
How to use the child and adolescent immunization
schedule
Recommended by the Advisory Committee on Immunization Practices (www.cdc.gov/vaccines/acip)
and approved by the Centers for Disease Control and Prevention (www.cdc.gov), American Academy
of Pediatrics (www.aap.org), American Academy of Family Physicians (www.aafp.org), American
College of Obstetricians and Gynecologists (www.acog.org), American College of Nurse-Midwives
(www.midwife.org), American Academy of Physician Associates (www.aapa.org), and National
Association of Pediatric Nurse Practitioners (www.napnap.org).
Vaccines and Other Immunizing Agents in the Child and Adolescent Immunization Schedule*
Monoclonal antibody Abbreviation(s) Trade name(s)
Respiratory syncytial virus monoclonal antibody (Nirsevimab) RSV-mAb Beyfortus™
Vaccine Abbreviation(s) Trade name(s)
COVID-19 1vCOV-mRNA Comirnaty®/Pzer-
BioNTech COVID-19
Vaccine
Spikevax®/Moderna
COVID-19 Vaccine
1vCOV-aPS Novavax COVID-19
Vaccine
Dengue vaccine DEN4CYD Dengvaxia®
Diphtheria, tetanus, and acellular pertussis vaccine DTaP Daptacel®
Infanrix®
Haemophilus inuenzae type b vaccine Hib (PRP-T)
Hib (PRP-OMP)
ActHIB®
Hiberix®
PedvaxHIB®
Hepatitis A vaccine HepA Havrix®
Vaqta®
Hepatitis B vaccine HepB Engerix-B®
Recombivax HB®
Human papillomavirus vaccine HPV Gardasil 9®
Inuenza vaccine (inactivated) IIV4 Multiple
Inuenza vaccine (live, attenuated) LAIV4 FluMist® Quadrivalent
Measles, mumps, and rubella vaccine MMR M-M-R II®
Priorix®
Meningococcal serogroups A, C, W, Y vaccine MenACWY-CRM Menveo®
MenACWY-TT MenQuad®
Meningococcal serogroup B vaccine MenB-4C Bexsero®
MenB-FHbp Trumenba®
Meningococcal serogroup A, B, C, W, Y vaccine MenACWY-TT/
MenB-FHbp
Penbraya™
Mpox vaccine Mpox Jynneos®
Pneumococcal conjugate vaccine PCV15
PCV20
Vaxneuvance™
Prevnar 20®
Pneumococcal polysaccharide vaccine PPSV23 Pneumovax 23®
Poliovirus vaccine (inactivated) IPV Ipol®
Respiratory syncytial virus vaccine RSV Abrysvo™
Rotavirus vaccine RV1
RV5
Rotarix®
RotaTeq®
Tetanus, diphtheria, and acellular pertussis vaccine Tdap Adacel®
Boostrix®
Tetanus and diphtheria vaccine Td Tenivac®
Tdvax™
Varicella vaccine VAR Varivax®
Combination vaccines (use combination vaccines instead of separate injections when appropriate)
DTaP, hepatitis B, and inactivated poliovirus vaccine DTaP-HepB-IPV Pediarix®
DTaP, inactivated poliovirus, and Haemophilus inuenzae type b vaccine DTaP-IPV/Hib Pentacel®
DTaP and inactivated poliovirus vaccine DTaP-IPV Kinrix®
Quadracel®
DTaP, inactivated poliovirus, Haemophilus inuenzae type b, and
hepatitis B vaccine
DTaP-IPV-Hib-
HepB
Vaxelis®
Measles, mumps, rubella, and varicella vaccine MMRV ProQuad®
* Administer recommended vaccines if immunization history is incomplete or unknown. Do not restart or add doses to vaccine series for
extended intervals between doses. When a vaccine is not administered at the recommended age, administer at a subsequent visit.
The use of trade names is for identication purposes only and does not imply endorsement by the ACIP or CDC.
Report
y Suspected cases of reportable vaccine-preventable diseases or outbreaks to your state or local health
department
y Clinically signicant adverse events to the Vaccine Adverse Event Reporting System (VAERS) at
www.vaers.hhs.gov or 800-822-7967
Questions or comments
Contact www.cdc.gov/cdc-info or 800-CDC-INFO (800-232-4636), in English or Spanish, 8 a.m.–8 p.m. ET,
Monday through Friday, excluding holidays
Helpful information
y Complete Advisory Committee on Immunization Practices (ACIP) recommendations:
www.cdc.gov/vaccines/hcp/acip-recs/index.html
y ACIP Shared Clinical Decision-Making Recommendations:
www.cdc.gov/vaccines/acip/acip-scdm-faqs.html
y General Best Practice Guidelines for Immunization (including contraindications and precautions):
www.cdc.gov/vaccines/hcp/acip-recs/general-recs/index.html
y Vaccine information statements:
www.cdc.gov/vaccines/hcp/vis/index.html
y Manual for the Surveillance of Vaccine-Preventable Diseases
(including case identication and outbreak response):
www.cdc.gov/vaccines/pubs/surv-manual
1
Determine
recommended
vaccine by age
(Table1)
2
Determine
recommended
interval for catch-
up vaccination
(Table2)
3
Assess need
for additional
recommended
vaccines
by medical
condition or
other indication
(Table3)
4
Review
vaccine types,
frequencies,
intervals, and
considerations
for special
situations
(Notes)
5
Review
contraindications
and precautions
for vaccine types
(Appendix)
6
Review new or
updated ACIP
guidance
(Addendum)
Download the CDC Vaccine Schedules app for providers at
www.cdc.gov/vaccines/schedules/hcp/schedule-app.html
CS310020-D
06/27/2024
Scan QR code
for access to
online schedule
Recommended Child and Adolescent Immunization Schedule
for ages 18 years or younger
UNITED STATES
2024
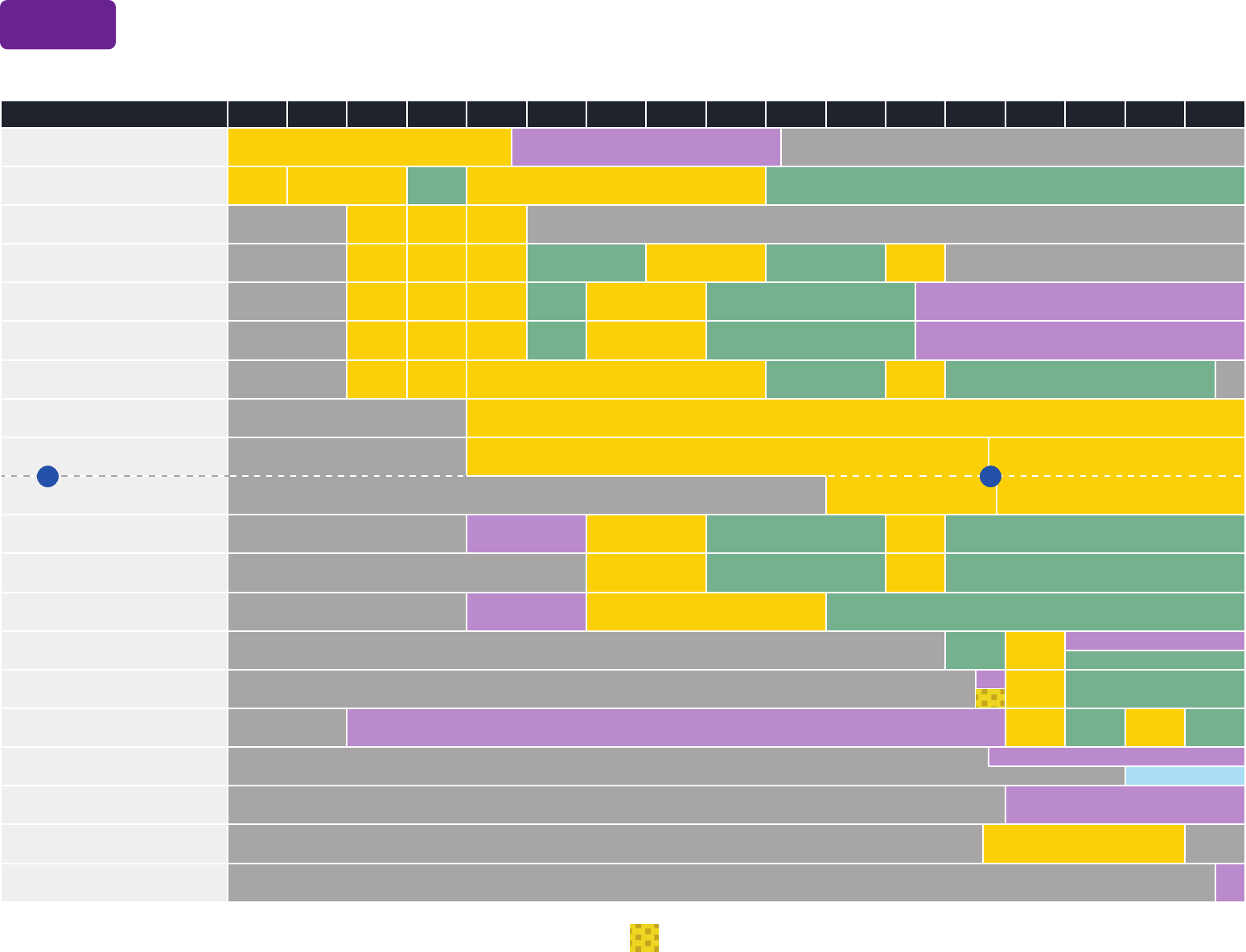
These recommendations must be read with the notes that follow. For those who fall behind or start late, provide catch-up vaccination at the earliest opportunity as indicated by the green bars.
To determine minimum intervals between doses, see the catch-up schedule (Table 2).
Vaccine and other immunizing agents Birth 1 mo 2 mos 4 mos 6 mos 9 mos 12 mos 15 mos 18 mos 19–23 mos 2–3 yrs 4–6 yrs 7–10 yrs 11–12 yrs 13–15 yrs 16 yrs 17–18 yrs
Respiratory syncytial virus
(RSV-mAb [Nirsevimab])
1 dose depending on maternal
RSV vaccination status, See Notes
1 dose (8 through 19 months), See Notes
Hepatitis B (HepB) 1
st
dose ----- 2
nd
dose ----- ---------------------------- 3
rd
dose ----------------------------
Rotavirus (RV): RV1 (2-dose series),
RV5 (3-dose series)
1
st
dose 2
nd
dose See Notes
Diphtheria, tetanus, acellular pertussis
(DTaP <7 yrs)
1
st
dose 2
nd
dose 3
rd
dose ----- 4
th
dose ------ 5
th
dose
Haemophilus inuenzae type b (Hib) 1
st
dose 2
nd
dose See Notes
3
rd
or 4
th
dose,
--
See Notes
--
Pneumococcal conjugate
(PCV15, PCV20)
1
st
dose 2
nd
dose 3
rd
dose ----- 4
th
dose -----
Inactivated poliovirus
(IPV <18yrs)
1
st
dose 2
nd
dose ---------------------------- 3
rd
dose ---------------------------- 4
th
dose
See
Notes
COVID-19 (1vCOV-mRNA, 1vCOV-aPS) 1 or more doses of updated (2023–2024 Formula) vaccine (See Notes)
Inuenza (IIV4) Annual vaccination 1 or 2 doses Annual vaccination 1 dose only
Inuenza (LAIV4)
Annual vaccination
1 or 2 doses
Annual vaccination 1 dose only
Measles, mumps, rubella (MMR) See Notes ----- 1
st
dose ----- 2
nd
dose
Varicella (VAR) ----- 1
st
dose ----- 2
nd
dose
Hepatitis A (HepA) See Notes 2-dose series, See Notes
Tetanus, diphtheria, acellular pertussis
(Tdap ≥7 yrs)
1 dose
Human papillomavirus (HPV)
See
Notes
Meningococcal (MenACWY-CRM ≥2 mos,
MenACWY-TT ≥2years)
See Notes 1
st
dose 2
nd
dose
Meningococcal B
(MenB-4C, MenB-FHbp)
See Notes
Respiratory syncytial virus vaccine
(RSV [Abrysvo])
Seasonal administration
during pregnancy, See Notes
Dengue (DEN4CYD; 9-16 yrs)
Seropositive in endemic
dengue areas (See Notes)
Mpox
oror
Range of recommended
ages for all children
Range of recommended ages
for catch-up vaccination
Range of recommended ages
for certain high-risk groups
Recommended vaccination
can begin in this age group
Recommended vaccination based
on shared clinical decision-making
No recommendation/
not applicable
Table 1
Recommended Child and Adolescent Immunization Schedule for Ages 18 Years or Younger, United States, 2024

The table below provides catch-up schedules and minimum intervals between doses for children whose vaccinations have been delayed. A vaccine series does not need to be restarted, regardless of the time that has elapsed
between doses. Use the section appropriate for the child’s age. Always use this table in conjunction with Table 1 and the Notes that follow.
