
Combine PDFs in Submission-ready Format Quick and Easy
Robin Wu, Lili Li, and Steven Huang, PTC Therapeutics
ABSTRACT
Portable Document Format (PDF) is an essential component in the regulatory submission package (U. S.
Food and Drug Administration, 2013; International Council for Harmonisation of Technical Requirements
for Pharmaceuticals for Human Use, 2017). The conventional approaches of combining PDFs include
using a SAS macro or utilizing a third-party stand-alone tool. Both approaches are usually time-
consuming, unstable, and technically challenging in real practices. In the meantime, however, very limited
research and utilities have been shared in the SAS community on how to combine PDFs properly and
efficiently in a submission-ready format. This paper will address the gap here by introducing a novel
approach that implements both a SAS macro and two third-party utilities (a Visual Basic application and a
Java application) to make the PDF combination quick, easy, and submission-ready.
KEYWORDS
Combine PDFs, Submission-Ready
INTRODUCTION
U. S. Food and Drug Administration (FDA) and International Council for Harmonisation of Technical
Requirements for Pharmaceuticals for Human Use (ICH) have very specific requirements for the
combined PDF files in submission packages (U. S. Food and Drug Administration, 2005; U. S. Food and
Drug Administration, 2013; International Council for Harmonisation of Technical Requirements for
Pharmaceuticals for Human Use, 2015; International Council for Harmonisation of Technical
Requirements for Pharmaceuticals for Human Use, 2017). For instance, both FDA and ICH request that
the combined PDF file needs to include a Table of Contents (TOCs) page where there are proper
hyperlinks for documents of more than five pages. In the combined PDF, FDA and ICH also require
bookmarks for each table, figure, or listing (TFL) listed in the TOCs. Even for the TFL outputs in
submission package that are produced in non-PDF file formats, FDA instructs that those files “should also
be provided in PDF format” (U. S. Food and Drug Administration, 2013, p. 2).
To convert and combine TFL outputs into one PDF file, conventional methods generally come in two
approaches. The first common approach is to directly combine in SAS. One frequently used method is to
produce SAS Item Store catalogs using the ODS Document statement while producing the TFLs. Then a
stand-alone SAS macro would be called to read in the SAS Item Store catalogs to be combined into one
PDF file at the end. The other common approach is to utilize third-party applications. For instance, Adobe
Acrobat is one of the most popular applications that allow the users to manually select, convert, and
combine individual files from WORD documents format into combined files in PDF format.
However, both conventional approaches pose significant challenges in practical uses. One most
outstanding challenge is that both approaches are time-consuming and resource-consuming, especially
when combining a large number of TFL outputs. It is not unusual for the SAS Item Store catalogs method
to take hours in converting and combining a couple of hundreds of TFL outputs. Similarly in combining
files using Adobe Acrobat, it is not only time-consuming but also very often unstable in processing due to
exhaustion of computing resources. The second challenge is in compliance with the regulatory guidelines.
For instance, when using third-party applications such as Adobe Acrobat, the final combined PDFs are
not submission-ready as it will not automatically generate the proper TOCs page required by the
regulatory agencies mentioned above.
Although it has been generally established that PDF is an integral part of the regulatory submissions
while there are many challenges in conventional approaches as discussed above, not much attention has
been shed on PDF creation for submission packages, especially on the combined PDF files (Poppe,
2005; Shao & Zhang, 2023; Xin & Wang, 2016). This paper introduces a SAS macro that utilizes a Visual
Basic application and a Java application to efficiently create the combined PDF document that complies
with regulatory requirements for submission purpose.
Compared to conventional methods, the new SAS macro is 1) highly efficient by significantly reducing the
processing time by at least 60% to 90%. The larger of combined file’s size, the more in reduction of the
processing time; 2) very easy to use as it removes all complex macro set-ups; 3) is resource-effective
where the processing does not require much computing resource, thus make it smooth and stable to
execute even with large number of TFL outputs to be converted and combined.
