
Soda Can Calorimeter
Energy Content of Food
Introduction
Have you ever noticed the nutrition label located on the packaging of the food you buy? One of the first things listed on
the label are the calories per serving. How is the calorie content of food determined? This activity will introduce the
concept of calorimetry and investigate the caloric content of snack foods.
Concepts
•Calorimetry • Conservation of energy • First law of thermodynamics
Background
The law of conservation of energy states that energy cannot be created or destroyed, only converted from one form to
another. This fundamental law was used by scientists to derive new laws in the field of thermodynamics—the study of heat
energy, temperature, and heat transfer. The First Law of Thermodynamics states that the heat energy lost by one body is
gained by another body. Heat is the energy that is transferred between objects when there is a difference in temperature.
Objects contain heat as a result of the small, rapid motion (vibrations, rotational motion, electron spin, etc.) that all atoms
experience. The temperature of an object is an indirect measurement of its heat. Particles in a hot object exhibit more rapid
motion than particles in a colder object. When a hot and cold object are placed in contact with one another, the faster
moving particles in the hot object will begin to bump into the slower moving particles in the colder object making them
move faster (vice versa, the faster particles will then move slower). Eventually, the two objects will reach the same
equilibrium temperature—the initially cold object will now be warmer, and the initially hot object will now be cooler. This
principle is the basis for calorimetry, or the measurement of heat transfer.
In the 1770s, Joseph Black (1728–1799) was one of the first scientists to conduct calorimetry experiments with different
materials. He discovered that not all materials are equal when it comes to heat transfer. He concluded that different
materials have their own unique ability to retain heat energy. Some materials, like water, can gain a large amount of heat
energy without a significant change in temperature, while other materials, such as metals, will have a more dramatic
temperature change for the same amount of heat energy gained. This property is based mainly on the structure of the
material, the size of the atoms and molecules, and the interactions between them. This is known as the specific heat of the
substance. The specific heat is defined as the heat energy required to raise the temperature of one gram of a substance by
one degree Celsius. The unit of energy commonly associated with heat is called a calorie. Water has a defined specific heat
of 1 cal/g °C so it takes one calorie of energy to raise the temperature of one gram of water by one degree Celsius. (The
reverse is also true, remove one calorie of heat from one gram of water, and the temperature will decrease by one degree
Celsius.) With the specific heat of a substance known, the amount of heat energy gained or lost by a substance can then be
calculated if the temperature change is measured.
In this experiment, the specific heat of water and its change in temperature will be used to determine the caloric content
of a food sample. The normal unit for measuring the energy content in food is called a Calorie (with an uppercase C). A
Calorie is really a kilocalorie, or 1000 calories (lowercase c). During calorimetry, food burns and its stored energy is quickly
converted into heat energy and products of combustion (carbon dioxide and water). The heat energy that is released is then
transferred into the water above it in the calorimeter. The temperature change in the water is then measured and used to
calculate the amount of heat energy released from the burning food. The heat energy is calculated using Equation 1.
Q = mCT Equation 1
where
Q = heat energy
m = mass of the water
C = specific heat of the water
T = change in water temperature, T
final
– T
initial
(“” is the Greek letter Delta which means “change in”)
© 2016 Flinn Scientific, Inc. All Rights Reserved. 1
Publication No. 10861
061616
SCIENTIFIC
FAX!
SCIENCE
PHYSICAL SCIENCE-FAX
. . .makes science teaching easier.
© 2017 Flinn Scientific, Inc. All Rights Reserved.
Soda Can Calorimeter continued
2
© 2016 Flinn Scientific, Inc. All Rights Reserved.
Materials
Balance (0.01-g precision) Snack foods (cheese puffs, popcorn, marshmallows, etc.)
Cork stopper Soda can, empty and clean
Butane safety lighter Stirring rod, glass
Graduated cylinder, 50-mL Support stand
Metal ring with clamp Thermometer
Pin, large straight Water, distilled or tap, 50 mL
Ruler, metric
Safety Precautions
Wear safety glasses when performing this or any lab that uses chemicals, heat or glassware. Care should be taken when handling or placing
food onto the pin point. Allow the food sample to cool before touching or discarding it. Use a glass stirring rod to stir the liquid; never stir
with a thermometer. Students should not be allowed to eat the snack foods once they are brought
into the lab. This lab should be performed in a well-ventilated room. Wash hands thoroughly
with soap and water before leaving the laboratory.
