September 2011
EMA/CHMP/ICH/309348/2008
ICH guideline E2F on development safety update report
Step 5
Transmission to CHMP
June 2008
Adoption by CHMP for release for consultation June 2008
End of consultation (deadline for comments) December 2008
Final adoption by CHMP September 2010
Date for coming into effect September 2011
7 Westferry Circus ● Canary Wharf ● London E14 4HB ● United Kingdom
An agency of the European Union
Telephone
+44 (0)20 7418 8400
Facsimile
+44 (0)20 7418 8416
E-mail
ich@ema.europa.eu
Website
www.ema.europa.eu
© European Medicines Agency, 2013. Reproduction is authorised provided the source is acknowledged.

E2F Development of safety update report
Table of contents
1. Introduction ............................................................................................ 4
1.1. Background ............................................................................................. 4
1.2. Objectives ............................................................................................... 4
1.3. Scope of the DSUR ................................................................................... 5
1.4. Relation of the DSUR to the periodic safety update report ............................. 5
1.5. Recipients of the DSUR ............................................................................. 6
2. General principles .................................................................................... 6
2.1. Single DSUR for an active substance .......................................................... 6
2.2. Periodicity and DSUR data lock point* ........................................................ 6
2.3. Duration of DSUR submissions ................................................................... 7
2.4. Responsibilities for preparing and submitting a DSUR ................................... 7
2.4.1. Sponsor’s responsibilities ....................................................................... 7
2.4.2. Responsibilities of multiple parties ........................................................... 7
2.5. DSURs for combination therapies ............................................................... 7
2.6. Reference safety information ..................................................................... 8
2.7. Format and presentation of DSUR .............................................................. 9
2.7.1. Format................................................................................................. 9
2.7.2. Presentation ......................................................................................... 9
3. Guidance on contents of DSUR .............................................................. 10
3.1. Introduction .......................................................................................... 11
3.2. Worldwide marketing approval status ....................................................... 11
3.3. Actions taken in the reporting period for safety reasons .............................. 11
3.4. Changes to reference safety information ................................................... 12
3.5. Inventory of clinical trials ongoing and completed during the reporting period 13
3.6. Estimated cumulative exposure ............................................................... 13
3.6.1. Cumulative subject exposure in the development programme ................... 14
3.6.2. Patient exposure from marketing experience .......................................... 14
3.7. Data in line listings and summary tabulations ............................................ 15
3.7.1. Reference information.......................................................................... 15
3.7.2. Line listings of serious adverse reactions during the reporting period
......... 15
3.7.3. Cumulative summary tabulations of serious adverse events ..................... 16
3.8. Significant findings from clinical trials during the reporting period ................ 17
3.8.1. Completed clinical trials ....................................................................... 17
3.8.2. Ongoing clinical trials ........................................................................... 17
3.8.3. Long-term follow-up ............................................................................ 17
3.8.4. Other therapeutic use of investigational drug .......................................... 17
3.8.5. New safety data related to combination therapies ................................... 17
3.9. Safety findings from non-interventional studies ......................................... 18
3.10. Other clinical trial/study safety information ............................................. 18
3.11. Safety findings from marketing experience .............................................. 18
3.12. Non-clinical data .................................................................................. 18
3.13. Literature ............................................................................................ 18
3.14. Other DSURs ....................................................................................... 18
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 2/35

3.15. Lack of efficacy .................................................................................... 19
3.16. Region-specific information ................................................................... 19
3.17. Late-breaking information ..................................................................... 20
3.18. Overall safety assessment ..................................................................... 20
3.18.1. Evaluation of the risks ........................................................................ 20
3.18.2. Benefit-risk considerations .................................................................. 21
3.19. Summary of important risks .................................................................. 21
3.20. Conclusions ......................................................................................... 22
4. Appendices to this guideline .................................................................. 22
APPENDIX A - Glossary ................................................................................. 23
APPENDIX B ─ Examples of tables and table headings for clinical trial listings ...... 27
APPENDIX C— Examples of the summary of important risks .............................. 30
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 3/35

1. Introduction
The Development Safety Update Report (DSUR) proposed in this guideline is intended to be a common
standard for periodic reporting on drugs under development (including marketed drugs that are under
further study) among the ICH regions. US and EU regulators consider that the DSUR, submitted
annually, would meet national and regional requirements currently met by the US IND Annual Report
and the EU Annual Safety Report, respectively, and can therefore take the place of these existing
reports.
1
This guideline defines the recommended content and format of a DSUR and provides an
outline of points to be considered in its preparation and submission.
Definitions of the technical terms used in the guideline are included in a glossary (Appendix A); the
first mention of a term in the guideline is identified with an asterisk (*).
1.1. Background
During the clinical development of an investigational drug,*
2
periodic analysis of safety information is
crucial to the ongoing assessment of risk to trial subjects.
3,4
It is also important to inform regulators
and other interested parties (e.g., ethics committees) at regular intervals about the results of such
analyses and the evolving safety profile of an investigational drug, and apprise them of actions
proposed or being taken to address safety concerns. Currently, laws and regulations of some ICH
countries and regions require submission of a periodic report to regulatory authorities to provide this
information. However, significant differences in the content, format and timing of these reports
highlight the importance of a common standard report in promoting consistency and enhancing
efficiency. Some national and regional laws and regulations also require a periodic report that
describes the status of ongoing individual investigations, manufacturing changes, and overall
development status and plans. To be broadly useful, the DSUR should also include this information, in
addition to safety-related information. The harmonisation of the content, format, and timing of periodic
safety reports will help to ensure that regulators in the three ICH regions receive a uniform, high-
quality, comprehensive report.
1.2. Objectives
The main objective of a DSUR is to present a comprehensive, thoughtful annual review and evaluation
of pertinent safety information collected during the reporting period related to a drug under
investigation, whether or not it is marketed, by: (1) examining whether the information obtained by
the sponsor during the reporting period is in accord with previous knowledge of the investigational
drug’s safety; (2) describing new safety issues that could have an impact on the protection of clinical
trial subjects; (3) summarising the current understanding and management of identified and potential
risks;* and (4) providing an update on the status of the clinical investigation/development programme
and study results.
A DSUR should be concise and provide information to assure regulators that sponsors are adequately
monitoring and evaluating the evolving safety profile of the investigational drug. All safety issues
discovered during the reporting period should be discussed in the text of the DSUR; however, it should
1
Japan will consider existing regulations on periodic safety reporting in implementing the DSUR.
2
The term “investigational drug” is used in this guideline to indicate only the experimental product under study or
development.
3
For detailed discussion see: The Development Safety Update Report (DSUR): Harmonizing the Format and Content for
Periodic Safety Reporting During Clinical Trials: Report of CIOMS Working Group VII, Geneva 2007.
4
ICH Topic E6 (R1) Guideline for Good Clinical Practice. http://www.ich.org/LOB/media/MEDIA482.pdf
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 4/35

not be used to provide the initial notification of significant new safety information or provide the means
by which new safety issues are detected.
1.3. Scope of the DSUR
The main focus of the DSUR is data and findings from interventional clinical trials* (hereafter referred
to as “clinical trials”) of drugs and biologicals that are under investigation, whether or not they have a
marketing approval. Because clinical development of a drug frequently continues following marketing
approval,
5
relevant information from post-marketing studies should also be included in the DSUR. The
DSUR should concentrate primarily on the investigational drug, providing information on comparators
only where relevant to the safety of trial subjects.
The DSUR should provide safety information from all ongoing clinical trials and other studies that the
sponsor is conducting or has completed during the review period including:
• Clinical trials using an investigational drug (i.e., human pharmacology, therapeutic exploratory
and therapeutic confirmatory trials [Phase I – III]);
6
• Clinical trials conducted using marketed drugs in approved indications (i.e., therapeutic use
trials (Phase IV));
• Therapeutic use of an investigational drug (e.g., expanded access programmes, compassionate
use programmes, particular patient use, single patient INDs, and treatment INDs); and
• Clinical trials conducted to support changes in the manufacturing process of medicinal
products.
The DSUR should also include significant other findings pertinent to the safety of the investigational
drug, including findings from:
• Observational or epidemiological studies;
• Non-clinical studies (toxicological and in vitro studies);
• Related DSURs, if applicable to the investigational drug;
• Manufacturing or microbiological changes;
• Studies recently published in the literature;
• Clinical trials with results indicating lack of efficacy that could have a direct impact on subject
safety (e.g., worsening of the underlying condition if the indication is serious or life-
threatening);
• Any other source of relevant safety findings for products in the same therapeutic class;
• Clinical trials conducted by a co-development partner, if permitted by the contractual
agreement.
1.4. Relation of the DSUR to the periodic safety update report
At present, some ICH countries and regions accept submission of a Periodic Safety Update Report
(PSUR) to fulfil national and regional requirements for periodic reporting on the safety of approved
5
For the purposes of this document, we use the term “authorisation/authorised” to refer to approvals of clinical trials, and
“approved/marketing approval” to refer to marketing authorisations.
6
For classification of clinical trials see ICH E8 General Considerations for Clinical Trials. Current Step 5 17 July 1997.
http://www.ich.org/LOB/media/MEDIA484.pdf
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 5/35

