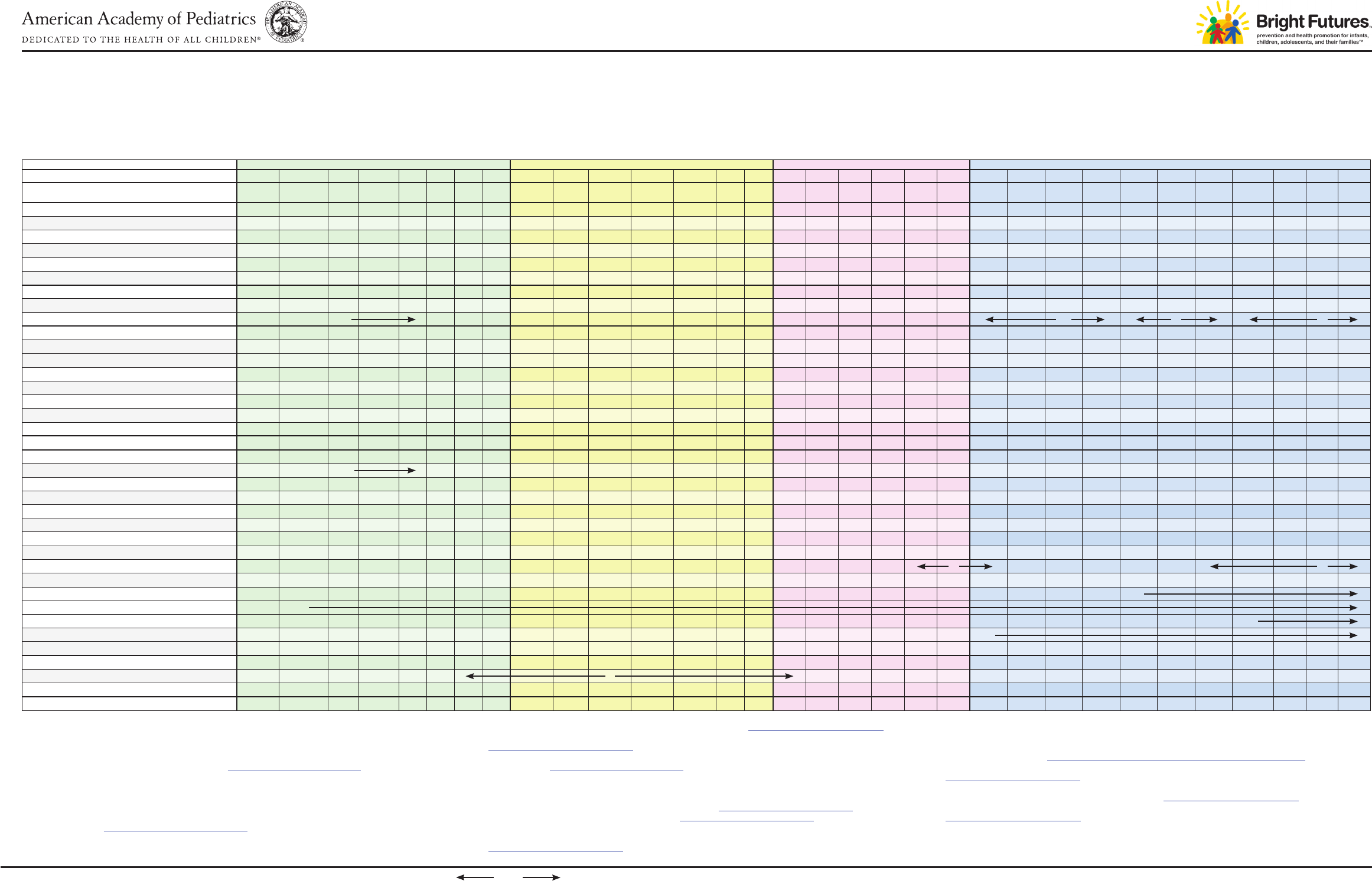
Recommendations for Preventive Pediatric Health Care
Bright Futures/American Academy of Pediatrics
KEY:
l
= to be performed
ê
= risk assessment to be performed with appropriate action to follow, if positive
ê
or
l
= range during which a service may be provided
Each child and family is unique; therefore, these Recommendations for Preventive Pediatric Health Care are designed
for the care of children who are receiving nurturing parenting, have no manifestations of any important health
problems, and are growing and developing in a satisfactory fashion. Developmental, psychosocial, and chronic
disease issues for children and adolescents may require more frequent counseling and treatment visits separate
from preventive care visits. Additional visits also may become necessary if circumstances suggest concerns.