Procedure
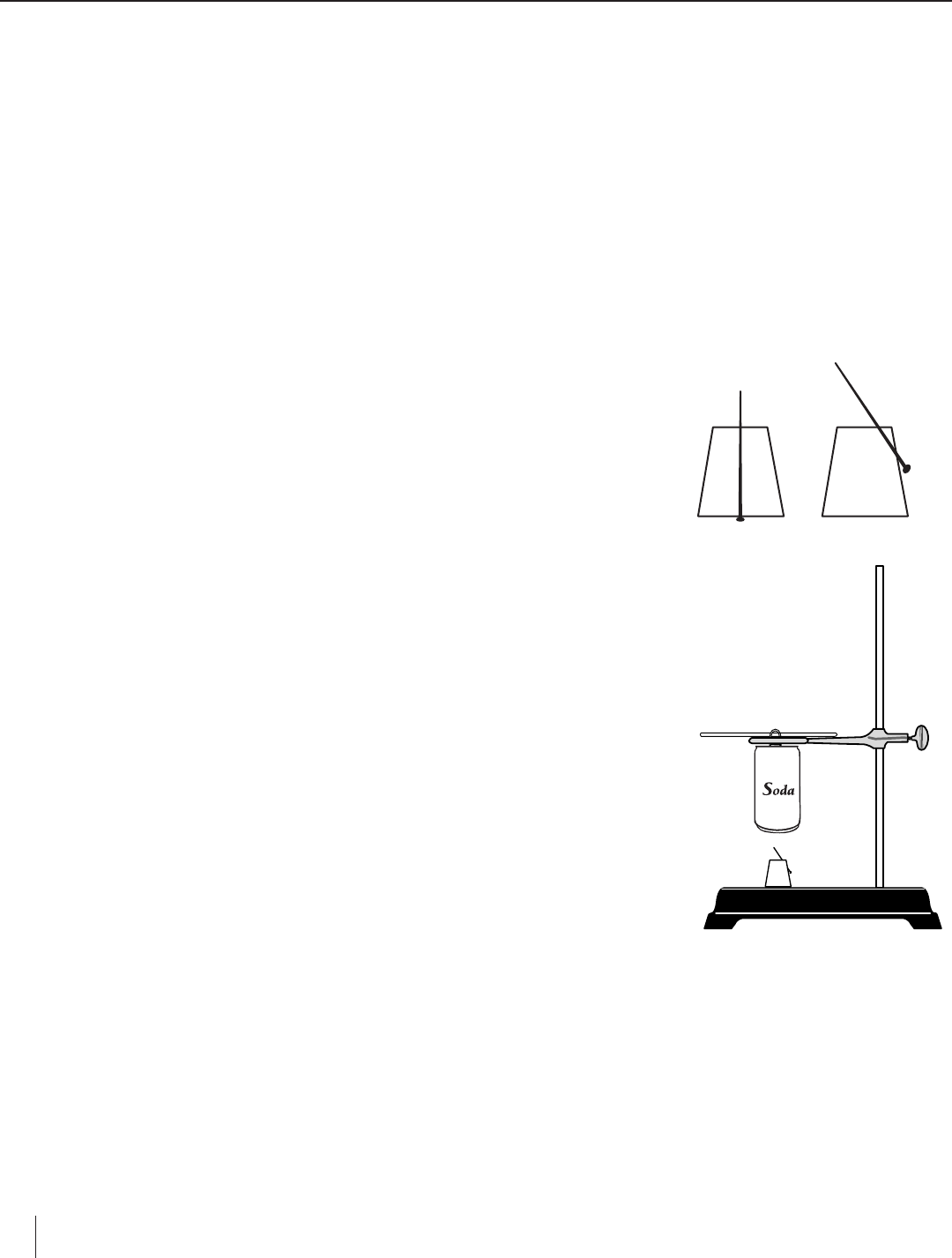
1. Push the pin through the cork so that the pin head is flush with the cork. If the pin
is large enough, try to go through the center. If this is hard to do, try to insert the
pin at an angle through the side and top of the cork (see Figure 1). Note: This setup
will now be referred to as the “Food Holder.”