drugs. Although the focus of the DSUR is on investigational drugs, there can be overlap between the
content of the DSUR and PSUR, and some repetition is expected. For example, information from
marketing experience (reported in the PSUR) might be relevant to clinical development, and therefore
reported in the DSUR. Safety findings from clinical trials conducted using marketed drugs would be
included in the DSUR, but would also be pertinent to post-marketing safety and would be reported in
the PSUR. Both the DSUR and PSUR should be comprehensive and stand alone
as they focus on
different subject matter and have differing periodicities and recipients.
1.5. Recipients of the DSUR
The DSUR is intended to serve as an annual report to regulatory authorities. Where national or regional
laws or regulations require submission of an annual safety report on an investigational drug to ethics
committees/institutional review boards, the DSUR Executive Summary might be appropriate,
supplemented with line listings of serious adverse reactions
7
(SARs) as warranted.
2. General principles
2.1. Single DSUR for an active substance
In order to promote a comprehensive analysis and presentation of the safety profile of the
investigational drug, a sponsor should prepare a single DSUR with data pertinent to all dosage forms
and strengths, all indications, and all patient populations under study with the investigational drug,
wherever feasible. If this is not possible (e.g., when the data are not available to the sponsor), an
explanation should be provided in the introduction section of the DSUR.
If more than one sponsor is involved in drug development, particularly in a co-development or other
contractual agreement, a single DSUR can be submitted (see section 2.4.2).
2.2. Periodicity and DSUR data lock point*
The “Development International Birth Date”* (DIBD) is used to determine the start of the annual
period for the DSUR. This date is the sponsor’s first authorisation to conduct a clinical trial in any
country worldwide. The start of the annual period for the DSUR is the month and date of the DIBD.
When the sponsor’s first clinical trial is conducted in a country without a formal authorisation process,
the sponsor should designate an appropriate date linked to the commencement of the first clinical trial.
Where clinical trials are ongoing in one country and are later initiated in another country, the original
DIBD should be maintained and used for all countries in preparing the DSUR.
The data lock point of the DSUR should be the last day of the one-year reporting period. For
administrative convenience, if desired by the sponsor, the data lock point of the DSUR can be
designated as the last day of the month prior to the month of the DIBD.
When clinical development of a drug continues following a marketing approval in any country
worldwide, both a PSUR and a DSUR should be submitted as specified by national or regional laws or
regulations. If desired by the sponsor, a DSUR can be prepared based on the PSUR International Birth
Date (IBD) so that the DSUR and the PSUR can be synchronised. In synchronising the data lock points
for the DSUR and PSUR, the period covered by the next DSUR should be no longer than one year.
7
“Serious adverse reaction,” “serious adverse event” and “adverse drug reaction” are defined in ICH E2A Clinical Safety
Data Management: Definitions and Standards for Expedited Reporting. October 1994.
http://www.ich.org/LOB/media/MEDIA436.pdf
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 6/35

The DSUR should be submitted to all concerned regulatory authorities no later than 60 calendar days
after the DSUR data lock point.
2.3. Duration of DSUR submissions
DSURs should continue to be submitted for as long as indicated by national or regional laws or
regulations.
8
When submission of an annual report is no longer required in an individual country or
region, the sponsor should indicate that the final DSUR serves as the last annual report for the
investigational drug in that country or region. The sponsor should also indicate whether or not clinical
trials are continuing elsewhere.
2.4. Responsibilities for preparing and submitting a DSUR
2.4.1. Sponsor’s responsibilities
The sponsor* of a clinical trial is considered responsible for the preparation, content and submission of
a DSUR. The sponsor can delegate the preparation of the DSUR to a third party (e.g., a contract
research organisation).
In situations where the sponsor does not have access to the information to be included in specific
sections (e.g., sponsor-investigators* might not have information on manufacturing issues, non-clinical
data, and marketing status), this should be stated in the DSUR.
2.4.2. Responsibilities of multiple parties
When there is more than one sponsor of a clinical trial or drug development programme, the parties
should arrange to prepare a single DSUR, if possible. This includes situations where a sponsor is in a
formal co-development or licensing relationship with one or more partners, or where individual clinical
trials or a drug development programme involve collaboration with public or private institutions,
business partners, or other parties. Written agreements should be in place specifying how data will be
exchanged and detailing the responsibilities for preparation and submission of the DSUR.
When a single DSUR cannot be arranged, multiple sponsors can agree to prepare separate DSURs for
the same investigational drug. This can occur where different indications, routes of administration, or
formulations are being investigated by different parties. In this situation, the rationale for separate
DSURs should be provided in each report.
2.5. DSURs for combination therapies
Given the potential complexities of clinical development involving combination therapies, it is not
possible to provide guidance that addresses all such situations. The sponsor should select the most
appropriate option based on judgement, taking into account patient population, indication, formulation,
etc., as well as the circumstances in which the clinical trials are being conducted and national or
regional laws or regulations. The rationale for this decision should be provided in the report.
In general, a single DSUR should be prepared for clinical trials involving a fixed combination product
(i.e., a product consisting of at least two active ingredients in a fixed dose that is administered in a
single dosage form). If the sponsor is also conducting clinical trials with individual component(s) of the
fixed combination product, separate DSUR(s) should be submitted for each component. Relevant
findings from each DSUR should be summarised in section 8.5 of the other DSUR(s).
8
For example, in the US, sponsors might keep an IND open even if no clinical trials are ongoing or planned. Annual reports
are submitted for as long as the IND remains open.
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 7/35

For trials involving multi-drug therapy, i.e., combinations of drugs that are not fixed, the sponsor can
prepare either:
(1) A DSUR for the multi-drug therapy, or
(2) DSUR(s) for one or more of the individual components; in this case information on the multi-
drug therapy trials can be included in the DSURs of one or all of the components.
The following table provides examples of strategies for preparation of DSURs for multi-drug therapies:
Multi-drug therapy used in clinical trial(s) DSUR
Investigational drug (A) + marketed
drug(s) (X, Y, Z)
Either a single DSUR focusing on (A+X+Y+Z)
or
A single DSUR focusing on (A)
including data on the multi-drug therapy
Two investigational drugs (A) + (B) Either a single DSUR focusing on (A + B)
or
Two separate DSURs (A) and (B), each including data on
the multi-drug therapy
Two (or more) marketed drugs as an
investigational drug combination (X, Y, Z)
A single DSUR focusing on the multi-drug therapy (X + Y
+ Z)
2.6. Reference safety information
The Investigator’s Brochure (IB) in effect at the start of the reporting period should serve as the
reference safety information to determine whether the information received during the reporting period
remains consistent with previous knowledge of the safety profile of the investigational drug. Section
7.1 of the DSUR should clearly indicate the version number and date of the IB used for this purpose.
When an IB is not required by national or regional laws or regulations, the applicable national or
regional product label
9
should serve as the reference safety information.
Usually, a single document should serve as the reference safety information. However, in certain
circumstances, it might be appropriate to use more than one reference document to support the DSUR
(e.g., for a DSUR providing information on an investigational drug used in combination and as
monotherapy).
If the IB has been revised during the reporting period and not previously submitted to the relevant
regulatory authority, the sponsor should provide a copy of the current version of the IB as an
attachment to the DSUR.
9
In the EU this would be the Summary of Product Characteristics (SmPC); in Japan this would be the Japanese Package
Insert; and in the US this would be the US Package Insert.
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 8/35

2.7. Format and presentation of DSUR
2.7.1. Format
The recommended format and content of the DSUR, including table of contents, section numbering,
and content of each section, is outlined below. For each heading where information is available, the
information should be presented concisely; when no information is available or a DSUR section is not
applicable, this should be stated.
If a sponsor intends to submit a DSUR in eCTD format, the sponsor should consult with the relevant
regulatory authority regarding the appropriate placement of the DSUR in the eCTD structure.
2.7.2. Presentation
The recommended table of contents, including section numbering, for the DSUR is provided below:
Title page
Executive Summary
Table of Contents
1. Introduction
2. Worldwide Marketing Approval Status
3. Actions Taken in the Reporting Period for Safety Reasons
4. Changes to Reference Safety Information
5. Inventory of Clinical Trials Ongoing and Completed during the Reporting Period
6. Estimated Cumulative Exposure
6.1 Cumulative Subject Exposure in the Development Programme
6.2 Patient Exposure from Marketing Experience
7. Data in Line Listings and Summary Tabulations
7.1 Reference Information
7.2 Line Listings of Serious Adverse Reactions during the Reporting
Period
7.3 Cumulative Summary Tabulations of Serious Adverse Events
8. Significant Findings from Clinical Trials during the Reporting Period
8.1 Completed Clinical Trials
8.2 Ongoing Clinical Trials
8.3 Long-term Follow-up
8.4 Other Therapeutic Use of Investigational Drug
8.5 New Safety Data Related to Combination Therapies
9. Safety Findings from Non-interventional Studies
10. Other Clinical Trial/Study Safety Information
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 9/35

11. Safety Findings from Marketing Experience
12. Non-clinical Data
13. Literature
14. Other DSURs
15. Lack of Efficacy
16. Region-Specific Information
17. Late-Breaking Information
18. Overall Safety Assessment
18.1. Evaluation of the Risks
18.2 Benefit-risk Considerations
19. Summary of Important Risks
20. Conclusions
Appendices to the DSUR
3. Guidance on contents of DSUR
All sections should be completed; when no information is available, this should be stated.
Title page
The title page of the DSUR should include the following information:
• DSUR number (reports should be numbered sequentially);
• Investigational drug(s);
• Reporting period;
• Date of the report;
• Sponsor(s) name(s) and address(es);
• Statement on the confidentiality of the information included in the DSUR;
• A cautionary statement that the DSUR includes unblinded information, if applicable.
Executive Summary
This section should provide a concise summary of the important information contained in the report.
Together with the title page, it can serve as a “stand-alone” document suitable for submission to ethics
committees and other stakeholders, if required by national or regional laws or regulations. The
following information should be included in the Executive Summary:
• Introduction – report number and reporting period;
• Investigational drug(s) – mode(s) of action, therapeutic class(es), indication(s), dose(s),
route(s) of administration, formulation(s);
• Estimated cumulative exposure of clinical trial subjects;
• Marketing approval(s)? (yes/no) – If yes, number of countries;
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 10/35

• Summary of overall safety assessment (based on section 18 of the DSUR);
• Summary of important risks (based on section 19 of the DSUR);
• Actions taken for safety reasons including significant changes to IB;
• Conclusions.
Table of Contents
3.1. Introduction
This section should include:
• DIBD or IBD (as applicable);
• Reporting period and sequential number of the report;
• Investigational drug(s) – mode(s) of action, therapeutic class(es), dose(s), route(s) of
administration, formulation(s);
• A brief description of the indication(s) and population(s) being studied;
• A short summary of the scope of the clinical trials covered by the report (e.g., all trials with the
investigational drug, indication-specific trials, trials with combination products);
• A brief description and explanation of any information that has not been included in the DSUR
(e.g., when written agreements with a partner company do not provide for exchange of all
safety data);
• The rationale for submission of multiple DSURs for the investigational drug, if applicable.
3.2. Worldwide marketing approval status
This section should provide a brief narrative overview including: date of first approval, indication(s),
approved dose(s), and where approved, if applicable.
3.3. Actions taken in the reporting period for safety reasons
This section should include a description of significant actions related to safety that have been taken
during the reporting period by the sponsor, regulators, data monitoring committees* (DMC) or ethics
committees that had an impact on the conduct of a specific clinical trial(s) or on the overall clinical
development programme.* The reason(s) for each action should be provided if known. Relevant
updates to previous actions should also be summarised in this section (e.g., resumption of a clinical
trial after suspension).
Changes to the Investigator’s Brochure should be discussed separately in the “Changes to Reference
Safety Information”; see section 3.4.
Examples of significant actions taken for safety reasons include:
Actions related to investigational drugs:
• Refusal to authorise a clinical trial for ethical or safety reasons;
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 11/35