These recommendations represent a consensus by the American Academy of Pediatrics (AAP) and Bright Futures.
The AAP continues to emphasize the great importance of continuity of care in comprehensive health supervision
and the need to avoid fragmentation of care.
Refer to the specic guidance by age as listed in the Bright Futures Guidelines (Hagan JF, Shaw JS, Duncan PM, eds.
Bright Futures: Guidelines for Health Supervision of Infants, Children, and Adolescents. 4th ed. American Academy
of Pediatrics; 2017).
The recommendations in this statement do not indicate an exclusive course of treatment or serve as a standard
of medical care. Variations, taking into account individual circumstances, may be appropriate.
The Bright Futures/American Academy of Pediatrics Recommendations for Preventive Pediatric Health Care are
updated annually.
Copyright © 2024 by the American Academy of Pediatrics, updated June 2024.
No part of this statement may be reproduced in any form or by any means without prior written permission from
the American Academy of Pediatrics except for one copy for personal use.
INFANCY EARLY CHILDHOOD MIDDLE CHILDHOOD ADOLESCENCE
AGE
1
Prenatal
2
Newborn
3
3-5 d
4
By 1 mo 2 mo 4 mo 6 mo 9 mo 12 mo 15 mo 18 mo 24 mo 30 mo 3 y 4 y 5 y 6 y 7 y 8 y 9 y 10 y 11 y 12 y 13 y 14 y 15 y 16 y 17 y 18 y 19 y 20 y 21 y
HISTORY
Initial/Interval
l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l
MEASUREMENTS
Length/Height and Weight
l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l
Head Circumference
l l l l l l l l l l l
Weight for Length
l l l l l l l l l l
Body Mass Index
5
l l l l l l l l l l l l l l l l l l l l l
Blood Pressure
6
ê ê ê ê ê ê ê ê ê ê ê ê
l l l l l l l l l l l l l l l l l l l
SENSORY SCREENING
Vision
7
ê ê ê ê ê ê ê ê ê ê ê ê l l l l ê l ê l
ê
l
ê ê
l
ê ê ê ê ê
ê
Hearing
l
8
l
9
ê ê ê ê ê ê ê ê ê l l l ê l ê l l
10
l l
DEVELOPMENTAL/SOCIAL/BEHAVIORAL/MENTAL HEALTH
Maternal Depression Screening
11
l l l l
Developmental Screening
12
l l l
Autism Spectrum Disorder Screening
13
l l
Developmental Surveillance
l l l l l l l l l l l l l l l l l l l l l l l l l l l l
Behavioral/Social/Emotional Screening
14
l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l
Tobacco, Alcohol, or Drug Use Assessment
15
ê ê ê ê ê ê ê ê ê ê ê
Depression and Suicide Risk Screening
16
l l l l l l l l l l
PHYSICAL EXAMINATION
17
l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l
PROCEDURES
18
Newborn Blood
l
19
l
20
Newborn Bilirubin
21
l
Critical Congenital Heart Defect
22
l
Immunization
23
l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l
Anemia
24
ê
l
ê ê ê ê ê ê ê ê ê ê ê ê ê ê ê ê ê ê ê ê ê ê ê
Lead
25
ê ê l
or
ê
26
ê l
or
ê
26
ê ê ê ê
Tuberculosis
27
ê ê
ê ê ê ê
ê ê ê ê ê ê ê ê ê
ê
ê ê ê ê ê ê ê
Dyslipidemia
28
ê ê
ê ê
l
ê ê ê ê ê
l
Sexually Transmitted Infections
29
ê ê ê ê ê ê ê ê ê ê ê
HIV
30
ê ê ê ê
l
Hepatitis B Virus Infection
31
ê
Hepatitis C Virus Infection
32
l
Sudden Cardiac Arrest/Death
33
ê
Cervical Dysplasia
34
l
ORAL HEALTH
35
l
36
l
36
ê ê ê ê
ê
ê
ê ê
Fluoride Varnish
37
l
Fluoride Supplementation
38
ê ê ê ê ê ê ê ê ê ê ê ê ê ê ê ê ê ê ê ê
ANTICIPATORY GUIDANCE
l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l
BFNC.2024.PSMAR
3-367/0324
continued)
1. If a child comes under care for the rst time at any point on the schedule, or if any items are not accomplished at the suggested
age, the schedule should be brought up to date at the earliest possible time.
