CODING FOR
Pediatric
Preventive
Care
TM
2022

1
Coding for Pediatric Preventive Care, 2022
is resource contains comprehensive listings of codes that may not
be used by your practice on a regular basis. We recommend that you
identify the codes most relevant to your practice and include those on
your encounter form or billing sheet.
Following are the Current Procedural Terminology (CPT®), Healthcare
Common Procedure Coding System (HCPCS) Level II, and International
Classication of Diseases, 10th Revision, Clinical Modication (ICD-10-CM)
codes most commonly reported by pediatricians in providing preventive
care services. e pediatrician, not the sta, is ultimately responsible for
the appropriate codes to report.
SYMBOL DESCRIPTION
•
A bullet at the beginning of a code means it is a new code for the current year.
+ A plus sign means the code is an add-on code.
~~ A lightning bolt indicates that a vaccine product code was approved, but the
vaccine product is still pending FDA approval.
The recommendations
in this publication do not indicate an exclusive course of treatment or
serve as a standard of medical care. Variations, taking into account individual circumstances,
may be appropriate.
© 2022 American Academy of Pediatrics
All rights reserved. No part of this publication may be reproduced, stored in a retrieval system, or
transmitted in any form or by any means—electronic, mechanical, photocopying, recording, or
otherwise—without prior written permission from the publisher.
Printed in the United States of America
Current Procedural Terminology (CPT®) 5-digit codes, nomenclature, and other data are copyright
2021 American Medical Association (AMA). All rights reserved. No fee schedules, basic units,
relative values, or related listings are included in CPT. The AMA assumes no liability for the
data contained herein.
The Bright Futures/American Academy of Pediatrics “Recommendations for Preventive Pediatric
Health Care,” also known as the “periodicity schedule,” is a schedule of screenings and assessments
recommended at each well-child visit from infancy through adolescence. The following services
and codes coincide with this schedule. For more details on the periodicity schedule, see
https://www.aap.org/en/practice-management/care-delivery-approaches/periodicity-schedule/.

2
PREVENTIVE MEDICINE SERVICE CODES
Services included under these codes include measurements (eg, length/
height, head circumference, weight, body mass index, blood pressure)
and age- and gender-appropriate examination and history (initial or
interval).
▶ Preventive medicine service codes are not time-based; therefore, time
spent during the visit is not relevant in selecting the appropriate
preventive medicine service code.
▶ If an illness or abnormality is discovered, or a preexisting problem
is addressed, in the process of performing the preventive medicine
service, and if the illness, abnormality, or problem is signicant
enough to require additional work to perform the components
of a problem-oriented evaluation and management (E/M) service
(ie, using medical decision making or time spent), the appropriate
oce or other outpatient service code (99202–99215) should be
reported in addition to the preventive medicine service code.
Append modier 25 to the oce or other outpatient service
code (eg, 99392 and 99213 25).
▶ An insignicant or trivial illness, abnormality, or problem encountered
in the process of performing the preventive medicine service should
not be separately reported.
▶ e comprehensive nature of the preventive medicine service
codes reects an age- and gender-appropriate history and physical
examination and is not synonymous with the comprehensive
examination required for some other E/M codes (eg, 99204,
99205, 99215).
▶ Immunization products and administration and ancillary studies
involving laboratory, radiology, or other procedures, or screening
tests (eg, vision, developmental, hearing) identied with a specic
CPT
code, are reported and paid for separately from the preventive
medicine service code.
▶ For more information on coding during the COVID-19 pandemic
(including telemedicine and telehealth), refer to the AAP's coding
site (www.aap.org/coding) and its page dedicated to this coding.
3
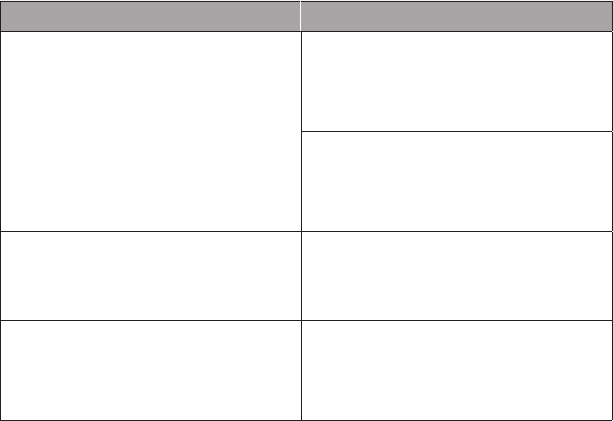
Preventive Medicine Services: New Patients
▶ Initial comprehensive preventive medicine E/M of an individual
includes an age- and gender-appropriate history; physical examination;
counseling, anticipatory guidance, or risk factor reduction interven-
tions; and the ordering of laboratory or diagnostic procedures.
▶ A new patient is dened as one who has not received any professional
face-to-face services rendered by physicians and other qualied health
care professionals (QHPs) who may report E/M services and reported
by a specic CPT code(s) from a physician/other QHP, or another
physician/other QHP of the exact same specialty and subspecialty
who belongs to the same group practice, within the past 3 years.
CPT Codes ICD-10-CM Codes
99381 Infant (younger than 1 year) Z00.110 Health supervision for newborn
under 8 days old or
Z00.111 Health supervision for newborn
8 to 28 days old or
Z00.121 Routine child health exam
with abnormal findings or
Z00.129 Routine child health exam
without abnormal findings
99382 Early childhood (age 1–4 years)
99383 Late childhood (age 5–11 years)
99384 Adolescent (age 12–17 years)
Z00.121
Z00.129
99385 18 years or older Z00.00 General adult medical exam
without abnormal findings
Z00.01 General adult medical exam
with abnormal findings
4
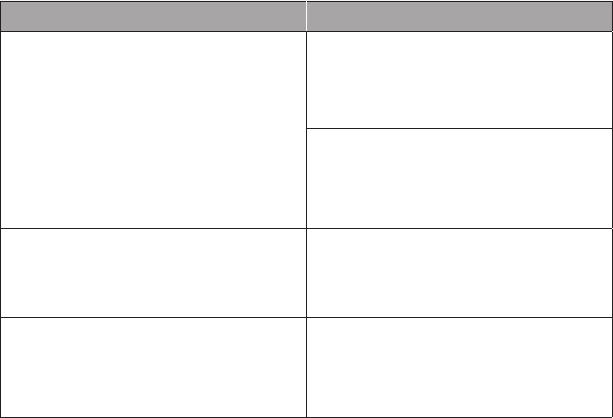
Preventive Medicine Services: Established Patients
Periodic comprehensive preventive medicine reevaluation and manage-
ment of an individual includes an age- and gender-appropriate history;
physical examination; counseling, anticipatory guidance, or risk factor
reduction interventions; and the ordering of laboratory or diagnostic
procedures.
CPT Codes ICD-10-CM Codes
99391 Infant (younger than 1 year) Z00.110 Health supervision for newborn
under 8 days old or
Z00.111 Health supervision for newborn
8 to 28 days old or
Z00.121 Routine child health exam
with abnormal findings or
Z00.129 Routine child health exam
without abnormal findings
99392 Early childhood (age 1–4 years)
99393 Late childhood (age 5–11 years)
99394 Adolescent (age 12–17 years)
Z00.121
Z00.129
99395 18 years or older Z00.00 General adult medical exam
without abnormal findings
Z00.01 General adult medical exam
with abnormal findings
Preventive Medicine Services: With And Without Abnormal Findings
e use of an ICD-10-CM code for with abnormal ndings (eg, Z00.121)
does not mean that an additional E/M service must or can be used.
Abnormal ndings can be trivial or incidental issues that do not require
additional work, but the condition is still documented or listed as con-
tributory. Examples of abnormal ndings include abnormal screening
results, new acute problem, or unstable or worsening chronic condition.
A stable chronic condition (whether addressed or not) would not warrant
the use of an abnormal ndings code. You can link an abnormal ndings
ICD-10-CM code to a screening if the screen is normal; the abnormality
will be identied with the appropriate ICD-10-CM code so the payer will
be aware.

5
COUNSELING, RISK FACTOR REDUCTION, AND
BEHAVIOR CHANGE INTERVENTION CODES
▶ Used to report services provided for the purpose of promoting health
and preventing illness or injury.
▶ ey are distinct from other E/M services that may be reported
separately when performed. However, one exception is you cannot
report counseling codes (99401–99404) in addition to preventive
medicine service codes (99381–99385 and 99391–99395).
▶ Counseling will vary with age and address such issues as family
dynamics, diet and exercise, sexual practices, injury prevention,
dental health, and diagnostic or laboratory test results available
at the time of the encounter.
▶ Codes are time-based, where the appropriate code is selected
according to the approximate time spent providing the service.
Codes may be reported when the midpoint for that time has
passed. For example, once 8 minutes are documented, one may
report 99401.
▶ Extent of counseling or risk factor reduction intervention must
be documented in the patient chart to qualify the service based
on time.
▶ Counseling or interventions are used for persons without a specic
illness for which the counseling might otherwise be used as part of
treatment.
▶ Cannot be reported with patients who have symptoms or established
illness.
▶ For counseling individual patients with symptoms or established
illness, report an oce or other outpatient service code (99202–
99215) instead.
▶ For counseling groups of patients with symptoms or established
illness, report 99078 (physician educational services rendered to
patients in a group setting) instead.