• Partial
10
or complete clinical trial suspension or early termination of an ongoing clinical trial
because of safety findings or lack of efficacy (see section 3.15);
• Recall of investigational drug or comparator;
• Failure to obtain marketing approval for a tested indication including voluntary withdrawal of a
marketing application;
• Risk management activities, including:
o Protocol modifications due to safety or efficacy concerns (e.g., dosage changes,
changes in study inclusion/exclusion criteria, intensification of subject monitoring,
limitation in trial duration);
o Restrictions in study population or indications;
o Changes to the informed consent document relating to safety issues;
o Formulation changes;
o Addition by regulators of a special safety-related reporting requirement;
o Issuance of a communication to investigators or healthcare professionals;
o Plans for new studies to address safety issues.
Actions related to marketed drugs:
• Failure to obtain a marketing approval renewal;
• Withdrawal or suspension of a marketing approval;
• Risk management activities including:
o Significant restrictions on distribution or introduction of other risk minimisation
measures;
o Significant safety-related changes in labelling documents that could affect the
development programme, including restrictions on use or population treated;
o Communications to health care professionals;
o New post-marketing study requirement(s) imposed by regulators.
This section should also summarise requests from regulatory authority(ies) that place a specific
limitation on current or future development (e.g., a request to conduct long-term animal studies before
initiating a long-term clinical trial, specification of a maximum dose to be evaluated, a request for
specific safety data before initiating trials in paediatric subjects). A cumulative listing of such requests
from regulatory authorities should be provided, including any updates if applicable. This can be
provided as a table, in an appendix, or in this section.
3.4. Changes to reference safety information
This section should list any significant safety-related changes to the IB or other reference safety
information within the reporting period. Such changes might include information relating to exclusion
criteria, contraindications, warnings, precautions, serious adverse drug reactions, adverse events of
10
“Partial suspension” might include several actions (e.g., suspension of repeat dose studies, but continuation of single dose
studies; suspension of trials in one indication, but continuation in another and/or suspension of a particular dosing regimen in a trial
but continuation of other doses).
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 12/35

special interest*, interactions, and any important findings from non-clinical studies (e.g.,
carcinogenicity studies). Specific information relevant to these changes should be provided in the
appropriate sections of the DSUR.
3.5. Inventory of clinical trials ongoing and completed during the reporting
period
This section should provide a brief overview of the clinical trials ongoing* and completed* by the
sponsor in the reporting period, with detailed information presented in a table as an appendix (see
examples in Appendix B, Table 1 of this guideline). Separate tables can be provided by indication,
formulation, and study population, if appropriate. In addition, where required by national or regional
laws or regulations, similar information should be provided for other therapeutic use of an
investigational drug in the reporting period. The table(s) should include the following information for
each clinical trial:
• Study ID (e.g., protocol number or other identifier);
• Phase (I, II, III, or IV);
• Status:
o Ongoing (clinical trial has begun; has begun but is currently on hold; has concluded but
clinical study report has not been finalised);
o Completed (clinical study report is finalised);
• Countries/regions where there is at least one investigational site for the protocol;
• Abbreviated study title;
• Design (uncontrolled, controlled, open, single blind, double blind, parallel, cross-over, etc.,
including treatment arms);
• Dose and regimen of investigational drug and any comparators;
• Study population as appropriate (age; sex; indication(s); specific patient groups, e.g., trial
subjects with impaired renal function or trial subjects resistant to treatment);
• Date of clinical trial start (as defined by the sponsor, e.g., first visit of first patient (FVFP));
• Planned enrolment for study as a whole;
• Estimates of cumulative numbers of exposed subjects for each treatment arm, where available.
The actual enrolment numbers for open or completed trials, and/or an estimate based on the
randomisation scheme for blinded trials, should be provided.
Appendix B, Table 1 of this guideline provides an example of the column headings for such tables.
3.6. Estimated cumulative exposure
Sections 6.1 and 6.2 of the DSUR should provide information on cumulative exposure in clinical trials
and the marketed setting, respectively.
An estimation of cumulative subject exposure can help provide context for the cumulative summary
tabulations of serious adverse events (SAEs), and the overall assessment of safety. The accuracy of
the estimation of clinical trial exposure might be limited because of a number of factors, including the
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 13/35

rapidity of subject enrolment and the number of ongoing trials where treatment assignment remains
blinded.
The optimal method of data presentation will depend on a number of factors, and the following general
points should be considered in the preparation of the estimated exposure for the DSUR:
• Data should be presented in tabular format;
• When there are important differences among trials in dose, route of administration, or patient
population, these differences can be noted in the tables, or separate tables can be considered;
• If the summary tabulations of SAEs are presented by indication, the exposure data should also
be presented by indication, when available;
• When there are substantial differences in time of exposure between subjects randomised to the
investigational drug and comparator(s), or disparities in length of exposure between clinical
trials, it can be useful to express exposure data in subject-time (subject-days, -months, or -
years);
• Investigational drug exposure in healthy volunteers might be less relevant to the overall safety
profile, particularly when volunteers are exposed to only a single dose. Such data can be
presented separately with explanation, when appropriate;
• For marketed drugs that are under clinical investigation, it might not be feasible or useful to
obtain precise cumulative clinical trial exposure data, e.g., when the drug has been marketed
for a number of years and/or has many indications. In these circumstances the sponsor should
provide an explanation.
3.6.1. Cumulative subject exposure in the development programme
This section should include the following information; in tabular format (see Appendix B, Tables 2-4 of
this guideline for examples):
• The cumulative number of subjects from ongoing and completed clinical trials; the number
exposed to the investigational drug, placebo, and/or active comparator(s) since the DIBD
(Note: When treatment assignment is blinded, numbers of subjects can be estimated based on
the randomisation scheme.);
• Cumulative number of subjects exposed to the investigational drug from ongoing and
completed clinical trials, subgrouped by age range, sex, and racial group for the development
programme when the data are available;
• Demographic characteristics for a single trial if the trial is of particular importance (e.g., a
pivotal Phase III trial).
The specific categorisation of age might be dependent on the subject population and indication.
This section should also include an explanation of the sponsor’s rationale for selecting the method to
estimate subject exposure, and the limitations of that method, based on the points above.
3.6.2. Patient exposure from marketing experience
If the investigational drug is marketed by the sponsor, the DSUR should include an estimate of the
cumulative patient exposure in the marketed setting, based on the information provided in the most
recent PSUR or other suitable data source, with an explanation of the method(s) used to determine the
estimate.
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 14/35

3.7. Data in line listings and summary tabulations
Sections 7.1-7.3 of the DSUR should present important clinical safety information through:
• Interval line listings of the SARs that were reported to the sponsor during the period covered
by the DSUR; and
• Cumulative summary tabulations of serious adverse events that have been reported to the
sponsor since the DIBD.
Although causality assessment is generally useful for the evaluation of individual rare adverse drug
reactions (ADRs) and for making decisions regarding expedited reporting, individual case causality
assessment has less value in the analysis of aggregate data, where group comparisons of rates are
possible. Therefore, the summary tabulations in a DSUR should include all SAEs and not just SARs for
the investigational drug and comparators.
The line listings and tabulations should include blinded and unblinded clinical trial data. Unblinded data
might originate from completed trials and individual cases that have been unblinded for safety-related
reasons (e.g., expedited reporting), if applicable. Sponsors should not unblind data for the specific
purpose of preparing the DSUR.
At the sponsor’s discretion, graphical displays can be used to illustrate specific aspects of the data
when useful to enhance understanding.
If the Medical Dictionary for Regulatory Activities (MedDRA) terminology is used for coding the adverse
event/reaction terms, the Preferred Term level should be presented in the line listings and summary
tabulations.
In general, the tabulation(s) of SAEs should include only those terms that were used in defining the
case as serious; they should not include non-serious events.
Certain adverse events can be excluded from the line listings and summary tabulations, but such
exclusions should be explained in the report. For example, adverse events that have been defined in
the protocol as “exempt” from special collection and entry into the safety database, and those that are
integral to efficacy endpoints, can be excluded (e.g., deaths reported in a trial of a drug for congestive
heart failure where all-cause mortality is the primary efficacy endpoint, disease progression in cancer
trials).
3.7.1. Reference information
This section of the DSUR should specify the version(s) of the coding dictionary used. If applicable, it
should also specify the document and version used as Reference Safety Information for determining
expectedness for the tabulations, where required by national or regional laws or regulations.
3.7.2. Line listings of serious adverse reactions during the reporting period
This section of the DSUR should summarise how case reports were selected for inclusion in the line
listings. This section should not serve to provide analyses or conclusions based on the SARs. The line
listings should be provided in an appendix (see Appendix B, Table 5 of this guideline).
The line listings should provide key information on all SARs (blinded and unblinded) reported from the
sponsor’s clinical trials during the reporting period. The data should be organised by trial and then by
System Organ Class (SOC).
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 15/35