2. A prenatal visit is recommended for parents who are at high risk, for rst-time parents, and for those who request a conference.
The prenatal visit should include anticipatory guidance, pertinent medical history, and a discussion of benets of breastfeeding
and planned method of feeding, per “The Prenatal Visit” (https://doi.org/10.1542/peds.2018-1218).
3. Newborns should have an evaluation after birth, and breastfeeding should be encouraged (and instruction and support
should be oered).
4. Newborns should have an evaluation within 3 to 5 days of birth and within 48 to 72 hours after discharge from the hospital
to include evaluation for feeding and jaundice. Breastfeeding newborns should receive formal breastfeeding evaluation, and
their mothers should receive encouragement and instruction, as recommended in “Policy Statement: Breastfeeding and the
Use of Human Milk” (https://doi.org/10.1542/peds.2022-057988). Newborns discharged less than 48hours after delivery must be
examined within
48 hours of discharge, per “Hospital Stay for Healthy Term Newborn Infants” (https://doi.org/10.1542/peds.2015-0699).
5. Screen, per “Clinical Practice Guideline for the Evaluation and Treatment of Children and Adolescents with Obesity”
(https://doi.org/10.1542/peds.2022-060640).
6. Screening should occur per “Clinical Practice Guideline for Screening and Management of High Blood Pressure in Children
and Adolescents” (https://doi.org/10.1542/peds.2017-1904). Blood pressure measurement in infants and children with
specic risk conditions should be performed at visits before age 3years.
7. A visual acuity screen is recommended at ages 4 and 5 years, as well as in cooperative 3-year-olds. Instrument-based screening
may be used to assess risk at ages 12 and 24 months, in addition to the well visits at 3 through 5 years of age. See “Visual System
Assessment in Infants, Children, and Young Adults by Pediatricians” (https://doi.org/10.1542/peds.2015-3596) and “Procedures
for the Evaluation of the Visual System by Pediatricians” (https://doi.org/10.1542/peds.2015-3597).
8. Conrm initial screen was completed, verify results, and follow up, as appropriate. Newborns should be screened,
per “Year 2007 Position Statement: Principles and Guidelines for Early Hearing Detection and Intervention Programs”
(https://doi.org/10.1542/peds.2007-2333).
9. Verify results as soon as possible, and follow up, as appropriate.
10. Screen with audiometry including 6,000 and 8,000 Hz high frequencies once between 11 and 14 years, once between
15 and 17 years, and once between 18 and 21 years. See “The Sensitivity of Adolescent Hearing Screens Signicantly Improves
by Adding High Frequencies” (https://www.sciencedirect.com/science/article/abs/pii/S1054139X16000483).
11. Screening should occur per “Incorporating Recognition and Management of Perinatal Depression Into Pediatric Practice”
(https://doi.org/10.1542/peds.2018-3259).
12. Screening should occur per “Promoting Optimal Development: Identifying Infants and Young Children With Developmental
Disorders Through Developmental Surveillance and Screening” (https://doi.org/10.1542/peds.2019-3449).