6
Preventive Medicine, Counseling
CPT Codes
99401 Preventive medicine counseling or risk factor reduction
intervention(s) provided to an individual; approximately
15 minutes
99402 approximately 30 minutes
99403 approximately 45 minutes
99404 approximately 60 minutes
99411 Preventive medicine counseling or risk factor reduction
intervention(s) provided to individuals in a group setting;
approximately 30 minutes
99412 approximately 60 minutes
ICD-10-CM Codes for Preventive Counseling
▶ e diagnosis codes reported for preventive counseling will vary
depending on the reason for the encounter.
▶ Remember that the patient cannot have symptoms or established
illness; therefore, the diagnosis codes reported cannot reect
symptoms or illnesses.
▶ Examples of some possible diagnosis codes include
Z28.3
Underimmunized status (Code also reason patient is behind,
eg, Z28.82 [caregiver refusal])
Z71.3
Dietary surveillance and counseling
Z71.82 Exercise counseling
Z71.84 Encounter for health counseling related to travel
Z71.85 Encounter for immunization safety counseling (Code also if
vaccine is not carried out [eg, Z28.82])
Z71.89
Other specied counseling
Z71.9 Counseling, unspecied
Behavior Change Interventions, Individual
▶ Used only when counseling a patient (not parent) on smoking
cessation (99406, 99407).
▶ If counseling a patient’s parent or guardian on smoking cessation, do
not report these codes (99406, 99407) under the patient; instead, refer

7
to preventive medicine counseling codes (99401–99404) if the patient
is not currently experiencing adverse eects (eg, illness), or include
under the problem-related E/M service if patient is present for a
sick visit (99202–99215).
▶ Codes 99406–99409 may be reported in addition to the preventive
medicine service codes.
CPT Codes
99406 Smoking and tobacco use cessation counseling visit;
intermediate, greater than 3 minutes up to 10 minutes
99407 intensive, greater than 10 minutes
99408 Alcohol or substance (other than tobacco) abuse structured
screening (eg, Alcohol Use Disorder Identication Test
[AUDIT], Drug Abuse Screening Test [DAST]) and brief
intervention (SBI) services; 15 to 30 minutes
99409 greater than 30 minutes
ICD-10-CM Codes for Risk Factor Reduction and Behavior Change
Interventions
F10.10 Alcohol abuse, uncomplicated
F11.10 Opioid abuse, uncomplicated
F12.10 Cannabis abuse, uncomplicated
F13.10 Sedative, hypnotic or anxiolytic abuse, uncomplicated
F13.90 Sedative, hypnotic, or anxiolytic use, unspecied, uncomplicated
F15.90 Other stimulant use, unspecied, uncomplicated
F16.90 Hallucinogen use, unspecied, uncomplicated
F17.290 Nicotine dependence, other tobacco products (Includes
Electronic nicotine delivery systems [ENDS]/vaping products)
Z71.41 Alcohol abuse counseling and surveillance of alcoholic
Z71.51 Drug abuse counseling and surveillance of drug abuser
Z71.6 Tobacco abuse counseling
Z87.891 Personal history of nicotine dependence
Z91.89 Other specied personal risk factors, presenting as hazards to
health not elsewhere classied

8
OTHER PREVENTIVE MEDICINE SERVICES
Oral Health
CPT Code
99188 Application of topical uoride varnish by a physician or other
qualied health care professional
Refer to page 15 for the denition of QHP.
ICD-10-CM Codes
Z00.121 Routine child health exam with abnormal ndings
Z00.129
Routine child health exam without abnormal ndings
Z29.3
Encounter for prophylactic uoride administration
Z91.841 Risk for dental caries, low
Z91.842 Risk for dental caries, moderate
Z91.843 Risk for dental caries, high
Z91.849 Unspecied risk for dental caries
Pelvic Examination
▶ Preventive medicine service codes (99381–99385 and 99391–99395)
include a pelvic examination as part of the age- and gender-appropriate
examination.
▶ If the patient is having a problem, the physician can report an oce
or other outpatient E/M service code (99212–99215) for the visit and
attach modier 25, which identies that the problem-oriented pelvic
visit is a separately identiable E/M service by the same physician on
the same date of service.
▶ Link the appropriate ICD-10-CM code for the well-child or well-adult
examination with abnormal ndings (Z00.121 or Z00.01) to the
preventive medicine service code, but link a dierent diagnosis code
(eg, N89.8 [vaginal discharge], N94.4 [primary dysmenorrhea]) to
the oce or other outpatient E/M service code (eg, 99212).
▶ Anticipatory or periodic contraceptive management is not a “problem”
and is therefore included in the preventive medicine service code;
however, if contraception creates a problem (eg, breakthrough
bleeding, vomiting), the service can be reported separately with
an oce or other outpatient service code.

9
ICD-10-CM Codes
Z01.411 Gynecological exam with abnormal ndings
Z01.419
Gynecological exam without abnormal ndings
Z11.51
Screening for human papillomavirus (HPV)
Z12.72 Screening for malignant neoplasm of vagina
Z30.011 Initial prescription of contraceptive pills
Z30.012 Prescription of emergency contraception
Z30.013 Initial prescription of injectable contraceptive
Z30.014 Initial prescription of intrauterine contraceptive device (IUD)
Z30.015 Encounter for initial prescription of vaginal ring hormonal
contraceptive
Z30.016 Encounter for initial prescription of transdermal patch
hormonal contraceptive device
Z30.017 Encounter for initial prescription of implantable subdermal
contraceptive
Z30.018 Encounter for initial prescription of other contraceptives
Z30.02 Counseling and instruction in natural family planning to
avoid pregnancy
Z30.09 General counseling and advice on contraception
Z30.40 Surveillance of contraceptives, unspecied
Z30.41 Surveillance of contraceptive pills
Z30.42 Surveillance of injectable contraceptive
Z30.430 Insertion of IUD
Z30.431 Routine checking of IUD
Z30.432 Removal of IUD
Z30.433 Removal and reinsertion of IUD
Z30.44 Encounter for surveillance of vaginal ring hormonal
contraceptive device
Z30.45 Encounter for surveillance of transdermal patch hormonal
contraceptive device
Z30.46 Encounter for surveillance of implantable subdermal
contraceptive
Z30.49 Surveillance of other contraceptives
10
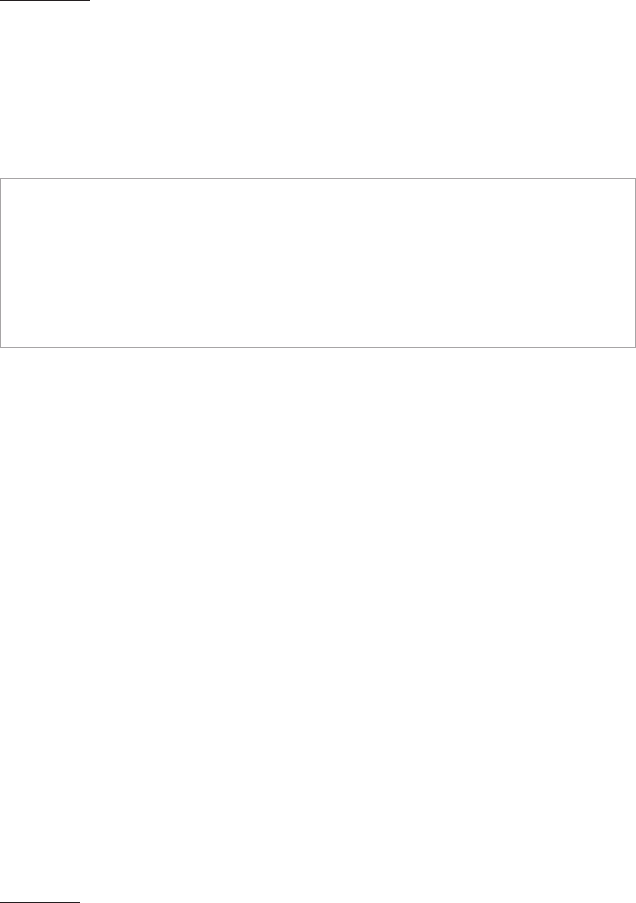
Health Risk Assessments
CPT Codes
96160 Administration of patient-focused health risk assessment
instrument (eg, health hazard appraisal) with scoring and
documentation, per standardized instrument
96161 Administration of caregiver-focused health risk assessment in-
strument (eg, depression inventory) for the benet of the patient,
with scoring and documentation, per standardized instrument
NOTE: Code 96161 can be reported for a postpartum screening administered
to a mother as part of a routine newborn check but billed under the baby’s
name. Link to ICD-10-CM code Z00.121 or Z00.129 for normal screening
results during a routine well-baby examination. Do not report ICD-10-CM
code Z13.31 or Z13.32 under the baby, as those are only for the maternal
record. Alternatively, payers may require G0442 (Annual alcohol misuse
screening, 15 minutes) in lieu of 96160 if screening for alcohol use.
▶ Used to report administration of standardized
health risk assessment instru-
ments on the patient (96160) or a primary caregiver (eg, parent) on behalf of
the patient (96161). Code 96160 would be used to report a standardized
suicide risk assessment linked to ICD-10-CM code Z13.39 (Encounter for
screening examination for other mental health and behavioral disorders).
Code 96161 requires that the questions and answers relate to the primary
caregiver’s health and behaviors, not the patient’s. For standardized ques-
tionnaires about the family's environment (that includes the patient), report
code 96160.
▶ CPT defines standardization as, “Standardized instruments are validated tests
that are administered and scored in a consistent or ‘standard’ manner
consistent with their validation.”
▶ A non-standardized screen or assessment is not separately reported, unless a
specific payer allows.
▶ At the time of publication, the recommended Sudden Cardiac Arrest/Death
risk assessment was not standardized, therefore no separate CPT code can be
reported.
Unlisted Preventive Medicine Service
CPT Code
99429 Unlisted preventive medicine service
Report code 99429 only when a more specic preventive medicine service
code does not exist.

