
R
Carbon
R
Synthesis of p-Nitroaniline via
a Multi-Step Sequence
Carbon

R
Carbon
Contents
Objectives 1
Introduction 1
Synthesis of acetophenone oxime 1
Synthesis of acetanilide: the Beckmann rearrangement 2
Nitration of acetanilide 5
Synthesis of p-nitroaniline 5
Manuscript prepared by Dr. Almas I. Zayya and Dr. A. Jonathan Singh. School of Chemical
and Physical Sciences, Victoria University of Wellington, New Zealand.
Carbon
R
Carbon
Introduction
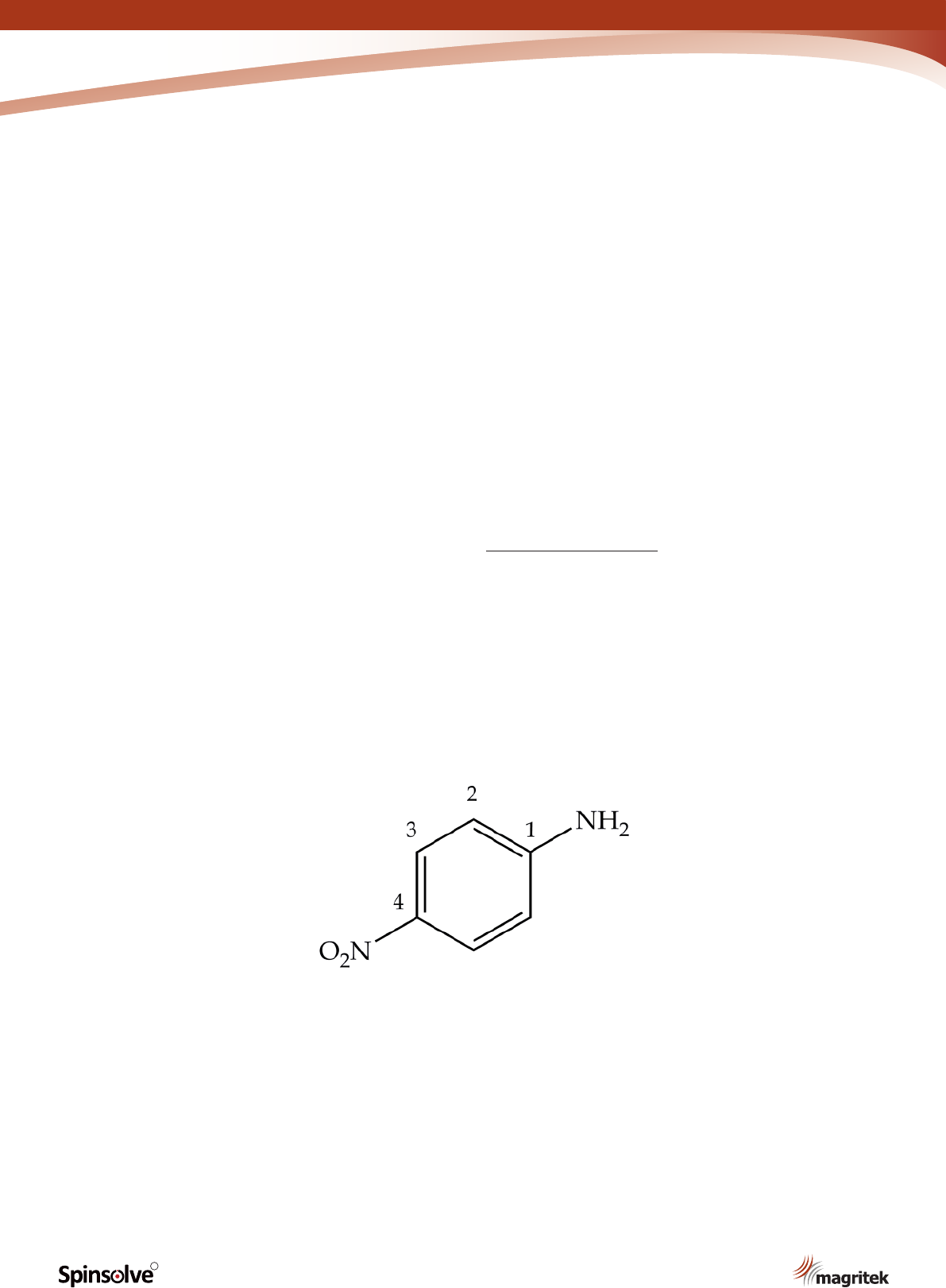
Nitroanilines are important chemical intermediates
in the manufacture of dyes.
1
In this series of
experiments, you will synthesise p-nitroaniline
(Figure 1) via a multi-step sequence.* This
particular compound is used in the synthesis of
the azo dye Para Red.
2
The synthetic sequence
to prepare p-nitroaniline from acetophenone
involves the transformation of one functional
group on a monosubstituted benzene into another
through chemical reactions, then performing
an electrophilic aromatic substitution reaction
to obtain the target compound. The various
compounds prepared will be characterised by
1
H NMR spectroscopy.
Figure 1. p-Nitroaniline.
Objectives
The principal aims of these experiments are to
provide experience in the synthesis, isolation,
purication and characterisation of simple
aromatic compounds. In particular, you will
study aromatic substitution reactions in which
functional groups greatly inuence further
substitution of monosubstituted benzenes. The
main characterisation technique utilised in these
experiments is
1
H NMR spectroscopy using the
benchtop Spinsolve NMR spectrometer.
1
*
In these experiments, ortho, meta and para (omp) nomenclature is used
to indicate the substituent position in disubstituted benzenes in place of
IUPAC (systematic) nomenclature.
