
1
COVID-19 Hospitalizations and Deaths
by Vaccination Status in Washington
State
Washington State Department of Health
December 12, 2023

2
Page Break
To request this document in another format, call 1-800-525-0127. Deaf or hard of hearing customers,
please call 711 (Washington Relay) or email civil.rights@doh.wa.gov.
Publication Number: 421-010
For more information or additional copies of this report:
Division of Prevention and Community Health
Office of Immunization
111 Israel Rd SE
Tumwater, WA 98501
Phone: 360-236-3595
Email: WAIISDataR[email protected]
3
Page break
COVID-19 Hospitalizations and Deaths by Vaccination Status in
Washington State
December 12, 2023
PLEASE NOTE: information about bivalent booster doses (authorized in the fall of 2022) or the updated
monovalent booster doses (authorized in September of 2023) is not included in this report.
Summary
From November 02 to November 29, 2023, unvaccinated individuals were
between 1.6 and 7.2 times more likely to be hospitalized with COVID-19
compared to those who received at least one booster dose*
From October 11 to November 07, 2023, unvaccinated individuals were
between 1.8 and 3.9 times more likely to die from COVID-19 compared to those
who received at least one booster dose*,**
*Booster here includes additional doses or monovalent booster doses (excluding bivalent booster doses
authorized on September 1, 2022)
**Due to a small number of COVID-19 deaths, death rates in certain age groups may be unstable.
Resulting rate ratios should be interpreted with caution.
4
Background
Vaccination is an important tool for controlling the COVID-19 pandemic. COVID-19 vaccines are safe and
effective at reducing the risk of severe infections, being hospitalized, and dying from COVID-19.
Approximately 70% of the Washington population ages 6 months and older has completed the primary
COVID-19 vaccine series and over half of the population has received at least one booster dose (shot).
Unvaccinated Washingtonians are at risk of serious outcomes such as being hospitalized and dying due
to COVID-19. Because vaccination rates vary across the state between different age and demographic
groups, some populations may be more vulnerable to these serious outcomes.
Although COVID-19 vaccines work well to protect against severe infections, being hospitalized, and
dying from COVID-19, some people who are vaccinated with the primary series or who have received a
booster dose will still get COVID-19 if they are exposed to the virus. As more individuals become
vaccinated it is natural to see more vaccinated individuals get COVID-19, and even be hospitalized or die
from COVID-19. However, because people who have completed the primary series and received at least
one booster dose are much less likely to be hospitalized or die compared to those who are
unvaccinated, increasing vaccination rates remains important to protect Washingtonians from severe
COVID-19 infections and save lives.
This report shows the impact of vaccination on COVID-19 in Washington by describing rates of COVID-19
hospitalizations and deaths among people with different vaccination statuses. This report breaks
vaccination status into three categories:
• Unvaccinated: those who have not received any dose of an authorized COVID-19 vaccine.
• Completed primary series: those who have reached two weeks after receiving the final
recommended dose of the primary series of an authorized COVID-19 vaccine.
• Boosted: those who have reached two weeks after receiving at least one booster dose or
additional dose of a monovalent COVID-19 vaccine. Individuals are not considered to have full
protection from the monovalent booster dose until two weeks have passed since receiving the
dose because of the time required for the body to build protection. COVID-19 vaccines
categorized as “monovalent” are the initial COVID-19 vaccines that were approved by the Food
and Drug Administration (FDA). Doses used for the primary series are monovalent vaccines.
These are different from the new booster doses authorized by the FDA for use beginning
September 1, 2022, which are referred to as “bivalent” vaccines. Data about doses of bivalent
boosters are not included in this report.
PLEASE NOTE:
• Information about bivalent boosters is not included in this report in order to avoid confusing the
impact of the bivalent boosters with the impact of the monovalent boosters. Individuals who
received a bivalent booster after August 31, 2022 are removed from the analyses presented
here. Information about the impact of bivalent boosters will be included in future iterations of
this report once a sufficient percentage of the Washington population has received the booster.