Where possible the line listing(s) should include each subject only once regardless of how many SAR
terms are reported for the case. If there is more than one reaction, they should all be mentioned but
the case should be listed under the most serious adverse reaction (sign, symptom or diagnosis), as
judged by the sponsor. It is possible that the same subject could experience different SARs on different
occasions (e.g., weeks apart during a clinical trial). Under such circumstances, the SARs can be listed
separately, and a single subject can be included in a line listing more than once.
The following information should be included in the line listings:
a) Study identification number and EudraCT number
11
as applicable;
b) Subject clinical trial identification number;
c) Sponsor’s adverse reaction case reference number;
d) Country in which case occurred;
e) Age and sex of trial subject;
f) Treatment group; identified as “blinded” if the blind has not been broken;
g) Dose and dosing interval of investigational drug (and, when relevant, dosage form and route of
administration);
h) Date of onset and/or time to onset of the most serious adverse reaction;
i) Dates of treatment and/or best estimate of treatment duration;
j) Serious adverse reaction(s); when MedDRA is used, the Preferred Term should be presented;
k) Outcome (e.g., resolved, fatal, improved, sequelae, unknown). This field should indicate the
consequences of the reaction(s) for the patient, using the worst of the different outcomes for
multiple reactions.
l) Comments, if relevant (e.g., causality assessment if the sponsor disagrees with the reporter;
concomitant medications suspected to play a role in the reactions directly or by interaction;
indication treated with suspect drug(s); dechallenge/rechallenge results if available).
Appendix B, Table 5 of this guideline provides an example of the headings for a line listing.
3.7.3. Cumulative summary tabulations of serious adverse events
This section should refer to an appendix that provides a cumulative summary tabulation of SAEs
reported in the sponsor’s clinical trials, from the DIBD to the data lock point of the current DSUR. The
sponsor should explain any omission of data (e.g., clinical trial data might not be available for products
marketed for many years or for products acquired through a business merger). The tabulation(s)
should be organised by SOC, for the investigational drug, as well as for the comparator arm(s) (active
comparators, placebo, and treatment unknown due to blinding) used in the programme. Data can be
integrated across the programme. Alternatively, when useful and feasible, tabulations of SAEs can be
presented by protocol, indication, route of administration, or other variables.
This section should not serve to provide analyses or conclusions based on the SAEs.
Appendix B, Table 6 of this guideline provides an example.
11
The EudraCT number is the unique identifier for trials authorised in the European Economic Area.
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 16/35

3.8. Significant findings from clinical trials during the reporting period
The information in this section can be provided by indication, when appropriate, and should address
the following topics, when applicable:
3.8.1. Completed clinical trials
This section of the DSUR should provide a brief summary of clinically important emerging efficacy and
safety findings obtained from clinical trials completed during the reporting period. This information can
be presented in narrative format or as a synopsis.
12
It could include information that supports or
refutes previously identified safety issues, as well as evidence of new safety signals.*
3.8.2. Ongoing clinical trials
If the sponsor is aware of clinically important information that has arisen from ongoing clinical trials
(e.g., learned through interim safety analyses or as a result of unblinding of subjects with adverse
events), this section should briefly summarise the issue(s). It could include information that supports
or refutes previously identified safety issues, as well as evidence of new safety signals.
3.8.3. Long-term follow-up
Where applicable, this section should provide information from long-term follow-up of subjects from
clinical trials of investigational drugs, particularly advanced therapy products (e.g., gene therapy, cell
therapy products and tissue engineered products). When the development programme is completed
and long-term follow-up is the only ongoing activity generating data for the DSUR, this could be the
only section where new information is presented.
3.8.4. Other therapeutic use of investigational drug
This section of the DSUR should include clinically important safety information from other programmes
conducted by the sponsor that follow a specific protocol, with solicited reporting as per ICH E2D (e.g.,
expanded access programmes, compassionate use programmes, particular patient use, single patient
INDs and treatment INDs).
3.8.5. New safety data related to combination therapies
If the DSUR is for an investigational drug that is also under development as a component of a fixed
combination product or a multi-drug regimen, this section should summarise important safety findings
from the combination therapy DSUR.
Conversely, if this DSUR is for a multi-drug therapy or fixed combination product, this section should
summarise important safety information arising from trials on the individual components.
Alternatively, the information specific to the combination can be incorporated into a separate
section(s) of the DSUR for one or all of the individual components of the combination.
General Principles, section 2.5, provides additional guidance on preparation of DSURs for combination
products.
12
Examples of synopses are provided in ICH E3 and CIOMS VII
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 17/35

3.9. Safety findings from non-interventional studies
This section should summarise relevant safety information from non-interventional studies* that
became available to the sponsor during the reporting period (e.g., observational studies,
epidemiological studies, registries* and active surveillance programmes).
3.10. Other clinical trial/study safety information
This section should summarise relevant safety information from any other clinical trial/study sources
that became available to the sponsor during the reporting period (e.g., results from pooled analyses or
meta-analyses of randomised clinical trials, safety information provided by co-development partners or
from investigator-initiated trials).
3.11. Safety findings from marketing experience
If the investigational drug has been approved for marketing in any country, this section should include
a concise summary of key safety findings that have arisen from marketing experience and that became
available to the sponsor during the reporting period, particularly if the findings resulted in changes to
the product labelling, Investigator’s Brochure, informed consent document or amendments to the
product’s risk management plan. This includes not only safety findings relating to approved use but
also off-label use, administration to special populations (e.g., pregnant women), medication errors,
overdose and abuse.
3.12. Non-clinical data
This section should summarise major safety findings from non-clinical in vivo and in vitro studies (e.g.,
carcinogenicity, reproduction, or immunotoxicity studies) ongoing or completed during the reporting
period. Implications of these findings should be discussed in the Overall Safety Assessment (see
section 3.18 of this Guideline).
3.13. Literature
This section should summarise new and significant safety findings, either published in the scientific
literature or available as unpublished manuscripts, relevant to the investigational drug that the sponsor
became aware of during the reporting period. This section should include information from non-clinical
and clinical studies and, if relevant and applicable, information on drugs of the same class. It should
also summarise significant new safety information presented at a scientific meeting and published as
an abstract; the sponsor should provide a copy of the abstract, if possible.
3.14. Other DSURs
A sponsor should prepare a single DSUR for a single investigational drug. However, if a sponsor
prepares multiple DSURs for a single investigational drug (e.g., covering different indications,
development programmes, or formulations), this section should summarise significant findings from
the other DSURs if they are not presented elsewhere within this report.
When available, the sponsor should summarise significant findings from DSURs provided by other
sponsors conducting clinical trials with the same investigational drug during the reporting period.
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 18/35

3.15. Lack of efficacy
Data indicating lack of efficacy, or lack of efficacy relative to established therapy(ies), for
investigational drugs intended to treat serious or life-threatening illnesses (e.g., excess cardiovascular
adverse events in a trial of a new anti-platelet drug for acute coronary syndromes) could reflect a
significant risk to clinical trial subjects and should be summarised in this section.
3.16. Region-specific information
The information in this section can be used to comply with national or regional requirements and can
be provided in appendices to the DSUR. Sponsors should refer to national or regional requirements to
determine which of the following sections should be included, as well as the scope of clinical trials that
should be covered by these sections. Examples include:
• Cumulative summary tabulation of serious adverse reactions
This cumulative summary tabulation of all SARs should specify the number of SARs by: a) SOC, b)
adverse reaction term and c) treatment arm, if applicable. Unexpected adverse reaction terms should
be identified.
• List of subjects who died during the reporting period
The list of subjects who died during participation in the clinical trials should include the following
information at a minimum: case number, assigned treatment (could still be blinded), and cause of
death of each subject. Any safety issues identified from a review of these deaths should be addressed
in section 18 of the DSUR as appropriate.
• List of subjects who dropped out of clinical trials in association with an adverse event during
the reporting period
This list should include all subjects who dropped out of clinical trials in association with adverse events
during the reporting period, whether or not thought to be drug-related. Any safety issues identified
from a review of these withdrawals should be addressed in section 18 of the DSUR as appropriate.
• Significant Phase I protocol modifications
This section should describe significant Phase I protocol modifications made during the reporting
period, if not previously submitted as a protocol amendment, as described in the US Code of Federal
Regulations.
• Significant manufacturing changes
This section should include a summary of significant manufacturing or microbiological changes during
the reporting period and discuss potential safety issues arising from these changes in Section 18 of the
DSUR, if applicable.
13
• Description of the general investigation plan for the coming year
This section should outline an investigational plan to replace that submitted for the previous year. US
IND holders should refer to the US Code of Federal Regulations.
14
• Log of outstanding business with respect to the US IND
13
In addition US IND holders should refer to: FDA Guidance for Industry: INDs for Phase 2 and Phase 3 Studies –
Chemistry, Manufacturing and Controls Information, May 2003.
http://www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/ucm070567.pdf
14
US Code of Federal Regulations 21 CFR 312.23(a)(3)(iv); revised April 2009. http://frwebgate.access.gpo.gov/cgi-bin/get-
cfr.cgi
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 19/35

If desired by the sponsor, this section can provide a log of any outstanding business with respect to
the US IND for which the sponsor requests or expects a reply, comment or meeting.
3.17. Late-breaking information
This section should summarise information on potentially important safety findings that arise after the
data lock point but while the DSUR is in preparation. Examples include clinically significant new case
reports, important follow-up data, clinically relevant toxicological findings and any action that the
sponsor, a DMC, or a regulatory authority has taken for safety reasons. The Overall Safety Assessment
(see section 3.18) should also take these new data into account.
3.18. Overall safety assessment
The overall safety assessment should be a concise, integrated evaluation of all new relevant clinical,
non-clinical, and epidemiologic information obtained during the reporting period relative to previous
knowledge of the investigational drug. This assessment should consider cumulative experience, new
information collected in the period covered by the DSUR and, for investigational drugs with a
marketing approval, clinically significant post-marketing data. It should not summarise or repeat
information presented in previous sections of the DSUR, but should provide an interpretation of the
information and its implications for the clinical trial population and the development programme. If
appropriate, separate assessments can be provided by therapeutic area, route of administration,
formulation and/or indication.
3.18.1. Evaluation of the risks
In evaluating the risks, particular emphasis should be placed on interpretation of data related to newly
identified safety concerns or providing significant new information relative to previously identified
safety concerns. Relevant points to consider include (where applicable):
• newly identified safety issues (detailed description of adverse events or reactions; associated
laboratory values; risk factors; relationship to dose, duration, time course of the treatment;
reversibility; factors that could be useful in predicting or preventing reactions);
• meaningful changes in previously identified adverse reactions (e.g., increased frequency or
severity, outcome, specific at-risk populations);
• symptoms, signs, and laboratory evidence of newly and previously identified clinically
significant toxicities, for example:
o hepatotoxicity;
o cardiovascular effects, including QT interval prolongation and results from thorough
QT/QTc studies;
o bone marrow toxicity;
o pulmonary toxicity;
o renal toxicity;
o central nervous system toxicity;
o immunogenicity and hypersensitivity;
• deaths that are an outcome of an adverse event;
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 20/35

