
[61] 2 826 66410
Belgium
Thierry Vanwelkenhuyzen
[32] 2 710 7422
Bolivia
César Lora
[591] 2 2408 230
Brazil (SOACAT)
Luis Madasi
[55] 11 3674 1520
Canada
Gord Jans
[1] 905 897 4527
China
Beatrijs van Liedekerke
[81] 10 6533 7223
Chile
Ricardo Arrano
[55] 31 3269 1551
Colombia
María Helena Díaz
[57] (1) 635 1125
Nacira Lamprea
[57] 1 634 0555
Czech Republic
Radmila Fortova
[420] 2 5115 2521
Denmark
Torben TOJ Jensen
[45] 3 945 9243
Erik Todbjerg
[45] 3 945 9433
Ecuador
Carlos Cruz
[593] 2 2562 288 130
Finland
Janne Rajalahti
[358] 3 3138 8016
Johan Kronberg
[358] 9 2280 1253
France
Jacques Denizeau
[33] 1 56 57 10 55
Germany
Volker Booten
[49] 89 5790 6347
India
Sharat Bansal
[91] 22 6669 1538
Indonesia
Eddy Rintis
[62] (21) 521 4048
Mirza Diran
[62] (21) 528 90950
Ireland
John M Kelly
[353] 1 792 6307
Enda McDonagh
[353] 1 792 8728
Israel
Assaf Shemer
[972] 3 795 4681
Italy
Massimo Dal Lago
[39] 045 8002561
Japan
Kenichiro Abe
[81] 80 3158 5929
Korea
Henry An
[82] (2) 3781 2594
Luxembourg
Laurent Probst
[352] 49 48 48 2199
Mexico
Jorge Luis Hernandez
[52] 55 5263 6106
Netherlands
Arwin van der Linden
[31] 20 5684712
Poland
Mariusz Ignatowicz
[48] 22 523 4795
Portugal
Ana Lopes
[351] 213 599 159
Peru
Felix Horna
[5] 11 211 6500
Russia
Alina Lavrentieva
[7] 495 967 6250
Singapore
Abhijit Ghosh
[65] 6236 3888
South Africa
Denis von Hoesslin
[27] 117 974 285
Spain
Rafael Rodríguez Alonso
[34] 91 568 4287
Sweden
Liselott Stenudd
[46] 8 555 33 405
Switzerland
Clive Bellingham
[41] 58 792 2822
Peter Kartscher
[41] 58 792 5630
Taiwan
Elliot Liao
[886] (2) 2729 6217
Thailand
Zoya Vassilieva
[66] (2) 344 1115
Turkey
Zeki Gündüz
[90] 212 326 64 10
United Kingdom
Andy Kemp
[44] 20 7804 4408
United States
Michael Swanick
[1] 267 330 6060

This publication has been prepared for general guidance on matters of interest only, and does not constitute professional advice. You should not act upon the information contained in this
publication without obtaining specic professional advice. No representation or warranty (express or implied) is given as to the accuracy or completeness of the information contained in this
publication, and, to the extent permitted by law, PricewaterhouseCoopers does not accept or assume any liability, responsibility or duty of care for any consequences of you or anyone else
acting, or refraining to act, in reliance on the information contained in this publication or for any decision based on it.
© 2010 PricewaterhouseCoopers. All rights reserved. "PricewaterhouseCoopers" and "PwC" refer to the network of member rms of PricewaterhouseCoopers International Limited (PwCIL).
Each member rm is a separate legal entity and does not act as agent of PwCIL or any other member rm. PwCIL does not provide any services to clients. PwCIL is not responsible or liable
for the acts or omissions of any of its member rms nor can it control the exercise of their professional judgment or bind them in any way. No member rm is responsible or liable for the acts or
omissions of any other member rm nor can it control the exercise of another member rm's professional judgment or bind another member rm or PwCIL in any way.
For further information, please contact:
pwc.com/pharma
Global
Simon Friend
Partner, Global Pharmaceutical & Life Sciences
Industry Leader
PricewaterhouseCoopers (UK)
[44] 20 7213 4875
Steve Arlington
Partner, Global Pharmaceutical & Life Sciences Advisory
Services Leader
PricewaterhouseCoopers (UK)
[44] 20 7804 3997
Michael Swanick
Partner, Global Pharmaceutical and Life Sciences Tax Leader
PricewaterhouseCoopers (US)
[1] 267 330 6060
Marketing
Sara Solomon
Marketing and Business Development
Global Pharmaceutical & Life Sciences
PricewaterhouseCoopers (UK)
[44] 20 7804 1014
India
Sujay Shetty
Director, India Pharmaceutical & Life Sciences
Industry Leader
PricewaterhouseCoopers (India)
sujay[email protected]
[91] 22 6669 1305
Nisha Vishwakarma
Manager, India Pharmaceutical & Life Sciences
PricewaterhouseCoopers (India)
[91] 22 6669 1100