R
Carbon
Synthesis of acetophenone oxime
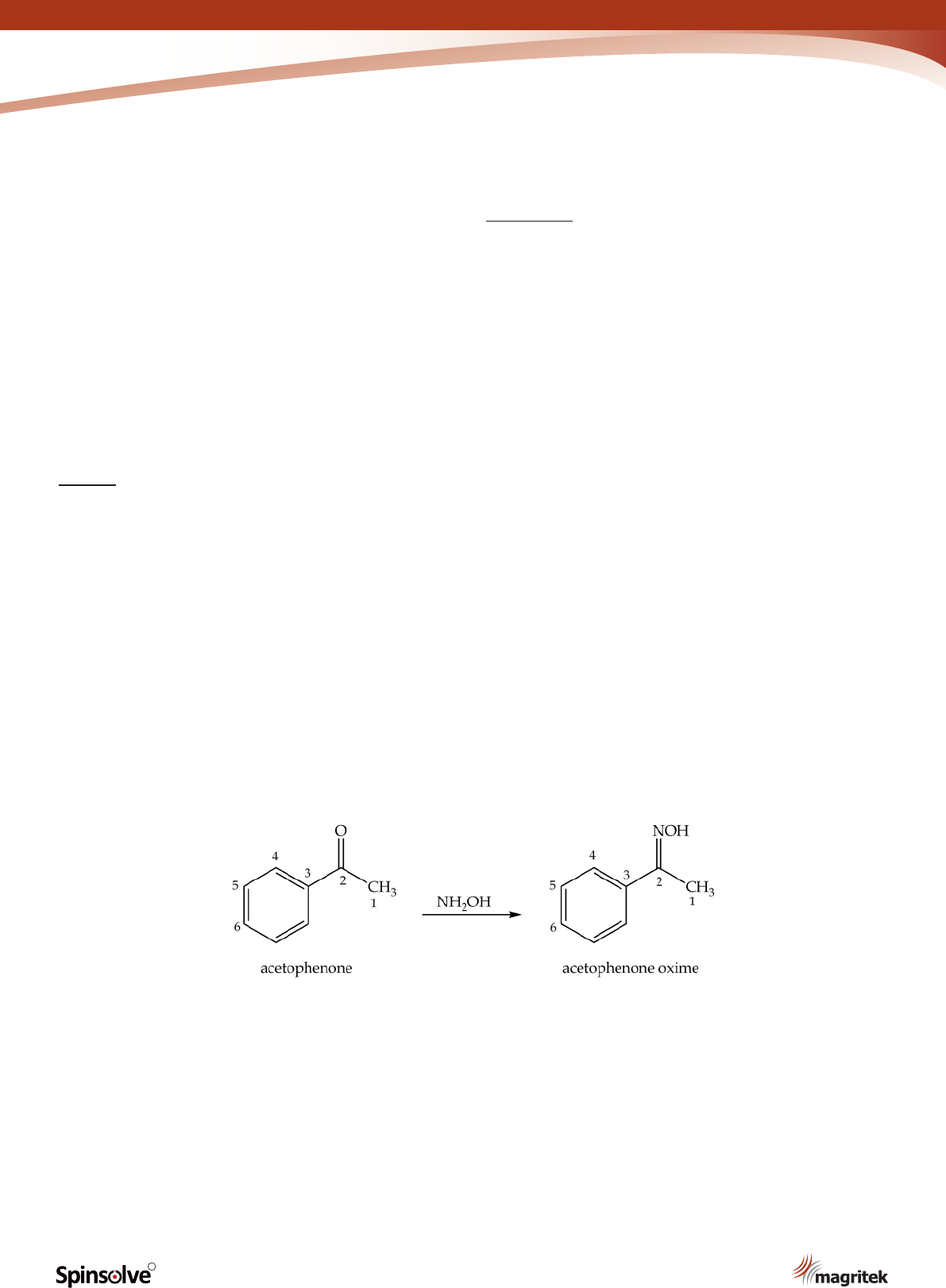
The rst step in the synthesis of p-nitroaniline
is the preparation of acetophenone oxime from
acetophenone (Scheme 1). Oximes are
highly crystalline compounds that feature a
carbon-nitrogen double bond, with an OH group
on the nitrogen atom (>C=N−OH).
3
They are used
extensively in synthetic organic chemistry for the
protection, purication and characterisation of
carbonyl compounds.
4
Oximes are also versatile
building blocks for the synthesis of nitrogen-
containing compounds.
3
Safety
Ethanol is ammable; handle with care.
Acetophenone, sodium acetate trihydrate
(CH
3
COONa.3H
2
O) and acetophenone oxime are
irritating to the skin, eyes and respiratory system.
Avoid contact and do not ingest or inhale.
Hydroxylamine hydrochloride (NH
2
OH.HCl) is
corrosive, avoid all contact and handle with
caution. Deuterochloroform (CDCl
3
) is toxic,
handle with care.
Scheme 1. Synthesis of acetophenone oxime.
2
Procedure
To a solution of water (30 mL) and ethanol (10 mL)
in a 100 mL round bottom ask, add acetophenone
(3.75 mL), hydrated sodium acetate crystals (7.50
g) and hydroxylamine hydrochloride (3.75 g).
Heat the reaction mixture with stirring on a hot
water bath for 10 min (Figure 2). Colourless oil
droplets should form on top of the solution. Cool
the mixture in an ice bath for 30 min, during which
time the oil should solidify. If necessary, induce
crystallisation by scratching the sides of the
ask with a glass rod. Collect the white solid by
ltration, wash with cold water and dry in the air.
Recrystallise the crude product from boiling water
(60 mL),
5
making sure that all oil droplets have
dissolved. Filter and dry the puried product, and
record your yield (Figure 3).

R
Carbon
3
Figure 2. Experimental setup for the synthesis of
acetophenone oxime.
Figure 3. Puried acetophenone oxime.
Tasks & Questions
• Calculate the theoretical and percentage yields of
acetophenone oxime.
• Record the
1
H NMR spectra of acetophenone
and acetophenone oxime using the Spinsolve
NMR spectrometer. Prepare the NMR samples
using 1 drop of acetophenone and 30 mg of
acetophenone oxime in 0.6 mL of CDCl
3
each.
• Record and assign the IR spectra of
acetophenone and acetophenone oxime.
• Record the melting points of acetophenone and
acetophenone oxime.
• Assign the
1
H NMR spectra of acetophenone and
acetophenone oxime.
• Give a mechanism for the transformation of
acetophenone into acetophenone oxime.
• Why is sodium acetate used in the reaction?
• Show how acetophenone oxime can exist as
geometric stereoisomers.