11
SCREENING CODES
Developmental/Autism Screening and Behavioral/Social/
Emotional Screening
CPT Codes ICD-10-CM Codes
96110 Developmental screening,
per instrument, scoring
and documentation
Z13.41 Encounter for autism screening
Z13.42 Encounter for screening for global
developmental delays (milestones)
96127 Brief emotional/behavioral
assessment (eg, depression
inventory) with scoring
and documentation, per
standardized instrument
Z13.31 Encounter for screening for
depression
Z13.39 Encounter for screening examination
for other mental health and
behavioral disorders
▶ U
sed to report administration of standardized developmental/autism
screening instruments (96110) or behavioral/social/emotional screening
instruments (96127). See page 10 for the definition of standardized.
▶ Often reported when performed in the context of preventive medicine
services but may also be reported when screening or assessment is
performed with other E/M services (eg, acute illness or follow-up office
visits).
▶ Clinical staff (eg, registered nurse) typically administers and scores the
completed instrument, while the physician incorporates the
interpretation component into the accompanying E/M service.
▶ When a standardized screening or assessment is administered along with
any E/M service (eg, preventive medicine service), both services should
be reported, and modifier 25 (significant, separately identifiable E/M
service by the same physician on the same day of the procedure or other
service) may need to be appended to the E/M code to show the E/M
service was distinct and necessary at the same visit.
▶ Alternatively, some payers may require the use of code G0444 (annual
depression screening, 15 minutes) in lieu of code 96127 for the routine
depression screening.
▶ For more information on reporting 96110 and 96127 instruments, refer to
https://downloads.aap.org/AAP/PDF/coding_factsheet_
developmentalscreeningtestingandEmotionalBehvioraassessment.pdf.
12
Hearing Screening
CPT Codes ICD-10-CM Codes
92551 Screening test, pure tone,
air only
Z00.121 Routine child health exam
with abnormal findings
92552 Pure tone audiometry
(threshold), air only
Z00.129 Routine child health exam
without abnormal findings
92567 Tympanometry
(impedance testing)
▶ Requires use of calibrated electronic equipment; tests using other
methods (eg, whispered voice, tuning fork) are not reported separately.
▶ Includes testing of both ears; append modier 52 when a test is applied
to only one ear.
▶ For newborn hearing screenings for young patients, including those
patients who are nonverbal or have developmental delays, other hearing
assessment methods may be more appropriate.
CPT Codes ICD-10-CM Codes
92558 Evoked otoacoustic emissions,
screening (qualitative measurement
of distortion product or transient
evoked otoacoustic emissions),
automated analysis
Z00.110 Health supervision for
newborn under 8 days old
or
Z00.111 Health supervision for
newborn 8 to 28 days old
or
Z00.121 Routine child health exam
with abnormal findings
Z00.129 Routine child health exam
without abnormal findings
P09.6 Abnormal findings on
neonatal screening
92650 Auditory evoked potentials;
screening of auditory potential with
broadband stimuli, automated
analysis
92587 Distortion product evoked
otoacoustic emissions; limited
evaluation (to confirm the presence
or absence of hearing disorder, 3-6
frequencies) or transient evoked
otoacoustic emissions, with
interpretation and report
92588 comprehensive diagnostic
evaluation, with interpretation
and report

13
▶ Codes Z01.10 (encounter for examination of ears and hearing without
abnormal ndings) and Z01.118 (encounter for examination of earsand
hearing with other abnormal ndings) are reported only when a patient
presents for an encounter specic to ears and hearing, not for a routine
well-child examination at which a hearing screening is performed.
▶ Failed hearing screenings will most likely result in a follow-up oce
visit (eg, 99212–99215). Code Z01.110 (encounter for hearing exami-
nation following failed hearing screening) is reported when a specic
disorder cannot be identied or when the follow-up hearing screening
ndings are normal. You can also report Z01.118 (encounter for exam-
ination of ears and hearing with other abnormal ndings) and include
the code for the abnormal ndings (eg, R94.120 [abnormal auditory
function study]).
Vision Screening
CPT Codes ICD-10-CM Codes
99173 Screening test of visual acuity
quantitative, bilateral
Z01.020 Encounter for examination of eyes
and vision following failed vision
screening without abnormal
findings
Z01.021 Encounter for examination of eyes
and vision following failed vision
screening with abnormal findings
Z00.121 Routine child health exam
with abnormal findings
99174 Instrument-based ocular
screening (eg, photoscreening,
automated-refraction), bilateral,
with remote-analysis and report
Z00.129 Routine child health exam
without abnormal findings
99177 Instrument-based ocular
screening (eg, photoscreening,
automated-refraction), bilateral,
with on-site analysis
Z01.00 and Z01.01 (examination of eyes and vision with and without
abnormal ndings) are reported only for routine examination of eyes and
vision, not when a vision screening is done during a routine well-child
examination.
14
▶ To report code 99173, you must employ graduate visual acuity stimuli
that allow a quantitative estimate of visual acuity (eg, Snellen chart).
▶ Codes 99174 and 99177 are reported for instrument-based ocular
screening for esotropia, exotropia, anisometropia, cataracts, ptosis,
hyperopia, and myopia.
▶ Code 99177 is reported in lieu of 99174 when the screening instrument
provides you with immediate pass or fail results.
▶ When acuity (99173) or instrument-based ocular screening (eg, 99174)
is measured as part of a general ophthalmologic service or an E/M
service of the eye (eg, for an eye-related problem or symptom), it
is considered part of the diagnostic examination of the oce or
other outpatient service code (99202–99215) and is not reported
separately.
▶ Other identiable services unrelated to the screening test provided
at the same time are reported separately (eg, preventive medicine
services).
▶ Failed vision screenings will most likely result in a follow-up oce
visit (eg, 99212–99215). Report the follow-up screening with
Z01.020 (encounter for examination of eyes and vision following
failed vision screening without abnormal ndings) if normal results
or Z01.021 (encounter for examination of eyes and vision following
failed vision screening with abnormal ndings) if abnormal results.
If abnormal, link to the diagnosis code for the reason for the failure
(eg, H52.1- [myopia]); when a specic disorder cannot be identied,
report R94.118 (abnormal results of other function studies of eye).

15
IMMUNIZATIONS
Immunization Administration (IA)
Pediatric IA Codes
90460 Immunization administration (IA) through 18 years of age
via any route of administration, with counseling by physician
or other qualied health care professional; rst or only
component of each vaccine or toxoid administered
+90461 each additional vaccine or toxoid component
administered (List separately in addition to code for
primary procedure.)
Report 90461 in conjunction with 90460.
▶ Component refers to all antigens in a vaccine that prevent diseases
caused by 1 organism. Multivalent antigens or multiple serotypes of
antigens against a single organism are considered a single component
of vaccines. Combination vaccines are vaccines that contain multiple
vaccine components. Conjugates or adjuvants contained in vaccines
are not considered to be component parts of the vaccine, as dened
previously.
▶ A QHP is an individual who by education, training, licensure/
regulation, facility credentialing (when applicable), and payer policy
is able to perform a professional service within his or her scope of
practice and to independently report a professional service. ese
professionals are distinct from clinical sta. A clinical sta member
is a person who works under the supervision of a physician or other
QHP and who is allowed by law, regulation, facility, and payer policy
to perform or assist in the performance of specied professional
services but does not individually report any professional services.
▶ Code 90460 is used to report the rst or only component in a single
vaccine given during an encounter. You can report 90460 more than
once during a single oce encounter. Code 90461 is considered an
add-on code to 90460 (hence the + symbol next to it). is means
that the provider will use 90461 in addition to 90460 if more than
1 component is contained within a single vaccine administered.
CPT codes 90460 and 90461 are reported regardless of route of
administration.