Children age 4 months through 6 years
Vaccine Minimum Age for
Dose 1
Minimum Interval Between Doses
Dose 1 to Dose 2 Dose 2 to Dose 3 Dose 3 to Dose 4 Dose 4 to Dose 5
Hepatitis B Birth 4 weeks 8 weeks and at least 16 weeks after rst dose
minimum age for the nal dose is 24 weeks
Rotavirus 6 weeks
Maximum age for rst
dose is 14weeks, 6 days.
4 weeks 4 weeks
maximum age for nal dose is 8 months, 0 days
Diphtheria, tetanus, and
acellular pertussis
6 weeks 4 weeks 4 weeks 6 months 6 months
A fifth dse is nt necessry
if the furth dse ws
dministered t ge 4 yers r
lder nd t lest 6 mnths fter
dse 3
Haemophilus inuenzae
typeb
6 weeks No further doses needed
if rst dose was administered at age 15
months or older.
4 weeks
if rst dose was administered before the
1
st
birthday.
8 weeks (as nal dose)
if rst dose was administered at age
12through 14 months.
No further doses needed
if previous dose was administered at age 15 months or older
4 weeks
if current age is younger than 12 months and rst dose was administered at younger than age 7 months and
at least 1 previous dose was PRP-T (ActHib®, Pentacel®, Hiberix®), Vaxelis® or unknown
8 weeks and age 12 through 59 months (as nal dose)
if current age is younger than 12 months and rst dose was administered at age 7 through 11 months; OR
if current age is 12 through 59 months and rst dose was administered before the 1
st
birthday and second
dose was administered at younger than 15 months; OR
if both doses were PedvaxHIB® and were administered before the 1st birthday
8 weeks (as nal dose)
This dose only necessary
for children age 12 through
59months who received
3doses before the
1
st
birthday.
Pneumococcal conjugate 6 weeks No further doses needed for healthy
children if rst dose was administered at
age 24 months or older
4 weeks
if rst dose was administered before the
1
st
birthday
8 weeks (as nal dose for healthy
children)
if rst dose was administered at the
1
st
birthday or after
No further doses needed
for healthy children if previous dose was administered at age 24 months or older
4 weeks
if current age is younger than 12 months and previous dose was administered at <7 months old
8 weeks (as nal dose for healthy children)
if previous dose was administered between 7–11 months (wait until at least 12 months old); OR
if current age is 12 months or older and at least 1 dose was administered before age 12 months
8 weeks (as nal dose)
This dose is only necessary
for children age 12 through
59 months regardless of risk,
or age 60 through 71 months
with any risk, who received 3
doses before age 12 months.
Inactivated poliovirus 6 weeks 4 weeks 4 weeks
if current age is <4 years
6 months (as nal dose)
if current age is 4 years or older
6 months (minimum age 4
years for nal dose)
Measles, mumps, rubella 12 months 4 weeks
Varicella 12 months 3 months
Hepatitis A 12 months 6 months
Meningococcal ACWY 2 months MenACWY-CRM
2 years MenACWY-TT
8 weeks See Notes See Notes
Children and adolescents age 7 through 18 years
Meningococcal ACWY Not applicable (N/A) 8 weeks
Tetanus, diphtheria;
tetanus, diphtheria, and
acellular pertussis
7 years 4 weeks 4 weeks
if rst dose of DTaP/DT was administered before the 1
st
birthday
6 months (as nal dose)
if rst dose of DTaP/DT or Tdap/Td was administered at or after the 1
st
birthday
6 months
if rst dose of DTaP/DT was
administered before the 1
st
birthday
Human papillomavirus 9 years Routine dosing intervals are
recommended.
Hepatitis A N/A 6 months
Hepatitis B N/A 4 weeks 8 weeks and at least 16 weeks after rst dose
Inactivated poliovirus N/A 4 weeks 6 months
A fourth dose is not necessary if the third dose was administered at age 4 years or older and at least 6months
after the previous dose.
A fourth dose of IPV is
indicated if all previous doses
were administered at <4
years OR if the third dose was
administered <6 months after
the second dose.
Measles, mumps, rubella N/A 4 weeks
Varicella N/A 3 months if younger than age 13 years.
4 weeks if age 13 years or older
Dengue 9 years 6 months 6 months
Table 2
Recommended Catch-up Immunization Schedule for Children and Adolescents Who Start Late or Who Are More
than 1 Month Behind, United States, 2024
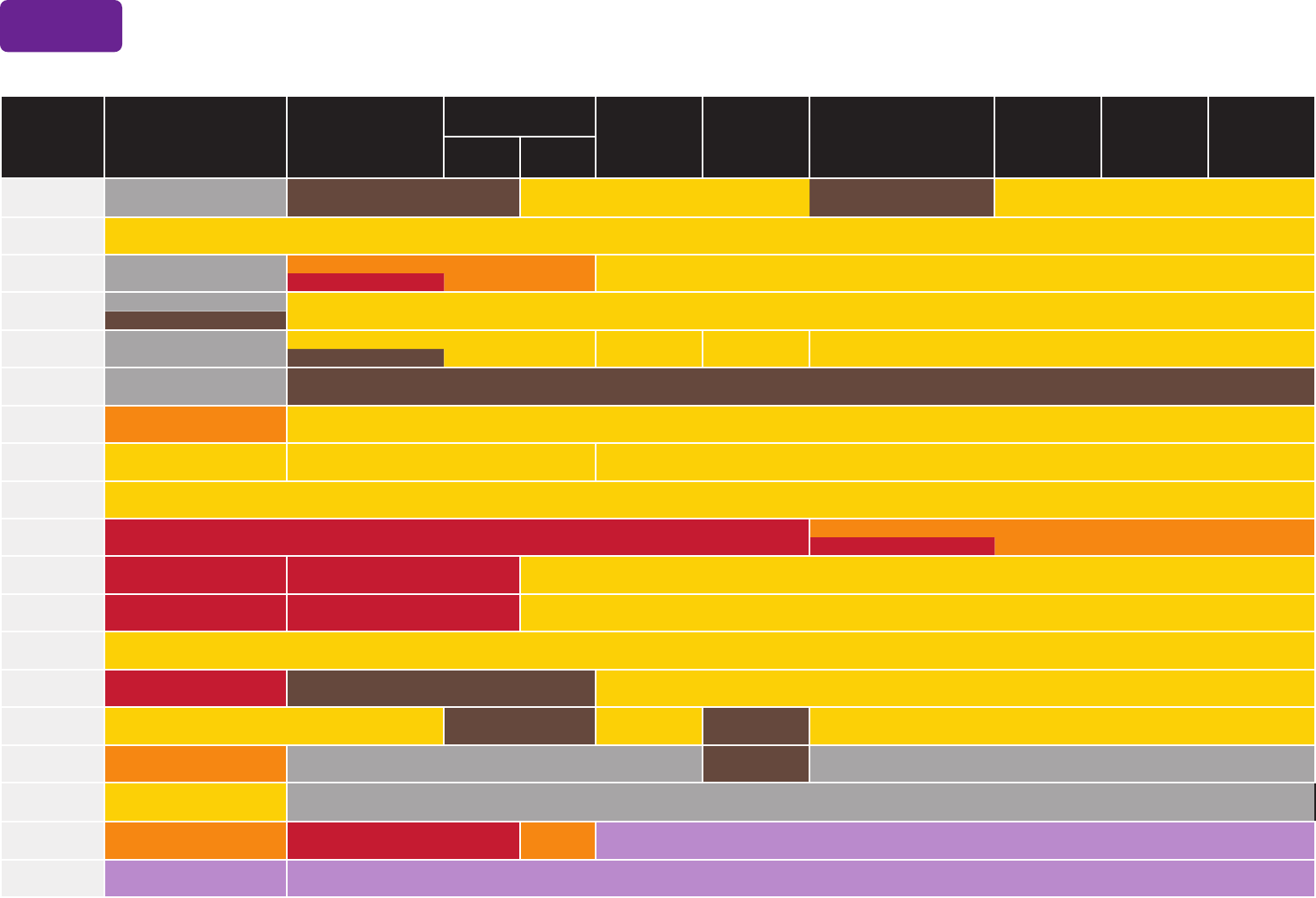
Always use this table in conjunction with Table 1 and the Notes that follow. Medical conditions are often not mutually exclusive. If multiple conditions are present, refer to guidance in all relevant
columns. See Notes for medical conditions not listed.
Vaccine
and other
immunizing
agents Pregnancy
Immunocompromised
(excluding HIV
infection)
HIV infection CD4
percentage and count
a
CSF leak or
cochlear
implant
Asplenia or
persistent
complement
component
deciencies
Heart disease or
chronic lung disease
Kidney failure,
End-stage
renal disease
or on Dialysis
Chronic liver
disease Diabetes
<15% or
<200mm
≥15% and
≥200mm
RSV-mAb
(nirsevimab)
2nd RSV season
1 dose depending on maternal
RSV vaccination status, See Notes
2nd RSV season for chronic
lung disease (See Notes)
1 dose depending on maternal
RSV vaccination status, See Notes
Hepatitis B
Rotavirus
SCID
b
DTaP/Tdap
DTaP
Tdap: 1 dose each pregnancy
Hib See Notes See Notes
HSCT: 3 doses
Pneumococcal
IPV
COVID-19 See Notes
IIV4
LAIV4
Asthma, wheezing: 2–4 years
c
MMR *
VAR *
Hepatitis A
HPV * 3 dose series. See Notes
MenACWY
MenB
RSV (Abrysvo)
Seasonal administration,
See Notes
Dengue
Mpox See Notes
Recommended for all age-
eligible children who lack
documentation of a complete
vaccination series
Not recommended for all children,
but is recommended for some
children based on increased risk for
or severe outcomes from disease
Recommended for all age-eligible
children, and additional doses may be
necessary based on medical condition
or other indications. See Notes.