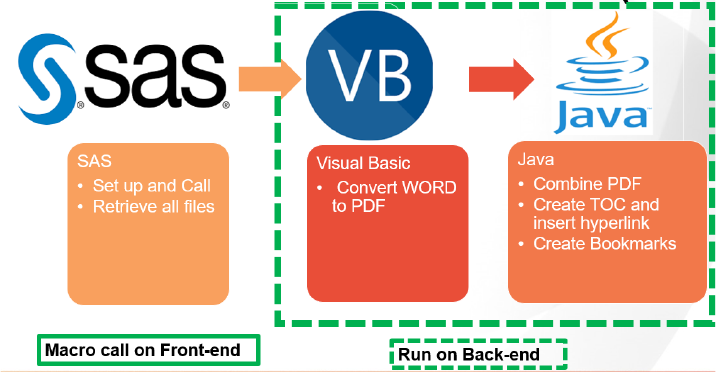
PROCESS FLOWCHART
The graph below demonstrates the process flow in and behind the SAS macro itself. The whole utility
here can be divided into two components: the SAS macro running on the front-end, and the Visual Basic
application and Java application running on the back-end. More details about each of the components will
be discussed below.
Figure 1. The Process of Combined PDF Produc�on
FRONT-END COMPONENT: SAS MACRO
The SAS macro is the front-end component in this application. As the first step of the process, it retrieves
all the TFL output files based in the designated folder path to determine if the TFL output files in WORD
document format have corresponding PDF files. If no corresponding PDF file to a particular WORD output
(in rft format) is found, the macro will report such case into a temporary data set (topdf) where all the TLF
reported will be converted into PDF format at a later step of the application. Details of the codes to realize
this function are shared below:
filename rtf pipe %sysfunc(quote(dir "%sysfunc(pathname(trtf))\*.rtf" /b));
filename pdf pipe %sysfunc(quote(dir "%sysfunc(pathname(trtf))\*.pdf" /b));
data dirrtf;
infile rtf pad ;
length fname $ 100 filex $100;
input fname $ char100.;
file=input(scan(fname, 1, ' '), ?? $char100.);
filex=tranwrd(file, '.rtf', '');
/*if ^missing(file);*/
if substr(fname, 1, 1)^='' ;
keep file filex ;
run;
proc sort; by filex; run;
data dirpdf;
infile dirpdf pad;
length fname $ 100 filex $100;
input fname $ char100.;
file=input(scan(fname, 1, ' '), ?? $char100.);
filex=tranwrd(file, '.pdf', '');
if substr(fname, 1, 1)^='' ;
keep file filex;
run;
proc sort; by filex; run;
data topdf;
merge dirrtf(in=a keep=filex file) dirpdf(in=b keep=filex);
by filex;
if a=1 and b=0;
run;
Note, trtf is path of the output’s location defined by the libname statement; Using pathname
function can convert it back into a macro variable to be directly used in the filename statement as
illustrated above.
Using a simple if a=1 and b=0 statement in the last data step above, the topdf data set will yield a
list of outputs where any WORD output is still missing corresponding PDF format files and need to be
converted into PDFs before combining all the PDF files. Details of rtf to PDF conversion will be further
discussed in the back-end components section below.
If WORD output needs to be converted into PDF, the next step of the application is to determine the
scope of PDF to be combined. This is achieved by a simple SAS macro to utilize an external excel
spreadsheet (TOC file) and generate a text file to be fed into the back-end Java API tool which will be
presented with more details below.
As shown in the codes below, only two macro parameters are needed. The tocfile denotes the file
name of the external excel spreadsheet where it contains all the outputs’ proper table number, table title,
and output names. The subset macro variable is optional only when a separate PDF file for a subset of
the outputs is needed.
The page header, font size, font color, and page number location are defined in the filename statement
after the back-end Java API tool (MergePdf.exe) is called in this macro. Details of this SAS macro codes
can be found below.
%macro combinePDF(tocfile=, subset= );
x %quote("P:");
x %quote(cd "P:\Macro_Library_Development\Combine_PDF\");
data temp;
set alltocx;
⊂
run;
data _null_;
set temp;
file "%sysfunc(pathname(trtf))\&tocfile..txt" lrecl=3000;
if _n_=1 then do;
put "File Name, TFL No., TFL Title";
end;
varlen=length(tocx);
put tocx $varying3000. varlen;
run;
filename cmppdf pipe
%sysfunc(quote(P:\Macro_Library_Development\Combine_PDF\MergePdf.exe -
s="%sysfunc(pathname(trtf))\&tocfile..txt" -
d="%sysfunc(pathname(trtf))\&tocfile._%sysfunc(today(), yymmdd10.).pdf"
-toc-title="%str(PTC Therapeutics, Inc. - &protocol)" -toc-fontsize=8 -
toc-fontfamily=%str(cour.ttf) %str(-toc -b) -add-toc-page=0));
data _null_;
infile cmppdf pad;
length fname $130;
input fname $char130.;
putlog fname;
run;
%mend;
BACK-END COMPONENTS: VB APPLICATION AND JAVA API TOOL
As illustrated in the process flowchart, there are two applications running on the back-end: the Visual
Basic application and the Java application utilizing i Te xt API.