13. Screening should occur per “Identication, Evaluation, and Management of Children With Autism Spectrum Disorder”
(https://doi.org/10.1542/peds.2019-3447).

Summary of Changes Made to the Bright Futures/AAP Recommendations
for Preventive Pediatric Health Care (Periodicity Schedule)
This schedule reects changes approved in December 2023 and published in June 2024. For updates and a list of previous changes made,
visit www.aap.org/periodicityschedule.
FOOTNOTE CHANGES MADE IN DECEMBER 2023
• 35 DAY VISIT
Footnote 4
This footnote reects the AAP “Policy Statement: Breastfeeding and the
Use of Human Milk”, published June 2022.
• BODY MASS INDEX
Footnote 5
This footnote reects the AAP “Clinical Practice Guideline for the
Evaluation and Treatment of Children and Adolescents with Obesity”,
published January 2023.
• BEHAVIORAL/SOCIAL/EMOTIONAL SCREENING
Footnote 14
This footnote reects the USPSTF “Anxiety in Children and Adolescents:
Screening” recommendations, published October 2022.
• TOBACCO, ALCOHOL, OR DRUG USE ASSESSMENT
(Footnote 15)
This footnote reects the Centers for Disease Control (CDC) and National
Institute of Drug Abuse (NIDA) guidance related to recommending and
prescribing Naloxone.
• NEWBORN BILIRUBIN SCREENING
Footnote 21
This footnote reects the AAP “Clinical Practice Guideline Revision:
Management of Hyperbilirubinemia in the Newborn Infant 35 or More
Weeks of Gestation”, published August 2022.
• ORAL HEALTH
Footnotes 35 and 36
These footnotes reect the AAP clinical report, “Maintaining and
Improving the Oral Health of Young Children”, published December 2022.
CHANGES MADE IN DECEMBER 2022
HIV
The HIV screening recommendation has been updated to extend the upper age
limit from 18 to 21 years (to account for the range in which the screening can
take place) to align with recommendations of the US Preventive Services Task
Force and AAP policy (“Adolescents and Young Adults: The Pediatrician’s Role
in HIV Testing and Pre- and Postexposure HIV Prophylaxis”).
• Footnote 30 has been updated to read as follows: “Screen adolescents
for HIV at least once between the ages of 15 and 21, making every
eort to preserve condentiality of the adolescent, as per ‘Human
Immunodeciency Virus (HIV) Infection: Screening’ (https://www.
uspreventiveservicestaskforce.org/uspstf/recommendation/human-
immunodeciency-virus-hiv-infection-screening); after initial screening,
youth at increased risk of HIV infection should be retested annually or more
frequently, as per ‘Adolescents and Young Adults: The Pediatrician’s Role in
HIV Testing and Pre- and Postexposure HIV Prophylaxis’
(https://doi.org/10.1542/peds.2021-055207)”
(continued)
This program is supported by the Health Resources and
Services Administration (HRSA) of the U.S. Department of
Health and Human Services (HHS) as part of an award totaling
$1,766,000 with 0% nanced with non-governmental sources.
The contents of this document are those of the author(s) and
do not necessarily represent the ocial views of, nor an
endorsement, by HRSA, HHS, or the U.S. Government.
For more information, please visit HRSA.gov.
14. Screen for behavioral and social-emotional problems per “Promoting
Optimal Development: Screening for Behavioral and Emotional Problems”
(https://doi.org/10.1542/peds.2014-3716), “Mental Health Competencies for
Pediatric Practice” (https://doi.org/10.1542/peds.2019-2757), “Clinical Practice
Guideline for the Assessment and Treatment of Children and Adolescents
With Anxiety Disorders” (https://pubmed.ncbi.nlm.nih.gov/32439401),
“Screening for Anxiety in Adolescent and Adult Women: A Recommendation
From the Women’s Preventive Services Initiative” (https://pubmed.ncbi.nlm.nih.