Precaution: Might be
indicated if benet of
protection outweighs
risk of adverse reaction
Contraindicated or not
recommended
*Vaccinate after pregnancy,
if indicated
No Guidance/
Not Applicable
Table 3
Recommended Child and Adolescent Immunization Schedule by Medical Indication, United States, 2024
a. For additional information regarding HIV laboratory parameters and use of live vaccines, see the General Best Practice Guidelines for Immunization,
“Altered Immunocompetence,” at www.cdc.gov/vaccines/hcp/acip-recs/general-recs/immunocompetence.html and Table 4-1 (footnote J) at
www.cdc.gov/vaccines/hcp/acip-recs/general-recs/contraindications.html.
b. Severe Combined Immunodeciency
c. LAIV4 contraindicated for children 2–4 years of age with
asthma or wheezing during the preceding 12 months

For vaccination recommendations for persons ages
19 years or older, see the Recommended
Adult Immunization Schedule, 2024.
Additional information
y For calculating intervals between doses, 4 weeks = 28 days.
Intervals of ≥4 months are determined by calendar months.
y Within a number range (e.g., 12–18), a dash (–) should
be read as “through.”
y Vaccine doses administered ≤4 days before the minimum
age or interval are considered valid. Doses of any vaccine
administered ≥5 days earlier than the minimum age or
minimum interval should not be counted as valid and
should be repeated as age appropriate. The repeat
dose should be spaced after the invalid dose by the
recommended minimum interval. For further details,
see Table 3-2, Recommended and minimum ages and
intervals between vaccine doses, in General Best Practice
Guidelines for Immunization at www.cdc.gov/vaccines/hcp/
acip-recs/general-recs/timing.html.
y Information on travel vaccination requirements and
recommendations is available at www.cdc.gov/travel/.
y For vaccination of persons with immunodeciencies, see
Table 8-1, Vaccination of persons with primary and secondary
immunodeciencies, in General Best Practice Guidelines for
Immunization at www.cdc.gov/vaccines/hcp/acip-recs/
general-recs/immunocompetence.html, and Immunization in
Special Clinical Circumstances (In: Kimberlin DW, Barnett ED,
Lyneld Ruth, Sawyer MH, eds. Red Book: 2021–2024 Report
of the Committee on Infectious Diseases. 32
nd
ed. Itasca, IL:
American Academy of Pediatrics; 2021:72–86).
y For information about vaccination in the setting of a vaccine-
preventable disease outbreak, contact your
state or local health department.
y The National Vaccine Injury Compensation Program (VICP)
is a no-fault alternative to the traditional legal system for
resolving vaccine injury claims. All vaccines included in
the child and adolescent vaccine schedule are covered by
VICP except dengue, PPSV23, RSV, Mpox and COVID-19
vaccines. Mpox and COVID-19 vaccines are covered by the
Countermeasures Injury Compensation Program (CICP). For
more information, see www.hrsa.gov/vaccinecompensation
or www.hrsa.gov/cicp.
Special situations
Persons who are moderately or severely immunocompromised**
Age 6 months–4 years
y Unvaccinated:
- 3-dose series of updated (2023–2024 Formula) Moderna at
0, 4, 8 weeks
- 3-dose series of updated (2023–2024 Formula) Pzer-
BioNTech at 0, 3, 11 weeks.
y Previously vaccinated* with 1 dose of any Moderna:
2-dose series of updated (2023–2024 Formula) Moderna at
0, 4 weeks (minimum interval between previous Moderna and
dose 1: 4 weeks).
y Previously vaccinated* with 2 doses of any Moderna:
1 dose of updated (2023–2024 Formula) Moderna at least
4 weeks after the most recent dose.
y Previously vaccinated* with 3 or more doses of any
Moderna: 1 dose of updated (2023–2024 Formula) Moderna
at least 8 weeks after the most recent dose.
y Previously vaccinated* with 1 dose of any Pzer-
BioNTech: 2-dose series of updated (2023–2024 Formula)
Pzer-BioNTech at 0, 8 weeks (minimum interval between
previous Pzer-BioNTech and dose 1: 3 weeks).
y Previously vaccinated* with 2 or more doses of any Pzer-
BioNTech: 1 dose of updated (2023–2024 Formula) Pzer-
BioNTech at least 8 weeks after the most recent dose.
Age 5–11 years
y Unvaccinated:
- 3-dose series of updated (2023–2024 Formula) Moderna
at 0, 4, 8 weeks
- 3-dose series updated (2023–2024 Formula) Pzer-BioNTech
at 0, 3, 7 weeks.
y Previously vaccinated* with 1 dose of any Moderna:
2-dose series of updated (2023–2024 Formula) Moderna at
0, 4 weeks (minimum interval between previous Moderna and
dose 1: 4 weeks).
y Previously vaccinated* with 2 doses of any Moderna:
1 dose of updated (2023–2024 Formula) Moderna at least
4 weeks after the most recent dose.
y Previously vaccinated* with 1 dose of any Pzer-
BioNTech: 2-dose series of updated (2023–2024 Formula)
Pzer-BioNTech at 0, 4 weeks (minimum interval between
previous Pzer-BioNTech and dose 1: 3 weeks)
y Previously vaccinated* with 2 doses of any Pzer-
BioNTech: 1 dose of 2023–2024 Pzer-BioNTech at least
4 weeks after the most recent dose.
COVID-19 vaccination
(minimum age: 6 months [Moderna and Pzer-
BioNTech COVID-19 vaccines], 12 years [Novavax
COVID-19 Vaccine])
Routine vaccination
Age 6 months–4 years
y Unvaccinated:
- 2-dose series of updated (2023–2024 Formula) Moderna at
0, 4-8 weeks
- 3-dose series of updated (2023–2024 Formula) Pzer-
BioNTech at 0, 3-8, 11-16 weeks
y Previously vaccinated* with 1 dose of any Moderna:
1 dose of updated (2023–2024 Formula) Moderna 4-8 weeks
after the most recent dose.
y Previously vaccinated* with 2 or more doses of any
Moderna: 1 dose of updated (2023–2024 Formula) Moderna
at least 8 weeks after the most recent dose.
y Previously vaccinated* with 1 dose of any Pzer-
BioNTech: 2-dose series of updated (2023–2024 Formula)
Pzer-BioNTech at 0, 8 weeks (minimum interval between
previous Pzer-BioNTech and dose 1: 3-8 weeks).
y Previously vaccinated* with 2 or more doses of any Pzer-
BioNTech: 1 dose of updated (2023–2024 Formula) Pzer-
BioNTech at least 8 weeks after the most recent dose.
Age 5–11 years
y Unvaccinated: 1 dose of updated (2023–2024 Formula)
Moderna or Pzer-BioNTech vaccine.
y Previously vaccinated* with 1 or more doses of Moderna
or Pzer-BioNTech: 1 dose of updated (2023–2024 Formula)
Moderna or Pzer-BioNTech at least 8 weeks after the most
recent dose.
Age 12–18 years
y Unvaccinated:
- 1 dose of updated (2023–2024 Formula) Moderna or Pzer-
BioNTech vaccine
- 2-dose series of updated (2023–2024 Formula) Novavax at
0, 3-8 weeks
y Previously vaccinated* with any COVID-19 vaccine(s):
1 dose of any updated (2023–2024 Formula) COVID-19
vaccine at least 8 weeks after the most recent dose.
Notes
Recommended Child and Adolescent Immunization Schedule for Ages 18 Years or Younger, United States, 2024

Notes
Recommended Child and Adolescent Immunization Schedule for Ages 18 Years or Younger, United States, 2024
y Previously vaccinated* with 3 or more doses of any
Moderna or Pzer-BioNTech: 1 dose of updated
(2023–2024 Formula) Moderna or Pzer-BioNTech at least
8 weeks after the most recent dose.
Age 12–18 years
y Unvaccinated:
- 3-dose series of updated (2023–2024 Formula) Moderna at
0, 4, 8 weeks
- 3-dose series of updated (2023–2024 Formula) Pzer-
BioNTech at 0, 3, 7 weeks
- 2-dose series of updated (2023–2024 Formula) Novavax at
0, 3 weeks
y Previously vaccinated* with 1 dose of any Moderna:
2-dose series of updated (2023–2024 Formula) Moderna at
0, 4 weeks (minimum interval between previous Moderna
dose and dose 1: 4 weeks).
y Previously vaccinated* with 2 doses of any Moderna:
1 dose of updated (2023–2024 Formula) Moderna at least
4 weeks after the most recent dose.
y Previously vaccinated* with 1 dose of any Pzer-
BioNTech: 2-dose series of updated (2023–2024 Formula)
Pzer-BioNTech at 0, 4 weeks (minimum interval between
previous Pzer-BioNTech dose and dose 1: 3 weeks).
y Previously vaccinated* with 2 doses of any Pzer-
BioNTech: 1 dose of updated (2023–2024 Formula) Pzer-
BioNTech at least 4 weeks after the most recent dose.
y Previously vaccinated* with 3 or more doses of any
Moderna or Pzer-BioNTech: 1 dose of any updated
(2023–2024 Formula) COVID-19 vaccine at least 8 weeks
after the most recent dose.
y Previously vaccinated* with 1 or more doses of Janssen
or Novavax or with or without dose(s) of any Original
monovalent or bivalent COVID-19 vaccine: 1 dose of any
updated (2023–2024 Formula) COVID-19 vaccine at least
8 weeks after the most recent dose.
There is no preferential recommendation for the use of
one COVID-19 vaccine over another when more than one
recommended age-appropriate vaccine is available.
Administer an age-appropriate COVID-19 vaccine product for
each dose. For information about transition from age 4 years
to age 5 years or age 11 years to age 12 years during COVID-19
vaccination series, see Tables 1 and 2 at www.cdc.gov/vaccines/
covid-19/clinical-considerations/interim-considerations-us.
html#covid-vaccines.
Current COVID-19 schedule and dosage formulation available
at www.cdc.gov/covidschedule. For more information on
Emergency Use Authorization (EUA) indications for COVID-19
vaccines, see www.fda.gov/emergency-preparedness-and-
response/coronavirus-disease-2019-covid-19/covid-19-vaccines
*Note: Previously vaccinated is dened as having received any
Original monovalent or bivalent COVID-19 vaccine (Janssen,
Moderna, Novavax, Pzer-BioNTech) prior to the updated
2023–2024 formulation.
**Note: Persons who are moderately or severely
immunocompromised have the option to receive one
additional dose of updated (2023–2024 Formula) COVID-19
vaccine at least 2 months following the last recommended
updated (2023 –2024 Formula) COVID-19 vaccine dose.
Further additional updated (2023–2024 Formula) COVID-19
vaccine dose(s) may be administered, informed by the clinical
judgement of a healthcare provider and personal preference
and circumstances. Any further additional doses should be
administered at least 2 months after the last updated
(2023–2024 Formula) COVID-19 vaccine dose. Moderately or
severely immunocompromised children 6 months–4 years of
age should receive homologous updated (2023–2024 Formula)
mRNA vaccine dose(s) if they receive additional doses.