2. Place a food sample on the food holder. Measure and record the combined mass of the
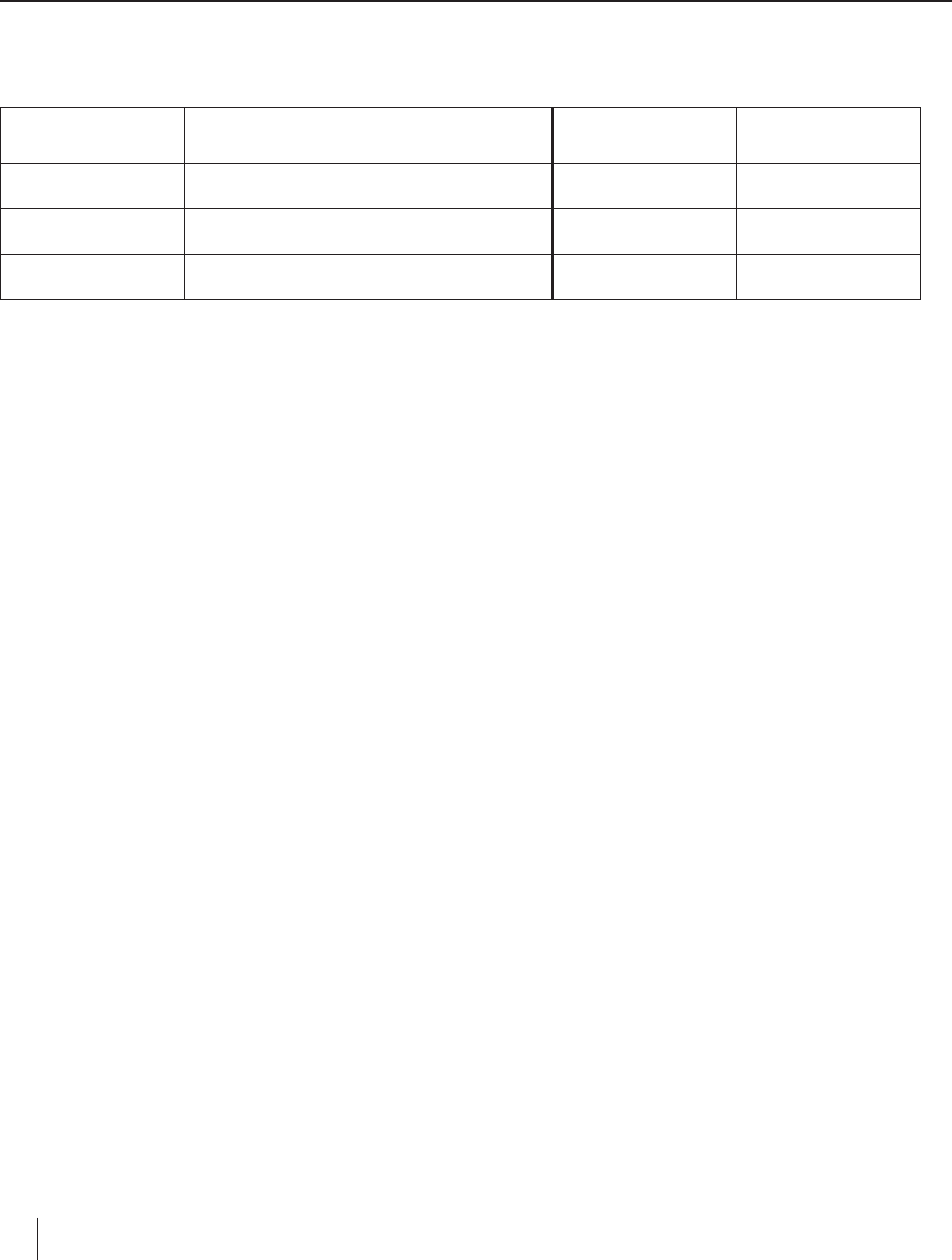
food holder and sample. Place the food holder on the base of a support stand.
3. Using a graduated cylinder, measure and add 50.0 mL of water to an empty, clean soda
can.
4. Bend the tab on the soda can and slide a glass stirring rod through the hole. Suspend
the can on a support stand using a metal ring (see Figure 2). Adjust the height of the
can so that it is about 2.5 cm above the food holder.
5. Insert a thermometer into the can. Measure and record the initial temperature of the
water.
6. Light the food sample and center it under the soda can. Allow the water to be heated
until the food sample stops burning. Record the maximum (final) temperature of the
water in the can.
7. Measure and record the final mass of the food holder and sample.
8. Allow the can and pin to cool, and then clean the bottom of the can and remove any
food residue from the food holder.
9. Repeat steps 1–8 two more times with two different snack food samples.
Disposal
Please consult your current Flinn Scientific Catalog/Reference Manual for general guidelines and specific procedures, and review
all federal, state and local regulations that may apply, before proceeding. Burned food samples should be allowed to cool and
may be disposed of in the trash according to Flinn Suggested Disposal Method #26a.
Figure 1.
Figure 2.
Soda Can Calorimeter continued
3
© 2016 Flinn Scientific, Inc. All Rights Reserved.
Sample Data Table (Student data may vary.)
Data Table — The Experiment
Food Sample
Initial Mass (food
sample and holder), g
Final Mass (food
sample and holder), g
Initial Temperature
of Water, °C
Final Temperature
of Water, °C
Cheese Puff 4.18 g 4.08 g 21.8 °C 27.1 °C
Marshmallow 6.08 g 6.00 g 22.0 °C 23.6 °C
Onion Ring 4.87 g 4.74 g 23.0 °C 30.1 °C
Analysis and Calculations (The sample calculations are for a cheese puff.)
1. Determine the change in temperature of the water by subtracting the initial water temperature from the final water
temperature.