gov/32510990), and “Anxiety in Children and Adolescents: Screening”
(https://www.uspreventiveservicestaskforce.org/uspstf/recommendation/screening-
anxiety-children-adolescents). The screening should be family centered and may
include asking about caregiver emotional and mental health concerns and social
determinants of health, racism, poverty, and relational health. See “Poverty and Child
Health in the United States” (https://doi.org/10.1542/peds.2016-0339), ”The Impact of
Racism on Child and Adolescent Health” (https://doi.org/10.1542/peds.2019-1765),
and “Preventing Childhood Toxic Stress: Partnering With Families and Communities
to Promote Relational Health” (https://doi.org/10.1542/peds.2021-052582).
15. A recommended tool to assess use of alcohol, tobacco and nicotine, marijuana, and
other substances, including opioids is available at http://crat.org. If there is a concern
for substance or opioid use, providers should consider recommending or prescribing
Naloxone (see https://www.cdc.gov/ore/search/pages/2018-evidence-based-strategies.
html and https://nida.nih.gov/publications/drugfacts/naloxone).
16. Screen adolescents for depression and suicide risk, making every eort to preserve
condentiality of the adolescent. See “Guidelines for Adolescent Depression in
Primary Care (GLAD-PC): Part I. Practice Preparation, Identication, Assessment,
and Initial Management” (https://doi.org/10.1542/peds.2017-4081), “Mental Health
Competencies for Pediatric Practice” (https://doi.org/10.1542/peds.2019-2757), “Suicide
and Suicide Attempts in Adolescents” (https://doi.org/10.1542/peds.2016-1420), and
“The 21st Century Cures Act & Adolescent Condentiality” (https://adolescenthealth.
org/press_release/naspag-sahm-statement-the-21st-century-cures-act-adolescent-
condentiality/).
17. At each visit, age-appropriate physical examination is essential, with infant
totally unclothed and older children undressed and suitably draped. See
“Use of Chaperones During the Physical Examination of the Pediatric Patient”
(https://doi.org/10.1542/peds.2011-0322).
18. These may be modied, depending on entry point into schedule and individual need.
19. Conrm initial screen was accomplished, verify results, and follow up, as
appropriate. The Recommended Uniform Screening Panel (https://www.hrsa.gov/
advisory-committees/heritable-disorders/rusp/index.html), as determined by The
Secretary’s Advisory Committee on Heritable Disorders in Newborns and Children, and
state newborn screening laws/regulations (https://www.babysrsttest.org/) establish
the criteria for and coverage of newborn screening procedures and programs.
20. Verify results as soon as possible, and follow up, as appropriate.
21. Conrm initial screening was accomplished, verify results, and follow up, as appropriate.
See “Clinical Practice Guideline Revision: Management of Hyperbilirubinemia in the
Newborn Infant 35 or More Weeks of Gestation” (https://doi.org/10.1542/peds.2022-
058859).
22. Screening for critical congenital heart disease using pulse oximetry should be
performed in newborns, after 24 hours of age, before discharge from the hospital,
per “Endorsement of Health and Human Services Recommendation for Pulse
Oximetry Screening for Critical Congenital Heart Disease”
(https://doi.org/10.1542/peds.2011-3211).
23. Schedules, per the AAP Committee on Infectious Diseases, are available at
https://publications.aap.org/redbook/pages/immunization-schedules. Every visit
should be an opportunity to update and complete a child’s immunizations.
24. Perform risk assessment or screening, as appropriate, per recommendations in
the current edition of the AAP Pediatric Nutrition: Policy of the American Academy
of Pediatrics (Iron chapter).
25. For children at risk of lead exposure, see “Prevention of Childhood Lead Toxicity”
(https://doi.org/10.1542/peds.2016-1493) and “Low Level Lead Exposure Harms Children:
A Renewed Call for Primary Prevention” (https://stacks.cdc.gov/view/cdc/11859).
26. Perform risk assessments or screenings as appropriate, based on universal screening
requirements for patients with Medicaid or in high prevalence areas.