Dengue vaccination
(minimum age: 9 years)
Routine vaccination
y Age 9–16 years living in areas with endemic dengue AND
have laboratory conrmation of previous dengue infection
- 3-dose series administered at 0, 6, and 12 months
y Endemic areas include Puerto Rico, American Samoa, US
Virgin Islands, Federated States of Micronesia, Republic of
Marshall Islands, and the Republic of Palau. For updated
guidance on dengue endemic areas and pre-vaccination
laboratory testing see www.cdc.gov/mmwr/volumes/70/rr/
rr7006a1.htm?s_cid=rr7006a1_w and www.cdc.gov/dengue/
vaccine/hcp/index.html
y Dengue vaccine should not be administered to children
traveling to or visiting endemic dengue areas.
Diphtheria, tetanus, and pertussis (DTaP)
vaccination (minimum age: 6 weeks [4 years
for Kinrix® or Quadracel®])
Routine vaccination
y 5-dose series (3-dose primary series at age 2, 4, and 6 months,
followed by a booster doses at ages 15–18 months and
4–6 years
- Prospectively: Dose 4 may be administered as early as age
12months if at least 6 months have elapsed since dose 3.
- Retrospectively: A 4
th
dose that was inadvertently
administered as early as age 12 months may be counted if at
least 4 months have elapsed since dose 3.
Catch-up vaccination
y Dose 5 is not necessary if dose 4 was administered at age
4years or older and at least 6 months after dose 3.
y For other catch-up guidance, see Table 2.
Special situations
y Wound management in children less than age 7 years with
history of 3 or more doses of tetanus-toxoid-containing
vaccine: For all wounds except clean and minor wounds,
administer DTaP if more than 5 years since last dose of
tetanus-toxoid-containing vaccine. For detailed information,
see www.cdc.gov/mmwr/volumes/67/rr/rr6702a1.htm.
Haemophilus inuenzae type b vaccination
(minimum age: 6 weeks)
Routine vaccination
y ActHIB®, Hiberix®, Pentacel®, or Vaxelis®: 4-dose series
(3-dose primary series at age 2, 4, and 6 months, followed by
a booster dose* at age 12–15 months)
- *Vaxelis® is not recommended for use as a booster dose.
A dierent Hib-containing vaccine should be used for the
booster dose.
y PedvaxHIB®: 3-dose series (2-dose primary series at age
2 and 4 months, followed by a booster dose at age 12–15
months)
Catch-up vaccination
y Dose 1 at age 7–11 months: Administer dose 2 at least
4 weeks later and dose 3 (nal dose) at age12–15 months or
8weeks after dose 2 (whichever is later).
y Dose 1 at age 12–14 months: Administer dose 2 (nal dose)
at least 8weeks after dose 1.
y Dose 1 before age 12 months and dose 2 before age
15 months: Administer dose 3 (nal dose) at least
8weeks after dose 2.
y 2 doses of PedvaxHIB® before age 12 months:
Administer dose 3 (nal dose) at age12–59 months and
at least 8weeks after dose 2.
y 1 dose administered at age 15 months or older:
No further doses needed
y Unvaccinated at age 15–59 months: Administer 1 dose.

Recommended Child and Adolescent Immunization Schedule for Ages 18 Years or Younger, United States, 2024
Notes
y Previously unvaccinated children age 60 months or
older who are not considered high risk: Do not
require catch-up vaccination
For other catch-up guidance, see Table 2. Vaxelis® can be used
for catch-up vaccination in children less than age 5 years.
Follow the catch-up schedule even if Vaxelis® is used for one
or more doses. For detailed information on use of Vaxelis® see
www.cdc.gov/mmwr/volumes/69/wr/mm6905a5.htm.
Special situations
y Chemotherapy or radiation treatment:
Age 12–59 months
- Unvaccinated or only 1 dose before age 12 months: 2doses,
8weeks apart
- 2 or more doses before age 12 months: 1 dose at least
8weeks after previous dose
Doses administered within 14 days of starting therapy or
during therapy should be repeated at least 3 months
after therapy completion.
y Hematopoietic stem cell transplant (HSCT):
- 3-dose series 4 weeks apart starting 6 to 12 months after
successful transplant, regardless of Hib vaccination history
y Anatomic or functional asplenia (including
sickle cell disease):
Age 12–59 months
- Unvaccinated or only 1 dose before age 12 months:
2 doses, 8weeks apart
- 2 or more doses before age 12 months:
1 dose at least 8weeks after previous dose
Unvaccinated* persons age 5 years or older
- 1 dose
y Elective splenectomy:
Unvaccinated* persons age 15 months or older
- 1 dose (preferably at least 14 days before procedure)
y HIV infection:
Age 12–59 months
- Unvaccinated or only 1 dose before age 12 months:
2doses, 8weeks apart
- 2 or more doses before age 12 months:
1 dose at least 8weeks after previous dose
Unvaccinated* persons age 5–18 years
- 1 dose
y Immunoglobulin deciency, early component
complement deciency:
Age 12–59 months
- Unvaccinated or only 1 dose before age 12 months:
2doses, 8weeks apart
- 2 or more doses before age 12 months:
1 dose at least 8weeks after previous dose
* Unvaccinated = Less than routine series (through age
14months) OR no doses (age 15 months or older)
Hepatitis A vaccination
(minimum age: 12 months for routine vaccination)
Routine vaccination
y 2-dose series (minimum interval: 6 months) at
age 12–23 months
Catch-up vaccination
y Unvaccinated persons through age 18 years should complete
a 2-dose series (minimum interval: 6 months).
y Persons who previously received 1 dose at age 12 months or
older should receive dose 2 at least 6 months after dose 1.
y Adolescents age 18 years or older may receive the combined
HepA and HepB vaccine, Twinrix®, as a 3-dose series (0, 1, and
6months) or 4-dose series (3 doses at 0, 7, and 21–30 days,
followed by a booster dose at 12 months).
International travel
y Persons traveling to or working in countries with high or
intermediate endemic hepatitis A (www.cdc.gov/travel/):
- Infants age 6–11 months: 1 dose before departure;
revaccinate with 2 doses (separated by at least 6 months)
between age 12–23 months.
- Unvaccinated age 12 months or older: Administer dose 1
as soon as travel is considered.
Hepatitis B vaccination
(minimum age: birth)
Routine vaccination
y 3-dose series at age 0, 1–2, 6–18 months (use monovalent
HepB vaccine for doses administered before age 6 weeks)
- Birth weight ≥2,000 grams: 1 dose within 24 hours of birth
if medically stable
- Birth weight <2,000 grams: 1 dose at chronological age
1 month or hospital discharge (whichever is earlier and
even if weight is still <2,000 grams).
y Infants who did not receive a birth dose should begin the
series as soon as possible (see Table 2 for minimum intervals).
y Administration of 4 doses is permitted when a combination
vaccine containing HepB is used after the birth dose.
y Minimum intervals (see Table 2): when 4 doses
are administered, substitute “dose 4” for “dose 3”
in these calculations
y Final (3rd or 4th) dose: age 6–18 months
(minimum age 24 weeks)
y Mother is HBsAg-positive
- Birth dose (monovalent HepB vaccine only): administer
HepB vaccine and hepatitis B immune globulin (HBIG)
(in separate limbs) within 12 hours of birth, regardless
of birth weight.
- Birth weight <2000 grams: administer 3 additional doses
of HepB vaccine beginning at age 1 month (total of 4 doses)
- Final (3rd or 4th) dose: administer at age 6 months
(minimum age 24 weeks)
- Test for HBsAg and anti-HBs at age 9–12 months. If HepB
series is delayed, test 1–2 months after nal dose. Do not
test before age 9 months.
y Mother is HBsAg-unknown
If other evidence suggestive of maternal hepatitis B infection
exists (e.g., presence of HBV DNA, HBeAg-positive, or mother
known to have chronic hepatitis B infection), manage infant
as if mother is HBsAg-positive
- Birth dose (monovalent HepB vaccine only):
Birth weight ≥2,000 grams: administer HepB vaccine
within 12 hours of birth. Determine mother’s HBsAg status
as soon as possible. If mother is determined to be HBsAg-
positive, administer HBIG as soon as possible (in separate
limb), but no later than 7 days of age.
Birth weight <2,000 grams: administer HepB vaccine
and HBIG (in separate limbs) within 12 hours of birth.
Administer 3 additional doses of HepB vaccine beginning
at age 1 month (total of 4 doses)
- Final (3rd or 4th) dose: administer at age 6 months
(minimum age 24 weeks)
- If mother is determined to be HBsAg-positive or if status
remains unknown, test for HBsAg and anti-HBs at
age 9–12 months. If HepB series is delayed, test 1–2 months
after nal dose. Do not test before age 9 months.
Catch-up vaccination
y Unvaccinated persons should complete a 3-dose series at
0, 1–2, 6 months. See Table 2 for minimum intervals
y Adolescents age 11–15 years may use an alternative
2-dose schedule with at least 4 months between doses
(adult formulation Recombivax HB® only).
y Adolescents age 18 years may receive:
- Heplisav-B®: 2-dose series at least 4 weeks apart
- PreHevbrio®: 3-dose series at 0, 1, and 6 months
- Combined HepA and HepB vaccine, Twinrix®: 3-dose series
(0, 1, and 6 months) or 4-dose series (3 doses at 0, 7, and
21–30 days, followed by a booster dose at 12 months).

Notes
Recommended Child and Adolescent Immunization Schedule for Ages 18 Years or Younger, United States, 2024
Special situations
y Revaccination is not generally recommended for persons
with a normal immune status who were vaccinated as infants,
children, adolescents, or adults.
y Post-vaccination serology testing and revaccination
(if anti-HBs <10mlU/mL) is recommended for certain
populations, including:
- Infants born to HBsAg-positive mothers
- Persons who are predialysis or on maintenance dialysis
- Other immunocompromised persons
- For detailed revaccination recommendations, see www.cdc.
gov/vaccines/hcp/acip-recs/vacc-specic/hepb.html.
Note: Heplisav-B and PreHevbrio are not recommended in
pregnancy due to lack of safety data in pregnant persons
Human papillomavirus vaccination
(minimum age: 9 years)
Routine and catch-up vaccination
y HPV vaccination routinely recommended at age 11–12 years
(can start at age 9 years) and catch-up HPV vaccination
recommended for all persons through age 18 years if not
adequately vaccinated
y 2- or 3-dose series depending on age at initial vaccination:
- Age 9–14 years at initial vaccination: 2-dose series at 0,
6–12 months (minimum interval: 5 months; repeat dose if
administered too soon)
- Age 15 years or older at initial vaccination: 3-dose series
at 0, 1–2 months, 6 months (minimum intervals: dose 1 to
dose 2: 4 weeks / dose 2 to dose 3: 12 weeks / dose 1 to dose
3: 5months; repeat dose if administered too soon)
y No additional dose recommended when any HPV
vaccine series of any valency has been completed using
recommended dosing intervals.
Special situations
y Immunocompromising conditions, including HIV
infection: 3-dose series, even for those who initiate
vaccination at age 9 through 14 years.
y History of sexual abuse or assault: Start at age 9 years
y Pregnancy: Pregnancy testing not needed before
vaccination; HPV vaccination not recommended
until after pregnancy; no intervention needed if
vaccinated while pregnant
Inuenza vaccination
(minimum age: 6 months [IIV], 2 years [LAIV4],
18years [recombinant inuenza vaccine, RIV4])
Routine vaccination
y Use any inuenza vaccine appropriate for age and health
status annually:
- Age 6 months–8 years who have received fewer than
2 inuenza vaccine doses before July 1, 2023, or whose
inuenza vaccination history is unknown: 2 doses, separated
by at least 4 weeks. Administer dose 2 even if the child turns
9 years between receipt of dose 1 and dose 2.