• Immunocompromised individuals are eligible for an additional dose following the primary series
before being eligible for a booster dose. Because DOH has very limited data about whether
someone is immunocompromised, all doses received after the primary series is completed are
categorized as “booster doses”.
5
Page break
Impact of vaccination on COVID-19 hospitalization and death rates
The following graphs show the 28-day COVID-19 hospitalization and death rates by age group for people
with different vaccination statuses. There are several important things to know when looking at the
data. First, individuals who have received at least one booster dose (represented by the blue line in the
following graphs) are less likely to be hospitalized or die of COVID-19 compared to those who are
unvaccinated (red line) as well as those who completed the primary series (yellow line). However,
because even very effective vaccines cannot prevent all infections, some individuals who have received
at least one booster dose may still get sick, be hospitalized with, or die from COVID-19. There may also
be other reasons these individuals experience serious outcomes even after receiving at least one
booster dose, including:
• Decreasing protection from the vaccine as more time passes from when individuals received
their most recent dose
• The spread of variants that are more contagious and that current vaccines are not as protective
against
• Individuals and society changing behaviors such as masking, social distancing, and seeking
testing
Interpreting the data is challenging because of several factors that are hard to measure in the
population, including:
• The number of unvaccinated individuals with some protection from COVID-19 because of a
previous infection
• The use of at-home tests and information about people who test positive not being reported to
the Washington State Department of Health
• Individuals with underlying health conditions potentially being more likely to both get
vaccinated and boosted, and also experience severe outcomes from COVID-19
• The potential for people who are vaccinated and boosted to be more likely to be tested
These limitations have a larger impact on case data compared to hospitalization and death data and
have also become more relevant in recent months. Therefore, case data are no longer included in this
report.

6
Page break
Trends in COVID-19 hospitalization and death rates by vaccination status and
age group
The following graphs show the 28-day COVID-19 hospitalization and death rates by age group for people