R
Carbon
1
H NMR Spectra
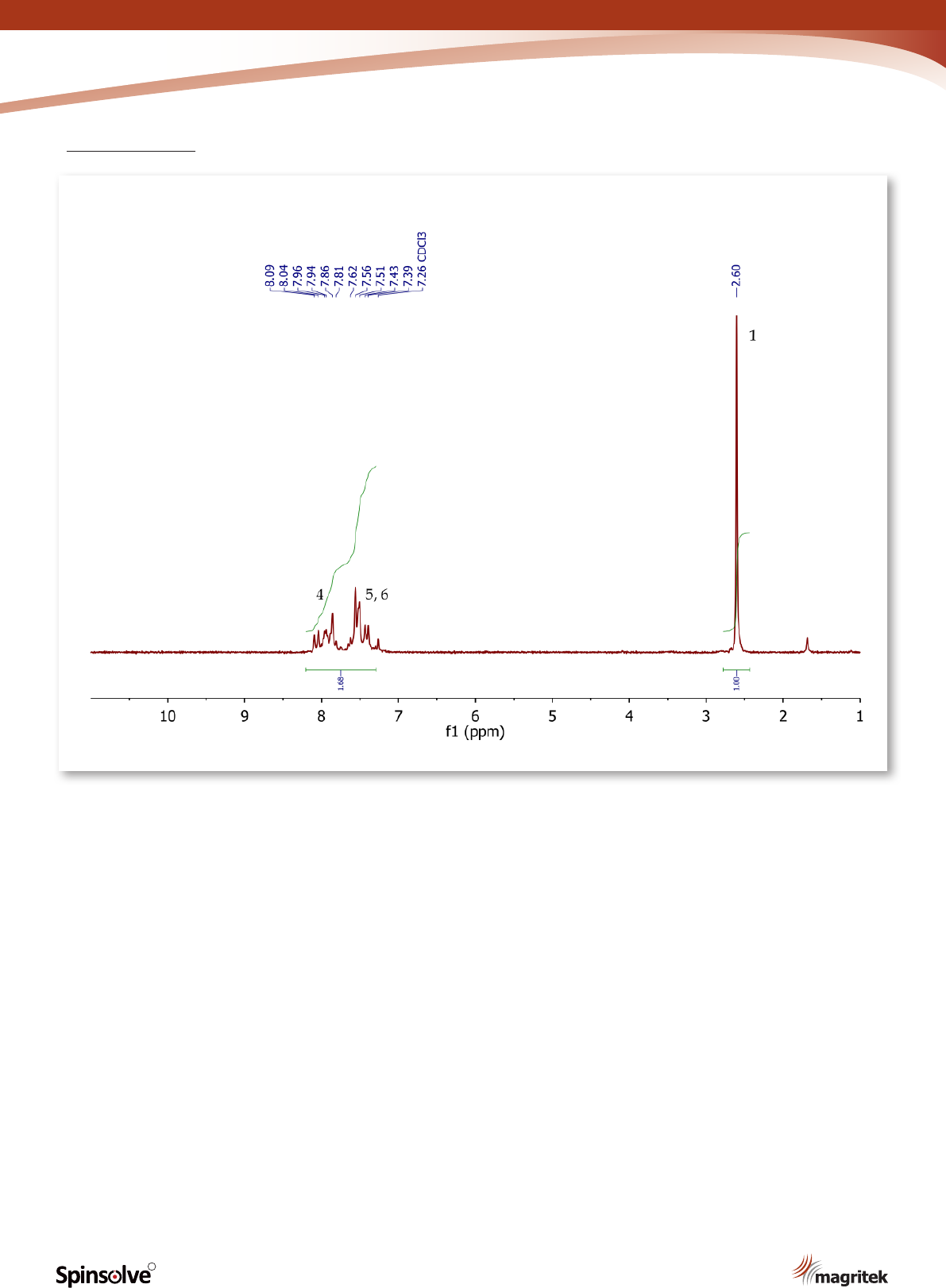
Figure 4.
1
H NMR spectrum of acetophenone, CDCl
3
.
4
The
1
H NMR spectrum of acetophenone (Figure 4)
shows a singlet (3H) at 2.60 ppm, corresponding
to the methyl group at position 1. The ve aromatic
protons at positions 4, 5 and 6 resonate as a
multiplet between 7.39-8.09 ppm.
R
Carbon
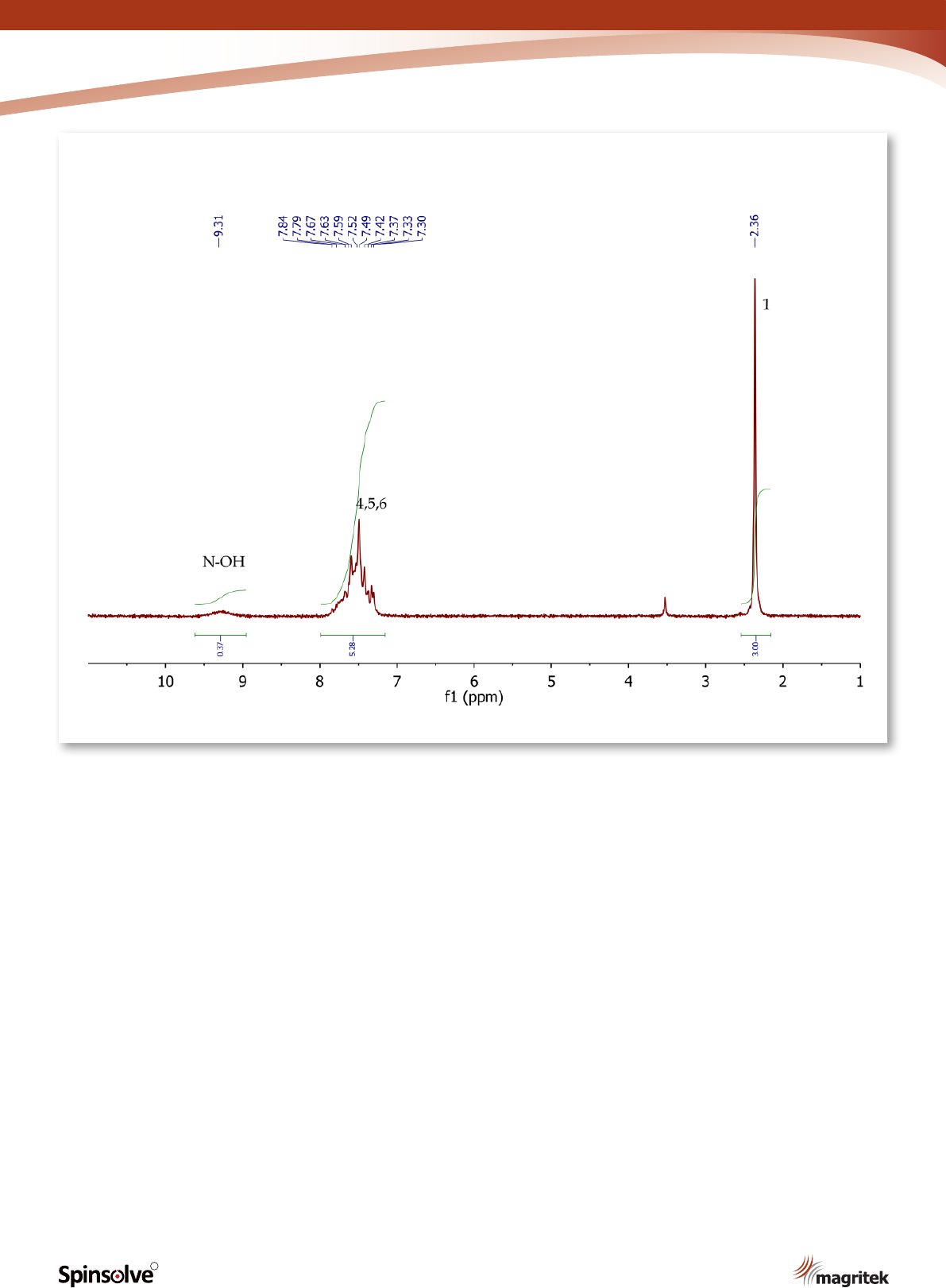
Figure 5.
1
H NMR spectrum of acetophenone oxime, CDCl
3
.
The
1
H NMR spectrum of acetophenone oxime
(Figure 5) shows a singlet (3H) at 2.36 ppm,
corresponding to the methyl group at position 1.