- Age 6 months–8 years who have received at least 2
inuenza vaccine doses before July 1, 2023: 1 dose
- Age 9 years or older: 1 dose
y For the 2023-2024 season, see www.cdc.gov/mmwr/
volumes/72/rr/rr7202a1.htm.
y For the 2024–25 season, see the 2024–25 ACIP inuenza
vaccine recommendations.
Special situations
y Close contacts (e.g., household contacts) of severely
immunosuppressed persons who require a protected
environment: should not receive LAIV4. If LAIV4 is given,
they should avoid contact with for such immunosuppressed
persons for 7 days after vaccination.
Note: Persons with an egg allergy can receive any inuenza
vaccine (egg-based and non-egg-based) appropriate for age
and health status.
Measles, mumps, and rubella vaccination
(minimum age: 12 months for routine vaccination)
Routine vaccination
y 2-dose series at age 12–15 months, age 4–6 years
y MMR or MMRV* may be administered
Note: For dose 1 in children age 12–47 months, it is
recommended to administer MMR and varicella vaccines
separately. MMRV* may be used if parents or caregivers
express a preference.
Catch-up vaccination
y Unvaccinated children and adolescents: 2-dose series
at least 4 weeks apart*
y The maximum age for use of MMRV* is 12 years.
Special situations
y International travel
- Infants age 6–11 months: 1 dose before departure;
revaccinate with 2-dose series at age 12–15 months
(12 months for children in high-risk areas) and dose 2
as early as 4 weeks later.*
- Unvaccinated children age 12 months or older:
2-dose series at least 4 weeks apart before departure*
y In mumps outbreak settings, for information about
additional doses of MMR (including 3rd dose of MMR), see
www.cdc.gov/mmwr/volumes/67/wr/mm6701a7.htm
*Note: If MMRV is used, the minimum interval between MMRV
doses is 3 months
Meningococcal serogroup A,C,W,Y vaccination
(minimum age: 2 months [MenACWY-CRM, Menveo],
2 years [MenACWY-TT, MenQuad]), 10 years
[MenACWY-TT/MenB-FHbp, Penbraya])
Routine vaccination
y 2-dose series at age 11–12 years; 16 years
Catch-up vaccination
y Age 13–15 years: 1 dose now and booster at age 16–18 years
(minimum interval: 8 weeks)
y Age 16–18 years: 1 dose
Special situations
Anatomic or functional asplenia (including sickle cell
disease), HIV infection, persistent complement
component deciency, complement inhibitor
(e.g., eculizumab, ravulizumab) use:
y Menveo®*
- Dose 1 at age 2 months: 4-dose series (additional 3 doses
at age 4, 6, and 12 months)
- Dose 1 at age 3–6 months: 3- or 4-dose series (dose 2
[and dose 3 if applicable] at least 8 weeks after previous
dose until a dose is received at age 7 months or older,
followed by an additional dose at least 12 weeks later
and after age 12 months)
- Dose 1 at age 7–23 months: 2-dose series (dose 2 at least
12weeks after dose 1 and after age 12 months)
- Dose 1 at age 24 months or older: 2-dose series
at least 8weeks apart
y MenQuad®
- Dose 1 at age 24 months or older: 2-dose series at least
8 weeks apart

Notes
Recommended Child and Adolescent Immunization Schedule for Ages 18 Years or Younger, United States, 2024
Travel to countries with hyperendemic or epidemic
meningococcal disease, including countries in the African
meningitis belt or during the Hajj (www.cdc.gov/travel/):
y Children less than age 24 months:
- Menveo®* (age 2–23 months)
Dose 1 at age 2 months: 4-dose series (additional 3 doses at
age 4, 6, and 12 months)
Dose 1 at age 3–6 months: 3- or 4-dose series (dose 2
[and dose 3 if applicable] at least 8 weeks after previous
dose until a dose is received at age 7 months or older,
followed by an additional dose at least 12 weeks later
and after age 12 months)
Dose 1 at age 7–23 months: 2-dose series (dose 2 at least
12 weeks after dose 1 and after age 12 months)
y Children age 2 years or older: 1 dose Menveo®*
or MenQuad®
First-year college students who live in residential housing
(if not previously vaccinated at age 16 years or older) or
military recruits:
y 1 dose Menveo®* or MenQuad®
Adolescent vaccination of children who received MenACWY
prior to age 10 years:
y Children for whom boosters are recommended because
of an ongoing increased risk of meningococcal disease
(e.g., those with complement component deciency, HIV,
or asplenia): Follow the booster schedule for persons at
increased risk.
y Children for whom boosters are not recommended
(e.g., a healthy child who received a single dose for travel
to a country where meningococcal disease is endemic):
Administer MenACWY according to the recommended
adolescent schedule with dose 1 at age 11–12 years and
dose 2 at age 16 years.
* Menveo has two formulations: lyophilized and liquid. The liquid
formulation should not be used before age 10 years. See www.
cdc.gov/vaccines/vpd/mening/downloads/menveo-single-vial-
presentation.pdf.
Note: For MenACWY booster dose recommendations for
groups listed under “Special situations” and in an outbreak
setting and additional meningococcal vaccination information,
see www.cdc.gov/mmwr/volumes/69/rr/rr6909a1.htm.
Children age 10 years or older may receive a single dose of
Penbraya™ as an alternative to separate administration of
MenACWY and MenB when both vaccines would be given
on the same clinic day (see “Meningococcal serogroup B
vaccination” section below for more information).
Meningococcal serogroup B vaccination
(minimum age: 10 years [MenB-4C, Bexsero®;
MenB-FHbp, Trumenba®; MenACWY-TT/MenB-FHbp,
Penbraya™])
Shared clinical decision-making
y Adolescents not at increased risk age 16–23 years
(preferred age 16–18 years) based on shared
clinical decision-making:
- Bexsero®: 2-dose series at least 1 month apart
- Trumenba®: 2-dose series at least 6 months apart (if dose 2
is administered earlier than 6 months, administer a 3
rd
dose
at least 4 months after dose 2)
For additional information on shared clinical decision-making
for MenB, see www.cdc.gov/vaccines/hcp/admin/downloads/
isd-job-aid-scdm-mening-b-shared-clinical-decision-making.pdf
Special situations
Anatomic or functional asplenia (including sickle cell
disease), persistent complement component deciency,
complement inhibitor (e.g., eculizumab, ravulizumab) use:
y Bexsero®: 2-dose series at least 1 month apart
y Trumenba®: 3-dose series at 0, 1–2, 6 months (if dose 2
was administered at least 6 months after dose 1, dose 3
not needed; if dose 3 is administered earlier than 4 months
after dose 2, a 4
th
dose should be administered at least
4 months after dose 3)
Note: Bexsero® and Trumenba® are not interchangeable;
the same product should be used for all doses in a series.
For MenB booster dose recommendations for groups listed
under “Special situations” and in an outbreak setting and
additional meningococcal vaccination information, see
www.cdc.gov/mmwr/volumes/69/rr/rr6909a1.htm.
Children age 10 years or older may receive a dose of Penbraya™
as an alternative to separate administration of MenACWY and
MenB when both vaccines would be given on the same clinic
day. For age-eligible children not at increased risk, if Penbraya™
is used for dose 1 MenB, MenB-FHbp (Trumenba) should be
administered for dose 2 MenB. For age-eligible children at
increased risk of meningococcal disease, Penbraya™ may be
used for additional MenACWY and MenB doses (including
booster doses) if both would be given on the same clinic
day and at least 6 months have elapsed since most recent
Penbraya™ dose.
Mpox vaccination
(minimum age: 18 years [Jynneos®])
Special situations
y Age 18 years and at risk for Mpox infection: 2-dose series,
28 days apart.
Risk factors for Mpox infection include:
- Persons who are gay, bisexual, and other MSM, transgender
or nonbinary people who in the past 6 months have had:
A new diagnosis of at least 1 sexually transmitted disease
More than 1 sex partner
Sex at a commercial sex venue
Sex in association with a large public event in a geographic
area where Mpox transmission is occurring
- Persons who are sexual partners of the persons described
above
- Persons who anticipate experiencing any of the situations
described above
y Pregnancy: There is currently no ACIP recommendation
for Jynneos use in pregnancy due to lack of safety data in
pregnant persons. Pregnant persons with any risk factor
described above may receive Jynneos.
For detailed information, see: www.cdc.gov/vaccines/acip/
meetings/downloads/slides-2023-10-25-26/04-MPOX-Rao-508.pdf
Pneumococcal vaccination
(minimum age: 6 weeks [PCV15], [PCV 20]; 2 years
[PPSV23])
Routine vaccination with PCV
y 4-dose series at 2, 4, 6, 12–15 months
Catch-up vaccination with PCV
y Healthy children ages 2–4 years with any incomplete*
PCV series: 1 dose PCV
y For other catch-up guidance, see Table 2.
Note: For children without risk conditions, PCV20 is not
indicated if they have received 4 doses of PCV13 or PCV15 or
another age appropriate complete PCV series.

Notes
Recommended Child and Adolescent Immunization Schedule for Ages 18 Years or Younger, United States, 2024
Special situations
Children and adolescents with cerebrospinal uid leak;
chronic heart disease; chronic kidney disease (excluding
maintenance dialysis and nephrotic syndrome); chronic
liver disease; chronic lung disease (including moderate
persistent or severe persistent asthma); cochlear implant;
or diabetes mellitus:
Age 2–5 years
y Any incomplete* PCV series with:
- 3 PCV doses: 1 dose PCV (at least 8 weeks after the most
recent PCV dose)
- Less than 3 PCV doses: 2 doses PCV (at least 8 weeks after
the most recent dose and administered at least 8 weeks
apart)
y Completed recommended PCV series but have not received
PPSV23
- Previously received at least 1 dose of PCV20: no further PCV
or PPSV23 doses needed
- Not previously received PCV20: administer 1 dose PCV20 OR
1 dose PPSV23 administer at least 8 weeks after the most
recent PCV dose.
Age 6–18 years
y Not previously received any dose of PCV13, PCV15, or PCV20:
administer 1 dose of PCV15 or PCV20. If PCV15 is used and no
previous receipt of PPSV23, administer 1 dose of PPSV23 at
least 8 weeks after the PCV15 dose.**
y Received PCV before age 6 years but have not received
PPSV23
- Previously received at least 1 dose of PCV20: no further PCV
or PPSV23 doses needed
- Not previously received PCV20: 1 dose PCV20 OR 1 dose
PPSV23 administer at least 8 weeks after the most recent
PCV dose.
y Received PCV13 only at or after age 6 years: administer 1 dose
PCV20 OR 1 dose PPSV23 at least 8 weeks after the most
recent PCV13 dose.
y Received 1 dose PCV13 and 1 dose PPSV23 at or after age
6 years: no further doses of any PCV or PPSV23 indicated.