16
▶ Pediatric IA codes (90460, 90461) are reported only when both of the
following requirements are met:
1. e patient must be 18 years or younger.
2. e physician or other QHP must perform face-to-face vaccine
counseling associated with the administration.
NOTE: e clinical sta can do the actual administration of the vaccine.
▶ If both of these requirements are not met, report a non–age-specic
IA code (90471–90474) instead.
Non–age-specific IA Codes
▶ Report a CPT code for both the administration and product and an
ICD-10-CM code for each vaccine administered during a patient
encounter.
90471 IA (includes percutaneous, intradermal, subcutaneous, or
intramuscular injections); one vaccine (single or combination
vaccine/toxoid)
Do not report 90471 in conjunction with 90473.
+90472 each additional vaccine (single or combination vaccine/
toxoid) (List separately to code for primary procedure.)
Use 90472 in conjunction with 90460, 90471, or 90473.
90473 IA (includes intranasal or oral administration); one vaccine
(single or combination vaccine/toxoid)
Do not report 90473 in conjunction with 90471.
+90474 each additional vaccine (single or combination vaccine/
toxoid) (List separately to code for primary procedure.)
Use 90474 in conjunction with 90460, 90471, or 90473.
▶ Codes 90471 and 90473 are used to code for the rst immunization
given during a single oce visit. Codes 90472 and 90474 are con-
sidered add-on codes (hence the + symbol next to them) to 90460,
90471, and 90473. is means that the provider will use 90472 or
90474 in addition to 90460, 90471, or 90473 if more than 1 vaccine
is administered during a visit. ere can be only 1 rst administration
during a given visit. (See vignettes 3, 4, and 5 on pages 21–23.)

17
▶ If during a single encounter for a patient 18 years or younger, a physi-
cian or other QHP only counsels on some of the vaccines, report code
90460 (and 90461 when applicable) for those counseled on and defer
to codes 90472 or 90474, as appropriate, for those that are not
counseled on.
▶ e following vignettes may help illustrate the correct use of the
administration codes (see pages 25–28 for a full list of vaccine
product codes):
NOTE: e coding vignettes are for teaching purposes only and do not
necessarily follow every payer’s reporting requirements.
Vignette 1
A 2-month-old established patient presents for her checkup. e follow-
ing vaccines are ordered: Pentacel (diphtheria-tetanus-acellular pertussis
[DTaP], Haemophilus inuenzae type b [Hib], inactivated poliovirus
[IPV]), pneumococcal, and rotavirus. e physician counsels the parents
on all of them, consent is obtained and the nurse administers them all.
How are the appropriate codes for this service selected?
Step 1: Select appropriate E/M code.
99391
Preventive medicine service, established patient, infant
(age younger than 1 year)
Step 2: Select appropriate vaccine product codes.
90698
DTaP-Hib-IPV (Pentacel) product
90670 Pneumococcal product
90680 Rotavirus vaccine, oral use
Step 3: Select appropriate IA codes by considering the following questions:
▶ Is the patient 18 years or younger?
▶ If the patient is younger than 18 years, did the physician or other QHP
perform the face-to-face vaccine counseling, discussing the specic
risks and benets of the vaccines?
If the answer to both questions is yes, select a code from the pediatric IA
code family (90460, 90461). If the answer to one of the questions is no,
select a code from the non–age-specic IA code family (90471–90474).
In this vignette, the answer to both questions is yes. erefore, IA codes
90460 and 90461 will be reported.
18
Step 4: Select the appropriate ICD-10-CM diagnosis codes.
Diagnosis codes are used along with CPT codes to reect the outcome
of a visit. e CPT codes tell a carrier what was done, and ICD-10-CM
codes tell a carrier why it was done.
e vaccine product CPT code and its corresponding IA CPT code are
always linked to the same ICD-10-CM code. is is because the vaccine
product and work that goes into administering that product are intended
to provide prophylactic vaccination against a certain type of disease.
ICD-10-CM lists only a single code to describe an encounter in which
a patient receives a vaccine. e code is Z23, and it is reported at any
encounter when a vaccine is given, including routine well-child or
adult examinations.
e diagnosis codes for the 3 vaccines and 3 IA codes used in this vignette
are as follows:
CPT Codes ICD-10-CM Codes
99391 25 Preventive medicine service, established patient,
<1 year
Z00.129
90698 DTaP-Hib-IPV (Pentacel) product Z23
90670 Pneumococcal product Z23
90680 Rotavirus vaccine, oral use Z23
90460 (×3) Pediatric IA (Pentacel, pneumococcal, rotavirus),
first component
Z23
90461 (×4) Pediatric IA (Pentacel), each additional
component
Z23
Vignette 2
A 5-year-old established patient is at a physician’s oce for her annual
well-child examination. e patient is scheduled to receive her rst
hepatitis A vaccine; her h DTaP vaccine; and the inuenza vaccine.
Aer distributing the Vaccine Information Statements and discussing
the risks and benets of immunizations with her parents, the physician
administers the vaccines.
How are the appropriate codes for this service selected?
19
Step 1: Select appropriate E/M code.
99393 Preventive medicine service, established patient, age 5 to
11 years
Step 2: Select appropriate vaccine product codes.
90633 Hepatitis A vaccine, pediatric/adolescent dosage
(2-dose schedule), for intramuscular use
90700 DTaP, for use in individuals younger than 7 years,
for intramuscular use
90686 Inuenza virus vaccine, quadrivalent (IIV4), split virus,
preservative free, 0.5 mL dosage, for IM use
Step 3: Select appropriate IA codes by considering the following questions:
▶ Is the patient 18 years or younger?
▶ If the patient is younger than 18 years, did the physician or other
qualied health care professional perform the face-to-face vaccine
counseling, discussing the specic risks and benets of the vaccines?
If the answer to both questions is yes, select a code from the pediatric IA
code family (90460, 90461). If the answer to one of the questions is no,
select a code from the non–age-specic IA code family (90471–90474).
In this vignette, the answer to both questions is yes. erefore, IA codes
90460 and 90461 will be reported.
Step 4: Select the appropriate ICD-10-CM diagnosis codes.
Diagnosis codes are used along with CPT codes to reect the outcome
of a visit. e CPT codes tell a carrier what was done, and ICD-10-CM
codes tell a carrier why it was done.
e vaccine product CPT code and its corresponding IA CPT code are
always linked to the same ICD-10-CM code. is is because the vaccine
product and work that goes into administering that product are intended
to provide prophylactic vaccination against a certain type of disease.
ICD-10-CM lists only a single code to describe an encounter in which
a patient receives a vaccine. e code is Z23, and it is reported at any
encounter when a vaccine is given, including routine well-child or
adult examinations.
20
e diagnosis codes for the 3 vaccines and 3 IA codes used in this
vignette are as follows:
CPT Codes ICD-10-CM Codes
99393 25 Preventive medicine service, established patient,
5–11 years
Z00.129
90633 Hepatitis A vaccine product Z23
90460 Pediatric IA (hepatitis A vaccine), first component Z23
90700 DTaP vaccine product Z23
90460 Pediatric IA (DTaP vaccine), first component Z23
90461 (×2) Pediatric IA (DTaP vaccine), each additional
component
Z23
90686 Influenza virus vaccine, quadrivalent, preservative
free, 0.5 mL dosage
Z23
90460 Pediatric IA (influenza vaccine), first component Z23
Alternative Coding
CPT Codes ICD-10-CM Codes
99393 25 Preventive medicine service, established patient,
5–11 years
Z00.129
90633 Hepatitis A vaccine product Z23
90700 DTaP vaccine product Z23
90686 Influenza virus vaccine, quadrivalent, preservative
free, 0.5 mL dosage
Z23
90460 (×3) Pediatric IA (hepatitis A, DTaP, influenza vaccines),
first component
Z23
90461 (×2) Pediatric IA (DTaP vaccine), second and third
components
Z23
NOTE: Most payers do not want multiple line items of codes 90460 or
90461; therefore, follow the alternative coding.