who are unvaccinated, people who have completed the primary series, and people who have received at
least one booster dose. The difference between rates in the unvaccinated population compared to the
population of those who have received at least one booster dose during the grayed out 28-day time
period is shown in text on each graph.
PLEASE NOTE: the Y-axes of the following graphs differ between age groups.
COVID-19 Hospitalizations
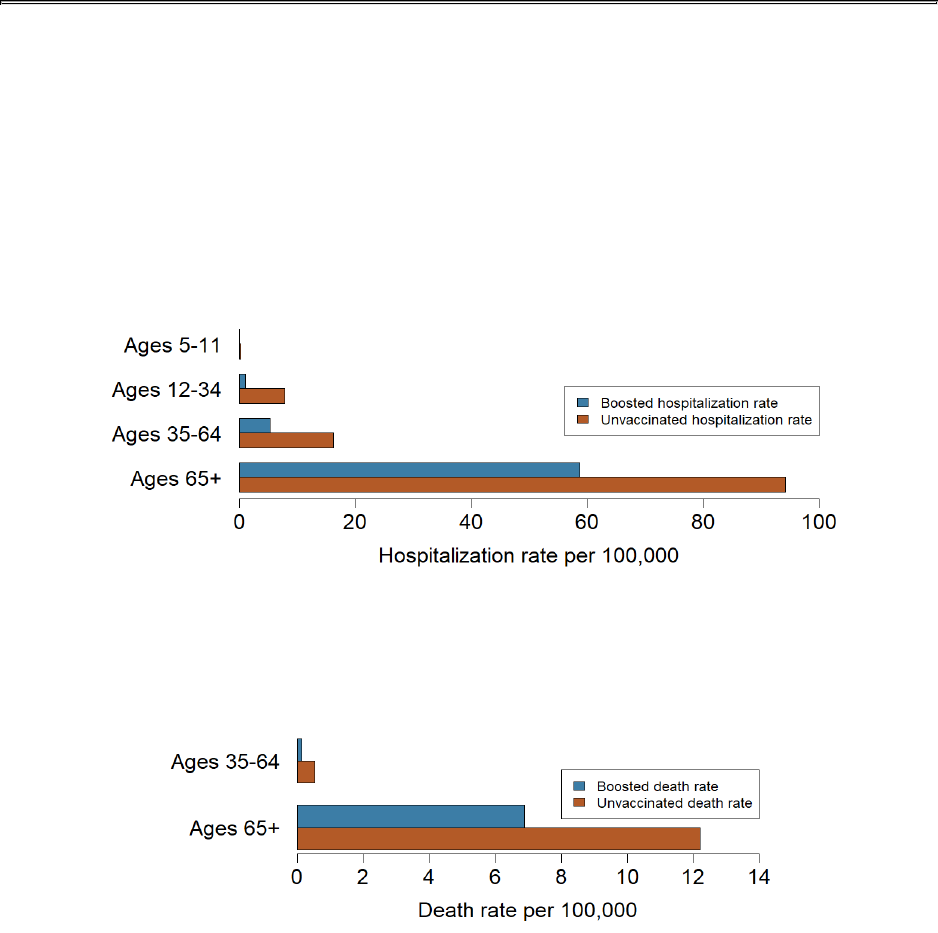
COVID-19 hospitalization rates per 100,000 population by age and vaccination
status, November 02 to November 29, 2023
Age group
Age-specific rate
per 100,000 among
unvaccinated
individuals
Age-specific rate
per 100,000 among
those who received
at least one booster
dose
Likelihood of unvaccinated
individuals being hospitalized with
COVID-19 compared to those who
received at least one booster dose
5 - 11
Not calculated*
Not calculated*
Not calculated*
12 - 34
7.9
1.1
7.2
35 - 64
16.3
5.3
3.1
65+
94.1
58.7
1.6
*Rates are not calculated when there are five or fewer hospitalizations in either the unvaccinated or
boosted group. The underlying number of hospitalizations is too small to analyze and draw meaningful
conclusions about the impact of booster doses on hospitalizations.
7
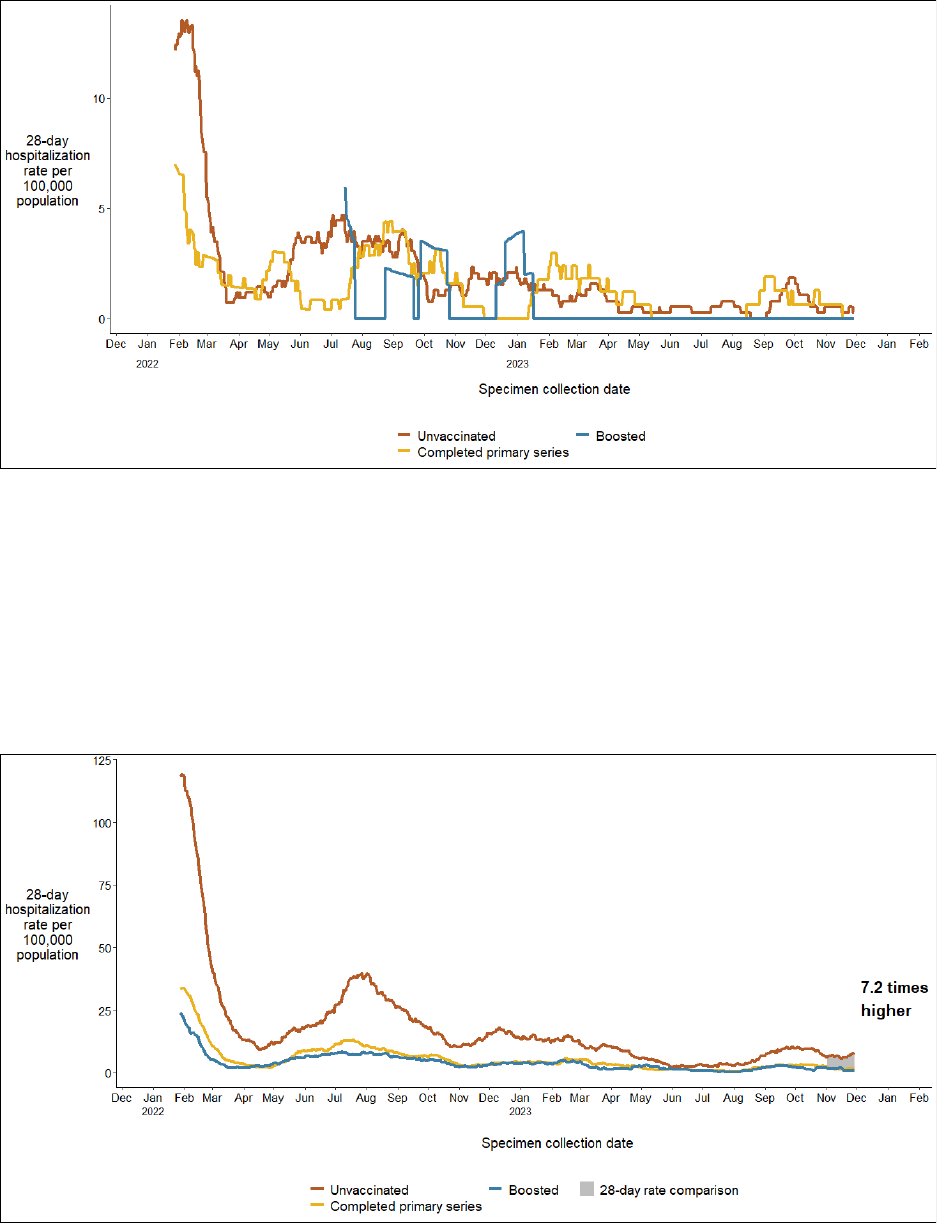
COVID-19 hospitalization rates among 5 - 11 year-olds were low across all