Children and adolescents on maintenance dialysis, or
with immunocompromising conditions such as nephrotic
syndrome; congenital or acquired asplenia or splenic
dysfunction; congenital or acquired immunodeciencies;
diseases and conditions treated with immunosuppressive
drugs or radiation therapy, including malignant
neoplasms, leukemias, lymphomas, Hodgkin disease, and
solid organ transplant; HIV infection; or sickle cell disease
or other hemoglobinopathies:
Age 2–5 years
y Any incomplete* PCV series:
- 3 PCV doses: 1 dose PCV (at least 8 weeks after the most
recent PCV dose)
- Less than 3 PCV doses: 2 doses PCV (at least 8 weeks after the
most recent dose and administered at least 8 weeks apart)
y Completed recommended PCV series but have not received
PPSV23
- Previously received at least 1 dose of PCV20: no further PCV
or PPSV23 doses needed
- Not previously received PCV20: administer 1 dose PCV20 OR
1 dose PPSV23 at least 8 weeks after the most recent PCV
dose. If PPSV23 is used, administer 1 dose of PCV20 or dose
2 PPSV23 at least 5 years after dose 1 PPSV23.
Age 6–18 years
y Not previously received any dose of PCV13, PCV15, or PCV20:
administer 1 dose of PCV15 or 1 dose of PCV20. If PCV15 is
used and no previous receipt of PPSV23, administer 1 dose of
PPSV23 at least 8 weeks after the PCV15 dose.**
y Received PCV before age 6 years but have not received
PPSV23
- Previously received at least 1 dose of PCV20: no additional
dose of PCV or PPSV23
- Not previously received PCV20: administer 1 dose PCV20 OR
1 dose PPSV23 at least 8 weeks after the most recent PCV
dose. If PPSV23 is used, administer either PCV20 or dose 2
PPSV23 at least 5 years after dose 1 PPSV23.
y Received PCV13 only at or after age 6 years: administer 1 dose
PCV20 OR 1 dose PPSV23 at least 8 weeks after the most
recent PCV13 dose. If PPSV23 is used, administer 1 dose of
PCV20 or dose 2 PPSV23 at least 5 years after dose 1 PPSV23.
y Received 1 dose PCV13 and 1 dose PPSV23 at or after age
6 years: administer 1 dose PCV20 OR 1 dose PPSV23 at least
8 weeks after the most recent PCV13 dose and at least 5 years
after dose 1 PPSV23.
*Incomplete series = Not having received all doses in either the
recommended series or an age-appropriate catch-up series.
See Table 2 in ACIP pneumococcal recommendations at
stacks.cdc.gov/view/cdc/133252
**When both PCV15 and PPSV23 are indicated, administer
all doses of PCV15 rst. PCV15 and PPSV23 should not be
administered during the same visit.
For guidance on determining which pneumococcal vaccines
a patient needs and when, please refer to the mobile app,
which can be downloaded here: www.cdc.gov/vaccines/vpd/
pneumo/hcp/pneumoapp.html
Poliovirus vaccination
(minimum age: 6 weeks)
Routine vaccination
y 4-dose series at ages 2, 4, 6–18 months, 4–6 years; administer
the nal dose on or after age 4 years and at least 6 months
after the previous dose.
y 4 or more doses of IPV can be administered before age 4 years
when a combination vaccine containing IPV is used. However,
a dose is still recommended on or after age 4 years and at
least 6 months after the previous dose.
Catch-up vaccination
y In the rst 6 months of life, use minimum ages and
intervals only for travel to a polio-endemic region
or during an outbreak.
y Adolescents age 18 years known or suspected to be
unvaccinated or incompletely vaccinated: administer
remaining doses (1, 2, or 3 IPV doses) to complete a 3-dose
primary series.* Unless there are specic reasons to believe
they were not vaccinated, most persons aged 18 years or
older born and raised in the United States can assume they
were vaccinated against polio as children.
Series containing oral poliovirus vaccine (OPV), either mixed
OPV-IPV or OPV-only series:
y Total number of doses needed to complete the series is the
same as that recommended for the U.S. IPV schedule. See
www.cdc.gov/mmwr/volumes/66/wr/mm6601a6.htm?s_%20
cid=mm6601a6_w.
y Only trivalent OPV (tOPV) counts toward the
U.S. vaccination requirements.
- Doses of OPV administered before April 1, 2016,
should be counted (unless specically noted as
administered during a campaign).
- Doses of OPV administered on or after April 1, 2016,
should not be counted.
- For guidance to assess doses documented as “OPV,” see
www.cdc.gov/mmwr/volumes/66/wr/mm6606a7.htm?s_
cid=mm6606a7_w.
y For other catch-up guidance, see Table 2.

Notes
Recommended Child and Adolescent Immunization Schedule for Ages 18 Years or Younger, United States, 2024
Special situations
y Adolescents aged 18 years at increased risk of exposure to
poliovirus and completed primary series*: may administer
one lifetime IPV booster
*Note: Complete primary series consist of at least 3 doses
of IPV or trivalent oral poliovirus vaccine (tOPV) in any
combination.
For detailed information, see: www.cdc.gov/vaccines/vpd/
polio/hcp/recommendations.html
Respiratory syncytial virus immunization
(minimum age: birth [Nirsevimab, RSV-mAb
(Beyfortus™)
Routine immunization
y Infants born October – March in most of the continental
United States*
- Mother did not receive RSV vaccine OR mother’s RSV
vaccination status is unknown: administer 1 dose nirsevimab
within 1 week of birth in hospital or outpatient setting
- Mother received RSV vaccine less than 14 days prior to
delivery: administer 1 dose nirsevimab within 1 week of
birth in hospital or outpatient setting
- Mother received RSV vaccine at least 14 days prior to
delivery: nirsevimab not needed but can be considered in
rare circumstances at the discretionof healthcare providers
(see special populations and situations at
www.cdc.gov/vaccines/vpd/rsv/hcp/child-faqs.html)
y Infants born April–September in most of the continental
United States*
- Mother did not receive RSV vaccine OR mother’s RSV
vaccination status is unknown: administer 1 dose nirsevimab
shortly before start of RSV season*
- Mother received RSV vaccine less than 14 days prior to
delivery: administer 1 dose nirsevimab shortly before start of
RSV season*
- Mother received RSV vaccine at least 14 days prior to
delivery: nirsevimab not needed but can be considered
in rare circumstances at the discretionof healthcare
providers(see special populations and situations at
www.cdc.gov/vaccines/vpd/rsv/hcp/child-faqs.html)
Infants with prolonged birth hospitalization** (e.g., for
prematurity) discharged October through March should be
immunized shortly before or promptly afterdischarge.
Special situations
y Ages 8–19 months with chronic lung disease of
prematurity requiring medical support (e.g.,
chronic corticosteroid therapy, diuretic therapy, or
supplemental oxygen) any time during the 6-month
period before the start of the second RSV season; severe
immunocompromise; cystic brosis with either weight
for length <10th percentile or manifestation of severe
lung disease (e.g., previous hospitalization for pulmonary
exacerbation in the rst year of life or abnormalities on
chest imaging that persist when stable)**:
- 1 dose nirsevimab shortly before start of second RSV
season*
y Ages 8–19 months who are American Indian or Alaska
Native:
- 1 dose nirsevimab shortly before start of second RSV
season*
y Age-eligible and undergoing cardiac surgery with
cardiopulmonary bypass**: 1 additional dose of nirsevimab
after surgery. For additional details see special populations
and situations at www.cdc.gov/vaccines/vpd/rsv/hcp/child-
faqs.html
*Note: While the timing of the onset and duration of RSV
season may vary, nirsevimab may be administered October
through March in most of the continental United States.
Providers in jurisdictions with RSV seasonality that diers from
most of the continental United States (e.g., Alaska, jurisdiction
with tropical climate) should follow guidance from public
health authorities (e.g., CDC, health departments) or regional
medical centers on timing of administration based on local
RSV seasonality. Although optimal timing of administration is
just before the start of the RSV season, nirsevimab may also
be administered during the RSV season to infants and children
who are age-eligible.
**Note: Nirsevimab can be administered to children who are
eligible to receive palivizumab. Children who have received
nirsevimab should not receive palivizumab for the same RSV
season.
For further guidance, see www.cdc.gov/mmwr/volumes/72/
wr/mm7234a4.htm and www.cdc.gov/vaccines/vpd/rsv/hcp/
child-faqs.html
Respiratory syncytial virus vaccination
(RSV [Abrysvo™])
Routine vaccination
y Pregnant at 32 weeks 0 days through 36 weeks and 6 days
gestation from September through January in most of the
continental United States*: 1 dose RSV vaccine (Abrysvo™).
Administer RSV vaccine regardless of previous RSV infection.
- Either maternal RSV vaccination or infant immunization with
nirsevimab (RSV monoclonal antibody) is recommended to
prevent respiratory syncytial virus lower respiratory tract
infection in infants.
y All other pregnant persons: RSV vaccine not recommended.
There is currently no ACIP recommendation for RSV vaccination
in subsequent pregnancies. No data are available to inform
whether additional doses are needed in later pregnancies.
*Note: Providers in jurisdictions with RSV seasonality that
diers from most of the continental United States (e.g., Alaska,
jurisdiction with tropical climate) should follow guidance from
public health authorities (e.g., CDC, health departments) or
regional medical centers on timing of administration based on
local RSV seasonality.
Rotavirus vaccination
(minimum age: 6 weeks)
Routine vaccination
y Rotarix®: 2-dose series at age 2 and 4 months
y RotaTeq®: 3-dose series at age 2, 4, and 6 months
y If any dose in the series is either RotaTeq® or unknown,
default to 3-dose series.
Catch-up vaccination
y Do not start the series on or after age 15 weeks, 0 days.
y The maximum age for the nal dose is 8 months, 0 days.
y For other catch-up guidance, see Table 2.

Notes
Recommended Child and Adolescent Immunization Schedule for Ages 18 Years or Younger, United States, 2024
11/16/2023 Centers for Disease Control and Prevention
|
Recommended Child and Adolescent Immunization Schedule, United States, 2024
Tetanus, diphtheria, and pertussis (Tdap)
vaccination
(minimum age: 11 years for routine vaccination,
7years for catch-up vaccination)
Routine vaccination
y Age 11–12 years: 1 dose Tdap (adolescent booster)
y Pregnancy: 1 dose Tdap during each pregnancy, preferably in
early part of gestational weeks 27–36.
Note: Tdap may be administered regardless of the interval
since the last tetanus- and diphtheria-toxoid-containing
vaccine.
Catch-up vaccination
y Age 13–18 years who have not received Tdap:
1 dose Tdap (adolescent booster)
y Age 7–18 years not fully vaccinated
*
with DTaP: 1dose
Tdap as part of the catch-up series (preferably the rst dose);
if additional doses are needed, use Td or Tdap.
y Tdap administered at age 7–10 years:
- Age 7–9 years who receive Tdap should receive the
adolescent Tdap booster dose at age 11–12 years.
- Age 10 years who receive Tdap do not need the adolescent
Tdap booster dose at age 11–12 years.
y DTaP inadvertently administered on or after age 7 years:
- Age 7–9 years: DTaP may count as part of catch-up series.
Administer adolescent Tdap booster dose at age 11–12 years.
- Age 10–18 years: Count dose of DTaP as the adolescent
Tdap booster dose.
y For other catch-up guidance, see Table 2.