21
Rationale
Because the patient is younger than 18 years and there is physician coun-
seling, pediatric IA codes are reported (90460 and 90461). Each vaccine
administered will be reported with its own 90460 (hepatitis A, DTaP,
and inuenza). e only vaccine with multiple components is DTaP.
Because the rst component (ie, diphtheria) was counted in 90460, only
the second and third components (ie, tetanus and acellular pertussis) are
reported with 90461 with 2 units.
Vignette 3
A 19-year-old patient presents to the oce to complete a college physical
examination (in college the patient will be living in a dormitory). He is due
for a tetanus-diphtheria-acellular pertussis (Tdap) booster, meningococcal
vaccine, and intranasal inuenza vaccine. e physician counsels the
patient on each, and the nurse administers each.
CPT Codes ICD-10-CM Codes
99395 25 Preventive medicine service, established patient,
18–39 years
Z02.0
90715 Tdap product Z23
90471 IA, first injection Z23
90734 Meningococcal conjugate vaccine (MenACWY-D
or MenACWY-CRM)
Z23
90472 IA, each additional injection Z23
90672 Influenza virus vaccine, quadrivalent, live (LAIV4),
for intranasal use
Z23
90474 IA, each additional oral or intranasal Z23
Rationale
e patient is older than 18 years; therefore, despite physician counseling,
pediatric IA codes cannot be reported. Instead, codes 90471 and 90474
must be used.
22
Vignette 4
A 17-year-old patient presents to the oce for her annual checkup and
to complete a college physical examination (in college the patient will be
living in a dormitory). e patient is healthy and due for a Tdap booster,
meningococcal vaccine, rst HPV (9-valent) vaccine, and inuenza
vaccine. e physician counsels the patient only on the meningococcal
and HPV vaccines, and the nurse administers each. e patient is asked
to return in 4 to 6 weeks for her second HPV vaccine.
CPT Codes
(First Visit Only)
ICD-10-CM Codes
(First Visit Only)
99395 25 Preventive medicine service, established patient,
12–17 years
Z00.0 and Z02.0
90734 Meningococcal (MCV4) product Z23
90651 HPV (9-valent) product Z23
90460 (×2) Pediatric IA (meningococcal and HPV), first
component
Z23
90715 Tdap product Z23
90472 (×2) IA, each additional injection (Tdap) Z23
90686 Influenza virus vaccine, quadrivalent, preservative
free, 0.5 mL dosage
Z23
Rationale
Because the physician documents counseling only for the meningococcal
and HPV vaccines, code 90460 can be reported only for those vaccines
because the patient meets the age criteria. For the Tdap and inuenza
vaccines, defer to non-pediatric IA codes (90471, 90472). In this case,
however, a rst vaccine code is already reported with code 90460, so
the additional IA code 90472 has to be reported. While ICD-10-CM
does not provide ocial ages for the “adult” ICD-10-CM codes (Z00.00
and Z00.01) in lieu of the well-child examination codes, many payers
use age 17 years as the cuto. Refer to specic payer policy for details.
Vignette 5
A 6-month-old patient presents to the oce for her routine checkup and
to receive vaccines. e patient is due for DTaP, pneumococcal, and hepa-
titis B vaccines. During the examination, the physician nds an upper
respiratory infection and fever. e physician counsels the parent on the
vaccines but decides to defer for 2 weeks. e physician completes the
well-baby checkup on that day.
Two weeks later, the patient returns. e patient is afebrile and asymptom-
atic and is seen only by the nurse. e DTaP, pneumococcal, and hepatitis
B vaccines are administered.
23
CPT Code
(First Visit)
ICD-10-CM Code
(First Visit)
99391 Preventive medicine service, established
patient, <1 year
Z00.121
An appropriate acute sick visit (eg, 99213) may be reported in addition with modifier 25
and linked to an appropriate ICD-10-CM code.
CPT Codes
(2 Weeks Later)
ICD-10-CM Codes
(2 Weeks Later)
90700 DTaP product Z23
90670 Pneumococcal product Z23
90744 Hepatitis B vaccine product Z23
90471 IA (DTaP), first vaccine Z23
90472 (×2) IA (pneumococcal, hepatitis B), each
additional vaccine
Z23
Rati
onale
If counseling occurs outside the IA service, there is no way to report it
separately. erefore, in this vignette, there is nothing separate to report
during the well-baby visit, and when the patient returns and sees only the
nurse, pediatric IA codes cannot be reported; defer to codes 90471–90474.
During the preventive medicine service, when an acute illness is detected,
a code from 99212–99215 can be reported if the service is signicant and
separately identiable. Code 9921x is reported with modier 25. When
the patient returns only for vaccines, an E/M service is not reported. e
ICD-10-CM code will be reported for with abnormal ndings (Z00.121)
because an abnormality was identified during the encounter.
For more information on IA codes, refer to the AAP's coding website
(www.aap.org/coding) and its page dedicated to vaccine coding.
How to Code When Immunizations Are Not Administered
ICD-CM-10 Codes
▶ For many reasons, immunizations are not given during routine pre-
ventive medicine services. Parents may refuse vaccines or defer them,
a patient may be ill at the time and it is counteractive to administer,
or the patient may already have had the disease or be immune.
▶ Because of tracking purposes and quality measures, it is important
to report non-administration as part of the ICD-10-CM codes. e
following ICD-10-CM codes were created to report why a vaccine is
not given:

24
Vaccination not carried out due to
Z28.01
Acute illness
Z28.02 Chronic illness or condition
Z28.03 Immunocompromised state
Z28.04 Allergy to vaccine or component
Z28.1 Religious reasons
Z28.20 Unspecied reason
Z28.21 Patient refusal
Z28.81 Patient had disease being vaccinated against
Z28.82 Caregiver refusal
Z28.83 Vaccine was unavailable (eg, manufacturer delay)
Z28.89 Other reason
Vignette
A 1-year-old presents for his routine well-child examination. He is
scheduled to receive his rst measles, mumps, rubella; hepatitis A; and
varicella vaccines. Because he had a documented case of varicella when
he was 9 months of age, the varicella vaccine is not given.
Report the following ICD-10-CM codes linked to the E/M service:
Z23
Encounter for immunization
Z28.81 Vaccination not carried out due to patient having had the
disease being vaccinated against
COVID-19 IA
As the public health emergency continues to evolve so does coding
for the COVID-19 vaccines. Refer to the online resource for more
information on reporting COVID-19 vaccines (https://www.aap.org/en/
pages/2019-novel-coronavirus-covid-19-infections/covid-19-vaccine-for-
children/covid-19-vaccine-administration-getting-paid/)
Tips:
▶ Do not charge for the product; however, you may report it if the payer
wants the product code included.
▶ Each specic vaccine and dose has its own unique administration code.
▶ If giving a COVID-19 vaccine in addition to routine vaccines,
a modier might be required to override any payer edits.
25
VACCINES FOR CHILDREN PROGRAM
e rules for reporting vaccines for patients who qualify for the Vaccines
for Children (VFC) program vary greatly. Some states require that the
product code be submitted, while others require the IA codes. Some
require the use of modiers, while others do not. Currently, the VFC
program does not recognize component-based vaccine counseling; there-
fore, you will not be paid for CPT
code 90461. e American Academy
of Pediatrics continues to work on changing this so pediatric providers
can be properly compensated for giving multiple-component vaccines.
Also be sure to check with your individual state Medicaid plan for vary-
ing rules, including, but not limted to, being able to report code 99211
in addition to IA codes for vaccine-only encounters. Be sure to get
these rules in writing.
Commonly Administered Pediatric Vaccines (Excludes COVID-19 vaccines -
refer to link on page 24)
Product
Code
Separately report the
administration with
codes 90460–90461
or 90471–90474. Manufacturer Brand
No. of
Vaccine
Components
90702 Diphtheria and tetanus
toxoids (DT), adsorbed when
administered to younger than
seven years, for IM use
SP Diphtheria
and Tetanus
Toxoids
Adsorbed
2
90700 Diphtheria, tetanus toxoids,
and acellular pertussis vaccine
(DTaP), when administered to
<7 years, for IM use
SP
GSK
DAPTACEL
INFANRIX
3
90696 Diphtheria, tetanus toxoids,
and acellular pertussis vaccine
and inactivated poliovirus
vaccine (DTaP-IPV), when
administered to children
4-6 years of age, for IM use
GSK
SP
KINRIX
Quadracel
4
90697 Diphtheria, tetanus toxoids,
acellular pertussis vaccine,
inactivated poliovirus vaccine,
w PRP-OMP conjugate
vaccine, and hepatitis B
vaccine (DTaP-IPV-Hib-HepB),
for IM use
Merck/SP VAXELIS 6
26
Commonly Administered Pediatric Vaccines (continued)
Product
Code
Separately report the
administration with
codes 90460–90461
or 90471–90474. Manufacturer Brand
No. of
Vaccine
Components
90698 Diphtheria, tetanus toxoids,
acellular pertussis vaccine,
Haemophilus influenzae type b,
and inactivated poliovirus
vaccine (DTaP-IPV/Hib),
for IM use
SP Pentacel 5
90723 Diphtheria, tetanus toxoids,
acellular pertussis vaccine,
Hepatitis B, and inactivated
poliovirus vaccine (DTaP-
Hep B- IPV), for IM use
GSK PEDIARIX 5
90633 Hepatitis A vaccine (Hep A),
pediatric/adolescent dosage,
2 dose, for IM use
GSK
Merck
HAVRIX
VAQTA
1
90740 Hepatitis B vaccine (Hep B),
dialysis or immunosuppressed
patient dosage, 3 dose,
for IM use
Merck RECOMBIVAX
HB
1
90743 Hepatitis B vaccine (Hep B),
adolescent, 2 dose, for IM use
Merck RECOMBIVAX
HB
1
90744 Hepatitis B vaccine (Hep B),
pediatric/adolescent dosage,
3 dose, for IM use
Merck
GSK
RECOMBIVAX
HB
ENERGIX-B
1
90746 Hepatitis B vaccine (Hep B),
adult dosage, for IM use
Merck
GSK
RECOMBIVAX
HB
ENERGIX-B
1
90747 Hepatitis B vaccine (Hep B),
dialysis or immunosuppressed
patient dosage, 4 dose,
for IM use
GSK ENERGIX-B 1
90647 Haemophilus influenzae type b
vaccine (Hib), PRP-OMP
conjugate, 3 dose, for IM use
Merck PedvaxHIB 1
90648 Haemophilus influenzae type b
vaccine (Hib), PRP-T conjugate,
4 dose, for IM use
SP
GSK
ActHIB
HIBERIX
1
90651 Human Papillomavirus vaccine
types 6, 11, 16, 18, 31, 33, 45,
52, 58, nonavalent (HPV), 2 or
3 dose schedule, for IM use
Merck GARDASIL 9 1
27
Product
Code
Separately report the
administration with
codes 90460–90461
or 90471–90474. Manufacturer Brand
No. of
Vaccine
Components
90707 Measles, mumps, and rubella
virus vaccine (MMR), live, for
subcutaneous use
Merck M-M-R II 3
90710 Measles, mumps, rubella, and
varicella vaccine (MMRV), live,
for subcutaneous use
Merck ProQuad 4
90619 Meningococcal conjugate
vaccine, serogroups A, C, W, Y,
quadrivalent, tetanus toxoid
carrier (MenACWY-TT), for
IM use
SP MenQuadfi 1
90620 Meningococcal recombinant
protein and outer membrane
vesicle vaccine, serogroup B
(MenB-4C), 2 dose schedule,
for IM use
GSK Bexsero 1
90621 Meningococcal recombinant
lipoprotein vaccine, serogroup B,
2 or 3 dose schedule, for IM use
Pfizer Trumenba 1
90734 Meningococcal conjugate
vaccine, serogroups A, C, W, Y,
quadrivalent, diphtheria toxoid
carrier (MenACWY-D) or CRM197
carrier(MenACWY-CRM), for
IM use
SP
GSK
Menactra
Menveo
1
90670 Pneumococcal conjugate
vaccine, 13 valent (PCV13),
for IM use
Pfizer PREVNAR 13 1
90732
Pneumococcal polysaccharide
vaccine, 23-valent (PPSV23),
adult or immunosuppressed
patient dosage, when adminis-
tered to 2 years or older, for
subcutaneous or IM use
Merck PNEUMOVAX
23
1
90713 Poliovirus vaccine (IPV),
inactivated, for subcutaneous
or IM use
SP IPOL 1
90680 Rotavirus vaccine, pentavalent
(RV5), 3 dose schedule, live,
for oral use
Merck RotaTeq 1
90681 Rotavirus vaccine, human,
attenuated (RV1), 2 dose
schedule, live, for oral use
GSK ROTARIX 1
28
Commonly Administered Pediatric Vaccines (continued)
Product
Code
Separately report the
administration with
codes 90460–90461
or 90471–90474.
Manufacturer Brand
No. of
Vaccine
Components
90714 Tetanus and diphtheria toxoids
(Td) adsorbed, preservative
free, when administered to
seven years or older, for IM use
MBL
SP
TDVAX
TENIVAC
2
90715 Tetanus, diphtheria toxoids
and acellular pertussis vaccine
(Tdap), when administered to
7 years or older, for IM use
SP
GSK
ADACEL
BOOSTRIX
3
90716 Varicella virus vaccine (VAR),
live, for subcutaneous use
Merck VARIVAX 1
90749 Unlisted vaccine or toxoid Please see CPT manual.
90672 Influenza virus vaccine, quad
(LAIV), live, intranasal use
AstraZeneca Flumist Quad 1
90674 Influenza virus vaccine, quad
(ccIIV4), derived from cell
cultures, subunit, preservative
and antibiotic free, 0.5 mL
dosage, IM
Seqirus Flucelvax 1
90682 Influenza virus vaccine,
quad (RIV4), derived from
recombinant DNA, HA protein
only, preservative and
antibiotic free, IM use
Seqirus Flublok Quad 1
90685 Influenza virus vaccine, quad
(IIV4), split virus, preservative
free, 0.25ml dose, for IM use
Seqirus
GSK
SP
Afluria
Fluarix
Fluzone Quad
1
90686 Influenza virus vaccine, quad
(IIV4), split virus, preservative
free, 0.5ml dosage, for IM use
Seqirus
GSK
GSK
SP
Afluria
FLUARIX Quad
FLULAVAL
Fluzone Quad
1
90687 Influenza virus vaccine,
quad (IIV4), split virus,
0.25ml dosage, for IM use
Seqirus
SP
Afluria Quad
Fluzone Quad
1
90688 Influenza virus vaccine,
quad (IIV4), split virus,
0.5ml dosage, for IM use
Seqirus
SP
Afluria
Fluzone Quad
1
90756 Influenza virus vaccine, quad
(ccIIV4), derived from cell
cultures, subunit, antibiotic
free, 0.5mL dosage, for IM use
Seqirus Flucelvax Quad 1
Current at time of publication. Developed and maintained by the American Academy of Pediatrics. Updated
periodically at https://downloads.aap.org/AAP/PDF/coding_vaccine_coding_table.pdf. For reporting purposes
only. Any vaccine products still US Food and Drug Administration pending are not listed in this resource.

29
LABORATORY
Two dierent practice models surround the conducting of laboratory
tests: blood is drawn in oce and specimen is sent to an outside laboratory
for analysis, or blood is drawn and laboratory tests are performed in the
physician’s practice. Never report the laboratory code for a laboratory test
that the practice does not run in-house or is not nancially responsible for
and billed by the outside laboratory. In those cases, report only the blood
draw and specimen handling, as appropriate.
Model 1: Blood is drawn in office and specimen is sent to an outside
laboratory for analysis.
CPT Code
99000 Handling and/or conveyance of specimen for transfer from
the physician’s oce to a laboratory
Venipuncture CPT Codes
36406 Venipuncture, younger than 3 years, necessitating physician’s
skill, not to be used for routine venipuncture
36410 Venipuncture, 3 years or older, necessitating physician’s skill,
for diagnostic or therapeutic purposes (not to be used for
routine venipuncture)
36415 Collection of venous blood by venipuncture
36416 Collection of capillary blood specimen (eg, nger, heel, ear stick)
Venipuncture ICD-10-CM Codes
Link to ICD-10-CM codes for the well-child examination or for specic
screening tests.
Model 2: Blood is drawn and laboratory tests are performed in the
physician’s practice.
Venipuncture CPT Codes
36406 Venipuncture, younger than 3 years, necessitating physician’s
skill, not to be used for routine venipuncture
36410 Venipuncture, 3 years or older, necessitating physician’s skill,
for diagnostic or therapeutic purposes (not to be used for
routine venipuncture)
36415 Collection of venous blood by venipuncture
36416 Collection of capillary blood specimen (eg, nger, heel, ear stick)