27. Tuberculosis testing per recommendations of the AAP Committee on Infectious
Diseases, published in the current edition of the AAP Red Book: Report of the Committee
on Infectious Diseases. Testing should be performed on recognition of high-risk factors.
28. See “Integrated Guidelines for Cardiovascular Health and Risk Reduction in Children
and Adolescents” (http://www.nhlbi.nih.gov/guidelines/cvd_ped/index.htm).
29. Adolescents should be screened for sexually transmitted infections (STIs) per
recommendations in the current edition of the AAP Red Book: Report of the
Committee on Infectious Diseases.
30. Screen adolescents for HIV at least once between the ages of 15 and 21, making every
eort to preserve condentiality of the adolescent, as per “Human Immunodeciency
Virus (HIV) Infection: Screening” (https://www.uspreventiveservicestaskforce.org/uspstf/
recommendation/human-immunodeciency-virus-hiv-infection-screening); after initial
screening, youth at increased risk of HIV infection should be retested annually or more
frequently, as per “Adolescents and Young Adults: The Pediatrician’s Role in
HIV Testing and Pre- and Postexposure HIV Prophylaxis” (https://doi.org/10.1542/
peds.2021-055207).
31. Perform a risk assessment for hepatitis B virus (HBV) infection according to
recommendations per the USPSTF (https://www.uspreventiveservicestaskforce.org/
uspstf/recommendation/hepatitis-b-virus-infection-screening) and in the 2021–2024
edition of the AAP Red Book: Report of the Committee on Infectious Diseases, making
every eort to preserve condentiality of the patient.
32. All individuals should be screened for hepatitis C virus (HCV) infection according
to the USPSTF (https://www.uspreventiveservicestaskforce.org/uspstf/
recommendation/hepatitis-c-screening) and Centers for Disease Control and Prevention
(CDC) recommendations (https://www.cdc.gov/mmwr/volumes/69/rr/rr6902a1.htm)
at least once between the ages of 18 and 79. Those at increased risk of HCV infection,
including those who are persons with past or current injection drug use, should be
tested for HCV infection and reassessed annually.
33. Perform a risk assessment, as appropriate, per “Sudden Death in the Young: Information
for the Primary Care Provider” (https://doi.org/10.1542/peds.2021-052044).
34. See USPSTF recommendations (https://www.uspreventiveservicestaskforce.org/uspstf/
recommendation/cervical-cancer-screening). Indications for pelvic examinations prior
to age 21 are noted in “Gynecologic Examination for Adolescents in the Pediatric Oce
Setting” (https://doi.org/10.1542/peds.2010-1564).
35. Assess whether the child has a dental home. If no dental home is identied, perform
a risk assessment (https://www.aap.org/en/patient-care/oral-health/oral-health-
practice-tools/) and refer to a dental home. Recommend brushing with uoride
toothpaste in the proper dosage for age. See “Maintaining and Improving the Oral
Health of Young Children” (https://doi.org/10.1542/peds.2022- 060417).
36. Perform a risk assessment (https://www.aap.org/en/patient-care/oral-health/oral-
health-practice-tools/). See “Maintaining and Improving the Oral Health of Young
Children” (https://doi.org/10.1542/peds.2022-060417).
37. The USPSTF recommends that primary care clinicians apply uoride varnish to the
primary teeth of all infants and children starting at the age of primary tooth eruption
(https://www.uspreventiveservicestaskforce.org/uspstf/recommendation/prevention-
of-dental-caries-in-children-younger-than-age-5-years-screening-and-interventions1).
Once teeth are present, apply uoride varnish to all children every 3 to 6 months in the
primary care or dental oce based on caries risk. Indications for uoride use are noted in
“Fluoride Use in Caries Prevention in the Primary Care Setting” (https://doi.org/10.1542/
peds.2020-034637).
38. If primary water source is decient in uoride, consider oral uoride supplementation.
See “Fluoride Use in Caries Prevention in the Primary Care Setting”
(https://doi.org/10.1542/peds.2020-034637).