The function of the VB application component is straightforward. When the SAS macro detects that there
are still WORD document files missing corresponding PDF format files, this application will be accessed
and called to convert those files into individual PDF format files.
Compared to other programming languages, VB application has its unique advantages as its high
compatibility and accessibility into the Window Office System. In this case, the VB application can best
reserve the original format from the WORD documents. When we were researching for possible solutions
and alternative programming languages, for instance, the blank space on the individual PDF page could
be compressed especially with graphic outputs. In other words, the individual PDF in the final combined
version would look more crammed than the original WORD output file. While on the other hand, the VB
application can preserve the exact original output format from the individual WORD document file when
combining into the final combined PDF file. In other words, what you see in WORD is what you get in the
final combined PD F. Sample VB codes to realize this function are included below for further references:
Sub SaveWordAsPDF(p_strFilePath, p_strPdfFolderPath)
'Save Word file as a PDF
'Initialise
Dim objWord, objDocument
Set objWord = CreateObject("Word.Application")
'Open the file
Set objDocument = objWord.Documents.Open(p_strFilePath)
'Save the PDF
objDocument.SaveAs PathOfPDF(p_strFilePath, p_strPdfFolderPath),
WORD_PDF
'Close the file and exit the application
objDocument.Close FALSE
objWord.Quit
End Sub
The Java application component performs a more complex function. As introduced in the SAS macro
component section above, when the scope of combination is defined as the TOC file being processed by
the SAS macro, the Java application generates a table of content page based on the TOC file and then
retrieves the individual PDF files in scope into combining them into the final PDF file. In the meantime, it
automatically inserts proper hyperlinks between the table of content page and the corresponding output
fil, as well as adds bookmarks for each TLF output for the convenience of reviewer per recommendation
by the FDA guideline.
The Java i Te x t API is publicly available. Therefore, this paper will avoid the redundancy here by providing
references links to where interested users can find more resources for further references and research
purpose:
• Sample codes for creating table of content page(s) when merging into PDFs:
https://kb.itextpdf.com/itext/merging-documents-and-create-a-table-of-contents
• Sample codes for adding bookmarks:
https://kb.itextpdf.com/itext/bookmark-examples
CONCLUSION
This paper shares a novel approach in combining individual output files into a submission-ready PDF file.
Compared to conventional methods, this new tool significantly reduces the processing time and
computing resources needed for converting and combining files into a submission-ready PDF file. It is
user-friendly to all levels of programmers or even non-programmers.

REFERENCES:
International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use.
(2015). Specification for Submission Formats for eCTD. Retrieved from
https://admin.ich.org/sites/default/files/inline-
files/Specification_for_Submission_Formats_for_eCTD_v1_2.pdf
International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use.
(2017). Specification for PDF Formatted Documents in Regulatory Submissions. Retrieved from
https://admin.ich.org/sites/default/files/inline-files/Specification_for_PDF_Format_v1_0.pdf
IText. iText Knowledge Base. (2024). https://kb.itextpdf.com/itext/
Poppe, F. (2005). Store Your Output and Digest It Later. SAS Users Group International 30, Philadelphia,
PA, United States.
Shao, W. & Zhang, L. (2023). A utility to combine study level outputs to one PDF file for development
safety update report (DSUR) regional appendices. PharmaSUG 2023, Beijing, China.
Xin, W. & Wang, J. (2016). Utilizing SAS® and Groovy to combine multiple RTF/PDF reports to one
bookmarked PDF document. PharmaSUG China 2016, Philadelphia, PA, United States.
U. S. Food and Drug Administration. (2013). Specifications for File Format Types Using eCTD
Specifications. Retrieved from https://www.fda.gov/media/85816/download
U. S. Food and Drug Administration. (2005). Portable Document Format (PDF) Specifications: Technical
Specifications Document. Retrieved from https://www.fda.gov/media/76797/download
CONTACT INFORMATION:
Your comments and questions are valued and encouraged. Contact the author at:
Robin WU
PTC Therapeutics
rwu@ptcbio.com
Lili LI
PTC Therapeutics
lli@ptcbio.com
Steven HUANG
PTC Therapeutics
steven.huang@ptcbio.com