• study drug discontinuations because of adverse events, including abnormal laboratory values
or investigations;
• drug–drug and other interactions;
• important non-clinical safety findings;
• manufacturing issues that could affect risk;
• lack of efficacy where this would place trial participants at risk;
• any specific safety issues related to special populations, such as the elderly, children, patients
with hepatic or renal impairment, or any other at-risk groups (e.g., slow or fast metabolisers);
• pregnancy and lactation exposure and outcomes;
• safety findings arising from experience with long-term treatment;
• evidence of clinically significant medication errors;
• evidence of lack of patient compliance;
• experience with overdose and its treatment;
• occurrences of drug misuse and abuse;
• any safety issues resulting from procedures required by the protocol (e.g., bronchoscopy,
biopsy, central line insertion) or associated with the conduct or design of a particular study
(e.g., inadequate subject monitoring schedule, excessive period without active treatment); and
• potential impact of significant new safety issues identified with another drug in the same class.
3.18.2. Benefit-risk considerations
This section should provide a succinct statement on the perceived balance between risks that have
been identified from cumulative safety data and anticipated efficacy/benefits* and should note whether
there have been any changes in this balance since the previous DSUR. This section is not intended to
be a full benefit-risk assessment of the investigational drug.
3.19. Summary of important risks
This section should provide a concise, cumulative, issue-by-issue list of important identified and
potential risks*, e.g., those that might lead to warnings, precautions, or contraindications in labelling.
Such risks might include, for example, toxicities known to be associated with a particular molecular
structure or drug class, or concerns based on accumulating non-clinical or clinical data. Each risk
should be re-evaluated annually and re-summarised as appropriate, based on the current state of
knowledge. New information should be highlighted. The appropriate level of detail is likely to be
dependent on the stage of drug development. For example, summaries covering drugs in early
development might include information on individual cases, whereas in later development, as more
knowledge and perspective are gained, the information on each risk might be less detailed.
The information in this section could provide the basis for the safety specification of a risk
management plan (ICH E2E).
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 21/35

Risks that have been fully addressed or resolved should remain in the summary and be briefly
described, e.g., findings from toxicology studies or early clinical trials that were not borne out by later
clinical data.
The information can be provided in either narrative or tabular format (see examples of both in
Appendix C of this guideline).
3.20. Conclusions
The conclusion should briefly describe any changes to the previous knowledge of efficacy and safety
resulting from information gained since the last DSUR. The conclusion should outline actions that have
been or will be taken to address emerging safety issues in the clinical development programme.
Appendices to the DSUR
The DSUR should be accompanied by the following appendices, as appropriate, numbered as follows:
1. Investigator’s Brochure (if required by national or regional laws or requirements);
2. Cumulative Table of Important Regulatory Requests;
3. Status of Ongoing and Completed Clinical Trials;
4. Cumulative Summary Tabulations of Demographic Data;
5. Line Listings of Serious Adverse Reactions;
6. Cumulative Summary Tabulation of Serious Adverse Events;
7. Scientific abstracts (if relevant).
The DSUR should also be accompanied by the following Regional Appendices, as appropriate (see
section 3.16):
o Cumulative summary tabulation of serious adverse reactions;
o List of subjects who died during the reporting period;
o List of subjects who dropped out of studies during the reporting period;
o Significant Phase I protocol modifications with respect to a US IND;
o Significant manufacturing changes;
o Description of the general investigation plan for the coming year with respect to a US IND;
o Log of outstanding business with respect to a US IND.
4. Appendices to this guideline
Appendix A Glossary
Appendix B Examples of Tables and Table Headings for Clinical Trial Data
Appendix C Examples of the Summary of Important Risks
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 22/35
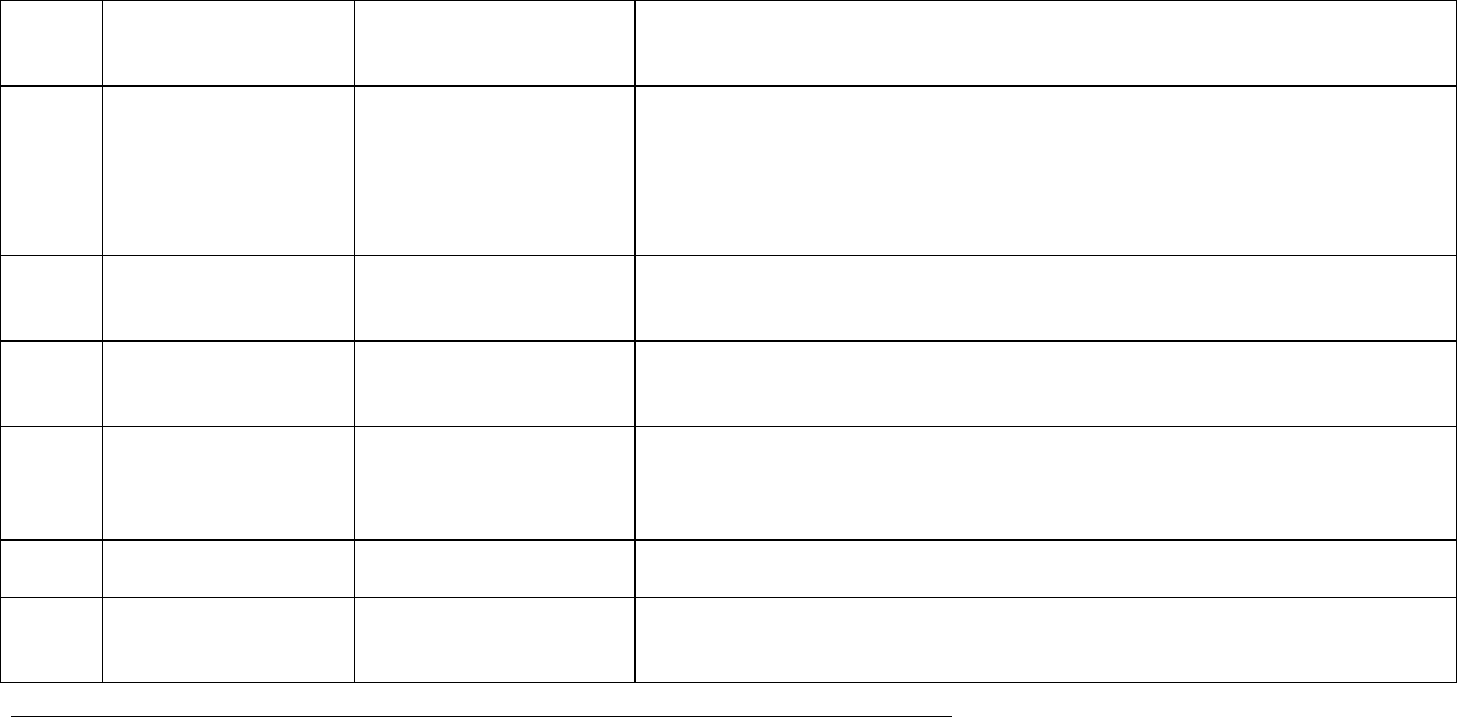
APPENDIX A - Glossary
Throughout this guideline the Working Group has used terms previously defined by ICH and other groups e.g., CIOMS. Generally, the definitions of terms
that were previously defined in ICH documents are not repeated in this glossary. However, the glossary includes several ICH terms of particular importance
to the DSUR, as well as terms defined by CIOMS and other groups.
Item Glossary Term Source of
Definition
Definition/Commentary
Adverse event of special
interest
Based on CIOMS VI An adverse event of special interest (serious or non-serious) is one of scientific and
medical concern specific to the sponsor’s product or programme, for which ongoing
monitoring and rapid communication by the investigator to the sponsor can be
appropriate. Such an event might warrant further investigation in order to characterise
and understand it. Depending on the nature of the event, rapid communication by the
trial sponsor to other parties (e.g., regulators) might also be warranted.
Anticipated
efficacy/benefit
Based on wording of CIOMS
VI definition of anticipated
risk
Efficacy/benefit that has not yet been established for the investigational drug, but
which is anticipated based on knowledge of the class of drugs or data from previous
clinical trials or non-clinical studies.
Clinical development
programme
ICH E2F This refers to all clinical trials being conducted with the same investigational drug,
regardless of indication or formulation.
Completed clinical trial CIOMS VII Study for which a final clinical study report is available. Note: For purposes of the
DSUR, any clinical trial for which enrolment has begun, but for which a final clinical
study report is not available, is considered to be ongoing (see “ongoing clinical trial”
definition).
Data lock point CIOMS VII The date (month and day) designated as the cut-off for data to be included in a DSUR.
It is based on the Development International Birth Date (DIBD).
Data Monitoring
Committee (synonyms:
Independent Data
ICH E6 An independent data monitoring committee that may be established by the sponsor to
assess at intervals the progress of a clinical trial, the safety data, and the critical
efficacy endpoints, and to recommend to the sponsor whether to continue, modify, or
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 23/35