The ve aromatic protons at positions 4, 5 and 6
5
resonate as a broad multiplet between 7.30-7.84
ppm. The exchangeable NOH proton is observed
at 9.31 ppm as a broad singlet with a low peak
integration value.
R
Carbon
6
(c)
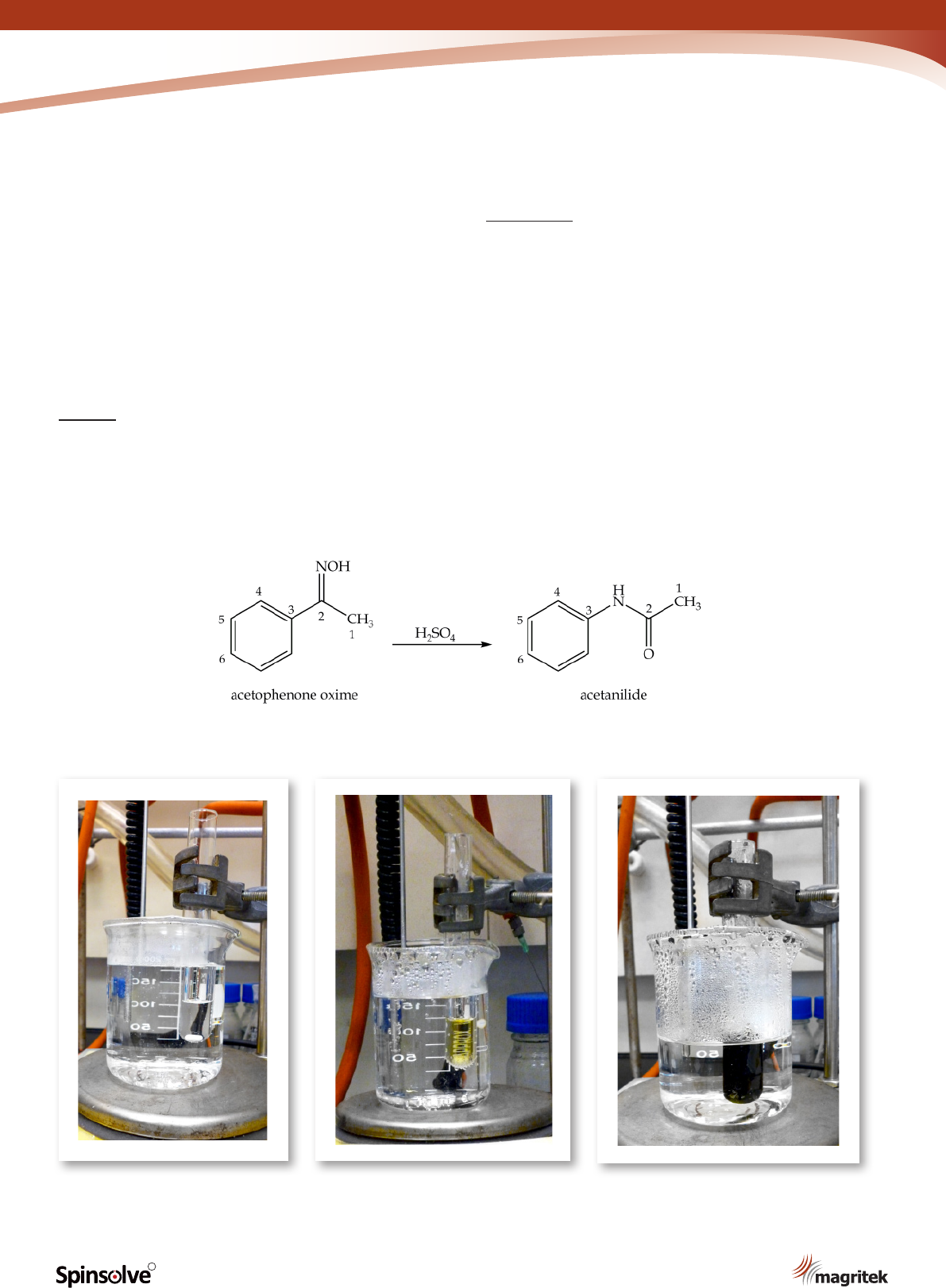
Synthesis of acetanilide: the
Beckmann rearrangement
The second step in the synthesis of p-nitroaniline is
the preparation of acetanilide from acetophenone
oxime (Scheme 2). In the presence of strong acids,
oximes can undergo molecular rearrangement to
form amides via the Beckmann rearrangement.
3
This isomerisation reaction provides a powerful
synthetic method to efciently incorporate a
nitrogen atom into compounds.
Safety
Sulfuric acid is highly corrosive; use with caution
and perform the experiment in a fume hood with
the protective glass door pulled down. Acetanilide
is an irritant, handle with care.
Procedure
Place concentrated sulfuric acid (3 mL) in a
boiling tube and heat in a hot water bath until the
temperature of the acid reaches approximately
90 °C. Add acetophenone oxime (3 g) in small
portions with stirring over a period of 20 min. Heat
and stir the reaction mixture for a further 15 min
(Figure 6). Pour the cool mixture onto crushed ice
(50 g) to precipitate the title compound. Collect
the solid by ltration and wash with cold water.
Recrystallise the crude product from 50 mL of
water and record your yield (Figure 7).
Scheme 2. Synthesis of acetanilide.
(a) (b)
Figure 6(a)-(c). Reaction mixture colour changes observed during the synthesis of acetanilide.

R
Carbon
7
(a) (b)
Figure 7(a)-(b). Crude and recrystallised acetanilide.
Tasks & Questions
• Calculate the theoretical and percentage yields
of acetanilide.
• Record the
1
H NMR spectrum of acetanilide
using the Spinsolve NMR spectrometer. Prepare
the NMR sample using 30 mg of acetanilide in
0.6 mL of CDCl
3
.
• Record the IR spectrum of acetanilide.
• Record the melting point of acetanilide.
• Assign the IR and
1
H NMR spectra of acetanilide.
• Give a mechanism for the transformation of
acetophenone oxime into acetanilide.