T = T
final
– T
inital
= 27.1 °C – 21.8 °C = 5.3 °C
2. Calculate the heat gained by the water using Equation 1 from the Background section. The mass of water used is 50.0 g and
the specific heat of water (C) is 1.0 cal/g °C. These values will give you the heat gained in calories.
Q = m × C × T = 50.0 g × 1.0 cal/g°C × 5.3 °C = 265 cal.
3. Convert the heat gained from calories to food Calories (kilocalories) by dividing the answer above by 1000.
265 cal. ÷ 1000 = 0.265 Cal.
4. Determine how much of the food burned by subtracting the final mass of the cork/pin/food assembly from the initial mass.
4.18 g – 4.08 g = 0.10 g
5. Calculate the energy content per gram of the food sample. This is done by dividing the heat gain of the water (in Calories),
by the change in mass of the food sample.
0.265 Cal. ÷ 0.1 g = 2.65 Cal./g
Note to Teacher: The total energy content in Calories per gram for all the foods will be lower than the actual energy content listed on
their nutrition label. This is due to the simplicity of the calorimeter used in this experiment. However, if the foods are ranked from
highest energy content to lowest energy content based on the class results, the relative ranking should be the same as an actual ranking
from the nutrition labels. You might want to summarize the results obtained above (in Cal/g) for three foods to show this is true.

Soda Can Calorimeter continued
4
© 2016 Flinn Scientific, Inc. All Rights Reserved.
NGSS Alignment
This laboratory activity relates to the following Next Generation Science Standards (2013):
Disciplinary Core Ideas: Middle School
MS-PS1 Matter and Its Interactions
PS1.A: Structure and Properties of Matter
MS-PS3 Energy
PS3.A: Definitions of Energy
PS3.B: Conservation of Energy and Energy
Transfer
PS3.C: Relationship between Energy and Forces
Disciplinary Core Ideas: High School
HS-PS1 Matter and Its Interactions
PS1.A: Structure and Properties of Matter
HS-PS3 Energy
PS3.A: Definitions of Energy
PS3.B: Conservation of Energy and Energy
Transfer
PS3.C: Relationship between Energy and Forces
Science and Engineering Practices
Planning and carrying out investigations
Analyzing and interpreting data
Using mathematics and computational
thinking
Crosscutting
Concepts
Cause and effect
Energy and matter
Tips
• A butane safety lighter (Flinn Catalog No. AP8960) is recommended instead of matches because it may take about 10
seconds for the food to ignite.
• For further concept development, try the Flinn Scientific “Calorimetry Basics—Specific Heat Laboratory Kit” (Catalog
No. AP5952).
• Have students pin the food piece at one of the ends so that the piece “points up” and the length is parallel to the pin.
• It may take about 10 seconds to get the food ignited, so some heat related to the burning food will be lost during this
process. A small flame on the food will spread and engulf it over time.
• Be sure that when the food sample burns, it is close to but not touching the soda can. If it is too close to the bottom of
the can, it may extinguish too early due to a lack of oxygen.
• Black carbon soot will deposit on the bottom of the can when the food burns. For best results, this soot should be wiped
off with a little water and a paper towel between trials.
• Have students try different samples of food in order to compare the caloric contents of different foods. Note: Avoid sugar
cookies, pretzels, soda crackers or other food samples with a high sugar content. They tend to get soft as they burn and
may fall off the pin. Walnuts, pecans, popped corn, and Cheetos
®
(or other puffed snacks) are good choices.
• Good ventilation is required since burning food can generate a large amount of smoke. Allow some time between trials so
that the smoke has time to dissipate.
References
Cesa, I. Flinn ChemTopic
™
Labs, Volume 10, Thermochemistry; Batavia, IL, 2002; pp 39–49.
Kotz, J. C.; Treichel, Jr., Paul. Chemistry and Chemical Reactivity, 3rd Ed.; Saunders College: New York, 1996; pp 264–271.
Tipler, P. A. Physics for Scientists and Engineers, 3rd Ed., Vol. 1; Worth: New York, 1990; pp 518–524, 534–537.

Soda Can Calorimeter continued
5
© 2016 Flinn Scientific, Inc. All Rights Reserved.
Materials for Soda Can Calorimeter are available from Flinn Scientific, Inc.
Catalog No. Description
OB2141 Flinn Scientific Electronic Balance (0.01-g precision)
AP8308 Cork Stopper, Size 8
AP8960 Butane Safety Lighter
GP2044 Graduated Cylinder, Borosilicate Glass, Plastic Base, 50 mL
AP8232 Metal Ring Support with Rod Clamp
AB1039 Pin, 20 Dissection
GP5075 Stirring Rod, Glass
AP8228 Support Stand
AP6049 Flinn Digital Pocket Thermometer
Consult your Flinn Scientific Catalog/Reference Manual for current prices.