Item Glossary Term Source of
Definition
Definition/Commentary
Monitoring Committee,
Data and Safety
Monitoring Board,
stop a trial.
Development
International Birth Date
CIOMS VII Date of first approval (or authorisation) for conducting an interventional clinical trial in
any country.
Identified risk Volume 9A Rules Governing
Medicinal Products in the EU
An untoward occurrence for which there is adequate evidence of an association with the
medicinal product of interest.
Examples of identified risks include:
an adverse reaction adequately demonstrated in non-clinical studies and confirmed by
clinical data;
an adverse reaction observed in well-designed clinical trials or epidemiological studies
for which the magnitude of the difference compared with the comparator group
(placebo or active substance) on a parameter of interest suggests a causal relationship;
an adverse reaction suggested by a number of well-documented spontaneous reports
where causality is strongly supported by temporal relationship and biological
plausibility, such as anaphylactic reactions or application site reactions.
Important identified risk;
important potential risk
Volume 9A Rules Governing
Medicinal Products in the EU
An identified risk or potential risk that could have an impact on the risk-benefit balance
of the product or have implications for public health.
Interventional clinical
trial
CIOMS VII
An interventional clinical trial is any research study that prospectively assigns people to
one or more health-related interventions (e.g., preventive care, drugs, surgical
procedures, behavioural treatments, etc.) to evaluate their effects on health-related
outcomes.
Investigational drug
CIOMS VII The term investigational drug is used in this guideline to indicate only the experimental
product under study or development. Note: This term is more specific than
“investigational medicinal product” which includes comparators and placebos.
Non-interventional
clinical study
EU Directive 2001/20/EC on
Clinical Trials
A study where the medicinal product(s) is (are) prescribed in the usual manner in
accordance with the terms of the marketing authorisation. The assignment of the
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 24/35
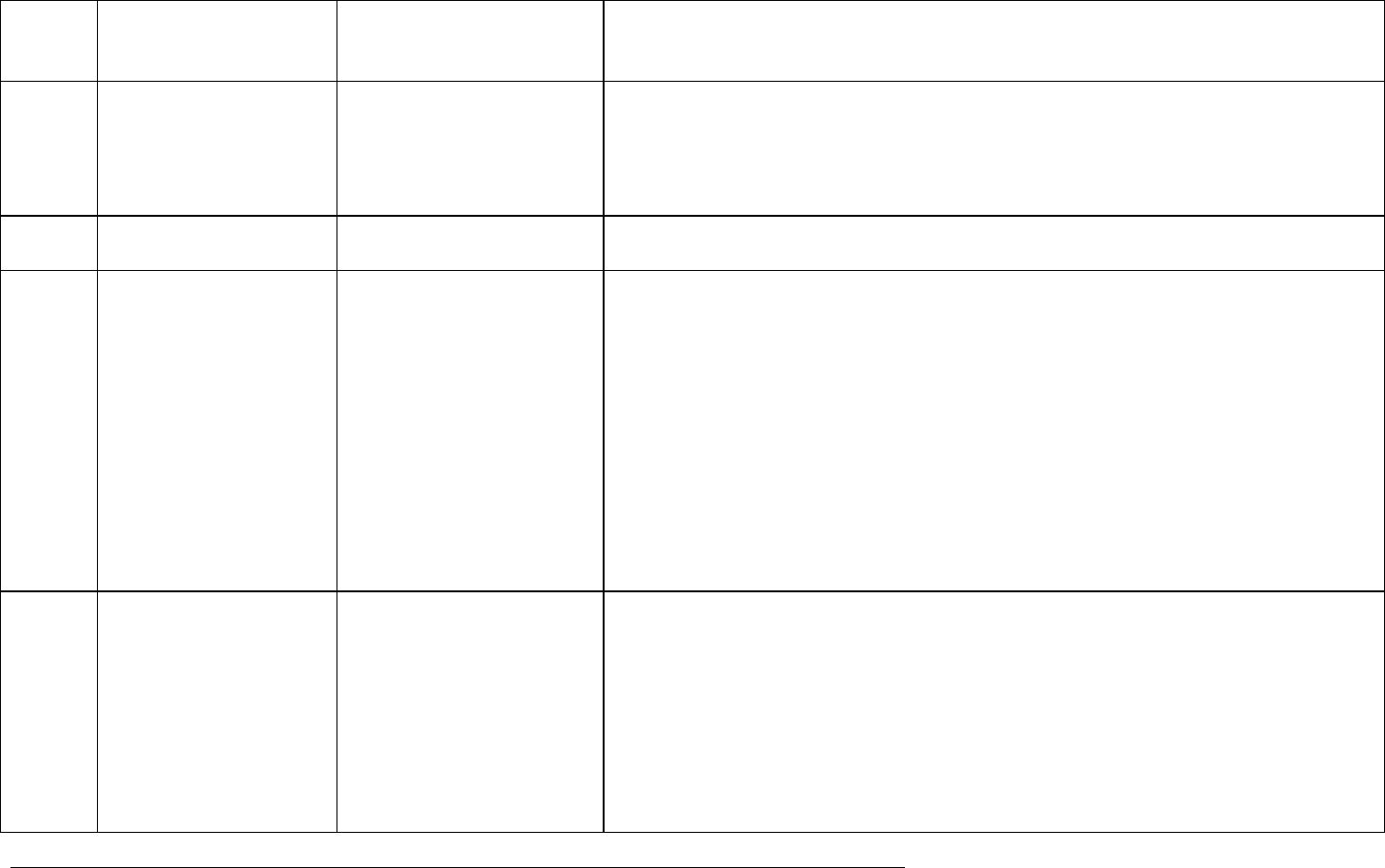
Item Glossary Term Source of
Definition
Definition/Commentary
patient to a particular therapeutic strategy is not decided in advance by a trial protocol
but falls within current practice and the prescription of the medicine is clearly separated
from the decision to include the patient in the study. No additional diagnostic or
monitoring procedures are applied to the patients and epidemiological methods are
used for the analysis of collected data.
Ongoing clinical trial CIOMS VII Trial where enrolment has begun, whether a hold is in place or analysis is complete,
but without a final clinical study report available.
Potential risk Volume 9A Rules Governing
Medicinal Products in the EU
An untoward occurrence for which there is some basis for suspicion of an association
with the medicinal product of interest but where this association has not been
confirmed.
Examples of potential risks include:
Non-clinical safety concerns that have not been observed or resolved in clinical studies;
Adverse events observed in clinical trials or epidemiological studies for which the
magnitude of the difference, compared with the comparator group (placebo or active
substance, or unexposed group), on the parameter of interest raises a suspicion of, but
is not large enough to suggest, a causal relationship;
A signal arising from a spontaneous adverse reaction reporting system;
An event which is known to be associated with other products of the same class or
which could be expected to occur based on the properties of the medicinal product.
Registry ICH E2E A registry is a list of patients presenting with the same characteristic(s). This
characteristic can be a disease (disease registry) or a specific exposure (drug registry).
Both types of registries, which only differ by the type of patient data of interest, can
collect a battery of information using standardised questionnaires in a prospective
fashion. Commentary: Exposure (drug) registries collect information over time on
populations exposed to drugs of interest and/or specific populations. Patients can be
included in a cohort study to collect data on adverse events using standardised
questionnaires. They can be useful for signal amplification, particularly of rare
outcomes.
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 25/35

Item Glossary Term Source of
Definition
Definition/Commentary
Signal CIOMS VI
A report or reports of an event with an unknown causal relationship to treatment that is
recognised as worthy of further exploration and continued surveillance.
Sponsor ICH E6 (R1) An individual, company, institution, or organisation which takes responsibility for the
initiation, management, and/or financing of a clinical trial.
Sponsor-investigator ICH E6 An individual who both initiates and conducts, alone or with others, a clinical trial, and
under whose immediate direction the investigational product is administered to,
dispensed to, or used by a subject. The term does not include any person other than an
individual (e.g., it does not include a corporation or an agency). The obligations of a
sponsor-investigator include both those of a sponsor and those of an investigator.
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 26/35
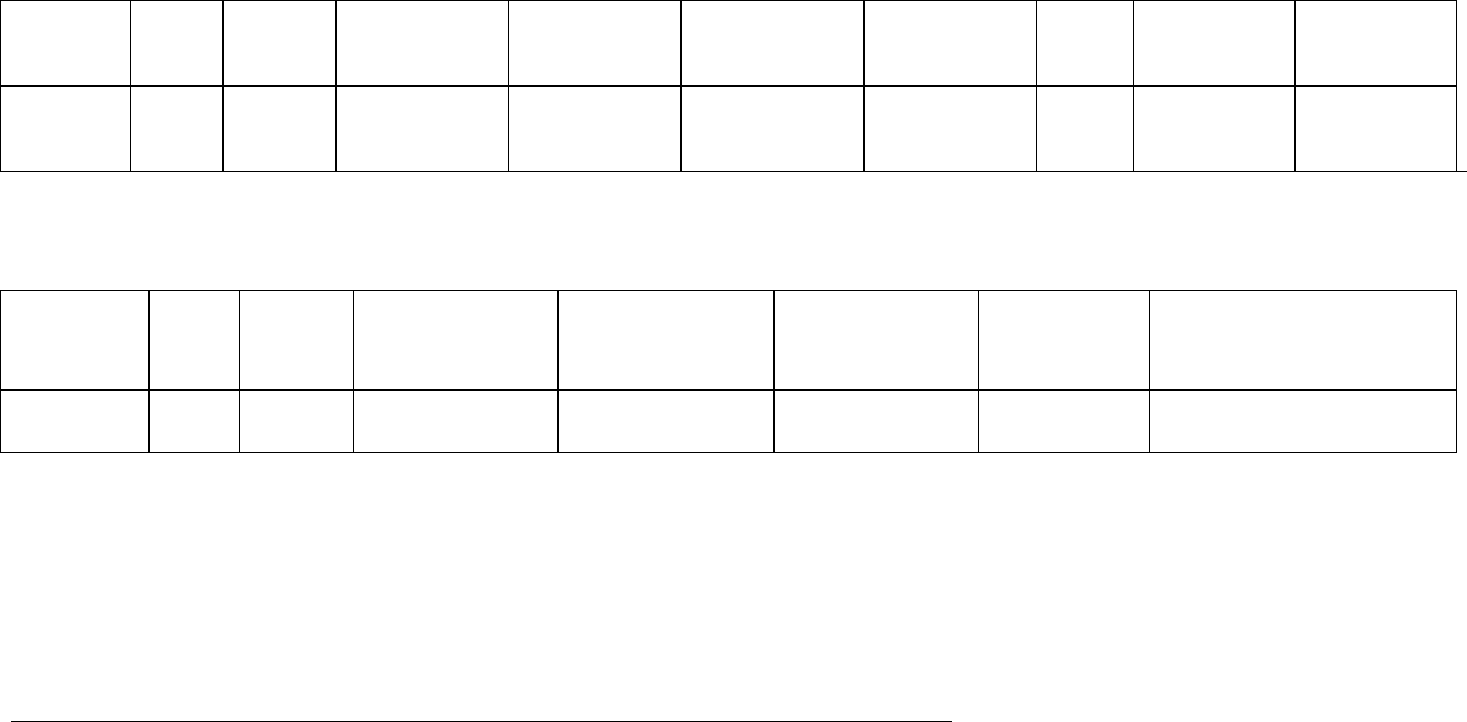
APPENDIX B ─ Examples of tables and table headings for clinical trial listings
Table 1. Status of ongoing and completed clinical trials
Overview of ongoing Studies [Study Drug]
Study ID
Phase Country Study Title Study design
Dosing regimen
Study
population
FVFP†
Planned
enrolment
Subject
exposure‡
† FVFP = first visit first patient
‡ Based upon total number of patients recruited as of [date] and applied randomisation schemes
Overview of Studies Completed During the DSUR Period [Study Drug]
Study ID
Phase Country Study Title Study design Dosing regimen
Study
population
Subject/patient patient
exposure per treatment arm
(M/F)
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 27/35