R
Carbon
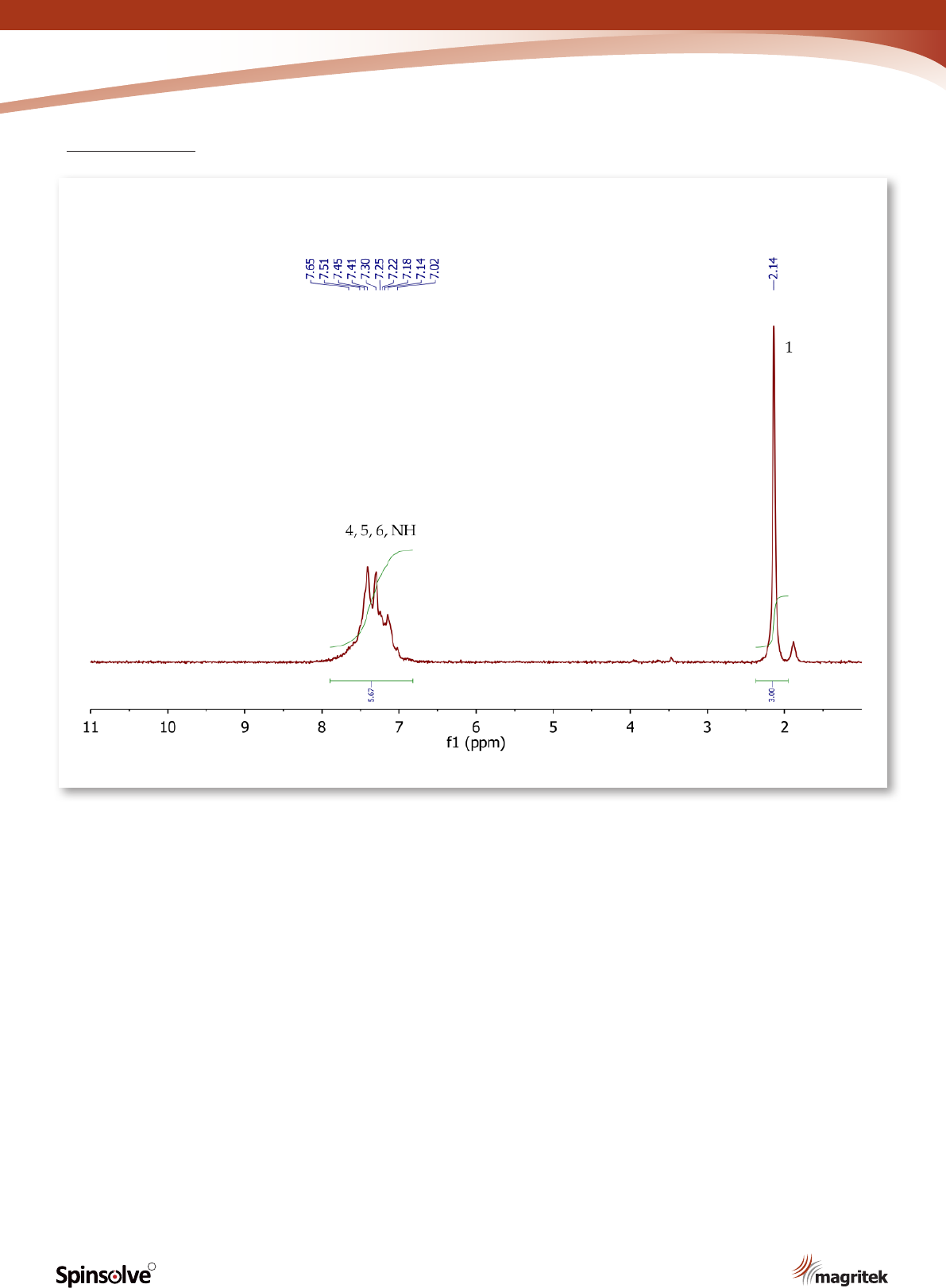
Figure 8.
1
H NMR spectrum of acetanilide, CDCl
3
.
The
1
H NMR spectrum of acetanilide (Figure 8)
shows a singlet (3H) at 2.14 ppm, corresponding
to the methyl group at position 1. The ve aromatic
protons at positions 4, 5 and 6 resonate as a broad
multiplet between 7.02-7.65 ppm. The signal
for the exchangeable NH proton may also be
overlapping with the multiplet as suggested by
the peak integration value.
8
1
H NMR Spectra
R
Carbon
9
Nitration of acetanilide
The third step in the synthesis of p-nitroaniline
is nitration of acetanilide using a mixture
of concentrated sulfuric and nitric acids to
obtain nitroacetanilide (Scheme 3). In this
electrophilic aromatic substitution reaction,
the acetamido group (−NHCOCH
3
) directs
the nitronium ion (
+
NO
2
) to the ortho and para
positions of the aromatic ring.
6
Thus, nitration
of acetanilide principally produces ortho- and
para-nitroacetanilides, with the para compound
being the major product. Separation of the
ortho- and para-nitroacetanilides is achieved by
recrystallistion from ethanol. The colourless major
product, p-nitroacetanilide, is almost insoluble in
ethanol and can be ltered out, while the yellow
ortho isomer remains in the ltrate.
Safety
Fuming nitric acid, sulfuric acid and glacial
acetic acid are highly corrosive, use with caution
and perform the experiment in a fume hood
with the protective glass door pulled down.
p-Nitroacetanilide is an irritant, avoid contact
with skin, eyes and clothing. Deuterated dimethyl
sulfoxide (DMSO-d
6
) is dangerous because it
increases the permeability of the skin to other
substances. Avoid all contact with skin and
clothing.
Procedure
Place glacial acetic acid (1.5 mL) in a boiling tube
and add 1.5 g of acetanilide. Stir the mixture and
add concentrated sulfuric acid (3 mL). Cool the
hot reaction mixture in an ice/salt bath until the
temperature drops to about 0.5 °C. With stirring,
slowly add fuming nitric acid (0.6 mL), making sure
that the temperature does not rise above 20 °C.
Once addition is complete, bring the reaction
mixture to room temperature and allow to stand
for 20 min. Pour the mixture onto ice (15 g) and
allow to stand for a further 20 min. Collect the
crude yellow solid by ltration, wash thoroughly
with water and dry in the air. Recrystallise from
the minimum amount of hot ethanol to obtain
p-nitroacetanilide as a cream-coloured crystalline
solid (Figure 9). Dry in the air and record your yield.
Scheme 3. Synthesis of p-nitroacetanilide.

R
Carbon
10
(a) Crude p-nitroacetanilide (b) Puried p-nitroacetanilide
Figure 9(a)-(b). Crude and recrystallised p-nitroacetanilide.
Tasks & Questions
• Calculate the theoretical and percentage yields
of p-nitroacetanilide.
• Record the
1
H NMR spectra of acetanilide and
p-nitroacetanilide using the Spinsolve NMR
spectrometer. Prepare the NMR samples using
30 mg of each compound in 0.6 mL of DMSO-d
6
.
• Record and assign the IR spectrum of
p-nitroacetanilide.
• Record the melting point of p-nitroacetanilide.
• Assign the
1
H NMR spectra of acetanilide and
p-nitroacetanilide.
• Give a mechanism for the formation of
p-nitroacetanilide from acetanilide.
• Why is o-nitroacetanilide the minor product in
this reaction?