vaccination statuses*
*The rate ratio is not included on this graph because the underlying number of hospitalizations is too
small to analyze and draw meaningful conclusions about the impact of booster doses on hospitalizations
in this age group
COVID-19 hospitalization rates among 12 - 34 year-olds are 7.2 times higher in
the unvaccinated population than in the population receiving at least one
booster
8
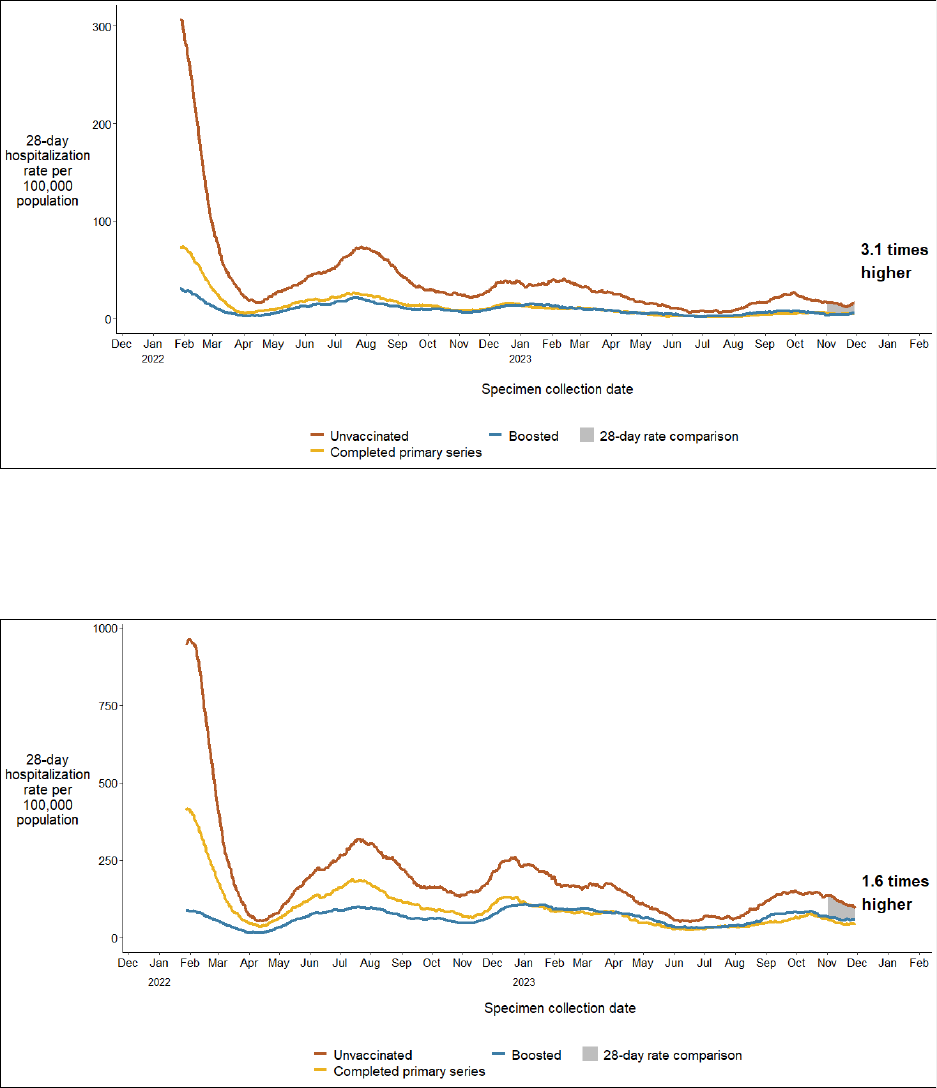
COVID-19 hospitalization rates among 35 - 64 year-olds are 3.1 times higher in
the unvaccinated population than in the population receiving at least one
booster
COVID-19 hospitalization rates among 65+ year-olds are 1.6 times higher in the
unvaccinated population than in the population receiving at least one booster
9
COVID-19 Deaths
Deaths are only shown for Washingtonians ages 35 years and older due to the relatively small number of
deaths in other age groups and associated instability in rates when assessing by vaccination status.
COVID-19 death rates per 100,000 population by age and vaccination status,
October 11 to November 07, 2023
Age group
Age-specific rate
per 100,000 among
unvaccinated
individuals
Age-specific rate
per 100,000 among
those who received
at least one booster
dose
Likelihood of unvaccinated