Special situations
y Wound management in persons age 7 years or older with
history of 3 or more doses of tetanus-toxoid-containing
vaccine: For clean and minor wounds, administer Tdap or
Td if more than 10 years since last dose of tetanus-toxoid-
containing vaccine; for all other wounds, administer Tdap
or Td if more than 5 years since last dose of tetanus-toxoid-
containing vaccine. Tdap is preferred for persons age 11 years
or older who have not previously received Tdap or whose
Tdap history is unknown. If a tetanus-toxoid-containing
vaccine is indicated for a pregnant adolescent, use Tdap.
y For detailed information, see www.cdc.gov/mmwr/
volumes/69/wr/mm6903a5.htm.
*Fully vaccinated = 5 valid doses of DTaP OR 4 valid doses of
DTaP if dose 4 was administered at age 4 years or older
Varicella vaccination
(minimum age: 12 months)
Routine vaccination
y 2-dose series at age 12–15 months, 4–6 years
y VAR or MMRV may be administered*
y Dose 2 may be administered as early as 3 months after dose 1
(a dose inadvertently administered after at least 4 weeks
may be counted as valid)
*Note: For dose 1 in children age 12–47 months, it is
recommended to administer MMR and varicella vaccines
separately. MMRV may be used if parents or caregivers
express a preference.
Catch-up vaccination
y Ensure persons age 7–18 years without evidence of immunity
(see MMWR at www.cdc.gov/mmwr/pdf/rr/rr5604.pdf)
have a 2-dose series:
- Age 7–12 years: Routine interval: 3 months
(a dose inadvertently administered after at least
4 weeks may be counted as valid)
- Age 13 years and older: Routine interval: 4–8weeks
(minimum interval: 4 weeks)
- The maximum age for use of MMRV is 12 years.

Vaccines and other
Immunizing Agents
Contraindicated or Not Recommended
1
Precautions
2
COVID-19 mRNA vaccines
[Pzer-BioNTech, Moderna]
• Severeallergic reaction (e.g., anaphylaxis) after a previous dose or to a component of
an mRNA COVID-19 vaccine
4
• Diagnosed non-severe allergy (e.g., urticaria beyond the injection site) to a
component of an mRNA COVID-19 vaccine
4
; or non-severe, immediate (onset
less than 4 hours) allergic reaction after administration of a previous dose of
an mRNA COVID-19 vaccine
• Myocarditis or pericarditis within 3 weeks after a dose ofany COVID-19 vaccine
• Multisystem inammatory syndrome in children (MIS-C) or multisystem
inammatory syndrome in adults (MIS-A)
• Moderate or severe acute illness, with or without fever
COVID-19 protein subunit
vaccine
[Novavax]
• Severe allergic reaction (e.g., anaphylaxis) after a previous dose or to a component of
a Novavax COVID-19 vaccine
4
• Diagnosed non-severe allergy (e.g., urticaria beyond the injection site) to a
component of Novavax COVID-19 vaccine
4
; or non-severe, immediate (onset
less than 4 hours) allergic reaction after administration of a previous dose of
a Novavax COVID-19 vaccine
• Myocarditis or pericarditis within 3 weeks after a dose ofany COVID-19 vaccine
• Multisystem inammatory syndrome in children (MIS-C) or multisystem
inammatory syndrome in adults (MIS-A)
• Moderate or severe acute illness, with or without fever
Inuenza, egg-based,
inactivated injectable (IIV4)
• Severe allergic reaction (e.g., anaphylaxis) after previous dose of any inuenza vaccine
(i.e., any egg-based IIV, ccIIV, RIV, or LAIV of any valency)
• Severe allergic reaction (e.g., anaphylaxis) to any vaccine component
3
(excluding egg)
• Guillain-Barré syndrome (GBS) within 6 weeks after a previous dose of any
type of inuenza vaccine
• Moderate or severe acute illness with or without fever
Inuenza, cell culture-based
inactivated injectable (ccIIV4)
[Flucelvax Quadrivalent]
• Severe allergic reaction (e.g., anaphylaxis) to any ccIIV of any valency, or to any component
3
of ccIIV4 • Guillain-Barré syndrome (GBS) within 6 weeks after a previous dose of any
type of inuenza vaccine
• Persons with a history of severe allergic reaction (e.g., anaphylaxis) after a previous
dose of any egg-based IIV, RIV, or LAIV of any valency. If using ccIV4, administer in
medical setting under supervision of health care provider who can recognize and
manage severe allergic reactions. May consult an allergist.
• Moderate or severe acute illness with or without fever
Inuenza, recombinant
injectable (RIV4)
[Flublok Quadrivalent]
• Severe allergic reaction (e.g., anaphylaxis) to any RIV of any valency, or to any component
3
of RIV4 • Guillain-Barré syndrome (GBS) within 6 weeks after a previous dose of any
type of inuenza vaccine
• Persons with a history of severe allergic reaction (e.g., anaphylaxis) after a previous
dose of any egg-based IIV, ccIIV, or LAIV of any valency. If using RIV4, administer in
medical setting under supervision of health care provider who can recognize and
manage severe allergic reactions. May consult an allergist.
• Moderate or severe acute illness with or without fever
Inuenza, live attenuated
(LAIV4)
[Flumist Quadrivalent]
• Severe allergic reaction (e.g., anaphylaxis) after previous dose of any inuenza vaccine
(i.e., any egg-based IIV, ccIIV, RIV, or LAIV of any valency)
• Severe allergic reaction (e.g., anaphylaxis) to any vaccine component
3
(excluding egg)
• Children age 2–4 years with a history of asthma or wheezing
• Anatomic or functional asplenia
• Immunocompromised due to any cause including, but not limited to, medications and HIV infection
• Close contacts or caregivers of severely immunosuppressed persons who require a protected environment
• Pregnancy
• Cochlear implant
• Active communication between the cerebrospinal uid (CSF) and the oropharynx, nasopharynx, nose,
ear or any other cranial CSF leak
• Children and adolescents receiving aspirin or salicylate-containing medications
• Received inuenza antiviral medications oseltamivir or zanamivir within the previous 48 hours,
peramivir within the previous 5 days, or baloxavir within the previous 17 days
• Guillain-Barré syndrome (GBS) within 6 weeks after a previous dose of any
type of inuenza vaccine
• Asthma in persons age 5 years old or older
• Persons with underlying medical conditions other than those listed under
contraindications that might predispose to complications after wild-type
inuenza virus infection, e.g., chronic pulmonary, cardiovascular (except isolated
hypertension), renal, hepatic, neurologic, hematologic, or metabolic disorders
(including diabetes mellitus)
• Moderate or severe acute illness with or without fever
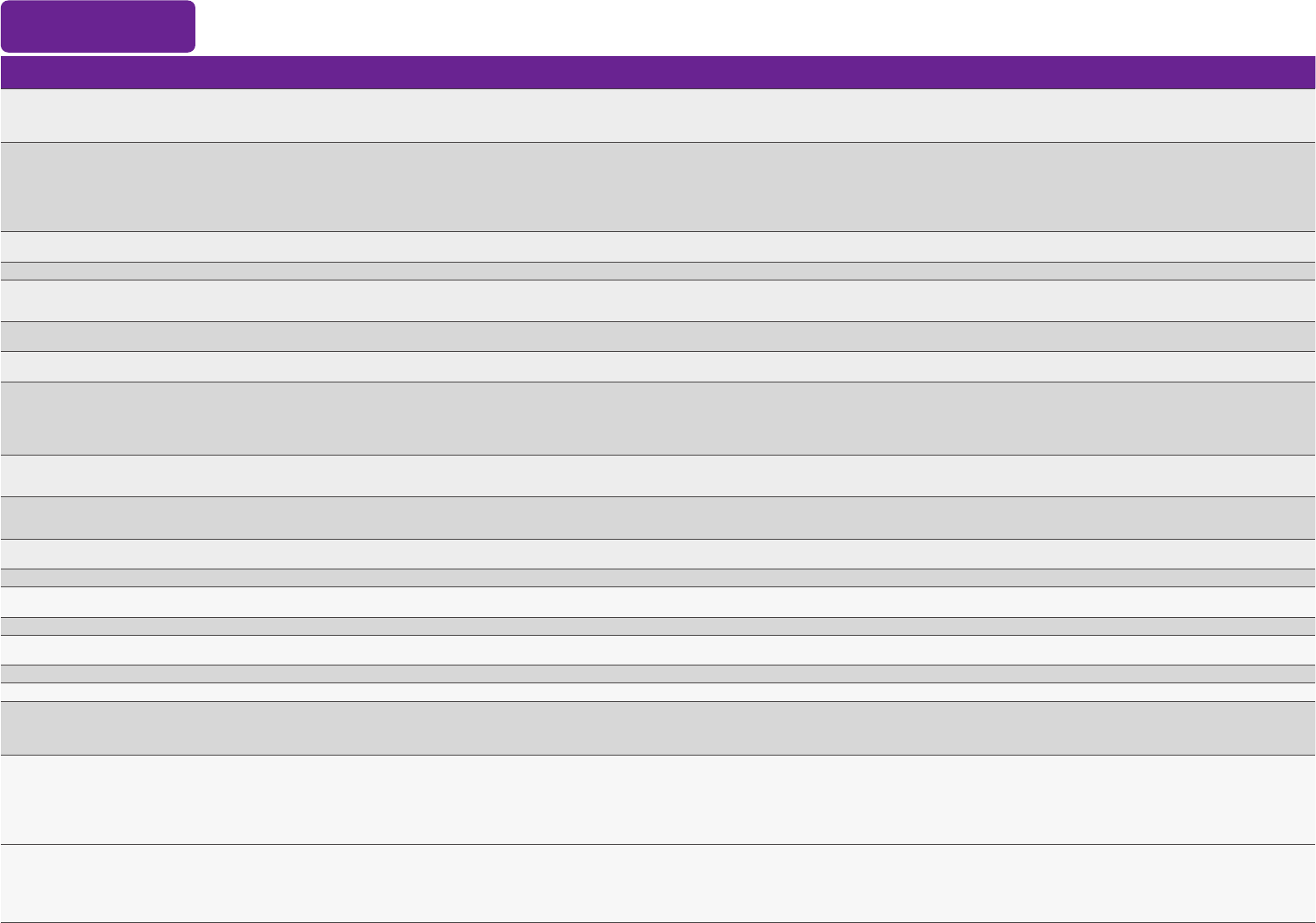
1. When a contraindication is present, a vaccine shouldNOTbe administered. Kroger A, Bahta L, Hunter P.ACIP General Best Practice Guidelines for Immunization.
2. When a precaution is present, vaccination should generally be deferred but might be indicated if the benet of protection from the vaccine outweighs the risk for an adverse reaction. Kroger A, Bahta L, Hunter P.ACIP General Best
Practice Guidelines for Immunization.
3. Vaccination providers should check FDA-approved prescribing information for the most complete and updated information, including contraindications, warnings, and precautions. SeePackage inserts for U.S.-licensed vaccines.
4. Seepackage insertsandFDA EUA fact sheetsfor a full list of vaccine ingredients. mRNA COVID-19 vaccines contain polyethylene glycol (PEG).