R
Carbon
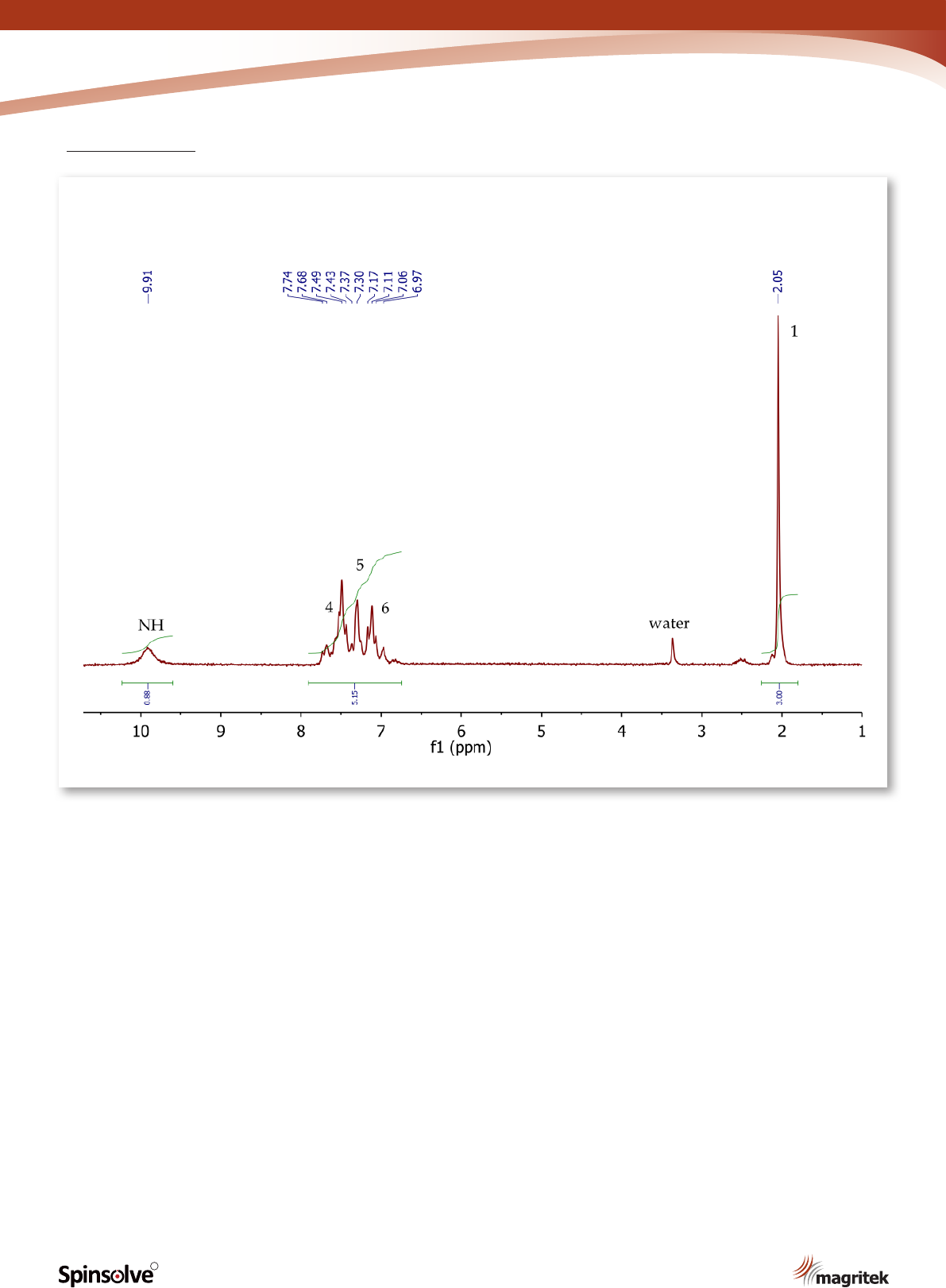
Figure 10.
1
H NMR spectrum of acetanilide, DMSO-d
6
.
The
1
H NMR spectrum of acetanilide (Figure 10)
shows a singlet (3H) at 2.05 ppm, corresponding
to the methyl group at position 1. The ve aromatic
protons at positions 4, 5 and 6 resonate as a
broad multiplet between 6.97-7.74 ppm. The
exchangeable NH proton is also observed in
DMSO-d
6
at 9.91 ppm as a broad singlet.
11
1
H NMR Spectra
R
Carbon
Figure 11.
1
H NMR spectrum of p-nitroacetanilide, DMSO-d
6
.
The
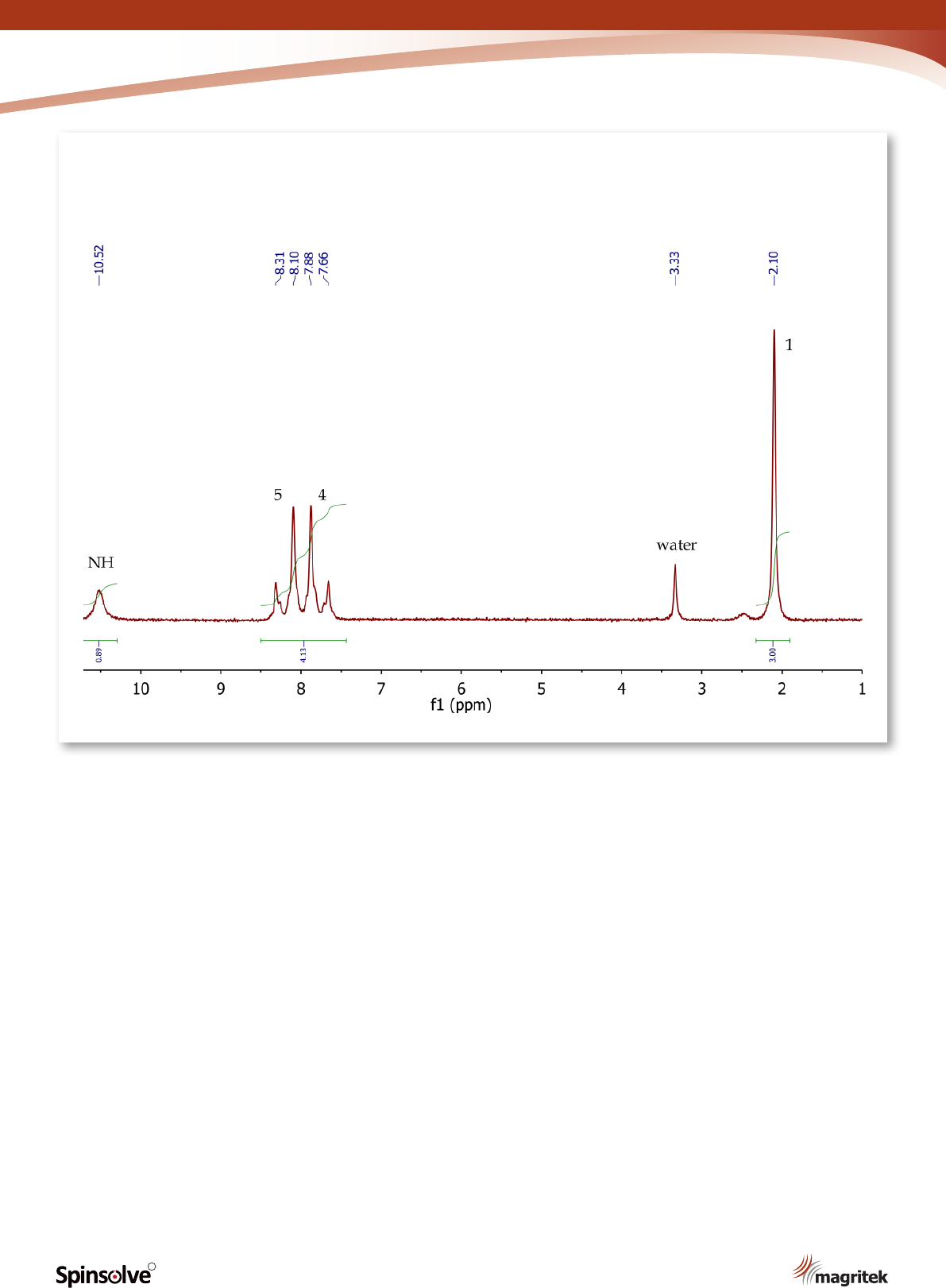
1
H NMR spectrum of p-nitroacetanilide
(Figure 11) shows a singlet (3H) at 2.10 ppm,
corresponding to the methyl group at position 1.