Table 2. Table 2 - Estimated Cumulative Subject Exposure
Estimates of cumulative subject exposure, based upon actual exposure data from completed clinical trials and the enrolment/randomisation schemes for
ongoing trials.
Treatment Number of subjects
drug
comparator
placebo
Table 3. Table 3 - Cumulative Subject Exposure to Investigational Drug from Completed Clinical Trials by Age and Sex*
Number of subjects
Age range
Male
Female
Total
* Data from completed trials as of [date]
Table 4. Table 4 – Cumulative Subject Exposure to Investigational Drug from Completed Clinical Trials by Racial Group*
Racial group Number of subjects
Asian
Black
Caucasian
Other
Unknown
Total
* Data from completed studies as of [date]
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 28/35
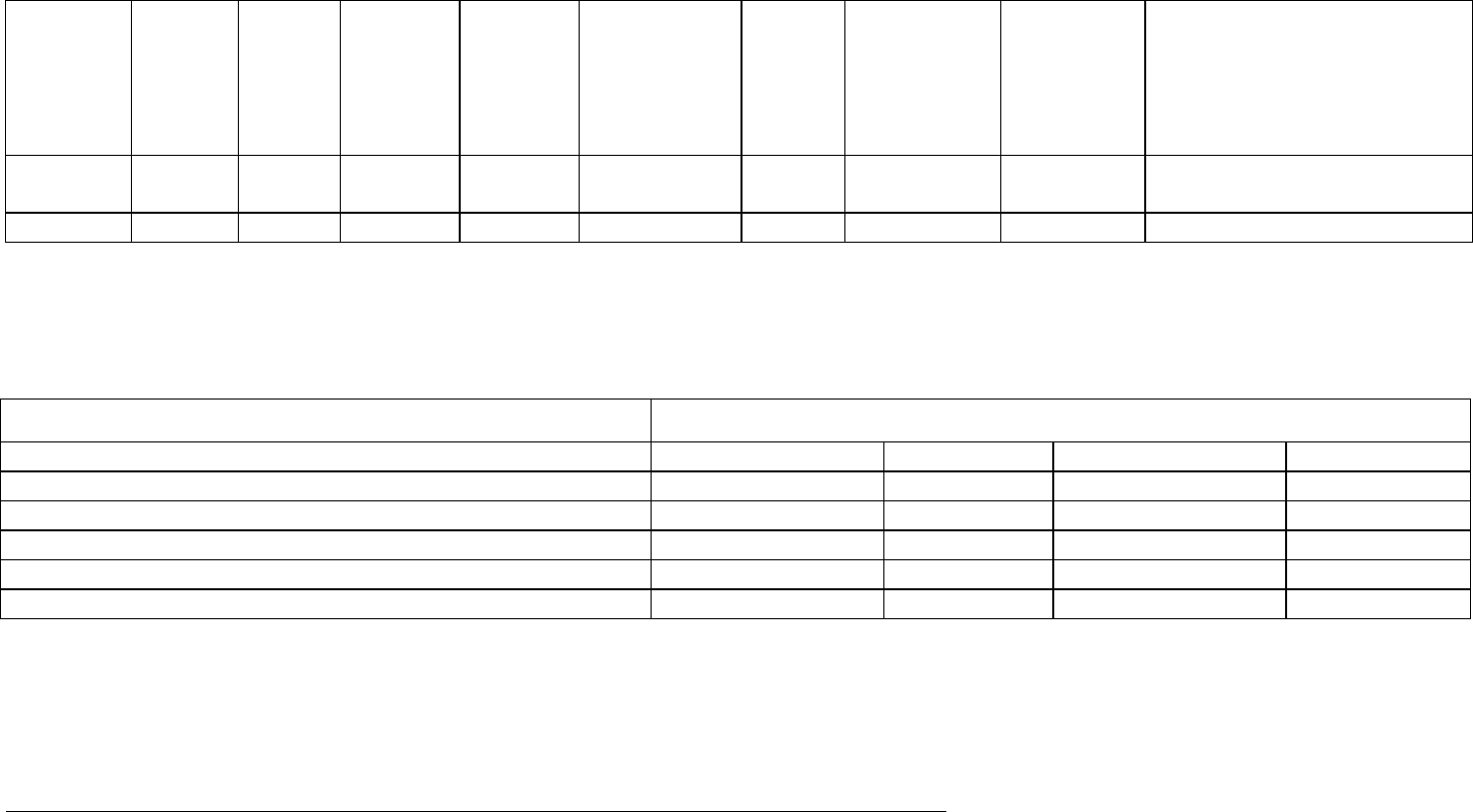
Table 5. Examples of Headings for Interval Line Listings of Serious Adverse Reactions
Interval Line Listings of Serious Adverse Reactions
† Study/centre/patient
‡ ‘Primary’ SAR only
Table 6. Examples of Cumulative Tabulations of Serious Adverse Events
Cumulative Summary Tabulation of Serious Adverse Events (SAEs)
Study ID
EudraCT
number
Case ID/
Subject
number†
Country
Gender
Age
Serious
adverse
drug
reactions
(SARs)
Outcome Date of onset‡
Time to onset‡
Suspect
Drug
Daily dose
Route
Formulation
Dates of
treatment
Treatment
duration
Comments
--------
--------
-------
--------
--------
--------
--------
--------
--------
System Organ Class Total up to 31-Dec-09
Preferred Term
[Study drug]
Blinded
Active comparator
Placebo
Investigations 18 4 7 2
Alanine aminotransferase increased
9
2
4
1
Aspartate aminotransferase increased
9
2
3
1
Nervous System Disorder
2
2
4
7
Syncope 2 2 4 7
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 29/35

APPENDIX C— Examples of the summary of important risks
This appendix depicts fictitious examples of the Summary of Important Risks, prepared in three 1
consecutive years, from 2012 to 2014. The Summary of Important Risks can be provided in either 2
narrative (Appendix C1) or tabular (Appendix C2) format. 3
APPENDIX C1: Narrative Format 4
DSUR 2012 5
19 Summary of Important Risks 6
New or updated risks are denoted with an asterisk. 7
1. Nephrotoxicity 8
Drug Z is a para-aminoglycolate that bears structural similarities to aminoglycosides, currently under 9
development for amelioration of angina pectoris in patients with stable coronary artery disease. Other 10
members of this class are known to be nephrotoxic, and there was evidence of nephrotoxicity in both 11
rats and rabbits at doses of 20 and 60 mg/kg/d, respectively. 12
In Phase I trials in healthy volunteers, 2 of 30 subjects (6.7%) who received the highest dose of drug 13
Z (100 mg po qd for 7 days) exhibited transient increases in serum creatinine associated with 14
proteinuria: subject 0127 had an increase in creatinine from 0.9 mg/dL at baseline to 1.8 mg/dL at 15
Day 7; subject 0139 had an increase from 1.0 mg/dL at baseline to 1.9 mg/dL at Day 7. Both subjects 16
had mild proteinuria (2+ by dipstick, 24-hour urinary protein not quantified). Urinalyses of both 17
subjects were unremarkable (minimal cells; no casts). By Day 21, serum creatinine had returned to 18
baseline in both subjects, and proteinuria had resolved (see sections 8.2 and 18.1 for details). None of 19
the other 28 healthy subjects who received drug Z 100 mg qd, and none of the 119 subjects who 20
received drug Z at lower doses (50 mg or less, including 72 subjects with coronary artery disease), 21
experienced proteinuria or significant increases in creatinine. 22
The increases in creatinine in the healthy volunteers who received the highest dose of drug Z (100 mg 23
QD) were thought likely to be drug-related, in part because of the known nephrotoxicity of the drug 24
class. It was decided, therefore, to reduce the maximum dose of drug Z in the Phase II trials to 50 mg. 25
In addition, subject monitoring was intensified: serum creatinine, eGFR, albumin/creatinine ratios, 26
blood urea nitrogen, and urinalysis are now performed at baseline, Weeks 1, 2, 4, 8, 16, and 24. 27
Twenty-four-hour urinary protein excretion will be determined for any subject who develops proteinuria 28
by dipstick. Study drug will be discontinued in subjects with creatinine increases of 0.5 mg/dL, a 30% 29
rise in creatinine, or a 25% decrease in eGFR (repeated 2 times over a 3-day period). The protocol, 30
informed consent document, and Investigator’s Brochure have been revised accordingly. 31
2. Hepatotoxicity 32
In rat study KR-102, 2 of 8 rats in the highest dose group (60 mg/kg/d) developed hepatic injury, with 33
centrilobular necrosis. None of the rats that received lower doses had evidence of hepatotoxicity, and 34
no hepatotoxicity was evident in rabbits at doses at high as 60 mg/kg/d. 35
One (1) subject (102-037) with coronary artery disease in study 102 who received 50 mg po qd drug Z 36
developed moderate elevation of alanine aminotransferase (ALT) and aspartate aminotransferase 37
(AST) on Day 14 (2.7 and 2.3 x the upper limits of normal, respectively), without increases in alkaline 38
phosphatase or bilirubin (see sections 8.2 and 18.1 for details). Drug Z was discontinued on Day 16, 39
and transaminases returned to normal by Day 28. The subject denied alcohol consumption, and all 40
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 30/35