individuals dying from COVID-19
compared to those who received
at least one booster dose
35 - 64
0.5
0.1
3.9
65+
12.2
6.9
1.8
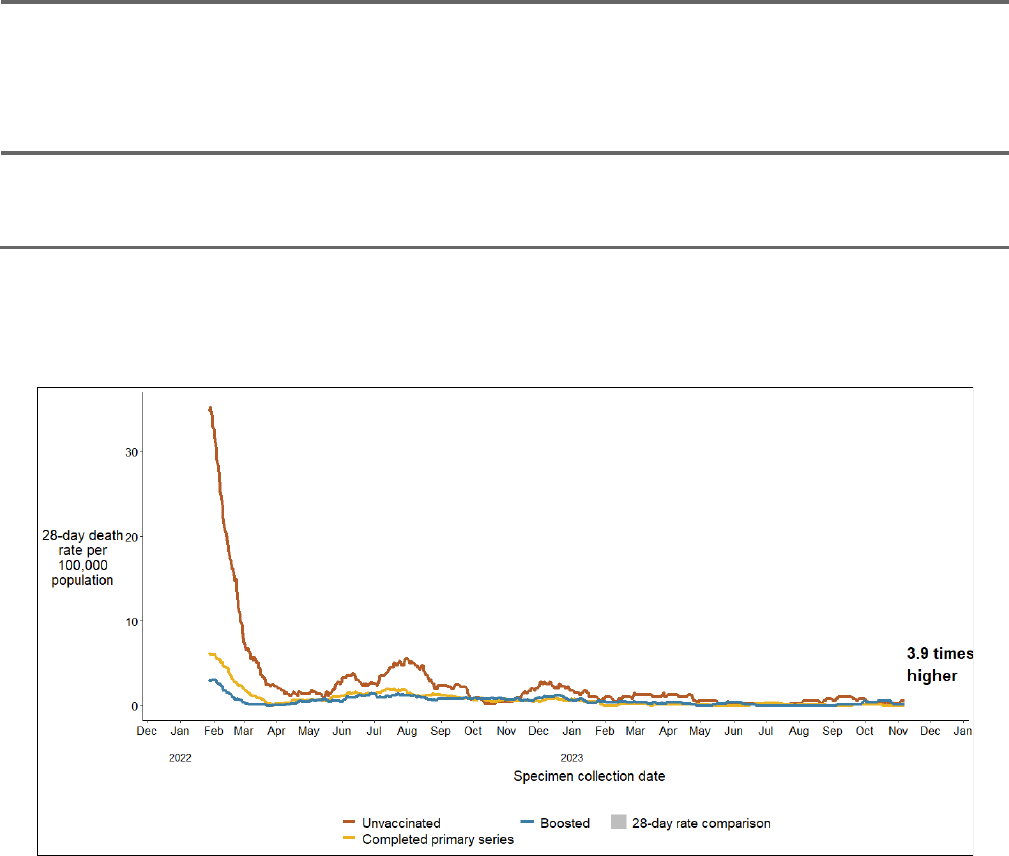
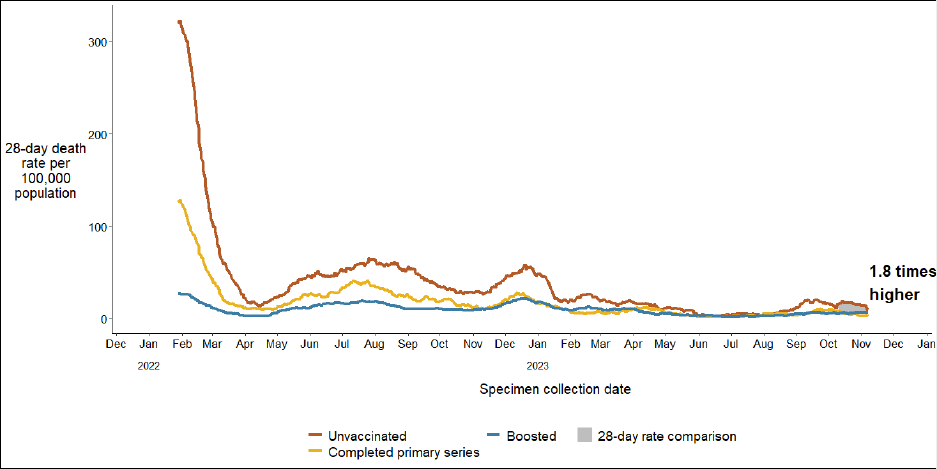
COVID-19 death rates among 35 - 64 year-olds are 3.9 times higher in the
unvaccinated population than in the population receiving at least one booster
10
COVID-19 death rates among 65+ year-olds are 1.8 times higher in the
unvaccinated population than in the population receiving at least one booster
11
Page break
Methods
Data sources
There are three data sources for these metrics:
• Washington Disease Reporting System (WDRS)
• Washington Immunization Information System (WAIIS)
• Washington Health and Life Events System (WHALES)
WDRS receives case, hospitalization, and death information for all COVID-19 cases in Washington
residents reported to the Washington State Department of Health. WAIIS collects COVID-19 vaccination
data from healthcare providers for people vaccinated in Washington, as well as Washington residents
vaccinated outside of the state. Not all federal correction facilities and federal healthcare organizations
(e.g., Department of Defense and Veterans Administration facilities) submit data to WAIIS. WAIIS may
not include vaccination data from all tribal health facilities.