The four aromatic protons at positions 4 and 5
appear as a second order AA’BB’ system, with
two multiplets centred at 7.77 and 8.21 ppm. The
exchangeable NH proton is observed at 10.52 ppm
as a broad singlet.
12
R
Carbon
Synthesis of p-nitroaniline
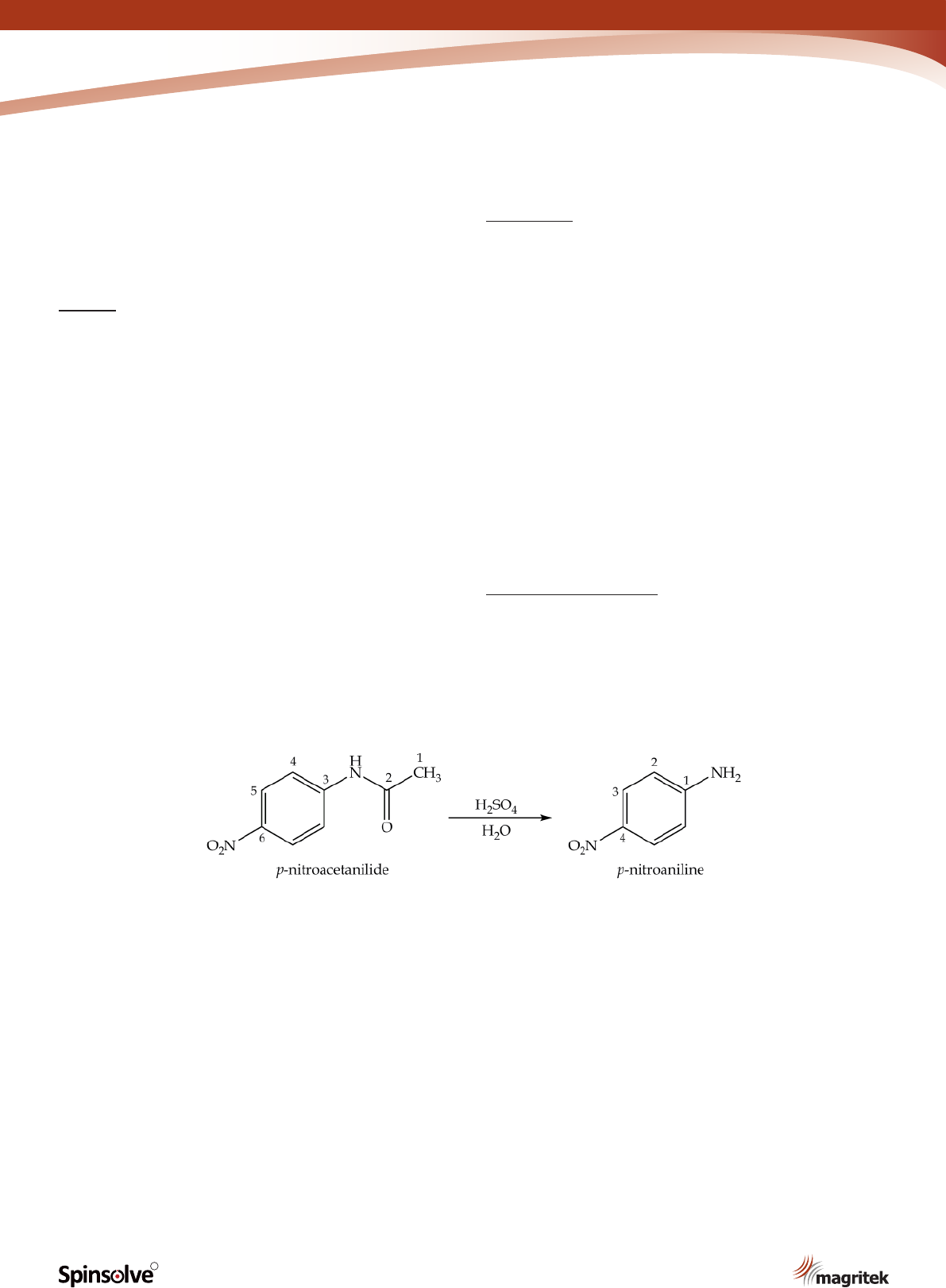
The nal step in the synthesis of p-nitroaniline is
the hydrolysis of p-nitroacetanilide under acidic
conditions (Scheme 4).
Safety
Perform the experiment in a fume hood with the
protective glass door pulled down since corrosive
sulfuric acid and sodium hydroxide solution are
being used. p-Nitroaniline is toxic, avoid contact
with skin, eyes and clothing and handle with care.
Procedure
Charge a 25 mL round bottom ask with a solution
of concentrated sulfuric acid (4 mL) and water
(3 mL).
+
Add p-nitroacetanilide (0.7 g) and heat the
reaction mixture gently under reux for 20 min.
Pour the hot mixture into cold water (20 mL),
and adjust the pH of the solution with sodium
hydroxide solution (2 M, approximately 120 mL)
until alkaline and a yellow precipitate is obtained
(Figure 12a-b). Cool the mixture in an ice bath.
Collect the crude yellow solid by ltration
(Figure 12c), wash thoroughly with water and dry
in the air. Recrystallise from 1:1 ethanol/water
mixture to obtain bright yellow crystals of the title
compound (Figure 12d). Record your yield.
Scheme 4. Synthesis of p-nitroacetanilide.
+
Sulfuric acid reacts violently with water in an exothermic reaction.