Guide to Contraindications and Precautions to Commonly Used Vaccines
Adapted from Table 4-1 inAdvisory Committee on Immunization Practices (ACIP) General Best Practice Guidelines for Immunization:Contraindication and Precautions, Prevention and Control of Seasonal Inuenza with Vaccines:
Recommendations of the Advisory Committee on Immunization Practices—United States, 2023–24 Inuenza Season | MMWR (cdc.gov), Contraindications and Precautions for COVID-19 Vaccination, and Contraindications and
Precautions for JYNNEOS Vaccination
Recommended Child and Adolescent Immunization Schedule for Ages 18 Years or Younger, United States, 2024
Appendix
Recommended Child and Adolescent Immunization Schedule for Ages 18 Years or Younger, United States, 2024
Appendix
Vaccines and other
Immunizing Agents
Contraindicated or Not Recommended
1
Precautions
2
Dengue (DEN4CYD) • Severe allergic reaction (e.g., anaphylaxis) after a previous dose or to a vaccine component
3
• Severe immunodeciency (e.g., hematologic and solid tumors, receipt of chemotherapy, congenital immunodeciency,
long-term immunosuppressive therapy or patients with HIV infection who are severely immunocompromised)
• Lack of laboratory conrmation of a previous Dengue infection
• Pregnancy
• HIV infection without evidence of severe immunosuppression
• Moderate or severe acute illness with or without fever
Diphtheria, tetanus, pertussis (DTaP) • Severe allergic reaction (e.g., anaphylaxis) after a previous dose or to a vaccine component
3
• For DTaP only: Encephalopathy (e.g., coma, decreased level of consciousness, prolonged seizures) not attributable to
another identiable cause within 7 days of administration of previous dose of DTP or DTaP
• Guillain-Barré syndrome (GBS) within 6 weeks after previous dose of tetanus-toxoid–containing vaccine
• History of Arthus-type hypersensitivity reactions after a previous dose of diphtheria-toxoid–containing
or tetanus-toxoid–containing vaccine; defer vaccination until at least 10 years have elapsed since the last
tetanus-toxoid-containing vaccine
• For DTaP only: Progressive neurologic disorder, including infantile spasms, uncontrolled epilepsy,
progressive encephalopathy; defer DTaP until neurologic status claried and stabilized
• Moderate or severe acute illness with or without fever
Haemophilus inuenzae type b (Hib) • Severe allergic reaction (e.g., anaphylaxis) after a previous dose or to a vaccine component
3
• Less than age 6 weeks
• Moderate or severe acute illness with or without fever
Hepatitis A (HepA) • Severe allergic reaction (e.g., anaphylaxis) after a previous dose or to a vaccine component
3
including neomycin • Moderate or severe acute illness with or without fever
Hepatitis B (HepB) • Severe allergic reaction (e.g., anaphylaxis) after a previous dose or to a vaccine component
3
including yeast
• Pregnancy: Heplisav-B and PreHevbrio are not recommended due to lack of safety data in pregnant persons. Use other hepatitis
B vaccines if HepB is indicated
4
.
• Moderate or severe acute illness with or without fever
Hepatitis A-Hepatitis B vaccine (HepA-HepB)
[Twinrix]
• Severe allergic reaction (e.g., anaphylaxis) after a previous dose or to a vaccine component
3
including neomycin and
yeast
• Moderate or severe acute illness with or without fever
Human papillomavirus (HPV) • Severe allergic reaction (e.g., anaphylaxis) after a previous dose or to a vaccine component
3
• Pregnancy: HPV vaccination not recommended.
• Moderate or severe acute illness with or without fever
Measles, mumps, rubella (MMR)
Measles, mumps, rubella, and varicella
(MMRV)
• Severe allergic reaction (e.g., anaphylaxis) after a previous dose or to a vaccine component
3
• Severe immunodeciency (e.g., hematologic and solid tumors, receipt of chemotherapy, congenital immunodeciency,
long-term immunosuppressive therapy or patients with HIV infection who are severely immunocompromised)
• Pregnancy
• Family history of altered immunocompetence, unless veried clinically or by laboratory testing as immunocompetent
• Recent (≤11 months) receipt of antibody-containing blood product (specic interval depends on product)
• History of thrombocytopenia or thrombocytopenic purpura
• Need for tuberculin skin testing or interferon-gamma release assay (IGRA) testing
• Moderate or severe acute illness with or without fever
• For MMRV only: Personal or family (i.e., sibling or parent) history of seizures of any etiology
Meningococcal ACWY (MenACWY)
MenACWY-CRM [Menveo]
MenACWY-TT [MenQuad]
• Severe allergic reaction (e.g., anaphylaxis) after a previous dose or to a vaccine component
3
• For Men ACWY-CRM only: severe allergic reaction to any diphtheria toxoid—or CRM197—containing vaccine
• For MenACWY-TT only: severe allergic reaction to a tetanus toxoid-containing vaccine
• For MenACWY-CRM only: Preterm birth if less than age 9 months
• Moderate or severe acute illness with or without fever
Meningococcal B (MenB)
MenB-4C [Bexsero]
MenB-FHbp [Trumenba]
• Severe allergic reaction (e.g., anaphylaxis) after a previous dose or to a vaccine component
3
• Pregnancy
• For MenB-4C only: Latex sensitivity
• Moderate or severe acute illness with or without fever
Meningococcal ABCWY
(MenACWY-TT/MenB-FHbp) [Penbraya]
• Severe allergic reaction (e.g., anaphylaxis) after a previous dose or to a vaccine component
3
• Severe allergic reaction to a tetanus toxoid-containing vaccine
• Moderate or severe acute illness, with or without fever
Mpox [Jynneos] • Severe allergic reaction (e.g., anaphylaxis) after a previous dose or to a vaccine component
3
• Moderate or severe acute illness, with or without fever
Pneumococcal conjugate (PCV) • Severe allergic reaction (e.g., anaphylaxis) after a previous dose or to a vaccine component
3
• Severe allergic reaction (e.g., anaphylaxis) to any diphtheria-toxoid-containing vaccine or its component
3
• Moderate or severe acute illness with or without fever
Pneumococcal polysaccharide (PPSV23) • Severe allergic reaction (e.g., anaphylaxis) after a previous dose or to a vaccine component
3
• Moderate or severe acute illness with or without fever
Poliovirus vaccine, inactivated (IPV) • Severe allergic reaction (e.g., anaphylaxis) after a previous dose or to a vaccine component
3
• Pregnancy
• Moderate or severe acute illness with or without fever
RSV monoclonal antibody (RSV-mAb) • Severe allergic reaction (e.g., anaphylaxis) after a previous dose or to a vaccine component
5
• Moderate or severe acute illness with or without fever
Respiratory syncytial virus vaccine (RSV) • Severe allergic reaction (e.g., anaphylaxis) after a previous dose or to a vaccine component
3
• Moderate or severe acute illness with or without fever
Rotavirus (RV)
RV1 [Rotarix]
RV5 [RotaTeq]
• Severe allergic reaction (e.g., anaphylaxis) after a previous dose or to a vaccine component
3
• Severe combined immunodeciency (SCID)
• History of intussusception
• Altered immunocompetence other than SCID
• Chronic gastrointestinal disease
• RV1 only: Spina bida or bladder exstrophy
• Moderate or severe acute illness with or without fever
Tetanus, diphtheria, and acellular pertussis
(Tdap)
Tetanus, diphtheria (Td)
• Severe allergic reaction (e.g., anaphylaxis) after a previous dose or to a vaccine component
3
• For Tdap only: Encephalopathy (e.g., coma, decreased level of consciousness, prolonged seizures) not attributable to
another identiable cause within 7 days of administration of previous dose of DTP, DTaP, or Tdap
• Guillain-Barré syndrome (GBS) within 6 weeks after a previous dose of tetanus-toxoid–containing vaccine
• History of Arthus-type hypersensitivity reactions after a previous dose of diphtheria-toxoid–containing
or tetanus-toxoid–containing vaccine; defer vaccination until at least 10 years have elapsed since the last
tetanus-toxoid–containing vaccine
• For Tdap only: Progressive or unstable neurological disorder, uncontrolled seizures, or progressive
encephalopathy until a treatment regimen has been established and the condition has stabilized
• Moderate or severe acute illness with or without fever
Varicella (VAR) • Severe allergic reaction (e.g., anaphylaxis) after a previous dose or to a vaccine component
3
• Severe immunodeciency (e.g., hematologic and solid tumors, receipt of chemotherapy, congenital immunodeciency,
long-term immunosuppressive therapy or patients with HIV infection who are severely immunocompromised)
• Pregnancy
• Family history of altered immunocompetence, unless veried clinically or by laboratory testing as immunocompetent
• Recent (≤11 months) receipt of antibody-containing blood product (specic interval depends on product)
• Receipt of specic antiviral drugs (acyclovir, famciclovir, or valacyclovir) 24 hours before vaccination (avoid
use of these antiviral drugs for 14 days after vaccination)
• Use of aspirin or aspirin-containing products
• Moderate or severe acute illness with or without fever
• If using MMRV, see MMR/MMRV for additional precautions
1. When a contraindication is present, a vaccine should NOT be administered. Kroger A, Bahta L, Hunter P. ACIP General Best Practice Guidelines for Immunization. www.cdc.gov/vaccines/hcp/acip-recs/general-recs/contraindications.html
2. When a precaution is present, vaccination should generally be deferred but might be indicated if the benet of protection from the vaccine outweighs the risk for an adverse reaction. Kroger A, Bahta L, Hunter P. ACIP General Best Practice Guidelines for
Immunization. www.cdc.gov/vaccines/hcp/acip-recs/general-recs/contraindications.html
3. Vaccination providers should check FDA-approved prescribing information for the most complete and updated information, including contraindications, warnings, and precautions. Package inserts for U.S.-licensed vaccines are available at
www.fda.gov/vaccines-blood-biologics/approved-products/vaccines-licensed-use-united-states.
4. For information on the pregnancy exposure registries for persons who were inadvertently vaccinated with Heplisav-B or PreHevbrio while pregnant, please visit heplisavbpregnancyregistry.com or www.prehevbrio.com/#safety.
5. Full prescribing information for BEYFORTUS (nirsevimab-alip) www.accessdata.fda.gov/drugsatfda_docs/label/2023/761328s000lbl.pdf
Recommended Child and Adolescent Immunization Schedule for Ages 18 Years or Younger, United States, 2024
Addendum
Vaccines Recommendations Eective Date of Recommendation*
COVID-19 (Moderna,
Pzer-BioNTech, Novavax)
• ACIP recommends 2024-2025 COVID-19 vaccines as authorized or approved by FDA in persons ≥6 months of age. June 27, 2024
Inuenza • ACIP rearms the recommendation for routine annual inuenza vaccination of all persons aged ≥6 months who do not
have contraindications.
• ACIP recommends high-dose inactivated (HD-IIV3) and adjuvanted inactivated (aIIV3) inuenza vaccines as acceptable options for
inuenza vaccination of solid organ transplant recipients aged 18 through 64 years who are on immunosuppressive medication
regimens, without a preference over other age-appropriate IIV3s or RIV3.
June 27, 2024
Vaxelis (DTaP-IPV-Hib-HepB) • ACIP recommends DTaP-IPV-Hib-HepB (Vaxelis®) should be included with PRP-OMP (PedvaxHIB®) in the preferential recommendation
for American Indian and Alaska Native infants based on the Haemophilus inuenzae type b (Hib) component.
June 26, 2024
In addition to the recommendations presented in the previous sections of this immunization schedule, ACIP has approved the following recommendations by majority vote since October 26, 2023. The following
recommendations have been adopted by the CDC Director and are now ocial. Links are provided if these recommendations have been published in Morbidity and Mortality Weekly Report (MMWR).
*The eective date is the date when the CDC director adopted the recommendation and when the ACIP recommendation became ocial.