30
Venipuncture ICD-10-CM Codes
Link to ICD-10-CM codes for the well-child examination or for specic
screening tests.
Anemia Screening CPT Code
85018 Blood count; hemoglobin
Anemia Screening ICD-10-CM Code
Z13.0 Encounter for screening for diseases of the blood and
blood-forming organs and certain disorders involving the
immune mechanism (eg, anemia)
Bilirubin CPT Codes
82247 Bilirubin, total
88720 Bilirubin, total, transcutaneous
Bilirubin ICD-10-CM Code
Z13.228 Encounter for screening for other metabolic disorder
Dyslipidemia Screening CPT Codes
80061 Lipid panel (includes total cholesterol, high-density lipoprotein
[HDL] cholesterol, and triglycerides)
82465 Cholesterol, serum, total
83718 Lipoprotein, direct measurement, high-density cholesterol
(HDL cholesterol)
84478 Triglycerides
Dyslipidemia Screening ICD-10-CM Code
Z13.220 Encounter for screening for lipid disorders
Hepatitis B Screening CPT Codes
86704 Hepatitis B core antibody (HBcAb); total
86705 IgM antibody
86706 Hepatitis B surface antibody (HBsAb)
86707 Hepatitis Be antibody (HBeAb)
87340 Infectious agent antigen detection by immunoassay technique
qualitative or semiquantitative hepatitis B surface antigen
(HBsAg)

31
Hepatitis B Screening ICD-10-CM Code
Z20.5 Contact with and (suspected) exposure to viral hepatitis
Z11.59 Encounter for screening for other viral diseases
Hepatitis C Screening CPT codes
86803 Hepatitis C antibody;
86804 Hepatitis C antibody; conrmatory test (eg, immunoblot)
87520 Infectious agent detection by nucleic acid (DNA or RNA);
hepatitis C, direct probe technique
87521 Infectious agent detection by nucleic acid (DNA or RNA);
hepatitis C, amplied probe technique, includes reverse
transcription when performed
Hepatitis C Screening ICD-10-CM Code
Z20.5 Contact with and (suspected) exposure to viral hepatitis
Z11.59 Encounter for screening for other viral diseases
Lead Screening CPT Code
83655 Lead
Lead Screening ICD-10-CM Code
Z13.88 Encounter for screening for disorder due to exposure to
contaminants
Newborn Metabolic Screening HCPCS Code
NOTE: See Healthcare Common Procedure Coding System Codes section
on page 34 for explanation of HCPCS codes.
S3620 Newborn metabolic screening panel, includes test kit, postage,
and the laboratory tests specied by the state for inclusion
in this panel (eg, galactose; hemoglobin, electrophoresis;
hydroxyprogesterone, 17-D; phenylalanine [phenylketonuria
(PKU)]; and thyroxine, total)
NOTE: Only report code S3620 if you are billing for the actual running of
the laboratory test or test kit. Otherwise only report the appropriate blood
collection code (eg, 36416).

32
Newborn Metabolic Screening ICD-10-CM Codes
Report the diagnosis codes for the state-specic newborn screening tests
conducted. Examples include
Z13.0
Encounter for screening for diseases of the blood and
blood-forming organs and certain disorders involving the
immune mechanism (eg, anemia, sickle cell)
Z13.21 Encounter for screening for nutritional disorder
Z13.228 Encounter for screening for other metabolic disorders
(eg, PKU, galactosemia)
Z13.29 Encounter for screening for other suspected endocrine
disorder (eg, thyroid)
Papanicolaou Smear HCPCS Code
NOTE: See Healthcare Common Procedure Coding System Codes section
on page 34 for explanation of HCPCS codes.
Q0091 Screening Papanicolaou smear; obtaining, preparing, and
conveyance of cervical or vaginal smear to laboratory
Papanicolaou Smear CPT Code
Collection of a cervical specimen via a pelvic examination is included in
the preventive medicine service code (99381–99385 and 99391–99395).
Papanicolaou Smear ICD-10-CM Codes
Z12.4 Encounter for screening for malignant neoplasm of cervix
(excludes HPV)
Z12.72 Encounter for screening for malignant neoplasm of vagina
Z12.79 Encounter for screening for malignant neoplasm of other
genitourinary organs
Z12.89 Encounter for screening for malignant neoplasms of other sites
Tuberculosis Testing (Mantoux/Purified Protein Derivative [PPD])
Administration of PPD Test
CPT Code ICD-10-CM Code
86580
Skin test; tuberculosis,
intradermal
Z11.1
Encounter for screening for
respiratory tuberculosis
NOTE: ere is no separate administration code for the PPD test.
Do not report one.

33
Reading of PPD Test
If patient returns to have a nurse read the test results, report
CPT Codes ICD-10-CM Codes
99211 Office or other outpatient
services (negative PPD
outcome)
Z11.1 Encounter for screening
for respiratory tuberculosis
(if test is negative)
99212– Office or outpatient services
99215 (physician service for positive
encounter)
R76.11 Nonspecific reaction to
tuberculin skin tuberculosis
(if test is positive)
Sexually Transmitted Infection and HIV Screening CPT Codes
86701 Antibody; HIV-1
86703 Antibody; HIV-1 and HIV-2; single assay
87490 Infectious agent detection by nucleic acid (DNA or RNA);
Chlamydia trachomatis, direct probe technique
87491
Infectious agent detection by nucleic acid (DNA or RNA);
C trachomatis, amplied probe technique
87590
Infectious agent detection by nucleic acid (DNA or RNA);
Neisseria gonorrhoeae, direct probe technique
87591
Infectious agent detection by nucleic acid (DNA or RNA);
N gonorrhoeae, amplied probe technique
87810
Infectious agent detection by immunoassay with direct
optical observation; C trachomatis
87850
Infectious agent detection by immunoassay with direct
optical observation; N gonorrhoeae
Sexually Transmitted Infection and HIV Screening ICD-10-CM Codes
Z11.3 Encounter for screening for infections with a predominantly
sexual mode of transmission (excludes HPV and HIV)
Z11.8 Encounter for screening for other infectious and parasitic
diseases (eg, chlamydia)

34
HEALTHCARE COMMON PROCEDURE CODING SYSTEM CODES
▶ e HCPCS Level II codes are procedure codes used to report services
and supplies not included in the CPT
nomenclature.
▶ Like CPT codes, HCPCS Level II codes are part of the standard
procedure code set under the Health Insurance Portability and
Accountability Act of 1996.
▶ Certain payers may require that HCPCS codes be reported in lieu of
or as a supplement to CPT codes.
▶ e HCPCS nomenclature contains many codes for reporting
nonphysician provider patient education, which can be an integral
service in the provision of pediatric preventive care.
▶ Examples of HCPCS Level II codes relevant to pediatric preventive
care include
S0302
Completed Early and Periodic Screening, Diagnosis, and
Treatment service (List in addition to code for appropriate
E/M service.)
S0610 Annual gynecologic examination; new patient
S0612 Annual gynecologic examination; established patient
S0613 Annual gynecologic examination, clinical breast examination
without pelvic examination
S0622 Routine examination for college, new or established patient
(List separately in addition to appropriate E/M code.)
S9444 Parenting classes, nonphysician provider, per session
S9445 Patient education, not otherwise classied, nonphysician
provider, individual, per session
S9446 Patient education, not otherwise classied, nonphysician
provider, group, per session
S9447 Infant safety (including cardiopulmonary resuscitation)
classes, nonphysician provider, per session
S9451 Exercise classes, nonphysician provider, per session
S9452 Nutrition classes, nonphysician provider, per session
S9454 Stress management classes, nonphysician provider, per session
35
Commonly Reported ICD-10-CM Codes for Pediatric Preventive Services
ICD-10-CM
Code Descriptor
Special Coding
Conventions
Encounter and Examination Codes
Z00.110 Newborn check under 8 days old Outpatient codes only
Z00.111 Newborn check 8 to 28 days old Outpatient codes only
Z00.121
Z00.129
Routine child health examination
with abnormal findings
without abnormal findings
First-listed ICD-10-CM
code only.
Z00.00
Z00.01
General adult medical examination
without abnormal findings
with abnormal findings
First-listed ICD-10-CM
code only.
Typically used for patients
18 years and older
(payer policy).
Z02.0 Examination for admission to
educational institution
Not required in addition
to a Z00 code
Z02.4 Examination for driving license
Z02.5 Examination for participation in sport
Z01.110 Hearing examination following
failed hearing screening
First-listed ICD-10-CM
code only. Do not report
as a secondary code or
in addition to a Z00 code.
Z23 Immunizations This is the only code in
ICD-10-CM for vaccines.
Link to both the product
and administration
CPT codes.
Z29.3 Encounter for prophylactic fluoride
administration
36
Commonly Reported ICD-10-CM Codes for Pediatric Preventive Services (continued)
ICD-10-CM
Code Descriptor
Special Coding
Conventions
Screening Codes
A screening code is not necessary if the screening is inherent to a routine examination,
but it can be reported and oftentimes payers require it.
Z11.1 Respiratory tuberculosis
Z11.3 Infections with a predominantly sexual mode of
transmission (excludes HPV and HIV)
Z12.4 Malignant neoplasm of cervix (excludes HPV)
Z11.51 Human papillomavirus (HPV)
Z11.59 Other viral diseases (eg, Hep B)
Z12.79 Malignant neoplasm of other genitourinary organs
Z12.89 Malignant neoplasms of other sites
Z13.0 Diseases of the blood and blood-forming organs
and certain disorders involving the immune
mechanism (eg, anemia, sickle cell)
Z13.1 Diabetes mellitus
Z13.21 Nutritional disorder
Z13.220 Lipid disorders
Z13.228 Other metabolic disorders (eg, inborn errors of
metabolism, galactosemia, PKU)
Z13.29 Other endocrine disorder
Z13.31 Depression screening
Z13.39 Encounter for screening examination for other
mental health and behavioral disorders
(eg, alcoholism, suicide ideation)
Z13.41 Autism screening
Z13.42 Global developmental delays (milestones)
screening
Z13.88 Disorder due to exposure to contaminants
(eg, lead)
Z13.89 Other specified disorders (not listed here)
37
ICD-10-CM
Code Descriptor
Special Coding
Conventions
Preventive Counseling
Z71.3 Dietary surveillance and counseling
Z71.82 Exercise counseling
Z71.84 Health counseling related to travel
Z71.89 Other specified counseling
Z71.9 Counseling, unspecified
Underimmunized Status
Z28.3 Underimmunized status A status code is
informative and may
affect the course
of treatment and
its outcome. Report
when this is the case.
Vaccines Not Given
Z28.01 Acute illness
Z28.04 Allergy to vaccine or components
Z28.82 Caregiver refusal
Z28.02 Chronic illness or condition
Z28.03 Immune compromised state
Z28.21 Patient refusal
Z28.81 Patient had disease being vaccinated for
Z28.1 Religious reasons
Z28.89 Other reason
Z28.83 Vaccine was unavailable (eg, manufacturer delay)
Z28.20 Unspecified reason