Prepare the solution by slowly adding the concentrated sulfuric acid
to water.
13

R
Carbon
14
(a) Alkaline reaction mixture
(c) Crude p-nitroaniline
(b) Yellow p-nitroaniline precipitate
(d) Puried p-nitroaniline
Figure 12(a)-(d). Precipitation and recrystallisation of p-nitroaniline.
R
Carbon
Tasks & Questions
• Calculate the theoretical and percentage yields
of p-nitroaniline.
• Record the
1
H NMR spectrum of p-nitroaniline
using the Spinsolve NMR spectrometer. Prepare
the NMR sample using 30 mg of p-nitroaniline in
0.6 mL of DMSO-d
6
.
• Record the IR spectrum of p-nitroaniline.
• Record the melting point of p-nitroaniline.
• Assign the
1
H NMR and IR spectra of
p-nitroaniline.
• Give a mechanism for the hydrolysis of
p-nitroacetanilide to give p-nitroaniline.
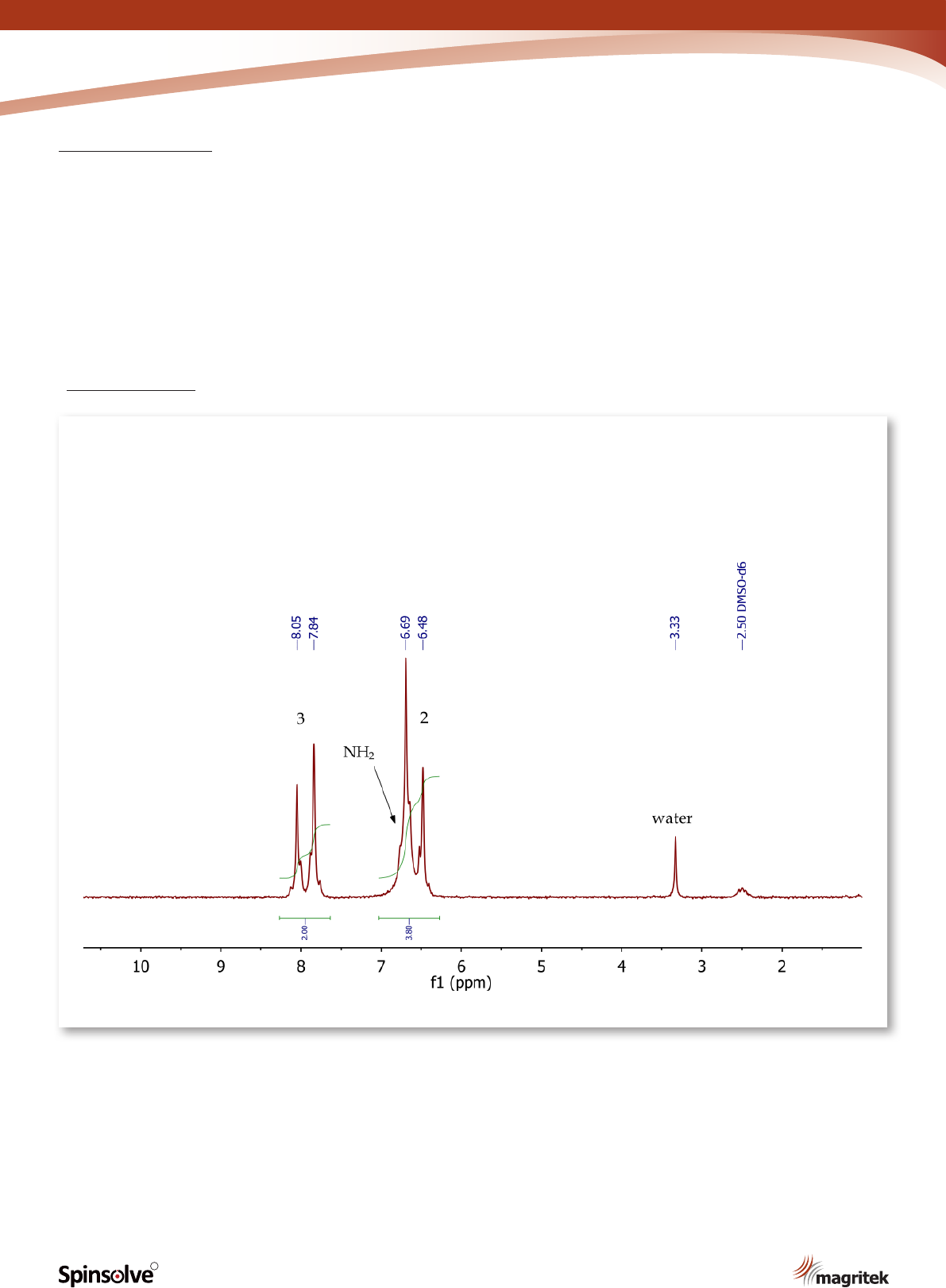
Figure 13.
1
H NMR spectrum of p-nitroanilide, DMSO-d
6
.
The
1
H NMR spectrum of p-nitroacetanilide
(Figure 13) shows two doublets at 6.59 and 7.95
ppm, corresponding to the four aromatic protons
at positions 2 and 3 respectively. The signal for the
exchangeable NH
2
protons is overlapping with the
doublet at 6.59 ppm, as indicated by the 2:1 peak
integration values.
1
H NMR Spectra
15

R
Carbon
CONTACT INFORMATION
For further information, please contact: [email protected]
GERMANY NEW ZEALAND UNITED STATES
Philipsstraße 8 6 Hurring Place, Unit 3 6440 Lusk Blvd (D108)
52068 Aachen, Germany Newlands, Wellington 6037, NZ San Diego, CA 92121, USA
Tel: +49 (241) 70525-6000 Tel: +64 4 477 7096 Tel: +1 (855) 667-6835
Fax: +49 (241) 963 1429 Fax: +64 4 471 4665 +1 (866) NMR-MTEK
Or visit our website www.magritek.com
R
References
1) Chudgar, R., J.; Oakes, J. Kirk-Othmar Encyclopedia of Chemical Technology, 2003.
2) Williamson, K., L. Macroscale and Microscale Organic Experiments; 2nd Ed; D. C.
Heath and Company, 1994.
3) Rappoport, Z.; Liebman, J., F. The Chemistry of Hydroxylamines, Oximes and
Hydroxamic Acids; John Wiley & Sons, Ltd., 2009.
4) Greene, T., W.; Wuts, P., G., M. Protective Groups in Organic Synthesis, 3rd Ed; John
Wiley & Sons, Inc., 1999.
5) Pavia, D., L.; Lampman, G., M.; Kriz, G., S.; Engel, R., G. Introduction to Organic
Laboratory Techniques: A Small Scale Approach; Thomson Brooks/Coles, 2005.
6) Jones, M., Jr. Organic Chemistry; 2nd Ed; W. W. Norton & Company, Inc., 1997.