All rates presented in this report are calculated using the Washington state population distribution
based on the Office of Financial Management’s (OFM) April 1, 2020 population estimates. To better
reflect the true rates of COVID-19 hospitalizations and deaths in this report, calculations include only the
5 years and older population because they were eligible to receive booster doses during the assessment
period.
Definitions
All case, hospitalization, and death data reported are based on positive molecular or antigen test results.
A person classified as a COVID-19 case who received at least one booster dose is defined as a person
with a positive molecular or antigen test result and a specimen collection date two or more weeks after
their first booster dose of a monovalent COVID-19 vaccine is administered. An individual is considered to
have received a booster dose two weeks after an age-appropriate booster dose is administered.
A COVID-19 hospitalization is a hospitalization of a Washington resident with COVID-19 who has been
identified using case investigation data in WDRS or links with Rapid Health Information Network (RHINO)
records as hospitalized with confirmed or probable COVID-19.
Deaths are reported to the state by health care providers, medical examiners or coroners, local health
departments, or others to the official vital records database, WHALES. COVID-19 deaths included in this
report are identified in WHALES where the cause of death was confirmed or suspected to have been
COVID-19, and require a known positive test result.
Timeframes
It takes up to 10 days from specimen collection date for DOH to receive information about 90% of
reported cases, 10 days for DOH to identify hospitalizations, and 32 days to identify deaths. For this
reason, we report time periods differently for hospitalizations and deaths to ensure we are reporting the
most complete data.
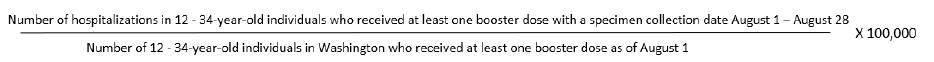
12
Linking methods
All information on COVID-19 cases, hospitalizations, and deaths in this report use WDRS data linked to
WAIIS COVID-19 vaccination data. The method to link data was updated in July 2022 to include more
robust linking methods. The links are based on a comparison of the following demographic information
present in both WDRS and WAIIS:
• first name
• last name
• middle initial
• date of birth
• sex at birth
• phone number
• zip code
The new methodology identifies matches even when there are slight variations in the spelling of names
or small errors in other demographic information between the two data sources. This method is a more
robust method of linking data than the previous method, which required exact matches of first name,
last name, and date of birth for records to be considered a match. COVID-19 cases with vaccines not
reported to WAIIS as described above are considered unvaccinated in this report.
28-day rate calculations
Rates are calculated as the total number of hospitalizations or deaths within the specified vaccination
and age groups with a specimen collection date during the 28-day period divided by the population that
falls in the same age group with the same vaccination status at the beginning of the 28-day period.
For example,
28-day COVID-19 hospitalization rates per 100,000 population among 12 - 34 year-old individuals who
received at least one booster dose =
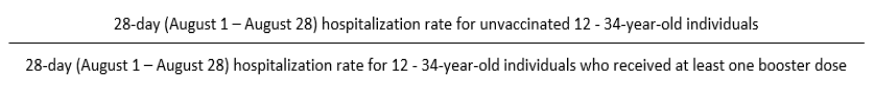
13
Page break
To compare rates in the population of individuals who received at least one booster dose to the
unvaccinated population, the rates for the most recent four-week (i.e., 28-day) period is calculated. The
28-day rate in the unvaccinated population is divided by the 28-day rate in the population of individuals
who received at least one booster dose to calculate a rate ratio.
For example,
The rate ratio for the boosted and unvaccinated COVID-19 hospitalization rates per 100,000 population
among 12 - 34 year-old individuals on August 28 =