38
Social Determinants of
Health
When identified during a routine preventive medicine service encounter, either through
a formal screening instrument or surveillance, they should be addressed as appropriate
and coded for. Listed below are a few of the SDOH codes in the ICD-10-CM code set;
however, always refer to the larger code set for others. For more information on coding
for SDOH issues and services visit https://downloads.aap.org/AAP/PDF/SDOH.pdf.
Social Determinants Of Health
Abuse
T74.02- Child neglect or abandonment
T74.12- Child physical abuse
T74.22- Child sexual abuse
T74.32- Child psychological abuse
T74.52- Child sexual exploitation
T74.62- Child forced labor exploitation
Z62.81- Personal history of abuse in childhood
Z69- Encounter for mental health services for victim of abuse
Economic
Z59.5 Extreme poverty
Z59.6 Low income
Z59.7 Insufficient social insurance and welfare support
Family Issues
Z63.31 Absence of family member due to military deployment
Z63.32 Other absence of family member
Z63.4 Disappearance and death of family member
Z63.5 Disruption of family by separation and divorce
Z63.72 Alcoholism and drug addiction in family
Z63.79 Other stressful life events affecting family and household
Z62.82- Parent-child conflict
Z62.890 Parent-child estrangement NEC
Food & Water Issues
Z59.41 Food insecurity
Z58.6 Unsafe drinking-water supply
Living situation
Z62.21 Child in welfare custody
Z59.0- Homelessness
Z59.81 Housing instability
39
Social Determinants Of Health
Z62.22 Institutional upbringing
Z62.29 Other upbringing away from parents
Social Issues
Z60.3 Acculturation difficulty
Z60.4 Social exclusion and rejection
Z60.5 Target of (perceived) adverse discrimination and persecution
- Indicates another character is required to complete the code.
Healthcare Effectiveness Data and Information Set Measures Related to
Pediatric Preventive Care
Measure Topic Measure Coding Options
Child and Adolescent
Well-Care Visits:
Well-Child Visits in
the First 15 Months
of Life (W15)
At least 6 well-child examinations
by 15 months of age
ICD-10-CM
Z00.110, Z00.111,
Z00.121, Z00.129
CPT
99381, 99382,
99391, 99392
Child and Adolescent
Well-Care Visits:
Well-Child Visits in
the Third, Fourth,
Fifth, and Sixth Years
of Life (W34)
One or more comprehensive
well-child visits with a PCP
(per year)
ICD-10-CM
Z00.121, Z00.129
CPT
99382, 99392
Child and Adolescent
Well-Care Visits:
Adolescent Well-Care
Visits (AWC)
At least one annual
comprehensive well-care
encounter (per year) for
adolescents and young
adults aged 12–21 years
ICD-10-CM
Z00.00, Z00.01,
Z00.121, Z00.129
CPT
99384, 99385,
99394, 99395
Lead Screening in
Children (LSC)
By age 2 years, have had one or
more capillary or venous lead
blood tests for lead poisoning
CPT
83655
Chlamydia Screening
in Women (CHL)
Sexually active women aged
16–24 years who received at
least one chlamydia test
each year
CPT
87110, 87270,
87320, 87490–87492,
87810
40
Healthcare Effectiveness Data and Information Set Measures Related to
Pediatric Preventive Care (continued)
Measure Topic Measure Coding Options
Childhood
Immunization
Status (CIS) and
Immunizations for
Adolescents (IMA)
By age 2 y, have
DTaP (4 doses)
IPV (3 doses)
MMR (1 dose)
Hib (3 doses)
Hep B (3 doses)
Varicella (1 dose)
Pneumococcal (4 doses)
Hep A (1 dose)
Rotavirus (2–3 doses)
Influenza (2 doses)
By 13th birthday, have
Meningococcal (1 dose)
(Ages 11–13 y)
Tdap (1 dose)
(Ages 10–13 y)
HPV (males/females) (2–3 doses)
(Ages 9–13 y)
Varies; refer to the
Commonly Administered
Pediatric Vaccines table
on pages 25–28 for
specific vaccine codes.
Weight Assessment
and Counseling for
Nutrition and Physical
Activity for Children/
Adolescents (WCC)
For those aged 3–17 years who
had an outpatient visit with a PCP
during the measurement year and
had evidence of BMI percentile
documentation and counseling for
nutrition and/or physical activity
ICD-10-CM
Z68.51–Z68.54,
a
Z71.3, Z02.5, Z71.82
CPT
3000F
a
Abbreviations: BMI, body mass index; CPT, Current Procedural Terminology; DTaP, diphtheria, tetanus,
acellular pertussis; Hep A, hepatitis A; Hep B, hepatitis B; Hib, Haemophilus influenzae type b; HPV, human
papillomavirus; ICD-10-CM, International Classification of Diseases, 10th Revision, Clinical Modification;
IPV, inactivated poliovirus; MMR, measles, mumps, rubella; PCP, primary care practitioner; Tdap, tetanus,
diphtheria, acellular pertussis.
a
Body mass index codes should only be reported when there is a related condition (eg, obesity). Payers need to
accept 3000F in lieu of BMI ICD-10-CM codes for the BMI measure unless the patient has a related condition.

This program is supported by the Health Resources and
Services Administration (HRSA) of the U.S. Department of
Health and Human Services (HHS) as part of an award totaling
$5,000,000 with 10 percent nanced with non-governmental
sources. The contents are those of the author(s) and do not
necessarily represent the ocial views of, nor an endorsement,
by HRSA, HHS, or the U.S. Government. For more information,
please visit HRSA.gov.
TM

BFNC.2022.CPPCFEB
3-362/0222
Also From Bright Futures!
Bright Futures Tool and Resource Kit, 2nd Edition
is online resource provides health care professionals with an organized and
integrated compilation of current forms and materials needed to perform a
well-child visit on one website. e new online platform allows easy access to
the core tools for health supervision visits, including previsit questionnaires,
visit documentation forms, parent and patient education handouts, and
additional documentation forms and AAP-developed educational resources.
Bright Futures: Guidelines for Health Supervision of Infants, Children,
and Adolescents, 4th Edition
is essential resource provides key background information and recommenda-
tions for themes critical to healthy child development along with well-child
supervision standards for 31 age-based visits—from newborn through 21 years.
Additional Pediatric Coding Resources From
the American Academy of Pediatrics
NEW EDITION!
Coding for Pediatrics 2022: A Manual for Pediatric Documentation and
Payment, 27th Edition
Code it right the first time—and avoid delays and costly denials—with this widely
used pediatric coding resource. This year’s updated 27th edition includes all
changes in CPT and ICD-10-CM codes—complete with expert guidance for their
application.
NEW EDITION!
Pediatric ICD-10-CM 2022: A Manual for Provider-Based Coding, 7th Edition
is convenient resource condenses the vast ICD-10-CM code set into 500+ pages
of pediatric-related codes and guidelines.
NEW EDITION!
AAP Pediatric Coding Newsletter™
is print and online monthly publication brings you proven coding strategies and
how-to help with your toughest coding and payment challenges!
To order these and other pediatric resources,
visit shop.aap.org.