serology was negative. The subject had been receiving drugs X and Y concomitantly on a chronic basis 41
for >2 years prior to enrolment. Neither drug was suspected of causing the elevation in transaminases. 42
Both were continued during the adverse event, making it very unlikely that they were the cause of the 43
transaminase elevations. Of note, subject 102-037 was found to have concentrations of drug Z (Cmax) 44
approximately 8-fold higher than the mean of the 50 mg cohort, suggesting an inability to metabolize 45
drug Z. This possibility remains under investigation. An additional 148 subjects were exposed to drug Z 46
in the Phase I program, and none exhibited transaminase elevations. 47
In light of the rat findings and the transaminase elevations in subject 102-037, more frequent 48
monitoring has been implemented for the Phase II study. Assessments at Weeks 4 and 16 have been 49
added, so that subjects now undergo assessment of ALT, AST, bilirubin, and alkaline phosphatase at 50
baseline, Weeks 1, 2, 4, 8, 16, and 24. The protocol, informed consent document, and Investigator’s 51
Brochure have been revised accordingly. 52
DSUR 2013 53
19 Summary of Important Risks 54
New or updated risks are denoted with an asterisk. 55
1. Nephrotoxicity* 56
Drug Z is a para-aminoglycolate, one of a class of drugs bearing structural similarities to 57
aminoglycosides, and known to be nephrotoxic. Two of 30 healthy volunteers (6.7%) in the Phase I 58
program who had received drug Z 100 mg PO qd developed transient increases in serum creatinine 59
associated with mild proteinuria (by dipstick), and the 100 mg dose was dropped from further 60
development. In the completed Phase II trial of drug Z in patients with coronary artery disease and 61
stable angina pectoris (Study 201), increases in creatinine >1.25 but <1.5 times baseline were
62
observed in 5 of 60 subjects (8.3%) in the 50 mg group, 5 of 62 subjects (8.1%) in the 25 mg group, 63
and 3 of 59 subjects (5.1%) in the 10 mg group, versus 6 of 61 subjects (10%) in the placebo group. 64
For all of these subjects, the study drug was continued (per protocol), and serum creatinine returned 65
to baseline within 2 weeks. Increases in creatinine >1.5 times baseline were observed in 1 of 60 66
subjects (1.7%) in the 50 mg group, 0 of 62 subjects in the 25 mg group, and 1 of 59 subjects (1.7%) 67
in the 10 mg group, versus 2 of 61 subjects (3.3%) in the placebo group. The study drug was 68
discontinued in all of these subjects (per protocol), and serum creatinine returned to baseline within 2 69
weeks. As explained in sections 8.2 and 18.1, the “recovery” of creatinine to normal (i.e., the slope of 70
the creatinine vs. time relationship) was the same in subjects who continued and discontinued the 71
study drug, suggesting that this was not a specific drug effect. Of note, 3 of the 4 drug Z-treated 72
subjects who developed creatinine elevations were taking concomitant diuretics. In ongoing studies 73
202 and 204, serum creatinine, eGFR, blood urea nitrogen, and urinalysis continue to be monitored at 74
baseline, Weeks 1, 2, 4, 12, 24, and 48. Twenty-four-hour urinary protein excretion is determined in 75
any subject who develops proteinuria by dipstick. 76
2. Hepatotoxicity* 77
Drug Z caused centrilobular necrosis at the highest dose tested (60 mg/kg/d) in rats (although there 78
was no evidence of liver damage at this dose in rabbits). One (1) of 149 subjects (0.7%) in the Phase I 79
program developed unexplained elevations of ALT and AST approximately 2.5 x the upper limit of 80
normal on Day 14; which resolved on discontinuation of the drug. Two drug Z-treated subjects in the 81
completed Phase II study #201 (2/181, 1.1%) had transaminase elevations (see section 8.2), but 82
these were mild and transient, and one subject in the placebo group (1/61, 1.6%) had more severe 83
elevations. Based on this information, the present monitoring plan seems appropriate, and no changes 84
have been made to the protocol, Investigator’s Brochure, or informed consent document. Of note, one 85
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 31/35

subject with elevated transaminases (102-037) had been thought to have had an unusually high 86
Cmax; however, this was subsequently determined to be a laboratory error. 87
3. Syncope* 88
Drug Z is thought to be a mild nitric oxide (NO)-dependent vasodilator, which could be partly 89
responsible for its anti-anginal effect. During this period there were 2 reports of syncope (2/81, 1.1%) 90
from study #201 that were considered by investigators to be causally-related to drug Z (subjects 201-91
119 and 201-212). The subjects had been receiving 10 and 25 mg of drug Z, respectively. Although 92
both subjects were predisposed to syncope (one was apparently very dehydrated; the other had a long 93
history of syncope), there is mechanistic plausibility and we will continue to focus on syncope as a 94
possible drug-related side effect. No specific changes have been were made to the monitoring plan as 95
a result of these adverse events. 96
DSUR 2014 97
19 Summary of Important Risks 98
New or updated risks are denoted with an asterisk. 99
1. Nephrotoxicity* 100
Drug Z is a para-aminoglycolate, one of a class of drugs bearing structural similarities to 101
aminoglycosides, and known to be nephrotoxic. A 100 mg dose, studied in Phase I, was dropped from 102
further development because of increases in creatinine and proteinuria in normal volunteers. In the 103
Phase II program, increases in creatinine >1.25 but <1.5 times baseline were observed in 7.8%, 104
6.8%, and 5.8% of subjects in the 50, 25, and 10 mg treatment groups, respectively, versus 6.3% in 105
the placebo group. Increases in creatinine >1.5 times baseline were observed in 1.5%, 0.5%, and 106
1.9% of subjects in the 50, 25, and 10 mg treatment groups, respectively, versus 2.7% in the placebo 107
group. As noted in sections 8.2 and 18.1, increases in creatinine seem to be associated with 108
dehydration and diuretic use. In addition, a number of the subjects with increases in creatinine of more 109
than 50% from baseline have had unusually low baseline values (i.e., <0.6 mg/dL). The clinical
110
meaning of this is unclear. 111
In the ongoing Phase III trial (301), serum creatinine, eGFR, blood urea nitrogen, and urinalysis are 112
monitored at baseline, Weeks 1, 4, 12, and 48. Twenty-four-hour urinary protein excretion is 113
determined in any subject who develops 3+ or greater proteinuria by dipstick. 114
2. Hepatotoxicity* 115
Drug Z caused centrilobular necrosis at the highest dose tested in rats. There was frequent monitoring 116
of ALT, AST, alkaline phosphatase, and bilirubin in the Phase I and II trials, and no consistent pattern 117
of laboratory abnormalities has emerged suggestive of liver injury. 118
In the ongoing Phase III study (301), the above laboratory tests of liver injury are monitored at 119
baseline, Weeks 1, 4, 12, and 48. 120
3. Syncope* 121
Drug Z is thought to be a nitric oxide (NO)-dependent vasodilator, which could be partly responsible 122
for its anti-anginal effect. There have been 21 reports of syncope in drug Z-treated subjects thus far in 123
the development program (21/632, 3.3%), versus 3 (1.4%) in placebo. Most of the cases were 124
orthostatic, and/or associated with coadministration of nitrates or vasodilators. In the Phase III 125
program, subjects are advised against concurrent administration of vasodilators (e.g., nitrates, 126
dihydropyridine calcium channel blockers), and given general precautions regarding orthostatic 127
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 32/35

dizziness. The protocol, informed consent document, and Investigator’s Brochure have been revised to 128
include this risk. 129
4. Pancreatitis* 130
Three case reports of pancreatitis have been reported from subjects in the completed Phase II and 131
ongoing Phase III trials (see sections 8.1 and 8.2, respectively). Although there were plausible 132
alternative explanations for each case, evidence of pancreatitis will be carefully sought thorough 133
laboratory monitoring: all subjects enrolled in the Phase III trial (301) undergo screening evaluations 134
of lipase and amylase, with repeat evaluations at Weeks 1 and 4. 135
APPENDIX C2: Tabular Format 136
19 Summary of Important Risks 137
This section summarises the important identified or potential risks that have been recognised during 138
the conduct of the Drug Z clinical development programme. At present, all are considered as potential 139
risks, with none characterised as identified risks associated with the administration of drug Z. 140
The following have been recognised as important potential risks during the reporting period: 141
• Nephrotoxicity 142
• Hepatotoxicity 143
• Syncope 144
• Pancreatitis 145
Additional details are provided in Table X, below: 146
Table X Summary of Important Risks 147
New or updated risks are denoted with an asterisk. 148
Risk Nonclinical data Clinical data Actions
Nephrotoxicity* Nephrotoxicity in rats
and rabbits at doses of
20 and 60 mg/kg/d,
respectively
Drug Z is a para-
aminoglycolate, in drug
class structurally similar to
aminoglycosides.
Nephrotoxicity well known.
Phase I: A 100 mg dose was
dropped from further
development because of
increases in creatinine and
proteinuria in normal
volunteers.
Phase II: Increases in
creatinine >1.25 but <1.5
times baseline were
observed in 7.8%, 6.8%,
and 5.8% of subjects in the
50, 25, and 10 mg
In Phase III trial (301),
serum creatinine, eGFR,
blood urea nitrogen, and
urinalysis monitored at
baseline, Weeks 1, 4, 12,
and 48.
24-hour urinary protein
excretion is determined in
subjects who develop >2+
or proteinuria by dipstick.
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 33/35

Risk Nonclinical data Clinical data Actions
treatment groups,
respectively, versus 6.3% in
the placebo group.
Increases in creatinine >1.5
times baseline were
observed in 1.5%, 0.5%,
and 1.9% of subjects in the
50, 25, and 10 mg
treatment groups,
respectively, versus 2.7% in
the placebo group.
Increases in creatinine seem
to be associated with
dehydration and diuretic
use. In addition, a number
of the subjects with
increases in creatinine of
more than 50% from
baseline have had unusually
low baseline values (i.e.,
<0.6 mg/dL). The clinical
meaning of this is unclear.
See sections 8.2 and 18.1, i
Hepatotoxicity* Rat study KR-102: 2 of
8 rats in the highest
dose group (60
mg/kg/d) developed
centrilobular necrosis.
At lower doses, no rats
had evidence of
hepatotoxicity.
No hepatotoxicity seen
in rabbits at doses < 60
mg/kg/d.
With frequent monitoring of
ALT, AST, alkaline
phosphatase, and bilirubin in
the Phase I and II trials, no
consistent pattern of
laboratory abnormalities
emerged suggestive of liver
injury.
Routine monitoring in
ongoing Phase III study
(301): ALT, AST, alkaline
phosphatase, and bilirubin
monitored at baseline,
Weeks 1, 4, 12, and 48.
Syncope* Published studies: drug
Z is a nitric oxide (NO)-
dependent vasodilator
in canine models.
21 reports of syncope in
drug Z-treated subjects to
date in the development
program (21/632, 3.3%),
versus 3 (1.4%) in placebo.
Most cases orthostatic,
and/or associated with
coadministration of nitrates
or vasodilators.
In Phase III program,
subjects advised against
concurrent administration
of vasodilators; given
precautions regarding
orthostatic dizziness.
Protocol, informed
consent, and
Investigator’s Brochure
revised to include this
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 34/35

Risk Nonclinical data Clinical data Actions
risk.
Pancreatitis* No findings.
3 cases of pancreatitis
reported from subjects in
the completed Phase II and
ongoing Phase III trials.
Causal relationship with
drug Z not determined -
plausible alternative
explanations for each case.
Subjects in the Phase III
trial (301) undergo
screening evaluations of
lipase and amylase, with
repeat evaluations at
Weeks 1 and 4.
ICH guideline E2F on development safety update report
EMA/CHMP/ICH/309348/2008 Page 35/35
